A Novel Two-Lipid Signature Is a Strong and Independent Prognostic Factor in Ovarian Cancer
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Cohorts and Samples
2.2. Lipidomic and Conventional Biomarker Analyses
2.3. Statistical Analyses
3. Results
3.1. Selection of a Prognostic Two-Lipid Signature
3.2. Prognostic Value of the Two-Lipid Signature Test
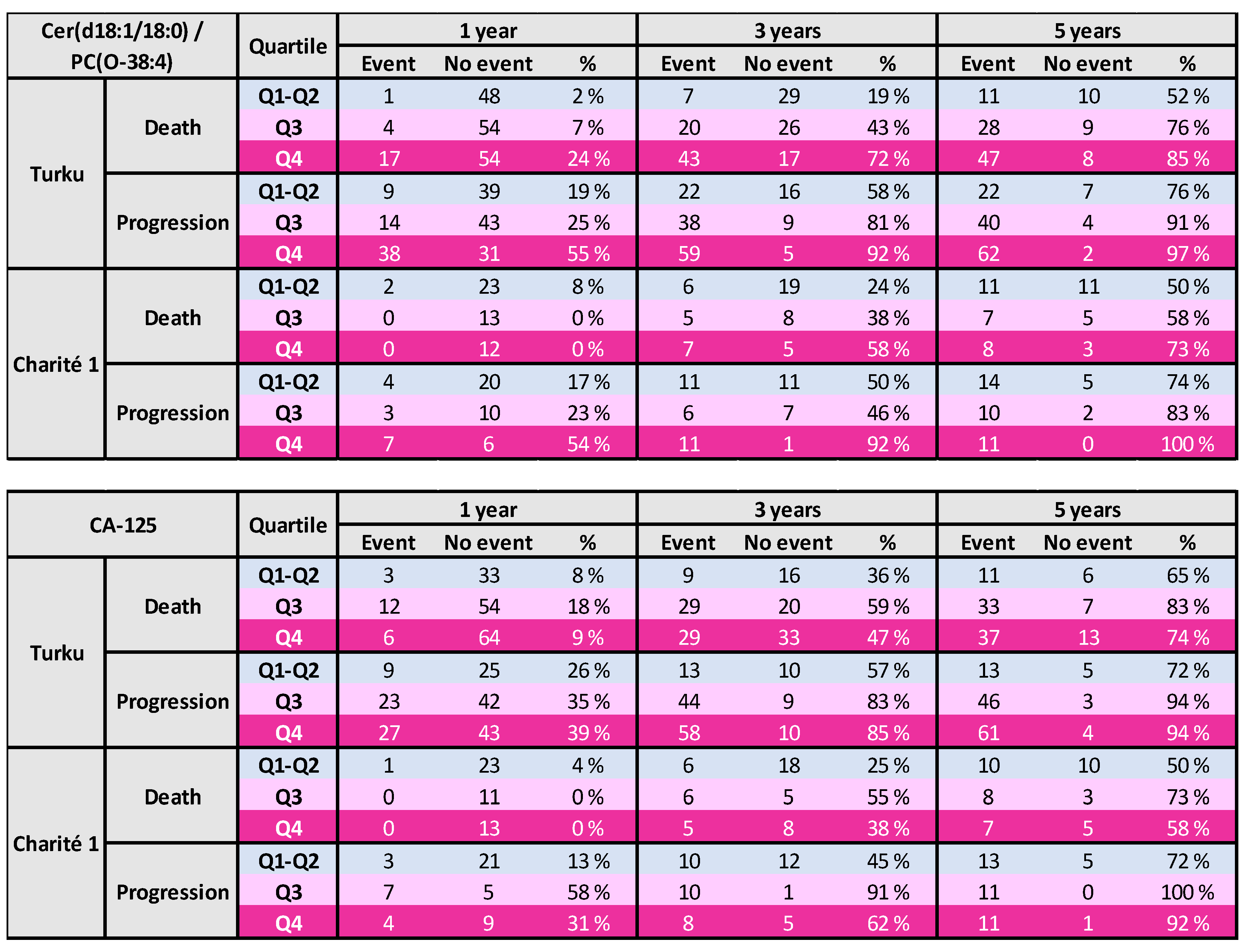
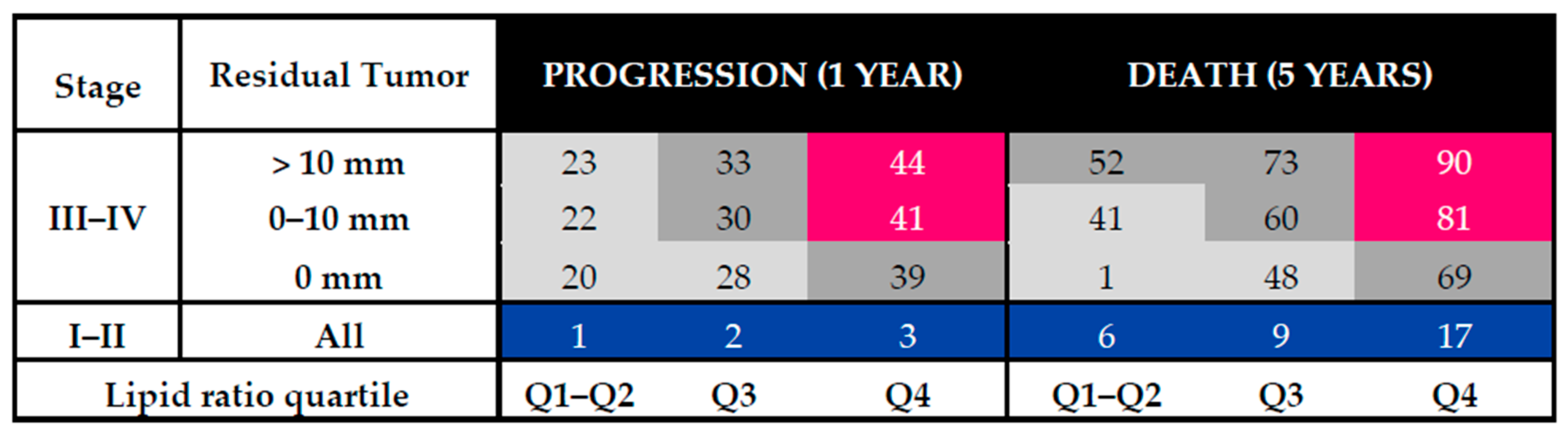
3.3. Risk Tables for Ovarian Cancer Patients
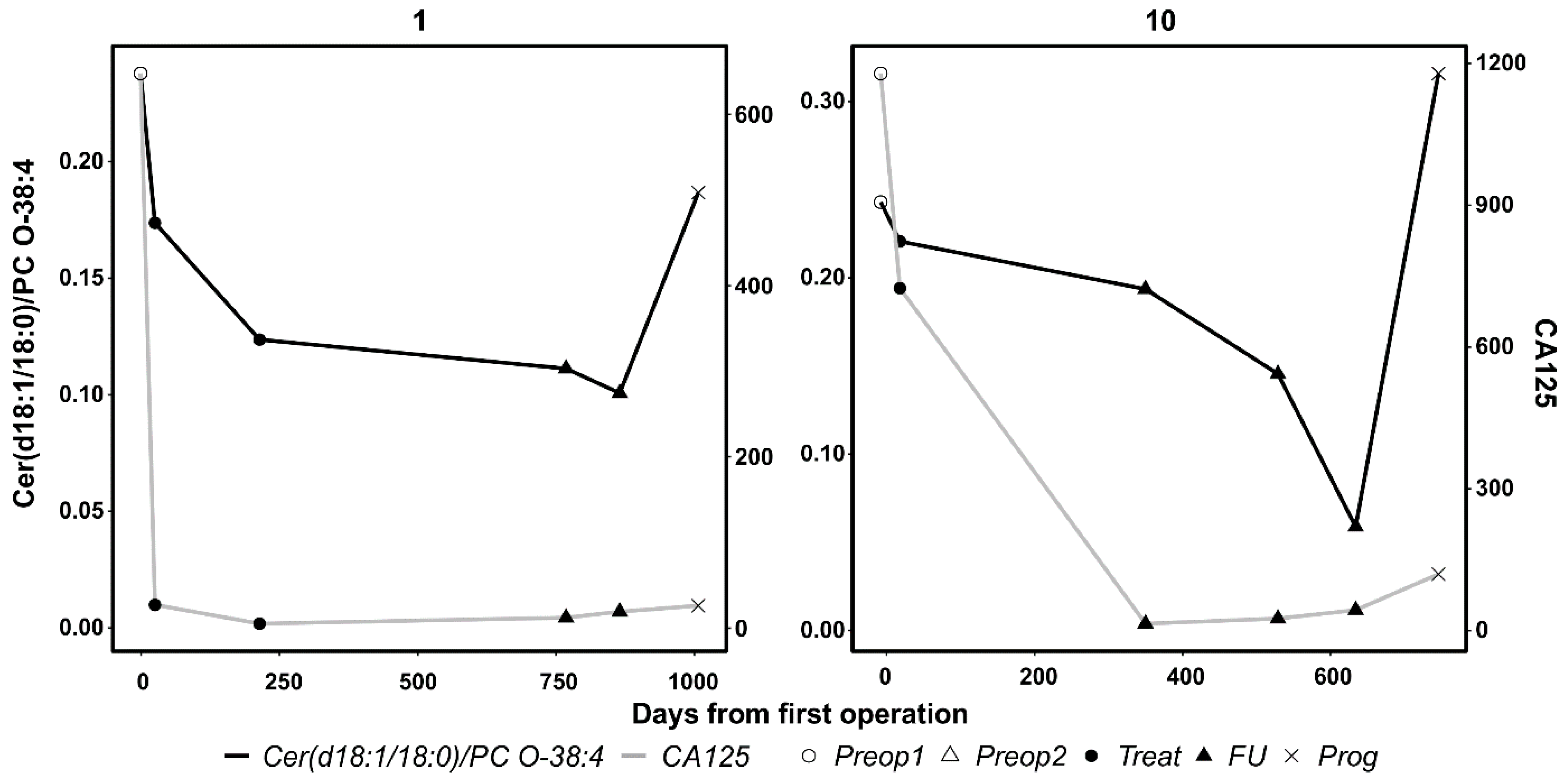
3.4. Results From Longitudinal Analyses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Perrotti, F.; Rosa, C.; Cicalini, I.; Sacchetta, P.; Del Boccio, P.; Genovesi, D.; Pieragostino, D. Advances in Lipidomics for Cancer Biomarkers Discovery. Int. J. Mol. Sci. 2016, 17, 1992. [Google Scholar] [CrossRef]
- Zhao, G.; Cardenas, H.; Matei, D. Ovarian Cancer—Why Lipids Matter. Cancers 2019, 11, 1870. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef]
- Yan, F.; Zhao, H.; Zeng, Y. Lipidomics: A promising cancer biomarker. Clin. Transl. Med. 2018, 21–23. [Google Scholar] [CrossRef] [PubMed]
- Ke, C.; Hou, Y.; Zhang, H.; Fan, L.; Ge, T.; Guo, B.; Zhang, F.; Yang, K.; Wang, J.; Lou, G.; et al. Large-scale profiling of metabolic dysregulation in ovarian cancer. Int. J. Cancer 2014. [Google Scholar] [CrossRef] [PubMed]
- Torre, L.A.; Trabert, B.; DeSantis, C.E.; Miller, K.D.; Samimi, G.; Runowicz, C.D.; Gaudet, M.M.; Jemal, A.; Siegel, R.L. Ovarian cancer statistics, 2018. CA Cancer J. Clin. 2018, 68, 284–296. [Google Scholar] [CrossRef] [PubMed]
- Karam, A.K.; Karlan, B.Y. Ovarian cancer: The duplicity of CA125 measurement. Nat. Rev. Clin. Oncol. 2010, 7, 335–339. [Google Scholar] [CrossRef]
