Magnetic Resonance Imaging-Guided Adaptive Radiotherapy for Colorectal Liver Metastases
Abstract
Simple Summary
Abstract
1. Introduction
2. The Evolution of External Beam Radiation Therapy for Liver Tumors
3. Liver SABR Treatment Planning and Delivery Considerations
4. Strategies to Overcome Limitations to SABR
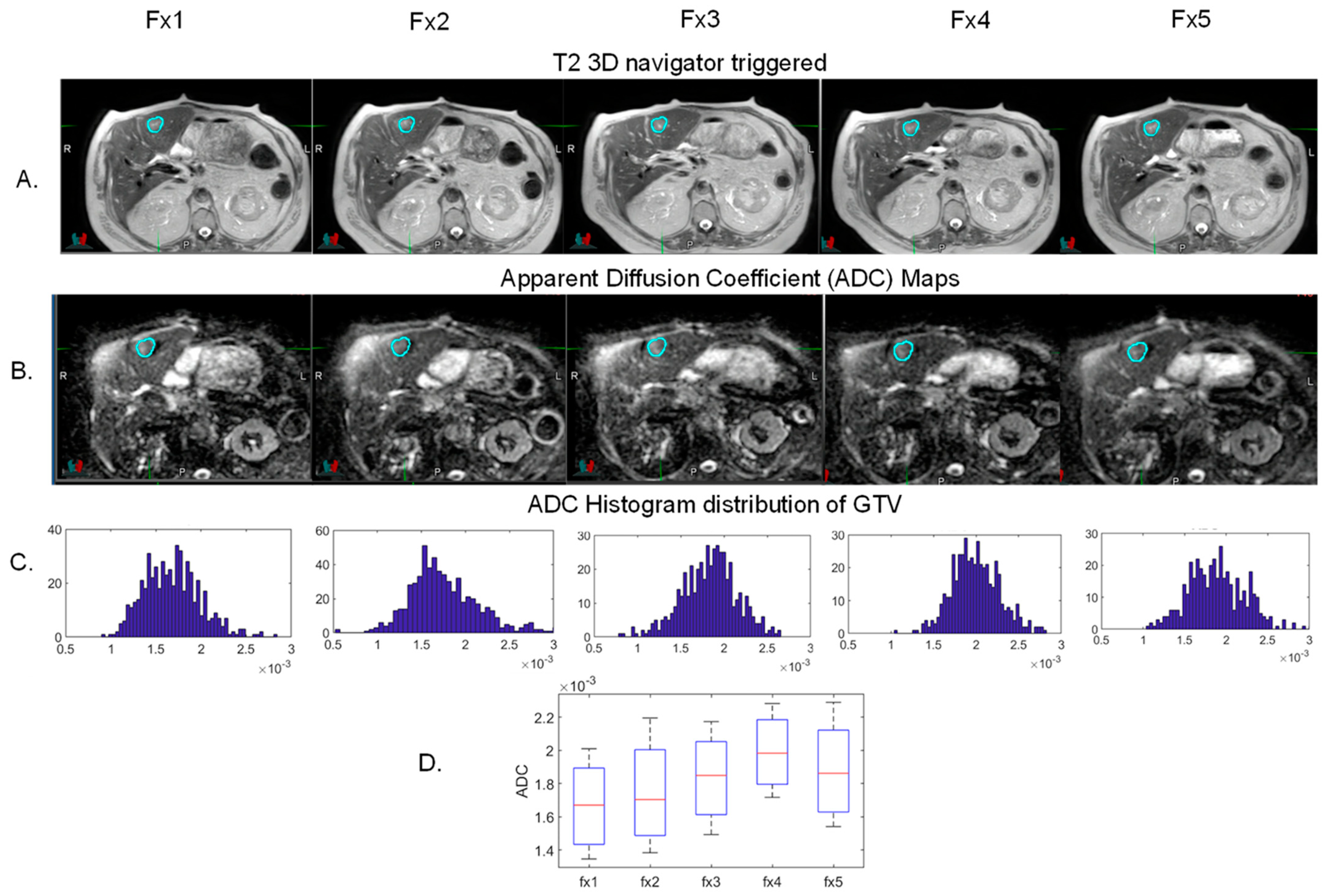
5. MR Guided Adaptive Radiation Therapy
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Aitken, K.L.; Hawkins, M.A. Stereotactic Body Radiotherapy for Liver Metastases. Clin. Oncol. 2015, 27, 307–315. [Google Scholar] [CrossRef] [PubMed]
- Goodman, K.A.; Kavanagh, B.D. Stereotactic Body Radiotherapy for Liver Metastases. Semin. Radiat. Oncol. 2017, 27, 240–246. [Google Scholar] [CrossRef] [PubMed]
- Romesser, P.B.; Neal, B.P.; Crane, C.H. External Beam Radiation Therapy for Liver Metastases. Surg. Oncol. Clin. N. Am. 2021, 30, 159–173. [Google Scholar] [CrossRef] [PubMed]
- Fong, Y.; Fortner, J.; Sun, R.L.; Brennan, M.F.; Blumgart, L.H. Clinical Score for Predicting Recurrence After Hepatic Resection for Metastatic Colorectal Cancer: Analysis of 1001 consecutive cases. Ann. Surg. 1999, 230, 309–318. [Google Scholar] [CrossRef]
- Morris, E.J.; Forman, D.; Thomas, J.D.; Quirke, P.; Taylor, E.F.; Fairley, L.; Cottier, B.; Poston, G. Surgical management and outcomes of colorectal cancer liver metastases. Br. J. Surg. 2010, 97, 1110–1118. [Google Scholar] [CrossRef]
- Nordlinger, B.; Guiguet, M.; Vaillant, J.C.; Balladur, P.; Boudjema, K.; Bachellier, P.; Jaeck, D. Surgical resection of colorectal carcinoma metastases to the liver: A prognostic scoring system to improve case selection, based on 1568 patients. Cancer Interdiscip. Int. J. Am. Cancer Soc. 1996, 77, 1254–1262. [Google Scholar] [CrossRef]
- Okumura, T.; Boku, N.; Hishida, T.; Ohde, Y.; Sakao, Y.; Yoshiya, K.; Higashiyama, M.; Hyodo, I.; Mori, K.; Kondo, H. Surgical Outcome and Prognostic Stratification for Pulmonary Metastasis from Colorectal Cancer. Ann. Thorac. Surg. 2017, 104, 979–987. [Google Scholar] [CrossRef]
- Onaitis, M.W.; Petersen, R.P.; Haney, J.C.; Saltz, L.; Park, B.; Flores, R.; Rizk, N.; Bains, M.S.; Dycoco, J.; D’Amico, T.A.; et al. Prognostic Factors for Recurrence After Pulmonary Resection of Colorectal Cancer Metastases. Ann. Thorac. Surg. 2009, 87, 1684–1688. [Google Scholar] [CrossRef]
- Choti, M.A.; Sitzmann, J.V.; Tiburi, M.F.; Sumetchotimetha, W.; Rangsin, R.; Schulick, R.D.; Lillemoe, K.D.; Yeo, C.J.; Cameron, J.L. Trends in Long-Term Survival Following Liver Resection for Hepatic Colorectal Metastases. Ann. Surg. 2002, 235, 759–766. [Google Scholar] [CrossRef]
- Lehnert, T.; Knaebel, H.P.; Dück, M.; Bülzebruck, H.; Herfarth, C. Sequential hepatic and pulmonary resections for metastatic colorectal cancer. Br. J. Surg. 1999, 86, 241–243. [Google Scholar] [CrossRef]
- Shah, S.A.; Haddad, R.; Al-Sukhni, W.; Kim, R.D.; Greig, P.D.; Grant, D.R.; Taylor, B.R.; Langer, B.; Gallinger, S.; Wei, A.C. Surgical Resection of Hepatic and Pulmonary Metastases from Colorectal Carcinoma. J. Am. Coll. Surg. 2006, 202, 468–475. [Google Scholar] [CrossRef]
- Johnson, B.; Jin, Z.; Truty, M.J.; Smoot, R.L.; Nagorney, D.M.; Kendrick, M.L.; Kipp, B.R.; Grothey, A. Impact of Metastasectomy in the Multimodality Approach forBRAFV600E Metastatic Colorectal Cancer: The Mayo Clinic Experience. Oncologist 2018, 23, 128–134. [Google Scholar] [CrossRef]
- McCormack, P.M.; Burt, M.E.; Bains, M.S.; Martini, N.; Rusch, V.W.; Ginsberg, R.J. Lung Resection for Colorectal Metastases. 10-year results. Arch. Surg. 1992, 127, 1403–1406. [Google Scholar] [CrossRef]
- Tepper, J.E.; O’Connell, M.; Hollis, D.; Niedzwiecki, D.; Cooke, E.; Mayer, R.J.; Intergroup, S. Analysis of Surgical Salvage after Failure of Primary Therapy in Rectal Cancer: Results from Intergroup Study 0114. J. Clin. Oncol. 2003, 21, 3623–3628. [Google Scholar] [CrossRef] [PubMed]
