Sonographic Features Differentiating Follicular Thyroid Cancer from Follicular Adenoma–A Meta-Analysis
Abstract
Simple Summary
Abstract
1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. Search Strategy
4.2. Data Extraction
4.3. Assessment of Methodological Quality
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Burman, K.D.; Wartofsky, L. CLINICAL PRACTICE. Thyroid Nodules. N. Engl. J. Med. 2015, 373, 2347–2356. [Google Scholar] [CrossRef]
- Borowczyk, M.; Szczepanek-Parulska, E.; Olejarz, M.; Wieckowska, B.; Verburg, F.A.; Debicki, S.; Budny, B.; Janicka-Jedynska, M.; Ziemnicka, K.; Ruchala, M. Evaluation of 167 Gene Expression Classifier (GEC) and ThyroSeq v2 Diagnostic Accuracy in the Preoperative Assessment of Indeterminate Thyroid Nodules: Bivariate/HROC Meta-analysis. Endocr. Pathol. 2019, 30, 8–15. [Google Scholar] [CrossRef]
- Cibas, E.S.; Ali, S.Z. The 2017 Bethesda System for Reporting Thyroid Cytopathology. Thyroid 2017, 27, 1341–1346. [Google Scholar] [CrossRef] [PubMed]
- Jarzab, B.; Dedecjus, M.; Slowinska-Klencka, D.; Lewinski, A.; Adamczewski, Z.; Anielski, R.; Baglaj, M.; Baldys-Waligorska, A.; Barczynski, M.; Bednarczuk, T.; et al. Guidelines of Polish National Societies Diagnostics and Treatment of Thyroid Carcinoma. 2018 Update. Endokrynol. Pol. 2018, 69, 34–74. [Google Scholar] [CrossRef] [PubMed]
- Munoz Perez, N.; Villar del Moral, J.M.; Muros Fuentes, M.A.; Lopez de la Torre, M.; Arcelus Martinez, J.I.; Becerra Massare, P.; Esteva Martinez, D.; Canadas Garre, M.; Coll Del Rey, E.; Bueno Larano, P.; et al. Could 18F-FDG-PET/CT avoid unnecessary thyroidectomies in patients with cytological diagnosis of follicular neoplasm? Langenbecks Arch. Surg 2013, 398, 709–716. [Google Scholar] [CrossRef]
- Ruchala, M.; Szczepanek, E. Thyroid ultrasound—A piece of cake? Endokrynol. Pol. 2010, 61, 330–344. [Google Scholar] [PubMed]
- Wolinski, K.; Szkudlarek, M.; Szczepanek-Parulska, E.; Ruchala, M. Usefulness of different ultrasound features of malignancy in predicting the type of thyroid lesions: A meta-analysis of prospective studies. Pol. Arch. Med. Wewn 2014, 124, 97–104. [Google Scholar] [CrossRef] [PubMed]
- Szczepanek-Parulska, E.; Wolinski, K.; Stangierski, A.; Gurgul, E.; Biczysko, M.; Majewski, P.; Rewaj-Losyk, M.; Ruchala, M. Comparison of diagnostic value of conventional ultrasonography and shear wave elastography in the prediction of thyroid lesions malignancy. PLoS ONE 2013, 8, e81532. [Google Scholar] [CrossRef] [PubMed]
- Brito, J.P.; Gionfriddo, M.R.; Al Nofal, A.; Boehmer, K.R.; Leppin, A.L.; Reading, C.; Callstrom, M.; Elraiyah, T.A.; Prokop, L.J.; Stan, M.N.; et al. The accuracy of thyroid nodule ultrasound to predict thyroid cancer: Systematic review and meta-analysis. J. Clin. Endocrinol. Metab. 2014, 99, 1253–1263. [Google Scholar] [CrossRef]
