Detection of IDH1 Mutations in Plasma Using BEAMing Technology in Patients with Gliomas
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
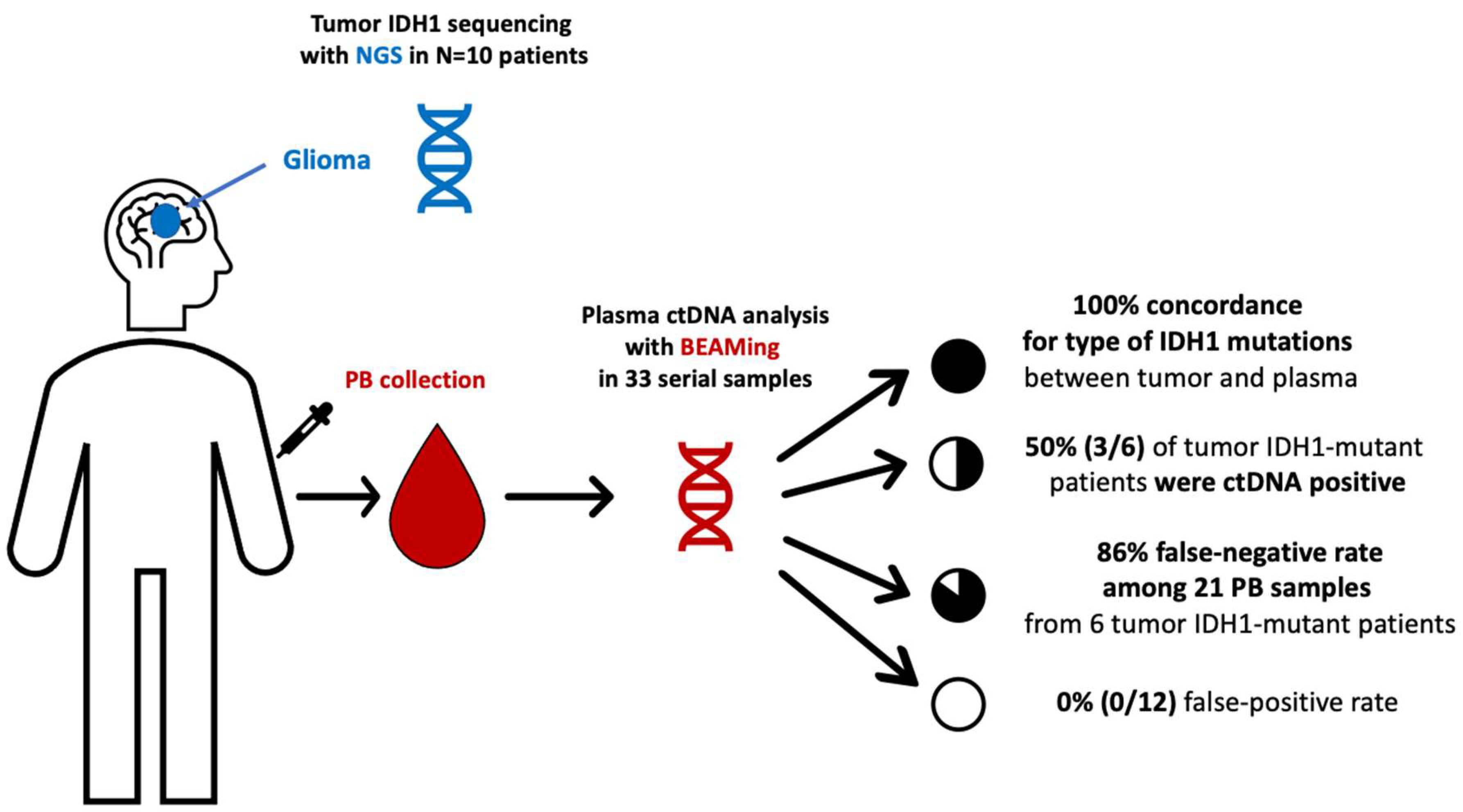
2.1. Patient Enrollment and Sampling
2.2. Immunohistochemistry
2.3. DNA Extraction from Tissue and Plasma Samples
2.4. Next Generation Sequencing (NGS) Study of IDH1 and IDH2 Mutations in Tumor Tissue
2.5. ctDNA BEAMing Digital PCR Analyses of IDH1 Mutations in Plasma
2.6. Tumor Response Assessment
2.7. Ethical Considerations
3. Results
3.1. Baseline Characteristics of the Patients
3.2. Immunohistochemistry Study of the IDH1-R132H Mutation in Tumor Tissue
3.3. NGS Study of IDH1 and IDH2 Mutations in Tumor Tissue
3.4. BEAMing Study of IDH1 Mutations in Plasma
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eckel-Passow, J.E.; Lachance, D.H.; Molinaro, A.M.; Walsh, K.M.; Decker, P.A.; Sicotte, H.; Pekmezci, M.; Rice, T.W.; Kosel, M.L.; Smirnov, I.V.; et al. Glioma Groups Based on 1p/19q, IDH, and TERT Promoter Mutations in Tumors. N. Engl. J. Med. 2015, 372, 2499–2508. [Google Scholar] [CrossRef] [Green Version]
- Cancer Genome Atlas Research Network. Comprehensive, Integrative Genomic Analysis of Diffuse Lower-Grade Gliomas. N. Engl. J. Med. 2015, 372, 2481–2498. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Louis, D.N.; Perry, A.; Wesseling, P.; Brat, D.J.; Cree, I.A.; Figarella-Branger, D.; Hawkins, C.; Ng, H.K.; Pfister, S.M.; Reifenberger, G.; et al. The 2021 WHO classification of tumors of the central nervous system: A summary. Neuro-Oncology 2021, 23, 1231–1251. [Google Scholar] [CrossRef] [PubMed]
- Pappula, A.L.; Rasheed, S.; Mirzaei, G.; Petreaca, R.C.; Bouley, R.A. A genome-wide profiling of glioma patients with an IDH1 mutation using the Catalogue of Somatic Mutations In Cancer Database. Cancers 2021, 13, 4299. [Google Scholar] [CrossRef]
- Hartmann, C.; Meyer, J.; Balss, J.; Capper, D.; Mueller, W.; Christians, A.; Felsberg, J.; Wolter, M.; Mawrin, C.; Wick, W.; et al. Type and frequency of IDH1 and IDH2 mutations are related to astrocytic and oligodendroglial differentiation and age: A study of 1010 diffuse gliomas. Acta Neuropathol. 2009, 118, 469–474. [Google Scholar] [CrossRef] [Green Version]
- Sulkowski, P.L.; Corso, C.D.; Robinson, N.D.; Scanlon, S.E.; Purshouse, K.R.; Bai, H.; Liu, Y.; Sundaram, R.K.; Hegan, D.C.; Fons, N.R.; et al. 2-Hydroxyglutarate produced by neomorphic IDH mutations suppresses homologous recombination and induces PARP inhibitor sensitivity. Sci. Transl. Med. 2017, 9, eaal2463. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Watanabe, T.; Nobusawa, S.; Kleihues, P.; Ohgaki, H. IDH1 mutations are early events in the development of astrocytomas and oligodendrogliomas. Am. J. Pathol. 2009, 174, 1149–1153. