Prognostic and Predictive Value of Integrated Qualitative and Quantitative Magnetic Resonance Imaging Analysis in Glioblastoma
Abstract
Simple Summary
Abstract
1. Introduction
2. Results
2.1. Patient and Tumor Characteristics
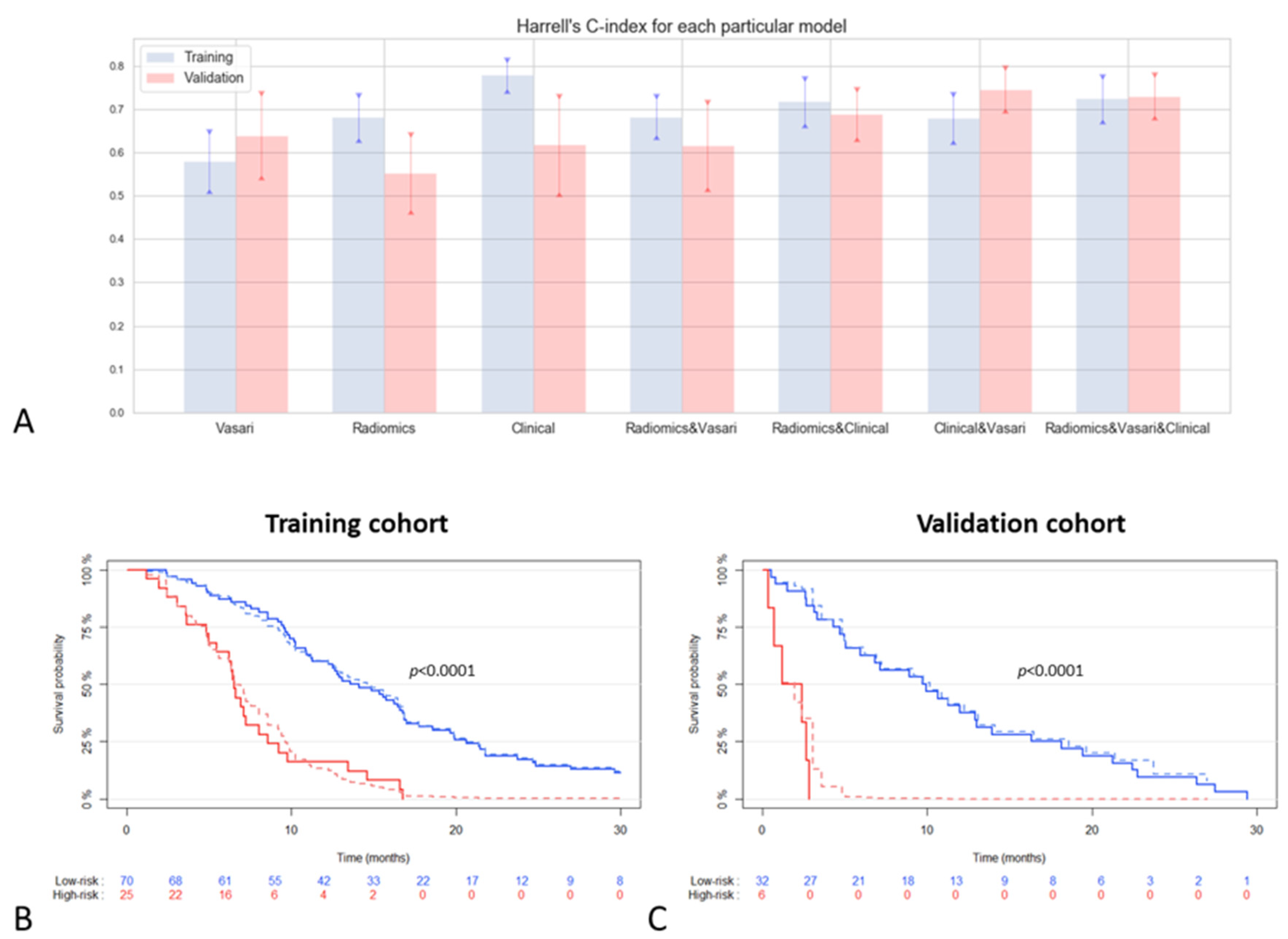
2.2. Prognostic Value of Integrative MRI Imaging Analysis in IDH-Wild Type GBM Population
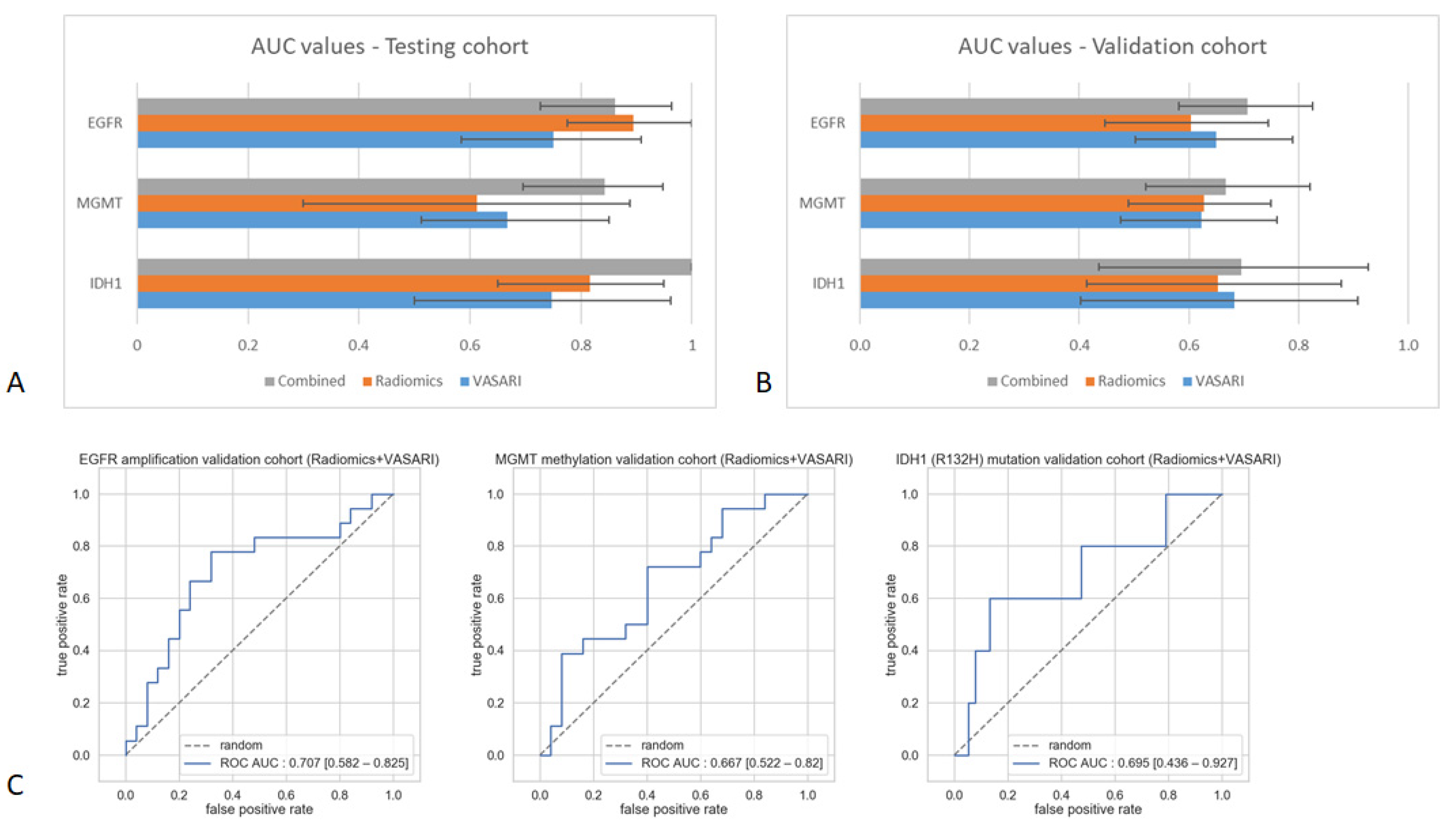
2.3. Predictive Value of Integrative Imaging Analysis
2.4. Transparent Reporting of a Multivariable Prediction Model for Individual Prognosis or Diagnosis (TRIPOD) Statement and Radiomics Quality Score
3. Discussion
4. Materials and Methods
4.1. Patient Population
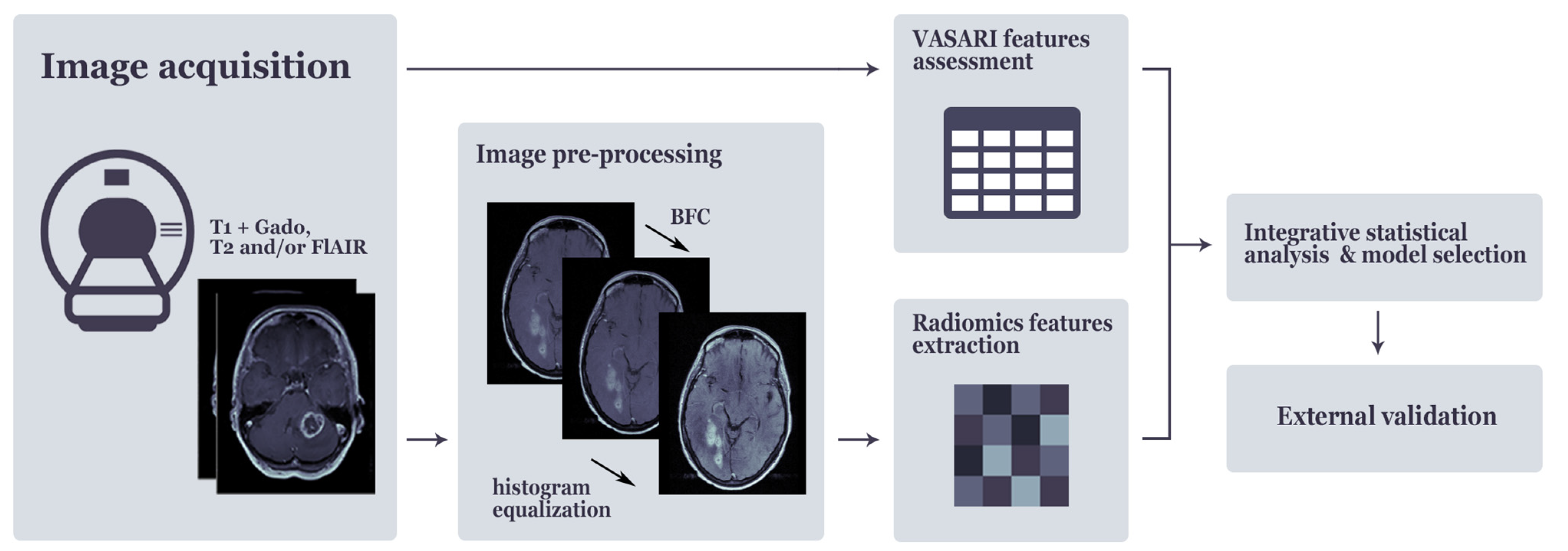
4.2. Image Acquisition and Qualitative Imaging Feature Assessment
4.3. Tumor Delineation, Image Pre-Processing and Extraction of Radiomics Features
4.4. Molecular Markers
4.5. Statistical Analysis
4.6. TRIPOD Statement and Radiomics Quality Score (RQS)
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Urbanska, K.; Sokolowska, J.; Szmidt, M.; Sysa, P. Glioblastoma multiforme—An overview. Contemp. Oncol. (Pozn) 2014, 18, 307–312. [Google Scholar] [CrossRef] [PubMed]
- Stupp, R.; Mason, W.P.; van den Bent, M.J.; Weller, M.; Fisher, B.; Taphoorn, M.J.; Belanger, K.; Brandes, A.A.; Marosi, C.; Bogdahn, U.; et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N. Engl. J. Med. 2005, 352, 987–996. [Google Scholar] [CrossRef]
- Wen, P.Y.; Weller, M.; Lee, E.Q.; Alexander, B.M.; Barnholtz-Sloan, J.S.; Barthel, F.P.; Batchelor, T.T.; Bindra, R.S.; Chang, S.M.; Chiocca, E.A.; et al. Glioblastoma in adults: A Society for Neuro-Oncology (SNO) and European Society of Neuro-Oncology (EANO) consensus review on current management and future directions. Neuro Oncol. 2020, 22, 1073–1113. [Google Scholar] [CrossRef]
- Louis, D.N.; Perry, A.; Reifenberger, G.; von Deimling, A.; Figarella-Branger, D.; Cavenee, W.K.; Ohgaki, H.; Wiestler, O.D.; Kleihues, P.; Ellison, D.W. The 2016 World Health Organization Classification of Tumors of the Central Nervous System: A summary. Acta Neuropathol. 2016, 131, 803–820. [Google Scholar] [CrossRef]
- Weller, M.; van den Bent, M.; Preusser, M.; Le Rhun, E.; Tonn, J.C.; Minniti, G.; Bendszus, M.; Balana, C.; Chinot, O.; Dirven, L.; et al. EANO guidelines on the diagnosis and treatment of diffuse gliomas of adulthood. Nat. Rev. Clin. Oncol. 2020. [Google Scholar] [CrossRef]
- Hegi, M.E.; Diserens, A.C.; Gorlia, T.; Hamou, M.F.; de Tribolet, N.; Weller, M.; Kros, J.M.; Hainfellner, J.A.; Mason, W.; Mariani, L.; et al. MGMT gene silencing and benefit from temozolomide in glioblastoma. N. Engl. J. Med. 2005, 352, 997–1003. [Google Scholar] [CrossRef]
- Hegi, M.E.; Genbrugge, E.; Gorlia, T.; Stupp, R.; Gilbert, M.R.; Chinot, O.L.; Nabors, L.B.; Jones, G.; Van Criekinge, W.; Straub, J.; et al. MGMT Promoter Methylation Cutoff with Safety Margin for Selecting Glioblastoma Patients into Trials Omitting Temozolomide: A Pooled Analysis of Four Clinical Trials. Clin. Cancer Res. 2019, 25, 1809–1816. [Google Scholar] [CrossRef]
- Dahlrot, R.H.; Kristensen, B.W.; Hjelmborg, J.; Herrstedt, J.; Hansen, S. A population-based study of high-grade gliomas and mutated isocitrate dehydrogenase 1. Int. J. Clin. Exp. Pathol. 2013, 6, 31–40. [Google Scholar]
- Lassman, A.B.; Aldape, K.D.; Ansell, P.J.; Bain, E.; Curran, W.J.; Eoli, M.; French, P.J.; Kinoshita, M.; Looman, J.; Mehta, M.; et al. Epidermal growth factor receptor (EGFR) amplification rates observed in screening patients for randomized trials in glioblastoma. J. Neuro Oncol. 2019, 144, 205–210. [Google Scholar] [CrossRef]
- Eskilsson, E.; Rosland, G.V.; Solecki, G.; Wang, Q.; Harter, P.N.; Graziani, G.; Verhaak, R.G.W.; Winkler, F.; Bjerkvig, R.; Miletic, H. EGFR heterogeneity and implications for therapeutic intervention in glioblastoma. Neuro Oncol. 2018, 20, 743–752. [Google Scholar] [CrossRef]