- Ledermann, J.A.; Raja, F.A.; Fotopoulou, C.; Gonzalez-Martin, A.; Colombo, N.; Sessa, C. Newly diagnosed and relapsed epithelial ovarian carcinoma: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up †. Ann. Oncol. 2013, 24, 24–32. [Google Scholar] [CrossRef]
- Norquist, B.M.; Brady, M.F.; Harrell, M.I.; Walsh, T.; Lee, M.K.; Gulsuner, S.; Bernards, S.S.; Casadei, S.; Burger, R.A.; Tewari, K.S.; et al. Mutations in homologous recombination genes and outcomes in ovarian carcinoma patients in GOG 218: An NRG oncology/Gynecologic oncology group study. Clin. Cancer Res. 2018, 24, 777–783. [Google Scholar] [CrossRef]
- Monk, B.J.; Pujade-Lauraine, E.; Burger, R.A. Integrating bevacizumab into the management of epithelial ovarian cancer: The controversy of front-line versus recurrent disease. Ann. Oncol. 2013, 24, x53–x58. [Google Scholar] [CrossRef]
- Colombo, N.; Sessa, C.; Du Bois, A.; Ledermann, J.; McCluggage, W.G.; McNeish, I.; Morice, P.; Pignata, S.; Ray-Coquard, I.; Vergote, I.; et al. ESMO-ESGO consensus conference recommendations on ovarian cancer: Pathology and molecular biology, early and advanced stages, borderline tumours and recurrent disease. Int. J. Gynecol. Cancer 2019, 29, 728–760. [Google Scholar] [CrossRef]
- Lin, H.M.; Mahon, K.L.; Weir, J.M.; Mundra, P.A.; Spielman, C.; Briscoe, K.; Gurney, H.; Mallesara, G.; Marx, G.; Stockler, M.R.; et al. A distinct plasma lipid signature associated with poor prognosis in castration-resistant prostate cancer. Int. J. Cancer 2017, 141, 2112–2120. [Google Scholar] [CrossRef]
- Cotte, A.K.; Cottet, V.; Aires, V.; Mouillot, T.; Rizk, M.; Vinault, S.; Binquet, C.; De Barros, J.P.P.; Hillon, P.; Delmas, D. Phospholipid profiles and hepatocellular carcinoma risk and prognosis in cirrhotic patients. Oncotarget 2019, 10, 2161–2172. [Google Scholar] [CrossRef]
