Novel Intraoperative Imaging of Gastric Tube Perfusion during Oncologic Esophagectomy—A Pilot Study Comparing Hyperspectral Imaging (HSI) and Fluorescence Imaging (FI) with Indocyanine Green (ICG)
Abstract
Simple Summary
Abstract
1. Introduction
2. Methods and Material
2.1. Study Design
2.2. Study Population
2.3. Surgical Procedure
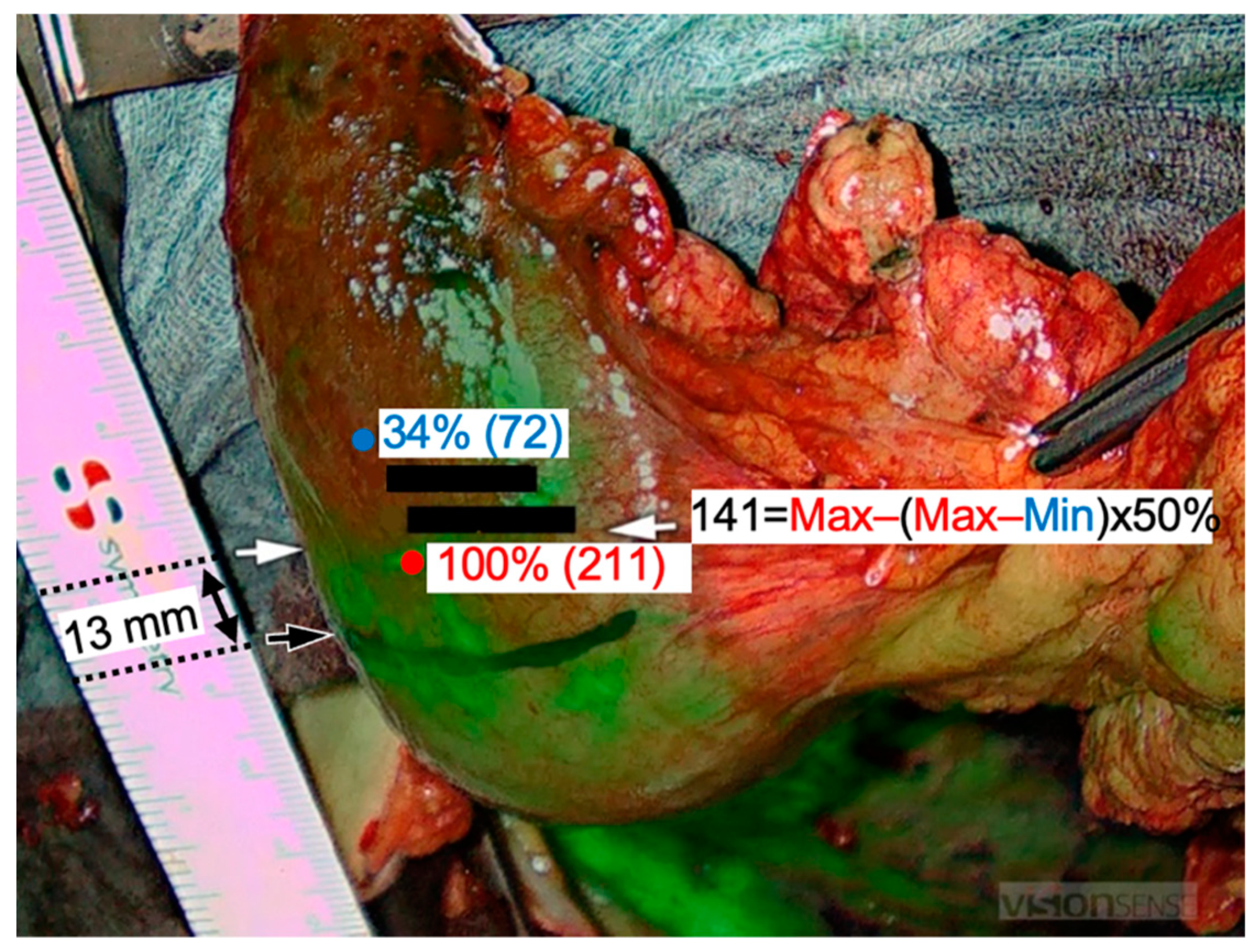
2.4. Hyperspectral Imaging (HSI)
2.5. Fluorescence Imaging (FI) with Indocyanine Green (ICG)
2.6. Postoperative Analysis
2.7. Follow-Up and Endpoints
2.8. Statistical Analysis
3. Results
3.1. Patients’ Characteristics
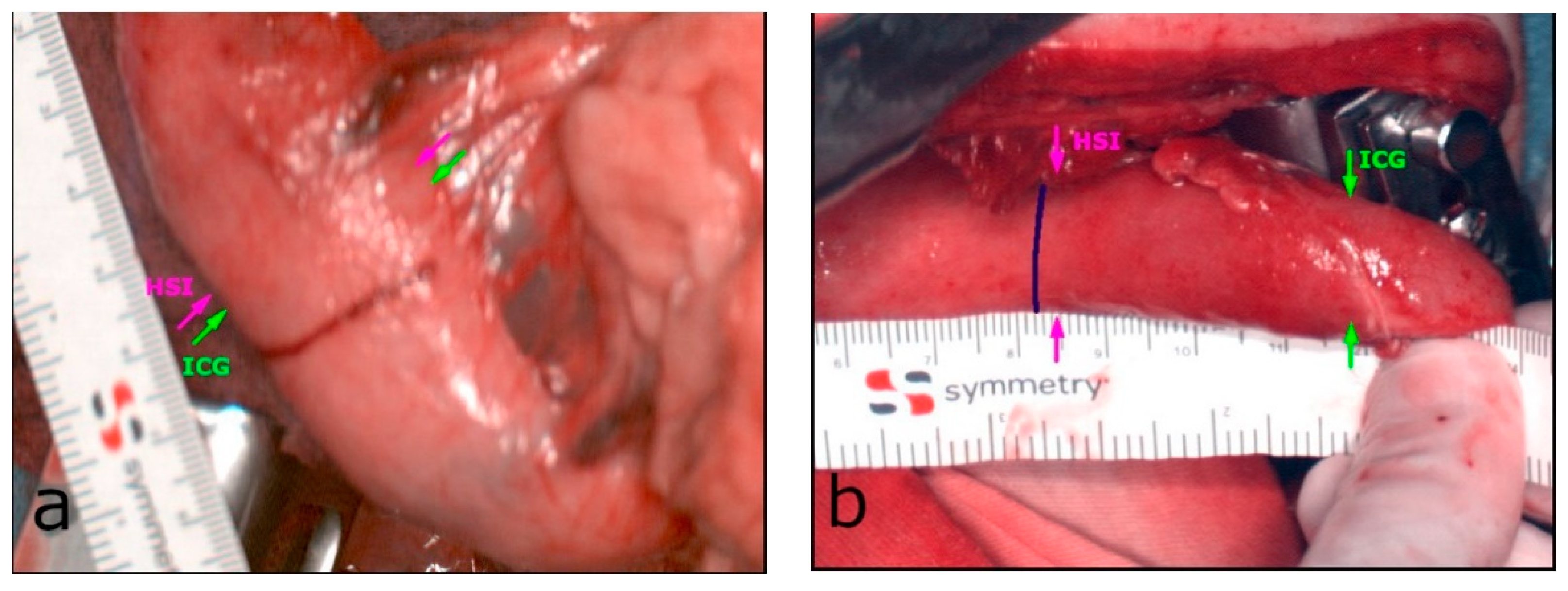
3.2. Intraoperative HSI and FI-ICG of the ROI (Gastric Tip)
3.3. Postoperative Findings and Follow-Up
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kassis, E.S.; Kosinski, A.S.; Ross, P.; Koppes, K.E.; Donahue, J.M.; Daniel, V.C. Predictors of Anastomotic Leak After Esophagectomy: An Analysis of The Society of Thoracic Surgeons General Thoracic Database. Ann. Thorac. Surg. 2013, 96, 1919–1926. [Google Scholar] [CrossRef] [PubMed]
- Paul, S.; Altorki, N. Outcomes in the Management of Esophageal Cancer: Management of Esophageal Cancer. J. Surg. Oncol. 2014, 110, 599–610. [Google Scholar] [CrossRef]
- Alanezi, K.; Urschel, J.D. Mortality Secondary to Esophageal Anastomotic Leak. Ann. Thorac. Cardiovasc. Surg. 2004, 10, 71–75. [Google Scholar]
- Makuuchi, R.; Irino, T.; Tanizawa, Y.; Bando, E.; Kawamura, T.; Terashima, M. Esophagojejunal Anastomotic Leakage Following Gastrectomy for Gastric Cancer. Surg. Today 2019, 49, 187–196. [Google Scholar] [CrossRef]
- Sierzega, M.; Kolodziejczyk, P.; Kulig, J. Impact of Anastomotic Leakage on Long-Term Survival after Total Gastrectomy for Carcinoma of the Stomach. Br. J Surg. 2010, 97, 1035–1042. [Google Scholar] [CrossRef]
- D’Cunha, J.; Rueth, N.M.; Groth, S.S.; Maddaus, M.A.; Andrade, R.S. Esophageal Stents for Anastomotic Leaks and Perforations. J. Thorac. Cardiovasc. Surg. 2011, 142, 39–46. [Google Scholar] [CrossRef]
