Enhance the Immune Checkpoint Inhibitors Efficacy with Radiotherapy Induced Immunogenic Cell Death: A Comprehensive Review and Latest Developments
Abstract
Simple Summary
Abstract
1. Introduction
2. The Biology of Radiotherapy-Induced ICD
2.1. ICD in Oncology
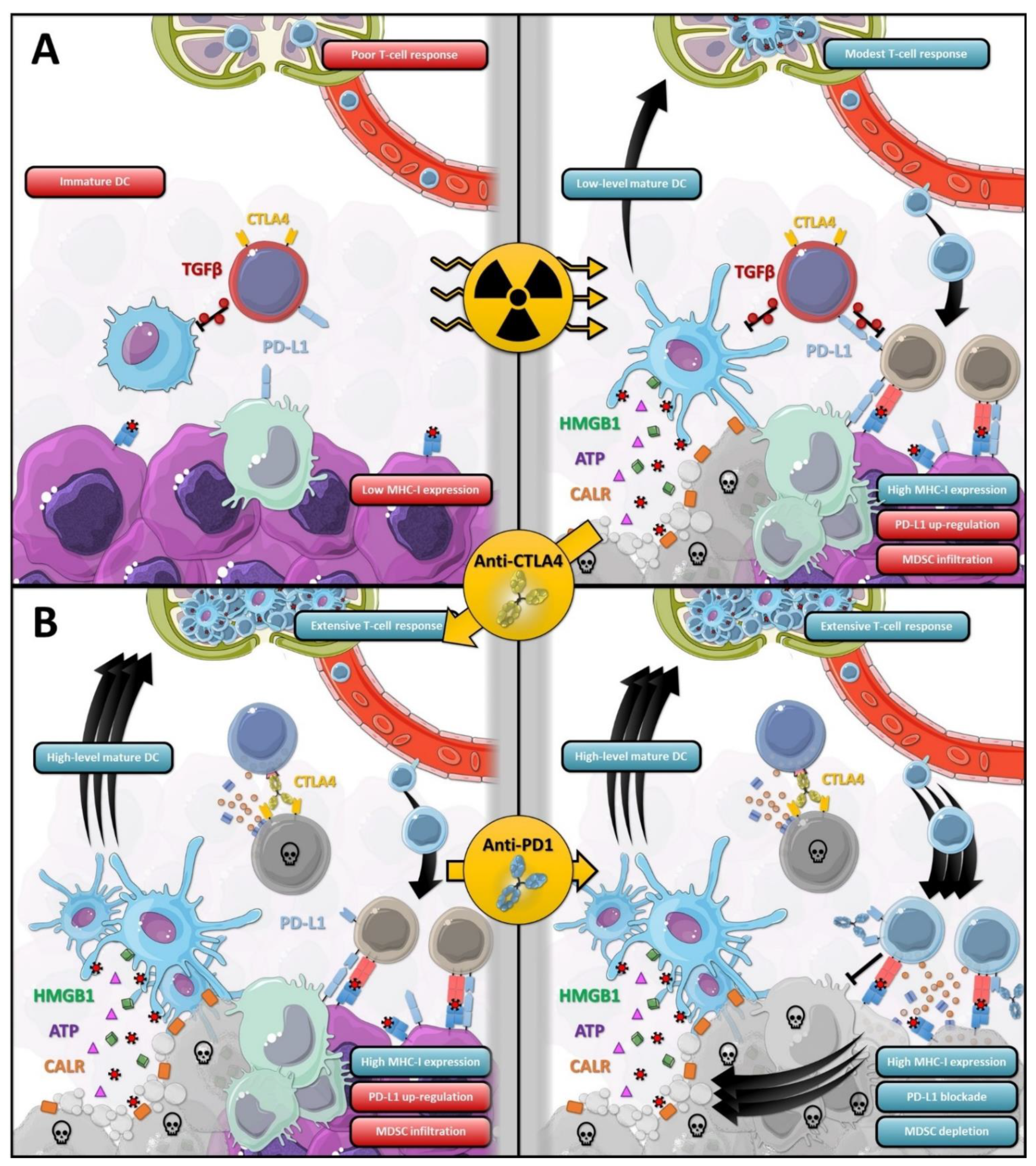
2.2. Radiotherapy as an ICD Inducer
2.3. Radiotherapy and ICI
3. RT and ICI Synergism: Emerging Clinical Evidence Related to Efficacy and Safety
3.1. Efficacy of Combined ICI and Radiation Therapy
3.1.1. Locally Advanced NSCLC (LA-NSCLC)
- Phase III—PACIFIC-4 (NCT03833154) evaluating durvalumab vs. placebo with Stereotactic Body Radiation Therapy (SBRT) in early-stage unresectable NSCLC (estimated enrollment: 706 patients).
- Phase II—KEYNOTE-799 (NCT03631784) evaluating pembrolizumab in combination with chemoradiotherapy in stage III NSCLC (enrollment: 216 patients).
- Phase II—NCT03663166 evaluating radiation and chemotherapy with ipilimumab followed by nivolumab for stage III unresectable NSCLC (estimated enrollment: 50 patients).
