Assessing Ethnic Inequalities in Diagnostic Interval of Common Cancers: A Population-Based UK Cohort Study
Abstract
Simple Summary
Abstract
1. Background
2. Methods
2.1. Study Design and Data Sources
2.2. Participants
2.3. Study Variables
2.4. Statistical Analyses
3. Results
3.1. Participant Characteristics
3.2. Index Features of Cancer
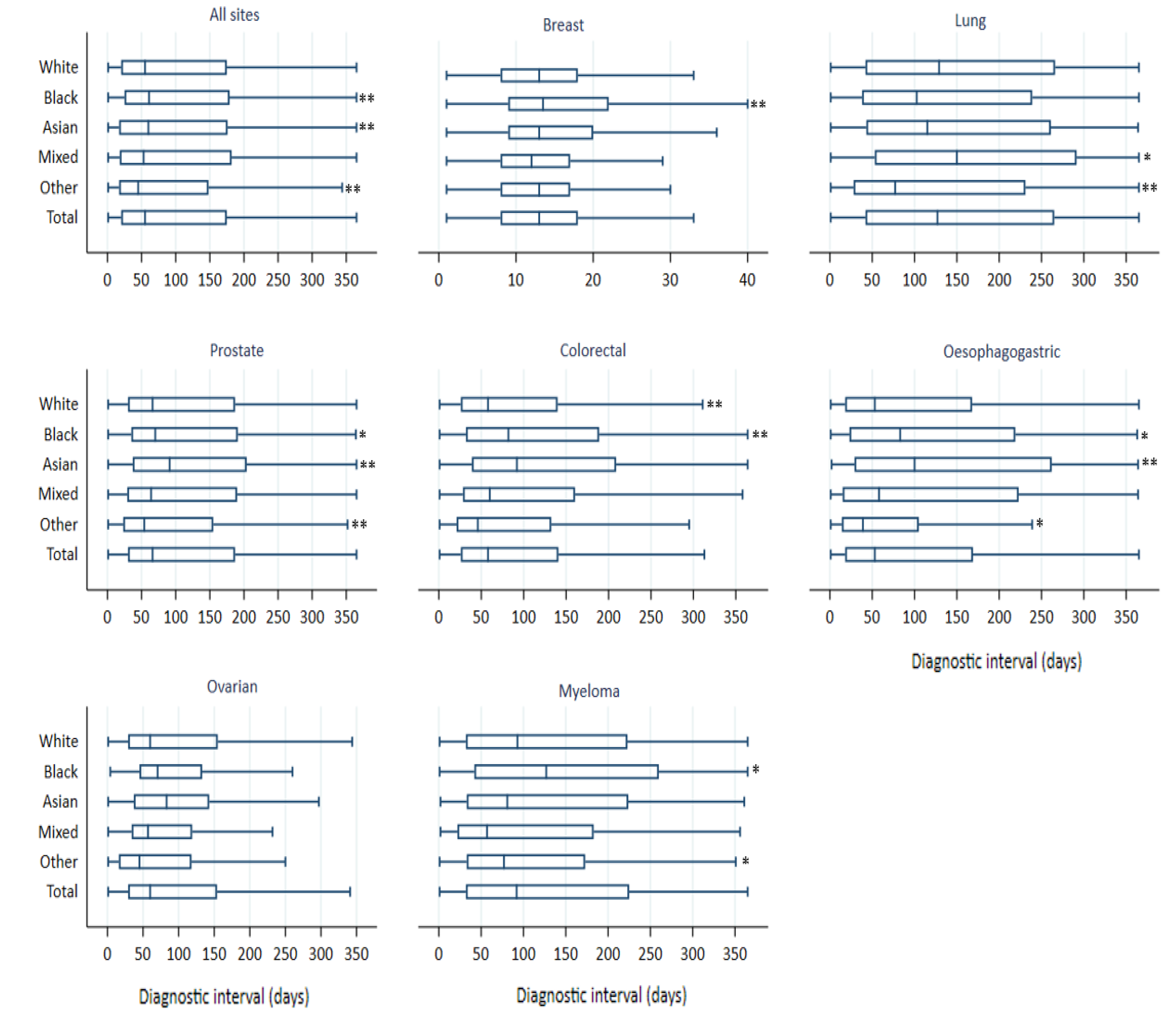
3.3. Diagnostic Interval
3.4. Ethnic Differences in Diagnostic Interval
4. Discussion
4.1. Strengths and Limitations
4.2. Comparison with Existing Evidence
4.3. Implications of the Findings
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- National Cancer Intelligence Network. Cancer Incidence and Survival by Major Ethnic Group, England, 2002–2006. London National Cancer Intelligence Network. 2009. Available online: http://www.ncin.org.uk/search/cancer+incidence+and+survival+by+major+ethnic+group (accessed on 1 June 2021).
