Video-Laparoscopic versus Open Surgery in Obese Patients with Colorectal Cancer: A Propensity Score Matching Study
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Population and Design
2.2. Demographic and Clinical Variables
2.3. Short-Term Postoperative Outcomes
2.4. Surgical Procedures
2.5. Statistical Analysis
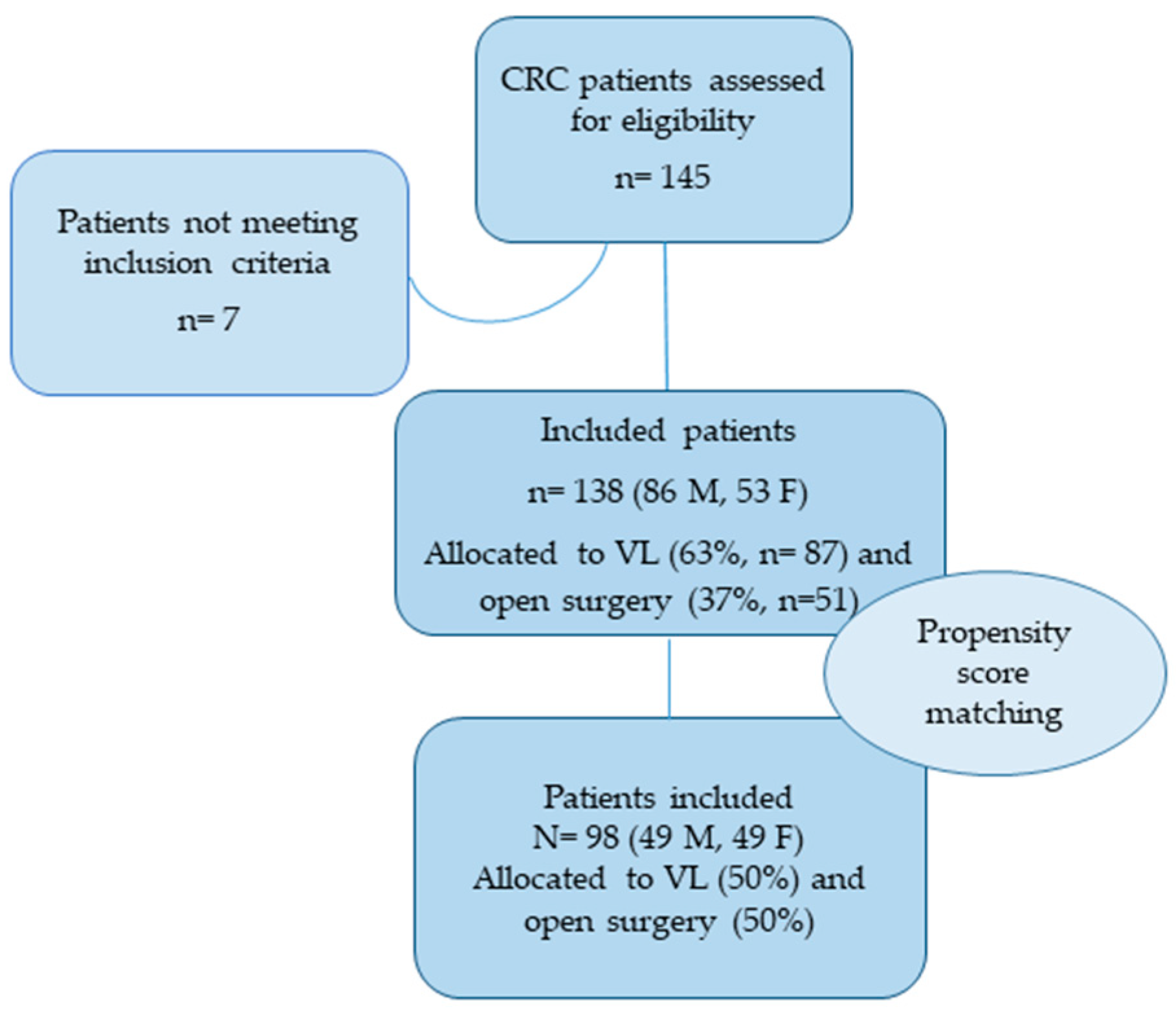
3. Results
3.1. Baseline Data
3.2. Propensity Score Matching
3.3. Multivariable Models
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Siegel, R.L.; Miller, K.D.; Fedewa, S.A.; Ahnen, D.J.; Meester, R.G.S.; Barzi, A.; Jemal, A. Colorectal cancer statistics, 2017. Cancer J. Clin. 2017, 67, 177–193. [Google Scholar] [CrossRef] [PubMed]
- Bardou, M.; Barkun, A.N.; Martel, M. Obesity and colorectal cancer. Gut 2013, 62, 933–947. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Jiang, W.; Wu, Y.; Jiang, X. Re: Television viewing and time spent sedentary in relation to cancer risk: A meta-analysis. JNCI J. Natl. Cancer Instit. 2014, 106, dju303. [Google Scholar] [CrossRef] [PubMed]
- Kerr, J.; Anderson, C.; Lippman, S.M. Physical activity, sedentary behaviour, diet, and cancer: An update and emerging new evidence. Lancet Oncol. 2017, 18, e457–e471. [Google Scholar] [CrossRef]
- Park, S.-Y.; Boushey, C.J.; Wilkens, L.R.; Haiman, C.A.; Le Marchand, L. High-quality diets associate with reduced risk of colorectal cancer: Analyses of diet quality indexes in the multiethnic cohort. Gastroenterology 2017, 153, 386–394.e2. [Google Scholar] [CrossRef] [PubMed]
- Tsilidis, K.K.; Kasimis, J.C.; Lopez, D.S.; Ntzani, E.E.; Ioannidis, J.P.A. Type 2 diabetes and cancer: Umbrella review of meta-analyses of observational studies. BMJ 2015, 350, g7607. [Google Scholar] [CrossRef] [PubMed]
- Chiu, C.C.; Lin, W.L.; Shi, H.Y.; Huang, C.C.; Chen, J.J.; Su, S.B.; Lai, C.C.; Chao, C.M.; Tsao, C.J.; Chen, S.H.; et al. Comparison of oncologic outcomes in laparoscopic versus open surgery for non-metastatic colorectal cancer: Personal experience in a single institution. J. Clin. Med. 2019, 8, 875. [Google Scholar] [CrossRef]
- Ma, Y.; Yang, Y.; Wang, F.; Zhang, P.; Shi, C.; Zou, Y.; Qin, H. Obesity and risk of colorectal cancer: A systematic review of prospective studies. PLoS ONE 2013, 8, e53916. [Google Scholar] [CrossRef]
- Cai, Y.; Zhou, Y.; Li, Z.; Xiang, J.; Chen, Z. Surgical outcome of laparoscopic colectomy for colorectal cancer in obese patients: A comparative study with open colectomy. Oncol. Lett. 2013, 6, 1057–1062. [Google Scholar] [CrossRef]
- Kyrgiou, M.; Kalliala, I.; Markozannes, G.; Gunter, M.J.; Paraskevaidis, E.; Gabra, H.; Martin-Hirsch, P.; Tsilidis, K.K. Adiposity and cancer at major anatomical sites: Umbrella review of the literature. BMJ 2017, 356, j477. [Google Scholar] [CrossRef]
