K3326X and Other C-Terminal BRCA2 Variants Implicated in Hereditary Cancer Syndromes: A Review
Simple Summary
Abstract
1. Introduction
2. The Homology Directed Repair Pathway
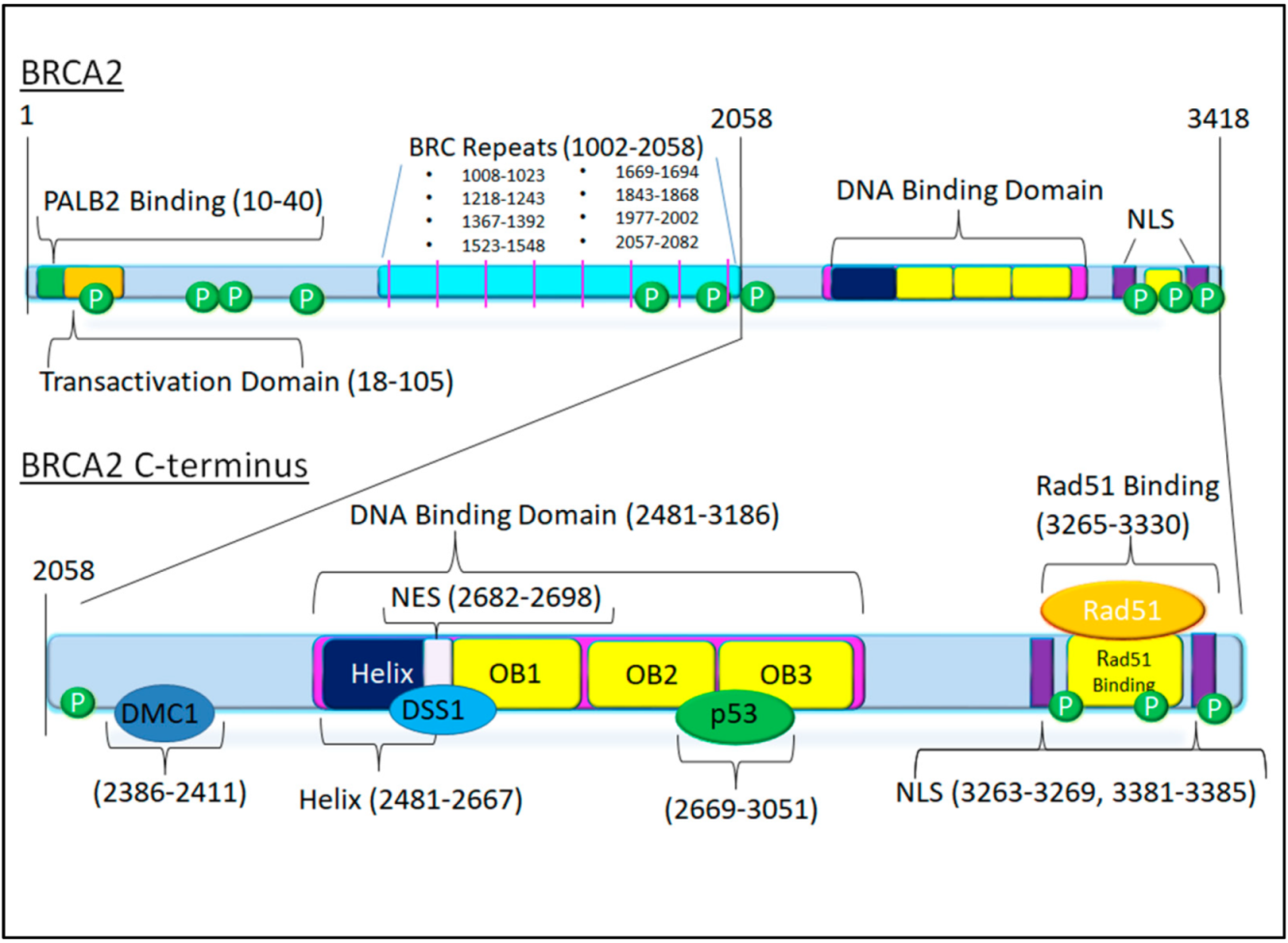
3. BRCA2 Structure and Biochemical Functions
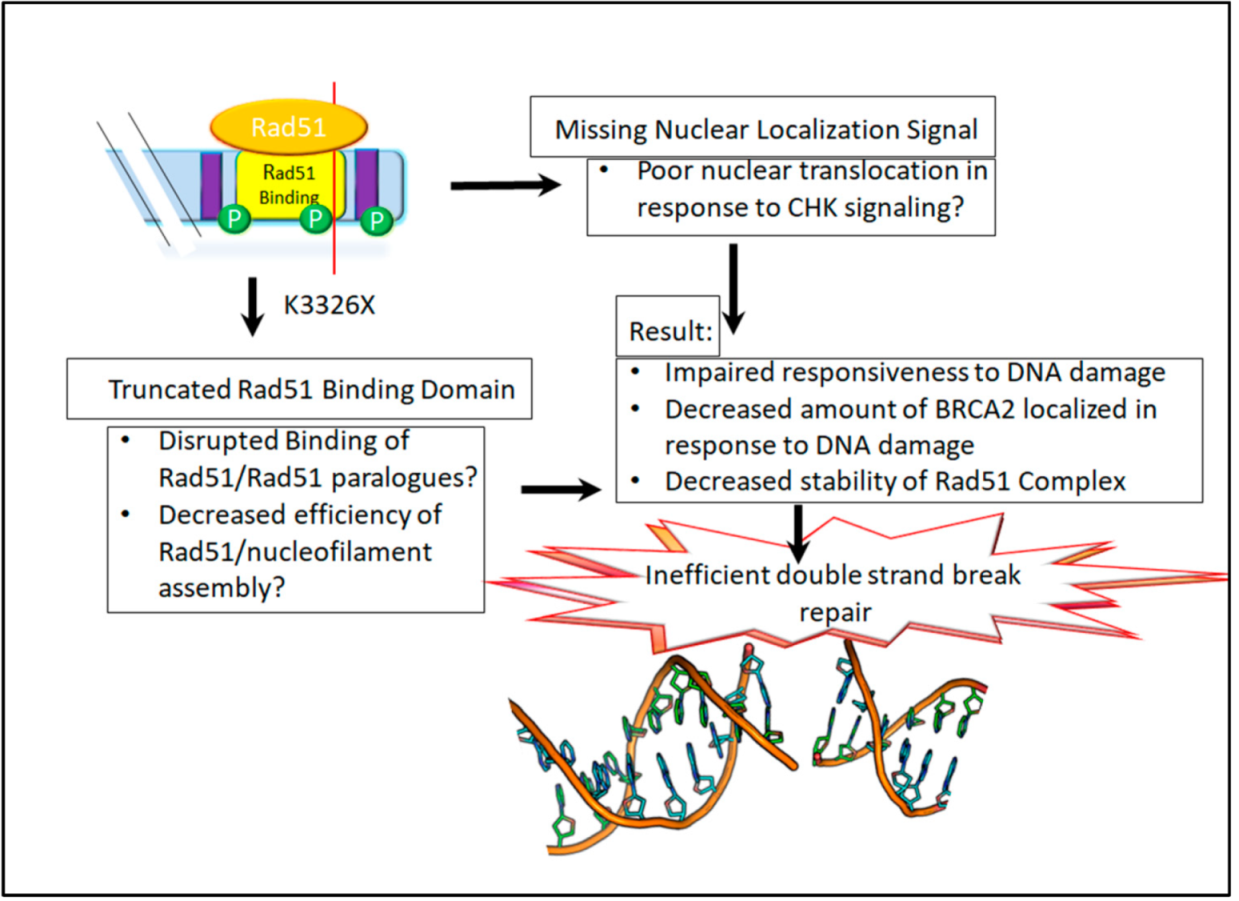
4. The K3326X Mutation
4.1. K3326X in Gynecologic Cancers
4.2. K3326X in Pancreatic Cancer
4.3. K3326X in Environmental Cancers
5. Summary of K3326X literature
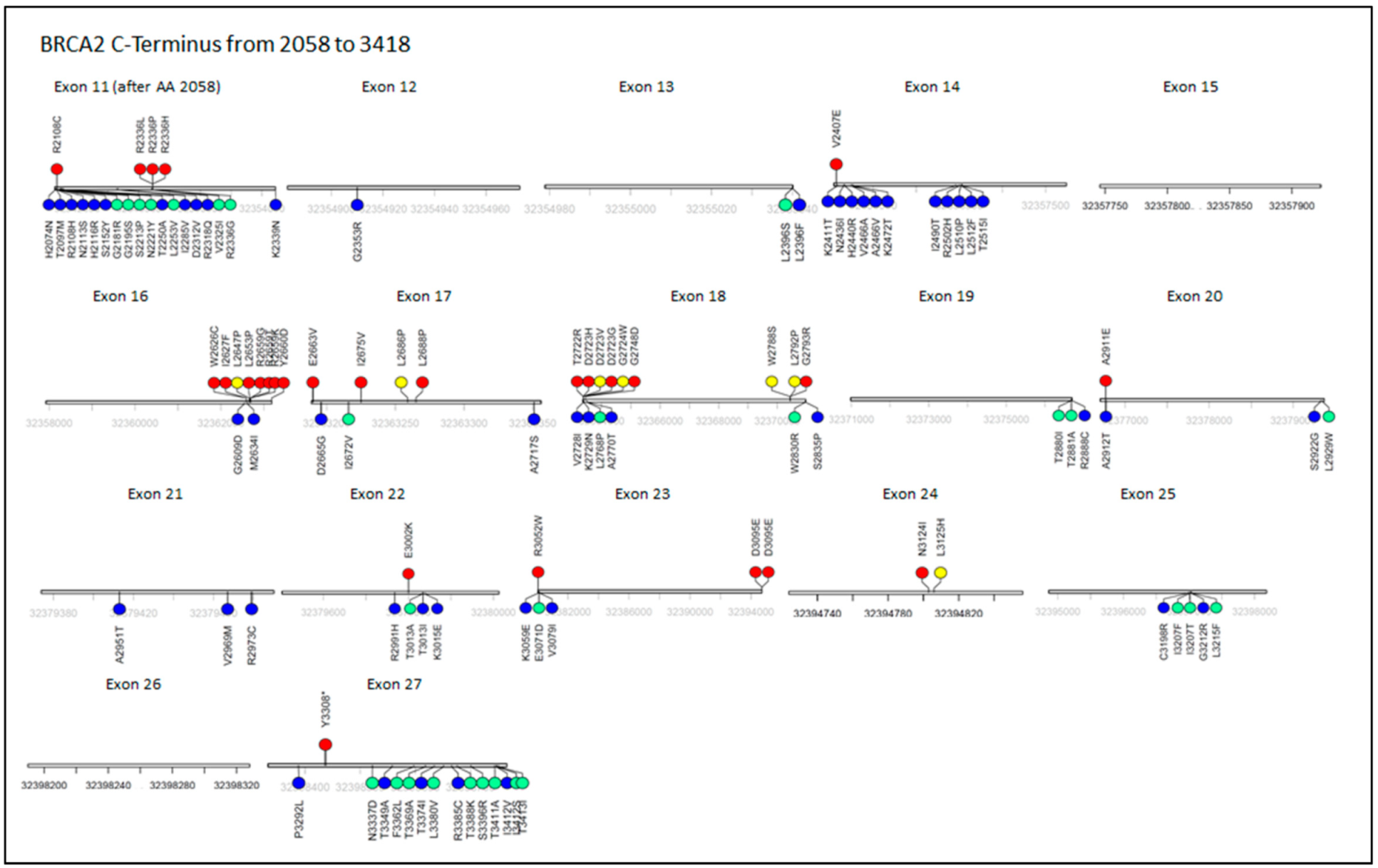
6. Other C-Terminal Mutations in BRCA2
7. New Ways to Analyze Mutations
8. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pennington, K.P.; Walsh, T.; Harrell, M.I.; Lee, M.K.; Pennil, C.C.; Rendi, M.H.; Thornton, A.; Norquist, B.M.; Casadei, S.; Nord, A.S.; et al. Germline and Somatic Mutations in Homologous Recombination Genes Predict Platinum Response and Survival in Ovarian, Fallopian Tube, and Peritoneal Carcinomas. Clin. Cancer Res. 2014, 20, 764–775. [Google Scholar] [CrossRef] [PubMed]
- Guidugli, L.; Carreira, A.; Caputo, S.M.; Ehlen, A.; Galli, A.; Monteiro, A.N.; Neuhausen, S.L.; Hansen, T.V.; Couch, F.J.; Vreeswijk, M.P.G.; et al. Functional Assays for Analysis of Variants of Uncertain Significance inBRCA2. Hum. Mutat. 2014, 35, 151–164. [Google Scholar] [CrossRef] [PubMed]
- Murphy, C.G.; Moynahan, M.E. BRCA Gene Structure and Function in Tumor Suppression. Cancer J. 2010, 16, 39–47. [Google Scholar] [CrossRef]
- Paull, T.T. Mechanisms of ATM Activation. Annu. Rev. Biochem. 2015, 84, 711–738. [Google Scholar] [CrossRef]
- Uziel, T.; Lerenthal, Y.; Moyal, L.; Andegeko, Y.; Mittelman, L.; Shiloh, Y. Requirement of the MRN complex for ATM activation by DNA damage. EMBO J. 2003, 22, 5612–5621. [Google Scholar] [CrossRef] [PubMed]
- Burma, S.; Chen, B.P.; Murphy, M.; Kurimasa, A.; Chen, D.J. ATM Phosphorylates Histone H2AX in Response to DNA Double-strand Breaks. J. Biol. Chem. 2001, 276, 42462–42467. [Google Scholar] [CrossRef] [PubMed]
- You, Z.; Chahwan, C.; Bailis, J.; Hunter, T.; Russell, P. ATM Activation and Its Recruitment to Damaged DNA Require Binding to the C Terminus of Nbs1. Mol. Cell. Biol. 2005, 25, 5363–5379. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Zhou, J.; Lim, C.U. The role of NBS1 in DNA double strand break repair, telomere stability, and cell cycle checkpoint control. Cell Res. 2006, 16, 45–54. [Google Scholar] [CrossRef] [PubMed]
- Lavin, M.F.; Kozlov, S.; Gatei, M.; Kijas, A.W. ATM-Dependent Phosphorylation of All Three Members of the MRN Complex: From Sensor to Adaptor. Biomolecules 2015, 5, 2877–2902. [Google Scholar] [CrossRef]