- Ryan, C.E.; Paniccia, A.; Meguid, R.A.; McCarter, M.D. Transthoracic Anastomotic Leak After Esophagectomy: Current Trends. Ann. Surg. Oncol. 2017, 24, 281–290. [Google Scholar] [CrossRef] [PubMed]
- Sauvanet, A.; Mariette, C.; Thomas, P.; Lozac’h, P.; Segol, P.; Tiret, E.; Delpero, J.-R.; Collet, D.; Leborgne, J.; Pradère, B.; et al. Mortality and Morbidity after Resection for Adenocarcinoma of the Gastroesophageal Junction: Predictive Factors. J. Am. Coll. Surg. 2005, 201, 253–262. [Google Scholar] [CrossRef]
- Duprée, A.; von Kroge, P.H.; Izbicki, J.R.; Wipper, S.H.; Mann, O. Fluoreszenzangiographie bei Ösophagusanastomosen: Perfusionsbeurteilung des Magenschlauchs mit Indocyaningrün. Chirurg 2019, 90, 875–879. [Google Scholar] [CrossRef]
- Urbanavičius, L. How to Assess Intestinal Viability during Surgery: A Review of Techniques. WJGS 2011, 3, 59. [Google Scholar] [CrossRef] [PubMed]
- Karliczek, A.; Harlaar, N.J.; Zeebregts, C.J.; Wiggers, T.; Baas, P.C.; van Dam, G.M. Surgeons Lack Predictive Accuracy for Anastomotic Leakage in Gastrointestinal Surgery. Int. J. Colorectal Dis. 2009, 24, 569–576. [Google Scholar] [CrossRef]
- Kumagai, Y.; Hatano, S.; Sobajima, J.; Ishiguro, T.; Fukuchi, M.; Ishibashi, K.-I.; Mochiki, E.; Nakajima, Y.; Ishida, H. Indocyanine Green Fluorescence Angiography of the Reconstructed Gastric Tube during Esophagectomy: Efficacy of the 90-Second Rule. Dis. Esophagus 2018, 31, doy052. [Google Scholar] [CrossRef] [PubMed]
- Koyanagi, K.; Ozawa, S.; Oguma, J.; Kazuno, A.; Yamazaki, Y.; Ninomiya, Y.; Ochiai, H.; Tachimori, Y. Blood Flow Speed of the Gastric Conduit Assessed by Indocyanine Green Fluorescence: New Predictive Evaluation of Anastomotic Leakage after Esophagectomy. Medicine 2016, 95, e4386. [Google Scholar] [CrossRef]
- Karampinis, I.; Ronellenfitsch, U.; Mertens, C.; Gerken, A.; Hetjens, S.; Post, S.; Kienle, P.; Nowak, K. Indocyanine Green Tissue Angiography Affects Anastomotic Leakage after Esophagectomy. A Retrospective, Case-Control Study. Int. J. Surg. 2017, 48, 210–214. [Google Scholar] [CrossRef] [PubMed]