3.1.2. Metastatic NSCLC
3.1.3. Melanoma
3.1.4. Head and Neck
3.1.5. Other Malignancies
3.2. Safety of Combined ICI and Radiation Therapy: Lessons Learned from NSCLC
3.2.1. Sequential Schedule
3.2.2. Concomitant Schedule
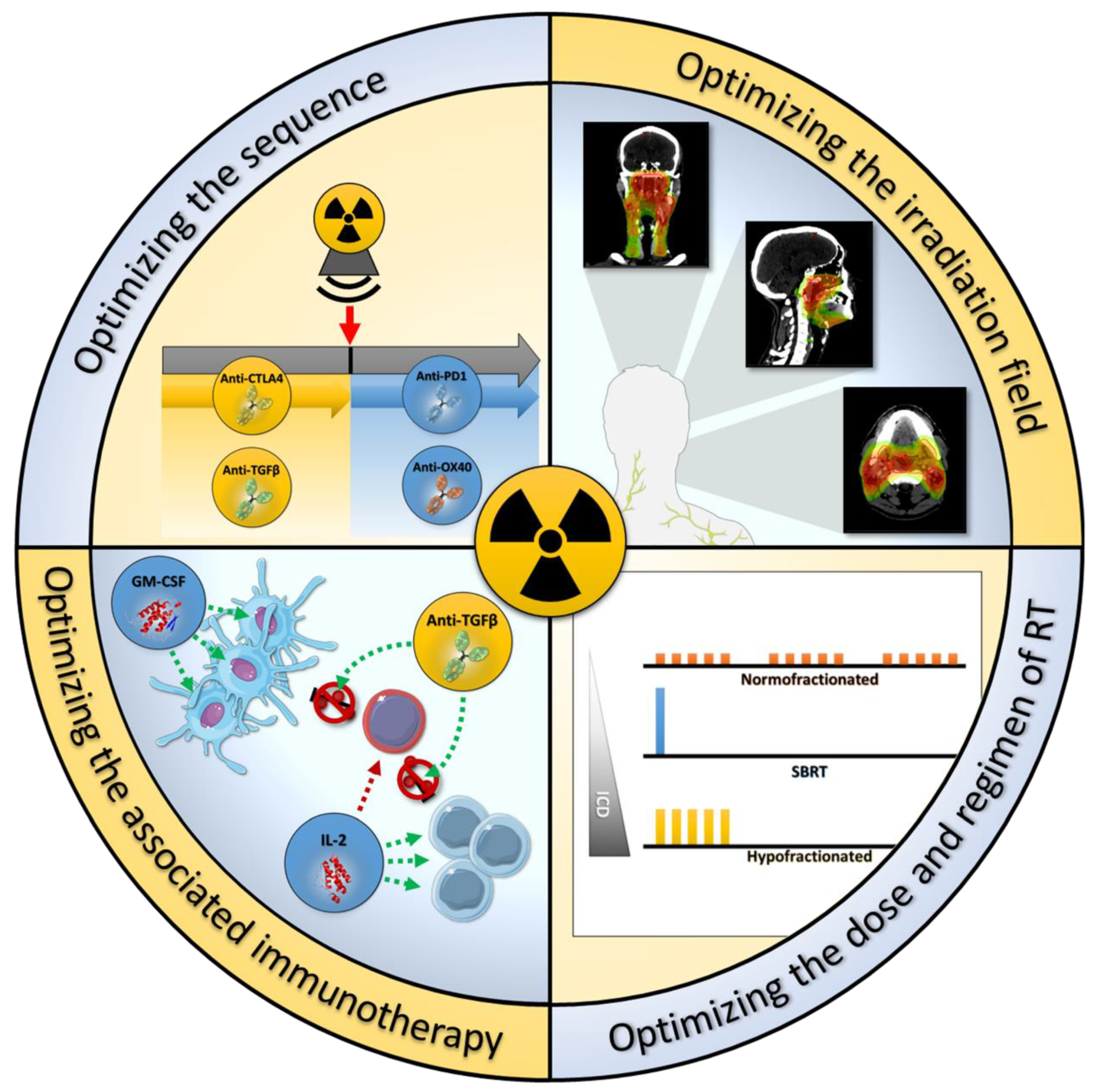
4. Increasing the RT and ICI Synergism: Schedule, ICI Partner and Sequence
4.1. Optimizing the Sequence and the Choice of ICI
4.2. Optimizing the Dose and Regimen of RT
4.3. Optimize the Irradiation Field
4.4. Combination of Radiotherapy and Other Immunotherapeutic Agents
4.4.1. GM-CSF (Granulocyte-Macrophage Colony-Stimulating Factor)
4.4.2. Interleukin-2 (IL2)
4.4.3. Anti-TGFß
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| ALC | Absolute Lymphocyte Count |
| APC | Antigen-Presenting Cell |
| ATP | Adenosine Triphosphate |
| BTLA | B and T Lymphocyte Attenuator |
| CALR | Calreticulin |
| CAR | Chimeric Antigen Receptor |
| CTLA4 | Cytotoxic T-Lymphocyte-Associated Protein |
| CRT | Chemoradiotherapy |
| DAMPs | Damage-Associated Molecular Patterns |
| DC | Dendritic Cell |
| HMGB1 | High Mobility Group Box 1 |
| HNSCC | Head and Neck Squamous Cell Carcinoma |
| HSP70/90 | Heat Shock Proteins 70/90 |
| ICD | Immunogenic Cell Death |
| ICI | Immune Checkpoint Inhibitor |
| irAEs | Immune-related Adverse Events |
| LA | Locally Advanced |
| LAG3 | Lymphocyte Activation Gene 3 |
| MDSC | Myeloid-Derived Suppressor Cell |
| MHC-1 | Major Histocompatibility Complex class 1 |
| NSCLC | Non-Small Cell Lung Carcinoma |
| OS | Overall Survival |
| PD-1 | Programmed cell death 1 |
| PD-L1 | Programmed death-ligand 1 |
| ROS | Reactive Oxygen Species |
| PFS | Progression Free Survival |
| RCC | Renal Cell Carcinoma |
| RT | Radiation Therapy |
| SBRT | Stereotactic Body Radiation Therapy |
| TGFβ | Transforming Growth Factor Beta |
| TIGIT | T cell Immunoglobulin and ITIM domain |
| TNFα | Tumor Necrosis Factor Alpha |
| Treg | Regulatory T cells |
| TREX1 | Three prime Repair Exonuclease 1 |
| TRAIL | TNF-related Apoptosis-Inducing Ligand |
| VISTA | V-Domain Ig Suppressor of T cell Activation |
References
- Dagenais, G.R.; Leong, D.P.; Rangarajan, S.; Lanas, F.; Lopez-Jaramillo, P.; Gupta, R.; Diaz, R.; Avezum, A.; Oliveira, G.B.F.; Wielgosz, A.; et al. Variations in Common Diseases, Hospital Admissions, and Deaths in Middle-Aged Adults in 21 Countries from Five Continents (PURE): A Prospective Cohort Study. Lancet 2020, 395, 785–794. [Google Scholar] [CrossRef]
- Pitt, J.M.; Vétizou, M.; Daillère, R.; Roberti, M.P.; Yamazaki, T.; Routy, B.; Lepage, P.; Boneca, I.G.; Chamaillard, M.; Kroemer, G.; et al. Resistance Mechanisms to Immune-Checkpoint Blockade in Cancer: Tumor-Intrinsic and -Extrinsic Factors. Immunity 2016, 44, 1255–1269. [Google Scholar] [CrossRef] [PubMed]
- Mole, R.H. Whole Body Irradiation—Radiobiology or Medicine? Br. J. Radiol. 1953, 26, 234–241. [Google Scholar] [CrossRef]
- Marconi, R.; Strolin, S.; Bossi, G.; Strigari, L. A Meta-Analysis of the Abscopal Effect in Preclinical Models: Is the Biologically Effective Dose a Relevant Physical Trigger? PLoS ONE 2017, 12, e0171559. [Google Scholar] [CrossRef] [PubMed]
- Demaria, S.; Ng, B.; Devitt, M.L.; Babb, J.S.; Kawashima, N.; Liebes, L.; Formenti, S.C. Ionizing Radiation Inhibition of Distant Untreated Tumors (Abscopal Effect) Is Immune Mediated. Int. J. Radiat. Oncol. 2004, 58, 862–870. [Google Scholar] [CrossRef]
- Rodriguez-Ruiz, M.E.; Rodriguez, I.; Garasa, S.; Barbes, B.; Solorzano, J.L.; Perez-Gracia, J.L.; Labiano, S.; Sanmamed, M.F.; Azpilikueta, A.; Bolaños, E.; et al. Abscopal Effects of Radiotherapy Are Enhanced by Combined Immunostimulatory MAbs and Are Dependent on CD8 T Cells and Crosspriming. Cancer Res. 2016, 76, 5994–6005. [Google Scholar] [CrossRef]
- Galluzzi, L.; Vitale, I.; Warren, S.; Adjemian, S.; Agostinis, P.; Martinez, A.B.; Chan, T.A.; Coukos, G.; Demaria, S.; Deutsch, E.; et al. Consensus Guidelines for the Definition, Detection and Interpretation of Immunogenic Cell Death. J. Immunother. Cancer 2020, 8. [Google Scholar] [CrossRef]
- Vanpouille-Box, C.; Alard, A.; Aryankalayil, M.J.; Sarfraz, Y.; Diamond, J.M.; Schneider, R.J.; Inghirami, G.; Coleman, C.N.; Formenti, S.C.; Demaria, S. DNA Exonuclease Trex1 Regulates Radiotherapy-Induced Tumour Immunogenicity. Nat. Commun. 2017, 8, 15618. [Google Scholar] [CrossRef]
- Galluzzi, L.; Vitale, I.; Aaronson, S.A.; Abrams, J.M.; Adam, D.; Agostinis, P.; Alnemri, E.S.; Altucci, L.; Amelio, I.; Andrews, D.W.; et al. Molecular Mechanisms of Cell Death: Recommendations of the Nomenclature Committee on Cell Death 2018. Cell Death Differ. 2018, 25, 486–541. [Google Scholar] [CrossRef]
- Tang, D.; Kang, R.; Berghe, T.V.; Vandenabeele, P.; Kroemer, G. The Molecular Machinery of Regulated Cell Death. Cell Res. 2019, 29, 347–364. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Wang, G.; Chen, Y.; Wang, H.; Hua, Y.; Cai, Z. Immunogenic Cell Death in Cancer Therapy: Present and Emerging Inducers. J. Cell. Mol. Med. 2019, 23, 4854–4865. [Google Scholar] [CrossRef] [PubMed]