- Kobayashi, J.; Antoccia, A.; Tauchi, H.; Matsuura, S.; Komatsu, K. NBS1 and its functional role in the DNA damage response. DNA Repair 2004, 3, 855–861. [Google Scholar] [CrossRef]
- Lamarche, B.J.; Orazio, N.I.; Weitzman, M.D. The MRN complex in double-strand break repair and telomere maintenance. FEBS Lett. 2010, 584, 3682–3695. [Google Scholar] [CrossRef] [PubMed]
- You, Z.; Shi, L.Z.; Zhu, Q.; Wu, P.; Zhang, Y.-W.; Basilio, A.; Tonnu, N.; Verma, I.M.; Berns, M.W.; Hunter, T. CtIP Links DNA Double-Strand Break Sensing to Resection. Mol. Cell 2009, 36, 954–969. [Google Scholar] [CrossRef] [PubMed]
- D’Amours, D.; Jackson, S.P. The MRE11 complex: At the crossroads of DNA repair and checkpoint signalling. Nat. Rev. Mol. Cell Biol. 2002, 3, 317–327. [Google Scholar] [CrossRef] [PubMed]
- You, Z.; Bailis, J.M. DNA damage and decisions: CtIP coordinates DNA repair and cell cycle checkpoints. Trends Cell Biol. 2010, 20, 402–409. [Google Scholar] [CrossRef] [PubMed]
- Ma, C.J.; Gibb, B.; Kwon, Y.; Sung, P.; Greene, E.C. Protein dynamics of human RPA and RAD51 on ssDNA during assembly and disassembly of the RAD51 filament. Nucleic Acids Res. 2017, 45, 749–761. [Google Scholar] [CrossRef] [PubMed]
- Jensen, R.B.; Carreira, A.; Kowalczykowski, S.C. Purified human BRCA2 stimulates RAD51-mediated recombination. Nat. Cell Biol. 2010, 467, 678–683. [Google Scholar] [CrossRef]
- Godin, S.K.; Sullivan, M.R.; Bernstein, K.A. Novel insights into RAD51 activity and regulation during homologous recombination and DNA replication. Biochem. Cell Biol. 2016, 94, 407–418. [Google Scholar] [CrossRef]
- Sullivan, M.R.; Bernstein, K.A. RAD-ical New Insights into RAD51 Regulation. Genes 2018, 9, 629. [Google Scholar] [CrossRef]
- Mailand, N.; Gibbs-Seymour, I.; Bekker-Jensen, S. Regulation of PCNA–protein interactions for genome stability. Nat. Rev. Mol. Cell Biol. 2013, 14, 269–282. [Google Scholar] [CrossRef]
- Kelman, Z. PCNA: Structure, functions and interactions. Oncogene 1997, 14, 629–640. [Google Scholar] [CrossRef]
- Choe, K.N.; Moldovan, G.-L. Forging Ahead through Darkness: PCNA, Still the Principal Conductor at the Replication Fork. Mol. Cell 2017, 65, 380–392. [Google Scholar] [CrossRef] [PubMed]
- Roy, R.; Chun, J.; Powell, S.N. BRCA1 and BRCA2: Different roles in a common pathway of genome protection. Nat. Rev. Cancer 2011, 12, 68–78. [Google Scholar] [CrossRef] [PubMed]
- Costanzo, V. Brca2, Rad51 and Mre11: Performing balancing acts on replication forks. DNA Repair 2011, 10, 1060–1065. [Google Scholar] [CrossRef]
- Evers, B.M.; Jonkers, J. Mouse models of BRCA1 and BRCA2 deficiency: Past lessons, current understanding and future prospects. Oncogene 2006, 25, 5885–5897. [Google Scholar] [CrossRef]
