Comparison of the Diagnostic Efficiency of Radial- and Convex-Arrayed Echoendoscopes for Indirect Findings of Pancreatic Cancer: A Retrospective Comparative Study Using Propensity Score Method
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients and Study Design
2.2. Main Outcome Measurements
2.3. Data Collection
2.4. Definition of Indirect Findings
2.5. Endoscopic Procedure and Endoscopists
2.6. Resolution of Endoscopes
2.7. Image Evaluation
2.8. Statistical Analysis
3. Results
3.1. Baseline Characteristics of Patients
3.2. Main Outcome Measurements
3.2.1. The Detection Rate for Indirect Findings
3.2.2. Final Diagnosis
3.2.3. Evaluation Using IPTW
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Warshaw, A.L.; Fernández-del Castillo, C. Pancreatic carcinoma. N. Engl. J. Med. 1992, 326, 455–465. [Google Scholar] [CrossRef]
- Rosewicz, S.; Wiedenmann, B. Pancreatic carcinoma. Lancet 1997, 349, 485–489. [Google Scholar] [CrossRef]
- Egawa, S.; Toma, H.; Ohigashi, H.; Okusaka, T.; Nakao, A.; Hatori, T.; Maguchi, H.; Yanagisawa, A.; Tanaka, M. Japan Pancreatic Cancer Registry; 30th year anniversary: Japan Pancreas Society. Pancreas 2012, 41, 985–992. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, H.; Kitano, M.; Suetomi, Y.; Maekawa, K.; Takeyama, Y.; Kudo, M. Utility of contrast-enhanced endoscopic ultrasonography for diagnosis of small pancreatic carcinomas. Ultrasound Med. Biol. 2008, 34, 525–532. [Google Scholar] [CrossRef]
- Canto, M.I.; Hruban, R.H.; Fishman, E.K.; Kamel, I.R.; Schulick, R.; Zhang, Z.; Topazian, M.; Takahashi, N.; Fletcher, J.; Petersen, G.; et al. Frequent detection of pancreatic lesions in asymptomatic high-risk individuals. Gastroenterology 2012, 142, 796–804. [Google Scholar] [CrossRef] [PubMed]
- Kamata, K.; Kitano, M.; Kudo, M.; Sakamoto, H.; Kadosaka, K.; Miyata, T.; Imai, H.; Maekawa, K.; Chikugo, T.; Kumano, M.; et al. Value of EUS in early detection of pancreatic ductal adenocarcinomas in patients with intraductal papillary mucinous neoplasms. Endoscopy 2014, 46, 22–29. [Google Scholar] [CrossRef] [PubMed]