- Obana, A.; Miki, T.; Hayashi, K.; Takeda, M.; Kawamura, A.; Mutoh, T.; Harino, S.; Fukushima, I.; Komatsu, H.; Takaku, Y.; et al. Survey of Complications of Indocyanine Green Angiography in Japan. Am. J. Ophthalmol. 1994, 118, 749–753. [Google Scholar] [CrossRef]
- Zhai, Q.; Wang, Y.; Tian, A. Severe Hemodynamic Instability after Indocyanine Green Injection during Off-Pump Coronary Artery Bypass Grafting: A Case Report. Medicine 2017, 96, e8766. [Google Scholar] [CrossRef] [PubMed]
- Köhler, H. Evaluation of Hyperspectral Imaging (HSI) for the Measurement of Ischemic Conditioning Effects of the Gastric Conduit during Esophagectomy. Surg. Endosc. 2019, 33, 3775–3782. [Google Scholar] [CrossRef]
- Barberio, M.; Longo, F.; Fiorillo, C.; Seeliger, B.; Mascagni, P.; Agnus, V.; Lindner, V.; Geny, B.; Charles, A.-L.; Gockel, I.; et al. HYPerspectral Enhanced Reality (HYPER): A Physiology-Based Surgical Guidance Tool. Surg. Endosc. 2020, 34, 1736–1744. [Google Scholar] [CrossRef]
- Jansen-Winkeln, B.; Germann, I.; Köhler, H.; Mehdorn, M.; Maktabi, M.; Sucher, R.; Barberio, M.; Chalopin, C.; Diana, M.; Moulla, Y.; et al. Comparison of Hyperspectral Imaging and Fluorescence Angiography for the Determination of the Transection Margin in Colorectal Resections—A Comparative Study. Int. J. Colorectal Dis. 2021, 36, 283–291. [Google Scholar] [CrossRef]
- Jansen-Winkeln, B.; Maktabi, M.; Takoh, J.P.; Rabe, S.M.; Barberio, M.; Köhler, H.; Neumuth, T.; Melzer, A.; Chalopin, C.; Gockel, I. Hyperspektral-Imaging bei gastrointestinalen Anastomosen. Chirurg 2018, 89, 717–725. [Google Scholar] [CrossRef]
- Gockel, I.; Jansen-Winkeln, B.; Holfert, N.; Rayes, N.; Thieme, R.; Maktabi, M.; Sucher, R.; Seehofer, D.; Barberio, M.; Diana, M.; et al. Möglichkeiten und Perspektiven der Hyperspektralbildgebung in der Viszeralchirurgie. Chirurg 2020, 91, 150–159. [Google Scholar] [CrossRef] [PubMed]