- Campanella, P.; Ianni, F.; Rota, C.A.; Corsello, S.M.; Pontecorvi, A. Quantification of cancer risk of each clinical and ultrasonographic suspicious feature of thyroid nodules: A systematic review and meta-analysis. Eur. J. Endocrinol. 2014, 170, R203–R211. [Google Scholar] [CrossRef]
- Wolinski, K.; Rewaj-Losyk, M.; Ruchala, M. Sonographic features of medullary thyroid carcinomas—A systematic review and meta-analysis. Endokrynol. Pol. 2014, 65, 314–318. [Google Scholar] [CrossRef]
- Cordes, M.; Kondrat, P.; Uder, M.; Kuwert, T.; Sasiadek, M. Differential diagnostic ultrasound criteria of papillary and follicular carcinomas: A multivariate analysis. Rofo 2014, 186, 489–495. [Google Scholar] [CrossRef] [PubMed]
- Sillery, J.C.; Reading, C.C.; Charboneau, J.W.; Henrichsen, T.L.; Hay, I.D.; Mandrekar, J.N. Thyroid follicular carcinoma: Sonographic features of 50 cases. AJR Am. J. Roentgenol. 2010, 194, 44–54. [Google Scholar] [CrossRef]
- Zhang, J.Z.; Hu, B. Sonographic features of thyroid follicular carcinoma in comparison with thyroid follicular adenoma. J. Ultrasound Med. 2014, 33, 221–227. [Google Scholar] [CrossRef] [PubMed]
- Seo, H.S.; Lee, D.H.; Park, S.H.; Min, H.S.; Na, D.G. Thyroid follicular neoplasms: Can sonography distinguish between adenomas and carcinomas? J. Clin. Ultrasound 2009, 37, 493–500. [Google Scholar] [CrossRef] [PubMed]
- Koike, E.; Noguchi, S.; Yamashita, H.; Murakami, T.; Ohshima, A.; Kawamoto, H.; Yamashita, H. Ultrasonographic characteristics of thyroid nodules: Prediction of malignancy. Arch. Surg. 2001, 136, 334–337. [Google Scholar] [CrossRef]
- Rago, T.; Di Coscio, G.; Basolo, F.; Scutari, M.; Elisei, R.; Berti, P.; Miccoli, P.; Romani, R.; Faviana, P.; Pinchera, A.; et al. Combined clinical, thyroid ultrasound and cytological features help to predict thyroid malignancy in follicular and Hupsilonrthle cell thyroid lesions: Results from a series of 505 consecutive patients. Clin. Endocrinol. (Oxf.) 2007, 66, 13–20. [Google Scholar] [CrossRef]
- Raber, W.; Kaserer, K.; Niederle, B.; Vierhapper, H. Risk factors for malignancy of thyroid nodules initially identified as follicular neoplasia by fine-needle aspiration: Results of a prospective study of one hundred twenty patients. Thyroid 2000, 10, 709–712. [Google Scholar] [CrossRef] [PubMed]
- Lin, J.D.; Hsueh, C.; Chao, T.C.; Weng, H.F.; Huang, B.Y. Thyroid follicular neoplasms diagnosed by high-resolution ultrasonography with fine needle aspiration cytology. Acta Cytol. 1997, 41, 687–691. [Google Scholar] [CrossRef]