- Saadeh, F.S.; Mahfouz, R.; Assi, H.I. EGFR as a clinical marker in glioblastomas and other gliomas. Int. J. Biol. Mark. 2018, 33, 22–32. [Google Scholar] [CrossRef] [PubMed]
- Armocida, D.; Pesce, A.; Frati, A.; Santoro, A.; Salvati, M. EGFR amplification is a real independent prognostic impact factor between young adults and adults over 45yo with wild-type glioblastoma? J. Neuro Oncol. 2020, 146, 275–284. [Google Scholar] [CrossRef]
- Hoffman, D.I.; Abdullah, K.G.; McCoskey, M.; Binder, Z.A.; O’Rourke, D.M.; Desai, A.S.; Nasrallah, M.P.; Bigdeli, A.; Morrissette, J.J.D.; Brem, S.; et al. Negative prognostic impact of epidermal growth factor receptor copy number gain in young adults with isocitrate dehydrogenase wild-type glioblastoma. J. Neuro Oncol. 2019, 145, 321–328. [Google Scholar] [CrossRef]
- Patel, A.P.; Tirosh, I.; Trombetta, J.J.; Shalek, A.K.; Gillespie, S.M.; Wakimoto, H.; Cahill, D.P.; Nahed, B.V.; Curry, W.T.; Martuza, R.L.; et al. Single-cell RNA-seq highlights intratumoral heterogeneity in primary glioblastoma. Science 2014, 344, 1396–1401. [Google Scholar] [CrossRef]
- Qazi, M.A.; Vora, P.; Venugopal, C.; Sidhu, S.S.; Moffat, J.; Swanton, C.; Singh, S.K. Intratumoral heterogeneity: Pathways to treatment resistance and relapse in human glioblastoma. Ann. Oncol. 2017, 28, 1448–1456. [Google Scholar] [CrossRef]
- Draaisma, K.; Chatzipli, A.; Taphoorn, M.; Kerkhof, M.; Weyerbrock, A.; Sanson, M.; Hoeben, A.; Lukacova, S.; Lombardi, G.; Leenstra, S.; et al. Molecular Evolution of IDH Wild-Type Glioblastomas Treated With Standard of Care Affects Survival and Design of Precision Medicine Trials: A Report From the EORTC 1542 Study. J. Clin. Oncol. 2020, 38, 81–99. [Google Scholar] [CrossRef]
- Wangaryattawanich, P.; Hatami, M.; Wang, J.; Thomas, G.; Flanders, A.; Kirby, J.; Wintermark, M.; Huang, E.S.; Bakhtiari, A.S.; Luedi, M.M.; et al. Multicenter imaging outcomes study of The Cancer Genome Atlas glioblastoma patient cohort: Imaging predictors of overall and progression-free survival. Neuro Oncol. 2015, 17, 1525–1537. [Google Scholar] [CrossRef]
- Aerts, H.J. The Potential of Radiomic-Based Phenotyping in Precision Medicine: A Review. JAMA Oncol. 2016, 2, 1636–1642. [Google Scholar] [CrossRef] [PubMed]
- Lambin, P.; Leijenaar, R.T.H.; Deist, T.M.; Peerlings, J.; de Jong, E.E.C.; van Timmeren, J.; Sanduleanu, S.; Larue, R.; Even, A.J.G.; Jochems, A.; et al. Radiomics: The bridge between medical imaging and personalized medicine. Nat. Rev. Clin. Oncol. 2017, 14, 749–762. [Google Scholar] [CrossRef]
- Rogers, W.; Thulasi Seetha, S.; Refaee, T.A.G.; Lieverse, R.I.Y.; Granzier, R.W.Y.; Ibrahim, A.; Keek, S.A.; Sanduleanu, S.; Primakov, S.P.; Beuque, M.P.L.; et al. Radiomics: From qualitative to quantitative imaging. Br. J. Radiol. 2020, 93, 20190948. [Google Scholar] [CrossRef]
- Chaddad, A.; Kucharczyk, M.J.; Daniel, P.; Sabri, S.; Jean-Claude, B.J.; Niazi, T.; Abdulkarim, B. Radiomics in Glioblastoma: Current Status and Challenges Facing Clinical Implementation. Front. Oncol. 2019, 9, 374. [Google Scholar] [CrossRef]
- Peeken, J.C.; Molina-Romero, M.; Diehl, C.; Menze, B.H.; Straube, C.; Meyer, B.; Zimmer, C.; Wiestler, B.; Combs, S.E. Deep learning derived tumor infiltration maps for personalized target definition in Glioblastoma radiotherapy. Radiother. Oncol. 2019, 138, 166–172. [Google Scholar] [CrossRef] [PubMed]
- Grossmann, P.; Stringfield, O.; El-Hachem, N.; Bui, M.M.; Rios Velazquez, E.; Parmar, C.; Leijenaar, R.T.; Haibe-Kains, B.; Lambin, P.; Gillies, R.J.; et al. Defining the biological basis of radiomic phenotypes in lung cancer. Elife 2017, 6. [Google Scholar] [CrossRef] [PubMed]