- Jack, R.H.; Davies, E.A.; Moller, H. Breast cancer incidence, stage, treatment and survival in ethnic groups in South East England. Br. J. Cancer 2009, 100, 545–550. [Google Scholar] [CrossRef]
- Metcalfe, C.; Evans, S.; Ibrahim, F.; Patel, B.; Anson, K.; Chinegwundoh, F.; Corbishley, C.; Gillatt, D.; Kirby, R.; Muir, G.; et al. Pathways to diagnosis for Black men and White men found to have prostate cancer: The PROCESS cohort study. Br. J. Cancer 2008, 99, 1040–1045. [Google Scholar] [CrossRef]
- Saunders, C.L.; Abel, G.A.; Lyratzopoulos, G. Inequalities in reported cancer patient experience by socio-demographic characteristic and cancer site: Evidence from respondents to the English Cancer Patient Experience Survey. Eur. J. Cancer Care 2015, 24, 85–98. [Google Scholar] [CrossRef]
- Lyratzopoulos, G.; Elliott, M.; Barbiere, J.M.; Henderson, A.; Staetsky, L.; Paddison, C.; Campbell, J.; Roland, M. Understanding ethnic and other socio-demographic differences in patient experience of primary care: Evidence from the English General Practice Patient Survey. BMJ Qual. Saf. 2012, 21, 21–29. [Google Scholar] [CrossRef]
- Roberts, M.J.; Campbell, J.L.; Abel, G.A.; Davey, A.F.; Elmore, N.L.; Maramba, I.; Carter, M.; Elliott, M.N.; Roland, M.O.; Burt, J. Understanding high and low patient experience scores in primary care: Analysis of patients’ survey data for general practices and individual doctors. BMJ Br. Med. J. 2014, 349, g6034. [Google Scholar] [CrossRef]
- El Turabi, A.; Abel, G.A.; Roland, M.; Lyratzopoulos, G. Variation in reported experience of involvement in cancer treatment decision making: Evidence from the National Cancer Patient Experience Survey. Br. J. Cancer 2013, 109, 780–787. [Google Scholar] [CrossRef]
- Waller, J.; Robb, K.; Stubbings, S.; Ramirez, A.; Macleod, U.; Austoker, J.; Hiom, S.; Wardle, J. Awareness of cancer symptoms and anticipated help seeking among ethnic minority groups in England. Br. J. Cancer 2009, 101 (Suppl. 2), S24–S30. [Google Scholar] [CrossRef]
- Marlow, L.A.V.; Robb, K.A.; Simon, A.E.; Waller, J.; Wardle, J. Awareness of cancer risk factors among ethnic minority groups in England. Public Health 2012, 126, 702–709. [Google Scholar] [CrossRef]
- Campbell, C.; Douglas, A.; Williams, L.; Cezard, G.; Brewster, D.H.; Buchanan, D.; Robb, K.; Stanners, G.; Weller, D.; Steele, R.J.C.; et al. Are there ethnic and religious variations in uptake of bowel cancer screening? A retrospective cohort study among 1.7 million people in Scotland. BMJ Open 2020, 10, e037011. [Google Scholar] [CrossRef]
- Niksic, M.; Rachet, B.; Warburton, F.G.; Forbes, L.J.L. Ethnic differences in cancer symptom awareness and barriers to seeking medical help in England. Br. J. Cancer 2016, 115, 136–144. [Google Scholar] [CrossRef]