- Bachmayr-Heyda, A.; Aust, S.; Auer, K.; Meier, S.M.; Schmetterer, K.G.; Dekan, S.; Gerner, C.; Pils, D. Integrative Systemic and Local Metabolomics with Impact on Survival in High-Grade Serous Ovarian Cancer. Clin. Cancer Res. 2017, 23, 2081–2092. [Google Scholar] [CrossRef] [PubMed]
- Braicu, E.I.; Darb-Esfahani, S.; Schmitt, W.D.; Koistinen, K.M.; Heiskanen, L.; Pöhö, P.; Budczies, J.; Kuhberg, M.; Dietel, M.; Frezza, C.; et al. High-grade serous carcinoma patients exhibit profound alterations in lipid metabolism. Oncotarget 2017, 8, 102912–102922. [Google Scholar] [CrossRef] [PubMed]
- Shida, D.; Takabe, K.; Kapitonov, D.; Milstien, S.; Spiegel, S. Targeting SphK1 as a New Strategy against Cancer. Curr. Drug Targets 2008, 9, 662–673. [Google Scholar] [CrossRef] [PubMed]
- Pyragius, C.E.; Fuller, M.; Ricciardelli, C.; Oehler, M.K. Aberrant lipid metabolism: An emerging diagnostic and therapeutic target in ovarian cancer. Int. J. Mol. Sci. 2013, 14, 7742–7756. [Google Scholar] [CrossRef] [PubMed]
- Salminen, L.; Nadeem, N.; Jain, S.; Grènman, S.; Carpén, O.; Hietanen, S.; Oksa, S.; Lamminmäki, U.; Pettersson, K.; Gidwani, K.; et al. A longitudinal analysis of CA125 glycoforms in the monitoring and follow up of high grade serous ovarian cancer. Gynecol. Oncol. 2020, 156, 689–694. [Google Scholar] [CrossRef]
- Harter, P.; Sehouli, J.; Lorusso, D.; Reuss, A.; Vergote, I.; Marth, C.; Kim, J.-W.; Raspagliesi, F.; Lampe, B.; Aletti, G.; et al. A Randomized Trial of Lymphadenectomy in Patients with Advanced Ovarian Neoplasms. N. Engl. J. Med. 2019, 380, 822–832. [Google Scholar] [CrossRef]
- Hilvo, M.; De Santiago, I.; Gopalacharyulu, P.; Schmitt, W.D.; Budczies, J.; Kuhberg, M.; Dietel, M.; Aittokallio, T.; Markowetz, F.; Denkert, C.; et al. Accumulated metabolites of hydroxybutyric acid serve as diagnostic and prognostic biomarkers of ovarian high-grade serous carcinomas. Cancer Res. 2016, 76. [Google Scholar] [CrossRef] [PubMed]