- Su, C.; Jiang, J.; Zhang, S.; Shi, J.; Xu, K.; Shen, N.; Zhang, J.; Li, L.; Zhao, L.; Zhang, J.; et al. Radiomics based on multicontrast MRI can precisely differentiate among glioma subtypes and predict tumour-proliferative behaviour. Eur. Radiol. 2019, 29, 1986–1996. [Google Scholar] [CrossRef]
- Park, J.E.; Kim, H.S.; Jo, Y.; Yoo, R.E.; Choi, S.H.; Nam, S.J.; Kim, J.H. Radiomics prognostication model in glioblastoma using diffusion- and perfusion-weighted MRI. Sci. Rep. 2020, 10, 4250. [Google Scholar] [CrossRef]
- Macyszyn, L.; Akbari, H.; Pisapia, J.M.; Da, X.; Attiah, M.; Pigrish, V.; Bi, Y.; Pal, S.; Davuluri, R.V.; Roccograndi, L.; et al. Imaging patterns predict patient survival and molecular subtype in glioblastoma via machine learning techniques. Neuro Oncol. 2016, 18, 417–425. [Google Scholar] [CrossRef]
- Aquilanti, E.; Miller, J.; Santagata, S.; Cahill, D.P.; Brastianos, P.K. Updates in prognostic markers for gliomas. Neuro Oncol. 2018, 20, vii17–vii26. [Google Scholar] [CrossRef]
- European Society of, R. ESR Statement on the Validation of Imaging Biomarkers. Insights Imaging 2020, 11, 76. [Google Scholar] [CrossRef]
- Radiomics Features. Available online: https://pyradiomics.readthedocs.io/en/latest/features.html (accessed on 28 January 2021).
- Aerts, H.J.; Velazquez, E.R.; Leijenaar, R.T.; Parmar, C.; Grossmann, P.; Carvalho, S.; Bussink, J.; Monshouwer, R.; Haibe-Kains, B.; Rietveld, D.; et al. Decoding tumour phenotype by noninvasive imaging using a quantitative radiomics approach. Nat. Commun. 2014, 5, 4006. [Google Scholar] [CrossRef]
- Gittleman, H.; Lim, D.; Kattan, M.W.; Chakravarti, A.; Gilbert, M.R.; Lassman, A.B.; Lo, S.S.; Machtay, M.; Sloan, A.E.; Sulman, E.P.; et al. An independently validated nomogram for individualized estimation of survival among patients with newly diagnosed glioblastoma: NRG Oncology RTOG 0525 and 0825. Neuro Oncol. 2017, 19, 669–677. [Google Scholar] [CrossRef]
- Peeken, J.C.; Goldberg, T.; Pyka, T.; Bernhofer, M.; Wiestler, B.; Kessel, K.A.; Tafti, P.D.; Nusslin, F.; Braun, A.E.; Zimmer, C.; et al. Combining multimodal imaging and treatment features improves machine learning-based prognostic assessment in patients with glioblastoma multiforme. Cancer Med. 2019, 8, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Mazurowski, M.A.; Desjardins, A.; Malof, J.M. Imaging descriptors improve the predictive power of survival models for glioblastoma patients. Neuro Oncol. 2013, 15, 1389–1394. [Google Scholar] [CrossRef]
- Nicolasjilwan, M.; Hu, Y.; Yan, C.; Meerzaman, D.; Holder, C.A.; Gutman, D.; Jain, R.; Colen, R.; Rubin, D.L.; Zinn, P.O.; et al. Addition of MR imaging features and genetic biomarkers strengthens glioblastoma survival prediction in TCGA patients. J. Neuro Radiol. 2015, 42, 212–221. [Google Scholar] [CrossRef]
- Pope, W.B.; Sayre, J.; Perlina, A.; Villablanca, J.P.; Mischel, P.S.; Cloughesy, T.F. MR imaging correlates of survival in patients with high-grade gliomas. Ajnr. Am. J. Neuro Radiol. 2005, 26, 2466–2474. [Google Scholar]
- Schoenegger, K.; Oberndorfer, S.; Wuschitz, B.; Struhal, W.; Hainfellner, J.; Prayer, D.; Heinzl, H.; Lahrmann, H.; Marosi, C.; Grisold, W. Peritumoral edema on MRI at initial diagnosis: An independent prognostic factor for glioblastoma? Eur. J. Neurol. 2009, 16, 874–878. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.X.; Lin, G.S.; Lin, Z.X.; Zhang, J.D.; Liu, S.Y.; Zhou, C.F. Peritumoral edema shown by MRI predicts poor clinical outcome in glioblastoma. World J. Surg. Oncol. 2015, 13, 97. [Google Scholar] [CrossRef] [PubMed]
- Henker, C.; Kriesen, T.; Glass, A.; Schneider, B.; Piek, J. Volumetric quantification of glioblastoma: Experiences with different measurement techniques and impact on survival. J. Neuro Oncol. 2017, 135, 391–402. [Google Scholar] [CrossRef]
