First-Line Atezolizumab Plus Bevacizumab versus Sorafenib in Hepatocellular Carcinoma: A Cost-Effectiveness Analysis
Abstract
Simple Summary
Abstract
1. Introduction
2. Methods
2.1. Modeling without Long-Term Outcome Data
2.2. Model Structure
2.3. Survival Estimates
2.4. Utility Estimates
2.5. Cost Estimates
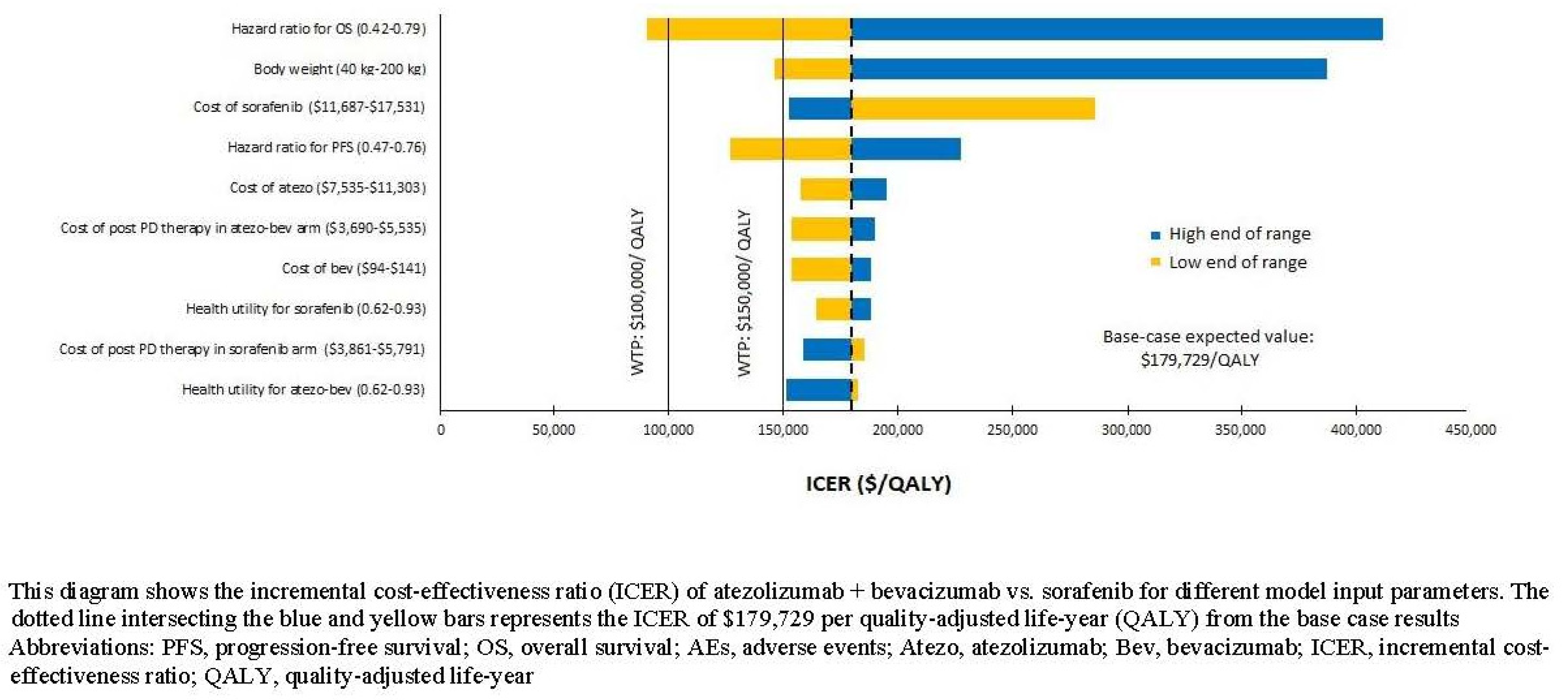
2.6. Sensitivity Analyses
3. Results
3.1. Base Case Analysis
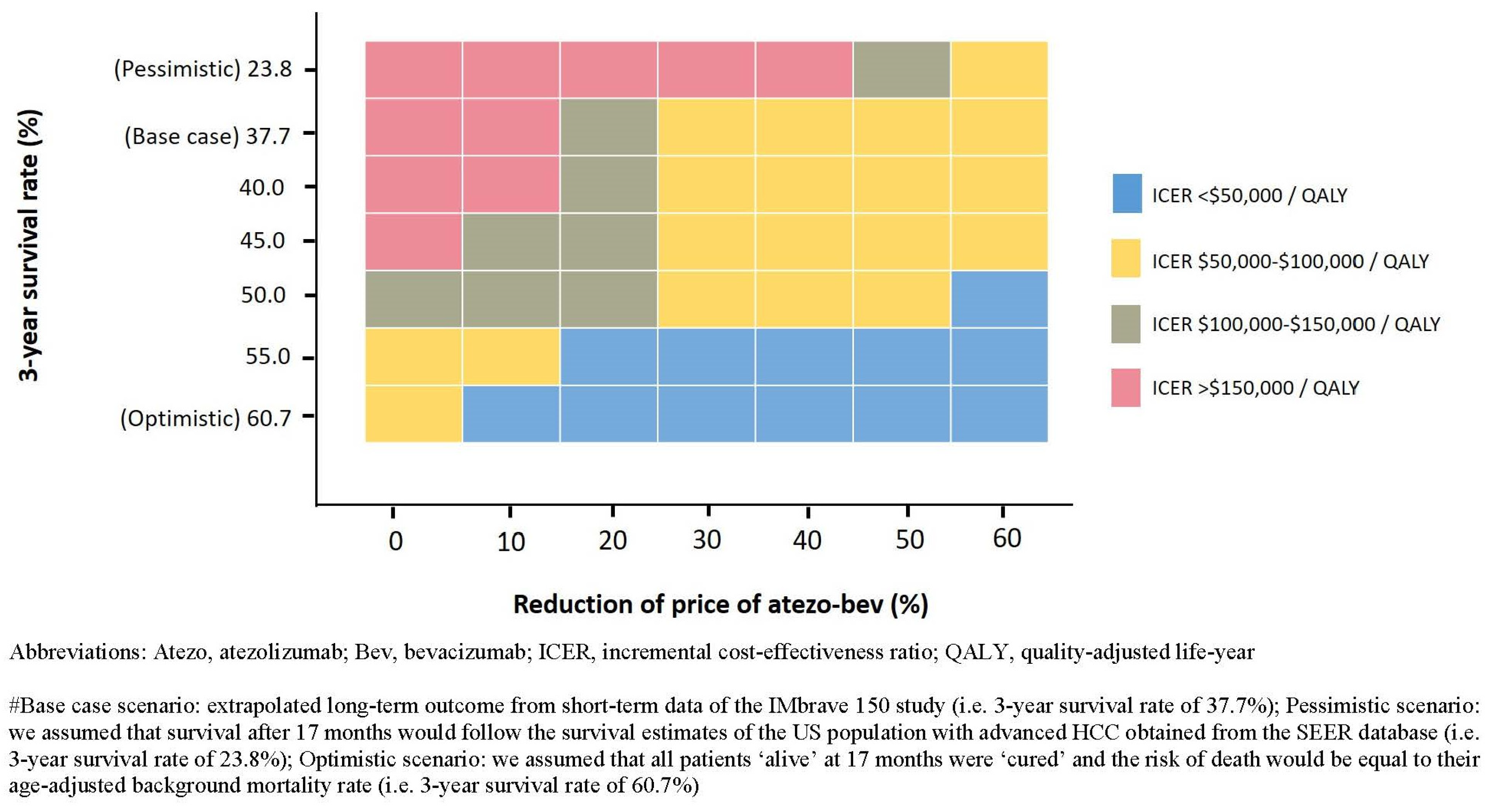
3.2. Scenario and Sensitivity Analyses
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Llovet, J.M.; Ricci, S.; Mazzaferro, V.; Hilgard, P.; Gane, E.; Blanc, J.F.; de Oliveira, A.C.; Santoro, A.; Raoul, J.L.; Forner, A.; et al. Sorafenib in advanced hepatocellular carcinoma. N. Engl. J. Med. 2008, 359, 378–390. [Google Scholar] [CrossRef]
- Kudo, M.; Finn, R.S.; Qin, S.; Han, K.H.; Ikeda, K.; Piscaglia, F.; Baron, A.; Park, J.W.; Han, G.; Jassem, J.; et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellu-lar carcinoma: A randomised phase 3 non-inferiority trial. Lancet 2018, 391, 1163–1173. [Google Scholar] [CrossRef]
- Zhu, A.X.; Finn, R.S.; Edeline, J.; Cattan, S.; Ogasawara, S.; Palmer, D.; Verslype, C.; Zagonel, V.; Fartoux, L.; Vogel, A.; et al. Pembrolizumab in patients with advanced hepatocellular carcinoma previously treated with sorafenib (KEYNOTE-224): A non-randomised, open-label phase 2 trial. Lancet Oncol. 2018, 19, 940–952. [Google Scholar] [CrossRef]
- El-Khoueiry, A.B.; Sangro, B.; Yau, T.; Crocenzi, T.S.; Kudo, M.; Hsu, C.; Kim, T.Y.; Choo, S.P.; Trojan, J.; Welling, T.H., 3rd; et al. Nivolumab in patients with advanced hepatocellular carcinoma (CheckMate 040): An open-label, non-comparative, phase 1/2 dose escalation and expansion trial. Lancet 2017, 389, 2492–2502. [Google Scholar] [CrossRef]