- Stoian, D.; Borcan, F.; Petre, I.; Mozos, I.; Varcus, F.; Ivan, V.; Cioca, A.; Apostol, A.; Dehelean, C.A. Strain Elastography as a Valuable Diagnosis Tool in Intermediate Cytology (Bethesda III) Thyroid Nodules. Diagnostics 2019, 9, 119. [Google Scholar] [CrossRef]
- Asteria, C.; Giovanardi, A.; Pizzocaro, A.; Cozzaglio, L.; Morabito, A.; Somalvico, F.; Zoppo, A. US-elastography in the differential diagnosis of benign and malignant thyroid nodules. Thyroid 2008, 18, 523–531. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.K.; Chung, K.W.; Min, H.S.; Kim, T.S.; Kim, T.H.; Ryu, J.S.; Jung, Y.S.; Kim, S.K.; Lee, Y.J. Preoperative serum thyroglobulin as a useful predictive marker to differentiate follicular thyroid cancer from benign nodules in indeterminate nodules. J. Korean Med. Sci. 2012, 27, 1014–1018. [Google Scholar] [CrossRef] [PubMed]
- Lai, X.J.; Zhang, B.; Jiang, Y.X.; Zhu, Q.L.; Yang, M.; Dai, Q.; Xia, Y.; Yang, X.; Zhao, R.N. Usefulness of ultrasonography in the differential diagnosis of thyroidal follicular tumor. Zhongguo Yi Xue Ke Xue Yuan Xue Bao 2013, 35, 483–487. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.H.; Shin, J.H.; Ko, E.S.; Hahn, S.Y.; Kim, J.S.; Kim, J.H.; Oh, Y.L. Predictive factors of malignancy in patients with cytologically suspicious for Hurthle cell neoplasm of thyroid nodules. Int. J. Surg. 2013, 11, 898–902. [Google Scholar] [CrossRef]
- Lee, S.H.; Baek, J.S.; Lee, J.Y.; Lim, J.A.; Cho, S.Y.; Lee, T.H.; Ku, Y.H.; Kim, H.I.; Kim, M.J. Predictive factors of malignancy in thyroid nodules with a cytological diagnosis of follicular neoplasm. Endocr. Pathol. 2013, 24, 177–183. [Google Scholar] [CrossRef]
- Pompili, G.; Tresoldi, S.; Primolevo, A.; De Pasquale, L.; Di Leo, G.; Cornalba, G. Management of thyroid follicular proliferation: An ultrasound-based malignancy score to opt for surgical or conservative treatment. Ultrasound Med. Biol. 2013, 39, 1350–1355. [Google Scholar] [CrossRef]
- Kamran, S.C.; Marqusee, E.; Kim, M.I.; Frates, M.C.; Ritner, J.; Peters, H.; Benson, C.B.; Doubilet, P.M.; Cibas, E.S.; Barletta, J.; et al. Thyroid nodule size and prediction of cancer. J. Clin. Endocrinol. Metab. 2013, 98, 564–570. [Google Scholar] [CrossRef] [PubMed]
- Tutuncu, Y.; Berker, D.; Isik, S.; Akbaba, G.; Ozuguz, U.; Kucukler, F.K.; Gocmen, E.; Yalcin, Y.; Aydin, Y.; Guler, S. The frequency of malignancy and the relationship between malignancy and ultrasonographic features of thyroid nodules with indeterminate cytology. Endocrine 2014, 45, 37–45. [Google Scholar] [CrossRef]
- Yoon, J.H.; Kim, E.K.; Youk, J.H.; Moon, H.J.; Kwak, J.Y. Better understanding in the differentiation of thyroid follicular adenoma, follicular carcinoma, and follicular variant of papillary carcinoma: A retrospective study. Int. J. Endocrinol. 2014, 2014, 321595. [Google Scholar] [CrossRef]