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Inoue, S.; Li, W.Y.; Tseng, A.; Beerman, I.; Elia, A.J.; Bendall, S.C.; Lemonnier, F.; Kron, K.J.; Cescon, D.W.; Hao, Z.; et al. Mutant IDH1 downregulates ATM and alters DNA repair and sensitivity to DNA damage independent of TET2. Cancer Cell 2016, 30, 337–348. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- DiNardo, C.D.; Stein, E.M.; de Botton, S.; Roboz, G.J.; Altman, J.K.; Mims, A.S.; Swords, R.; Collins, R.H.; Mannis, G.N.; Pollyea, D.A.; et al. Durable Remissions with Ivosidenib in IDH1 -Mutated Relapsed or Refractory AML. New Engl. J. Med. 2018, 378, 2386–2398. [Google Scholar] [CrossRef]
- Stein, E.M.; Dinardo, C.D.; Pollyea, D.A.; Fathi, A.T.; Roboz, G.J.; Altman, J.K.; Stone, R.M.; DeAngelo, D.J.; Levine, R.L.; Flinn, I.W.; et al. Enasidenib in mutant IDH2 relapsed or refractory acute myeloid leukemia. Blood 2017, 130, 722–731. [Google Scholar] [CrossRef] [PubMed]
- Mellinghoff, I.K.; Ellingson, B.M.; Touat, M.; Maher, E.; De La Fuente, M.I.; Holdhoff, M.; Cote, G.M.; Burris, H.; Janku, F.; Young, R.J.; et al. Ivosidenib in Isocitrate Dehydrogenase 1-Mutated Advanced Glioma. J. Clin. Oncol. 2020, 38, 3398–3406. [Google Scholar] [CrossRef] [PubMed]
- Tejera, D.; Kushnirsky, M.; Gultekin, S.H.; Lu, M.; Steelman, L.; de la Fuente, M.I. Ivosidenib, an IDH1 inhibitor, in a patient with recurrent, IDH1-mutant glioblastoma: A case report from a Phase I study. CNS Oncol. 2020, 8, CNS62. [Google Scholar] [CrossRef]
- Boisselier, B.; Pérez-Larraya, J.G.; Rossetto, M.; Labussière, M.; Ciccarino, P.; Marie, Y.; Delattre, J.-Y.; Sanson, M. Detection of IDH1 mutation in the plasma of patients with glioma. Neurology 2012, 79, 1693–1698. [Google Scholar] [CrossRef] [PubMed]
- García-Romero, N.; Carrión-Navarro, J.; Esteban-Rubio, S.; Lázaro-Ibáñez, E.; Peris-Celda, M.; Alonso, M.M.; Guzmán-De-Villoria, J.; Fernández-Carballal, C.; de Mendivil, A.O.; García-Duque, S.; et al. DNA sequences within glioma-derived extracellular vesicles can cross the intact blood-brain barrier and be detected in peripheral blood of patients. Oncotarget 2017, 8, 1416–1428. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miller, A.; Shah, R.; Pentsova, E.I.; Pourmaleki, M.; Briggs, S.; Distefano, N.; Zheng, Y.; Skakodub, A.; Mehta, S.A.; Campos, C.; et al. Tracking tumour evolution in glioma through liquid biopsies of cerebrospinal fluid. Nature 2019, 565, 654–658. [Google Scholar] [CrossRef] [PubMed]
- Eibl, R.H.; Schneemann, M. Liquid Biopsy and Primary Brain Tumors. Cancers 2021, 13, 5429. [Google Scholar] [CrossRef] [PubMed]