- Kopetz, S.; Chang, G.J.; Overman, M.J.; Eng, C.; Sargent, D.J.; Larson, D.W.; Grothey, A.; Vauthey, J.-N.; Nagorney, D.M.; McWilliams, R.R. Improved Survival in Metastatic Colorectal Cancer Is Associated With Adoption of Hepatic Resection and Improved Chemotherapy. J. Clin. Oncol. 2009, 27, 3677–3683. [Google Scholar] [CrossRef]
- Hewish, M.; Cunningham, D. First-line treatment of advanced colorectal cancer. Lancet 2011, 377, 2060–2062. [Google Scholar] [CrossRef]
- Garcea, G.; Lloyd, T.D.; Aylott, C.; Maddern, G.; Berry, D.P. The emergent role of focal liver ablation techniques in the treatment of primary and secondary liver tumours. Eur. J. Cancer 2003, 39, 2150–2164. [Google Scholar] [CrossRef]
- Ruers, T.; Van Coevorden, F.; Punt, C.J.; Pierie, J.; Borel-Rinkes, I.; Ledermann, J.A.; Poston, G.; Bechstein, W.; Lentz, M.A.; Mauer, M.; et al. Local Treatment of Unresectable Colorectal Liver Metastases: Results of a Randomized Phase II Trial. J. Natl. Cancer Inst. 2017, 109. [Google Scholar] [CrossRef]
- Ruers, T.; Punt, C.; Van Coevorden, F.; Pierie, J.P.E.N.; Borel-Rinkes, I.; Ledermann, J.A.; Poston, G.; Bechstein, W.; Lentz, M.A.; Mauer, M.; et al. Radiofrequency ablation combined with systemic treatment versus systemic treatment alone in patients with non-resectable colorectal liver metastases: A randomized EORTC Intergroup phase II study (EORTC 40004). Ann. Oncol. 2012, 23, 2619–2626. [Google Scholar] [CrossRef]
- Wong, S.L.; Mangu, P.B.; Choti, M.A.; Crocenzi, T.S.; Dodd, G.D., 3rd; Dorfman, G.S.; Eng, C.; Fong, Y.; Giusti, A.F.; Lu, D.; et al. American Society of Clinical Oncology 2009 Clinical Evidence Review on Radiofrequency Ablation of Hepatic Metastases From Colorectal Cancer. J. Clin. Oncol. 2010, 28, 493–508. [Google Scholar] [CrossRef]
- Høyer, M.; Swaminath, A.; Bydder, S.; Lock, M.; Romero, A.M.; Kavanagh, B.; Goodman, K.A.; Okunieff, P.; Dawson, L.A. Radiotherapy for Liver Metastases: A Review of Evidence. Int. J. Radiat. Oncol. Biol. Phys. 2012, 82, 1047–1057. [Google Scholar] [CrossRef] [PubMed]
- Chmura, S.J.; Winter, K.A.; Salama, J.K.; Robinson, C.G.; Pisansky, T.M.; Borges, V.; Al-Hallaq, H.A.; Matuszak, M.M.; Park, S.S.; Gonzalez, V.J.; et al. Phase I Trial of Stereotactic Body Radiation Therapy (SBRT) to Multiple Metastatic Sites: A NRG Oncology Study. Int. J. Radiat. Oncol. 2018, 102, S68–S69. [Google Scholar] [CrossRef]
- Al-Hallaq, H.A.; Chmura, S.; Salama, J.K.; Winter, K.A.; Robinson, C.G.; Pisansky, T.M.; Borges, V.; Lowenstein, J.R.; McNulty, S.; Galvin, J.M.; et al. Rationale of technical requirements for NRG-BR001: The first NCI-sponsored trial of SBRT for the treatment of multiple metastases. Pract. Radiat. Oncol. 2016, 6, e291–e298. [Google Scholar] [CrossRef] [PubMed]
- Al-Hallaq, H.A.; Chmura, S.J.; Salama, J.K.; Lowenstein, J.R.; McNulty, S.; Galvin, J.M.; Followill, D.S.; Robinson, C.G.; Pisansky, T.M.; Winter, K.A.; et al. Benchmark Credentialing Results for NRG-BR001: The First National Cancer Institute-Sponsored Trial of Stereotactic Body Radiation Therapy for Multiple Metastases. Int. J. Radiat. Oncol. Biol. Phys. 2017, 97, 155–163. [Google Scholar] [CrossRef]
- Palma, D.A.; Olson, R.; Harrow, S.; Gaede, S.; Louie, A.V.; Haasbeek, C.; Mulroy, L.; Lock, M.; Rodrigues, G.B.; Yaremko, B.P.; et al. Stereotactic ablative radiotherapy versus standard of care palliative treatment in patients with oligometastatic cancers (SABR-COMET): A randomised, phase 2, open-label trial. Lancet 2019, 393, 2051–2058. [Google Scholar] [CrossRef]
- Palma, D.A.; Olson, R.; Harrow, S.; Gaede, S.; Louie, A.V.; Haasbeek, C.; Mulroy, L.; Lock, M.; Rodrigues, G.B.; Yaremko, B.P.; et al. Stereotactic Ablative Radiotherapy for the Comprehensive Treatment of Oligometastatic Cancers: Long-Term Results of the SABR-COMET Phase II Randomized Trial. J. Clin. Oncol. 2020, 38, 2830–2838. [Google Scholar] [CrossRef]