- Apetoh, L.; Ghiringhelli, F.; Tesniere, A.; Obeid, M.; Ortiz, C.; Criollo, A.; Mignot, G.; Maiuri, M.C.; Ullrich, E.; Saulnier, P.; et al. Toll-like Receptor 4–Dependent Contribution of the Immune System to Anticancer Chemotherapy and Radiotherapy. Nat. Med. 2007, 13, 1050–1059. [Google Scholar] [CrossRef] [PubMed]
- Kroemer, G.; Galluzzi, L.; Kepp, O.; Zitvogel, L. Immunogenic Cell Death in Cancer Therapy. Annu. Rev. Immunol. 2013, 31, 51–72. [Google Scholar] [CrossRef]
- Jiang, W.; Chan, C.K.; Weissman, I.L.; Kim, B.Y.S.; Hahn, S.M. Immune Priming of the Tumor Microenvironment by Radiation. Trends Cancer 2016, 2, 638–645. [Google Scholar] [CrossRef]
- Kobie, J.J.; Wu, R.S.; Kurt, R.A.; Lou, S.; Adelman, M.K.; Whitesell, L.J.; Ramanathapuram, L.V.; Arteaga, C.L.; Akporiaye, E.T. Transforming Growth Factor Beta Inhibits the Antigen-Presenting Functions and Antitumor Activity of Dendritic Cell Vaccines. Cancer Res. 2003, 63, 1860–1864. [Google Scholar] [PubMed]
- Zhao, X.; Shao, C. Radiotherapy-Mediated Immunomodulation and Anti-Tumor Abscopal Effect Combining Immune Checkpoint Blockade. Cancers 2020, 12, 2762. [Google Scholar] [CrossRef]
- Weichselbaum, R.R.; Liang, H.; Deng, L.; Fu, Y.-X. Radiotherapy and Immunotherapy: A Beneficial Liaison? Nat. Rev. Clin. Oncol. 2017, 14, 365–379. [Google Scholar] [CrossRef]
- Deng, L.; Liang, H.; Xu, M.; Yang, X.; Burnette, B.; Arina, A.; Li, X.-D.; Mauceri, H.; Beckett, M.; Darga, T.; et al. STING-Dependent Cytosolic DNA Sensing Promotes Radiation-Induced Type I Interferon-Dependent Antitumor Immunity in Immunogenic Tumors. Immunity 2014, 41, 843–852. [Google Scholar] [CrossRef]
- Reits, E.A.; Hodge, J.W.; Herberts, C.A.; Groothuis, T.A.; Chakraborty, M.; Wansley, E.K.; Camphausen, K.; Luiten, R.M.; de Ru, A.H.; Neijssen, J.; et al. Radiation Modulates the Peptide Repertoire, Enhances MHC Class I Expression, and Induces Successful Antitumor Immunotherapy. J. Exp. Med. 2006, 203, 1259–1271. [Google Scholar] [CrossRef]
- Ganss, R.; Ryschich, E.; Klar, E.; Arnold, B.; Hämmerling, G.J. Combination of T-Cell Therapy and Trigger of Inflammation Induces Remodeling of the Vasculature and Tumor Eradication. Cancer Res. 2002, 62, 1462–1470. [Google Scholar]
- Deng, L.; Liang, H.; Burnette, B.; Beckett, M.; Darga, T.; Weichselbaum, R.R.; Fu, Y.-X. Irradiation and Anti–PD-L1 Treatment Synergistically Promote Antitumor Immunity in Mice. J. Clin. Investig. 2014, 124, 687–695. [Google Scholar] [CrossRef] [PubMed]
- Kachikwu, E.L.; Iwamoto, K.S.; Liao, Y.-P.; DeMarco, J.J.; Agazaryan, N.; Economou, J.S.; McBride, W.H.; Schaue, D. Radiation Enhances Regulatory T Cell Representation. Int. J. Radiat. Oncol. 2011, 81, 1128–1135. [Google Scholar] [CrossRef]
- Dovedi, S.J.; Illidge, T.M. The Antitumor Immune Response Generated by Fractionated Radiation Therapy May Be Limited by Tumor Cell Adaptive Resistance and Can Be Circumvented by PD-L1 Blockade. Oncoimmunology 2015, 4, e1016709. [Google Scholar] [CrossRef] [PubMed]
- Roux, C.; Jafari, S.M.; Shinde, R.; Duncan, G.; Cescon, D.W.; Silvester, J.; Chu, M.F.; Hodgson, K.; Berger, T.; Wakeham, A.; et al. Reactive Oxygen Species Modulate Macrophage Immunosuppressive Phenotype through the Up-Regulation of PD-L1. Proc. Natl. Acad. Sci. USA 2019, 116, 4326–4335. [Google Scholar] [CrossRef] [PubMed]
- Jobling, M.F.; Mott, J.D.; Finnegan, M.T.; Jurukovski, V.; Erickson, A.C.; Walian, P.J.; Taylor, S.E.; Ledbetter, S.; Lawrence, C.M.; Rifkin, D.B.; et al. Isoform-Specific Activation of Latent Transforming Growth Factor β (LTGF-β) by Reactive Oxygen Species. Radiat. Res. 2006, 166, 839–848. [Google Scholar] [CrossRef] [PubMed]
- Park, S.S.; Dong, H.; Liu, X.; Harrington, S.M.; Krco, C.J.; Grams, M.P.; Mansfield, A.S.; Furutani, K.M.; Olivier, K.R.; Kwon, E.D. PD-1 Restrains Radiotherapy-Induced Abscopal Effect. Cancer Immunol. Res. 2015, 3, 610–619. [Google Scholar] [CrossRef]
- Skinner, H.D.; Giri, U.; Yang, L.P.; Kumar, M.; Liu, Y.; Story, M.D.; Pickering, C.R.; Byers, L.A.; Williams, M.D.; Wang, J.; et al. Integrative Analysis Identifies a Novel AXL–PI3 Kinase–PD-L1 Signaling Axis Associated with Radiation Resistance in Head and Neck Cancer. Clin. Cancer Res. 2017, 23, 2713–2722. [Google Scholar] [CrossRef]
- Formenti, S.C.; Demaria, S. Combining Radiotherapy and Cancer Immunotherapy: A Paradigm Shift. JNCI J. Natl. Cancer Inst. 2013, 105, 256–265. [Google Scholar] [CrossRef] [PubMed]
- Kalbasi, A.; June, C.H.; Haas, N.; Vapiwala, N. Radiation and Immunotherapy: A Synergistic Combination. J. Clin. Investig. 2013, 123, 2756–2763. [Google Scholar] [CrossRef]
- Demaria, S.; Formenti, S.C. Radiation as an Immunological Adjuvant: Current Evidence on Dose and Fractionation. Front. Oncol. 2012, 2. [Google Scholar] [CrossRef]