- Finn, R.S.; Ryoo, B.Y.; Merle, P.; Kudo, M.; Bouattour, M.; Lim, H.Y.; Breder, V.; Edeline, J.; Chao, Y.; Ogasawara, S.; et al. Pembrolizumab As Second-Line Therapy in Patients with Advanced Hepatocellular Carci-noma in KEYNOTE-240: A Randomized, Double-Blind, Phase III Trial. J. Clin. Oncol. 2020, 38, 193–202. [Google Scholar] [CrossRef] [PubMed]
- Yau, T.; Park, J.W.; Finn, R.S.; Cheng, A.L.; Mathurin, P.; Edeline, J.; Kudo, M.; Han, K.H.; Hardling, J.J.; Merle, P.; et al. CheckMate 459: A Randomized, Multi-Center Phase 3 Study of Nivolumab (NIVO) vs Soraf-enib (SOR) as First-Line (1L) Treatment in Patients (pts) With Advanced Hepatocellular Carcinoma (aHCC). Ann. Oncol. 2019, 30 (Suppl. S5), v851–v934. [Google Scholar] [CrossRef]
- Chen, D.S.; Hurwitz, H. Combinations of Bevacizumab with Cancer Immunotherapy. Cancer J. 2018, 24, 193–204. [Google Scholar] [CrossRef] [PubMed]
- Herbst, R.S.; Soria, J.C.; Kowanetz, M.; Fine, G.D.; Hamid, O.; Gordon, M.S.; Sosman, J.A.; McDermott, D.F.; Powderly, J.D.; Gettinger, S.N.; et al. Predictive correlates of response to the anti-PD-L1 antibody MPDL3280A in cancer patients. Nature 2014, 515, 563–567. [Google Scholar] [CrossRef] [PubMed]
- Morse, M.A.; Sun, W.; Kim, R.; He, A.R.; Abada, P.B.; Mynderse, M.; Finn, R.S. The Role of Angiogenesis in Hepatocellular Carcinoma. Clin. Cancer Res. 2019, 25, 912–920. [Google Scholar] [CrossRef]
- Zhu, A.X.; Duda, D.G.; Sahani, D.V.; Jain, R.K. HCC and angiogenesis: Possible targets and future directions. Nat. Rev. Clin. Oncol. 2011, 8, 292–301. [Google Scholar] [CrossRef] [PubMed]
- Wallin, J.J.; Bendell, J.C.; Funke, R.; Sznol, M.; Korski, K.; Jones, S.; Hernandez, G.; Mier, J.; He, X.; Hodi, F.S.; et al. Atezolizumab in combination with bevacizumab enhances antigen-specific T-cell mi-gration in metastatic renal cell carcinoma. Nat. Commun. 2016, 7, 12624. [Google Scholar] [CrossRef]