- Sakamoto, H.; Kitano, M.; Takeyama, Y. Early diagnosis of pancreatic cancer: Approach to the diagnosis of very small pancreatic cancer with emphasis of US, MDCT and EUS. Tan. Sui. 2009, 30, 335–341. [Google Scholar]
- Byrne, M.F.; Jowell, P.S. Gastrointestinal imaging: Endoscopic ultrasound. Gastroenterology 2002, 122, 1631–1648. [Google Scholar] [CrossRef]
- Tierney, W.M.; Adler, D.G.; Chand, B.; Conway, J.D.; Croffie, J.M.; DiSario, J.A.; Mishkin, D.S.; Shah, R.J.; Somogyi, L.; Wong Kee Song, L.M.; et al. Echoendoscopes. Gastrointest. Endosc. 2007, 66, 435–442. [Google Scholar] [CrossRef]
- Kaneko, M.; Katanuma, A.; Maguchi, H.; Takahashi, K.; Osanai, M.; Yane, K.; Hashigo, S.; Harada, R.; Kato, S.; Kato, R.; et al. Prospective, randomized, comparative study of delineation capability of radial scanning and curved linear array endoscopic ultrasound for the pancreaticobiliary region. Endosc. Int. Open 2014, 2, E160–E170. [Google Scholar] [CrossRef]
- Fusaroli, P.; Serrani, M.; Lisotti, A.; D’Ercole, M.C.; Ceroni, L.; Caletti, G. Performance of the forward-view echoendoscope for pancreaticobiliary examination in patients with status post-upper gastrointestinal surgery. Endosc. Ultrasound 2015, 4, 336–341. [Google Scholar] [CrossRef] [PubMed]
- Kongkam, P.; Linlawan, S.; Aniwan, S.; Lakananurak, N.; Khemnark, S.; Sahakitrungruang, C.; Pattanaarun, J.; Khomvilai, S.; Wisedopas, N.; Ridtitid, W.; et al. Forward-viewing radial-array echoendoscope for staging of colon cancer beyond the rectum. World J. Gastroenterol. 2014, 20, 2681–2687. [Google Scholar] [CrossRef]
- Katanuma, A.; Maguchi, H.; Osanai, M.; Takahashi, K. The difference in the capability of delineation between convex and radial arrayed echoendoscope for pancreas and biliary tract; case reports from the standpoint of both convex and radial arrayed echoendoscope. Dig. Endosc. 2011, 23 (Suppl. 1), 2–8. [Google Scholar] [CrossRef]