- Cordes, M.; Nagel, H.; Horstrup, K.; Sasiadek, M.; Kuwert, T. Ultrasound characteristics of thyroid nodules diagnosed as follicular neoplasms by fine-needle aspiration cytology. A prospective study with histological correlation. Nuklearmedizin 2016, 55, 93–98. [Google Scholar] [CrossRef]
- Jeong, S.H.; Hong, H.S.; Lee, E.H. Can Nodular Hyperplasia of the Thyroid Gland be Differentiated From Follicular Adenoma and Follicular Carcinoma by Ultrasonography? Ultrasound Q 2016, 32, 349–355. [Google Scholar] [CrossRef]
- Kobayashi, K.; Hirokawa, M.; Yabuta, T.; Masuoka, H.; Fukushima, M.; Kihara, M.; Higashiyama, T.; Ito, Y.; Miya, A.; Amino, N.; et al. Tumor protrusion with intensive blood signals on ultrasonography is a strongly suggestive finding of follicular thyroid carcinoma. Med. Ultrason. 2016, 18, 25–29. [Google Scholar] [CrossRef]
- Yang, G.C.H.; Fried, K.O. Most Thyroid Cancers Detected by Sonography Lack Intranodular Vascularity on Color Doppler Imaging: Review of the Literature and Sonographic-Pathologic Correlations for 698 Thyroid Neoplasms. J. Ultrasound Med. 2017, 36, 89–94. [Google Scholar] [CrossRef]
- Kuru, B.; Kefeli, M. Risk factors associated with malignancy and with triage to surgery in thyroid nodules classified as Bethesda category IV (FN/SFN). Diagn. Cytopathol. 2018, 46, 489–494. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Han, M.; Lee, J.H.; Song, D.E.; Kim, K.; Baek, J.H.; Shong, Y.K.; Kim, W.G. Tumour growth rate of follicular thyroid carcinoma is not different from that of follicular adenoma. Clin. Endocrinol. (Oxf.) 2018, 88, 936–942. [Google Scholar] [CrossRef] [PubMed]
- Kuo, T.C.; Wu, M.H.; Chen, K.Y.; Hsieh, M.S.; Chen, A.; Chen, C.N. Ultrasonographic features for differentiating follicular thyroid carcinoma and follicular adenoma. Asian J. Surg. 2020, 43, 339–346. [Google Scholar] [CrossRef]
- Liu, B.J.; Zhang, Y.F.; Zhao, C.K.; Wang, H.X.; Li, M.X.; Xu, H.X. Conventional ultrasound characteristics, TI-RADS category and shear wave speed measurement between follicular adenoma and follicular thyroid carcinoma. Clin. Hemorheol. Microcirc. 2020, 75, 291–301. [Google Scholar] [CrossRef]
- Aschebrook-Kilfoy, B.; Grogan, R.H.; Ward, M.H.; Kaplan, E.; Devesa, S.S. Follicular thyroid cancer incidence patterns in the United States, 1980-2009. Thyroid 2013, 23, 1015–1021. [Google Scholar] [CrossRef] [PubMed]
- Slowinska-Klencka, D.; Klencki, M.; Sporny, S.; Lewinski, A. Fine-needle aspiration biopsy of the thyroid in an area of endemic goitre: Influence of restored sufficient iodine supplementation on the clinical significance of cytological results. Eur. J. Endocrinol. 2002, 146, 19–26. [Google Scholar] [CrossRef] [PubMed]