- Hegde, P.S.; Wallin, J.J.; Mancao, C. Predictive markers of anti-VEGF and emerging role of angiogenesis inhibitors as immuno-therapeutics. Semin. Cancer Biol. 2018, 52, 117–124. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.; Ryoo, B.; Hsu, C.; Numata, K.; Stein, S.; Verret, W.; Hack, S.; Spahn, J.; Liu, B.; Abdullah, H.; et al. Randomised Efficacy and Safety Results for Atezolizumab (Atezo) + Bevacizumab (Bev) in Pa-tients (pts) With Previously Untreated, Unresectable Hepatocellular Carcinoma (HCC). Ann. Oncol. 2019, 30 (Suppl. S5), v851–v934. [Google Scholar] [CrossRef]
- Finn, R.S.; Qin, S.; Ikeda, M.; Galle, P.R.; Ducreux, M.; Kim, T.Y.; Kudo, M.; Breder, V.; Merle, P.; Kaseb, A.O.; et al. Atezolizumab plus Bevacizumab in Unresectable Hepatocellular Carcinoma. N. Engl. J. Med. 2020, 382, 1894–1905. [Google Scholar] [CrossRef]
- Casak, S.J.; Donoghue, M.; Fashoyin-Aje, L.; Jiang, X.; Rodriguez, L.; Shen, Y.L.; Xu, Y.; Jiang, X.; Liu, J.; Zhao, H.; et al. FDA Approval Summary: Atezolizumab Plus Bevacizumab for the Treatment of Patients with Advanced Unresectable or Metastatic Hepatocellular Carcinoma. Clin. Cancer Res. 2020. [Google Scholar] [CrossRef]
- Yang, J.D.; Hainaut, P.; Gores, G.J.; Amadou, A.; Plymoth, A.; Roberts, L.R. A global view of hepatocellular carcinoma: Trends, risk, prevention and management. Nat. Rev. Gastroenterol. Hepatol. 2019, 16, 589–604. [Google Scholar] [CrossRef]
- Yang, X.; Wang, D.; Lin, J.; Yang, X.; Zhao, H. Atezolizumab plus bevacizumab for unresectable hepatocellular carcinoma. Lancet Oncol. 2020, 21, 9. [Google Scholar] [CrossRef]