- Katanuma, A.; Maguchi, H.; Hashigo, S.; Kaneko, M.; Kin, T.; Yane, K.; Kato, R.; Kato, S.; Harada, R.; Osanai, M.; et al. Tumor seeding after endoscopic ultrasound-guided fine-needle aspiration of cancer in the body of the pancreas. Endoscopy 2012, 44 (Suppl. 2), E160–E161. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, K.; Okusaka, T.; Shimizu, K.; Furuse, J.; Ito, Y.; Hanada, K.; Shimosegawa, T.; Yamaguchi, K.; Okusaka, T.; Shimizu, K.; et al. EBM-based Clinical Guidelines for Pancreatic Cancer (2013) issued by the Japan Pancreas Society: A synopsis. Jpn. J. Clin. Oncol. 2014, 44, 883–888. [Google Scholar] [CrossRef] [PubMed]
- Hanada, K.; Okazaki, A.; Hirano, N.; Izumi, Y.; Teraoka, Y.; Ikemoto, J.; Kanemitsu, K.; Hino, F.; Fukuda, T.; Yonehara, S. Diagnostic strategies for early pancreatic cancer. J. Gastroenterol. 2015, 50, 147–154. [Google Scholar] [CrossRef]
- Takaori, K.; Matsusue, S.; Fujikawa, T.; Kobashi, Y.; Ito, T.; Matsuo, Y.; Oishi, H.; Takeda, H. Carcinoma in situ of the pancreas associated with localized fibrosis: A clue to early detection of neoplastic lesions arising from pancreatic ducts. Pancreas 1998, 17, 102–105. [Google Scholar] [CrossRef] [PubMed]
- Izumi, Y.; Hanada, K.; Okazaki, A.; Minami, T.; Hirano, N.; Ikemoto, J.; Kanemitsu, K.; Nakadoi, K.; Shishido, T.; Katamura, Y.; et al. Endoscopic ultrasound findings and pathological features of pancreatic carcinoma in situ. Endosc. Int. Open 2019, 7, E585–E593. [Google Scholar] [CrossRef] [PubMed]
- Koo, T.K.; Li, M.Y. A Guideline of Selecting and Reporting Intraclass Correlation Coefficients for Reliability Research. J. Chiropr Med. 2016, 15, 155–163. [Google Scholar] [CrossRef]
- Austin, P.C.; Schuster, T. The performance of different propensity score methods for estimating absolute effects of treatments on survival outcomes: A simulation study. Stat. Methods Med. Res. 2016, 25, 2214–2237. [Google Scholar] [CrossRef]
- Pezzi, A.; Cavo, M.; Biggeri, A.; Zamagni, E.; Nanni, O. Inverse probability weighting to estimate causal effect of a singular phase in a multiphase randomized clinical trial for multiple myeloma. BMC Med. Res. Methodol. 2016, 16, 150. [Google Scholar] [CrossRef] [PubMed]