- Martinez, J.S.; Baldeyron, C.; Carreira, A. Molding BRCA2 function through its interacting partners. Cell Cycle 2015, 14, 3389–3395. [Google Scholar] [CrossRef] [PubMed]
- Krejci, L.; Altmannova, V.; Spirek, M.; Zhao, X. Homologous recombination and its regulation. Nucleic Acids Res. 2012, 40, 5795–5818. [Google Scholar] [CrossRef]
- Tavtigian, S.V.; Simard, J.; Rommens, J.M.; Couch, F.J.; Shattuck-Eidens, D.; Neuhausen, S.L.; Merajver, S.D.; Thorlacius, S.; Offit, K.; Stoppalyonnet, D.; et al. The complete BRCA2 gene and mutations in chromosome 13q-linked kindreds. Nat. Genet. 1996, 12, 333–337. [Google Scholar] [CrossRef]
- Chatterjee, G.; Jimenez-Sainz, J.; Presti, T.; Nguyen, T.; Jensen, R.B. Distinct binding of BRCA2 BRC repeats to RAD51 generates differential DNA damage sensitivity. Nucleic Acids Res. 2016, 44, 5256–5270. [Google Scholar] [CrossRef]
- Davies, O.R.; Pellegrini, L. Interaction with the BRCA2 C terminus protects RAD51–DNA filaments from disassembly by BRC repeats. Nat. Struct. Mol. Biol. 2007, 14, 475–483. [Google Scholar] [CrossRef]
- Kolinjivadi, A.M.; Sannino, V.; De Antoni, A.; Técher, H.; Baldi, G.; Costanzo, V. Moonlighting at replication forks - a new life for homologous recombination proteins BRCA1, BRCA2 and RAD51. FEBS Lett. 2017, 591, 1083–1100. [Google Scholar] [CrossRef]
- Yano, K.-I.; Morotomi, K.; Saito, H.; Kato, M.; Matsuo, F.; Miki, Y. Nuclear Localization Signals of the BRCA2 Protein. Biochem. Biophys. Res. Commun. 2000, 270, 171–175. [Google Scholar] [CrossRef] [PubMed]
- Spain, B.H.; Larson, C.J.; Shihabuddin, L.S.; Gage, F.H.; Verma, I.M. Truncated BRCA2 is cytoplasmic: Implications for cancer-linked mutations. Proc. Natl. Acad. Sci. USA 1999, 96, 13920–13925. [Google Scholar] [CrossRef] [PubMed]
- Bahassi, E.M.; Ovesen, J.L.; Riesenberg, A.L.; Bernstein, W.Z.; E Hasty, P.; Stambrook, P.J. The checkpoint kinases Chk1 and Chk2 regulate the functional associations between hBRCA2 and Rad51 in response to DNA damage. Oncogene 2008, 27, 3977–3985. [Google Scholar] [CrossRef] [PubMed]
- Rajagopalan, S.; Andreeva, A.; Rutherford, T.J.; Fersht, A.R. Mapping the physical and functional interactions between the tumor suppressors p53 and BRCA2. Proc. Natl. Acad. Sci. USA 2010, 107, 8587–8592. [Google Scholar] [CrossRef] [PubMed]
- Chandramouly, G.; Willis, N.; Scully, R. A protective role for BRCA2 at stalled replication forks. Breast Cancer Res. 2011, 13, 314. [Google Scholar] [CrossRef]