- Lewis, S.L.; Porceddu, S.; Nakamura, N.; Palma, D.A.; Lo, S.S.; Hoskin, P.; Moghanaki, D.; Chmura, S.J.; Salama, J.K. Definitive Stereotactic Body Radiotherapy (SBRT) for Extracranial Oligometastases: An International Survey of >1000 Radiation Oncologists. Am. J. Clin. Oncol. 2017, 40, 418–422. [Google Scholar] [CrossRef]
- Bydder, S.; Spry, N.A.; Christie, D.R.; Roos, D.; Burmeister, B.H.; Krawitz, H.; Davis, S.; Joseph, D.J.; Poulsen, M.; Berry, M. A prospective trial of short-fractionation radiotherapy for the palliation of liver metastases. Australas. Radiol. 2003, 47, 284–288. [Google Scholar] [CrossRef]
- Borgelt, B.B.; Gelber, R.; Brady, L.W.; Griffin, T.; Hendrickson, F.R. The palliation of hepatic metastases: Results of the radiation therapy oncology group pilot study. Int. J. Radiat. Oncol. Biol. Phys. 1981, 7, 587–591. [Google Scholar] [CrossRef]
- Soliman, H.; Ringash, J.; Jiang, H.; Singh, K.; Kim, J.; Dinniwell, R.; Brade, A.; Wong, R.; Brierley, J.; Cummings, B.; et al. Phase II Trial of Palliative Radiotherapy for Hepatocellular Carcinoma and Liver Metastases. J. Clin. Oncol. 2013, 31, 3980–3986. [Google Scholar] [CrossRef]
- Toesca, D.A.S.; Ibragimov, B.; Koong, A.J.; Xing, L.; Koong, A.C.; Chang, D.T. Strategies for prediction and mitigation of radiation-induced liver toxicity. J. Radiat. Res. 2018, 59, i40–i49. [Google Scholar] [CrossRef] [PubMed]
- Pan, C.C.; Kavanagh, B.D.; Dawson, L.A.; Li, X.A.; Das, S.K.; Miften, M.; Haken, R.K.T. Radiation-Associated Liver Injury. Int. J. Radiat. Oncol. Biol. Phys. 2010, 76, S94–S100. [Google Scholar] [CrossRef] [PubMed]
- Lawrence, T.S.; Robertson, J.M.; Anscher, M.S.; Jirtle, R.L.; Ensminger, W.D.; Fajardo, L.F. Hepatic toxicity resulting from cancer treatment. Int. J. Radiat. Oncol. 1995, 31, 1237–1248. [Google Scholar] [CrossRef]
- Cheng, J.C.; Wu, J.K.; Huang, C.M.; Huang, D.Y.; Cheng, S.H.; Lin, Y.-M.; Jian, J.J.; Yang, P.-S.; Chuang, V.P.; Huang, A.T. Radiation-induced liver disease after radiotherapy for hepatocellular carcinoma: Clinical manifestation and dosimetric description. Radiother. Oncol. 2002, 63, 41–45. [Google Scholar] [CrossRef]
- Cheng, J.C.; Wu, J.K.; Lee, P.C.T.; Liu, H.S.; Jian, J.J.; Lin, Y.M.; Sung, J.L.; Jan, G.J. Biologic susceptibility of hepatocellular carcinoma patients treated with radiotherapy to radiation-induced liver disease. Int. J. Radiat. Oncol. Biol. Phys. 2004, 60, 1502–1509. [Google Scholar] [CrossRef]
- Liang, S.-X.; Zhu, X.-D.; Xu, Z.-Y.; Zhu, J.; Zhao, J.-D.; Lu, H.-J.; Yang, Y.-L.; Chen, L.; Wang, A.-Y.; Fu, X.-L.; et al. Radiation-induced liver disease in three-dimensional conformal radiation therapy for primary liver carcinoma: The risk factors and hepatic radiation tolerance. Int. J. Radiat. Oncol. Biol. Phys. 2006, 65, 426–434. [Google Scholar] [CrossRef]
- Lawrence, T.S.; Haken, R.K.T.; Kessler, M.L.; Robertson, J.M.; Lyman, J.T.; Lavigne, M.L.; Brown, M.B.; DuRoss, D.J.; Andrews, J.C.; Ensminger, W.D.; et al. The use of 3-D dose volume analysis to predict radiation hepatitis. Int. J. Radiat. Oncol. Biol. Phys. 1992, 23, 781–788. [Google Scholar] [CrossRef]
- Dawson, L.A.; McGinn, C.J.; Normolle, D.; Haken, R.K.T.; Walker, S.; Ensminger, W.; Lawrence, T.S. Escalated Focal Liver Radiation and Concurrent Hepatic Artery Fluorodeoxyuridine for Unresectable Intrahepatic Malignancies. J. Clin. Oncol. 2000, 18, 2210–2218. [Google Scholar] [CrossRef]
- Dawson, L.A.; Normolle, D.; Balter, J.M.; McGinn, C.J.; Lawrence, T.S.; Haken, R.K.T. Analysis of radiation-induced liver disease using the Lyman NTCP model. Int. J. Radiat. Oncol. Biol. Phys. 2002, 53, 810–821. [Google Scholar] [CrossRef]