- Okusaka, T.; Nakamura, M.; Yoshida, M.; Kitano, M.; Uesaka, K.; Ito, Y.; Furuse, J.; Hanada, K.; Okazaki, K. Clinical Practice Guidelines for Pancreatic Cancer 2019 From the Japan Pancreas Society: A Synopsis. Pancreas 2020, 49, 326–335. [Google Scholar] [CrossRef]
- Shin, E.J.; Topazian, M.; Goggins, M.G.; Syngal, S.; Saltzman, J.R.; Lee, J.H.; Farrell, J.J.; Canto, M.I. Linear-array EUS improves detection of pancreatic lesions in high-risk individuals: A randomized tandem study. Gastrointest. Endosc. 2015, 82, 812–818. [Google Scholar] [CrossRef]
- Gress, F.; Savides, T.; Cummings, O.; Sherman, S.; Lehman, G.; Zaidi, S.; Hawes, R. Radial scanning and linear array endosonography for staging pancreatic cancer: A prospective randomized comparison. Gastrointest. Endosc. 1997, 45, 138–142. [Google Scholar] [CrossRef]
- Irisawa, A. Current role of radial and curved-linear arrayed EUS scopes for diagnosis of pancreatic abnormalities in Japan. Dig. Endosc. 2011, 23 (Suppl. 1), 9–11. [Google Scholar] [CrossRef]
- Yokohata, K.; Shirahane, K.; Yonemasu, H.; Nabae, T.; Inoue, K.; Ohtsuka, T.; Yamaguchi, K.; Chijiiwa, K.; Tanaka, M. Focal ductal branch dilatation on magnetic resonance cholangiopancreatography: A hint for early diagnosis of pancreatic carcinoma. Scand. J. Gastroenterol. 2000, 35, 1229–1232. [Google Scholar] [CrossRef]
- Uehara, H.; Ikezawa, K.; Kawada, N.; Fukutake, N.; Katayama, K.; Takakura, R.; Takano, Y.; Ishikawa, O.; Takenaka, A. Diagnostic accuracy of endoscopic ultrasound-guided fine needle aspiration for suspected pancreatic malignancy in relation to the size of lesions. J. Gastroenterol. Hepatol. 2011, 26, 1256–1261. [Google Scholar] [CrossRef] [PubMed]
- Iiboshi, T.; Hanada, K.; Fukuda, T.; Yonehara, S.; Sasaki, T.; Chayama, K. Value of cytodiagnosis using endoscopic nasopancreatic drainage for early diagnosis of pancreatic cancer: Establishing a new method for the early detection of pancreatic carcinoma in situ. Pancreas 2012, 41, 523–529. [Google Scholar] [CrossRef] [PubMed]