- John, G.; Rustin, S.; Vergote, I.; Eisenhauer, E.; Pujade-lauraine, E.; Quinn, M.; Thigpen, T.; Bois, A.; Kristensen, G. Definitions for Response and Progression in Ovarian Cancer Clinical Trials Incorporating RECIST 1.1 and CA 125 Agreed by the Gynecological Cancer Intergroup ( GCIG ). Int. J. Gynecol. Cancer 2011, 21, 419–423. [Google Scholar] [CrossRef]
- Folch, J.; Lees, M.; Sloane Stanley, G.H. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem. 1957, 226, 497–509. [Google Scholar] [CrossRef]
- Weir, J.M.; Wong, G.; Barlow, C.K.; Greeve, M.A.; Kowalczyk, A.; Almasy, L.; Comuzzie, A.G.; Mahaney, M.C.; Jowett, J.B.M.; Shaw, J.; et al. Plasma lipid profiling in a large population-based cohort. J. Lipid Res. 2013, 54, 2898–2908. [Google Scholar] [CrossRef] [PubMed]
- Laaksonen, R.; Ekroos, K.; Sysi-Aho, M.; Hilvo, M.; Vihervaara, T.; Kauhanen, D.; Suoniemi, M.; Hurme, R.; März, W.; Scharnagl, H.; et al. Plasma ceramides predict cardiovascular death in patients with stable coronary artery disease and acute coronary syndromes beyond LDL-cholesterol. Eur. Heart J. 2016, 37, 1967–1976. [Google Scholar] [CrossRef]
- Hilvo, M.; Meikle, P.J.; Pedersen, E.R.; Tell, G.S.; Dhar, I.; Brenner, H.; Schöttker, B.; Lääperi, M.; Kauhanen, D.; Koistinen, K.M.; et al. Development and validation of a ceramide- and phospholipid-based cardiovascular risk estimation score for coronary artery disease patients. Eur. Heart J. 2020, 41, 371–380. [Google Scholar] [CrossRef] [PubMed]
- Takaya, H.; Nakai, H.; Takamatsu, S.; Mandai, M.; Matsumura, N. Homologous recombination deficiency status-based classification of high-grade serous ovarian carcinoma. Sci. Rep. 2020, 10, 1–8. [Google Scholar] [CrossRef]