- Parikh, N.D.; Singal, A.G.; Hutton, D.W. Cost-effectiveness of regorafenib as second-line therapy for patients with advanced hepatocellular carcinoma. Cancer 2017, 123, 3725–3731. [Google Scholar] [CrossRef]
- Soto-Perez-de-Celis, E.; Aguiar, P.N.; Cordon, M.L.; Chavarri-Guerra, Y.; Lopes, G.L. Cost-effectiveness of cabozantinib in the second-line treatment of advanced hepatocellular carcinoma. J. Natl. Compr. Cancer Netw. 2019, 17, 669–675. [Google Scholar] [CrossRef]
- Cammà, C.; Cabibbo, G.; Petta, S.; Enea, M.; Iavarone, M.; Grieco, A.; Gasbarrini, A.; Villa, E.; Zavaglia, C.; Bruno, R.; et al. Cost-effectiveness of sorafenib treatment in field practice for patients with hepatocellular carcinoma. Hepatology 2013, 57, 1046–1054. [Google Scholar] [CrossRef] [PubMed]
- Chiang, C.L.; Chan, S.K.; Lee, S.F.; Wong, I.O.; Choi, H.C. Cost-effectiveness of Pembrolizumab as a Second-Line Therapy for Hepatocellular Carcinoma. JAMA Netw. Open 2021, 4, e2033761. [Google Scholar] [CrossRef]
- Othus, M.; Bansal, A.; Koepl, L.; Wagner, S.; Ramsey, S. Accounting for Cured Patients in Cost-Effectiveness Analysis. Value Health 2017, 20, 705–709. [Google Scholar] [CrossRef]
- Bullement, A.; Latimer, N.R.; Bell Gorrod, H. Survival Extrapolation in Cancer Immunotherapy: A Validation-Based Case Study. Value Health 2019, 22, 276–283. [Google Scholar] [CrossRef]
- Prasad, V.; Mailankody, S. How should we assess the value of innovative drugs in oncology? Lessons from cost-effectiveness analyses. Blood 2015, 126, 1860–1861. [Google Scholar] [CrossRef] [PubMed]
- Surveillance, Epidemiology, and End Results (SEER) Program. SEER*Stat Database: Incidence—SEER 9 Regs Research Data. Linked to County Attributes—Total U.S., 1975–2017 Counties, National Cancer Institute, DCCPS, Surveillance Research Program. 2020. Available online: https://seer.cancer.gov (accessed on 31 October 2020).