- Lemee, J.M.; Clavreul, A.; Menei, P. Intratumoral heterogeneity in glioblastoma: Don’t forget the peritumoral brain zone. Neuro Oncol. 2015, 17, 1322–1332. [Google Scholar] [CrossRef]
- Petrecca, K.; Guiot, M.C.; Panet-Raymond, V.; Souhami, L. Failure pattern following complete resection plus radiotherapy and temozolomide is at the resection margin in patients with glioblastoma. J. Neuro Oncol. 2013, 111, 19–23. [Google Scholar] [CrossRef] [PubMed]
- Prasanna, P.; Patel, J.; Partovi, S.; Madabhushi, A.; Tiwari, P. Radiomic features from the peritumoral brain parenchyma on treatment-naive multi-parametric MR imaging predict long versus short-term survival in glioblastoma multiforme: Preliminary findings. Eur. Radiol. 2017, 27, 4188–4197. [Google Scholar] [CrossRef]
- Shi, J.; Yang, S.; Wang, J.; Huang, S.; Yao, Y.; Zhang, S.; Zhu, W.; Shao, J. Analysis of heterogeneity of peritumoral T2 hyperintensity in patients with pretreatment glioblastoma: Prognostic value of MRI-based radiomics. Eur. J. Radiol. 2019, 120, 108642. [Google Scholar] [CrossRef]
- Leijenaar, R.T.; Carvalho, S.; Hoebers, F.J.; Aerts, H.J.; van Elmpt, W.J.; Huang, S.H.; Chan, B.; Waldron, J.N.; O′Sullivan, B.; Lambin, P. External validation of a prognostic CT-based radiomic signature in oropharyngeal squamous cell carcinoma. Acta Oncol. 2015, 54, 1423–1429. [Google Scholar] [CrossRef]
- Chen, X.; Fang, M.; Dong, D.; Liu, L.; Xu, X.; Wei, X.; Jiang, X.; Qin, L.; Liu, Z. Development and Validation of a MRI-Based Radiomics Prognostic Classifier in Patients with Primary Glioblastoma Multiforme. Acad. Radiol. 2019, 26, 1292–1300. [Google Scholar] [CrossRef]
- Kickingereder, P.; Neuberger, U.; Bonekamp, D.; Piechotta, P.L.; Gotz, M.; Wick, A.; Sill, M.; Kratz, A.; Shinohara, R.T.; Jones, D.T.W.; et al. Radiomic subtyping improves disease stratification beyond key molecular, clinical, and standard imaging characteristics in patients with glioblastoma. Neuro Oncol. 2018, 20, 848–857. [Google Scholar] [CrossRef]
- Kickingereder, P.; Burth, S.; Wick, A.; Gotz, M.; Eidel, O.; Schlemmer, H.P.; Maier-Hein, K.H.; Wick, W.; Bendszus, M.; Radbruch, A.; et al. Radiomic Profiling of Glioblastoma: Identifying an Imaging Predictor of Patient Survival with Improved Performance over Established Clinical and Radiologic Risk Models. Radiology 2016, 280, 880–889. [Google Scholar] [CrossRef]
- Choi, Y.; Nam, Y.; Jang, J.; Shin, N.Y.; Lee, Y.S.; Ahn, K.J.; Kim, B.S.; Park, J.S.; Jeon, S.S.; Hong, Y.G. Radiomics may increase the prognostic value for survival in glioblastoma patients when combined with conventional clinical and genetic prognostic models. Eur. Radiol. 2020. [Google Scholar] [CrossRef]
- Kickingereder, P.; Bonekamp, D.; Nowosielski, M.; Kratz, A.; Sill, M.; Burth, S.; Wick, A.; Eidel, O.; Schlemmer, H.P.; Radbruch, A.; et al. Radiogenomics of Glioblastoma: Machine Learning-based Classification of Molecular Characteristics by Using Multiparametric and Multiregional MR Imaging Features. Radiology 2016, 281, 907–918. [Google Scholar] [CrossRef]
- Gupta, A.; Young, R.J.; Shah, A.D.; Schweitzer, A.D.; Graber, J.J.; Shi, W.; Zhang, Z.; Huse, J.; Omuro, A.M. Pretreatment Dynamic Susceptibility Contrast MRI Perfusion in Glioblastoma: Prediction of EGFR Gene Amplification. Clin. Neuro Radiol. 2015, 25, 143–150. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.S.; Ning, S.; Eschbacher, J.M.; Baxter, L.C.; Gaw, N.; Ranjbar, S.; Plasencia, J.; Dueck, A.C.; Peng, S.; Smith, K.A.; et al. Radiogenomics to characterize regional genetic heterogeneity in glioblastoma. Neuro Oncol. 2017, 19, 128–137. [Google Scholar] [CrossRef]
