Exploratory Analysis of Serial 18F-fluciclovine PET-CT and Multiparametric MRI during Chemoradiation for Glioblastoma
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Selection and Assessment
2.2. Clinical Imaging
2.2.1. Imaging Timeline
2.2.2. MRI Acquisition
2.2.3. PET-CT Acquisition
2.2.4. Image Post-Processing and Analysis
2.2.5. Tumour Volume Analysis
2.3. Pre-Clinical Glioma Model
2.4. In Vivo Imaging Study Design
2.4.1. MRI Acquisition
2.4.2. Radiotracer Availability and PET-CT Acquisition
2.5. Immunohistochemistry
3. Results
3.1. Demographic and Oncological Outcomes
3.2. Clinical Imaging
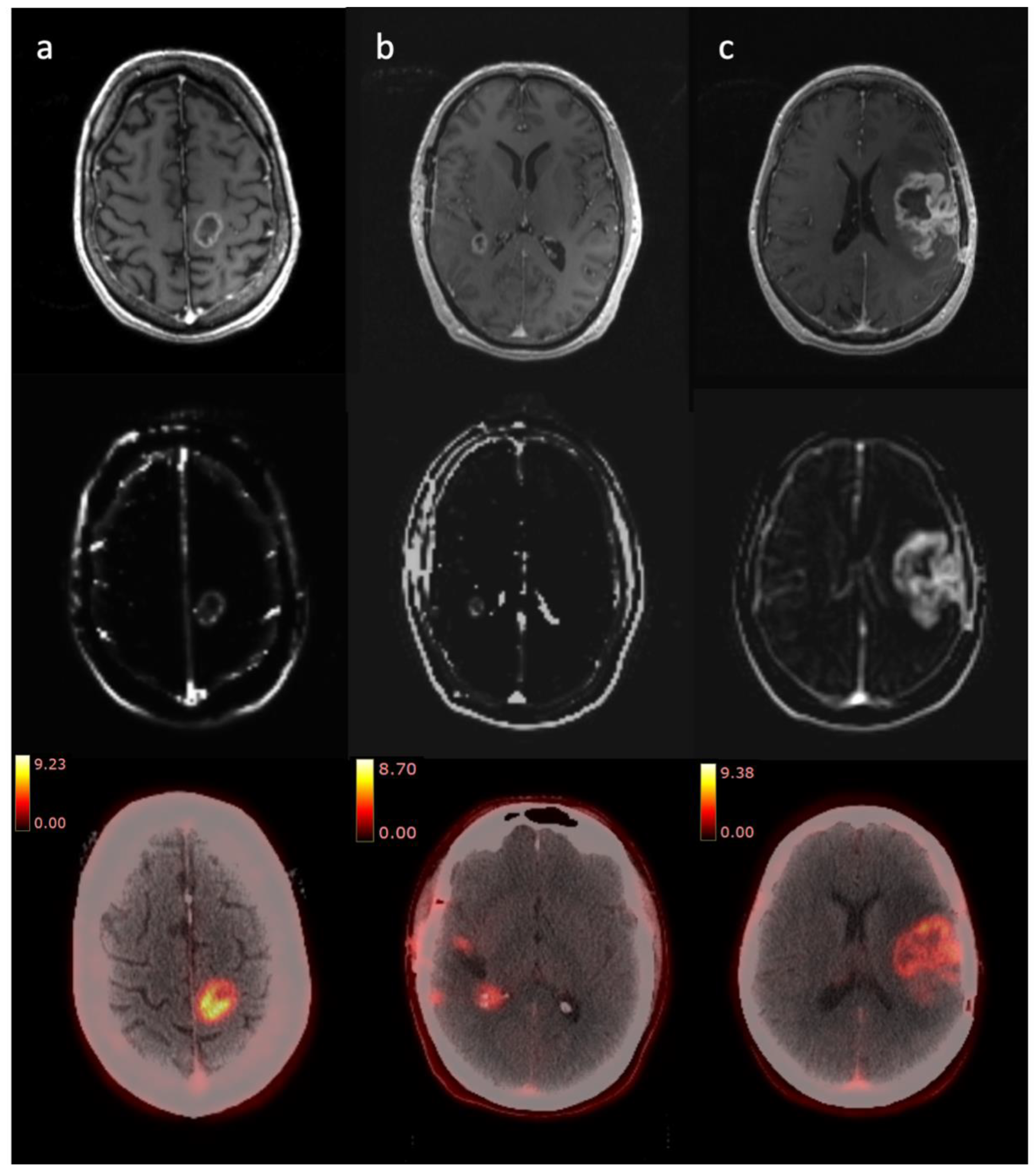
3.2.1. Radiologist Assessment
3.2.2. Tumour Volume Analysis
Short Survival
Long Survival
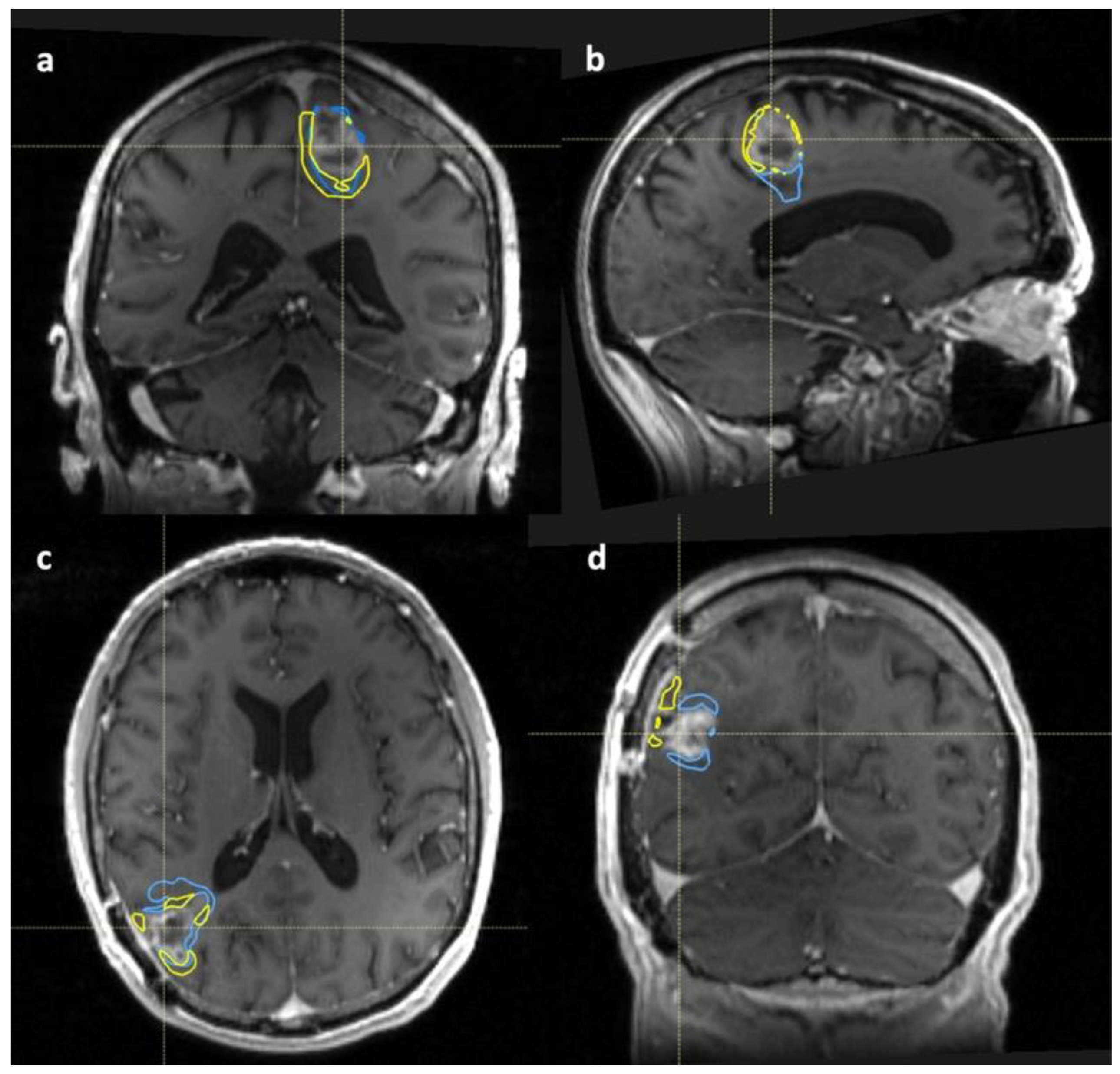
Subtracted Volumes
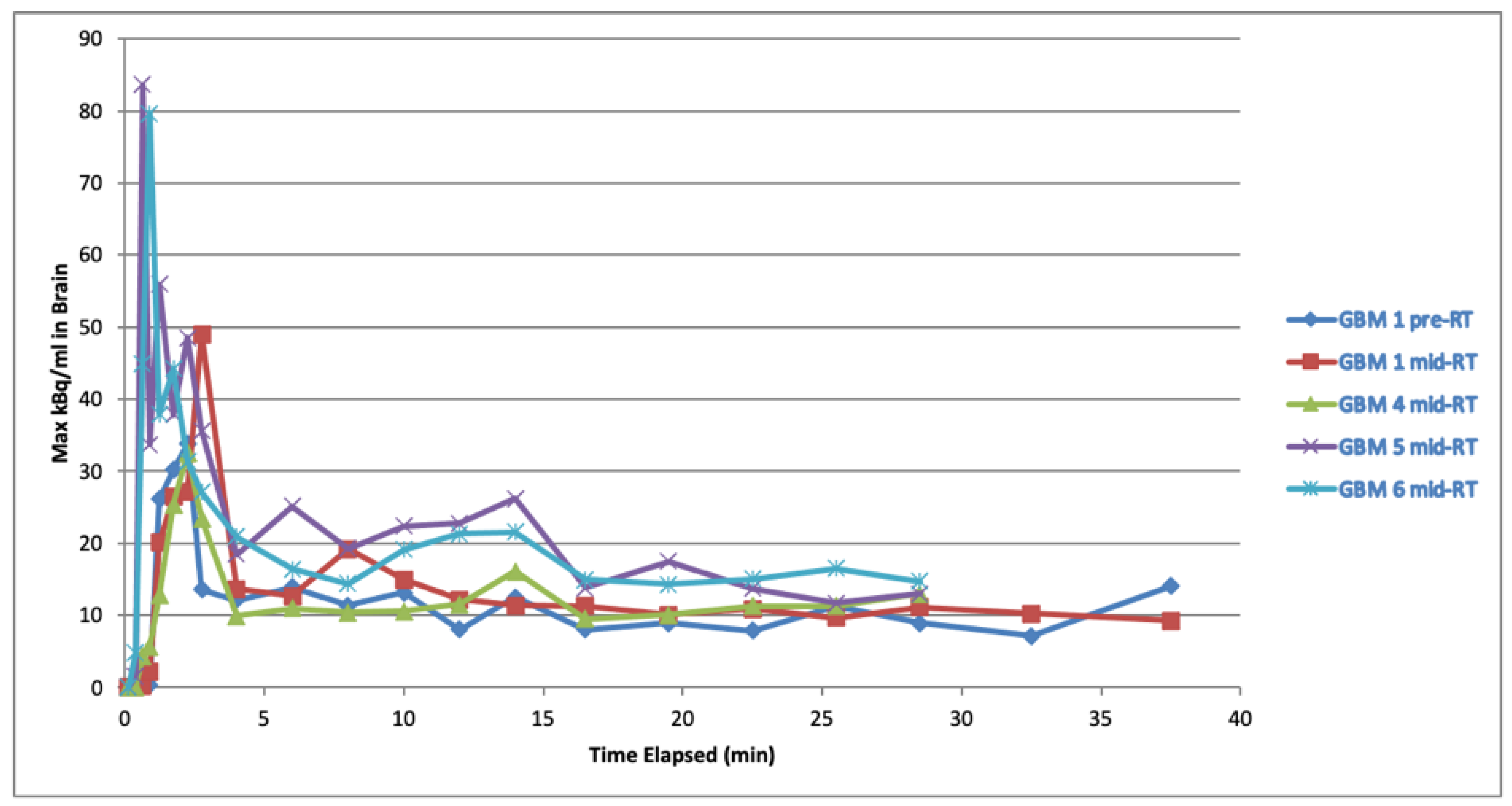
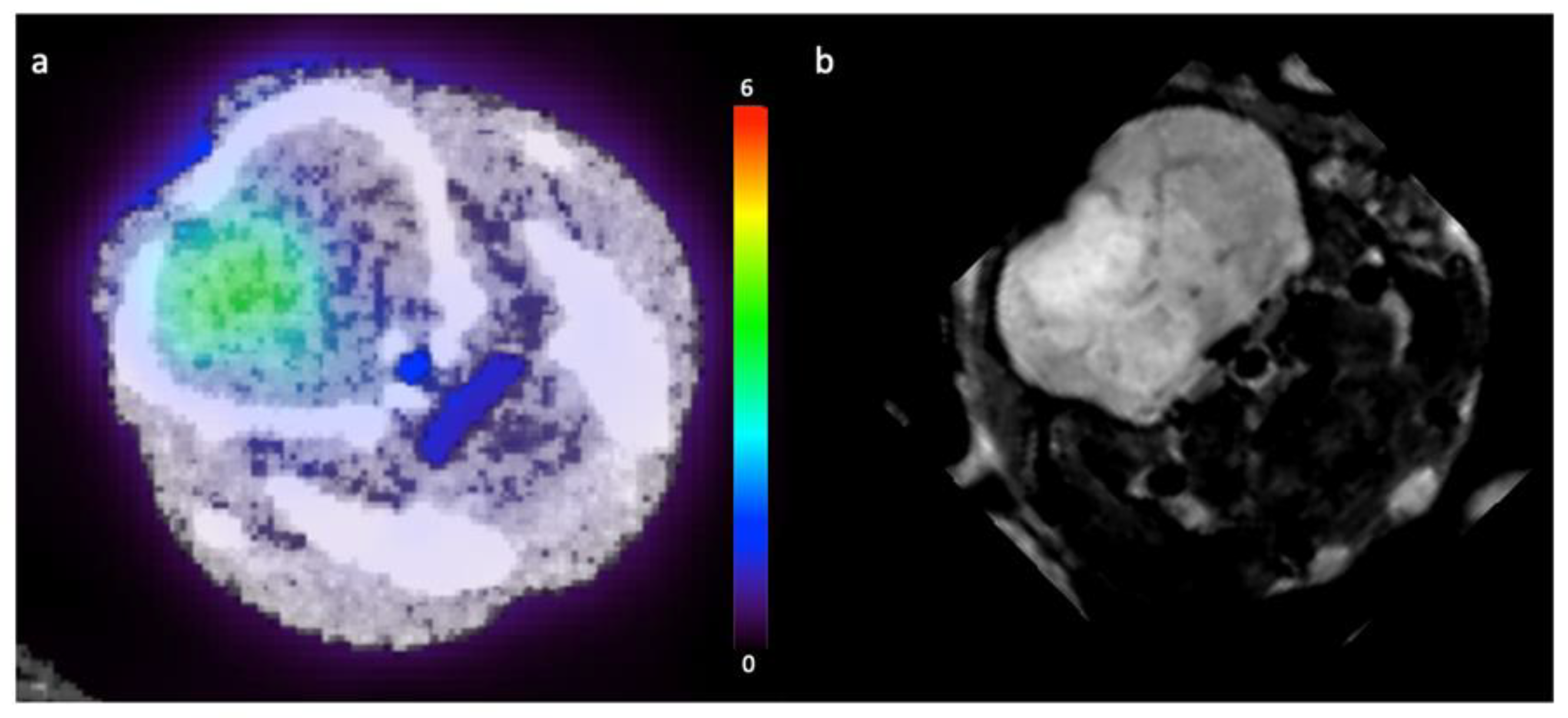
3.3. Pre-Clinical Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Watanabe, M.; Tanaka, R.; Takeda, N. Magnetic Resonance Imaging and Histopathology of Cerebral Gliomas. Neuroradiology 1992, 34, 463–469. [Google Scholar] [CrossRef] [PubMed]
- Lemée, J.M.; Clavreul, A.; Aubry, M.; Com, E.; de Tayrac, M.; Eliat, P.A.; Henry, C.; Rousseau, A.; Mosser, J.; Menei, P. Characterizing the Peritumoral Brain Zone in Glioblastoma: A Multidisciplinary Analysis. J. Neurooncol. 2015, 122, 53–61. [Google Scholar] [CrossRef] [PubMed]
- Shoup, T.M.; Olson, J.; Hoffman, J.M.; Votaw, J.; Eshima, D.; Eshima, L.; Camp, V.M.; Stabin, M.; Votaw, D.; Goodman, M.M. Synthesis and Evaluation of [18F]1-Amino-3-Fluorocyclobutane-1-Carboxylic Acid to Image Brain Tumors. J. Nucl. Med. 1999, 40, 331–338. [Google Scholar] [PubMed]
- Kondo, A.; Ishii, H.; Aoki, S.; Suzuki, M.; Nagasawa, H.; Kubota, K.; Minamimoto, R.; Arakawa, A.; Tominaga, M.; Arai, H. Phase IIa Clinical Study of [18F]Fluciclovine: Efficacy and Safety of a New PET Tracer for Brain Tumors. Ann. Nucl. Med. 2016, 30, 608–618. [Google Scholar] [CrossRef] [PubMed]
- Albano, D.; Tomasini, D.; Bonù, M.; Giubbini, R.; Bertagna, F. 18F-Fluciclovine (18F-FACBC) PET/CT or PET/MRI in Gliomas/Glioblastomas. Ann. Nucl. Med. 2020, 34, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Ono, M.; Oka, S.; Okudaira, H.; Schuster, D.M.; Goodman, M.M.; Kawai, K.; Shirakami, Y. Comparative Evaluation of Transport Mechanisms of Trans-1-Amino-3-[ 18F]Fluorocyclobutanecarboxylic Acid and l-[Methyl- 11C]Methionine in Human Glioma Cell Lines. Brain Res. 2013, 1535, 24–37. [Google Scholar] [CrossRef]