- Ben-Josef, E.; Normolle, D.; Ensminger, W.D.; Walker, S.; Tatro, D.; Haken, R.K.T.; Knol, J.; Dawson, L.A.; Pan, C.; Lawrence, T.S. Phase II Trial of High-Dose Conformal Radiation Therapy with Concurrent Hepatic Artery Floxuridine for Unresectable Intrahepatic Malignancies. J. Clin. Oncol. 2005, 23, 8739–8747. [Google Scholar] [CrossRef]
- Blomgren, H.; Lax, I.; Näslund, I.; Svanström, R. Stereotactic High Dose Fraction Radiation Therapy of Extracranial Tumors Using an Accelerator: Clinical experience of the first thirty-one patients. Acta Oncol. 1995, 34, 861–870. [Google Scholar] [CrossRef] [PubMed]
- Herfarth, K.K.; Debus, J.; Lohr, F.; Bahner, M.L.; Rhein, B.; Fritz, P.; Höss, A.; Schlegel, W.; Wannenmacher, M.F. Stereotactic Single-Dose Radiation Therapy of Liver Tumors: Results of a Phase I/II Trial. J. Clin. Oncol. 2001, 19, 164–170. [Google Scholar] [CrossRef] [PubMed]
- Romero, A.M.; Wunderink, W.; Hussain, S.M.; De Pooter, J.A.; Heijmen, B.J.; Nowak, P.C.; Nuyttens, J.J.; Brandwijk, R.P.; Verhoef, C.; Ijzermans, J.N.; et al. Stereotactic body radiation therapy for primary and metastatic liver tumors: A single institution phase i–ii study. Acta Oncol. 2006, 45, 831–837. [Google Scholar] [CrossRef] [PubMed]
- Rusthoven, K.E.; Kavanagh, B.D.; Cardenes, H.; Stieber, V.W.; Burri, S.H.; Feigenberg, S.J.; Chidel, M.A.; Pugh, T.J.; Franklin, W.; Kane, M.; et al. Multi-Institutional Phase I/II Trial of Stereotactic Body Radiation Therapy for Liver Metastases. J. Clin. Oncol. 2009, 27, 1572–1578. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.T.; Kim, J.J.; Dinniwell, R.; Brierley, J.; Lockwood, G.; Wong, R.; Cummings, B.; Ringash, J.; Tse, R.V.; Knox, J.J.; et al. Phase I Study of Individualized Stereotactic Body Radiotherapy of Liver Metastases. J. Clin. Oncol. 2009, 27, 1585–1591. [Google Scholar] [CrossRef]
- Rule, W.; Timmerman, R.; Tong, L.; Abdulrahman, R.; Meyer, J.; Boike, T.; Schwarz, R.E.; Weatherall, P.; Chinsoo Cho, L. Phase I Dose-Escalation Study of Stereotactic Body Radiotherapy in Patients with Hepatic Metastases. Ann. Surg. Oncol. 2010, 18, 1081–1087. [Google Scholar] [CrossRef]
- Comito, T.; Cozzi, L.; Clerici, E.; Campisi, M.C.; Liardo, R.L.; Navarria, P.; Ascolese, A.; Tozzi, A.; Iftode, C.; De Rose, F.; et al. Stereotactic Ablative Radiotherapy (SABR) in inoperable oligometastatic disease from colorectal cancer: A safe and effective approach. BMC Cancer 2014, 14, 619. [Google Scholar] [CrossRef]
- Scorsetti, M.; Comito, T.; Tozzi, A.; Navarria, P.; Fogliata, A.; Clerici, E.; Mancosu, P.; Reggiori, G.; Rimassa, L.; Torzilli, G.; et al. Final results of a phase II trial for stereotactic body radiation therapy for patients with inoperable liver metastases from colorectal cancer. J. Cancer Res. Clin. Oncol. 2015, 141, 543–553. [Google Scholar] [CrossRef]
- Goodman, K.A.; Wiegner, E.A.; Maturen, K.E.; Zhang, Z.; Mo, Q.; Yang, G.; Gibbs, I.C.; Fisher, G.A.; Koong, A.C. Dose-Escalation Study of Single-Fraction Stereotactic Body Radiotherapy for Liver Malignancies. Int. J. Radiat. Oncol. Biol. Phys. 2010, 78, 486–493. [Google Scholar] [CrossRef]
- Meyer, J.J.; Foster, R.D.; Lev-Cohain, N.; Yokoo, T.; Dong, Y.; Schwarz, R.E.; Rule, W.; Tian, J.; Xie, Y.; Hannan, R.; et al. A Phase I Dose-Escalation Trial of Single-Fraction Stereotactic Radiation Therapy for Liver Metastases. Ann. Surg. Oncol. 2016, 23, 218–224. [Google Scholar] [CrossRef]
- Hong, T.S.; Wo, J.Y.; Borger, D.R.; Yeap, B.Y.; McDonnell, E.I.; Willers, H.; Blaszkowsky, L.S.; Kwak, E.L.; Allen, J.N.; Clark, J.W.; et al. Phase II Study of Proton-Based Stereotactic Body Radiation Therapy for Liver Metastases: Importance of Tumor Genotype. J. Natl. Cancer Inst. 2017, 109. [Google Scholar] [CrossRef]