- Beynon, C.; Wei, S.; Radbruch, A.; Capper, D.; Unterberg, A.W.; Kiening, K.L. Preoperative assessment of haemostasis in patients undergoing stereotactic brain biopsy. J. Clin. Neurosci. 2018, 53, 112–116. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Ricarte, F.; Mayor, R.; Martínez-Sáez, E.; Rubio-Pérez, C.; Pineda, E.; Cordero, E.; Cicuéndez, M.; Poca, M.A.; López-Bigas, N.; Cajal, S.R.Y.; et al. Molecular diagnosis of diffuse gliomas through sequencing of cell-free circulating tumor DNA from cerebrospinal fluid. Clin. Cancer Res. 2018, 24, 2812–2819. [Google Scholar] [CrossRef] [Green Version]
- Juratli, T.A.; Stasik, S.; Zolal, A.; Schuster, C.; Richter, S.; Daubner, D.; Juratli, M.A.; Thowe, R.T.; Hennig, S.; Makina, M.; et al. TERT promoter mutation detection in cell-free tumor-derived DNA in patients with IDH wild-type glioblastomas: A pilot prospective study. Clin. Cancer Res. 2018, 24, 5282–5291. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Panditharatna, E.; Kilburn, L.B.; Aboian, M.S.; Kambhampati, M.; Gordish-Dressman, H.; Magge, S.N.; Gupta, N.; Myseros, J.S.; Hwang, E.I.; Kline, C.; et al. Clinically relevant and minimally invasive tumor surveillance of pediatric diffuse midline gliomas using patient-derived liquid biopsy. Clin. Cancer Res. 2018, 24, 5850–5859. [Google Scholar] [CrossRef] [Green Version]
- Nassiri, F.; Chakravarthy, A.; Feng, S.; Shen, S.Y.; Nejad, R.; Zuccato, J.A.; Voisin, M.R.; Patil, V.; Horbinski, C.; Aldape, K.; et al. Detection and discrimination of intracranial tumors using plasma cell-free DNA methylomes. Nat. Med. 2020, 26, 1044–1047. [Google Scholar] [CrossRef] [PubMed]
- Mirimanoff, R.-O.; Gorlia, T.; Mason, W.; Bent, M.V.D.; Kortmann, R.-D.; Fisher, B.; Reni, M.; Brandes, A.; Curschmann, J.; Villà, S.; et al. Radiotherapy and temozolomide for newly diagnosed glioblastoma: Recursive partitioning analysis of the EORTC 26981/22981-NCIC CE3 phase III randomized trial. J. Clin. Oncol. 2006, 24, 2563–2569. [Google Scholar] [CrossRef] [PubMed]
- Müller, C.; Holtschmidt, J.; Auer, M.; Heitzer, E.; Lamszus, K.; Schulte, A.; Matschke, J.; Langer-Freitag, S.; Gasch, C.; Stoupiec, M.; et al. Hematogenous dissemination of glioblastoma multiforme. Sci. Transl. Med. 2014, 6, 247ra101. [Google Scholar] [CrossRef] [PubMed]
- Krol, I.; Castro-Giner, F.; Maurer, M.; Gkountela, S.; Szczerba, B.M.; Scherrer, R.; Coleman, N.; Carreira, S.; Bachmann, F.; Anderson, S.; et al. Detection of circulating tumour cell clusters in human glioblastoma. Br. J. Cancer 2018, 119, 487–491. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bang-Christensen, S.R.; Pedersen, R.S.; Pereira, M.A.; Clausen, T.M.; Løppke, C.; Sand, N.T.; Ahrens, T.D.; Jørgensen, A.M.; Lim, Y.C.; Goksøyr, L.; et al. Capture and Detection of Circulating Glioma Cells Using the Recombinant VAR2CSA Malaria Protein. Cells 2019, 8, 998. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, T.; Xu, H.; Huang, M.; Ma, W.; Saxena, D.; Lustig, R.A.; Alonso-Basanta, M.; Zhang, Z.; O’Rourke, D.M.; Zhang, L.; et al. Circulating glioma cells exhibit stem cell-like properties. Cancer Res. 2018, 78, 6632–6642. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sullivan, J.P.; Nahed, B.V.; Madden, M.W.; Oliveira, S.M.; Springer, S.; Bhere, D.; Chi, A.S.; Wakimoto, H.; Rothenberg, S.M.; Sequist, L.V.; et al. Brain tumor cells in circulation are enriched for mesenchymal gene expression. Cancer Discov. 2014, 4, 1299–1309. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schwaederle, M.; Chattopadhyay, R.; Kato, S.; Fanta, P.T.; Banks, K.; Choi, I.S.; Piccioni, D.E.; Ikeda, S.; Talasaz, A.; Lanman, R.B.; et al. Genomic alterations in circulating tumor DNA from diverse cancer patients identified by next-generation sequencing. Cancer Res. 2017, 77, 5419–5427. [Google Scholar] [CrossRef] [Green Version]
- Bettegowda, C.; Sausen, M.; Leary, R.J.; Kinde, I.; Wang, Y.; Agrawal, N.; Bartlett, B.R.; Wang, H.; Luber, B.; Alani, R.M.; et al. Detection of circulating tumor DNA in early- and late-stage human malignancies. Sci. Transl. Med. 2014, 6, 224. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lavon, I.; Refael, M.; Zelikovitch, B.; Shalom, E.; Siegal, T. Serum DNA can define tumor-specific genetic and Epigenetic Markers in Gliomas of Various Grades. Neuro-Oncology 2010, 12, 173–180. [Google Scholar] [CrossRef] [Green Version]
- Diehl, F.; Schmidt, K.; Choti, M.A.; Romans, K.; Goodman, S.; Li, M.; Thornton, K.; Agrawal, N.; Sokoll, L.; Szabo, S.A.; et al. Circulating mutant DNA to assess tumor dynamics. Nat. Med. 2008, 14, 985–990. [Google Scholar] [CrossRef]
- Vidal, J.; Muinelo, L.; Dalmases, A.; Jones, F.; Edelstein, D.; Iglesias, M.; Orrillo, M.; Abalo, A.; Rodríguez, C.; Brozos, E.; et al. Plasma ctDNA RAS mutation analysis for the diagnosis and treatment monitoring of metastatic colorectal cancer patients. Ann. Oncol. 2017, 28, 1325–1332. [Google Scholar] [CrossRef]
- García-Foncillas, J.; Tabernero, J.; Élez, E.; Aranda, E.; Benavides, M.; Camps, C.; Jantus-Lewintre, E.; López, R.; Muinelo-Romay, L.; Montagut, C.; et al. Prospective multicenter real-world RAS mutation comparison between OncoBEAM-based liquid biopsy and tissue analysis in metastatic colorectal cancer. Br. J. Cancer 2018, 119, 1464–1470. [Google Scholar] [CrossRef]
- Vivancos, A.; Aranda, E.; Benavides, M.; Elez, E.; Gómez-España, M.A.; Toledano, M.; Alvarez, M.; Parrado, M.R.C.; García-Barberán, V.; Diaz-Rubio, E. Comparison of the Clinical Sensitivity of the Idylla Platform and the OncoBEAM RAS CRC Assay for KRAS Mutation Detection in Liquid Biopsy Samples. Sci. Rep. 2019, 9, 8976. [Google Scholar] [CrossRef] [Green Version]
- Tabernero, J.; Lenz, H.-J.; Siena, S.; Sobrero, A.; Falcone, A.; Ychou, M.; Humblet, Y.; Bouché, O.; Mineur, L.; Barone, C.; et al. Analysis of circulating DNA and protein biomarkers to predict the clinical activity of regorafenib and assess prognosis in patients with metastatic colorectal cancer: A retrospective, exploratory analysis of the CORRECT trial. Lancet Oncol. 2015, 16, 937–948. [Google Scholar] [CrossRef]