- Han, Y.; Yan, L.F.; Wang, X.B.; Sun, Y.Z.; Zhang, X.; Liu, Z.C.; Nan, H.Y.; Hu, Y.C.; Yang, Y.; Zhang, J.; et al. Structural and advanced imaging in predicting MGMT promoter methylation of primary glioblastoma: A region of interest based analysis. BMC Cancer 2018, 18, 215. [Google Scholar] [CrossRef] [PubMed]
- Leather, T.; Jenkinson, M.D.; Das, K.; Poptani, H. Magnetic Resonance Spectroscopy for Detection of 2-Hydroxyglutarate as a Biomarker for IDH Mutation in Gliomas. Metabolites 2017, 7, 29. [Google Scholar] [CrossRef] [PubMed]
- Lohmann, P.; Lerche, C.; Bauer, E.K.; Steger, J.; Stoffels, G.; Blau, T.; Dunkl, V.; Kocher, M.; Viswanathan, S.; Filss, C.P.; et al. Predicting IDH genotype in gliomas using FET PET radiomics. Sci. Rep. 2018, 8, 13328. [Google Scholar] [CrossRef] [PubMed]
- Park, J.E.; Kim, H.S.; Kim, D.; Park, S.Y.; Kim, J.Y.; Cho, S.J.; Kim, J.H. A systematic review reporting quality of radiomics research in neuro-oncology: Toward clinical utility and quality improvement using high-dimensional imaging features. BMC Cancer 2020, 20, 29. [Google Scholar] [CrossRef] [PubMed]
- Peeken, J.C.; Hesse, J.; Haller, B.; Kessel, K.A.; Nusslin, F.; Combs, S.E. Semantic imaging features predict disease progression and survival in glioblastoma multiforme patients. Strahlenther Onkol. 2018, 194, 580–590. [Google Scholar] [CrossRef] [PubMed]
- Bae, S.; Choi, Y.S.; Ahn, S.S.; Chang, J.H.; Kang, S.G.; Kim, E.H.; Kim, S.H.; Lee, S.K. Radiomic MRI Phenotyping of Glioblastoma: Improving Survival Prediction. Radiology 2018, 289, 797–806. [Google Scholar] [CrossRef]
- Moradmand, H.; Aghamiri, S.M.R.; Ghaderi, R. Impact of image preprocessing methods on reproducibility of radiomic features in multimodal magnetic resonance imaging in glioblastoma. J. Appl. Clin. Med. Phys. 2020, 21, 179–190. [Google Scholar] [CrossRef] [PubMed]
- Shiri, I.; Hajianfar, G.; Sohrabi, A.; Abdollahi, H.; Shayesteh, S.P.; Geramifar, P.; Zaidi, H.; Oveisi, M.; Rahmim, A. Repeatability of Radiomic Features in Magnetic Resonance Imaging of Glioblastoma: Test-Retest and Image Registration Analyses. Med. Phys. 2020. [Google Scholar] [CrossRef]
- Um, H.; Tixier, F.; Bermudez, D.; Deasy, J.O.; Young, R.J.; Veeraraghavan, H. Impact of image preprocessing on the scanner dependence of multi-parametric MRI radiomic features and covariate shift in multi-institutional glioblastoma datasets. Phys. Med. Biol. 2019, 64, 165011. [Google Scholar] [CrossRef]
- Rai, R.; Holloway, L.C.; Brink, C.; Field, M.; Christiansen, R.L.; Sun, Y.; Barton, M.B.; Liney, G.P. Multicenter evaluation of MRI-based radiomic features: A phantom study. Med. Phys. 2020. [Google Scholar] [CrossRef]
- Niyazi, M.; Brada, M.; Chalmers, A.J.; Combs, S.E.; Erridge, S.C.; Fiorentino, A.; Grosu, A.L.; Lagerwaard, F.J.; Minniti, G.; Mirimanoff, R.O.; et al. ESTRO-ACROP guideline “target delineation of glioblastomas”. Radiother. Oncol. 2016, 118, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Pavic, M.; Bogowicz, M.; Wurms, X.; Glatz, S.; Finazzi, T.; Riesterer, O.; Roesch, J.; Rudofsky, L.; Friess, M.; Veit-Haibach, P.; et al. Influence of inter-observer delineation variability on radiomics stability in different tumor sites. Acta Oncol. 2018, 57, 1070–1074. [Google Scholar] [CrossRef]
- Chang, K.; Beers, A.L.; Bai, H.X.; Brown, J.M.; Ly, K.I.; Li, X.; Senders, J.T.; Kavouridis, V.K.; Boaro, A.; Su, C.; et al. Automatic assessment of glioma burden: A deep learning algorithm for fully automated volumetric and bidimensional measurement. Neuro Oncol. 2019, 21, 1412–1422. [Google Scholar] [CrossRef] [PubMed]
- Rios Velazquez, E.; Meier, R.; Dunn, W.D., Jr.; Alexander, B.; Wiest, R.; Bauer, S.; Gutman, D.A.; Reyes, M.; Aerts, H.J. Fully automatic GBM segmentation in the TCGA-GBM dataset: Prognosis and correlation with VASARI features. Sci. Rep. 2015, 5, 16822. [Google Scholar] [CrossRef] [PubMed]