- Russ, G.; Bonnema, S.J.; Erdogan, M.F.; Durante, C.; Ngu, R.; Leenhardt, L. European Thyroid Association Guidelines for Ultrasound Malignancy Risk Stratification of Thyroid Nodules in Adults: The EU-TIRADS. Eur. Thyroid J. 2017, 6, 225–237. [Google Scholar] [CrossRef]
- Kunt, M.; Cirit, E.; Eray, I.C.; Yalay, O.; Parsak, C.K.; Sakmann, G. Parameters predicting follicular carcinoma in thyroid nodules with indeterminate cytology. Ann. Ital. Chir. 2015, 86, 301–305; discussion 306. [Google Scholar] [PubMed]
- Chng, C.L.; Kurzawinski, T.R.; Beale, T. Value of sonographic features in predicting malignancy in thyroid nodules diagnosed as follicular neoplasm on cytology. Clin. Endocrinol. (Oxf.) 2015, 83, 711–716. [Google Scholar] [CrossRef] [PubMed]
- Maia, F.F.; Matos, P.S.; Pavin, E.J.; Vassallo, J.; Zantut-Wittmann, D.E. Value of ultrasound and cytological classification system to predict the malignancy of thyroid nodules with indeterminate cytology. Endocr. Pathol. 2011, 22, 66–73. [Google Scholar] [CrossRef]
- Russ, G.; Bigorgne, C.; Royer, B.; Rouxel, A.; Bienvenu-Perrard, M. The Thyroid Imaging Reporting and Data System (TIRADS) for ultrasound of the thyroid. J. Radiol. 2011, 92, 701–713. [Google Scholar] [CrossRef] [PubMed]
- Verburg, F.A.; Mader, U.; Luster, M.; Reiners, C. Histology does not influence prognosis in differentiated thyroid carcinoma when accounting for age, tumour diameter, invasive growth and metastases. Eur. J. Endocrinol. 2009, 160, 619–624. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Borowczyk, M.; Szczepanek-Parulska, E.; Debicki, S.; Budny, B.; Verburg, F.A.; Filipowicz, D.; Wieckowska, B.; Janicka-Jedynska, M.; Gil, L.; Ziemnicka, K.; et al. Differences in Mutational Profile between Follicular Thyroid Carcinoma and Follicular Thyroid Adenoma Identified Using Next Generation Sequencing. Int. J. Mol. Sci. 2019, 20, 3126. [Google Scholar] [CrossRef] [PubMed]
- Frates, M.C.; Benson, C.B.; Doubilet, P.M.; Cibas, E.S.; Marqusee, E. Can color Doppler sonography aid in the prediction of malignancy of thyroid nodules? J. Ultrasound Med. 2003, 22, 127–131. [Google Scholar] [CrossRef]
- Fukunari, N.; Nagahama, M.; Sugino, K.; Mimura, T.; Ito, K.; Ito, K. Clinical evaluation of color Doppler imaging for the differential diagnosis of thyroid follicular lesions. World J. Surg. 2004, 28, 1261–1265. [Google Scholar] [CrossRef]
- Rago, T.; Scutari, M.; Santini, F.; Loiacono, V.; Piaggi, P.; Di Coscio, G.; Basolo, F.; Berti, P.; Pinchera, A.; Vitti, P. Real-time elastosonography: Useful tool for refining the presurgical diagnosis in thyroid nodules with indeterminate or nondiagnostic cytology. J. Clin. Endocrinol. Metab. 2010, 95, 5274–5280. [Google Scholar] [CrossRef]