- McShane, L.M.; Altman, D.G.; Sauerbrei, W.; Taube, S.E.; Gion, M.; Clark, G.M. REporting recommendations for tumour MARKer prognostic studies (REMARK). Eur. J. Cancer 2005, 41, 1690–1696. [Google Scholar] [CrossRef] [Green Version]
- Capper, D.; Weißert, S.; Balss, J.; Habel, A.; Meyer, J.; Jã¤Ger, D.; Ackermann, U.; Tessmer, C.; Korshunov, A.; Zentgraf, H.; et al. Capper D et al. Characterization of R132H mutation-specific IDH1 antibody binding in brain tumors. Brain Pathol. 2010, 20, 245–254. [Google Scholar] [CrossRef]
- Ravi, R.K.; Walton, K.; Khosroheidari, M. MiSeq: A Next Generation Sequencing Platform for Genomic Analysis. Methods Mol. Biol. 2018, 1706, 223–232. [Google Scholar] [CrossRef]
- Dressman, D.; Yan, H.; Traverso, G.; Kinzler, K.W.; Vogelstein, B. Transforming single DNA molecules into fluorescent magnetic particles for detection and enumeration of genetic variations. Proc. Natl. Acad. Sci. USA 2003, 100, 8817–8822. [Google Scholar] [CrossRef] [Green Version]
- Roboz, G.J.; Dinardo, C.D.; Stein, E.M.; De Botton, S.; Mims, A.S.; Prince, G.T.; Altman, J.K.; Arellano, M.L.; Donnellan, W.; Erba, H.P.; et al. Ivosidenib induces deep durable remissions in patients with newly diagnosed IDH1-mutant acute myeloid leukemia. Blood 2020, 135, 463–471. [Google Scholar] [CrossRef] [Green Version]
- Wen, P.Y.; Chang, S.M.; Bent, M.V.D.; Vogelbaum, M.A.; Macdonald, D.R.; Lee, E.Q. Response assessment in neuro-oncology clinical trials. J. Clin. Oncol. 2017, 35, 2439–2449. [Google Scholar] [CrossRef]
- Chukwueke, U.N.; Wen, P.Y. Use of the Response Assessment in Neuro-Oncology (RANO) criteria in clinical trials and clinical practice. CNS Oncol. 2019, 8, CNS28. [Google Scholar] [CrossRef] [Green Version]
- Eichinger, P.; Alberts, E.; Delbridge, C.; Trebeschi, S.; Valentinitsch, A.; Bette, S.; Huber, T.; Gempt, J.; Meyer, B.; Schlegel, J.; et al. Diffusion tensor image features predict IDH genotype in newly diagnosed WHO grade II/III gliomas. Sci. Rep. 2017, 7, 13396. [Google Scholar] [CrossRef]
- Bell, E.H.; Zhang, P.; Shaw, E.G.; Buckner, J.C.; Barger, G.R.; Bullard, D.E.; Mehta, M.P.; Gilbert, M.R.; Brown, P.D.; Stelzer, K.J.; et al. Comprehensive Genomic Analysis in NRG Oncology/RTOG 9802: A Phase III Trial of Radiation Versus Radiation Plus Procarbazine, Lomustine (CCNU), and Vincristine in High-Risk Low-Grade Glioma. J. Clin. Oncol. 2020, 38, 3407–3417. [Google Scholar] [CrossRef]
- Intlekofer, A.M.; Shih, A.H.; Wang, B.; Nazir, A.; Rustenburg, A.S.; Albanese, S.K.; Patel, M.; Famulare, C.; Correa, F.M.; Takemoto, N.; et al. Acquired resistance to IDH inhibition through trans or cis dimer-interface mutations. Nature 2018, 559, 125–129. [Google Scholar] [CrossRef]