- Hotouras, A.; Ribas, Y.; Zakeri, S.A.; Nunes, Q.M.; Murphy, J.; Bhan, C.; Wexner, S.D. The influence of obesity and body mass index on the outcome of laparoscopic colorectal surgery: A systematic literature review. Colorectal Dis. 2016, 18, O337–O366. [Google Scholar] [CrossRef]
- Ye, X.Z.; Chen, X.Y.; Ruan, X.J.; Chen, W.Z.; Ma, L.L.; Dong, Q.T.; Cai, H.Y.; Yu, Z.; Chen, X.L. Laparoscopic-assisted colorectal surgery benefits visceral obesity patients. Eur. J. Gastroenterol. Hepatol. 2019, 31, 786–791. [Google Scholar] [CrossRef]
- Vignali, A.; De Nardi, P.; Ghirardelli, L.; Di Palo, S.; Staudacher, C. Short and long-term outcomes of laparoscopic colectomy in obese patients. World J. Gastroenterol. 2013, 19, 7405–7411. [Google Scholar] [CrossRef] [PubMed]
- Whitlock, K.; Gill, R.S.; Birch, D.W.; Karmali, S. The Association between Obesity and Colorectal Cancer. Gastroenterol. Res. Pract. 2012, 2012, 768247. [Google Scholar] [CrossRef]
- Kazama, K.; Numata, M.; Aoyama, T.; Onodeara, A.; Hara, K.; Atsumi, Y.; Tamagawa, H.; Godai, T.; Saeki, H.; Saigusa, Y.; et al. Laparoscopic vs. open surgery for stage II/III colon cancer patients with body mass index >25 kg/m2. In Vivo 2020, 34, 2079–2085. [Google Scholar] [CrossRef] [PubMed]
- Delaney, C.P.; Pokala, N.; Senagore, A.J.; Casillas, S.; Kiran, R.P.; Brady, K.M.; Fazio, V.W. Is laparoscopic colectomy applicable to patients with body mass index >30? A case-matched comparative study with open colectomy. Dis. Colon Rectum 2005, 48, 975–981. [Google Scholar] [CrossRef]
- Scheidbach, H.; Benedix, F.; Hügel, O.; Kose, D.; Köckerling, F.; Lippert, H. Laparoscopic approach to colorectal procedures in the obese patient: Risk factor or benefit? Obes. Surg. 2008, 18, 66–70. [Google Scholar] [CrossRef] [PubMed]
- Gendall, K.A.; Raniga, S.; Kennedy, R.; Frizelle, F.A. The impact of obesity on outcome after major colorectal surgery. Dis. Colon Rectum 2007, 50, 2223–2237. [Google Scholar] [CrossRef] [PubMed]
- Kitano, S.; Inomata, M.; Mizusawa, J.; Katayama, H.; Watanabe, M.; Yamamoto, S.; Ito, M.; Saito, S.; Fujii, S.; Konishi, F.; et al. Survival outcomes following laparoscopic versus open D3 dissection for stage II or III colon cancer (JCOG0404): A phase 3, randomised controlled trial. Lancet Gastroenterol. Hepatol. 2017, 2, 261–268. [Google Scholar] [CrossRef]
- Ye, S.P.; Qiu, H.; Liao, S.J.; Ai, J.H.; Shi, J. Mini-invasive vs. open resection of colorectal cancer and liver metastases: A meta-analysis. World J. Gastroenterol. 2019, 25, 2819–2832. [Google Scholar] [CrossRef] [PubMed]
- Engstrom, P.F.; Benson, A.B., 3rd; Saltz, L. National comprehensive cancer network colon cancer. Clinical practice guidelines in oncology. J. Natl. Compr. Canc. Netw. 2003, 1, 40–53. [Google Scholar] [PubMed]
- Ulijaszek, S.J. Obesity: Preventing and managing the global epidemic. Report of a WHO consultation. J. Siosoc. Sci. 2003, 35, 624–625. [Google Scholar]