- Trimboli, P.; Castellana, M.; Piccardo, A.; Romanelli, F.; Grani, G.; Giovanella, L.; Durante, C. The ultrasound risk stratification systems for thyroid nodule have been evaluated against papillary carcinoma. A meta-analysis. Rev. Endocr. Metab. Disord. 2020. [Google Scholar] [CrossRef] [PubMed]
- Koc, A.M.; Adibelli, Z.H.; Erkul, Z.; Sahin, Y.; Dilek, I. Comparison of diagnostic accuracy of ACR-TIRADS, American Thyroid Association (ATA), and EU-TIRADS guidelines in detecting thyroid malignancy. Eur. J. Radiol. 2020, 133, 109390. [Google Scholar] [CrossRef]
- Migda, B.; Migda, M.; Migda, A.M.; Bierca, J.; Slowniska-Srzednicka, J.; Jakubowski, W.; Slapa, R.Z. Evaluation of Four Variants of the Thyroid Imaging Reporting and Data System (TIRADS) Classification in Patients with Multinodular Goitre—Initial study. Endokrynol. Pol. 2018, 69, 156–162. [Google Scholar] [CrossRef]
- Grant, E.G.; Tessler, F.N.; Hoang, J.K.; Langer, J.E.; Beland, M.D.; Berland, L.L.; Cronan, J.J.; Desser, T.S.; Frates, M.C.; Hamper, U.M.; et al. Thyroid Ultrasound Reporting Lexicon: White Paper of the ACR Thyroid Imaging, Reporting and Data System (TIRADS) Committee. J. Am. Coll. Radiol. 2015, 12, 1272–1279. [Google Scholar] [CrossRef]
- Singaporewalla, R.M.; Hwee, J.; Lang, T.U.; Desai, V. Clinico-pathological Correlation of Thyroid Nodule Ultrasound and Cytology Using the TIRADS and Bethesda Classifications. World J. Surg. 2017, 41, 1807–1811. [Google Scholar] [CrossRef] [PubMed]
- Shayganfar, A.; Hashemi, P.; Esfahani, M.M.; Ghanei, A.M.; Moghadam, N.A.; Ebrahimian, S. Prediction of thyroid nodule malignancy using thyroid imaging reporting and data system (TIRADS) and nodule size. Clin. Imaging 2020, 60, 222–227. [Google Scholar] [CrossRef] [PubMed]
- Slowinska-Klencka, D.; Wysocka-Konieczna, K.; Klencki, M.; Popowicz, B. Diagnostic Value of Six Thyroid Imaging Reporting and Data Systems (TIRADS) in Cytologically Equivocal Thyroid Nodules. J. Clin. Med. 2020, 9, 2281. [Google Scholar] [CrossRef] [PubMed]
- Chaigneau, E.; Russ, G.; Royer, B.; Bigorgne, C.; Bienvenu-Perrard, M.; Rouxel, A.; Leenhardt, L.; Belin, L.; Buffet, C. TIRADS score is of limited clinical value for risk stratification of indeterminate cytological results. Eur. J. Endocrinol. 2018, 179, 13–20. [Google Scholar] [CrossRef]
- Shamseer, L.; Moher, D.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A.; Group, P.-P. Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015: Elaboration and explanation. BMJ 2015, 350, g7647. [Google Scholar] [CrossRef]
- Lo, C.K.; Mertz, D.; Loeb, M. Newcastle-Ottawa Scale: Comparing reviewers’ to authors’ assessments. BMC Med. Res. Methodol. 2014, 14, 45. [Google Scholar] [CrossRef] [PubMed]