- Jabbour, S.K.; Berman, A.T.; Decker, R.H.; Lin, Y.; Feigenberg, S.J.; Gettinger, S.N.; Aggarwal, C.; Langer, C.J.; Simone, C.B.; Bradley, J.D.; et al. Phase 1 Trial of Pembrolizumab Administered Concurrently With Chemoradiotherapy for Locally Advanced Non–Small Cell Lung Cancer: A Nonrandomized Controlled Trial. JAMA Oncol. 2020, 6, 848. [Google Scholar] [CrossRef] [PubMed]
- Gray, J.E.; Villegas, A.; Daniel, D.; Vicente, D.; Murakami, S.; Hui, R.; Kurata, T.; Chiappori, A.; Lee, K.H.; Cho, B.C.; et al. Three-Year Overall Survival with Durvalumab after Chemoradiotherapy in Stage III NSCLC—Update from PACIFIC. J. Thorac. Oncol. 2020, 15, 288–293. [Google Scholar] [CrossRef]
- Antonia, S.J.; Villegas, A.; Daniel, D.; Vicente, D.; Murakami, S.; Hui, R.; Yokoi, T.; Chiappori, A.; Lee, K.H.; de Wit, M.; et al. Durvalumab after Chemoradiotherapy in Stage III Non–Small-Cell Lung Cancer. N. Engl. J. Med. 2017, 377, 1919–1929. [Google Scholar] [CrossRef] [PubMed]
- Antonia, S.J.; Villegas, A.; Daniel, D.; Vicente, D.; Murakami, S.; Hui, R.; Kurata, T.; Chiappori, A.; Lee, K.H.; de Wit, M.; et al. Overall Survival with Durvalumab after Chemoradiotherapy in Stage III NSCLC. N. Engl. J. Med. 2018, 379, 2342–2350. [Google Scholar] [CrossRef] [PubMed]
- Durm, G.A.; Jabbour, S.K.; Althouse, S.K.; Liu, Z.; Sadiq, A.A.; Zon, R.T.; Jalal, S.I.; Kloecker, G.H.; Williamson, M.J.; Reckamp, K.L.; et al. A Phase 2 Trial of Consolidation Pembrolizumab Following Concurrent Chemoradiation for Patients with Unresectable Stage III Non–Small Cell Lung Cancer: Hoosier Cancer Research Network LUN 14-179. Cancer 2020, 126, 4353–4361. [Google Scholar] [CrossRef]
- Bauml, J.M.; Mick, R.; Ciunci, C.; Aggarwal, C.; Davis, C.; Evans, T.; Deshpande, C.; Miller, L.; Patel, P.; Alley, E.; et al. Pembrolizumab After Completion of Locally Ablative Therapy for Oligometastatic Non–Small Cell Lung Cancer: A Phase 2 Trial. JAMA Oncol. 2019, 5, 1283. [Google Scholar] [CrossRef]
- Powell, S.F.; Gold, K.A.; Gitau, M.M.; Sumey, C.J.; Lohr, M.M.; McGraw, S.C.; Nowak, R.K.; Jensen, A.W.; Blanchard, M.J.; Fischer, C.D.; et al. Safety and Efficacy of Pembrolizumab With Chemoradiotherapy in Locally Advanced Head and Neck Squamous Cell Carcinoma: A Phase IB Study. J. Clin. Oncol. 2020, 38, 2427–2437. [Google Scholar] [CrossRef]
- Cohen, E.E.; Ferris, R.L.; Psyrri, A.; Haddad, R.; Tahara, M.; Bourhis, J.; Harrington, K.J.; Chang, P.M.-H.; Lin, J.-C.; Razaq, M.; et al. 910O Primary Results of the Phase III JAVELIN Head & Neck 100 Trial: Avelumab plus Chemoradiotherapy (CRT) Followed by Avelumab Maintenance vs CRT in Patients with Locally Advanced Squamous Cell Carcinoma of the Head and Neck (LA SCCHN). Ann. Oncol. 2020, 31, S658. [Google Scholar] [CrossRef]
- Bourhis, J.; Sire, C.; Tao, Y.; Martin, L.; Alfonsi, M.; Prevost, J.B.; Rives, M.; Lafond, C.; Tourani, J.M.; Biau, J.; et al. LBA38 Pembrolizumab versus Cetuximab, Concomitant with Radiotherapy (RT) in Locally Advanced Head and Neck Squamous Cell Carcinoma (LA-HNSCC): Results of the GORTEC 2015-01 “PembroRad” Randomized Trial. Ann. Oncol. 2020, 31, S1168. [Google Scholar] [CrossRef]
- Hammers, H.J.; Vonmerveldt, D.; Ahn, C.; Nadal, R.M.; Drake, C.G.; Folkert, M.R.; Laine, A.M.; Courtney, K.D.; Brugarolas, J.; Song, D.Y.; et al. Combination of Dual Immune Checkpoint Inhibition (ICI) with Stereotactic Radiation (SBRT) in Metastatic Renal Cell Carcinoma (MRCC) (RADVAX RCC). J. Clin. Oncol. 2020, 38, 614. [Google Scholar] [CrossRef]
- Masini, C.; Iotti, C.; De Giorgi, U.; Bellia, R.S.; Buti, S.; Salaroli, F.; Zampiva, I.; Mazzarotto, R.; Mucciarini, C.; Baldessari, C.; et al. Nivolumab (NIVO) in Combination with Stereotactic Body Radiotherapy (SBRT) in Pretreated Patients (Pts) with Metastatic Renal Cell Carcinoma (MRCC): First Results of Phase II NIVES Study. J. Clin. Oncol. 2020, 38, 613. [Google Scholar] [CrossRef]
- Twyman-Saint Victor, C.; Rech, A.J.; Maity, A.; Rengan, R.; Pauken, K.E.; Stelekati, E.; Benci, J.L.; Xu, B.; Dada, H.; Odorizzi, P.M.; et al. Radiation and Dual Checkpoint Blockade Activate Non-Redundant Immune Mechanisms in Cancer. Nature 2015, 520, 373–377. [Google Scholar] [CrossRef]
- Hiniker, S.M.; Reddy, S.A.; Maecker, H.T.; Subrahmanyam, P.B.; Rosenberg-Hasson, Y.; Swetter, S.M.; Saha, S.; Shura, L.; Knox, S.J. A Prospective Clinical Trial Combining Radiation Therapy With Systemic Immunotherapy in Metastatic Melanoma. Int. J. Radiat. Oncol. 2016, 96, 578–588. [Google Scholar] [CrossRef]
- Postow, M.A.; Knox, S.J.; Goldman, D.A.; Elhanati, Y.; Mavinkurve, V.; Wong, P.; Halpenny, D.; Reddy, S.K.; Vado, K.; McCabe, D.; et al. A Prospective, Phase 1 Trial of Nivolumab, Ipilimumab, and Radiotherapy in Patients with Advanced Melanoma. Clin. Cancer Res. 2020, 26, 3193–3201. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Jin, T.; Li, M.; Xue, J.; Lu, B. Synergistic Effect of Immunotherapy and Radiotherapy in Non-Small Cell Lung Cancer: Current Clinical Trials and Prospective Challenges. Precis. Clin. Med. 2019, 2, 57–70. [Google Scholar] [CrossRef]