- Quirke, P.; Durdey, P.; Dixon, M.F.; Williams, N.S. Local recurrence of rectal adenocarcinoma due to inadequate surgical resection. Histopathological study of lateral tumour spread and surgical excision. Lancet 1986, 2, 996–999. [Google Scholar] [CrossRef]
- Adam, I.J.; Mohamdee, M.O.; Martin, I.G.; Scott, N.; Finan, P.J.; Johnston, D.; Dixon, M.F.; Quirke, P. Role of circumferential margin involvement in the local recurrence of rectal cancer. Lancet 1994, 344, 707–711. [Google Scholar] [CrossRef]
- Pollett, W.G.; Nicholls, R.J.; Chir, M. The relationship between the extent of distal clearance and survival and local recurrence rates after curative anterior resection for carcinoma of the rectum. Ann. Surg. 1983, 198, 159–163. [Google Scholar] [CrossRef]
- Edge, S.B.; Compton, C.C. The american joint committee on cancer: The 7th edition of the AJCC cancer staging manual and the future of TNM. Ann. Surg. Oncol. 2010, 17, 1471–1474. [Google Scholar] [CrossRef]
- Ueno, H.; Kajiwara, Y.; Shimazaki, H.; Shinto, E.; Hashiguchi, Y.; Nakanishi, K.; Maekawa, K.; Katsurada, Y.; Nakamura, T.; Mochizuki, H.; et al. New criteria for histologic grading of colorectal cancer. Am. J. Surg. Pathol. 2012, 36, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Dindo, D.; Demartines, N.; Clavien, P.A. Classification of surgical complications: A new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann. Surg. 2004, 240, 205. [Google Scholar] [CrossRef]
- Heald, R.J.; Ryall, R.D. Recurrence and survival after total mesorectal excision for rectal cancer. Lancet 1986, 1, 1479–1482. [Google Scholar] [CrossRef]
- Knight, C.D.; Griffen, F.D. An improved technique for low anterior resection of the rectum using the EEA stapler. Surgery 1980, 88, 710–714. [Google Scholar]
- Gastinger, I.; Marusch, F.; Koch, A.; Meyer, F.; Nestler, G.; Schmidt, U.; Meyer, J.; Eggert, A.; Albrecht, R.; Köckerling, F.; et al. Hartmann’s procedure indication in colorectal carcinoma. Chirurg 2004, 75, 1191–1196. [Google Scholar] [CrossRef]
- Miles, W.E. A method of performing abdomino-perineal excision for carcinoma of the rectum and of the terminal portion of the pelvic colon (1908). CA Cancer J. Clin. 1971, 21, 361–364. [Google Scholar] [CrossRef]
- Rosenbaum, P.R.; Rubin, D.B. Constructing a control group using multivariate matched sampling methods that incorporate the propensity score. Am. Stat. 1985, 39, 33–38. [Google Scholar]
- Zhang, Z.; Kim, H.J.; Lonjon, G.; Zhu, Y. Balance diagnostics after propensity score matching. Ann. Trans. Med. 2019, 7, 16. [Google Scholar] [CrossRef] [PubMed]
- Steyerberg, E.W.; Vergouwe, Y. Towards better clinical prediction models: Seven steps for development and an ABCD for validation. Eur. Heart J. 2014, 35, 1925–1931. [Google Scholar] [CrossRef] [PubMed]