- Fuchs, B.C.; Bode, B.P. Amino Acid Transporters ASCT2 and LAT1 in Cancer: Partners in Crime? Semin. Cancer Biol. 2005, 15, 254–266. [Google Scholar] [CrossRef]
- Bogsrud, T.V.; Londalen, A.; Brandal, P.; Leske, H.; Panagopoulos, I.; Borghammer, P.; Bach-Gansmo, T. 18F-Fluciclovine PET/CT in Suspected Residual or Recurrent High-Grade Glioma. Clin. Nucl. Med. 2019, 44, 605–611. [Google Scholar] [CrossRef]
- Karlberg, A.; Berntsen, E.M.; Johansen, H.; Skjulsvik, A.J.; Reinertsen, I.; Dai, H.Y.; Xiao, Y.; Rivaz, H.; Borghammer, P.; Solheim, O.; et al. 18F-FACBC PET/MRI in Diagnostic Assessment and Neurosurgery of Gliomas. Clin. Nucl. Med. 2019, 44, 550–559. [Google Scholar] [CrossRef] [Green Version]
- Wakabayashi, T.; Hirose, Y.; Miyake, K.; Arakawa, Y.; Kagawa, N.; Nariai, T.; Narita, Y.; Nishikawa, R.; Tsuyuguchi, N.; Fukami, T.; et al. Determining the Extent of Tumor Resection at Surgical Planning with 18F-Fluciclovine PET/CT in Patients with Suspected Glioma: Multicenter Phase III Trials. Ann. Nucl. Med. 2021, 35, 1279–1292. [Google Scholar] [CrossRef]
- Doi, Y.; Kanagawa, M.; Maya, Y.; Tanaka, A.; Oka, S.; Nakata, N.; Toyama, M.; Matsumoto, H.; Shirakami, Y. Evaluation of Trans-1-Amino-3-18F-Fluorocyclobutanecarboxylic Acid Accumulation in Low-Grade Glioma in Chemically Induced Rat Models: PET and Autoradiography Compared with Morphological Images and Histopathological Findings. Nucl. Med. Biol. 2015, 42, 664–672. [Google Scholar] [CrossRef] [PubMed]
- Jensen, R.L.; Mumert, M.L.; Gillespie, D.L.; Kinney, A.Y.; Schabel, M.C.; Salzman, K.L. Preoperative Dynamic Contrast-Enhanced MRI Correlates with Molecular Markers of Hypoxia and Vascularity in Specific Areas of Intratumoral Microenvironment and Is Predictive of Patient Outcome. Neuro-Oncology 2014, 16, 280–291. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Keil, V.C.; Gielen, G.H.; Pintea, B.; Baumgarten, P.; Datsi, A.; Hittatiya, K.; Simon, M.; Hattingen, E. DCE-MRI in Glioma, Infiltration Zone and Healthy Brain to Assess Angiogenesis: A Biopsy Study. Clin. Neuroradiol. 2021, 31, 1049–1058. [Google Scholar] [CrossRef]