- Mikata, R.; Ishihara, T.; Tada, M.; Tawada, K.; Saito, M.; Kurosawa, J.; Sugiyama, H.; Sakai, Y.; Tsuyuguchi, T.; Miyazaki, M.; et al. Clinical usefulness of repeated pancreatic juice cytology via endoscopic naso-pancreatic drainage tube in patients with pancreatic cancer. J. Gastroenterol. 2013, 48, 866–873. [Google Scholar] [CrossRef] [PubMed]
- Robins, J.M.; Hernán, M.A.; Brumback, B. Marginal structural models and causal inference in epidemiology. Epidemiology 2000, 11, 550–560. [Google Scholar] [CrossRef]
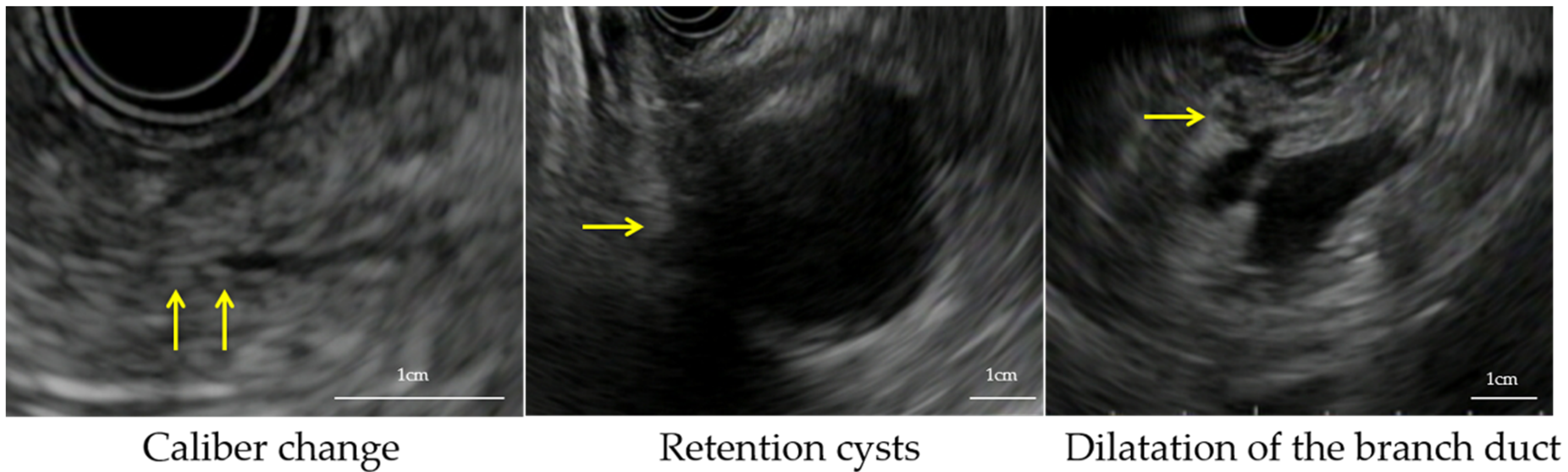
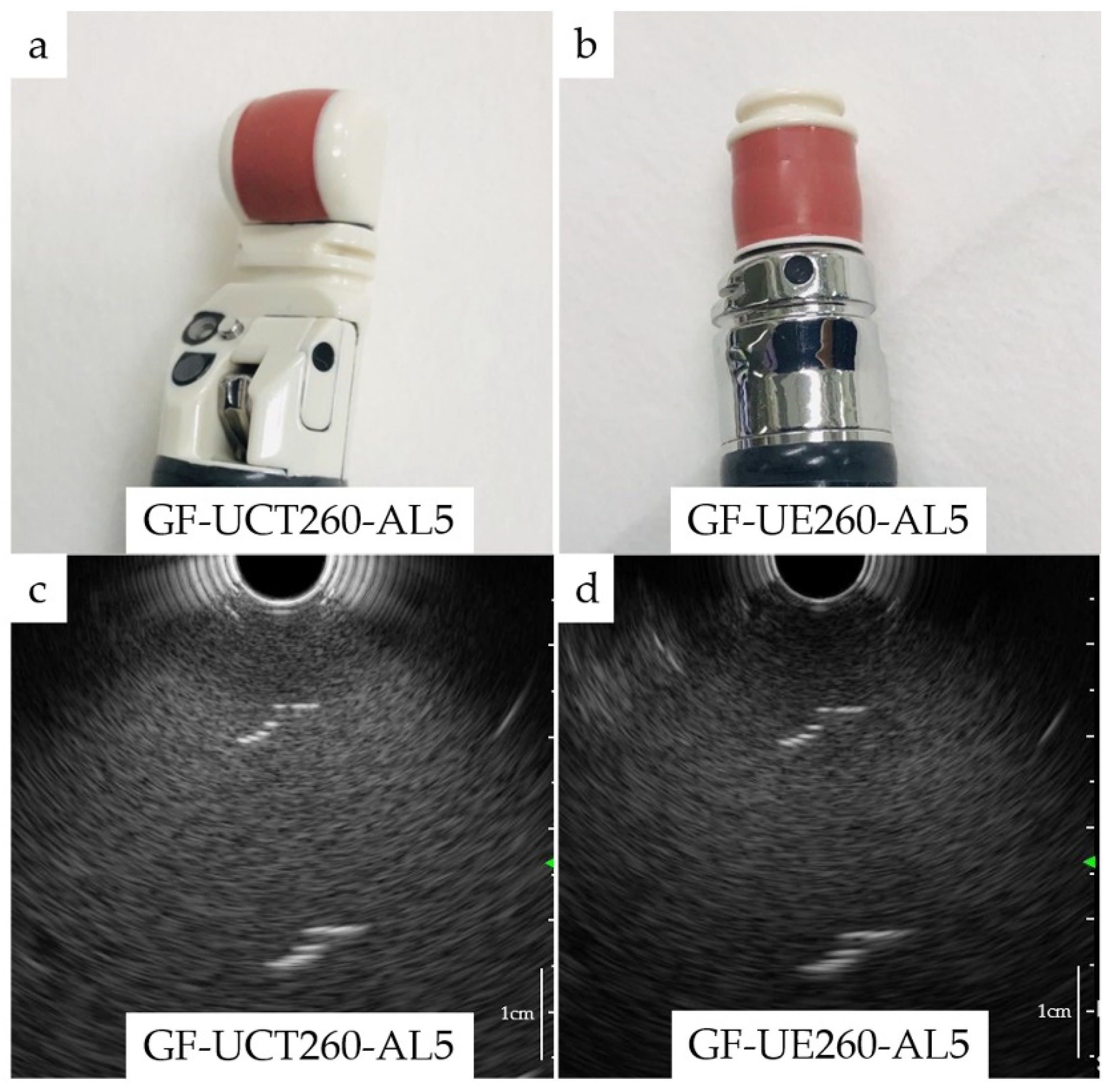
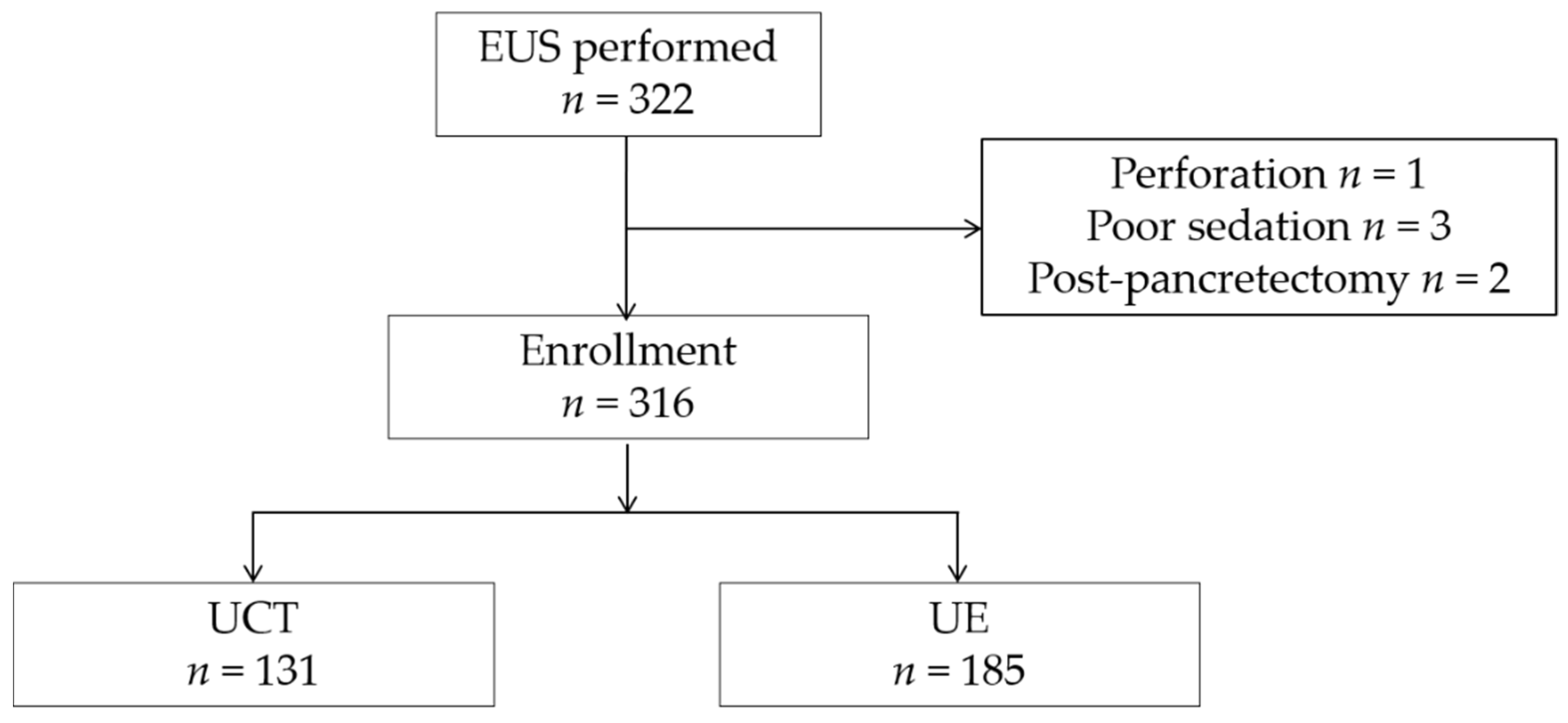
| Characteristics | Category | Radial-Type n = 185, (%) | Convex-Type n = 131, (%) | p Value |
|---|---|---|---|---|
| Age (median) | 71 | 70 | 0.60 | |
| Sex | male | 83 (44.9) | 64 (48.9) | 0.49 |
| Symptom | yes | 20 (10.8) | 22 (16.8) | 0.13 |
| Smoking habit | yes | 36 (19.5) | 34 (26.0) | 0.22 |
| Drinking habit | yes | 43 (23.2) | 39 (29.8) | 0.20 |
| DM | yes | 42 (22.7) | 25 (19.1) | 0.49 |
| Previous CT | yes | 112 (60.5) | 91 (69.5) | 0.12 |
| Previous MRI | yes | 109 (58.9) | 80 (61.1) | 0.73 |
| Part of the main lesion | 0.16 | |||
| all | 6 | 0 | ||
| uncinate process | 12 | 15 | ||
| head | 57 | 37 | ||
| neck | 20 | 15 | ||
| body | 51 | 42 | ||
| tail | 39 | 22 | ||
| Early chronic pancreatitis | yes | 30 (16.2) | 14 (10.1) | 0.19 |
| Chronic pancreatitis | yes | 16 (8.6) | 5 (3.8) | 0.11 |
| Scopist | expert | 8 (4.3) | 9 (6.9) | 0.33 |
| Indirect Findings | Radial-Type n = 185, (%) | Convex-Type n = 131, (%) | p Value |
|---|---|---|---|
| Patients based indirect findings | |||
| Indirect findings | 17 (9.2) | 3 (2.3) | 0.02 |
| Lesions based indirect findings | |||
| Caliber change | 15 (8.1) | 2 (1.5) | 0.01 |
| Branch duct dilatation | 8 (4.3) | 1 (0.8) | 0.09 |
| Retention cyst | 1 (0.5) | 0 | 1.00 |
| Slightly low echoic lesion | 14 (7.6) | 2 (1.5) | 0.02 |