- Schwenk, W.; Neudecker, J.; Haase, O.; Müller, J.M. Short term benefits for laparoscopic colorectal resection. Cochrane Database Syst. Rev. 2001. [Google Scholar] [CrossRef]
- Vargas, G.M.; Sieloff, E.P.; Parmar, A.D.; Tamirisa, N.P.; Mehta, H.B.; Riall, T.S. Laparoscopy decreases complications for obese patients undergoing elective rectal surgery. Surg. Endosc. 2016, 30, 1826–1832. [Google Scholar] [CrossRef][Green Version]
- Panteleimonitis, S.; Popeskou, S.; Harper, M.; Kandala, N.; Figueiredo, N.; Qureshi, T.; Parvaiz, A. Minimally invasive colorectal surgery in the morbid obese: Does size really matter? Surg. Endosc. 2018, 32, 3486–3494. [Google Scholar] [CrossRef]
- Di, B.; Li, Y.; Wei, K.; Xiao, X.; Shi, J.; Zhang, Y.; Yang, X.; Gao, P.; Zhang, K.; Yuan, Y.; et al. Laparoscopic versus open surgery for colon cancer: A meta-analysis of 5-year follow-up outcomes. Surg. Oncol. 2013, 22, 39–43. [Google Scholar] [CrossRef]
- Miles, R.H.; Carballo, R.E.; Prinz, R.A.; McMahon, M.; Pulawski, G.; Olen, R.N.; Dahlinghaus, D.L. Laparoscopy: The preferred method of cholecystectomy in the morbidly obese. Surgery 1992, 112, 818–822. [Google Scholar]
- Law, W.L.; Poon, J.T.C.; Fan, J.K.M.; Lo, O.S.H. Survival following laparoscopic versus open resection for colorectal cancer. Int. J. Colorectal Dis. 2012, 27, 1077–1085. [Google Scholar] [CrossRef] [PubMed]
- Balentine, C.J.; Marshall, C.; Robinson, C.; Wilks, J.; Anaya, D.; Albo, D.; Berger, D.H. Obese patients benefit from minimally invasive colorectal cancer surgery. J. Surg. Res. 2010, 163, 29–34. [Google Scholar] [CrossRef] [PubMed]
- Hotta, T.; Takifuji, K.; Yokoyama, S.; Matsuda, K.; Oku, Y.; Nasu, T.; Tamura, K.; Ieda, J.; Yamamoto, N.; Yamaue, H. The impact of obesity on learning laparoscopic surgery for colon cancer. J. Laparoendosc. Adv. Surg. Tech. A 2012, 22, 635–640. [Google Scholar] [CrossRef]
- Abraham, N.S.; Young, J.M.; Solomon, M.J. Meta-analysis of short-term outcomes after laparoscopic resection for colorectal cancer. Br. J. Surg. 2004, 91, 1111–1124. [Google Scholar] [CrossRef]
- Leroy, J.; Ananian, P.; Rubino, F.; Claudon, B.; Mutter, D.; Marescaux, J. The impact of obesity on technical feasibility and postoperative outcomes of laparoscopic left colectomy. Ann. Surg. 2005, 241, 69–76. [Google Scholar] [CrossRef] [PubMed]
- Martin, S.T.; Stocchi, L. Laparoscopic colorectal resection in the obese patient. Clin. Colon Rectal Surg. 2011, 24, 263–273. [Google Scholar] [CrossRef]
- He, Y.; Wang, J.; Bian, H.; Deng, X.; Wang, Z. BMI as a predictor for perioperative outcome of laparoscopic colorectal surgery: A pooled analysis of comparative studies. Dis. Colon Rectum 2017, 60, 433–445. [Google Scholar] [CrossRef]