- Chaddad, A.; Sabri, S.; Niazi, T.; Abdulkarim, B. Prediction of survival with multi-scale radiomic analysis in glioblastoma patients. Med. Biol. Eng. Comput. 2018, 56, 2287–2300. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.C.; Bai, H.; Sun, Q.; Zhao, Y.; Lv, Y.; Zhou, J.; Liang, C.; Chen, Y.; Liang, D.; Zheng, H. Multiregional radiomics profiling from multiparametric MRI: Identifying an imaging predictor of IDH1 mutation status in glioblastoma. Cancer Med. 2018, 7, 5999–6009. [Google Scholar] [CrossRef] [PubMed]
- Park, J.E.; Kim, H.S.; Kim, N.; Park, S.Y.; Kim, Y.H.; Kim, J.H. Spatiotemporal Heterogeneity in Multiparametric Physiologic MRI Are Associated with Patient Outcomes in IDH-wildtype Glioblastoma. Clin. Cancer Res. 2020. [Google Scholar] [CrossRef] [PubMed]
- Tustison, N.J.; Avants, B.B.; Cook, P.A.; Zheng, Y.; Egan, A.; Yushkevich, P.A.; Gee, J.C. N4ITK: Improved N3 bias correction. IEEE Trans. Med. Imaging 2010, 29, 1310–1320. [Google Scholar] [CrossRef]
- van der Walt, S.; Schonberger, J.L.; Nunez-Iglesias, J.; Boulogne, F.; Warner, J.D.; Yager, N.; Gouillart, E.; Yu, T. scikit-image: Image processing in Python. PeerJ 2014, 2, e453. [Google Scholar] [CrossRef] [PubMed]
- Kaur, H.; Rani, J. MRI brain image enhancement using Histogram Equalization techniques. In Proceedings of the 2016 International Conference on Wireless Communications, Signal Processing and Networking (WiSPNET), Chennai, India, 23–25 March 2016; pp. 770–773. [Google Scholar]
- Reinhold, J.C.; Dewey, B.E.; Carass, A.; Prince, J.L. Evaluating the Impact of Intensity Normalization on MR Image Synthesis. Proc. SPIE Int. Soc. Opt. Eng. 2019, 10949. [Google Scholar] [CrossRef]
- van Griethuysen, J.J.M.; Fedorov, A.; Parmar, C.; Hosny, A.; Aucoin, N.; Narayan, V.; Beets-Tan, R.G.H.; Fillion-Robin, J.C.; Pieper, S.; Aerts, H. Computational Radiomics System to Decode the Radiographic Phenotype. Cancer Res. 2017, 77, e104–e107. [Google Scholar] [CrossRef]
- de Leng, W.W.; Gadellaa-van Hooijdonk, C.G.; Barendregt-Smouter, F.A.; Koudijs, M.J.; Nijman, I.; Hinrichs, J.W.; Cuppen, E.; van Lieshout, S.; Loberg, R.D.; de Jonge, M.; et al. Targeted Next Generation Sequencing as a Reliable Diagnostic Assay for the Detection of Somatic Mutations in Tumours Using Minimal DNA Amounts from Formalin Fixed Paraffin Embedded Material. PLoS ONE 2016, 11, e0149405. [Google Scholar] [CrossRef]
- van Riet, J.; Krol, N.M.G.; Atmodimedjo, P.N.; Brosens, E.; van, I.W.F.J.; Jansen, M.; Martens, J.W.M.; Looijenga, L.H.; Jenster, G.; Dubbink, H.J.; et al. SNPitty: An Intuitive Web Application for Interactive B-Allele Frequency and Copy Number Visualization of Next-Generation Sequencing Data. J. Mol. Diagn 2018, 20, 166–176. [Google Scholar] [CrossRef] [PubMed]
- Jeuken, J.W.; Cornelissen, S.J.; Vriezen, M.; Dekkers, M.M.; Errami, A.; Sijben, A.; Boots-Sprenger, S.H.; Wesseling, P. MS-MLPA: An attractive alternative laboratory assay for robust, reliable, and semiquantitative detection of MGMT promoter hypermethylation in gliomas. Lab. Investig. 2007, 87, 1055–1065. [Google Scholar] [CrossRef]
- Mogensen, U.B.; Ishwaran, H.; Gerds, T.A. Evaluating Random Forests for Survival Analysis using Prediction Error Curves. J. Stat. Softw. 2012, 50, 1–23. [Google Scholar] [CrossRef] [PubMed]
- van Timmeren, J.E.; Leijenaar, R.T.H.; van Elmpt, W.; Reymen, B.; Oberije, C.; Monshouwer, R.; Bussink, J.; Brink, C.; Hansen, O.; Lambin, P. Survival prediction of non-small cell lung cancer patients using radiomics analyses of cone-beam CT images. Radiother. Oncol. 2017, 123, 363–369. [Google Scholar] [CrossRef] [PubMed]
- Royston, P.; Altman, D.G. External validation of a Cox prognostic model: Principles and methods. BMC Med. Res. Methodol. 2013, 13, 33. [Google Scholar] [CrossRef] [PubMed]