- Neal, R.D.; Allgar, V.L. Sociodemographic factors and delays in the diagnosis of six cancers: Analysis of data from the “National Survey of NHS Patients: Cancer”. Br. J. Cancer 2005, 92, 1971–1975. [Google Scholar] [CrossRef] [PubMed]
- Martins, T.; Hamilton, W.; Ukoumunne, O.C. Ethnic inequalities in time to diagnosis of cancer: A systematic review. BMC Fam. Pract. 2013, 14, 197. [Google Scholar] [CrossRef] [PubMed]
- Martins, T.; Walter, F.M.; Penfold, C.; Abel, G.; Hamilton, W. Primary care use by men with symptoms of possible prostate cancer: A multi-method study with an ethnically diverse sample in London. Eur. J. Cancer Care 2021, 30, e13482. [Google Scholar] [CrossRef] [PubMed]
- Lyratzopoulos, G.; Abel, G.A.; Barbiere, J.M.; Brown, C.H.; Rous, B.A.; Greenberg, D.C. Variation in advanced stage at diagnosis of lung and female breast cancer in an English region 2006–2009. Br. J. Cancer 2012, 106, 1068–1075. [Google Scholar] [CrossRef]
- Mendonca, S.C.; Abel, G.A.; Lyratzopoulos, G. Pre-referral GP consultations in patients subsequently diagnosed with rarer cancers: A study of patient-reported data. Br. J. Gen. Pract. 2016, 66, e171–e181. [Google Scholar] [CrossRef]
- Martins, T.; Ukoumunne, O.C.; Banks, J.; Raine, R.; Hamilton, W. Ethnic differences in patients’ preferences for prostate cancer investigation: A vignette-based survey in primary care. Br. J. Gen. Pract. 2015, 65, e161–e170. [Google Scholar] [CrossRef]
- Sadler, G.J.; Jothimani, D.; Zanetto, U.; Anderson, M.R. The effect of ethnicity on the presentation and management of oesophageal and gastric cancers: A UK perspective. Eur. J. Gastroenterol. Hepatol. 2009, 21, 996–1000. [Google Scholar] [CrossRef]
- Wolf, A.; Dedman, D.; Campbell, J.; Booth, H.; Lunn, D.; Chapman, J.; Myles, P. Data resource profile: Clinical Practice Research Datalink (CPRD) Aurum. Int. J. Epidemiol. 2019, 48, 1740g. [Google Scholar] [CrossRef]
- Herbert, A.; Wijlaars, L.; Zylbersztejn, A.; Cromwell, D.; Hardelid, P. Data Resource Profile: Hospital Episode Statistics Admitted Patient Care (HES APC). Int. J. Epidemiol. 2017, 46, 1093i. [Google Scholar] [CrossRef]
- Henson, K.E.; Elliss-Brookes, L.; Coupland, V.H.; Payne, E.; Vernon, S.; Rous, B.; Rashbass, J. Data Resource Profile: National Cancer Registration Dataset in England. Int. J. Epidemiol. 2020, 49, 16h. [Google Scholar] [CrossRef]
- Herrett, E.; Gallagher, A.M.; Bhaskaran, K.; Forbes, H.; Mathur, R.; van Staa, T.; Smeeth, L. Data Resource Profile: Clinical Practice Research Datalink (CPRD). Int. J. Epidemiol. 2015, 44, 827–836. [Google Scholar] [CrossRef] [PubMed]
- CPRD Linked Data. 2019. Available online: https://cprd.com/linked-data (accessed on 6 December 2020).