- Martinez, J.S.; Von Nicolai, C.; Kim, T.; Ehlén, Å.; Mazin, A.V.; Kowalczykowski, S.C.; Carreira, A. BRCA2 regulates DMC1-mediated recombination through the BRC repeats. Proc. Natl. Acad. Sci. USA 2016, 113, 3515–3520. [Google Scholar] [CrossRef]
- Thorslund, T.; Esashi, F.; West, S.C. Interactions between human BRCA2 protein and the meiosis-specific recombinase DMC1. EMBO J. 2007, 26, 2915–2922. [Google Scholar] [CrossRef]
- Jeyasekharan, A.D.; Liu, Y.; Hattori, H.; Pisupati, V.; Jonsdottir, A.B.; Rajendra, E.; Lee, M.; Sundaramoorthy, E.; Schlachter, S.; Kaminski, C.F.; et al. A cancer-associated BRCA2 mutation reveals masked nuclear export signals controlling localization. Nat. Struct. Mol. Biol. 2013, 20, 1191–1198. [Google Scholar] [CrossRef]
- Landrum, M.J.; Lee, J.M.; Benson, M.; Brown, G.R.; Chao, C.; Chitipiralla, S.; Gu, B.; Hart, J.; Hoffman, D.; Jang, W.; et al. ClinVar: Improving access to variant interpretations and supporting evidence. Nucleic Acids Res. 2018, 46, D1062–D1067. [Google Scholar] [CrossRef]
- Kim, T.M.; Son, M.Y.; Dodds, S.G.; Hu, L.; Hasty, P. Deletion of BRCA2 exon 27 causes defects in response to both stalled and collapsed replication forks. Mutat. Res. Mol. Mech. Mutagen. 2014, 767, 66–72. [Google Scholar] [CrossRef]
- Schlacher, K.; Christ, N.; Siaud, N.; Egashira, A.; Wu, H.; Jasin, M. Double-Strand Break Repair-Independent Role for BRCA2 in Blocking Stalled Replication Fork Degradation by MRE11. Cell 2011, 145, 529–542. [Google Scholar] [CrossRef] [PubMed]
- Meeks, H.D.; Song, H.; Michailidou, K.; Bolla, M.K.; Dennis, J.; Wang, Q.; Barrowdale, D.; Frost, D.; McGuffog, L.; Ellis, S.; et al. BRCA2 Polymorphic Stop Codon K3326X and the Risk of Breast, Prostate, and Ovarian Cancers. J. Natl. Cancer Inst. 2015, 108, djv315. [Google Scholar] [CrossRef] [PubMed]
- Arbustini, E.; Sgarella, A.; Ferrari, A.; Grasso, D.; Cassani, C.; Lucioni, M.; Di Giulio, G.; Grasso, M. RE: BRCA2 Polymorphic Stop Codon K3326X and the Risk of Breast, Prostate, and Ovarian Cancers. J. Natl. Cancer Inst. 2016, 108, djw172. [Google Scholar] [CrossRef]
- Palmirotta, R.; Lovero, D.; Stucci, L.S.; Silvestris, E.; Quaresmini, D.; Cardascia, A.; Silvestris, F. Double Heterozygosity for BRCA1 Pathogenic Variant and BRCA2 Polymorphic Stop Codon K3326X: A Case Report in a Southern Italian Family. Int. J. Mol. Sci. 2018, 19, 285. [Google Scholar] [CrossRef] [PubMed]
- A McAllister, K.; Bennett, L.M.; Houle, C.D.; Ward, T.; Malphurs, J.; Collins, N.K.; Cachafeiro, C.; Haseman, J.; Goulding, E.H.; Bunch, D.; et al. Cancer susceptibility of mice with a homozygous deletion in the COOH-terminal domain of the Brca2 gene. Cancer Res. 2002, 62, 990–994. [Google Scholar]
