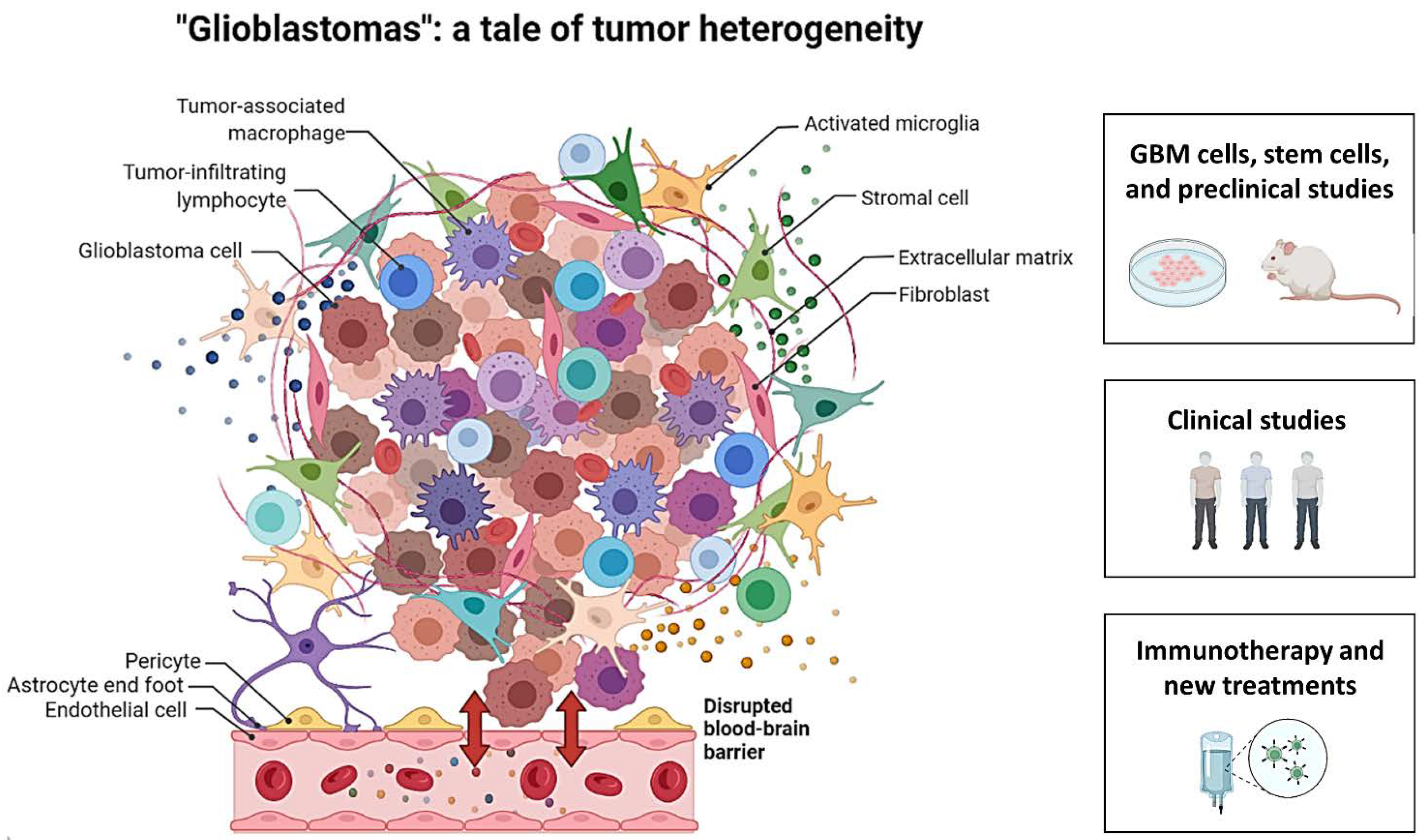
Glioblastomas
Author Contributions
Funding
Conflicts of Interest
References
- Patel, A.P.; Tirosh, I.; Trombetta, J.J.; Shalek, A.K.; Gillespie, S.M.; Wakimoto, H.; Cahill, D.P.; Nahed, B.V.; Curry, W.T.; Martuza, R.L.; et al. Single-cell RNA-seq highlights intratumoral heterogeneity in primary glioblastoma. Science 2014, 344, 1396–1401. [Google Scholar] [CrossRef] [Green Version]
- Arthurs, A.L.; Keating, D.J.; Stringer, B.W.; Conn, S.J. The suitability of glioblastoma cell lines as models for primary glioblastoma cell metabolism. Cancers 2020, 12, 3722. [Google Scholar] [CrossRef] [PubMed]
- Costa, B.; Fletcher, M.N.C.; Boskovic, P.; Ivanova, E.L.; Eisemann, T.; Lohr, S.; Bunse, L.; Löwer, M.; Burchard, S.; Korshunov, A.; et al. A Set of Cell Lines Derived from a Genetic Murine Glioblastoma Model Recapitulates Molecular and Morphological Characteristics of Human Tumors. Cancers 2021, 13, 230. [Google Scholar] [CrossRef] [PubMed]
- Guelfi, S.; Orsetti, B.; Deleuze, V.; Rigau, V.; Bauchet, L.; Duffau, H.; Rothhut, B.; Hugnot, J.-P. SLUG and truncated TAL1 reduce glioblastoma stem cell growth downstream of notch1 and define distinct vascular subpopulations in glioblastoma multiforme. Cancers 2021, 13, 5393. [Google Scholar] [CrossRef]
- Khan, S.; Mahalingam, R.; Sen, S.; Martinez-Ledesma, E.; Khan, A.; Gandy, K.; Lang, F.F.; Sulman, E.P.; Alfaro-Munoz, K.D.; Majd, N.K.; et al. Intrinsic interferon signaling regulates the cell death and mesenchymal phenotype of glioblastoma stem cells. Cancers 2021, 13, 5284. [Google Scholar] [CrossRef]
- Panza, S.; Malivindi, R.; Caruso, A.; Russo, U.; Giordano, F.; Győrffy, B.; Gelsomino, L.; De Amicis, F.; Barone, I.; Conforti, F.L.; et al. Novel Insights into the Antagonistic Effects of Losartan against Angiotensin II/AGTR1 Signaling in Glioblastoma Cells. Cancers 2021, 13, 4555. [Google Scholar] [CrossRef]
- Jabbarli, R.; Ahmadipour, Y.; Rauschenbach, L.; Santos, A.N.; Darkwah Oppong, M.; Pierscianek, D.; Quesada, C.M.; Kebir, S.; Dammann, P.; Guberina, N.; et al. How about Levetiracetam in Glioblastoma? An Institutional Experience and Meta-Analysis. Cancers 2021, 13, 3770. [Google Scholar] [CrossRef]