- Moulla, Y.; Reifenrath, M.; Rehmet, K.; Niebisch, S.; Jansen-Winkeln, B.; Sucher, R.; Hoffmeister, A.; Kreuser, N.; Köhler, H.; Gockel, I. Hybridösophagektomie mit intraoperativem Hyperspektral-Imaging: Videobeitrag. Chirurg 2020, 91, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Bludau, M.; Hölscher, A.H.; Vallböhmer, D.; Gutschow, C.; Schröder, W. Ischemic Conditioning of the Gastric Conduit Prior to Esophagectomy Improves Mucosal Oxygen Saturation. Ann. Thorac. Surg. 2010, 90, 1121–1126. [Google Scholar] [CrossRef]
- Holmer, A.; Marotz, J.; Wahl, P.; Dau, M.; Kämmerer, P.W. Hyperspectral Imaging in Perfusion and Wound Diagnostics—Methods and Algorithms for the Determination of Tissue Parameters. Biomed. Eng./Biomed. Tech. 2018, 63, 547–556. [Google Scholar] [CrossRef]
- Köhler, H.; Kulcke, A.; Maktabi, M.; Moulla, Y.; Jansen-Winkeln, B.; Barberio, M.; Diana, M.; Gockel, I.; Neumuth, T.; Chalopin, C. Laparoscopic System for Simultaneous High-Resolution Video and Rapid Hyperspectral Imaging in the Visible and near-Infrared Spectral Range. J. Biomed. Opt. 2020, 25, 086004. [Google Scholar] [CrossRef]
- Baiocchi, G.L.; Diana, M.; Boni, L. Indocyanine Green-Based Fluorescence Imaging in Visceral and Hepatobiliary and Pancreatic Surgery: State of the Art and Future Directions. WJG 2018, 24, 2921–2930. [Google Scholar] [CrossRef] [PubMed]
- Mascagni, P.; Longo, F.; Barberio, M.; Seeliger, B.; Agnus, V.; Saccomandi, P.; Hostettler, A.; Marescaux, J.; Diana, M. New Intraoperative Imaging Technologies: Innovating the Surgeon’s Eye toward Surgical Precision. J. Surg. Oncol. 2018, 118, 265–282. [Google Scholar] [CrossRef]
- Abdel Salam, A.R.; Drummond, G.B.; Bauld, H.W.; Scott, D.B. Clearance of indocyanine green as an index of liver function during cyclopropane anaesthesia and induced hypotension. Br. J. Anaesth. 1976, 48, 231–238. [Google Scholar] [CrossRef][Green Version]
- Cherrick, G.R.; Stein, S.W.; Leevy, C.M.; Davidson, C.S. Indocyanine green: Observation on its physical properties, plasma decay, and hepatic extraction *. J. Clin. Investig. 1960, 39, 592–600. [Google Scholar] [CrossRef]