- Mathur, R.; Bhaskaran, K.; Chaturvedi, N.; Leon, D.A.; vanStaa, T.; Grundy, E.; Smeeth, L. Completeness and usability of ethnicity data in UK-based primary care and hospital databases. J. Public Health 2014, 36, 684–692. [Google Scholar] [CrossRef] [PubMed]
- Tanimola Martins, G.A.; Ukoumunne, O.C.; Mounce, L.; Price, S.; Lyratzopoulos, G.; Chinegwundoh, F.; Hamilton, W. Ethnic inequalities in routes to diagnosis of cancer: A population-based UK cohort study. Br. J. Cancer 2022, in press. [Google Scholar]
- Mathur, R.; Palla, L.; Farmer, R.E.; Chaturvedi, N.; Smeeth, L. Ethnic differences in the severity and clinical management of type 2 diabetes at time of diagnosis: A cohort study in the UK Clinical Practice Research Datalink. Diabetes Res. Clin. Pract. 2020, 160, 108006. [Google Scholar] [CrossRef]
- Watson, J.; Nicholson, B.D.; Hamilton, W.; Price, S. Identifying clinical features in primary care electronic health record studies: Methods for codelist development. BMJ Open 2017, 7, e019637. [Google Scholar] [CrossRef]
- National Institute for Health and Care Excellence. Suspected Cancer: Recognition and Referral; NG12; National Institute for Health and Care Excellence (NICE): London, UK, 2015. [Google Scholar]
- National Institute for Health and Care Excellence. Referral Guidelines for Suspected Cancer; National Institute for Health and Clinical Excellence: London, UK, 2005. [Google Scholar]
- Price, S.; Spencer, A.; Zhang, X.; Ball, S.; Lyratzopoulos, G.; Mujica-Mota, R.; Stapley, S.; Ukoumunne, O.C.; Hamilton, W. Trends in time to cancer diagnosis around the period of changing national guidance on referral of symptomatic patients: A serial cross-sectional study using UK electronic healthcare records from 2006–17. Cancer Epidemiol. 2020, 69, 101805. [Google Scholar] [CrossRef] [PubMed]
- White, B.; Renzi, C.; Rafiq, M.; Abel, G.A.; Jensen, H.; Lyratzopoulos, G. Does changing healthcare use signal opportunities for earlier detection of cancer? A review of studies using information from electronic patient records. Cancer Epidemiol. 2022, 76, 102072. [Google Scholar] [CrossRef]
- Weller, D.; Vedsted, P.; Rubin, G.; Walter, F.M.; Emery, J.; Scott, S.; Campbell, C.; Andersen, R.S.; Hamilton, W.; Olesen, F.; et al. The Aarhus statement: Improving design and reporting of studies on early cancer diagnosis. Br. J. Cancer 2012, 106, 1262–1267. [Google Scholar] [CrossRef]
- Cassell, A.; Edwards, D.; Harshfield, A.; Rhodes, K.; Brimicombe, J.; Payne, R.; Griffin, S. The epidemiology of multimorbidity in primary care: A retrospective cohort study. Br. J. Gen. Pract. 2018, 68, e245–e251. [Google Scholar] [CrossRef]
- Payne, R.A.; Mendonca, S.C.; Elliott, M.N.; Saunders, C.L.; Edwards, D.A.; Marshall, M.; Roland, M. Development and validation of the Cambridge Multimorbidity Score. Cmaj 2020, 192, E107–E114. [Google Scholar] [CrossRef]
- Din, N.U.; Ukoumunne, O.C.; Rubin, G.; Hamilton, W.; Carter, B.; Stapley, S.; Neal, R.D. Age and Gender Variations in Cancer Diagnostic Intervals in 15 Cancers: Analysis of Data from the UK Clinical Practice Research Datalink. PLoS ONE 2015, 10, e0127717. [Google Scholar]