- Lopes, J.L.; Dyson, G.; Levin, N.K.; Chaudhry, S.; Rosati, R.; Kalpage, H.; Wernette, C.; Petrucelli, N.; Simon, M.S.; Tainsky, M.A. Reanalysis of BRCA1/2 negative high risk ovarian cancer patients reveals novel germline risk loci and insights into missing heritability. PLoS ONE 2017, 12, e0178450. [Google Scholar] [CrossRef]
- Martin, S.T.; Matsubayashi, H.; Rogers, C.D.; Philips, J.; Couch, F.J.; Brune, K.; Yeo, C.J.; E Kern, S.; Hruban, R.H.; Goggins, M. Increased prevalence of the BRCA2 polymorphic stop codon K3326X among individuals with familial pancreatic cancer. Oncogene 2005, 24, 3652–3656. [Google Scholar] [CrossRef]
- Obazee, O.; Archibugi, L.; Andriulli, A.; Soucek, P.; Małecka-Panas, E.; Ivanauskas, A.; Johnson, T.; Gazouli, M.; Pausch, T.; Lawlor, R.T.; et al. Germline BRCA2 K3326X and CHEK2 I157T mutations increase risk for sporadic pancreatic ductal adenocarcinoma. Int. J. Cancer 2019, 145, 686–693. [Google Scholar] [CrossRef]
- Rafnar, T.; Sigurjonsdottir, G.R.; Stacey, S.N.; Halldorsson, G.H.; Sulem, P.; Pardo, L.M.; Helgason, H.; Sigurdsson, S.T.; Gudjonsson, T.; Tryggvadottir, L.; et al. Association of BRCA2 K3326* With Small Cell Lung Cancer and Squamous Cell Cancer of the Skin. J. Natl. Cancer Inst. 2018, 110, 967–974. [Google Scholar] [CrossRef]
- Delahaye-Sourdeix, M.; Anantharaman, D.; Timofeeva, M.N.; Gaborieau, V.; Chabrier, A.; Vallée, M.P.; Lagiou, P.; Holcátová, I.; Richiardi, L.; Kjaerheim, K.; et al. A Rare Truncating BRCA2 Variant and Genetic Susceptibility to Upper Aerodigestive Tract Cancer. J. Natl. Cancer Inst. 2015, 107. [Google Scholar] [CrossRef]
- Sarin, K.Y.; Lin, Y.; Daneshjou, R.; Ziyatdinov, A.; Thorleifsson, G.; Rubin, A.; Pardo, L.M.; Wu, W.; Khavari, P.A.; Uitterlinden, A.G.; et al. Genome-wide meta-analysis identifies eight new susceptibility loci for cutaneous squamous cell carcinoma. Nat. Commun. 2020, 11, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Akbari, M.R.; Malekzadeh, R.; Lepage, P.; Roquis, D.; Sadjadi, A.R.; Aghcheli, K.; Yazdanbod, A.; Shakeri, R.; Bashiri, J.; Sotoudeh, M.; et al. Mutations in Fanconi anemia genes and the risk of esophageal cancer. Qual. Life Res. 2011, 129, 573–582. [Google Scholar] [CrossRef] [PubMed]
- Ko, J.M.-Y.; Ning, L.; Zhao, X.; Chai, A.W.Y.; Lei, L.C.; Choi, S.S.A.; Tao, L.; Law, S.; Kwong, A.; Lee, N.P.; et al. BRCA2 loss-of-function germline mutations are associated with esophageal squamous cell carcinoma risk in Chinese. Int. J. Cancer 2020, 146, 1042–1051. [Google Scholar] [CrossRef] [PubMed]
- Rudd, M.F.; Webb, E.L.; Matakidou, A.; Sellick, G.S.; Williams, R.D.; Bridle, H.; Eisen, T.; Houlston, R.S. Variants in the GH-IGF axis confer susceptibilityto lung cancer. Genome Res. 2006, 16, 693–701. [Google Scholar] [CrossRef] [PubMed]