- Dovedi, S.J.; Adlard, A.L.; Lipowska-Bhalla, G.; McKenna, C.; Jones, S.; Cheadle, E.J.; Stratford, I.J.; Poon, E.; Morrow, M.; Stewart, R.; et al. Acquired Resistance to Fractionated Radiotherapy Can Be Overcome by Concurrent PD-L1 Blockade. Cancer Res. 2014, 74, 5458–5468. [Google Scholar] [CrossRef] [PubMed]
- Spaas, M.; Lievens, Y. Is the Combination of Immunotherapy and Radiotherapy in Non-Small Cell Lung Cancer a Feasible and Effective Approach? Front. Med. 2019, 6, 244. [Google Scholar] [CrossRef]
- Knisely, J.P.S.; Yu, J.B.; Flanigan, J.; Sznol, M.; Kluger, H.M.; Chiang, V.L.S. Radiosurgery for Melanoma Brain Metastases in the Ipilimumab Era and the Possibility of Longer Survival: Clinical Article. J. Neurosurg. 2012, 117, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Kiess, A.P.; Wolchok, J.D.; Barker, C.A.; Postow, M.A.; Tabar, V.; Huse, J.T.; Chan, T.A.; Yamada, Y.; Beal, K. Stereotactic Radiosurgery for Melanoma Brain Metastases in Patients Receiving Ipilimumab: Safety Profile and Efficacy of Combined Treatment. Int. J. Radiat. Oncol. 2015, 92, 368–375. [Google Scholar] [CrossRef]
- Kabiljo, J.; Harpain, F.; Carotta, S.; Bergmann, M. Radiotherapy as a Backbone for Novel Concepts in Cancer Immunotherapy. Cancers 2019, 12, 79. [Google Scholar] [CrossRef]
- Stokes, W.A.; Binder, D.C.; Jones, B.L.; Oweida, A.J.; Liu, A.K.; Rusthoven, C.G.; Karam, S.D. Impact of Immunotherapy among Patients with Melanoma Brain Metastases Managed with Radiotherapy. J. Neuroimmunol. 2017, 313, 118–122. [Google Scholar] [CrossRef] [PubMed]
- Qian, J.M.; Yu, J.B.; Kluger, H.M.; Chiang, V.L.S. Timing and Type of Immune Checkpoint Therapy Affect the Early Radiographic Response of Melanoma Brain Metastases to Stereotactic Radiosurgery: Immunotherapy and SRS in Brain Metastases. Cancer 2016, 122, 3051–3058. [Google Scholar] [CrossRef]
- Ahmed, K.A.; Stallworth, D.G.; Kim, Y.; Johnstone, P.A.S.; Harrison, L.B.; Caudell, J.J.; Yu, H.H.M.; Etame, A.B.; Weber, J.S.; Gibney, G.T. Clinical Outcomes of Melanoma Brain Metastases Treated with Stereotactic Radiation and Anti-PD-1 Therapy. Ann. Oncol. 2016, 27, 434–441. [Google Scholar] [CrossRef]
- Komatsu, T.; Konishi, K.; Aoshima, M.; Tokura, Y.; Nakamura, K. Combined Radiotherapy with Nivolumab for Extracranial Metastatic Malignant Melanoma. Jpn. J. Radiol. 2018, 36, 712–718. [Google Scholar] [CrossRef]
- Nardin, C.; Mateus, C.; Texier, M.; Lanoy, E.; Hibat-Allah, S.; Ammari, S.; Robert, C.; Dhermain, F. Tolerance and Outcomes of Stereotactic Radiosurgery Combined with Anti-Programmed Cell Death-1 (Pembrolizumab) for Melanoma Brain Metastases. Melanoma Res. 2018, 28, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Aboudaram, A.; Modesto, A.; Chaltiel, L.; Gomez-Roca, C.; Boulinguez, S.; Sibaud, V.; Delord, J.-P.; Chira, C.; Delannes, M.; Moyal, E.; et al. Concurrent Radiotherapy for Patients with Metastatic Melanoma and Receiving Anti-Programmed-Death 1 Therapy: A Safe and Effective Combination. Melanoma Res. 2017, 27, 485–491. [Google Scholar] [CrossRef] [PubMed]
- Miyauchi, S.; Kim, S.S.; Pang, J.; Gold, K.A.; Gutkind, J.S.; Califano, J.A.; Mell, L.K.; Cohen, E.E.W.; Sharabi, A.B. Immune Modulation of Head and Neck Squamous Cell Carcinoma and the Tumor Microenvironment by Conventional Therapeutics. Clin. Cancer Res. 2019, 25, 4211–4223. [Google Scholar] [CrossRef] [PubMed]
- Ettinger, D.S.; Wood, D.E.; Aggarwal, C.; Aisner, D.L.; Akerley, W.; Bauman, J.R.; Bharat, A.; Bruno, D.S.; Chang, J.Y.; Chirieac, L.R.; et al. NCCN Guidelines Insights: Non–Small Cell Lung Cancer, Version 1.2020. J. Natl. Compr. Canc. Netw. 2019, 17, 1464–1472. [Google Scholar] [CrossRef]
- Shaverdian, N.; Lisberg, A.E.; Bornazyan, K.; Veruttipong, D.; Goldman, J.W.; Formenti, S.C.; Garon, E.B.; Lee, P. Previous Radiotherapy and the Clinical Activity and Toxicity of Pembrolizumab in the Treatment of Non-Small-Cell Lung Cancer: A Secondary Analysis of the KEYNOTE-001 Phase 1 Trial. Lancet Oncol. 2017, 18, 895–903. [Google Scholar] [CrossRef]
- Peters, S.; Felip, E.; Dafni, U.; Tufman, A.; Guckenberger, M.; Irigoyen, A.; Nadal, E.; Becker, A.; Vees, H.; Pless, M.; et al. Efficacy Evaluation of Concurrent Nivolumab Addition to a First-Line, Concurrent Chemo-Radiotherapy Regimen in Unresectable Locally Advanced NSCLC: Results from the European Thoracic Oncology Platform (ETOP 6-14) NICOLAS Phase II Trial. Ann. Oncol. 2019, 30, v591. [Google Scholar] [CrossRef]
- Lin, S.; Lin, X.; Clay, D.; Yao, L.; Mok, I.; Gomez, D.; Kurie, J.; Simon, G.; Blumenschein, G.; Young, J.; et al. OA01.06 DETERRED: Phase II Trial Combining Atezolizumab Concurrently with Chemoradiation Therapy in Locally Advanced Non-Small Cell Lung Cancer. J. Thorac. Oncol. 2018, 13, S320–S321. [Google Scholar] [CrossRef]