- Scorsetti, M.; Comito, T.; Clerici, E.; Franzese, C.; Tozzi, A.; Iftode, C.; Di Brina, L.; Navarria, P.; Mancosu, P.; Reggiori, G.; et al. Phase II trial on SBRT for unresectable liver metastases: Long-term outcome and prognostic factors of survival after 5 years of follow-up. Radiat. Oncol. 2018, 13, 234. [Google Scholar] [CrossRef]
- Kang, J.I.; Sufficool, D.C.; Hsueh, C.-T.; Wroe, A.J.; Patyal, B.; Reeves, M.E.; Slater, J.D.; Yang, G.Y. A phase I trial of Proton stereotactic body radiation therapy for liver metastases. J. Gastrointest. Oncol. 2019, 10, 112–117. [Google Scholar] [CrossRef]
- Dawson, L.A.; Winter, K.A.; Katz, A.W.; Schell, M.C.; Brierley, J.; Chen, Y.; Kopek, N.; Crane, C.H.; Willett, C.G. NRG Oncology/RTOG 0438: A Phase 1 Trial of Highly Conformal Radiation Therapy for Liver Metastases. Pract. Radiat. Oncol. 2019, 9, e386–e393. [Google Scholar] [CrossRef]
- Schefter, T.E.; Kavanagh, B.D.; Timmerman, R.D.; Cardenes, H.R.; Baron, A.; Gaspar, L.E. A phase I trial of stereotactic body radiation therapy (SBRT) for liver metastases. Int. J. Radiat. Oncol. Biol. Phys. 2005, 62, 1371–1378. [Google Scholar] [CrossRef] [PubMed]
- Klement, R.J. Radiobiological parameters of liver and lung metastases derived from tumor control data of 3719 metastases. Radiother. Oncol. 2017, 123, 218–226. [Google Scholar] [CrossRef] [PubMed]
- Mahadevan, A.; Blanck, O.; Lanciano, R.; Peddada, A.; Sundararaman, S.; D’Ambrosio, D.; Sharma, S.; Perry, D.; Kolker, J.; Davis, J. Stereotactic Body Radiotherapy (SBRT) for liver metastasis—Clinical outcomes from the international multi-institutional RSSearch® Patient Registry. Radiat. Oncol. 2018, 13, 26. [Google Scholar] [CrossRef] [PubMed]
- Wurm, R.E.; Gum, F.; Erbel, S.; Schlenger, L.; Scheffler, D.; Agaoglu, D.; Schild, R.; Gebauer, B.; Rogalla, P.; Plotkin, M.; et al. Image guided respiratory gated hypofractionated Stereotactic Body Radiation Therapy (H-SBRT) for liver and lung tumors: Initial experience. Acta Oncol. 2006, 45, 881–889. [Google Scholar] [CrossRef]
- Kirilova, A.; Lockwood, G.; Choi, P.; Bana, N.; Haider, M.A.; Brock, K.K.; Eccles, C.; Dawson, L.A. Three-Dimensional Motion of Liver Tumors Using Cine-Magnetic Resonance Imaging. Int. J. Radiat. Oncol. Biol. Phys. 2008, 71, 1189–1195. [Google Scholar] [CrossRef]
- Worm, E.S.; Høyer, M.; Fledelius, W.; Poulsen, P.R. Three-dimensional, Time-Resolved, Intrafraction Motion Monitoring Throughout Stereotactic Liver Radiation Therapy on a Conventional Linear Accelerator. Int. J. Radiat. Oncol. Biol. Phys. 2013, 86, 190–197. [Google Scholar] [CrossRef]
- Vogel, L.; Sihono, D.S.K.; Weiss, C.; Lohr, F.; Stieler, F.; Wertz, H.; Von Swietochowski, S.; Simeonova-Chergou, A.; Wenz, F.; Blessing, M.; et al. Intra-breath-hold residual motion of image-guided DIBH liver-SBRT: An estimation by ultrasound-based monitoring correlated with diaphragm position in CBCT. Radiother. Oncol. 2018, 129, 441–448. [Google Scholar] [CrossRef] [PubMed]
- Briere, T.M.; Beddar, S.; Balter, P.; Murthy, R.; Gupta, S.; Nelson, C.; Starkschall, G.; Gillin, M.T.; Krishnan, S. Respiratory gating with EPID-based verification: The MDACC experience. Phys. Med. Biol. 2009, 54, 3379–3391. [Google Scholar] [CrossRef] [PubMed]
- Heinzerling, J.H.; Anderson, J.F.; Papiez, L.; Boike, T.; Chien, S.; Zhang, G.; Abdulrahman, R.; Timmerman, R. Four-Dimensional Computed Tomography Scan Analysis of Tumor and Organ Motion at Varying Levels of Abdominal Compression During Stereotactic Treatment of Lung and Liver. Int. J. Radiat. Oncol. Biol. Phys. 2008, 70, 1571–1578. [Google Scholar] [CrossRef]
- Case, R.B.; Moseley, D.J.; Sonke, J.J.; Eccles, C.L.; Dinniwell, R.E.; Kim, J.; Bezjak, A.; Milosevic, M.; Brock, K.K.; Dawson, L.A. Interfraction and Intrafraction Changes in Amplitude of Breathing Motion in Stereotactic Liver Radiotherapy. Int. J. Radiat. Oncol. Biol. Phys. 2010, 77, 918–925. [Google Scholar] [CrossRef] [PubMed]