- Thompson, E.R.; Gorringe, K.; Rowley, S.M.; Li, N.; McInerny, S.; Wong-Brown, M.; Devereux, L.; Li, J.; Trainer, A.H.; Mitchell, G.; et al. Reevaluation of the BRCA2 truncating allele c.9976A > T (p.Lys3326Ter) in a familial breast cancer context. Sci. Rep. 2015, 5, 14800. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; McKay, J.D.; Rafnar, T.; Wang, Z.; Timofeeva, M.N.; Broderick, P.; Zong, X.; Laplana, M.; Wei, Y.; Han, Y.; et al. Rare variants of large effect in BRCA2 and CHEK2 affect risk of lung cancer. Nat. Genet. 2014, 46, 736–741. [Google Scholar] [CrossRef]
- Cerami, E.; Gao, J.; Dogrusoz, U.; Gross, B.E.; Sumer, S.O.; Aksoy, B.A.; Jacobsen, A.; Byrne, C.J.; Heuer, M.L.; Larsson, E.; et al. The cBio Cancer Genomics Portal: An Open Platform for Exploring Multidimensional Cancer Genomics Data: Figure 1. Cancer Discov. 2012, 2, 401–404. [Google Scholar] [CrossRef]
- Gao, J.; Aksoy, B.A.; Dogrusoz, U.; Dresdner, G.; Gross, B.; Sumer, S.O.; Sun, Y.; Jacobsen, A.; Sinha, R.; Larsson, E.; et al. Integrative Analysis of Complex Cancer Genomics and Clinical Profiles Using the cBioPortal. Sci. Signal. 2013, 6, pl1. [Google Scholar] [CrossRef]
- Carvalho, S.D.C.E.S.; Cury, N.M.; Brotto, D.B.; De Araujo, L.F.; Rosa, R.C.A.; Texeira, L.A.; Plaça, J.R.; Marques, A.A.; Peronni, K.C.; Ruy, P.D.C.; et al. Germline variants in DNA repair genes associated with hereditary breast and ovarian cancer syndrome: Analysis of a 21 gene panel in the Brazilian population. BMC Med. Genom. 2020, 13, 1–24. [Google Scholar] [CrossRef]
- Kawahara, M.; Sakayori, M.; Shiraishi, K.; Nomizu, T.; Takeda, M.; Abe, R.; Ohuchi, N.; Takenoshita, S.; Ishioka, C. Identification and evaluation of 55 genetic variations in the BRCA1 and the BRCA2 genes of patients from 50 Japanese breast cancer families. J. Hum. Genet. 2004, 49, 391–395. [Google Scholar] [CrossRef][Green Version]
- Konecny, M.; Milly, M.; Zavodna, K.; Weismanová, E.; Gregorova, J.; Mlkva, I.; Ilencikova, D.; Kausitz, J.; Bartosova, Z. Comprehensive genetic characterization of hereditary breast/ovarian cancer families from Slovakia. Breast Cancer Res. Treat. 2011, 126, 119–130. [Google Scholar] [CrossRef][Green Version]
- Hansen, T.V.O.; Jønson, L.; Albrechtsen, A.; Andersen, M.K.; Ejlertsen, B.; Nielsen, F.C. Large BRCA1 and BRCA2 genomic rearrangements in Danish high risk breast-ovarian cancer families. Breast Cancer Res. Treat. 2008, 115, 315–323. [Google Scholar] [CrossRef]