- Wu, A.; Ruiz Colón, G.; Aslakson, R.; Pollom, E.; Patel, C.B. Palliative care service utilization and advance care planning for adult glioblastoma patients: A systematic review. Cancers 2021, 13, 2867. [Google Scholar] [CrossRef] [PubMed]
- Jin, K.; Mao, C.; Chen, L.; Wang, L.; Liu, Y.; Yuan, J. Adenosinergic pathway: A hope in the immunotherapy of glioblastoma. Cancers 2021, 13, 229. [Google Scholar] [CrossRef] [PubMed]
- Andersen, R.S.; Anand, A.; Harwood, D.S.L.; Kristensen, B.W. Tumor-Associated Microglia and Macrophages in the Glioblastoma Microenvironment and Their Implications for Therapy. Cancers 2021, 13, 4255. [Google Scholar] [CrossRef] [PubMed]
- Aguilar, A.A.; Ho, M.C.; Chang, E.; Carlson, K.W.; Natarajan, A.; Marciano, T.; Bomzon, Z.; Patel, C.B. Permeabilizing Cell Membranes with Electric Fields. Cancers 2021, 13, 2283. [Google Scholar] [CrossRef] [PubMed]
- Ranjan, A.; Pang, Y.; Butler, M.; Merchant, M.; Kim, O.; Yu, G.; Su, Y.-T.; Gilbert, M.R.; Levens, D.; Wu, J. Targeting CDK9 for the treatment of glioblastoma. Cancers 2021, 13, 3039. [Google Scholar] [CrossRef] [PubMed]
- Koppenol, W.H.; Bounds, P.L.; Dang, C.V. Otto Warburg’s contributions to current concepts of cancer metabolism. Nat. Rev. Cancer 2011, 11, 325–337. [Google Scholar] [CrossRef] [PubMed]
- Costa, B.; Eisemann, T.; Strelau, J.; Spaan, I.; Korshunov, A.; Liu, H.-K.; Bugert, P.; Angel, P.; Peterziel, H. Intratumoral platelet aggregate formation in a murine preclinical glioma model depends on podoplanin expression on tumor cells. Blood Adv. 2019, 3, 1092–1102. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, K.; Zhou, J.; Chen, Y.; Chen, Y.; Chen, L.; Zhang, P.; Ma, L.; Jiang, Z.; Bian, J.; Yin, W. Angiotensin II contributes to intratumoral immunosuppressionvia induction of PD-L1 expression in non-small cell lung carcinoma. Int. Immunopharmacol. 2020, 84, 106507. [Google Scholar] [CrossRef] [PubMed]
- Arrieta, O.; Guevara, P.; Escobar, E.; García-Navarrete, R.; Pineda, B.; Sotelo, J. Blockage of angiotensin II type I receptor decreases the synthesis of growth factors and induces apoptosis in C6 cultured cells and C6 rat glioma. Br. J. Cancer 2005, 92, 1247–1252. [Google Scholar] [CrossRef] [PubMed]