- Bisdas, S.; Smrdel, U.; Bajrovic, F.F.; Surlan-Popovic, K. Assessment of Progression-Free-Survival in Glioblastomas by Intratreatment Dynamic Contrast-Enhanced MRI. Clin. Neuroradiol. 2016, 26, 39–45. [Google Scholar] [CrossRef] [PubMed]
- Larsson, C.; Groote, I.; Vardal, J.; Kleppestø, M.; Odland, A.; Brandal, P.; Due-Tønnessen, P.; Holme, S.S.; Hope, T.R.; Meling, T.R.; et al. Prediction of Survival and Progression in Glioblastoma Patients Using Temporal Perfusion Changes during Radiochemotherapy. Magn. Reson. Imaging 2020, 68, 106–112. [Google Scholar] [CrossRef]
- Kim, M.M.; Aryal, M.P.; Sun, Y.; Parmar, H.A.; Li, P.; Schipper, M.; Wahl, D.R.; Lawrence, T.S.; Cao, Y. Response Assessment during Chemoradiation Using a Hypercellular/Hyperperfused Imaging Phenotype Predicts Survival in Patients with Newly Diagnosed Glioblastoma. Neuro-Oncology 2021, 23, 1537–1546. [Google Scholar] [CrossRef]
- Tofts, P.S.; Brix, G.; Buckley, D.L.; Evelhoch, J.L.; Henderson, E.; Knopp, M.V.; Larsson, H.B.W.; Lee, T.Y.; Mayr, N.A.; Parker, G.J.M.; et al. Estimating Kinetic Parameters from Dynamic Contrast-Enhanced T1- Weighted MRI of a Diffusable Tracer: Standardized Quantities and Symbols. J. Magn. Reson. Imaging 1999, 10, 223–232. [Google Scholar] [CrossRef]
- Mason, D.; Scaramallion; Rhaxton; Mrbean-Bremen; Suever, J.; Vanessasaurus; Lemaitre, G.; Orfanos, D.P.; Panchal, A.; Rothberg, A.; et al. Pydicom: An Open Source DICOM Library 2.1.2. Available online: https://github.com/pydicom/pydicom (accessed on 12 July 2021).
- Harris, C.R.; Millman, K.J.; van der Walt, S.J.; Gommers, R.; Virtanen, P.; Cournapeau, D.; Wieser, E.; Taylor, J.; Berg, S.; Smith, N.J.; et al. Array Programming with NumPy. Nature 2020, 585, 357–362. [Google Scholar] [CrossRef]
- Van Der Walt, S.; Schönberger, J.L.; Nunez-Iglesias, J.; Boulogne, F.; Warner, J.D.; Yager, N.; Gouillart, E.; Yu, T. Scikit-Image: Image Processing in Python. PeerJ 2014, 2, e453. [Google Scholar] [CrossRef]
- Virtanen, P.; Gommers, R.; Oliphant, T.E.; Haberland, M.; Reddy, T.; Cournapeau, D.; Burovski, E.; Peterson, P.; Weckesser, W.; Bright, J.; et al. SciPy 1.0: Fundamental Algorithms for Scientific Computing in Python. Nat. Methods 2020, 17, 261–272. [Google Scholar] [CrossRef] [Green Version]