- Egger, M.; Davey Smith, G.; Schneider, M.; Minder, C. Bias in meta-analysis detected by a simple, graphical test. BMJ 1997, 315, 629–634. [Google Scholar] [CrossRef]
- Sterne, J.A.; Egger, M.; Smith, G.D. Systematic reviews in health care: Investigating and dealing with publication and other biases in meta-analysis. BMJ 2001, 323, 101–105. [Google Scholar] [CrossRef] [PubMed]
- Newcombe, R.G. Two-sided confidence intervals for the single proportion: Comparison of seven methods. Stat. Med. 1998, 17, 857–872. [Google Scholar] [CrossRef]

| Author | Year | Number
of Nodules (FTC/FTA); Malignancy Rate (%) | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Seo HS et al. [15] | 2009 | 126 (66/60) 52.4 | ||||||||||||||
| Sillery JC et al. [13] | 2010 | 102 (50/52); 49.0 | ||||||||||||||
| Lee EK et al. [22] | 2012 | 110 (33/77); 30.0 | ||||||||||||||
| Lai X et al. [23] | 2013 | 111 (37/74); 33.3 | ||||||||||||||
| Lee KH et al. [24] | 2013 | 75 (11/64); 14.7 | ||||||||||||||
| Lee SH et al. [25] | 2013 | 66 (16/50), 24.2 | ||||||||||||||
| Pompili G et al. [26] | 2013 | 102 (14/88); 13.7 | ||||||||||||||
| Kamran SC et al. [27] | 2013 | 7348 (927/6421); 12.6 | ||||||||||||||
| Tutuncu J et al. [28] | 2014 | 88 (6/82); 6.8 | ||||||||||||||
| Cordes M et al. [12] | 2014 | 57 (24/33); 42.1 | ||||||||||||||
| Yoon JH et al. [29] | 2014 | 177 (25/152); 14.1 | ||||||||||||||
| Zhang JZ et al. [14] | 2014 | 88 (36/52); 40.9 | ||||||||||||||
| Cordes M et al. [30] | 2016 | 200 (100/100); 50 | ||||||||||||||
| Jeong SH et al. [31] | 2016 | 178 (22/156); 12.4 | ||||||||||||||
| Kobayashi K et al. [32] | 2016 | 531 (184/347); 34.7 | ||||||||||||||
| Yang GCH et al. [33] | 2016 | 279 (6/273); 2.2 | ||||||||||||||
| Kuru B.et al. [34] | 2018 | 139 (51/88). 36.7 | ||||||||||||||
| Kim M et al. [35] | 2018 | 160 (50/110); 31.3 | ||||||||||||||
| Kuo TC et al. [36] | 2020 | 188 (49/139); 26.1 | ||||||||||||||
| Liu BJ et al. [37] | 2020 | 90 (28/62); 31.1 |
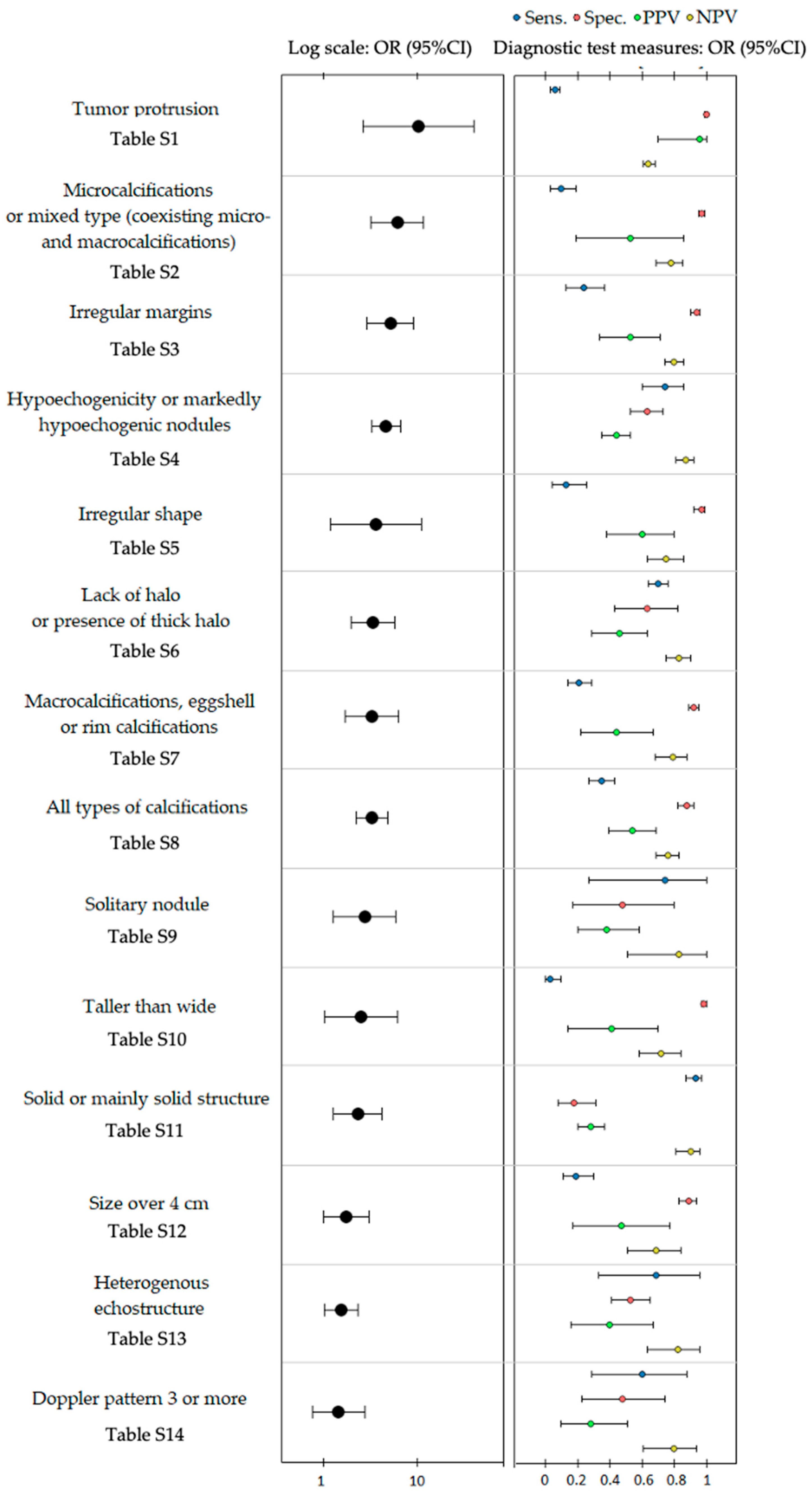
| Sonographic Feature | OR (95% CI) | Sensitivity (95% CI) | Specificity (95% CI) | PPV (95% CI) | NPV (95% CI) |
|---|---|---|---|---|---|
| Tumor protrusion Table S1 | 10.19 (2.62–39.71) | 0.06 (0.03–0.09) | 1.00 (0.99–1.00) | 0.96 (0.7–1.00) | 0.64 (0.61–0.68) |
| Microcalcifications or mixed type (coexisting micro- and macrocalcifications) Table S2 | 6.09 (3.22–11.5) | 0.10 (0.03–0.19) | 0.97 (0.95–0.99) | 0.53 (0.19–0.86) | 0.78 (0.69–0.85) |
| Irregular margins Table S3 | 5.11 (2.9–8.99) | 0.24 (0.13–0.37) | 0.94 (0.90–0.96) | 0.53 (0.34–0.71) | 0.80 (0.74–0.86) |