- Cabibbo, G.; Cucchetti, A.; Cammà, C.; Casadei-Gardini, A.; Celsa, C.; Emanuele Maria Rizzo, G.; Johnson, P.; Ercolani, G. Outcomes of hepatocellular carcinoma patients treated with sorafenib: A meta-analysis of Phase III trials. Future Oncol. 2019, 15, 3411–3422. [Google Scholar] [CrossRef] [PubMed]
- Husereau, D.; Drummond, M.; Petrou, S.; Carswell, C.; Moher, D.; Greenberg, D.; Augustovski, F.; Briggs, A.H.; Mauskopf, J.; Loder, E.; et al. Consolidated Health Economic Evaluation Reporting Standards (CHEERS)—Explanation and elaboration: A report of the ISPOR Health Economic Evaluation Publication Guidelines Good Reporting Practices Task Force. Value Health 2013, 16, 231–250. [Google Scholar] [CrossRef]
- Hoyle, M.W.; Henley, W. Improved curve fits to summary survival data: Application to economic evaluation of health tech-nologies. BMC Med. Res. Methodol. 2011, 11, 139. [Google Scholar] [CrossRef]
- Wan, X.; Peng, L.; Li, Y. A Review and Comparison of Methods for Recreating Individual Patient Data from Published Kaplan-Meier Survival Curves for Economic Evaluations: A Simulation Study. PLoS ONE 2015, 10, e0121353. [Google Scholar] [CrossRef]
- Atkins, M.B.; Rini, B.I.; Motzer, R.J.; Powles, T.; McDermott, D.F.; Suarez, C.; Bracarda, S.; Stadler, W.M.; Donskov, F.; Gurney, H.; et al. Patient-Reported Outcomes from the Phase III Randomized IMmotion151 Trial: Ate-zolizumab + Bevacizumab versus Sunitinib in Treatment-Naïve Metastatic Renal Cell Carcinoma. Clin. Cancer Res. 2020, 26, 2506–2514. [Google Scholar] [PubMed]
- Sanders, G.D.; Neumann, P.J.; Basu, A.; Brock, D.W.; Feeny, D.; Krahn, M.; Kuntz, K.M.; Meltzer, D.O.; Owens, D.K.; Prosser, L.A.; et al. Recommendations for Conduct, Methodological Practices, and Reporting of Cost-effectiveness Analyses: Second Panel on Cost-Effectiveness in Health and Medicine. JAMA 2016, 316, 1093–1103. [Google Scholar] [CrossRef]
- May, P.; Normand, C.; Cassel, J.B.; Del Fabbro, E.; Fine, R.L.; Menz, R.; Morrison, C.A.; Penrod, J.D.; Robinson, C.; Morrison, R.S. Economics of palliative care for hospitalized adults with serious illness: A meta-analysis. JAMA Intern. Med. 2018, 178, 820–829. [Google Scholar] [CrossRef] [PubMed]
- Academy of Managed Care Pharmacy. Guide to Pharmaceutical Payment Methods; Academy of Managed Care Pharmacy: Alexandria, VA, USA, 2019. [Google Scholar]
- Centers for Medicare & Medicaid Services: 2020 ASP Drug Pricing Files. Available online: https://www.cms.gov/medicare/medicare-part-b-drug-average-sales-price/2020-asp-drug-pricing-files (accessed on 31 October 2020).
- U.S. Department of Veterans Affairs. Office of Procurement, Acquisition and Logistics (OPAL) Price Data; Department of Veterans Affairs: Lanham, MD, USA, 2020.
- U.S. Centers for Medicare & Medicaid Services. Physician Fee Schedule: CY 2020 Physician Fee Schedule Final Rule. Available online: https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/PhysicianFeeSched/ (accessed on 31 October 2020).