- Pozzessere, C.; Bouchaab, H.; Jumeau, R.; Letovanec, I.; Daccord, C.; Bourhis, J.; Prior, J.O.; Peters, S.; Lazor, R.; Beigelman-Aubry, C. Relationship between Pneumonitis Induced by Immune Checkpoint Inhibitors and the Underlying Parenchymal Status: A Retrospective Study. ERJ Open Res. 2020, 6, 00165–02019. [Google Scholar] [CrossRef]
- Sha, C.M.; Lehrer, E.J.; Hwang, C.; Trifiletti, D.M.; Mackley, H.B.; Drabick, J.J.; Zaorsky, N.G. Toxicity in Combination Immune Checkpoint Inhibitor and Radiation Therapy: A Systematic Review and Meta-Analysis. Radiother. Oncol. 2020, 151, 141–148. [Google Scholar] [CrossRef]
- Quezada, S.A.; Peggs, K.S. Lost in Translation: Deciphering the Mechanism of Action of Anti-Human CTLA-4. Clin. Cancer Res. 2019, 25, 1130–1132. [Google Scholar] [CrossRef]
- Simpson, T.R.; Li, F.; Montalvo-Ortiz, W.; Sepulveda, M.A.; Bergerhoff, K.; Arce, F.; Roddie, C.; Henry, J.Y.; Yagita, H.; Wolchok, J.D.; et al. Fc-Dependent Depletion of Tumor-Infiltrating Regulatory T Cells Co-Defines the Efficacy of Anti–CTLA-4 Therapy against Melanoma. J. Exp. Med. 2013, 210, 1695–1710. [Google Scholar] [CrossRef]
- Vanpouille-Box, C.; Diamond, J.M.; Pilones, K.A.; Zavadil, J.; Babb, J.S.; Formenti, S.C.; Barcellos-Hoff, M.H.; Demaria, S. TGFβ Is a Master Regulator of Radiation Therapy-Induced Antitumor Immunity. Cancer Res. 2015, 75, 2232–2242. [Google Scholar] [CrossRef] [PubMed]
- Young, K.H.; Baird, J.R.; Savage, T.; Cottam, B.; Friedman, D.; Bambina, S.; Messenheimer, D.J.; Fox, B.; Newell, P.; Bahjat, K.S.; et al. Optimizing Timing of Immunotherapy Improves Control of Tumors by Hypofractionated Radiation Therapy. PLoS ONE 2016, 11, e0157164. [Google Scholar] [CrossRef]
- Barker, C.A.; Postow, M.A.; Khan, S.A.; Beal, K.; Parhar, P.K.; Yamada, Y.; Lee, N.Y.; Wolchok, J.D. Concurrent Radiotherapy and Ipilimumab Immunotherapy for Patients with Melanoma. Cancer Immunol. Res. 2013, 1, 92–98. [Google Scholar] [CrossRef] [PubMed]
- Kwon, E.D.; Drake, C.G.; Scher, H.I.; Fizazi, K.; Bossi, A.; van den Eertwegh, A.J.M.; Krainer, M.; Houede, N.; Santos, R.; Mahammedi, H.; et al. Ipilimumab versus Placebo after Radiotherapy in Patients with Metastatic Castration-Resistant Prostate Cancer That Had Progressed after Docetaxel Chemotherapy (CA184-043): A Multicentre, Randomised, Double-Blind, Phase 3 Trial. Lancet Oncol. 2014, 15, 700–712. [Google Scholar] [CrossRef]
- Tang, C.; Welsh, J.W.; de Groot, P.; Massarelli, E.; Chang, J.Y.; Hess, K.R.; Basu, S.; Curran, M.A.; Cabanillas, M.E.; Subbiah, V.; et al. Ipilimumab with Stereotactic Ablative Radiation Therapy: Phase I Results and Immunologic Correlates from Peripheral T Cells. Clin. Cancer Res. 2017, 23, 1388–1396. [Google Scholar] [CrossRef] [PubMed]
- Verbrugge, I.; Hagekyriakou, J.; Sharp, L.L.; Galli, M.; West, A.; McLaughlin, N.M.; Duret, H.; Yagita, H.; Johnstone, R.W.; Smyth, M.J.; et al. Radiotherapy Increases the Permissiveness of Established Mammary Tumors to Rejection by Immunomodulatory Antibodies. Cancer Res. 2012, 72, 3163–3174. [Google Scholar] [CrossRef] [PubMed]
- Cao, Y.; Li, W.; Wang, Z.; Pang, H. Potential and Unsolved Problems of Anti-PD-1/PD-L1 Therapy Combined with Radiotherapy. Tumori J. 2020. [Google Scholar] [CrossRef]
- Hwang, W.L.; Pike, L.R.G.; Royce, T.J.; Mahal, B.A.; Loeffler, J.S. Safety of Combining Radiotherapy with Immune-Checkpoint Inhibition. Nat. Rev. Clin. Oncol. 2018, 15, 477–494. [Google Scholar] [CrossRef]
- Deutsch, E.; Chargari, C.; Galluzzi, L.; Kroemer, G. Optimising Efficacy and Reducing Toxicity of Anticancer Radioimmunotherapy. Lancet Oncol. 2019, 20, e452–e463. [Google Scholar] [CrossRef]
- Maker, A.V.; Yang, J.C.; Sherry, R.M.; Topalian, S.L.; Kammula, U.S.; Royal, R.E.; Hughes, M.; Yellin, M.J.; Haworth, L.R.; Levy, C.; et al. Intrapatient Dose Escalation of Anti-CTLA-4 Antibody in Patients with Metastatic Melanoma. J. Immunother. Hagerstown Md 1997 2006, 29, 455–463. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.E.; Patel, M.A.; Mangraviti, A.; Kim, E.S.; Theodros, D.; Velarde, E.; Liu, A.; Sankey, E.W.; Tam, A.; Xu, H.; et al. Combination Therapy with Anti-PD-1, Anti-TIM-3, and Focal Radiation Results in Regression of Murine Gliomas. Clin. Cancer Res. 2017, 23, 124–136. [Google Scholar] [CrossRef]
- Dewan, M.Z.; Galloway, A.E.; Kawashima, N.; Dewyngaert, J.K.; Babb, J.S.; Formenti, S.C.; Demaria, S. Fractionated but Not Single-Dose Radiotherapy Induces an Immune-Mediated Abscopal Effect When Combined with Anti-CTLA-4 Antibody. Clin. Cancer Res. 2009, 15, 5379–5388. [Google Scholar] [CrossRef]
- Lugade, A.A.; Moran, J.P.; Gerber, S.A.; Rose, R.C.; Frelinger, J.G.; Lord, E.M. Local Radiation Therapy of B16 Melanoma Tumors Increases the Generation of Tumor Antigen-Specific Effector Cells That Traffic to the Tumor. J. Immunol. 2005, 174, 7516–7523. [Google Scholar] [CrossRef]