- LASA Education, Training and Ethics Section. Guiding Principles for Preparing for and Undertaking Aseptic Surgery; LASA: Tamworth, UK, 2017; Volume 17. [Google Scholar]
- Wick, W.; Weller, M.; van den Bent, M.; Sanson, M.; Weiler, M.; von Deimling, A.; Plass, C.; Hegi, M.; Platten, M.; Reifenberger, G. MGMT Testing—The Challenges for Biomarker-Based Glioma Treatment. Nat. Rev. Neurol. 2014, 10, 372–385. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wakabayashi, T.; Iuchi, T.; Tsuyuguchi, N.; Nishikawa, R.; Arakawa, Y.; Sasayama, T.; Miyake, K.; Nariai, T.; Narita, Y.; Hashimoto, N.; et al. Diagnostic Performance and Safety of Positron Emission Tomography Using 18F-Fluciclovine in Patients with Clinically Suspected High- or Low-Grade Gliomas: A Multicenter Phase IIb Trial. Asia Ocean. J. Nucl. Med. Biol. 2017, 5, 10–21. [Google Scholar] [CrossRef] [PubMed]
- Michaud, L.; Beattie, B.J.; Akhurst, T.; Dunphy, M.; Zanzonico, P.; Finn, R.; Mauguen, A.; Schöder, H.; Weber, W.A.; Lassman, A.B.; et al. 18F-Fluciclovine (18F-FACBC) PET Imaging of Recurrent Brain Tumors. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 1353–1367. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nabavizadeh, S.A.; Doot, R.K.; Young, A.J.; Bagley, S.J.; Ware, J.B.; Schubert, E.; Henderson, F., Jr.; Pantel, A.; Chen, H.I.; Lee, J.Y.K.; et al. NIMG-18. [18F]Fluciclovine Pet to Distinguish Pseudoprogression from Tumor Progression in Post-Treatment Glioblastoma. Neuro-Oncology 2021, 23, vi131. [Google Scholar] [CrossRef]
- Kotecha, R.; Aboian, M.; Nabavizadeh, S.A.; Parent, E.E.; Trifiletti, D.M.; Chao, S.T. Letter Regarding “Contribution of PET Imaging to Radiotherapy Planning and Monitoring in Glioma Patients—A Report of the PET/RANO Group”: 18F-Fluciclovine and Target Volume Delineation. Neuro-Oncology 2021, 23, 1408–1409. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Liu, H.; Tong, H.; Wang, S.; Yang, Y.; Liu, G.; Zhang, W. Clinical Applications of Contrast-Enhanced Perfusion MRI Techniques in Gliomas: Recent Advances and Current Challenges. Contrast Media Mol. Imaging 2017, 2017, 7064120. [Google Scholar] [CrossRef] [Green Version]