- Mounce, L.T.A.; Price, S.; Valderas, J.M.; Hamilton, W. Comorbid conditions delay diagnosis of colorectal cancer: A cohort study using electronic primary care records. Br. J. Cancer 2017, 116, 1536–1543. [Google Scholar] [CrossRef] [PubMed]
- Renzi, C.; Lyratzopoulos, G. Comorbidity and the diagnosis of symptomatic-but-as-yet-undiagnosed cancer. Br. J. Gen. Pract. 2020, 70, e598–e599. [Google Scholar] [CrossRef] [PubMed]
- Hansen, R.P.; Olesen, F.; Sørensen, H.T.; Sokolowski, I.; Søndergaard, J. Socioeconomic patient characteristics predict delay in cancer diagnosis: A Danish cohort study. BMC Health Serv. Res. 2008, 8, 49. [Google Scholar] [CrossRef]
- Williams, P.; Murchie, P.; Bond, C. Patient and primary care delays in the diagnostic pathway of gynaecological cancers: A systematic review of influencing factors. Br. J. Gen. Pract. 2019, 69, e106–e111. [Google Scholar] [CrossRef]
- von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement: Guidelines for reporting observational studies. Prev. Med. 2007, 45, 247–251. [Google Scholar] [CrossRef]
- Saunders, C.L.; Abel, G.A.; El Turabi, A.; Ahmed, F.; Lyratzopoulos, G. Accuracy of routinely recorded ethnic group information compared with self-reported ethnicity: Evidence from the English Cancer Patient Experience survey. BMJ Open 2013, 3, e002882. [Google Scholar] [CrossRef]
- Swann, R.; McPhail, S.; Witt, J.; Shand, B.; Abel, G.A.; Hiom, S.; Rashbass, J.; Lyratzopoulos, G.; Rubin, G.; The National Cancer Diagnosis Audit Steering Group; et al. Diagnosing cancer in primary care: Results from the National Cancer Diagnosis Audit. Br. J. Gen. Pract. 2018, 68, e63–e72. [Google Scholar] [CrossRef]
- Neal, R.D.; Din, N.U.; Hamilton, W.; Ukoumunne, O.C.; Carter, B.; Stapley, S.; Rubin, G. Comparison of cancer diagnostic intervals before and after implementation of NICE guidelines: Analysis of data from the UK General Practice Research Database. Br. J. Cancer 2014, 110, 584–592. [Google Scholar] [CrossRef]
- Velikova, G.; Booth, L.; Johnston, C.; Forman, D.; Selby, P. Breast cancer outcomes in South Asian population of West Yorkshire. Br. J. Cancer 2004, 90, 1926–1932. [Google Scholar] [CrossRef][Green Version]
- National Health Services. The NHS Long Term Plan 2019. Available online: https://www.longtermplan.nhs.uk/publication/nhs-long-term-plan/ (accessed on 14 December 2021).
- Calanzani, N.; Chang, A.; Van Melle, M.; Pannebakker, M.M.; Funston, G.; Walter, F.M. Recognising Colorectal Cancer in Primary Care. Adv. Ther. 2021, 38, 2732–2746. [Google Scholar] [CrossRef] [PubMed]
- The National Institute for Health and Care Excellence (NICE). Quantitative Faecal Immunochemical Tests to Guide Referral for Colorectal Cancer in Primary Care (DG30). 2017. Available online: https://www.nice.org.uk/guidance/dg30 (accessed on 14 December 2021).