- Case, R.B.; Sonke, J.-J.; Moseley, D.J.; Kim, J.; Brock, K.K.; Dawson, L.A. Inter- and Intrafraction Variability in Liver Position in Non-Breath-Hold Stereotactic Body Radiotherapy. Int. J. Radiat. Oncol. Biol. Phys. 2009, 75, 302–308. [Google Scholar] [CrossRef]
- Yang, J.; Cai, J.; Wang, H.; Chang, Z.; Czito, B.G.; Bashir, M.R.; Palta, M.; Yin, F.-F. Is Diaphragm Motion a Good Surrogate for Liver Tumor Motion? Int. J. Radiat. Oncol. Biol. Phys. 2014, 90, 952–958. [Google Scholar] [CrossRef]
- Wunderink, W.; Romero, A.M.; Seppenwoolde, Y.; De Boer, H.; Levendag, P.; Heijmen, B. Potentials and Limitations of Guiding Liver Stereotactic Body Radiation Therapy Set-Up on Liver-Implanted Fiducial Markers. Int. J. Radiat. Oncol. Biol. Phys. 2010, 77, 1573–1583. [Google Scholar] [CrossRef]
- Seppenwoolde, Y.; Wunderink, W.; Wunderink-van Veen, S.R.; Storchi, P.; Romero, A.M.; Heijmen, B.J. Treatment precision of image-guided liver SBRT using implanted fiducial markers depends on marker-tumour distance. Phys. Med. Biol. 2011, 56, 5445–5468. [Google Scholar] [CrossRef]
- Crane, C.H.; Koay, E.J. Solutions that enable ablative radiotherapy for large liver tumors: Fractionated dose painting, simultaneous integrated protection, motion management, and computed tomography image guidance. Cancer 2016, 122, 1974–1986. [Google Scholar] [CrossRef]
- Bujold, A.; Massey, C.A.; Kim, J.J.; Brierley, J.; Cho, C.; Wong, R.K.; Dinniwell, R.E.; Kassam, Z.; Ringash, J.; Cummings, B.; et al. Sequential Phase I and II Trials of Stereotactic Body Radiotherapy for Locally Advanced Hepatocellular Carcinoma. J. Clin. Oncol. 2013, 31, 1631–1639. [Google Scholar] [CrossRef]
- Mondlane, G.; Gubanski, M.; Lind, P.A.; Henry, T.; Ureba, A.; Siegbahn, A. Dosimetric Comparison of Plans for Photon- or Proton-Beam Based Radiosurgery of Liver Metastases. Int. J. Part. Ther. 2016, 3, 277–284. [Google Scholar] [CrossRef] [PubMed]
- Colbert, L.E.; Cloyd, J.M.; Koay, E.J.; Crane, C.H.; Vauthey, J.-N. Proton beam radiation as salvage therapy for bilateral colorectal liver metastases not amenable to second-stage hepatectomy. Surgery 2017, 161, 1543–1548. [Google Scholar] [CrossRef] [PubMed]
- Sanford, N.N.; Pursley, J.; Noe, B.; Yeap, B.Y.; Goyal, L.; Clark, J.W.; Allen, J.N.; Blaszkowsky, L.S.; Ryan, D.P.; Ferrone, C.R.; et al. Protons versus Photons for Unresectable Hepatocellular Carcinoma: Liver Decompensation and Overall Survival. Int. J. Radiat. Oncol. Biol. Phys. 2019, 105, 64–72. [Google Scholar] [CrossRef] [PubMed]
- Hall, W.A.; Paulson, E.S.; van der Heide, U.A.; Fuller, C.D.; Raaymakers, B.W.; Lagendijk, J.J.W.; Li, X.A.; Jaffray, D.A.; Dawson, L.A.; Erickson, B.; et al. The transformation of radiation oncology using real-time magnetic resonance guidance: A review. Eur. J. Cancer 2019, 122, 42–52. [Google Scholar] [CrossRef]
- Owrangi, A.M.; Greer, P.B.; Glide-Hurst, C.K. MRI-only treatment planning: Benefits and challenges. Phys. Med. Biol. 2018, 63, 05TR01. [Google Scholar] [CrossRef]
- Liu, L.; Johansson, A.; Cao, Y.; Dow, J.; Lawrence, T.S.; Balter, J.M. Abdominal synthetic CT generation from MR Dixon images using a U-net trained with ‘semi-synthetic’ CT data. Phys. Med. Biol. 2020, 65, 125001. [Google Scholar] [CrossRef]
- Sahin, B.; Mustafayev, T.Z.; Gungor, G.; Aydin, G.; Yapici, B.; Atalar, B.; Ozyar, E. First 500 Fractions Delivered with a Magnetic Resonance-guided Radiotherapy System: Initial Experience. Cureus 2019, 11, e6457. [Google Scholar] [CrossRef]
- Winkel, D.; Bol, G.H.; Kroon, P.S.; Van Asselen, B.; Hackett, S.S.; Werensteijn-Honingh, A.M.; Intven, M.P.W.; Eppinga, W.S.C.; Tijssen, R.H.N.; Kerkmeijer, L.G.W.; et al. Adaptive radiotherapy: The Elekta Unity MR-linac concept. Clin. Transl. Radiat. Oncol. 2019, 18, 54–59. [Google Scholar] [CrossRef]