- Cherbal, F.; Salhi, N.; Bakour, R.; Adane, S.; Boualga, K.; Maillet, P. BRCA1 and BRCA2 Unclassified Variants and Missense Polymorphisms in Algerian Breast/Ovarian Cancer Families. Dis. Markers 2012, 32, 343–353. [Google Scholar] [CrossRef]
- Agata, S.; Tognazzo, S.; Alducci, E.; Matricardi, L.; Moserle, L.; Barana, D.; Montagna, M. Segregation analysis of the BRCA2 c.9227G>T variant in multiple families suggests a pathogenic role in breast and ovarian cancer predisposition. Sci. Rep. 2020, 10, 1–6. [Google Scholar] [CrossRef]
- Balabanski, L.; Antov, G.; Dimova, I.; Ivanov, S.; Nacheva, M.; Gavrilov, I.; Nesheva, D.; Rukova, B.; Hadjidekova, S.; Malinov, M.; et al. Next-generation sequencing of BRCA1 and BRCA2 in breast cancer patients and control subjects. Mol. Clin. Oncol. 2014, 2, 435–439. [Google Scholar] [CrossRef][Green Version]
- Federici, G.; Soddu, S. Variants of uncertain significance in the era of high-throughput genome sequencing: A lesson from breast and ovary cancers. J. Exp. Clin. Cancer Res. 2020, 39, 1–12. [Google Scholar] [CrossRef]
- Moynahan, M.E.; Pierce, A.J.; Jasin, M. BRCA2 Is Required for Homology-Directed Repair of Chromosomal Breaks. Mol. Cell 2001, 7, 263–272. [Google Scholar] [CrossRef]
- Wu, K.; Hinson, S.R.; Ohashi, A.; Farrugia, D.; Wendt, P.; Tavtigian, S.V.; Deffenbaugh, A.; Goldgar, D.; Couch, F.J. Functional evaluation and cancer risk assessment of BRCA2 unclassified variants. Cancer Res. 2005, 65, 417–426. [Google Scholar]
- Kuznetsov, S.G.; Liu, P.; Sharan, S.K. Mouse embryonic stem cell–based functional assay to evaluate mutations in BRCA2. Nat. Med. 2008, 14, 875–881. [Google Scholar] [CrossRef]
- Alanazi, I.O.; Al Shehri, Z.S.; Ebrahimie, E.; Giahi, H.; Mohammadi-Dehcheshmeh, M. Non-coding and coding genomic variants distinguish prostate cancer, castration-resistant prostate cancer, familial prostate cancer, and metastatic castration-resistant prostate cancer from each other. Mol. Carcinog. 2019, 58, 862–874. [Google Scholar] [CrossRef]
- Wunderle, M.; Olmes, G.; Nabieva, N.; Häberle, L.; Jud, S.M.; Hein, A.; Rauh, C.; Hack, C.C.; Erber, R.; Ekici, A.B.; et al. Risk, Prediction and Prevention of Hereditary Breast Cancer – Large-Scale Genomic Studies in Times of Big and Smart Data. Geburtshilfe und Frauenheilkd. 2018, 78, 481–492. [Google Scholar] [CrossRef] [PubMed]
- Ingham, S.L.; Warwick, J.; Byers, H.; Lalloo, F.; Newman, W.; Evans, D.; Newman, W.G.; Evans, D.G. Is multiple SNP testing inBRCA2andBRCA1female carriers ready for use in clinical practice? Results from a large Genetic Centre in the UK. Clin. Genet. 2012, 84, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Prosperi, M.; Ingham, S.L.; Howell, A.; Lalloo, F.; Buchan, I.; Evans, D.G. Can multiple SNP testing in BRCA2 and BRCA1 female carriers be used to improve risk prediction models in conjunction with clinical assessment? BMC Med. Informatics Decis. Mak. 2014, 14, 87. [Google Scholar] [CrossRef] [PubMed]