- Stupp, R.; Hegi, M.E.; Mason, W.P.; van den Bent, M.J.; Taphoorn, M.J.B.; Janzer, R.C.; Ludwin, S.K.; Allgeier, A.; Fisher, B.; Belanger, K.; et al. National Cancer Institute of Canada Clinical Trials Group Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol. 2009, 10, 459–466. [Google Scholar] [CrossRef]
- Bobustuc, G.C.; Baker, C.H.; Limaye, A.; Jenkins, W.D.; Pearl, G.; Avgeropoulos, N.G.; Konduri, S.D. Levetiracetam enhances p53-mediated MGMT inhibition and sensitizes glioblastoma cells to temozolomide. Neuro Oncol. 2010, 12, 917–927. [Google Scholar] [CrossRef] [Green Version]
- Happold, C.; Gorlia, T.; Chinot, O.; Gilbert, M.R.; Nabors, L.B.; Wick, W.; Pugh, S.L.; Hegi, M.; Cloughesy, T.; Roth, P.; et al. Does valproic acid or levetiracetam improve survival in glioblastoma? A pooled analysis of prospective clinical trials in newly diagnosed glioblastoma. J. Clin. Oncol. 2016, 34, 731–739. [Google Scholar] [CrossRef] [PubMed]
- Walbert, T.; Puduvalli, V.K.; Taphoorn, M.J.B.; Taylor, A.R.; Jalali, R. International patterns of palliative care in neuro-oncology: A survey of physician members of the Asian Society for Neuro-Oncology, the European Association of Neuro-Oncology, and the Society for Neuro-Oncology. Neurooncol. Pract. 2015, 2, 62–69. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stupp, R.; Mason, W.P.; van den Bent, M.J.; Weller, M.; Fisher, B.; Taphoorn, M.J.B.; Belanger, K.; Brandes, A.A.; Marosi, C.; Bogdahn, U.; et al. European Organisation for Research and Treatment of Cancer Brain Tumor and Radiotherapy Groups; National Cancer Institute of Canada Clinical Trials Group Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N. Engl. J. Med. 2005, 352, 987–996. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Finocchiaro, G.; Berzero, G. Glioblastomas. Cancers 2022, 14, 104. https://doi.org/10.3390/cancers14010104
Finocchiaro G, Berzero G. Glioblastomas. Cancers. 2022; 14(1):104. https://doi.org/10.3390/cancers14010104
Chicago/Turabian StyleFinocchiaro, Gaetano, and Giulia Berzero. 2022. "Glioblastomas" Cancers 14, no. 1: 104. https://doi.org/10.3390/cancers14010104
APA StyleFinocchiaro, G., & Berzero, G. (2022). Glioblastomas. Cancers, 14(1), 104. https://doi.org/10.3390/cancers14010104