- Rosenberg, S.A.; Henke, L.E.; Shaverdian, N.; Mittauer, K.; Wojcieszynski, A.P.; Hullett, C.R.; Kamrava, M.; Lamb, J.; Cao, M.; Green, O.L.; et al. A Multi-Institutional Experience of MR-Guided Liver Stereotactic Body Radiation Therapy. Adv. Radiat. Oncol. 2019, 4, 142–149. [Google Scholar] [CrossRef]
- Hal, W.A.; Straza, M.W.; Chen, X.; Mickevicius, N.; Erickson, B.; Schultz, C.; Awan, M.; Ahunbay, E.; Li, X.A.; Paulson, E.S. Initial clinical experience of Stereotactic Body Radiation Therapy (SBRT) for liver metastases, primary liver malignancy, and pancreatic cancer with 4D-MRI based online adaptation and real-time MRI monitoring using a 1.5 Tesla MR-Linac. PLoS ONE 2020, 15, e0236570. [Google Scholar] [CrossRef]
- Fast, M.; van de Schoot, A.; van de Lindt, T.; Carbaat, C.; van der Heide, U.; Sonke, J.-J. Tumor Trailing for Liver SBRT on the MR-Linac. Int. J. Radiat. Oncol. Biol. Phys. 2019, 103, 468–478. [Google Scholar] [CrossRef]
- Feldman, A.M.; Modh, A.; Glide-Hurst, C.; Chetty, I.J.; Movsas, B. Real-time Magnetic Resonance-guided Liver Stereotactic Body Radiation Therapy: An Institutional Report Using a Magnetic Resonance-Linac System. Cureus 2019, 11, e5774. [Google Scholar] [CrossRef]
- Güngör, G.; Serbez, I.; Temur, B.; Gür, G.; Kayalılar, N.; Mustafayev, T.Z.; Korkmaz, L.; Aydın, G.; Yapıcı, B.; Atalar, B.; et al. Time Analysis of Online Adaptive Magnetic Resonance-Guided Radiation Therapy Workflow According to Anatomical Sites. Pract. Radiat. Oncol. 2021, 11, e11–e21. [Google Scholar] [CrossRef]
- Chetty, I.J.; Rosu-Bubulac, M. Deformable Registration for Dose Accumulation. Semin. Radiat. Oncol. 2019, 29, 198–208. [Google Scholar] [CrossRef]
- Paulson, E.S.; Ahunbay, E.; Chen, X.; Mickevicius, N.J.; Chen, G.-P.; Schultz, C.; Erickson, B.; Straza, M.; Hall, W.A.; Li, X.A. 4D-MRI driven MR-guided online adaptive radiotherapy for abdominal stereotactic body radiation therapy on a high field MR-Linac: Implementation and initial clinical experience. Clin. Transl. Radiat. Oncol. 2020, 23, 72–79. [Google Scholar] [CrossRef] [PubMed]
- Bruijnen, T.; Stemkens, B.; Lagendijk, J.J.W.; van den Berg, C.A.T.; Tijssen, R.H.N. Multiresolution radial MRI to reduce IDLE time in pre-beam imaging on an MR-Linac (MR-RIDDLE). Phys. Med. Biol. 2019, 64, 055011. [Google Scholar] [CrossRef] [PubMed]
- Fiz, F.; Viganò, L.; Gennaro, N.; Costa, G.; La Bella, L.; Boichuk, A.; Cavinato, L.; Sollini, M.; Politi, L.S.; Chiti, A.; et al. Radiomics of Liver Metastases: A Systematic Review. Cancers 2020, 12, 2881. [Google Scholar] [CrossRef] [PubMed]
- Shaverdian, N.; Yang, Y.; Hu, P.; Hart, S.; Sheng, K.; Lamb, J.; Cao, M.; Agazaryan, N.; Thomas, D.; Steinberg, M.; et al. Feasibility evaluation of diffusion-weighted imaging using an integrated MRI-radiotherapy system for response assessment to neoadjuvant therapy in rectal cancer. Br. J. Radiol. 2017, 90, 20160739. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Cao, M.; Sheng, K.; Gao, Y.; Chen, A.; Kamrava, M.; Lee, P.; Agazaryan, N.; Lamb, J.; Thomas, D.; et al. Longitudinal diffusion MRI for treatment response assessment: Preliminary experience using an MRI-guided tri-cobalt 60 radiotherapy system. Med. Phys. 2016, 43, 1369–1373. [Google Scholar] [CrossRef] [PubMed]
- Kooreman, E.S.; van Houdt, P.J.; Nowee, M.E.; van Pelt, V.W.J.; Tijssen, R.H.N.; Paulson, E.S.; Gurney-Champion, O.J.; Wang, J.; Koetsveld, F.; van Buuren, L.D.; et al. Feasibility and accuracy of quantitative imaging on a 1.5 T MR-linear accelerator. Radiother. Oncol. 2019, 133, 156–162. [Google Scholar] [CrossRef] [PubMed]
- Cusumano, D.; Boldrini, L.; Yadav, P.; Casà, C.; Lee, S.L.; Romano, A.; Piras, A.; Chiloiro, G.; Placidi, L.; Catucci, F.; et al. Delta Radiomics Analysis for Local Control Prediction in Pancreatic Cancer Patients Treated Using Magnetic Resonance Guided Radiotherapy. Diagnostics 2021, 11, 72. [Google Scholar] [CrossRef] [PubMed]