- Du Bois, A.; Reuss, A.; Pujade-Lauraine, E.; Harter, P.; Ray-Coquard, I.; Pfisterer, J. Role of surgical outcome as prognostic factor in advanced epithelial ovarian cancer: A combined exploratory analysis of 3 prospectively randomized phase 3 multicenter trials: By the arbeitsgemeinschaft gynaekologische onkologie studiengruppe ovarialkarzin. Cancer 2009, 115, 1234–1244. [Google Scholar] [CrossRef] [PubMed]
- Aletti, G.D.; Dowdy, S.C.; Gostout, B.S.; Jones, M.B.; Stanhope, C.R.; Wilson, T.O.; Podratz, K.C.; Cliby, W.A. Aggressive Surgical Effort and Improved Survival in Advanced-Stage Ovarian Cancer. Obstet. Gynecol. 2006, 107, 77–85. [Google Scholar] [CrossRef] [PubMed]
- Messias, M.C.F.; Mecatti, G.C.; Priolli, D.G.; De Oliveira Carvalho, P. Plasmalogen lipids: Functional mechanism and their involvement in gastrointestinal cancer. Lipids Health Dis. 2018, 17, 1–12. [Google Scholar] [CrossRef]
- Clifford, C.; Vitkin, N.; Nersesian, S.; Reid-Schachter, G.; Francis, J.A.; Koti, M. Multi-omics in high-grade serous ovarian cancer: Biomarkers from genome to the immunome. Am. J. Reprod. Immunol. 2018, 80, 1–10. [Google Scholar] [CrossRef]
- Kauhanen, D.; Sysi-Aho, M.; Koistinen, K.M.; Laaksonen, R.; Sinisalo, J.; Ekroos, K. Development and validation of a high-throughput LC-MS/MS assay for routine measurement of molecular ceramides. Anal. Bioanal. Chem. 2016, 408, 3475–3483. [Google Scholar] [CrossRef] [PubMed]
| Characteristic | Subgroup | Turku | Charité 1 | Charité 2 | Charité 3 |
|---|---|---|---|---|---|
| Malignant (N) | 197 | 51 | 104 | 147 | |
| Age (years) | 66 (59–72) | 61 (56–68) | 65 (57–70) | 59 (50–67) | |
| Histology | Serous | 156 | 48 | 95 | 147 |
| Mucinous | 13 | 1 | |||
| Endometroid | 16 | 1 | 2 | ||
| Clear-cell | 12 | 1 | |||
| Other/unknown | 2 | 5 | |||
| Stage | I | 31 | 2 | ||
| II | 12 | 1 | 5 | ||
| III | 102 | 51 | 67 | 99 | |
| IV | 50 | 24 | 31 | ||
| NA | 2 | 12 | 10 | ||
| Follow-up time (years) | Death | 2.6 (1.5–3.9) | 3.6 (1.9–6.0) | 1.6 (0.9–2.3) | 3.2 (1.7–4.3) |