- Wong, W.; Yim, Y.M.; Kim, A.; Cloutier, M.; Gauthier-Loiselle, M.; Gagnon-Sanschagrin, P.; Guerin, A. Assessment of costs associated with adverse events in patients with cancer. PLoS ONE 2018, 13, e0196007. [Google Scholar] [CrossRef]
- Goldstein, D.A.; Ahmad, B.B.; Chen, Q.; Ayer, T.; Howard, D.H.; Lipscomb, J.; El-Rayes, B.F.; Flowers, C.R. Cost-Effectiveness Analysis of Regorafenib for Metastatic Colorectal Cancer. J. Clin. Oncol. 2015, 33, 3727–3732. [Google Scholar] [CrossRef] [PubMed]
- Criss, S.D.; Mooradian, M.J.; Watson, T.R.; Gainor, J.F.; Reynolds, K.L.; Kong, C.Y. Cost-effectiveness of Atezolizumab Combination Therapy for First-Line Treat-ment of Metastatic Nonsquamous Non–Small Cell Lung Cancer in the United States. JAMA Netw. Open 2019, 2, e1911952. [Google Scholar] [CrossRef]
- Wan, X.; Luo, X.; Tan, C.; Zeng, X.; Zhang, Y.; Peng, L. First-line atezolizumab in addition to bevacizumab plus chemotherapy for metastatic, nonsquamous non-small cell lung cancer: A United States-based cost-effectiveness analysis. Cancer 2019, 125, 3526–3534. [Google Scholar] [CrossRef]
- Wan, X.; Zhang, Y.; Tan, C.; Zeng, X.; Peng, L. First-line Nivolumab Plus Ipilimumab vs Sunitinib for Metastatic Renal Cell Carcinoma: A Cost-effectiveness Analysis. JAMA Oncol. 2019, 5, 491–496. [Google Scholar] [CrossRef] [PubMed]
- Li, C.; Zeliadt, S.B.; Hall, I.J.; Smith, J.L.; Ekwueme, D.U.; Moinpour, C.M.; Penson, D.F.; Thompson, I.M.; Keane, T.E.; Ramsey, S.D. Burden among partner caregivers of patients diagnosed with localized prostate cancer within 1 year after diagnosis: An economic perspective. Support Care Cancer 2013, 21, 3461–3469. [Google Scholar] [CrossRef]
- Hopkins, R.B.; Goeree, R.; Longo, C.J. Estimating the national wage loss from cancer in Canada. Curr. Oncol. 2010, 17, 40–49. [Google Scholar] [CrossRef] [PubMed][Green Version]
- De Almeida, J.R.; Moskowitz, A.J.; Miles, B.A.; Goldstein, D.P.; Teng, M.S.; Sikora, A.G.; Gupta, V.; Posner, M.; Genden, E.M. Cost-effectiveness of transoral robotic surgery versus (chemo)radiotherapy for early T classification oropharyngeal carcinoma: A cost-utility analysis. Head Neck 2016, 38, 589–600. [Google Scholar] [CrossRef] [PubMed]
- Spigel, D.R.; McLeod, M.; Hussein, M.A.; Waterhouse, D.M.; Einhorn, L.; Horn, L.; Creelan, B.; Babu, S.; Leighl, N.B.; Couture, F.; et al. Randomized results of fixed-duration (1-yr) vs continuous nivolumab in patients (pts) with advanced non-small cell lung cancer (NSCLC). Ann. Oncol. 2017, 28 (Suppl. S5), v461. [Google Scholar] [CrossRef]
- Hou, Y.; Wu, B. Atezolizumab plus bevacizumab versus sorafenib as first-line treatment for unresectable hepatocellular carcinoma: A cost-effectiveness analysis. Cancer Commun. 2020, 40, 743–745. [Google Scholar] [CrossRef]
- Dubois de Gennes, C.; Mazaleyrat, B.; Sanchez Alvarez, J.; Cawston, H. Preliminary Results of a Cost Effectiveness MODEL of Atezolizumab PLUS Bevacizumab in Unresectable Hepatocellular Carcinoma (HCC) in France. Value Health 2020, 23 (Suppl. S2), S436. [Google Scholar] [CrossRef]
- Jansen, Y.J.L.; Rozeman, E.A.; Mason, R.; Goldinger, S.M.; Foppen, M.G.; Hoejberg, L.; Schmidt, H.; Van Thienen, J.V.; Haanen, J.B.A.G.; Tiainen, L.; et al. Discontinuation of anti-PD-1 antibody therapy in the absence of disease progres-sion or treatment limiting toxicity: Clinical outcomes in advanced melanoma. Ann. Oncol. 2019, 30, 1154–1161. [Google Scholar] [CrossRef] [PubMed]