- Welch, M.L.; McIntosh, C.; Haibe-Kains, B.; Milosevic, M.F.; Wee, L.; Dekker, A.; Huang, S.H.; Purdie, T.G.; O’Sullivan, B.; Aerts, H.; et al. Vulnerabilities of radiomic signature development: The need for safeguards. Radiother. Oncol. 2019, 130, 2–9. [Google Scholar] [CrossRef] [PubMed]
- Sanduleanu, S.; Woodruff, H.C.; de Jong, E.E.C.; van Timmeren, J.E.; Jochems, A.; Dubois, L.; Lambin, P. Tracking tumor biology with radiomics: A systematic review utilizing a radiomics quality score. Radiother. Oncol. 2018, 127, 349–360. [Google Scholar] [CrossRef]
- Collins, G.S.; Reitsma, J.B.; Altman, D.G.; Moons, K.G. Transparent Reporting of a multivariable prediction model for Individual Prognosis or Diagnosis (TRIPOD): The TRIPOD statement. Ann. Intern. Med. 2015, 162, 55–63. [Google Scholar] [CrossRef]
| Demographics | Training Cohort (n = 142) | Validation Cohort (n = 46) | p-Value |
|---|---|---|---|
| Median age at diagnosis (range) | 61.4 years (15–85) | 61.7 years (18–81) | 0.991 |
| Sex (%) | Male: 85 (59.9%) Female: 57 (40.1%) | Male: 29 (63.0%) Female: 17 (37.0%) | 0.258 |
| Treatment characteristics | |||
| Surgical treatment (%) | Biopsy: 54 (38.0%) Debulking: 88 (62.0%) | Biopsy: 17 (37.0%) Debulking: 29 (63.0%) | 0.112 |
| Adjuvant treatment (%) | STUPP completed: 67 (47.2%) STUPP not completed or Non-STUPP regimen: 75 (52.8%) | STUPP completed: 17 (37.0%) STUPP not completed or Non-STUPP regimen: 16 (34.8%) Missing: 13 (28.2%) | 0.288 |
| Tumor characteristics | |||
| Isocitrate dehydrogenase (IDH1) (R132H) mutation status (%) | IDH1/2-WT: 129 (91.5%) IDH1-mutation: 12 (8.5%) Missing: 1 | IDH1/2-WT: 39 (84.8%) IDH1-mutation: 5 (10.9%) Missing: 2 | 1.000 |
| Methylguanine methyltransferase (MGMT)-methylation status (%) | MGMT-methylated: 37 (26.2%) MGMT non-methylated: 104 (73.8%) Missing: 1 | MGMT-methylated: 18 (39.1%) MGMT non-methylated: 26 (56.5%) Missing: 2 | 0.045 |
| Epidermal growth factor receptor (EGFR) amplification status (%) | EGFR amplified: 47 (37.3%) EGFR non-amplified: 79 (62.7%) Missing: 16 | EGFR amplified: 20 (43.5%) EGFR non-amplified: 26 (56.5%) Missing: 0 | 0.738 |
| Prognostic Model Variables | Hazard Ratio (95% CI) | p-Value |
|---|---|---|
| Model 1: VASARI features model (n = 129) | ||
| Involvement of eloquent cortex | 1.28 (0.88–1.87) | 0.198 |
| Multifocality | 1.72 (0.97–3.05) | 0.064 |
| Subependymal extension | 1.75 (1.21–2.53) | 0.003 |
| Low proportion of edema | Reference | Reference |
| Medium proportion of edema | 1.09 (0.75–1.61) | 0.653 |
| High proportion of edema | 0.45 (0.24–0.83) | 0.011 |
| Increased T1FLAIR-ratio | 0.59 (0.37–0.94) | 0.026 |
| Model 2: Radiomics features model (n = 95) | ||
| T1_wavelet.HHH_firstorder_Median | 1.04 (0.81–1.3) | 0.754 |
| T2_log.sigma.2.0.mm.3D_glszm_LargeAreaLowGrayLevelEmphasis | 1.00 (0.83–1.2) | 0.958 |
| T2_log.sigma.3.0.mm.3D_glszm_LargeAreaLowGrayLevelEmphasis | 1.33 (1.07–1.6) | 0.009 |
| T2_wavelet.LLH_firstorder_Mean | 1.70 (1.32–2.2) | 0.001 |
| T2_wavelet.HHL_glszm_LargeAreaLowGrayLevelEmphasis | 0.92 (0.75–1.1) | 0.404 |
| Model 3: Clinical features model (n = 95) | ||
| Sex (male vs. female) | 1.12 (0.70–1.77) | 0.644 |
| Type of surgery (resection vs. biopsy) | 0.48 (0.31–0.76)) | 0.002 |
| Age at diagnosis (>70 vs. <70) | 1.10 (0.60–2.02) | 0.749 |
| Adjuvant treatment (non-STUPP vs. STUPP) | 4.92 (2.79–8.67) | 0.001 |
| Methylguanine methyltransferase (MGMT)-methylation | 0.61 (0.35–1.06) | 0.082 |
| Model 4: Integrated imaging model (VASARI + radiomics) (n = 95) | ||
| VASARI prognostic score | 2.2 (1.4–3.4) | <0.001 |
| Radiomics prognostic score | 2.92 (1.9–4.5) | <0.001 |
| Model 5: Integrated VASARI and clinical model (n = 95) | ||