- Gurunathan, U.; Ramsay, S.; Mitrić, G.; Way, M.; Wockner, L.; Myles, P. Association between obesity and wound infection following colorectal surgery: Systematic review and meta-analysis. J. Gastrointest. Surg. 2017, 21, 1700–1712. [Google Scholar] [CrossRef] [PubMed]
- Stracci, F.; Bianconi, F.; Leite, S.; Liso, A.; La Rosa, F.; Lancellotta, V.; van de Velde, C.J.H.; Aristei, C. Linking surgical specimen length and examined lymph nodes in colorectal cancer patients. Eur. J. Surg. Oncol. 2016, 42, 260–265. [Google Scholar] [CrossRef]
- Rivard, J.D.; Hochman, D.J. Equivalency of lymph node harvest in laparoscopic versus open colorectal cancer procedures: An early surgeon’s perspective. Surg. Laparosc. Endosc. Percutaneous Tech. 2011, 21, 14–16. [Google Scholar] [CrossRef] [PubMed]
- Benson, A.B.; Venook, A.P.; Al-Hawary, M.M.; Arain, M.A.; Chen, Y.J.; Ciombor, K.K.; Cohen, S.; Cooper, H.S.; Deming, D.; Farkas, L.; et al. Colon cancer, version 2.2021, NCCN clinical practice guidelines in oncology. J. Natl. Compr. Cancer Netw. 2021, 19, 329–359. [Google Scholar] [CrossRef] [PubMed]
- Araujo, S.E.A.; da Silva e Sousa, A.H., Jr.; de Campos, F.G.C.M.; Habr-Gama, A.; Dumarco, R.B.; de Paris Caravatto, P.P.; Nahas, S.C.; da Silva, J.H.; Kiss, D.R.; Gama-Rodrigues, J.J. Conventional approach x laparoscopic abdominoperineal resection for rectal cancer treatment after neoadjuvant chemoradiation: Results of a prospective randomized trial. Rev. Hosp. Clín. 2003, 58, 133–140. [Google Scholar] [CrossRef]
- Hardiman, K.; Chang, E.T.; Diggs, B.S.; Lu, K.C. Laparoscopic colectomy reduces morbidity and mortality in obese patients. Surg. Endosc. 2013, 27, 2907–2910. [Google Scholar] [CrossRef] [PubMed]
| Before Matching | After Matching | |||||||
|---|---|---|---|---|---|---|---|---|
| Open | Video | Effect Size | p * | Open | Video | Effect Size | p * | |
| Laparoscopic | Laparoscopic | |||||||
| Surgery | Surgery | Surgery | Surgery | |||||
| Frequency (%) | 51 (37.00) | 87 (63.00) | 49 (50.00) | 49 (50.00) | ||||
| Age (years) | 72 ± 9.02 | 66.83 ± 10.27 | 0.535 | 0.001 | 71.78 ± 9.1 | 63.12 ± 11.02 | 0.856 | 0.001 ‡ |
| Sex | ||||||||
| Female | 25 (49.00) | 27 (31.00) | 0.373 | 0.029 | 24 (49.00) | 12 (24.50) | 0.525 | 0.001 |
| Male | 26 (51.00) | 60 (69.00) | 25 (51.00) | 37 (75.50) | ||||
| BMI (Kg/m2) | 35.13 ± 5.45 | 32.98 ± 3.56 | 0.467 | 0.001 | 35.23 ± 5.52 | 32.05 ± 2.55 | 0.741 | 0.001 |
| Grading | ||||||||
| G1 | 9 (22.00) | 16 (20.50) | 0.684 | 0.001 | 8 (20.00) | 9 (19.60) | 0.702 | 0.001 |
| G2 | 18 (43.90) | 55 (70.50) | 18 (45.00) | 33 (71.70) | ||||
| G3 | 14 (34.10) | 7 (9.00) | 14 (35.00) | 4 (8.70) | ||||
| Tumor location | ||||||||
| Right | 19 (37.30) | 22 (25.30) | 0.101 | 19 (38.80) | 3 (6.10) | 1.036 | 0.001 | |
| Left | 10 (19.60) | 31 (35.60) | 0.385 | 9 (18.40) | 27 (55.10) | |||
| Rectum | 22 (22.43) | 34 (39.10) | 21 (42.90) | 19 (38.80) | ||||