| Hypoechogenicity or marked hypoechogenicity Table S4 | 4.59 (3.23–6.54) | 0.74 (0.6–0.86) | 0.63 (0.53–0.73) | 0.44 (0.35–0.53) | 0.87 (0.81–0.92) |
| Irregular shape Table S5 | 3.6 (1.19–10.92) | 0.13 (0.04–0.26) | 0.97 (0.92–0.99) | 0.60 (0.38–0.8) | 0.75 (0.63–0.86) |
| Lack of halo or presence of thick halo Table S6 | 3.34 (1.95–5.73) | 0.70 (0.64–0.76) | 0.63 (0.43–0.82) | 0.46 (0.29–0.63) | 0.83 (0.75–0.90) |
| Macrocalcifications, eggshell or rim calcifications Table S7 | 3.28 (1.69–6.35) | 0.21 (0.14–0.29) | 0.92 (0.89–0.95) | 0.44 (0.22–0.67) | 0.79 (0.68–0.88) |
| All types of calcifications Table S8 | 3.26 (2.20–4.83) | 0.35 (0.27–0.43) | 0.88 (0.82–0.92) | 0.54 (0.39–0.69) | 0.76 (0.69–0.83) |
| Solitary nodule Table S9 | 2.72 (1.26–5.86) | 0.74 (0.27–1.00) | 0.48 (0.17–0.80) | 0.38 (0.20–0.58) | 0.83 (0.51–1.00) |
| Taller than wide Table S10 | 2.52 (1.02–6.19) | 0.03 (0.00–0.10) | 0.98 (0.97–1.00) | 0.41 (0.14–0.70) | 0.72 (0.58–0.84) |
| Solid or mainly solid structure Table S11 | 2.3 (1.27–4.17) | 0.93 (0.87–0.97) | 0.18 (0.08–0.31) | 0.28 (0.20–0.37) | 0.9 (0.81–0.96) |
| Size over 4 cm Table S12 | 1.73 (0.99–3.00) | 0.19 (0.11–0.30) | 0.89 (0.83–0.94) | 0.47 (0.17–0.77) | 0.69 (0.51–0.84) |
| Heterogenous echostructure Table S13 | 1.53 (1.02–2.30) | 0.69 (0.33–0.96) | 0.53 (0.41–0.65) | 0.4 (0.16–0.67) | 0.82 (0.63–0.96) |
| Doppler pattern 3 or more Table S14 | 1.44 (0.76–2.74) | 0.60 (0.29–0.88) | 0.48 (0.23–0.74) | 0.28 (0.10–0.51) | 0.80 (0.61–0.94) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Borowczyk, M.; Woliński, K.; Więckowska, B.; Jodłowska-Siewert, E.; Szczepanek-Parulska, E.; Verburg, F.A.; Ruchała, M. Sonographic Features Differentiating Follicular Thyroid Cancer from Follicular Adenoma–A Meta-Analysis. Cancers 2021, 13, 938. https://doi.org/10.3390/cancers13050938
Borowczyk M, Woliński K, Więckowska B, Jodłowska-Siewert E, Szczepanek-Parulska E, Verburg FA, Ruchała M. Sonographic Features Differentiating Follicular Thyroid Cancer from Follicular Adenoma–A Meta-Analysis. Cancers. 2021; 13(5):938. https://doi.org/10.3390/cancers13050938
Chicago/Turabian StyleBorowczyk, Martyna, Kosma Woliński, Barbara Więckowska, Elżbieta Jodłowska-Siewert, Ewelina Szczepanek-Parulska, Frederik A. Verburg, and Marek Ruchała. 2021. "Sonographic Features Differentiating Follicular Thyroid Cancer from Follicular Adenoma–A Meta-Analysis" Cancers 13, no. 5: 938. https://doi.org/10.3390/cancers13050938
APA StyleBorowczyk, M., Woliński, K., Więckowska, B., Jodłowska-Siewert, E., Szczepanek-Parulska, E., Verburg, F. A., & Ruchała, M. (2021). Sonographic Features Differentiating Follicular Thyroid Cancer from Follicular Adenoma–A Meta-Analysis. Cancers, 13(5), 938. https://doi.org/10.3390/cancers13050938