| Variable | Univariate Analysis | Multivariate Analysis | ||
|---|---|---|---|---|
| Crude OR (95% CI) | p Value | Crude OR (95% CI) | p Value | |
| Age | 1.00 (0.97–1.05) | 0.73 | ||
| Sex (male) | 1.79 (0.71–4.50) | 0.22 | ||
| Smoking habit | 0.87 (0.28–2.70) | 0.81 | ||
| Drinking habit | 0.87 (0.23–2.15) | 0.53 | ||
| DM | 1.26 (0.44–3.60) | 0.67 | ||
| Symptom | 1.16 (0.33–4.15) | 0.82 | ||
| Previous CT | 0.83 (0.33–2.08) | 0.68 | 1.13 (0.43–2.97) | 0.81 |
| Previous MRI | 2.10 (0.75–5.94) | 0.16 | 2.26 (0.77–6.64) | 0.14 |
| Early chronic pancreatitis | 0.67 (0.15–3.00) | 0.60 | ||
| Chronic pancreatitis | 1.62 (0.35–7.54) | 0.54 | ||
| Scope (radial) | 4.32 (1.24–15.10) | 0.02 | 4.48 (1.27–15.70) | 0.02 |
| Scopist (expert) | 0.92 (0.12–7.31) | 0.94 | ||
| Variable | Odds Ratio (95% CI) | p Value |
|---|---|---|
| Before IPTW | ||
| Unadjusted (radial/convex) | 4.32 (1.24–15.10) | 0.02 |
| Adjusted for previous CT, previous MRI | 4.48 (1.27–15.70) | 0.02 |
| After IPTW | ||
| Unadjusted (radial/convex) | 5.94 (1.68–21.10) | 0.01 |
| Adjusted for previous CT, previous MRI | 6.04 (1.74–21.00) | 0.01 |
| Case | Age (Years) | Sex | Symptom | Opportunity | Part of the Main Lesion | Type of Echoendoscopes | Background(Early Chronic Pancreatitis/ Chronic Pancreatitis) | Indirect Findings | Further Examination (Single Cytology, SPACE, EUS-FNA, Follow Up) | Final Diagnosis | Undetected Indirect Findings by Previous CT/MRI | ||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Part | Caliber Change | Retention Cyst | Branch Duct Dilatation | Slightly Low Echoic Lesion (mm) | |||||||||||
| 1 | 70 | Male | No | PC | Body | Convex | No/No | Body | + | − | − | 10 | SPACE | PC (pT1b) | None |
| 2 | 68 | Female | Yes | BD-IPMN | Tail | Radial | No/No | Tail | + | − | + | 3 | Single cytology | IPMN and retention cyst | Slightly low echoic lesion |
| 3 | 69 | Female | No | PC | Body | Radial | No/No | Body | + | − | − | 11.2 | EUS-FNA | PC (pT2) | None |