| Progression | 1.3 (0.8–2.2) | 2.0 (1.0–3.6) | 1.2 (0.8–1.8) | 1.5 (0.8–2.8) | |
| Benign (N) | 114 | 98 | |||
| Age (years) | 55 (45-68) | 41 (31–55) |
| Endpoint | Study | (Sub)Group | Cer(d18:1/18:0)/PC(O-38:4) | CA-125 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Ev+ | Ev− | HR (95% CI) a | C-Statistic a | HR (95% CI) b | C-Statistic b | Ev+ | Ev− | HR (95% CI) a | C-Statistic a | HR (95% CI) b | C-Statistic b | |||
| Death | Turku | All | 90 | 93 | 1.79 (1.40, 2.29) | 0.707 | 1.72 (1.32, 2.25) | 0.717 | 85 | 92 | 1.02 (0.79, 1.32) | 0.486 | 0.79 (0.60, 1.06) | 0.655 |
| Turku | No residual tumor | 21 | 58 | 2.12 (1.26, 3.55) | 0.648 | 2.16 (1.20, 3.86) | 0.735 | 21 | 58 | 0.86 (0.52, 1.44) | 0.532 | 0.60 (0.35, 1.04) | 0.703 | |
| Charité 1 | Stage III, no residual tumor | 33 | 17 | 1.59 (1.08, 2.35) | 0.592 | 1.67 (1.12, 2.47) | 0.626 | 32 | 16 | 1.21 (0.82, 1.78) | 0.534 | 1.38 (0.91, 2.08) | 0.628 | |
| Charité 2 | All | 28 | 76 | 1.95 (1.31, 2.88) | 0.694 | 1.73 (1.11, 2.71) | 0.706 | 25 | 74 | 1.39 (0.94, 2.05) | 0.421 | 1.26 (0.81, 1.98) | 0.580 | |
| Charité 3 | All | 77 | 66 | 1.40 (1.12, 1.74) | 0.630 | 1.10 (0.88, 1.38) | 0.722 | 76 | 64 | 1.18 (0.92, 1.50) | 0.544 | 1.02 (0.78, 1.32) | 0.706 | |
| Charité 3 | No residual tumor | 42 | 46 | 1.40 (1.03, 1.91) | 0.621 | 1.23 (0.91, 1.68) | 0.672 | 42 | 44 | 1.21 (0.87, 1.68) | 0.550 | 1.17 (0.84, 1.64) | 0.633 | |
| Progression | Turku | All | 122 | 61 | 1.40 (1.14, 1.71) | 0.644 | 1.28 (1.04, 1.57) | 0.700 | 118 | 59 | 1.35 (1.08, 1.69) | 0.585 | 1.01 (0.76, 1.34) | 0.667 |
| Turku | No residual tumor | 34 | 45 | 1.28 (0.88, 1.87) | 0.524 | 1.13 (0.71, 1.78) | 0.747 | 34 | 45 | 1.29 (0.87, 1.92) | 0.418 | 0.83 (0.50, 1.37) | 0.740 | |
| Charité 1 | Stage III, no residual tumor | 34 | 16 | 1.53 (1.02, 2.30) | 0.615 | 1.55 (1.03, 2.32) | 0.629 | 34 | 14 | 1.39 (0.95, 2.05) | 0.592 | 1.51 (1.00, 2.28) | 0.603 | |