- Falk, A.T.; Barrière, J.; François, E.; Follana, P. Bevacizumab: A dose review. Crit. Rev. Oncol. Hematol. 2015, 94, 311–322. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.J.; McFarlane, T.; Tully, S.; Wong, W.W.L. Lenvatinib versus Sorafenib as First-Line Treatment of Unresectable Hepatocellular Carcinoma: A Cost-Utility Analysis. Oncologist 2020, 25, e512–e519. [Google Scholar] [CrossRef] [PubMed]
- Finn, R.S.; Qin, S.; Ikeda, M.; Galle, P.R.; Ducreux, M.; Kim, T.Y.; Lim, H.Y.; Kudo, M.; Breder, V.V.; Merle, P.; et al. IMbrave150: Updated overall survival (OS) data from a global, randomized, open-label phase III study of atezolizumab (atezo) + bevacizumab (bev) vs sorafenib (sor) in patients (pts) with unresectable hepatocellular carcinoma (HCC). J. Clin. Oncol. 2021, 39 (Suppl. S3), 267. [Google Scholar] [CrossRef]
| Parameters | Base Case Value | Range | Reference | Distribution | |
|---|---|---|---|---|---|
| Minimum | Maximum | ||||
| Clinical effectiveness [15] | |||||
| HR for OS (atezo + bev vs.sorafenib) | 0.58 | 0.42 | 0.79 | [15] | Normal |
| HR for PFS (atezo + bev vs. sorafenib) | 0.59 | 0.47 | 0.76 | [15] | Normal |
| Weibull OS model with sorafenib | λ = 0.027, γ = 1.286 | - | - | [15] | - |
| Weibull PFS model with sorafenib | λ = 0.093, γ = 1.312 | - | - | [15] | - |
| Background mortality rate | Age-specific | ||||
| Rate of treatment discontinuation due to AEs [15] | |||||
| Atezo + Bev | 0.07 | 0.056 | 0.084 | [15] | Binomial |
| Sorafenib | 0.10 | 0.08 | 0.12 | [15] | Binomial |
| Proportion of patients with grade 3–4 AEs [15] | |||||
| Atezo + Bev | |||||
| Diarrhea | 0.018 | 0.014 | 0.022 | [15] | Binomial |
| Hand–foot syndrome | 0.0 | 0.0 | 0.0 | [15] | Binomial |
| Hypertension | 0.152 | 0.122 | 0.182 | [15] | Binomial |
| Increased bilirubin | 0.024 | 0.019 | 0.029 | [15] | Binomial |
| Sorafenib | |||||
| Diarrhea | 0.051 | 0.041 | 0.061 | [15] | Binomial |
| Hand–foot syndrome | 0.083 | 0.066 | 0.1 | [15] | Binomial |
| Hypertension | 0.122 | 0.098 | 0.146 | [15] | Binomial |
| Increased bilirubin | 0.064 | 0.051 | 0.077 | [15] | Binomial |
| Patient weight, kg | 70 | 40 | 200 | [42] | Triangular |
| Cost parameters, USD * | |||||
| Atezolizumab (1200 mg) (fixed) (every 3 weeks) | 9419.16 | 7535.33 | 11,302.99 | [4,5] | Triangular |
| Bevacizumab (15 mg/kg) (every 3 weeks) | 117.60 | 94.08 | 141.12 | [34,35,36] | Triangular |
| Sorafenib (every 3 weeks) | 14,609.28 | 11,687.42 | 17,531.14 | [34,35,36] | Triangular |
| Drug administration (every 3 weeks) | 435.04 | 348.03 | 522.05 | [34,35,36] | Triangular |
| CT imaging (every 6 weeks) | 1543 | 1235 | 1852 | [35] | Triangular |
| Other care (every week) | 174.5 | 139.6 | 209.4 | [20] | Triangular |
| In-patient EOL care | 7360.16 | 6208.85 | 9313.77 | [31] | Triangular |
| Post-progression therapy cost per cycle in atezo + bev | 4612.66 | 3690.13 | 5535.20 | [15] | Triangular |
| Post-progression therapy cost per cycle in sorafenib | 4825.93 | 3860.74 | 5791.11 | [15] | Triangular |
| Societal costs, USD * | |||||
| Caregiver (every 3 weeks) | 382.21 | 305.77 | 458.65 | [43] | Triangular |
| Patient time | 896.2 | 716.96 | 1075.44 | [43,44] | Triangular |
| Parking/meals/travel | 329.5 | 263.6 | 395.4 | [43,45] | Triangular |
| Management of Grade 3–4 AEs, USD * | |||||
| Diarrhea | 88.38 | 70.70 | 106.05 | [38,39,40,41] | Triangular |
| Hand–foot syndrome | 145.65 | 116.51 | 174.78 | [38,39,40,41] | Triangular |
| Hypertension | 64.01 | 51.21 | 76.81 | [38,39,40,41] | Triangular |