- Schaue, D.; Ratikan, J.A.; Iwamoto, K.S.; McBride, W.H. Maximizing Tumor Immunity With Fractionated Radiation. Int. J. Radiat. Oncol. 2012, 83, 1306–1310. [Google Scholar] [CrossRef]
- Qin, Q.; Nan, X.; Miller, T.; Fisher, R.; Teh, B.; Pandita, S.; Farach, A.M.; Pingali, S.R.; Pandita, R.K.; Butler, E.B.; et al. Complete Local and Abscopal Responses from a Combination of Radiation and Nivolumab in Refractory Hodgkin’s Lymphoma. Radiat. Res. 2018, 190, 322. [Google Scholar] [CrossRef]
- Quéro, L.; Gilardin, L.; Fumagalli, I.; Martin, V.; Guillerm, S.; Bauduceau, O.; Kirova, Y.M.; Hennequin, C.; Brice, P. Anti-PD-1 Immunotherapy in Combination with Sequential Involved-Site Radiotherapy in Heavily Pretreated Refractory Hodgkin Lymphoma. Cancer/Radiothérapie 2019, 23, 132–137. [Google Scholar] [CrossRef]
- Bernstein, M.B.; Krishnan, S.; Hodge, J.W.; Chang, J.Y. Immunotherapy and Stereotactic Ablative Radiotherapy (ISABR): A Curative Approach? Nat. Rev. Clin. Oncol. 2016, 13, 516–524. [Google Scholar] [CrossRef]
- DeSelm, C.; Palomba, M.L.; Yahalom, J.; Hamieh, M.; Eyquem, J.; Rajasekhar, V.K.; Sadelain, M. Low-Dose Radiation Conditioning Enables CAR T Cells to Mitigate Antigen Escape. Mol. Ther. 2018, 26, 2542–2552. [Google Scholar] [CrossRef] [PubMed]
- Deloch, L.; Derer, A.; Hartmann, J.; Frey, B.; Fietkau, R.; Gaipl, U.S. Modern Radiotherapy Concepts and the Impact of Radiation on Immune Activation. Front. Oncol. 2016, 6. [Google Scholar] [CrossRef]
- Venkatesulu, B.P.; Mallick, S.; Lin, S.H.; Krishnan, S. A Systematic Review of the Influence of Radiation-Induced Lymphopenia on Survival Outcomes in Solid Tumors. Crit. Rev. Oncol. Hematol. 2018, 123, 42–51. [Google Scholar] [CrossRef]
- Chen, D.; Verma, V.; Patel, R.R.; Barsoumian, H.B.; Cortez, M.A.; Welsh, J.W. Absolute Lymphocyte Count Predicts Abscopal Responses and Outcomes in Patients Receiving Combined Immunotherapy and Radiation Therapy: Analysis of 3 Phase 1/2 Trials. Int. J. Radiat. Oncol. 2020, 108, 196–203. [Google Scholar] [CrossRef]
- Marciscano, A.E.; Ghasemzadeh, A.; Nirschl, T.R.; Theodros, D.; Kochel, C.M.; Francica, B.J.; Muroyama, Y.; Anders, R.A.; Sharabi, A.B.; Velarde, E.; et al. Elective Nodal Irradiation Attenuates the Combinatorial Efficacy of Stereotactic Radiation Therapy and Immunotherapy. Clin. Cancer Res. 2018, 24, 5058–5071. [Google Scholar] [CrossRef]
- Golden, E.B.; Chhabra, A.; Chachoua, A.; Adams, S.; Donach, M.; Fenton-Kerimian, M.; Friedman, K.; Ponzo, F.; Babb, J.S.; Goldberg, J.; et al. Local Radiotherapy and Granulocyte-Macrophage Colony-Stimulating Factor to Generate Abscopal Responses in Patients with Metastatic Solid Tumours: A Proof-of-Principle Trial. Lancet Oncol. 2015, 16, 795–803. [Google Scholar] [CrossRef]
- Liu, M.; Cai, X.; Zeng, Y. EP1.04-28 The Abscopal Effects of the Combination of Radiotherapy and GM-CSF for Patients with Metastatic Thoracic Cancers. J. Thorac. Oncol. 2019, 14, S980. [Google Scholar] [CrossRef]
- Kwek, S.S.; Kahn, J.; Greaney, S.K.; Lewis, J.; Cha, E.; Zhang, L.; Weber, R.W.; Leonard, L.; Markovic, S.N.; Fong, L.; et al. GM-CSF and Ipilimumab Therapy in Metastatic Melanoma: Clinical Outcomes and Immunologic Responses. OncoImmunology 2016, 5, e1101204. [Google Scholar] [CrossRef]
- Seung, S.K.; Curti, B.D.; Crittenden, M.; Walker, E.; Coffey, T.; Siebert, J.C.; Miller, W.; Payne, R.; Glenn, L.; Bageac, A.; et al. Phase 1 Study of Stereotactic Body Radiotherapy and Interleukin-2--Tumor and Immunological Responses. Sci. Transl. Med. 2012, 4, 137ra74. [Google Scholar] [CrossRef]
- Bouquet, F.; Pal, A.; Pilones, K.A.; Demaria, S.; Hann, B.; Akhurst, R.J.; Babb, J.S.; Lonning, S.M.; DeWyngaert, J.K.; Formenti, S.C.; et al. TGF 1 Inhibition Increases the Radiosensitivity of Breast Cancer Cells In Vitro and Promotes Tumor Control by Radiation In Vivo. Clin. Cancer Res. 2011, 17, 6754–6765. [Google Scholar] [CrossRef] [PubMed]
- Spiotto, M.; Fu, Y.-X.; Weichselbaum, R.R. The Intersection of Radiotherapy and Immunotherapy: Mechanisms and Clinical Implications. Sci. Immunol. 2016, 1, eaag1266. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Ruiz, M.E.; Rodríguez, I.; Mayorga, L.; Labiano, T.; Barbes, B.; Etxeberria, I.; Ponz-Sarvise, M.; Azpilikueta, A.; Bolaños, E.; Sanmamed, M.F.; et al. TGFβ Blockade Enhances Radiotherapy Abscopal Efficacy Effects in Combination with Anti-PD1 and Anti-CD137 Immunostimulatory Monoclonal Antibodies. Mol. Cancer Ther. 2019, 18, 621–631. [Google Scholar] [CrossRef]
- Neelapu, S.S.; Tummala, S.; Kebriaei, P.; Wierda, W.; Gutierrez, C.; Locke, F.L.; Komanduri, K.V.; Lin, Y.; Jain, N.; Daver, N.; et al. Chimeric Antigen Receptor T-Cell Therapy—Assessment and Management of Toxicities. Nat. Rev. Clin. Oncol. 2018, 15, 47–62. [Google Scholar] [CrossRef] [PubMed]