- Lal, A.; Peters, H.; St, B.; Haroon, Z.A.; Mark, W.; Strausberg, R.L.; Kaanders, J.H.A.M.; Van Der Kogel, J.; Riggins, G.J. Transcriptional Response to Hypoxia in Human Tumors These Observations Raise the Question: Which Cancer Cells Adapt to Hypoxia and An Initial Molecular Response to Hyp-Regions within Solid Tumors Is Associ- Serial Analysis of Gene Expression HIF-1 Trans. J. Natl. Cancer Inst. 2001, 93, 1337–1343. [Google Scholar] [CrossRef] [Green Version]
- Hambardzumyan, D.; Bergers, G. Glioblastoma: Defining Tumor Niches. Trends Cancer 2015, 1, 252–265. [Google Scholar] [CrossRef] [Green Version]
- Jiang, S.; Eberhart, C.G.; Lim, M.; Heo, H.Y.; Zhang, Y.; Blair, L.; Wen, Z.; Holdhoff, M.; Lin, D.; Huang, P.; et al. Identifying Recurrent Malignant Glioma after Treatment Using Amide Proton Transfer-Weighted MR Imaging: A Validation Study with Image-Guided Stereotactic Biopsy. Clin. Cancer Res. 2019, 25, 552–561. [Google Scholar] [CrossRef] [Green Version]
- Yamauchi, T.; Kitai, R.; Kodera, T.; Arishima, H.; Matsuda, K.; Isozaki, M.; Ishida, S.; Matta, Y.; Kanamoto, M.; Kimura, H.; et al. Comparison of Amide Proton Transfer Imaging with Perfusion Imaging of Using Arterial Spin-Labeling for Evidence of Tumor Invasion in Glioblastoma. Interdiscip. Neurosurg. Adv. Tech. Case Manag. 2022, 28, 101461. [Google Scholar] [CrossRef]
- Jiang, S.; Eberhart, C.G.; Zhang, Y.; Heo, H.Y.; Wen, Z.; Blair, L.; Qin, H.; Lim, M.; Quinones-Hinojosa, A.; Weingart, J.D.; et al. Amide Proton Transfer-Weighted Magnetic Resonance Image-Guided Stereotactic Biopsy in Patients with Newly Diagnosed Gliomas. Eur. J. Cancer 2017, 83, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Yoo, R.E.; Choi, S.H.; Kim, T.M.; Park, C.K.; Park, S.H.; Won, J.K.; Kim, I.H.; Lee, S.T.; Choi, H.J.; You, S.H.; et al. Dynamic Contrast-Enhanced MR Imaging in Predicting Progression of Enhancing Lesions Persisting after Standard Treatment in Glioblastoma Patients: A Prospective Study. Eur. Radiol. 2017, 27, 3156–3166. [Google Scholar] [CrossRef] [PubMed]
- Brandes, A.A.; Tosoni, A.; Spagnolli, F.; Frezza, G.; Leonardi, M.; Calbucci, F.; Franceschi, E. Disease Progression or Pseudoprogression after Concomitant Radiochemotherapy Treatment: Pitfalls in Neurooncology. Neuro-Oncology 2008, 10, 361–367. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Patient | ||||||