| Increased bilirubin | 0 | 0 | 0 | Estimates | Triangular |
| Utilities and dis-utilities | |||||
| In atezo–bev first-line therapy | 0.78 | 0.624 | 0.936 | [31] | Triangular |
| In sorafenib first-line therapy | 0.78 | 0.624 | 0.936 | [21] | Triangular |
| In second-line therapy | 0.68 | 0.54 | 0.82 | [21] | Triangular |
| Dis-utilities | |||||
| Diarrhea | −0.103 | −0.082 | −0.123 | [46] | Triangular |
| Hand–foot syndrome | −0.116 | −0.093 | −0.139 | [46] | Triangular |
| Hypertension | −0 | −0 | −0 | [46] | Triangular |
| Increased bilirubin | −0 | −0 | −0 | [46] | Triangular |
| Parameters | Atevo + Bev | Sorafenib |
|---|---|---|
| Treatment duration, months | Atezo: 7.4; Bev: 6.9 | 2.8 |
| Cost, USD | ||
| Total cost | 713,742 | 634,668 |
| Clinical effectiveness, months | ||
| Median PFS | 6.8 | 4.3 |
| Median OS | NE | 13.2 |
| Median post-progression survival | NE | 8.9 |
| Utilities | ||
| Utility while on first-line treatment | 0.78 | 0.78 |
| AEs | −0.013 | −0.013 |
| Utility at progression | 0.68 | 0.68 |
| QALYs | 1.426 | 0.987 |
| QALY gain | 0.440 | |
| Life-years | 2.02 | 1.51 |
| Incremental life-year | 0.51 | |
| ICER, USD | ||
| Per life-year | 155,047 (142,953–174,815) * | |
| Per QALY | 179,729 (163,932–191,054) * | |
| Cost-Effectiveness Acceptability at WTP, % | ||||||
|---|---|---|---|---|---|---|
| Scenario | Life-Years | QALYs | Cost, 2020 US USD | ICER, USD/QALY | USD 100,000/ QALY | USD 150,000/ QALY |
| Atezo–bev | ||||||
| Base case * | 2.02 | 1.426 | 713,742 | 179,729 | 24.5 | 45.6 |
| Pessimistic survival * | 1.78 | 1.227 | 727,557 | 385,857 | 4.3 | 9.7 |
| Optimistic survival * | 2.48 | 1.825 | 679,811 | 53,854 | 78.1 | 92.7 |
| Sorafenib | 1.51 | 0.987 | 634,668 | NA | NA | NA |
| Parameters | ICER (USD /QALY) |
|---|---|
| Base case analysis | 179,729 |
| Model Perspective | |
| Third-party perspective (base case) | 179,729 |
| Societal perspective | 219,058 |
| Scenario analysis | |
| Duration of atezo–bev | |
| Until disease progression (base case) | 179,729 |
| Max 24 months | 176,626 |
| Max 18 months | 167,058 |
| Max 12 months | 136,205 |
| Max 6 months | 18,598 |
| Dosage of bev (assuming a body weight of 70 kg) | |
| 15 mg/kg (base case) | 179,729 |
| 12.5 mg/kg | 167,405 |
| 10 mg/kg | 122,286 |
| 7.5 mg/kg | 107,969 |
| 5 mg/kg | 81,952 |
| Time horizon | |
| 5 years (base case) | 179,729 |
| 10 years | 156,988 |
| Lifetime | 156,710 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chiang, C.-L.; Chan, S.-K.; Lee, S.-F.; Choi, H.C.-W. First-Line Atezolizumab Plus Bevacizumab versus Sorafenib in Hepatocellular Carcinoma: A Cost-Effectiveness Analysis. Cancers 2021, 13, 931. https://doi.org/10.3390/cancers13050931
Chiang C-L, Chan S-K, Lee S-F, Choi HC-W. First-Line Atezolizumab Plus Bevacizumab versus Sorafenib in Hepatocellular Carcinoma: A Cost-Effectiveness Analysis. Cancers. 2021; 13(5):931. https://doi.org/10.3390/cancers13050931
Chicago/Turabian StyleChiang, Chi-Leung, Sik-Kwan Chan, Shing-Fung Lee, and Horace Cheuk-Wai Choi. 2021. "First-Line Atezolizumab Plus Bevacizumab versus Sorafenib in Hepatocellular Carcinoma: A Cost-Effectiveness Analysis" Cancers 13, no. 5: 931. https://doi.org/10.3390/cancers13050931
APA StyleChiang, C.-L., Chan, S.-K., Lee, S.-F., & Choi, H. C.-W. (2021). First-Line Atezolizumab Plus Bevacizumab versus Sorafenib in Hepatocellular Carcinoma: A Cost-Effectiveness Analysis. Cancers, 13(5), 931. https://doi.org/10.3390/cancers13050931