| Type of surgery Anastomosis | ||||||||
| Hartmann Technique | 4 (7.80) | 1 (1.10) | 0.398 | 0.064 a | 4 (8.20) | 1 (2.00) | 0.354 | 0.219 a |
| Mile Techniques | 1 (2.00) | 1 (1.10) | 1 (2.00) | -- | ||||
| Postoperative hospital stay (days) | 10.16 ± 7.65 | 7.07 ± 3.05 | 0.103 | 0.001 | 10.16 ± 7.65 | 6.33 ± 1.25 | 0.700 | 0.001 |
| Harvested Lymph Nodes (n) | 19.09 ± 9.4 | 16.82 ± 9.75 | 0.237 | 0.101 | 19.18 ± 9.49 | 15 ± 8.23 | 0.470 | 0.019 |
| Staging | ||||||||
| 0 | 1 (2.50) | -- | 0.201 | 0.192 a | 1 (2.50) | -- | 0.538 | 0.652 a |
| I | 9 (22.50) | 24 (31.20) | 9 (22–50) | 13 (27.70) | ||||
| II | 12 (30.00) | 28 (36.40) | 12 (30.00) | 17 (36.20) | ||||
| III | 14 (35.00) | 23 (29.90) | 14 (35.00) | 15 (31.90) | ||||
| IV | 4 (10.00) | 2 (2.60) | 4 (10.00) | 2 (4.30) | ||||
| V | -- | -- | -- | -- | ||||
| Operative time (min) | 140.11 ± 50.80 | 144.94 ± 42.33 | 0.103 | 0.332 | 139.22 ± 51.01 | 147.81 ± 41.84 | 0.184 | 0.192 |
| Previous surgery (yes) | 30 (58.80) | 50 (57.50) | 0.027 | 0.871 | 28 (57.10) | 24 (49.00) | 0.164 | 0.411 |
| Readmission (yes) | 6 (11.80) | 10 (11.50) | 0.008 | 0.962 | 6 (12.20) | 5 (10.20) | 0.065 | 0.742 |
| Re-intervention (yes) | 7 (13.70) | 19 (21.80) | 0.213 | 0.231 | 6 (12.20) | 13 (26.50) | 0.367 | 0.073 |
| Length of Specimen (cm) | 34.11 ± 13.8 | 29.85 ± 10.69 | 0.345 | 0.042 | 34.18 ± 13.95 | 28.84 ± 7.65 | 0.475 | 0.010 |
| Clearance (cm) | 6.62 ± 6.60 | 5.35 ± 4.02 | 0.232 | 0.991 | 6.71 ± 6.66 | 5.11 ± 4.12 | 0.290 | 0.742 |
| CRM (<1 mm) | 2 (10.50) | 2 (6.70) | 0.138 | 0.623 | 2 (11.10) | 2 (11.10) | 0.001 | 0.901 |
| Time to flatus (days) | 3.12 ± 1.08 | 2.31 ± 0.74 | 0.880 | 0.001 | 3.12 ± 1.08 | 2.3 ± 0.68 | 0.903 | 0.001 |
| Time to canalization | ||||||||
| Anastomosis (days) | 4.59 ± 1.31 | 3.77 ± 1.16 | 0.660 | 0.001 | 4.59 ± 1.31 | 3.67 ± 1.12 | 0.757 | 0.001 |
| Stoma (days) | 2.27 ± 1.27 | 1.57 ± 0.728 | 0.683 | 0.082 | 2.27 ± 1.27 | 1.47 ± 0.8 | 0.755 | 0.039 |
| Liquid oral diet (days) | 2.77 ± 1.2 | 1.96 ± 0.99 | 0.736 | 0.001 | 2.77 ± 1.2 | 1.86 ± 0.89 | 0.868 | 0.001 |
| Solid oral diet (days) | 4.41 ± 1.24 | 3.45 ± 1.15 | 0.804 | 0.001 | 4.41 ± 1.24 | 3.2 ± 1.12 | 1.019 | 0.001 |
| Postoperative complications | ||||||||
| Dehiscence (yes) | 11 (21.60) | 5 (5.70) | 0.473 | 0.001 | 10 (20.40) | 2 (4.10) | 0.514 | 0.001 |
| (Clavien–Dindo) | 1.36 ± 1.37 | 0.77 ± 1.37 | 0.471 | 0.010 | 1.22 ± 1.22 | 0.63 ± 0.95 | 0.538 | 0.021 |
| 0 | 19 (40.40) | 51 (60.00) | 0.505 | 0.152 a | 19 (42.20) | 32 (65.30) | 0.556 | 0.14 a |
| I | 5 (10.60) | 10 (11.80) | 5 (11.10) | 5 (10.20) | ||||
| II | 15 (31.90) | 19 (22.40) | 15 (33.30) | 10 (20.40) | ||||
| III | 4 (8.50) | 2 (2.40) | 4 (8.90) | 2 (4.10) | ||||
| IV | 3 (6.40) | 3 (3.50) | 2 (4.40) | -- | ||||
| V | 1 (2.10) | -- | -- | -- | ||||
| Matching Variables | Type | Diff. Adj. | M. Threshold |