- Choe, S.; Wang, H.; Dinardo, C.D.; Stein, E.M.; De Botton, S.; Roboz, G.J.; Altman, J.K.; Mims, A.S.; Watts, J.M.; Pollyea, D.A.; et al. Molecular mechanisms mediating relapse following ivosidenib monotherapy in IDH1-mutant relapsed or refractory AML. Blood Adv. 2020, 4, 1894–1905. [Google Scholar] [CrossRef]
- Yan, H.; Parsons, D.W.; Jin, G.; McLendon, R.; Rasheed, B.A.; Yuan, W.; Kos, I.; Batinic-Haberle, I.; Jones, S.; Riggins, G.J.; et al. IDH1 and IDH2 mutations in gliomas. N. Engl. J. Med. 2009, 360, 765–773. [Google Scholar] [CrossRef]
| Variable | Value | ||
|---|---|---|---|
| N | 10 | ||
| Male:Female | 6:4 | ||
| Median Age (range) | At initial diagnosis | 51 (31–78) | |
| At blood collection | 58 (34–78) | ||
| Histology † | Glioblastoma, IDH-wild-type | 4 | |
| Astrocytoma (grade 4), IDH-mutant | 3 | ||
| Astrocytoma (grade 3), IDH-mutant | 1 | ||
| Astrocytoma (grade 2), IDH-mutant | 1 | ||
| Oligodendroglioma (grade 2), IDH-mutant, and 1p/19q-codeleted | 1 | ||
| IDH R132H (IHC) | MUT | 4 | |
| WT | 6 | ||
| IDH1 & IDH2 status (NGS) | MUT | 6 (IDH1-R132H: 5; IDH1-R132G: 1; IDH1-R132C: 1 ‡) | |
| WT | 4 | ||
| MGMT (GB, IDH-WT) | Methylated | 1 | |
| Unmethylated | 2 | ||
| Unknown | 1 | ||
| Treatment received prior to or during blood collection | Surgery | GTR | 3 |
| Subtotal Resection | 2 | ||
| Partial Resection | 4 | ||
| Biopsy | 2 | ||
| Radiotherapy | RT alone | 2 | |
| RT + TMZ | 7 | ||
| Adjuvant TMZ (median No. of cycles) | 8 | ||
| Lobe/Area | Frontal | 5 | |
| Temporal | 5 | ||
| Parietal/Occipital | 0 | ||
| Cerebellar | 1 | ||
| Side | Left | 7 | |
| Right | 3 | ||
| Bilateral | 1 | ||
| Scenario | Newly diagnosed | 6 | |
| Recurrent | 5 | ||
| Tumor NGS and BEAMing ctDNA analyses * | No. of peripheral blood draws | Total No. of PB draws | 33 |
| Median PB per patient (range) | 3 (1–7) | ||
| Tumor NGS (+) | 21/33 | ||
| Tumor NGS (−) | 12/33 | ||
| NGS (+) and Untreated/PD disease | 3/18 (17%) | ||
| NGS (+) Treated/non-PD disease | 15/18 (83%) | ||
| Pts with concordance NGS (+) and ctDNA | 3/6 (50%) | ||
| Pts with concordance NGS (−) and ctDNA | 4/4 (100%) | ||
| ctDNA (+) rate in NGS (+) pts (true-positive rate) | 3/21 (14.3%) | ||
| ctDNA (−) rate in NGS (+) pts (false-negative rate) | 18/21 (86.4%) | ||
| Rate of NGS (+) ctDNA (+) and Untreated/PD | 3/6 (50%) | ||
| Rate of NGS (+) ctDNA (−) and Treated/non-PD | 15/15 (100%) | ||
| Patient No. | Histology (Grade) † | Age/Current Disease Setting/Treatment History | Time From IDx to ctDNA (Months) | Tumor IHC IDH1-R132H | Tumor NGS IDH1 (VAF) | Plasma ctDNA BEAMing IDH1 (VAF)(RANO) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| T1 | T2 | T3 | T4 | T5 | T6 | T7 | ||||||
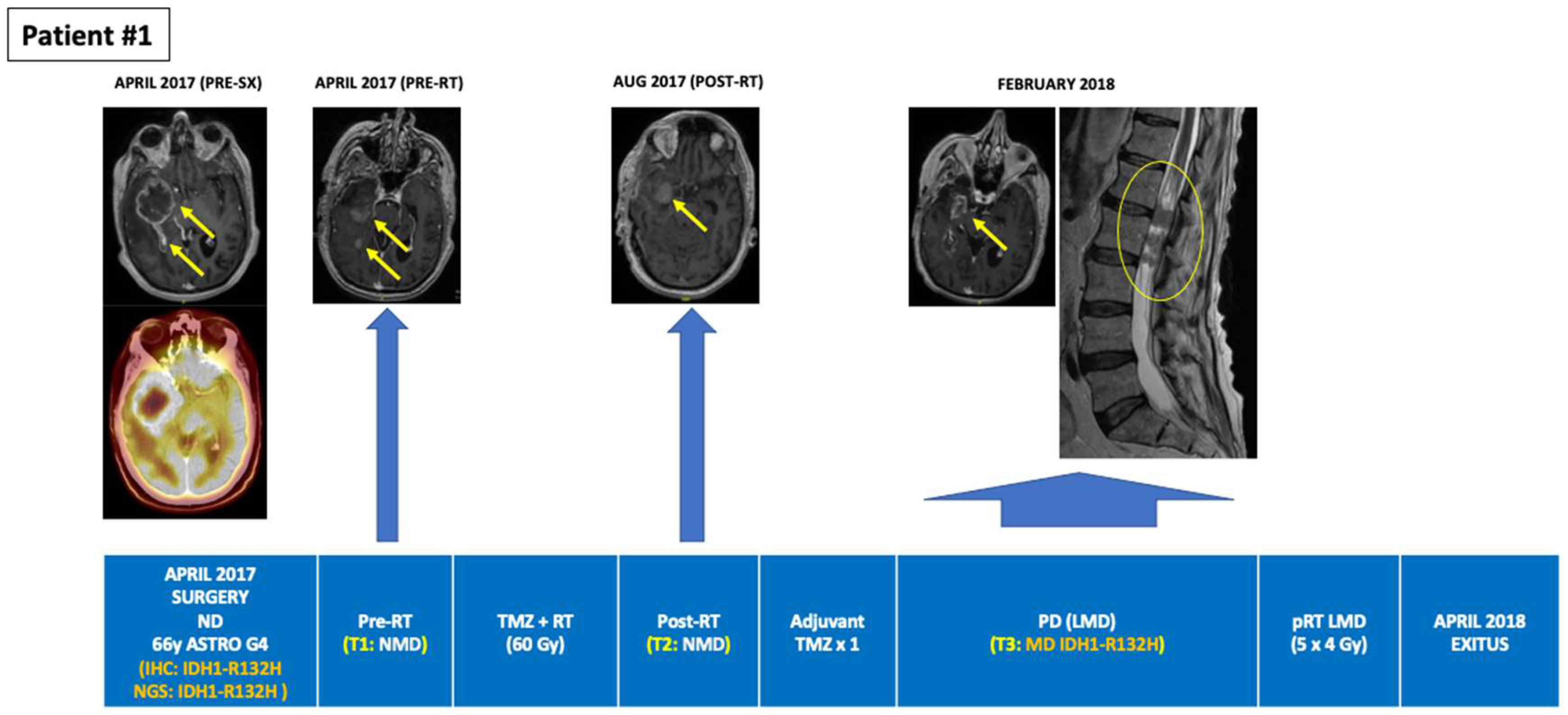
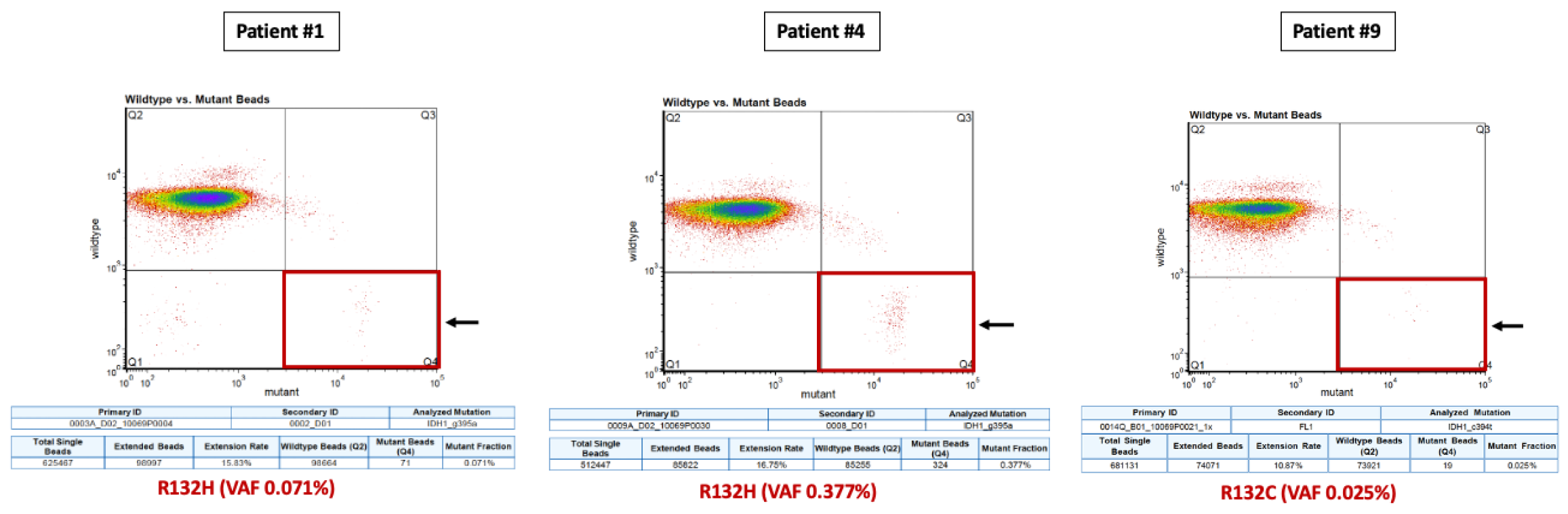