| 4 | 54 | Male | No | PC | Neck | Radial | No/No | Neck | + | − | − | 10.7 | EUS-FNA | PC (pT1c) | None |
| 5 | 59 | Female | No | BD-IPMN | Body | Radial | No/No | Tail | − | − | + | 13.6 | Follow up | IPMN | Slightly low echoic lesion |
| 6 | 85 | Male | No | BD-IPMN | Body | Radial | No/No | Body | + | − | + | − | Follow up | IPMN | - |
| 7 | 83 | Female | No | BD-IPMN | Head | Radial | No/No | Tail | + | − | − | 12 | SPACE | IPMN | Slightly low echoic lesion |
| 8 | 78 | Male | No | BD-IPMN | Head | Radial | No/Yes | Body | + | − | + | 3.9 | SPACE | IPMN | Caliber change, Slightly low echoic lesion |
| 9 | 88 | Male | No | BD-IPMN+IPNB | Tail | Radial | No/No | Tail | + | − | − | 7.5 | Single cytology | IPMN | Slightly low echoic lesion |
| 10 | 51 | Male | No | Pancreatic pseudocyst | Body | Radial | No/Yes | Body | + | − | − | 10.7 | SPACE | Pancreatic pseudocyst | Slightly low echoic lesion |
| 11 | 59 | Male | No | BD-IPMN | Neck | Convex | No/No | Neck | − | − | + | 7.5 | Follow up | IPMN | Slightly low echoic lesion |
| 12 | 86 | Female | No | Mixed-IPMN | Neck | Radial | Yes/No | Neck | + | − | + | − | SPACE | IPMN | None |
| 13 | 75 | Female | No | BD-IPMN | Head | Radial | No/No | Tail | + | − | + | 4 | Follow up | IPMN | Caliber change, Branch duct dilatation, Slightly low echoic lesion |
| 14 | 52 | Male | No | BD-IPMN | Tail | Radial | Yes/No | Tail | − | + | − | 8 | SPACE | IPMN | Slightly low echoic lesion |
| 15 | 77 | Male | Yes | Mixed-IPMN | Body | Convex | No/No | Body | + | + | − | − | Single cytology | IPMN | None |
| 16 | 54 | Male | No | Pancreatitis | Head | Radial | No/No | Body | + | − | − | 4.6 | SPACE | No remarkable findings | Caliber change, Slightly low echoic lesion |
| 17 | 72 | Female | No | MD-IPMN | Body | Radial | No/No | Body | + | − | − | 4 | SPACE | IPMA | Slightly low echoic lesion |
| 18 | 67 | Male | No | Mixed-IPMN | Tail | Radial | No/No | Body | + | − | − | 9.1 | Surgery | IPMC (pTis) | Slightly low echoic lesion |