|---|---|---|---|
| Age (years) | Continue | −0.8422 | Not Balanced, >0.1 |
| Sex (Male) | Binary | 0.2449 | Not Balanced, >0.1 |
| BMI (Kg/m2) | Continue | −0.8955 | Not Balanced, >0.1 |
| Tumor Location | |||
| Right | Binary | −0.3265 | Not Balanced, >0.1 |
| Rectum | Binary | −0.0408 | Balanced, <0.1 |
| Left | Binary | 0.3673 | Not Balanced, >0.1 |
| Covariates | Coefficient | Std. Err. | CI 95% | p | p-Adjusted “Bonferroni” |
|---|---|---|---|---|---|
| Model 1 | |||||
| Treatment (VL) | −3.09 | 0.93 | −4.91 to −1.28 | 0.001 | 0.011 |
| Model 2 | |||||
| Treatment (VL) | −2.84 | 1.00 | −4.81 to −0.88 | 0.005 | 0.019 |
| Age (years) | 0.03 | 0.05 | −0.06 to 0.12 | 0.525 | 0.992 |
| Sex (Male) | −0.84 | 0.98 | −2.76 to 1.09 | 0.396 | 0.993 |
| BMI (Kg/m2) | −0.02 | 0.11 | −0.23 to 0.19 | 0.855 | 0.992 |
| Model 3 | |||||
| Treatment (VL) | −3.09 | 0.99 | −5.02 to −1.15 | 0.002 | 0.012 |
| Age (years) | 0.05 | 0.05 | −0.04 to 0.13 | 0.323 | 0.993 |
| Sex (Male) | −0.87 | 0.96 | −2.75 to 1.02 | 0.369 | 0.992 |
| BMI (Kg/m2) | −0.01 | 0.11 | −0.21 to 0.20 | 0.958 | 0.991 |
| Location (Rectum) | 2.91 | 1.06 | 0.83 to 4.99 | 0.006 | 0.039 |
| Location (Left) | 2.72 | 1.17 | 0.43 to 5.02 | 0.021 | 0.141 |
| Model 4 | |||||
| Treatment (VL) | −1.89 | 0.79 | −3.44 to −0.33 | 0.019 | 0.241 |
| Age (years) | 0.03 | 0.03 | −0.03 to 0.10 | 0.348 | 0.992 |
| Sex (Male) | −1.06 | 0.73 | −2.48 to 0.36 | 0.147 | 0.991 |
| BMI (Kg/m2) | −0.08 | 0.08 | −0.24 to 0.07 | 0.286 | 0.072 |
| Location (Rectum) | 2.70 | 0.97 | 0.80 to 4.59 | 0.006 | 0.991 |
| Location (Left) | 1.45 | 0.91 | −0.33 to 3.23 | 0.113 | 0.992 |
| Length of Specimen (cm) | −0.01 | 0.03 | −0.07 to 0.05 | 0.709 | 0.992 |
| Previous surgery (yes) | 0.29 | 0.66 | −1.00 to 1.58 | 0.662 | 0.993 |
| Staging | 0.35 | 0.37 | −0.39 to 1.08 | 0.369 | 0.992 |
| Clearance < 1 cm/CRM < 1 mm | −1.67 | 1.27 | −4.17 to 0.82 | 0.191 | 0.991 |
| Harvested Lymph nodes (n) | 0.03 | 0.04 | −0.05 to 0.11 | 0.444 | 0.993 |
| Ileostomy | 0.59 | 0.95 | −1.28 to 2.46 | 0.535 | 0.994 |
| Colostomy | −1.03 | 1.86 | −4.67 to 2.62 | 0.582 | 0.992 |
| Covariates | OR | CI 95% | p * | p-Adjusted “Bonferroni” |
|---|---|---|---|---|
| Model 1 | ||||
| Treatment (VL) | 0.41 | 0.20 to 0.81 | 0.011 | 0.012 |
| Model 2 | ||||
| Treatment (VL) | 0.41 | 0.19 to 0.86 | 0.019 | 0.072 |
| Age (years) | 0.99 | 0.96 to 1.03 | 0.980 | 0.992 |
| Sex (Male) | 1.13 | 0.55 to 2.35 | 0.733 | 0.991 |
| BMI (Kg/m2) | 1.01 | 0.93 to 1.09 | 0.714 | 0.993 |
| Model 3 | ||||
| Treatment (VL) | 0.35 | 0.16 to 0.76 | 0.008 | 0.050 |
| Age (years) | 1.01 | 0.97 to 1.04 | 0.777 | 0.991 |
| Sex (Male) | 1.15 | 0.56 to 2.40 | 0.702 | 0.992 |
| BMI (Kg/m2) | 1.02 | 0.94 to 1.09 | 0.690 | 0.993 |
| Location (Rectum) | 2.20 | 0.97 to 5.10 | 0.060 | 0.362 |