- Buccafusca, G.; Proserpio, I.; Tralongo, A.C.; Rametta Giuliano, S.; Tralongo, P. Early colorectal cancer: Diagnosis, treatment and survivorship care. Crit. Rev. Oncol. Hematol. 2019, 136, 20–30. [Google Scholar] [CrossRef] [PubMed]
- Mendonca, S.C.; Abel, G.A.; Gildea, C.; McPhail, S.; Peake, M.D.; Rubin, G.; Singh, H.; Hamilton, W.; Walter, F.M.; Roland, M.O.; et al. Associations between general practice characteristics with use of urgent referrals for suspected cancer and endoscopies: A cross-sectional ecological study. Fam. Pract. 2018, 36, 573–580. [Google Scholar] [CrossRef] [PubMed]
- Morris, S.; Baio, G.; Kendall, E.; von Wagner, C.; Wardle, J.; Atkin, W.; Halloran, S.P.; Handley, G.; Logan, R.F.; Obichere, A.; et al. Socioeconomic variation in uptake of colonoscopy following a positive faecal occult blood test result: A retrospective analysis of the NHS Bowel Cancer Screening Programme. Br. J. Cancer 2012, 107, 765–771. [Google Scholar] [CrossRef]
- Sud, A.; Torr, B.; Jones, M.E.; Broggio, J.; Scott, S.; Loveday, C.; Garrett, A.; Gronthoud, F.; Nicol, D.L.; Jhanji, S.; et al. Effect of delays in the 2-week-wait cancer referral pathway during the COVID-19 pandemic on cancer survival in the UK: A modelling study. Lancet Oncol. 2020, 21, 1035–1044. [Google Scholar] [CrossRef]
| Cancer Site | NICE Features |
|---|---|
| Breast | Breast pain, breast lump, breast skin changes (peau d’orange), nipple discharge, nipple retraction, lymphadenopathy (axilla). |
| Lung | Appetite loss, chest infection, chest pain, chest signs consistent with lung cancer, cough, dyspnoea, fatigue, features suggestive of lung metastases’ finger clubbing, haemoptysis, hoarseness, lymphadenopathy (supraclavicular, cervical), shoulder pain, signs of superior vena cava obstruction, stridor, thrombocytosis, weight loss, x-ray findings suggestive of lung cancer. |
| Prostate | Abnormal digital rectal examination, erectile dysfunction, haematuria (visible), nocturia, raised prostate specific antigen (PSA) above age-specific value, urinary frequency, urinary hesitancy, urinary retention, Urinary urgency. |
| Colorectal | Abdominal mass, abdominal pain, change in bowel habit, faecal occult blood, iron-deficiency anaemia, rectal bleeding, rectal mass, weight loss. |
| Oesophagogastric | Back pain, dyspepsia, dysphagia, haematemesis, gastrointestinal bleeding, low haemoglobin, nausea, reflux, suspicious barium meal results, thrombocytosis, upper abdominal mass, upper abdominal pain, vomiting, weight loss. |
| Ovary | Fatigue, abdominal distension/bloating, abdominal or pelvic mass, abdominal pain, abdominal/pelvic mass, appetite loss, ascites, back pain, change in bowel habit, constipation, pelvic pain, raised ca125, urinary urgency, urinary frequency, weight loss. |
| Myeloma | Bone pain, back pain, Bence-jones protein, abnormal erythrocyte sedimentation rate, hypercalcaemia, abnormal white cell count, pathological fracture, plasma viscosity consistent with myeloma, protein electrophoresis suggesting myeloma, spinal cord compression suspected of being caused by myeloma. |