| Charité 2 | All | 49 | 55 | 1.27 (0.90, 1.80) | 0.589 | 1.31 (0.90, 1.90) | 0.625 | 46 | 53 | 1.22 (0.88, 1.67) | 0.560 | 1.45 (1.01, 2.09) | 0.592 | |
| Charité 3 | All | 84 | 59 | 1.22 (0.99, 1.51) | 0.563 | 1.09 (0.87, 1.37) | 0.633 | 82 | 58 | 1.15 (0.91, 1.44) | 0.541 | 1.04 (0.82, 1.32) | 0.580 | |
| Charité 3 | No residual tumor | 58 | 30 | 1.32 (1.00, 1.73) | 0.567 | 1.22 (0.92, 1.62) | 0.632 | 57 | 29 | 1.09 (0.84, 1.42) | 0.476 | 1.02 (0.78, 1.34) | 0.544 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salminen, L.; Braicu, E.I.; Lääperi, M.; Jylhä, A.; Oksa, S.; Hietanen, S.; Sehouli, J.; Kulbe, H.; Bois, A.d.; Mahner, S.; et al. A Novel Two-Lipid Signature Is a Strong and Independent Prognostic Factor in Ovarian Cancer. Cancers 2021, 13, 1764. https://doi.org/10.3390/cancers13081764
Salminen L, Braicu EI, Lääperi M, Jylhä A, Oksa S, Hietanen S, Sehouli J, Kulbe H, Bois Ad, Mahner S, et al. A Novel Two-Lipid Signature Is a Strong and Independent Prognostic Factor in Ovarian Cancer. Cancers. 2021; 13(8):1764. https://doi.org/10.3390/cancers13081764
Chicago/Turabian StyleSalminen, Liina, Elena Ioana Braicu, Mitja Lääperi, Antti Jylhä, Sinikka Oksa, Sakari Hietanen, Jalid Sehouli, Hagen Kulbe, Andreas du Bois, Sven Mahner, and et al. 2021. "A Novel Two-Lipid Signature Is a Strong and Independent Prognostic Factor in Ovarian Cancer" Cancers 13, no. 8: 1764. https://doi.org/10.3390/cancers13081764
APA StyleSalminen, L., Braicu, E. I., Lääperi, M., Jylhä, A., Oksa, S., Hietanen, S., Sehouli, J., Kulbe, H., Bois, A. d., Mahner, S., Harter, P., Carpén, O., Huhtinen, K., Hynninen, J., & Hilvo, M. (2021). A Novel Two-Lipid Signature Is a Strong and Independent Prognostic Factor in Ovarian Cancer. Cancers, 13(8), 1764. https://doi.org/10.3390/cancers13081764