| Location (Left) | 2.25 | 0.89 to 5.82 | 0.086 | 0.513 |
| Model 4 | ||||
| Treatment (VL) | 0.37 | 0.14 to 0.95 | 0.041 | 0.531 |
| Age (years) | 0.99 | 0.95 to 1.03 | 0.625 | 0.992 |
| Sex (Male) | 0.67 | 0.28 to 1.62 | 0.381 | 0.993 |
| BMI (Kg/m2) | 1.01 | 0.92 to 1.09 | 0.912 | 0.992 |
| Location (Rectum) | 2.65 | 0.82 to 8.87 | 0.106 | 0.991 |
| Location (Left) | 2.18 | 0.69 to 7.25 | 0.190 | 0.993 |
| Length of Specimen (cm) | 0.99 | 0.96 to 1.03 | 0.856 | 0.994 |
| Previous surgery (yes) | 1.78 | 0.79 to 4.09 | 0.162 | 0.993 |
| Staging | 1.35 | 0.84 to 2.19 | 0.205 | 0.992 |
| Clearance < 1 cm/CRM < 1 mm | 0.17 | 0.02 to 0.88 | 0.062 | 0.681 |
| Lymph nodes | 1.02 | 0.97 to 1.07 | 0.326 | 0.993 |
| Ileostomy | 2.14 | 0.71 to 6.56 | 0.177 | 0.994 |
| Colostomy | 4.81 | 0.55 to 41.39 | 0.147 | 0.993 |
| Models | Res. Df. | RSS | Df. | Sum of Sq. | F | Pr (>F) |
|---|---|---|---|---|---|---|
| Restricted model | 135 | 3912.4 | ||||
| Model 1 | 134 | 3612.3 | 1 | 300.12 | 11.133 | 0.001 |
| Restricted model | 135 | 3912.4 | ||||
| Model 2 | 131 | 3580.1 | 4 | 332.29 | 3.039 | 0.019 |
| Restricted model | 135 | 3912.4 | ||||
| Model 3 | 129 | 3357.2 | 6 | 555.19 | 3.555 | 0.002 |
| Restricted model | 111 | 1335.1 | ||||
| Model 4 | 98 | 1068.9 | 13 | 266.18 | 1.877 | 0.041 |
| Models | Res. Df. | Df. | Chisq. | Pr (Chisq) |
|---|---|---|---|---|
| Restricted model | 127 | |||
| Model 1 | 126 | 1 | 6.413 | 0.001 |
| Restricted model | 127 | |||
| Model 2 | 123 | 4 | 6.594 | 0.15 |
| Restricted model | 127 | |||
| Model 3 | 121 | 6 | 10.376 | 0.10 |
| Restricted model | 108 | |||
| Model 4 | 95 | 13 | 17.214 | 0.18 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bizzoca, C.; Zupo, R.; Aquilino, F.; Castellana, F.; Fiore, F.; Sardone, R.; Vincenti, L. Video-Laparoscopic versus Open Surgery in Obese Patients with Colorectal Cancer: A Propensity Score Matching Study. Cancers 2021, 13, 1844. https://doi.org/10.3390/cancers13081844
Bizzoca C, Zupo R, Aquilino F, Castellana F, Fiore F, Sardone R, Vincenti L. Video-Laparoscopic versus Open Surgery in Obese Patients with Colorectal Cancer: A Propensity Score Matching Study. Cancers. 2021; 13(8):1844. https://doi.org/10.3390/cancers13081844
Chicago/Turabian StyleBizzoca, Cinzia, Roberta Zupo, Fabrizio Aquilino, Fabio Castellana, Felicia Fiore, Rodolfo Sardone, and Leonardo Vincenti. 2021. "Video-Laparoscopic versus Open Surgery in Obese Patients with Colorectal Cancer: A Propensity Score Matching Study" Cancers 13, no. 8: 1844. https://doi.org/10.3390/cancers13081844
APA StyleBizzoca, C., Zupo, R., Aquilino, F., Castellana, F., Fiore, F., Sardone, R., & Vincenti, L. (2021). Video-Laparoscopic versus Open Surgery in Obese Patients with Colorectal Cancer: A Propensity Score Matching Study. Cancers, 13(8), 1844. https://doi.org/10.3390/cancers13081844