| White | Black | Asian | Mixed | Other | All | ||
|---|---|---|---|---|---|---|---|
| Age | Median (IQR Years) | 72 (63–80) | 67 (56–76) | 67 (56–75) | 70 (62–78) | 72 (63–81) | 72 (63–80) |
| Sex | Male n (%) | 67,263 (57.6) | 1768 (70.1) | 1175 (54.4) | 1352 (58.2) | 1650 (55.3) | 73,208 (57.8) |
| IMD n (%) * | 1 (least deprived) | 28,413 (24.4) | 122 (4.84) | 339 (15.7) | 453 (19.5) | 735 (24.6) | 30,062 (23.8) |
| 2 | 26,240 (22.5) | 164 (6.51) | 360 (16.7) | 424 (18.3) | 706 (23.7) | 27,894 (22.0) | |
| 3 | 23,353 (19.9) | 424 (16.8) | 465 (21.5) | 471 (20.3) | 587 (19.7) | 25,200 (19.9) | |
| 4 | 20,029 (17.2) | 717 (28.4) | 462 (21.4) | 502 (21.6) | 512 (17.2) | 22,222 (17.6) | |
| 5 (most deprived) | 18,662 (16.0) | 1094 (43.4) | 533 (24.7) | 471 (20.3) | 443 (14.9) | 21,203 (16.8) | |
| Morbidity score ** n (%) | 0–None | 7814 (6.70) | 144 (5.71) | 135 (6.25) | 115 (4.95) | 349 (11.7) | 8557 (6.76) |
| 1 | 17,901 (15.4) | 395 (15.7) | 299 (13.9) | 347 (14.9) | 560 (18.8) | 19,502 (15.4) | |
| 2 | 26,338 (22.6) | 668 (26.5) | 503 (23.3) | 451 (19.4) | 825 (27.6) | 28,785 (22.7) | |
| 3 | 29,912 (25.5) | 683 (27.1) | 641 (29.7) | 622 (26.8) | 718 (24.1) | 32,576 (25.7) | |
| 4 (most score) | 34,675 (29.7) | 632 (25.1) | 581 (26.9) | 786 (33.9) | 533 (17.9) | 37,207 (29.4) | |
| Sites | Breast | 18,280 (15.7) | 378 (14.9) | 538 (24.9) | 441 (19.0) | 365 (12.2) | 20,002 (15.8) |
| Lung | 27,926 (23.9) | 282 (11.2) | 371 (17.2) | 460 (19.8) | 939 (31.5) | 29,978 (23.7) | |
| Prostate | 33,256 (28.5) | 1205 (47.8) | 572 (26.5) | 777 (33.5) | 642 (21.5) | 36,452 (28.8) | |
| Colorectal | 20,586 (17.7) | 311 (12.3) | 342 (15.8) | 388 (16.7) | 524 (17.5) | 22,151 (17.5) | |
| Oesophagogastric | 10,102 (8.66) | 174 (6.90) | 161 (7.46) | 135 (5.82) | 355 (11.9) | 10,927 (8.63) | |
| Ovarian | 3,644 (3.12) | 32 (1.27) | 94 (4.35) | 65 (2.80) | 99 (3.32) | 3,934 (3.11) | |
| Myeloma | 2,846 (2.44) | 140 (5.55) | 81 (3.75) | 55 (2.37) | 61 (2.04) | 3,183 (2.51) | |
| Total | 116,640 (92.1) | 2522 (1.99) | 2159 (1.71) | 2321 (1.83) | 2985 (2.36) | 126,627 (100) |
| Sites | Ethnicity | N | Crude Time Ratio | Adjusted Time Ratio | 95% CI | p–Value | Average Marginal Difference | 95% CI |
|---|---|---|---|---|---|---|---|---|
| All sites | White | 116,640 | ||||||
| Black | 2522 | 1.05 | 1.10 | 1.05–1.14 | <0.001 | 6.57 | 3.54–9.60 | |
| Asian | 2159 | 1.02 | 1.16 | 1.10–1.22 | <0.001 | 11.0 | 6.99–15.0 | |
| Mixed | 2321 | 1.00 | 1.02 | 0.98–1.07 | 0.29 | 1.65 | −1.47–4.78 | |
| Other | 2985 | 0.88 | 0.88 | 0.84–0.91 | <0.001 | −8.57 | −11.0––6.08 | |
| Breast | White | 18,280 | ||||||
| Black | 378 | 1.15 | 1.12 | 1.04–1.21 | 0.004 | 1.55 | 0.42–2.67 | |
| Asian | 538 | 1.09 | 1.06 | 0.99–1.13 | 0.11 | 0.72 | −0.19–1.62 | |
| Mixed | 441 | 1.00 | 0.99 | 0.92–1.06 | 0.81 | −0.11 | −0.99–0.77 | |
| Other | 365 | 0.97 | 0.99 | 0.92–1.06 | 0.72 | −0.18 | −1.11–0.76 | |
| Lung | White | 27,926 | ||||||
| Black | 282 | 0.90 | 0.94 | 0.84–1.04 | 0.23 | −7.26 | −18.6–4.23 | |