- Vandecaveye, V.; Dirix, P.; De Keyzer, F.; De Beeck, K.O.; Vander Poorten, V.; Roebben, I.; Nuyts, S.; Hermans, R. Predictive value of diffusion-weighted magnetic resonance imaging during chemoradiotherapy for head and neck squamous cell carcinoma. Eur. Radiol. 2010, 20, 1703–1714. [Google Scholar] [CrossRef] [PubMed]
- Cai, G.; Xu, Y.; Zhu, J.; Gu, W.-L.; Zhang, S.; Ma, X.-J.; Cai, S.-J.; Zhang, Z. Diffusion-weighted magnetic resonance imaging for predicting the response of rectal cancer to neoadjuvant concurrent chemoradiation. World J. Gastroenterol. 2013, 19, 5520–5527. [Google Scholar] [CrossRef] [PubMed]
- Van Rossum, P.S.; Van Lier, A.L.; Van Vulpen, M.; Reerink, O.; Lagendijk, J.J.; Lin, S.H.; Van Hillegersberg, R.; Ruurda, J.P.; Meijer, G.J.; Lips, I.M. Diffusion-weighted magnetic resonance imaging for the prediction of pathologic response to neoadjuvant chemoradiotherapy in esophageal cancer. Radiother. Oncol. 2015, 115, 163–170. [Google Scholar] [CrossRef] [PubMed]

| Study | Study Type | Patient # | Lesion # | Primary Histology | Dose (Gy)/ Fraction Number | BED (a/b = 10) | Toxicity | Local Tumor Control | Overall Survival |
|---|---|---|---|---|---|---|---|---|---|
| Herfarth et al. [42] | Phase I/II | 37 | 60 | Mixed | 14-26/1 | 33–94 | 71% @ 1 year | 72% @ 1 year | |
| Mendez–Romero et al. [43] | Phase I/II | 17 | 39 | Mixed | 37.5/3 | 84 | 12% G3 Liver | 100% @ 1 year 86% @ 2 years | 85% @ 1 year 62% @ 2 years |
| Rusthoven et al. [44] | Phase I/II | 47 | 63 | Mixed | 36–60/3 | 79–180 | 1.5% G3 (dermatitis) | 95% @ 1 year 92% @ 2 years | 30% @ 2 years |
| Lee et al. [45] | Dose escalation, phase I | 68 | 142 | Mixed | 28–60/6 | 41–120 | 10% G3+ | 71% @ 1 year | 47% @ 1.5 years |
| Rule et al. [46] | Dose escalation, phase I | 27 | 37 | Mixed | 30/3–50–60/5 | 60–132 | 4% G3 Liver | 100%, 89%, 56% * @ 2 years | 50%, 67%, 56% * @ 2 years |
| Comito et al. [47] | Observational | 42 | 52 | Colorectal | 75/3 | 263 | 60% G2, 0%G3 | 95% @ 1 year 85% @ 2 years | 85% @ 1 year 65% @ 2 years |
| Scorsetti et al. [48] | Phase II | 42 | 52 | Colorectal | 75/3 | 263 | 25% G2 Liver, 0% G3 | 95% @ 1 year 91% @ 2 years 85% @ 3 years | 65% @ 2 years |
| Goodman et al. [49] | Dose escalation, phase I | 26 | 40 | Mixed | 18–30/1 | 50–120 | 8% GI bleeding | 77% @ 1 year | 50% @ 2 years |
| Meyer et al. [50] | Dose escalation, phase I | 14 | 17 | Mixed | 35–40/1 | 158–200 | 6% G2 | 100% @ 2.5 years | 78% @ 2 years |
| Hong et al. [51] | Phase II | 89 | 143 | Mixed | 30–50/5 | 48–100 | No G3+ | 72% @ 1 year 61% @ 3 years | 66% @ 1 year 21% @ 3 years |
| Scorsetti et al. [52] | Phase II | 61 | 76 | Mixed | 75/3 | 263 | 2% G3 chest wall pain | 94% @ 1 year 78% @ 3 years 78% @ 5 years | 85% @ 1 year 31% @ 3 years 18% @ 5 years |
| Kang et al. [53] | Phase I | 9 | 14 | Mixed | 36–60/3 | 79–180 | No G3+ | NR | NR |
| Dawson et al. [54] | Dose escalation, phase I, multicenter | 26 | 37 | Mixed | 35–50/10 | 47–75 | 7.7% G3 GI | NR | NR |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Romesser, P.B.; Tyagi, N.; Crane, C.H. Magnetic Resonance Imaging-Guided Adaptive Radiotherapy for Colorectal Liver Metastases. Cancers 2021, 13, 1636. https://doi.org/10.3390/cancers13071636
Romesser PB, Tyagi N, Crane CH. Magnetic Resonance Imaging-Guided Adaptive Radiotherapy for Colorectal Liver Metastases. Cancers. 2021; 13(7):1636. https://doi.org/10.3390/cancers13071636
Chicago/Turabian StyleRomesser, Paul B., Neelam Tyagi, and Christopher H. Crane. 2021. "Magnetic Resonance Imaging-Guided Adaptive Radiotherapy for Colorectal Liver Metastases" Cancers 13, no. 7: 1636. https://doi.org/10.3390/cancers13071636
APA StyleRomesser, P. B., Tyagi, N., & Crane, C. H. (2021). Magnetic Resonance Imaging-Guided Adaptive Radiotherapy for Colorectal Liver Metastases. Cancers, 13(7), 1636. https://doi.org/10.3390/cancers13071636