|---|---|---|---|---|---|---|
| GBM 1 | GBM 2 | GBM 3 | GBM 4 | GBM 5 | GBM 6 | |
| Age at surgery (years) | 72 | 47 | 54 | 67 | 57 | 68 |
| Gender | M | M | M | F | M | F |
| PET-CT studies (number) | 2 | 2 | 3 | 3 | 3 | 3 |
| Multiparametric MRI studies (number) | 3 | 3 | 3 | 3 | 3 | 3 |
| Status | Deceased | Deceased | Deceased | Alive | Alive | Deceased |
| Overall survival (days) | 249 | 193 | 292 | 910 * | 903 * | 554 |
| Surgery | Stereotactic biopsy | Resection | Resection | Resection | Resection | Resection |
| Radiotherapy | 60Gy/30 fractions | 60Gy/30 fractions | 60Gy/30 fractions | 60Gy/30 fractions | 60Gy/30 fractions | 60Gy/30 fractions |
| Adjuvant temozolomide | None | None | 4 cycles | 6 cycles | 6 cycles | 6 cycles |
| Histology | Glioblastoma | Glioblastoma | Glioblastoma | Glioblastoma | Glioblastoma | Glioblastoma |
| Cytogenetic analysis | ||||||
| IDH1/2 | Wild type | Wild type | Wild type | Wild type | Wild type | Failed |
| MGMT | Unmethylated | Unmethylated | Unmethylated | Methylated | Methylated | Failed |
| TERT promoter | Mutated | Mutated | Mutated | Mutated | Wild type | Failed |
| 1p/19q co-deletion | Wild-type | Wild-type | Wild-type | Wild-type | Wild-type | Failed |
| GBM 1 | ||||||||
| PET-CT | MRI | |||||||
| Timepoint | 2 × SUVmax | 3 × SUVmax | 4 × SUVmax | Radiologist Assessment | Gd-T1 | Ktrans | ve | Radiologist Assessment |
| Pre-RT | 16.0 | 11.1 | 8.9 | Avid tumour | 6.3 | 7.3 | 7.8 | Stable |
| Mid-RT | 14.9 | 10.6 | 7.0 | Stable | 6.8 | 7.1 | 5.6 | Stable |
| Post-RT | - | - | - | - | 13.5 | 14.1 | 15.5 | Progression |
| GBM 2 | ||||||||
| PET-CT | MRI | |||||||
| Timepoint | 2 × SUVmax | 3 × SUVmax | 4 × SUVmax | Radiologist Assessment | Gd-T1 | Ktrans | ve | Radiologist Assessment |
| Pre-RT | 47.7 | 36.2 | 29.0 | Avid tumour | 21.4 | 23.3 | 25.8 | Progression |
| Mid-RT | 71.8 | 57.4 | 44.5 | Progression | 47.9 | 53.1 | 57.4 | Progression/Pseudoprogression |
| Post-RT | - | - | - | - | 84.3 | 84.8 | 96.9 | Progression |
| GBM 3 | ||||||||
| PET-CT | MRI | |||||||
| Timepoint | 2 × SUVmax | 3 × SUVmax | 4 × SUVmax | Radiologist Assessment | Gd-T1 | Ktrans | ve | Radiologist Assessment |
| Pre-RT | 12.1 | 6.1 | 3.4 | Multifocal avid tumour | 3.4 | 2.8 | 3.9 | Mixed picture |