- Low, D.E.; Alderson, D.; Cecconello, I.; Chang, A.C.; Darling, G.E.; D’Journo, X.B.; Griffin, S.M.; Hölscher, A.H.; Hofstetter, W.L.; Jobe, B.A.; et al. International Consensus on Standardization of Data Collection for Complications Associated With Esophagectomy: Esophagectomy Complications Consensus Group (ECCG). Ann. Surg. 2015, 262, 286–294. [Google Scholar] [CrossRef]
- Dindo, D.; Demartines, N.; Clavien, P.-A. Classification of Surgical Complications: A New Proposal With Evaluation in a Cohort of 6336 Patients and Results of a Survey. Ann. Surg. 2004, 240, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Nerup, N.; Svendsen, M.B.S.; Svendsen, L.B.; Achiam, M.P. Feasibility and Usability of Real-Time Intraoperative Quantitative Fluorescent-Guided Perfusion Assessment during Resection of Gastroesophageal Junction Cancer. Langenbeck's Arch. Surg. 2020, 405, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Diana, M.; Agnus, V.; Halvax, P.; Liu, Y.-Y.; Dallemagne, B.; Schlagowski, A.-I.; Geny, B.; Diemunsch, P.; Lindner, V.; Marescaux, J. Intraoperative Fluorescence-Based Enhanced Reality Laparoscopic Real-Time Imaging to Assess Bowel Perfusion at the Anastomotic Site in an Experimental Model: Intraoperative Fluorescence-Based Enhanced Reality Laparoscopic Real-Time Imaging. J. Br. Surg. 2015, 102, e169–e176. [Google Scholar] [CrossRef] [PubMed]
- Lütken, C.D.; Achiam, M.P.; Osterkamp, J.; Svendsen, M.B.; Nerup, N. Quantification of Fluorescence Angiography: Toward a Reliable Intraoperative Assessment of Tissue Perfusion—A Narrative Review. Langenbeck's Arch. Surg. 2020, 406, 251–259. [Google Scholar] [CrossRef] [PubMed]

| Variables | Number of Cases (%) |
|---|---|
| Sex | |
| male | 12 (92.3) |
| female | 1 (7.7) |
| ASA * | |
| I | 1 (7.7) |
| II | 7 (53.8) |
| III | 4 (30.8) |
| IV | 1 (7.7) |
| Neoadjuvanttherapy | |
| none | 1 (7.7) |
| chemotherapy (FLOT) | 7 (53.8) |
| radiochemotherapy (CROSS) ** | 5 (38.5) |
| Histopathologicentity | |
| squamous cell carcinoma | 3 (23.0) |
| adenocarcinoma | 8 (61.5) |
| MANEC, adenosquamous | 2 (15.4) |
| Riskfactors | |