| 1 | Astrocytoma (grade 4), IDH-mutant | 65/ND/SX→RT + TMZ→TMZ × 2→PD (Feb 2018) →Exitus 12 m postND | 2 | MUT | R132H (13.3%) | - | NMD (PR; Post-SX) | NMD (SD; Post-RT) | - | - | R132H (0.071%) (PD; 10-m post-SX) | - |
| 2 | Glioblastoma, IDH-WT | 48/ND/SX→RT + TMZ→TMZ × 6→W&S→PD→SX→Exitus 12 m postND | 2 | WT | WT | NMD (SD; 2 m Post-RT) | NMD (SD; 4 m Post-RT) | - | NMD (PD; 7 m post-SX) | NMD (PD; 9 m post-SX) | NMD (PD; 10 m post-SX) | NMD (PD; 12-m post-SX) |
| 3 | Astrocytoma (grade 3), IDH-mutant | 70/RD/SX (Jun 2015)→SX (2018)→TMZ × 12→W&S (Jan 2020)→SD (ongoing) | 36 | MUT | R132H (14.35%) | NMD (PD; Pre-BX) | NMD (SD; 1 m Post-BX) | - | NMD (SD; Post 2nd TMZ) | NMD (SD; Post 4th TMZ) | NMD (SD; Post 5th TMZ) | NMD (SD; Post 6th TMZ) |
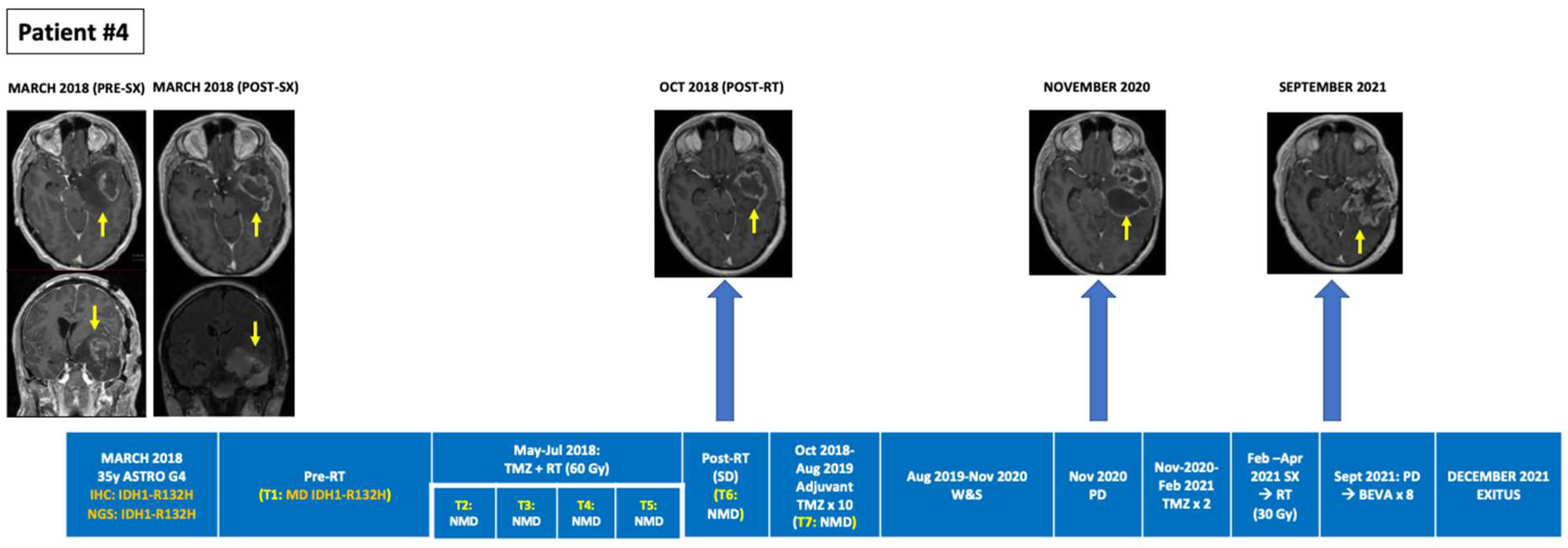
| 4 | Astrocytoma (grade 4), IDH-mutant | 34/ND/SX (PR May 2018)→RT + TMZ→TMZ × 10→W&S (12 m)→TMZ × 2→SX (2021)→RT→PD (Oct 2021)→BEV (ongoing) | 1 | MUT | R132H (31.1%) | R132H (0.377%) (ND; Pre-RT) | NMD (Intra-RT) | NMD (Intra-RT) | NMD (1 m Post-RT) | NMD (2 m Post-RT) | NMD (PsPD; Post 2nd TMZ) | NMD (Post 3rd TMZ) |
| 5 | Glioblastoma, IDH-WT | 50/ND/SX→Exitus 2 m post-SX | 0 | WT | WT | NMD (ND; Pre-BX) | - | - | - | - | - | - |
| 6 | Glioblastoma, IDH-WT | 55/RD/SX (2013)→RT + TMZ→TMZ × 6→SX (RD; 2017)→TMZ × 6→W&S (6 m)→BEV × 6 m→Exitus 6y postND | 48 | WT | WT | NMD (RD; Pre-SX) | NMD (RD; Pre-SX) | - | - | - | - | - |
| 7 | Glioblastoma, IDH-WT | 72/ND/SX (2017)→RT + TMZ→TMZ × 3→SD→W&S (SD ongoing) | 0 | WT | WT | NMD (PD; Pre-SX) | NMD (PD; Pre-RT) | NMD (PsPD; 1 m Post-RT) | - | - | - | - |
| 8 | Astrocytoma (grade 2), IDH-mutant | 34/RD/SX (2015)→RT + TMZ→TMZ ×→SX (RD; 2017)→BEV × 30→PD→Exitus 38 m postND | 26 | WT | R132G (20.5%) | NMD (PR; Post-SX) | - | - | - | - | - | - |
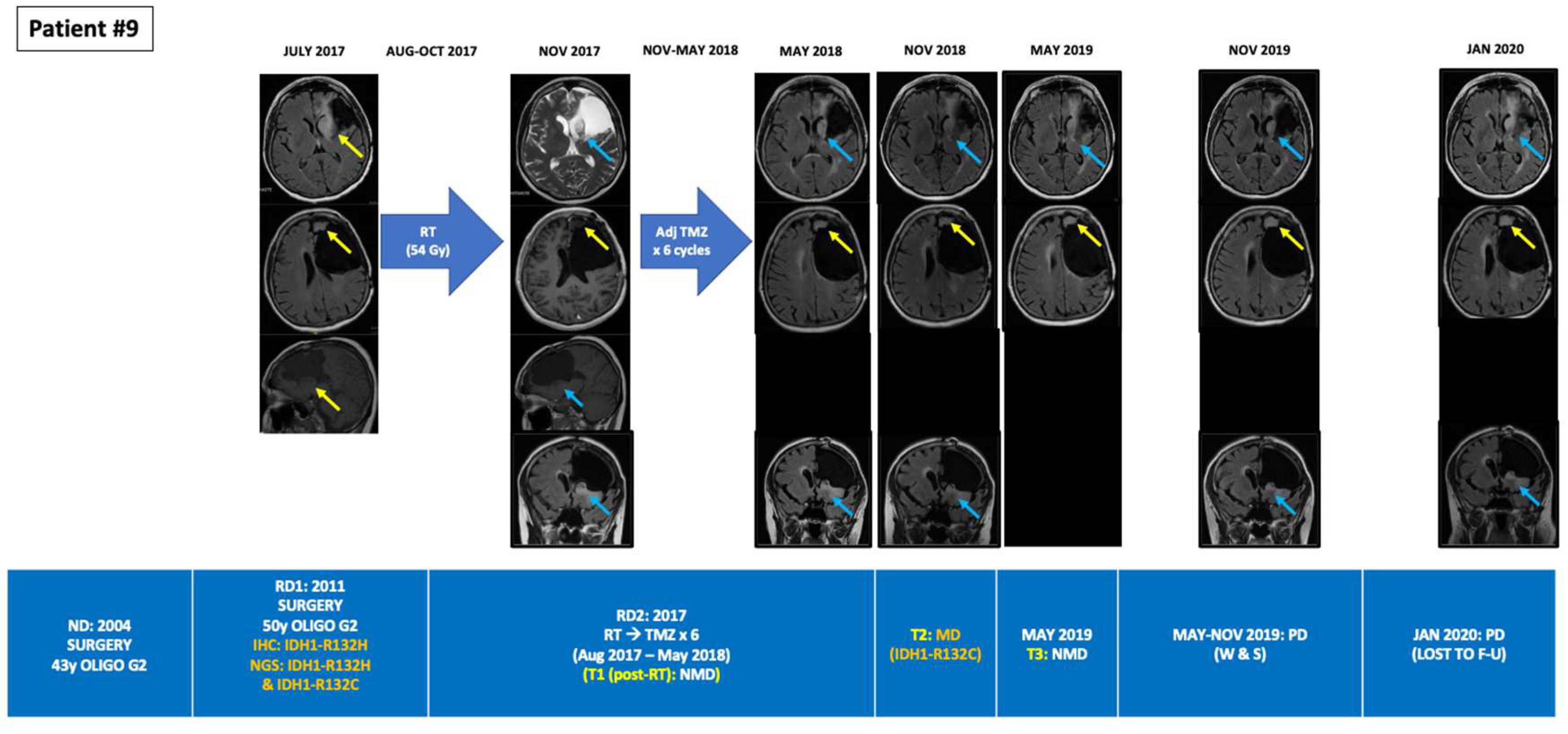
| 9 | Oligodendroglioma (grade 2), IDH-mutant, and 1p/19q-codeleted | 58/RD2/SX (ND; 2004)→SX (RD1; 2011)→RT (RD2; 2017)→TMZ × 6→W&S→PD & lost to FU in Jan 2020 | 156 | MUT | R132H (44.8%) R132C (0.75%) | NMD (PD; Pre-RT) | R132C (0.025%) (SD *; 5 m Post-RT) | NMD (SD *; 11 m Post-RT) | - | - | - | - |
| 10 | Astrocytoma (grade 4), IDH-mutant | 78/ND/SX (2018)→RT + TMZ→TMZ × 3→PD→Exitus 7-m postSX | 0 | WT | R132H (5.9%) | NMD (ND; Pre-BX) | - | - | - | - | - | - |
| Author (Year) | N | ctDNA Reservoir | ctDNA Detection Method | % of ctDNA-Positive Patients | % of ctDNA Positive Controls | Type of ctDNA Mutations Detected | Main Findings |
|---|---|---|---|---|---|---|---|
| Lavon (2010) [31] | Astro: N = 41 Oligo: N = 34 | Plasma | 10q LOH MGMT and PTEN methylation | Astrocytomas: -10q LOH: 51% -MGMTmet: 24% -PTENmet: 0% Oligodendrogliomas: -10q LOH: 79% -1p LOH: 17% -19q LOH: 4% -MGMTmet: 24% | 24% serum positive with no viable tumor on MRI | - | Moderate sensitivity and specificity for LGG and HGG |