| Mid-RT | 13.0 | 6.5 | 3.6 | Stable | 4.9 | 5.1 | 4.5 | Progression |
| Post-RT | 31.0 | 18.4 | 12.8 | Progression | 13.4 | 23.3 | 12.5 | Progression |
| GBM 4 | ||||||||
| PET-CT | MRI | |||||||
| Timepoint | 2 × SUVmax | 3 × SUVmax | 4 × SUVmax | Radiologist Assessment | Gd-T1 | Ktrans | ve | Radiologist Assessment |
| Pre-RT | 0.6 | 0.1 | 0 | Likely remnant tumour | 0.4 | 0.6 | 0.8 | Small volume enhancement |
| Mid-RT | 0.2 | 0 | 0 | Stable | 0.1 | 0.2 | 0.1 | Stable |
| Post-RT | 0.1 | 0 | 0 | Stable | 0.1 | 0.1 | 0.03 | Stable |
| GBM 5 | ||||||||
| PET-CT | MRI | |||||||
| Timepoint | 2 × SUVmax | 3 × SUVmax | 4 × SUVmax | Radiologist Assessment | Gd-T1 | Ktrans | ve | Radiologist Assessment |
| Pre-RT | 12.4 | 3.2 | 0.5 | Uptake at margins | 6.3 | 11.6 | 9.9 | Stable |
| Mid-RT | 12.8 | 5.1 | 1.4 | Stable | 5.1 | 4.0 | 5.3 | Stable |
| Post-RT | 7.2 | 1.8 | 0.2 | Stable | 5.1 | 4.6 | 5.5 | Progression/pseudoprogression |
| GBM 6 | ||||||||
| PET-CT | MRI | |||||||
| Timepoint | 2 × SUVmax | 3 × SUVmax | 4 × SUVmax | Radiologist Assessment | Gd-T1 | Ktrans | ve | Radiologist Assessment |
| Pre-RT | 44.4 | 35.0 | 27.5 | Large avid tumour | 46.6 | 51.0 | 48.3 | Stable/Large residuum |
| Mid-RT | 43.9 | 33.7 | 26.3 | Partial response | 32.9 | 36.6 | 34.6 | Stable |
| Post-RT | 46.8 | 36.8 | 28.6 | Stable tumour | 35.4 | 40.0 | 39.3 | Likely progression |
| GBM 1 | |||||
| Volume (cm3) | Dice Similarity Coefficient (DSC) | ||||
| Timepoint | PET (3 × SUVmax) | Ktrans | ve | PET vs. Ktrans | PET vs. ve |
| Pre-RT | 5.1 | 1.4 | 1.6 | 0.4 | 0.3 |
| Mid-RT | 4.1 | 2.1 | 1.1 | 0.2 | 0.2 |
| Post-RT | - | 1.9 | 2.6 | - | - |
| GBM 2 | |||||
| Volume (cm3) | Dice Similarity Coefficient (DSC) | ||||
| Timepoint | PET (3 × SUVmax) | Ktrans | ve | PET vs. Ktrans | PET vs. ve |
| Pre-RT | 15.7 | 5.4 | 6.6 | 0.5 | 0.5 |
| Mid-RT | 12.0 | 9.5 | 12.6 | 0.5 | 0.5 |
| Post-RT | - | 7.3 | 16.1 | - | - |
| GBM 3 | |||||
| Volume (cm3) | Dice Similarity Coefficient (DSC) | ||||
| Timepoint | PET (3 × SUVmax) | Ktrans | ve | PET vs. Ktrans | PET vs. ve |
| Pre-RT | 3.8 | 1.0 | 2.3 | 0.2 | 0.3 |
| Mid-RT | 2.9 | 2.3 | 1.3 | 0.3 | 0.3 |
| Post-RT | 1.0 | 10.4 | 3.9 | 0.1 | 0.1 |
| GBM 4 | |||||
| Volume (cm3) | Dice Similarity Coefficient (DSC) | ||||