| VASARI prognostic score | 2.0 (1.3–3.2) | 0.003 |
| Clinical prognostic score | 2.7 (1.8–3.9) | <0.001 |
| Model 6: Integrated Radiomics and clinical model (n = 95) | ||
| Radiomics prognostic score | 2.6 (1.7–4.0) | <0.001 |
| Clinical prognostic score | 2.8 (1.9–4.1) | <0.001 |
| Model 7: Integrated imaging and clinical model (n = 95) | ||
| VASARI prognostic score | 2.1 (1.4–3.3) | <0.001 |
| Radiomics prognostic score | 3.0 (1.9–4.7) | <0.001 |
| Clinical prognostic score | 2.1 (1.4–3.3) | <0.001 |
| VASARI Features | Radiomics Features |
|---|---|
| EGFR amplification (n = 64) | |
| Size: Major Axis, Minor Axis, Mean Minor Axis, Median Major Axis Location: Eloquent location, Midline cross of enhancing tumor Morphology: Proportion necrosis Tumor characteristics: Hemorrhage, Subependymal Extension, Pial invasion, Definition enhancing margin | T1+Gado: wavelet-HLH_glcm_Correlation T2: log-sigma-2-0-mm-3D_gldm_LargeDependenceLowGrayLevelEmphasis; wavelet-LLH_glcm_ClusterShade; wavelet-LLH_firstorder_Skewness |
| MGMT-methylation (n = 74) | |
| Size: Major Axis, Minor Axis, Median Major Axis, Mean Major Axis Morphology: Proportion non-enhancing tumor Tumor characteristics: Deep white matter invasion, Subependymal extension | T1+Gado: no features selected T2: wavelet-HLL_gldm_LargeDependenceHighGrayLevelEmphasis; log-sigma-5-0-mm-3D_glrlm_HighGrayLevelRunEmphasis; log-sigma-5-0-mm-3D_glszm_SmallAreaHighGrayLevelEmphasis |
| IDH1 mutation (n = 72) | |
| Size: Minor Axis, Major Axis Location: Tumor side, Eloquent location Morphology: Proportion non-enhancing tumor, Proportion Edema Tumor characteristics: Pial invasion, Thickness Enhancing Margin, Definition enhancing margin, T1-FLAIR-ratio | T1+Gado: wavelet-HLL_glcm_Contrast; wavelet-HLL_glcm_DifferenceAverage T2: log-sigma-2-0-mm-3D_firstorder_90Percentile; Original_glrlm_LongRunHighGrayLevelEmphasis; log-sigma-3-0-mm-3D_firstorder_90Percentile; original_firstorder_10Percentile; log-sigma-4-0-mm-3D_firstorder_Uniformity; wavelet-HLL_gldm_DependenceEntropy; wavelet-HLL_glcm_Correlation |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Verduin, M.; Primakov, S.; Compter, I.; Woodruff, H.C.; van Kuijk, S.M.J.; Ramaekers, B.L.T.; te Dorsthorst, M.; Revenich, E.G.M.; ter Laan, M.; Pegge, S.A.H.; et al. Prognostic and Predictive Value of Integrated Qualitative and Quantitative Magnetic Resonance Imaging Analysis in Glioblastoma. Cancers 2021, 13, 722. https://doi.org/10.3390/cancers13040722
Verduin M, Primakov S, Compter I, Woodruff HC, van Kuijk SMJ, Ramaekers BLT, te Dorsthorst M, Revenich EGM, ter Laan M, Pegge SAH, et al. Prognostic and Predictive Value of Integrated Qualitative and Quantitative Magnetic Resonance Imaging Analysis in Glioblastoma. Cancers. 2021; 13(4):722. https://doi.org/10.3390/cancers13040722
Chicago/Turabian StyleVerduin, Maikel, Sergey Primakov, Inge Compter, Henry C. Woodruff, Sander M. J. van Kuijk, Bram L. T. Ramaekers, Maarten te Dorsthorst, Elles G. M. Revenich, Mark ter Laan, Sjoert A. H. Pegge, and et al. 2021. "Prognostic and Predictive Value of Integrated Qualitative and Quantitative Magnetic Resonance Imaging Analysis in Glioblastoma" Cancers 13, no. 4: 722. https://doi.org/10.3390/cancers13040722
APA StyleVerduin, M., Primakov, S., Compter, I., Woodruff, H. C., van Kuijk, S. M. J., Ramaekers, B. L. T., te Dorsthorst, M., Revenich, E. G. M., ter Laan, M., Pegge, S. A. H., Meijer, F. J. A., Beckervordersandforth, J., Speel, E. J., Kusters, B., de Leng, W. W. J., Anten, M. M., Broen, M. P. G., Ackermans, L., Schijns, O. E. M. G., ... Hoeben, A. (2021). Prognostic and Predictive Value of Integrated Qualitative and Quantitative Magnetic Resonance Imaging Analysis in Glioblastoma. Cancers, 13(4), 722. https://doi.org/10.3390/cancers13040722