| Boisselier (2012) [14] | Gliomas: N = 39 (n = 25 IDH1m) HC: N = 14 | Plasma | COLD dPCR (IDH1-R132H) | Total: 60% (15/25) LGG: 37.5% (3/8) HGG: 70.6% (12/17) | 0 HC (0/14) | IDH1-R132H | Higher detection rate among HGG vs. LGG (70.6% vs. 37.5%) Higher detection rate and higher DNA concentration among high vs. low volume tumors |
| Bettegowda (2014) [30] | Intra- and extracranial cancers: N = 177 Gliomas: N = 27 | Plasma | NGS | <10% | - | - | Extracranial malignancies: ctDNA detected in 82% Intracranial malignancies: ctDNA detected in <50% of MB and in >10% of gliomas |
| Schwaederle (2017) [29] | Intra- and extra-cranial cancers: N = 670 Gliomas: N = 152 | Plasma | NGS | 15% (pts with characterized actionable alterations) | - | - | Extra- and intra-cranial malignancies: ctDNA detected in 48% Gliomas: ctDNA detected in 15% |
| Nassiri (2021) [22] | IDHm gliomas: N = 70 IDHwt gliomas: N = 52 Menin- giomas: N = 60 Hemangio- pericytomas: N = 9 Low-grade glial-neuronal tumors: N = 14 Brain mets of UK primary: N = 9 | Plasma | cfMeDIP-seq | - | - | - | High sensitivity and discriminative capacity between: Gliomas vs. other cancers vs. HC IDHm vs. IDHwt HGG vs. LGG High correlation between plasma and tumor methylation signatures |
| Current study (2022) | N = 10 (6 IDH1m, 4 IDH1wt) | Plasma | BEAMing for IDH1m | 50% (3/6) | 0 IDH1wt (0/4) | IDH1-R132H (n = 2) IDH1-R132C (n = 1) | Same mutant loci detected in IDH1 plasma (BEAMing) and tumor (NGS) All ctDNA+ pts had active disease on MRI In 1 pt BEAMing detected in plasma 1 of the 2 co-existing IDH1 mutations in tissue (R132H, R132C). |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cabezas-Camarero, S.; García-Barberán, V.; Pérez-Alfayate, R.; Casado-Fariñas, I.; Sloane, H.; Jones, F.S.; Pérez-Segura, P. Detection of IDH1 Mutations in Plasma Using BEAMing Technology in Patients with Gliomas. Cancers 2022, 14, 2891. https://doi.org/10.3390/cancers14122891
Cabezas-Camarero S, García-Barberán V, Pérez-Alfayate R, Casado-Fariñas I, Sloane H, Jones FS, Pérez-Segura P. Detection of IDH1 Mutations in Plasma Using BEAMing Technology in Patients with Gliomas. Cancers. 2022; 14(12):2891. https://doi.org/10.3390/cancers14122891
Chicago/Turabian StyleCabezas-Camarero, Santiago, Vanesa García-Barberán, Rebeca Pérez-Alfayate, Isabel Casado-Fariñas, Hillary Sloane, Frederick S. Jones, and Pedro Pérez-Segura. 2022. "Detection of IDH1 Mutations in Plasma Using BEAMing Technology in Patients with Gliomas" Cancers 14, no. 12: 2891. https://doi.org/10.3390/cancers14122891
APA StyleCabezas-Camarero, S., García-Barberán, V., Pérez-Alfayate, R., Casado-Fariñas, I., Sloane, H., Jones, F. S., & Pérez-Segura, P. (2022). Detection of IDH1 Mutations in Plasma Using BEAMing Technology in Patients with Gliomas. Cancers, 14(12), 2891. https://doi.org/10.3390/cancers14122891