| Population | Reference | Phase | Intervention | Results |
|---|---|---|---|---|
| Locally advanced NSCLC | [31] | I | Pembrolizumab + chemoradiotherapy | 6-mo PFS rate = 81% |
| 12-mo PFS rate = 69.7% | ||||
| Median PFS = 18.7 mo | ||||
| Locally advanced NSCLC | PACIFIC [32,33,34] | III | Durvalumab (12 mo) as consolidation therapy vs. placebo (12 mo) | ORR = 28.4% vs. 16.0% (p < 0.001) |
| Median PFS = 16.8 mo vs. 5.6 (p < 0.001) | ||||
| 36 months OS = 55.3% vs. 43.5% | ||||
| Locally advanced NSCLC | [35] | II | Chemoradiation + pembrolizumab (12 mo) as consolidation therapy | Time to metastatic disease = 30.7 mo |
| PFS = 18.7 mo | ||||
| OS = 35.8 mo | ||||
| 1–4 metastatic sites NSCLC | [36] | II | Pembrolizumab within 4–12 weeks after locally ablative therapy | Median PFS from the start of locally ablative therapy = 19.1 mo |
| Locally advanced HNSCC | [37] | I | Cisplatin-based chemoradiotherapy + pembrolizumab (concurrently + as maintenance) | CR (HPV+) = 85.3% |
| CR (HPV−) = 78% | ||||
| Locally advanced HNSCC | JAVELIN H&N 100 [38] | III | Avelumab + chemoradiotherapy + avelumab maintenance vs. Placebo + chemoradiotherapy + placebo maintenance | At the time of the interim analysis: no significant improvement in PFS or OS |
| Locally advanced HNSCC (cisplatin-unfit patients) | PembroRad [39] | II | Once-daily RT up to 69.9 Gy associated with: Cetuximab vs. pembrolizumab | Loco-regional-control at 15 mo = 59% vs. 50% (p = 0.91) |
| 24-mo PFS = 40% vs. 42% (p = 0.41) | ||||
| 24-mo OS = 55% vs. 62% (p = 0.5) | ||||
| Stage III/IV RCC | RADVAX RCC [40] | II | Nivolumab + ipilimumab + SBRT (40–50 Gy in 5 fractions) | PR = 56% |
| SD = 24% | ||||
| PD = 16% | ||||
| 12-mo PFS rate = 36% | ||||
| 2nd or 3rd line RCC | NIVES [41] | II | Nivolumab + SBRT (10 Gy × 3 fractions 7 days after the 1st infusion of nivolumab) | ORR = 17.4% |
| 12-mo median OS = 73.4% | ||||
| Metastatic Melanoma | [42] | I | RT (6–8 Gy, 2–3 times) followed by ipilimumab injections | PR = 18% |
| SD = 18% | ||||
| Metastatic Melanoma | [43] | I | Ipilimumab + RT (between 18–50 Gy, in 1–15 fractions) | Clinical benefit = 50% |
| PR = 15% | ||||
| CR = 15% | ||||
| Metastatic Melanoma | [44] | I | Nivolumab + ipilimumab + extracranial RT (30 Gy in 10 fractions or 27 Gy in 3 fractions) | PR outside of the irradiated volume: 6/19 No progression of irradiated metastases |
| Trial Name | Study Phase | Enrollment | Stage | Experimental/Control Arm | Status |
|---|---|---|---|---|---|
| NCT03867175 | Phase III | 116 | III | Consolidative immunotherapy with pembrolizumab +/− SBRT after first line systemic therapy | Recruiting |
| NCT03774732 NIRVANA-lung | Phase III | 688 | III/IV | Anti PD1 +/− RT 15 d after the beginning of ICI, 18 Gy | Recruiting |
| NCT03391869 LONESTAR | Phase III | 116 | IV | Local consolidation therapy: 14 d after nivolumab + ipilimumab for ICI-naive patients with metastatic NSCLC | Recruiting |
| Trial Name | Study Phase | Enrollment | Stage | Experimental/Control Arm | Status |
|---|---|---|---|---|---|
| NCT03380394 | Phase II | 122 | III | RT + pembrolizumab vs. RT + cisplatin | Recruiting |
| NCT03673735 | Phase III | 650 | III | RT + cisplatin +/− durvalumab HPV-negative HNSCC only | Not yet recruiting |
| NCT02999087 | Phase III | 688 | III | RT + cisplatin vs. RT + cetuximab vs. RT + cetuximab + avelumab | Recruiting |
| NCT03349710 | Phase III | 1046 | III/IV | RT + cetuximab +/− nivolumab vs. RT + cisplatin +/− nivolumab | Recruiting |
| NCT03426657 | Phase II | 120 | III | RT + durvalumab + tremelimumab | Not yet recruiting |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Procureur, A.; Simonaggio, A.; Bibault, J.-E.; Oudard, S.; Vano, Y.-A. Enhance the Immune Checkpoint Inhibitors Efficacy with Radiotherapy Induced Immunogenic Cell Death: A Comprehensive Review and Latest Developments. Cancers 2021, 13, 678. https://doi.org/10.3390/cancers13040678
Procureur A, Simonaggio A, Bibault J-E, Oudard S, Vano Y-A. Enhance the Immune Checkpoint Inhibitors Efficacy with Radiotherapy Induced Immunogenic Cell Death: A Comprehensive Review and Latest Developments. Cancers. 2021; 13(4):678. https://doi.org/10.3390/cancers13040678
Chicago/Turabian StyleProcureur, Adrien, Audrey Simonaggio, Jean-Emmanuel Bibault, Stéphane Oudard, and Yann-Alexandre Vano. 2021. "Enhance the Immune Checkpoint Inhibitors Efficacy with Radiotherapy Induced Immunogenic Cell Death: A Comprehensive Review and Latest Developments" Cancers 13, no. 4: 678. https://doi.org/10.3390/cancers13040678
APA StyleProcureur, A., Simonaggio, A., Bibault, J.-E., Oudard, S., & Vano, Y.-A. (2021). Enhance the Immune Checkpoint Inhibitors Efficacy with Radiotherapy Induced Immunogenic Cell Death: A Comprehensive Review and Latest Developments. Cancers, 13(4), 678. https://doi.org/10.3390/cancers13040678