| Timepoint | PET (3 × SUVmax) | Ktrans | ve | PET vs. Ktrans | PET vs. ve |
| Pre-RT | 0.1 | 0.4 | 0.5 | 0 | 0 |
| Mid-RT | - | 0.1 | 0.04 | - | - |
| Post-RT | - | 0.04 | 0.03 | - | - |
| GBM 5 | |||||
| Volume (cm3) | Dice Similarity Coefficient (DSC) | ||||
| Timepoint | PET (3 × SUVmax) | Ktrans | ve | PET vs. Ktrans | PET vs. ve |
| Pre-RT | 1.9 | 6.0 | 5.0 | 0.2 | 0.2 |
| Mid-RT | 2.3 | 0.7 | 1.4 | 0.2 | 0.3 |
| Post-RT | 2.3 | 1.2 | 1.8 | 0.1 | 0.1 |
| GBM 6 | |||||
| Volume (cm3) | Dice Similarity Coefficient (DSC) | ||||
| Timepoint | PET (3 × SUVmax) | Ktrans | ve | PET vs. Ktrans | PET vs. ve |
| Pre-RT | 0.7 | 7.6 | 7.1 | 0.1 | 0.1 |
| Mid-RT | 4.4 | 7.3 | 6.3 | 0.5 | 0.5 |
| Post-RT | 4.9 | 7.1 | 6.1 | 0.3 | 0.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fatania, K.; Frood, R.; Tyyger, M.; McDermott, G.; Fernandez, S.; Shaw, G.C.; Boissinot, M.; Salvatore, D.; Ottobrini, L.; Teh, I.; et al. Exploratory Analysis of Serial 18F-fluciclovine PET-CT and Multiparametric MRI during Chemoradiation for Glioblastoma. Cancers 2022, 14, 3485. https://doi.org/10.3390/cancers14143485
Fatania K, Frood R, Tyyger M, McDermott G, Fernandez S, Shaw GC, Boissinot M, Salvatore D, Ottobrini L, Teh I, et al. Exploratory Analysis of Serial 18F-fluciclovine PET-CT and Multiparametric MRI during Chemoradiation for Glioblastoma. Cancers. 2022; 14(14):3485. https://doi.org/10.3390/cancers14143485
Chicago/Turabian StyleFatania, Kavi, Russell Frood, Marcus Tyyger, Garry McDermott, Sharon Fernandez, Gary C. Shaw, Marjorie Boissinot, Daniela Salvatore, Luisa Ottobrini, Irvin Teh, and et al. 2022. "Exploratory Analysis of Serial 18F-fluciclovine PET-CT and Multiparametric MRI during Chemoradiation for Glioblastoma" Cancers 14, no. 14: 3485. https://doi.org/10.3390/cancers14143485
APA StyleFatania, K., Frood, R., Tyyger, M., McDermott, G., Fernandez, S., Shaw, G. C., Boissinot, M., Salvatore, D., Ottobrini, L., Teh, I., Wright, J., Bailey, M. A., Koch-Paszkowski, J., Schneider, J. E., Buckley, D. L., Murray, L., Scarsbrook, A., Short, S. C., & Currie, S. (2022). Exploratory Analysis of Serial 18F-fluciclovine PET-CT and Multiparametric MRI during Chemoradiation for Glioblastoma. Cancers, 14(14), 3485. https://doi.org/10.3390/cancers14143485