| 19 | 71 | Male | No | MD-IPMN | Head | Radial | No/No | Head | + | − | + | − | Single cytology | IPMN | None |
| 20 | 52 | Male | Yes | PC | Tail | Radial | No/No | Tail | + | − | + | 6.7 | SPACE | PC (pT3) | Slightly low echoic lesion |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ishikawa-Kakiya, Y.; Maruyama, H.; Yamamoto, K.; Yamamura, M.; Tanoue, K.; Higashimori, A.; Ominami, M.; Nadatani, Y.; Fukunaga, S.; Otani, K.; et al. Comparison of the Diagnostic Efficiency of Radial- and Convex-Arrayed Echoendoscopes for Indirect Findings of Pancreatic Cancer: A Retrospective Comparative Study Using Propensity Score Method. Cancers 2021, 13, 1217. https://doi.org/10.3390/cancers13061217
Ishikawa-Kakiya Y, Maruyama H, Yamamoto K, Yamamura M, Tanoue K, Higashimori A, Ominami M, Nadatani Y, Fukunaga S, Otani K, et al. Comparison of the Diagnostic Efficiency of Radial- and Convex-Arrayed Echoendoscopes for Indirect Findings of Pancreatic Cancer: A Retrospective Comparative Study Using Propensity Score Method. Cancers. 2021; 13(6):1217. https://doi.org/10.3390/cancers13061217
Chicago/Turabian StyleIshikawa-Kakiya, Yuki, Hirotsugu Maruyama, Kei Yamamoto, Masafumi Yamamura, Kojiro Tanoue, Akira Higashimori, Masaki Ominami, Yuji Nadatani, Shusei Fukunaga, Koji Otani, and et al. 2021. "Comparison of the Diagnostic Efficiency of Radial- and Convex-Arrayed Echoendoscopes for Indirect Findings of Pancreatic Cancer: A Retrospective Comparative Study Using Propensity Score Method" Cancers 13, no. 6: 1217. https://doi.org/10.3390/cancers13061217
APA StyleIshikawa-Kakiya, Y., Maruyama, H., Yamamoto, K., Yamamura, M., Tanoue, K., Higashimori, A., Ominami, M., Nadatani, Y., Fukunaga, S., Otani, K., Hosomi, S., Tanaka, F., Kamata, N., Nagami, Y., Taira, K., Shiba, M., Watanabe, T., & Fujiwara, Y. (2021). Comparison of the Diagnostic Efficiency of Radial- and Convex-Arrayed Echoendoscopes for Indirect Findings of Pancreatic Cancer: A Retrospective Comparative Study Using Propensity Score Method. Cancers, 13(6), 1217. https://doi.org/10.3390/cancers13061217