| Asian | 371 | 0.97 | 0.98 | 0.91–1.05 | 0.51 | −2.83 | −11.2–5.54 | |
| Mixed | 460 | 1.09 | 1.06 | 0.99–1.12 | 0.08 | 6.75 | −0.85–13.9 | |
| Other | 939 | 0.83 | 0.90 | 0.85–0.95 | <0.001 | −11.5 | −17.3––5.62 | |
| Prostate | White | 33,256 | ||||||
| Black | 1205 | 1.03 | 1.09 | 1.03–1.15 | 0.005 | 6.63 | 1.86–11.4 | |
| Asian | 572 | 1.16 | 1.17 | 1.09–1.27 | <0.001 | 13.2 | 6.27–20.0 | |
| Mixed | 777 | 0.99 | 1.00 | 0.93–1.08 | 0.92 | 0.31 | −5.52–6.15 | |
| Other | 642 | 0.87 | 0.87 | 0.80–0.95 | 0.002 | −9.68 | −15.5–3.88 | |
| Colorectal | White | 20,586 | ||||||
| Black | 311 | 1.22 | 1.22 | 1.09–1.35 | <0.001 | 14.1 | 5.69–22.4 | |
| Asian | 342 | 1.33 | 1.37 | 1.24–1.51 | <0.001 | 24.1 | 15.5–32.8 | |
| Mixed | 388 | 1.08 | 1.06 | 0.96–1.17 | 0.27 | 3.85 | −3.25–10.9 | |
| Other | 524 | 0.88 | 0.93 | 0.85–1.02 | 0.14 | −4.51 | −10.3–1.24 | |
| Oesophago– gastric | White | 10,102 | ||||||
| Black | 174 | 1.26 | 1.21 | 1.05–1.41 | 0.01 | 13.1 | 2.07–24.1 | |
| Asian | 161 | 1.41 | 1.33 | 1.14–1.55 | <0.001 | 19.9 | 7.51–32.4 | |
| Mixed | 135 | 1.12 | 1.04 | 0.86–1.25 | 0.73 | 2.11 | −9.98–14.2 | |
| Other | 355 | 0.77 | 0.84 | 0.74–0.97 | 0.01 | −9.54 | −16.5––2.56 | |
| Ovary | White | 3,644 | ||||||
| Black | 32 | 0.99 | 0.87 | 0.66–1.18 | 0.39 | −8.78 | −27.4–9.83 | |
| Asian | 94 | 1.09 | 1.04 | 0.88–1.24 | 0.65 | 2.91 | −9.99–15.8 | |
| Mixed | 65 | 0.96 | 0.96 | 0.73–1.25 | 0.73 | −3.29 | −21.8–15.2 | |
| Other | 99 | 0.83 | 0.87 | 0.70–1.09 | 0.22 | −9.13 | −22.8–4.59 | |
| Myeloma | White | 2846 | ||||||
| Black | 140 | 1.15 | 1.16 | 1.00–1.35 | 0.05 | 15.3 | −0.99–31.5 | |
| Asian | 81 | 0.94 | 0.99 | 0.82–1.20 | 0.92 | −0.90 | −18.9–17.1 | |
| Mixed | 55 | 0.86 | 0.82 | 0.62–1.07 | 0.14 | −17.2 | −38.1–3.68 | |
| Other | 61 | 0.84 | 0.83 | 0.66–1.03 | 0.09 | −16.3 | −33.6–0.91 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martins, T.; Abel, G.; Ukoumunne, O.C.; Price, S.; Lyratzopoulos, G.; Chinegwundoh, F.; Hamilton, W. Assessing Ethnic Inequalities in Diagnostic Interval of Common Cancers: A Population-Based UK Cohort Study. Cancers 2022, 14, 3085. https://doi.org/10.3390/cancers14133085
Martins T, Abel G, Ukoumunne OC, Price S, Lyratzopoulos G, Chinegwundoh F, Hamilton W. Assessing Ethnic Inequalities in Diagnostic Interval of Common Cancers: A Population-Based UK Cohort Study. Cancers. 2022; 14(13):3085. https://doi.org/10.3390/cancers14133085
Chicago/Turabian StyleMartins, Tanimola, Gary Abel, Obioha C. Ukoumunne, Sarah Price, Georgios Lyratzopoulos, Frank Chinegwundoh, and William Hamilton. 2022. "Assessing Ethnic Inequalities in Diagnostic Interval of Common Cancers: A Population-Based UK Cohort Study" Cancers 14, no. 13: 3085. https://doi.org/10.3390/cancers14133085
APA StyleMartins, T., Abel, G., Ukoumunne, O. C., Price, S., Lyratzopoulos, G., Chinegwundoh, F., & Hamilton, W. (2022). Assessing Ethnic Inequalities in Diagnostic Interval of Common Cancers: A Population-Based UK Cohort Study. Cancers, 14(13), 3085. https://doi.org/10.3390/cancers14133085