| smoking | 8 (61.5) |
| COPD *** Gold II | 6 (46.2) |
| diabetes mellitus type II | 1 (7.7) |
| arterial hypertension | 5 (38.5) |
| pulmonary embolism | 1 (7.7) |
| renal insufficiency | 1 (7.7) |
| Step 1 Laparoscopic/Robotic Gastrolysis | Step 2 (after 3–6 Days) Ivor Lewis Esophagectomy |
|---|---|
|
|
|
|
|
|
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hennig, S.; Jansen-Winkeln, B.; Köhler, H.; Knospe, L.; Chalopin, C.; Maktabi, M.; Pfahl, A.; Hoffmann, J.; Kwast, S.; Gockel, I.; et al. Novel Intraoperative Imaging of Gastric Tube Perfusion during Oncologic Esophagectomy—A Pilot Study Comparing Hyperspectral Imaging (HSI) and Fluorescence Imaging (FI) with Indocyanine Green (ICG). Cancers 2022, 14, 97. https://doi.org/10.3390/cancers14010097
Hennig S, Jansen-Winkeln B, Köhler H, Knospe L, Chalopin C, Maktabi M, Pfahl A, Hoffmann J, Kwast S, Gockel I, et al. Novel Intraoperative Imaging of Gastric Tube Perfusion during Oncologic Esophagectomy—A Pilot Study Comparing Hyperspectral Imaging (HSI) and Fluorescence Imaging (FI) with Indocyanine Green (ICG). Cancers. 2022; 14(1):97. https://doi.org/10.3390/cancers14010097
Chicago/Turabian StyleHennig, Sebastian, Boris Jansen-Winkeln, Hannes Köhler, Luise Knospe, Claire Chalopin, Marianne Maktabi, Annekatrin Pfahl, Jana Hoffmann, Stefan Kwast, Ines Gockel, and et al. 2022. "Novel Intraoperative Imaging of Gastric Tube Perfusion during Oncologic Esophagectomy—A Pilot Study Comparing Hyperspectral Imaging (HSI) and Fluorescence Imaging (FI) with Indocyanine Green (ICG)" Cancers 14, no. 1: 97. https://doi.org/10.3390/cancers14010097
APA StyleHennig, S., Jansen-Winkeln, B., Köhler, H., Knospe, L., Chalopin, C., Maktabi, M., Pfahl, A., Hoffmann, J., Kwast, S., Gockel, I., & Moulla, Y. (2022). Novel Intraoperative Imaging of Gastric Tube Perfusion during Oncologic Esophagectomy—A Pilot Study Comparing Hyperspectral Imaging (HSI) and Fluorescence Imaging (FI) with Indocyanine Green (ICG). Cancers, 14(1), 97. https://doi.org/10.3390/cancers14010097