- Basu, N.N.; Ingham, S.; Hodson, J.; Lalloo, F.; Bulman, M.; Howell, A.; Evans, D.G. Risk of contralateral breast cancer in BRCA1 and BRCA2 mutation carriers: A 30-year semi-prospective analysis. Fam. Cancer 2015, 14, 531–538. [Google Scholar] [CrossRef]
- Ikegami, M.; Kohsaka, S.; Ueno, T.; Momozawa, Y.; Inoue, S.; Tamura, K.; Shimomura, A.; Hosoya, N.; Kobayashi, H.; Tanaka, S.; et al. High-throughput functional evaluation of BRCA2 variants of unknown significance. Nat. Commun. 2020, 11, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Mesman, R.L.S.; Msc, F.M.G.R.C.; Hendriks, G.; Morolli, B.; Misovic, B.; Devilee, P.; Van Asperen, C.J.; Vrieling, H.; Vreeswijk, M.P.G. The functional impact of variants of uncertain significance in BRCA2. Genet. Med. 2019, 21, 293–302. [Google Scholar] [CrossRef] [PubMed]
- Biswas, K.; Lipton, G.B.; Stauffer, S.; Sullivan, T.; Cleveland, L.; Southon, E.; Reid, S.; Magidson, V.; Iversen, E.S.; Sharan, S.K. A computational model for classification of BRCA2 variants using mouse embryonic stem cell-based functional assays. npj Genom. Med. 2020, 5, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Yadegari, F.; Majidzadeh, K. In silico analysis for determining the deleterious nonsynonymous single nucleotide polymorphisms of BRCA genes. Mol. Biol. Res. Commun. 2019, 8, 141–150. [Google Scholar]
- Hirotsu, Y.; Schmidt-Edelkraut, U.; Nakagomi, H.; Sakamoto, I.; Hartenfeller, M.; Narang, R.; Soldatos, T.G.; Kaduthanam, S.; Wang, X.; Hettich, S.; et al. Consolidated BRCA1/2 Variant Interpretation by MH BRCA Correlates with Predicted PARP Inhibitor Efficacy Association by MH Guide. Int. J. Mol. Sci. 2020, 21, 3895. [Google Scholar] [CrossRef]
- Riahi, A.; Messaoudi, A.; Mrad, R.; Fourati, A.; Chabouni-Bouhamed, H.; Kharrat, M. Molecular characterization, homology modeling and docking studies of the R2787H missense variation in BRCA2 gene: Association with breast cancer. J. Theor. Biol. 2016, 403, 188–196. [Google Scholar] [CrossRef]
- Esposito, M.V.; Minopoli, G.; Esposito, L.; D’Argenio, V.; Di Maggio, F.; Sasso, E.; D’Aiuto, M.; Zambrano, N.; Salvatore, F. A Functional Analysis of the Unclassified Pro2767Ser BRCA2 Variant Reveals Its Potential Pathogenicity that Acts by Hampering DNA Binding and Homology-Mediated DNA Repair. Cancers 2019, 11, 1454. [Google Scholar] [CrossRef] [PubMed]
- Dines, J.N.; Shirts, B.H.; Slavin, T.P.; Walsh, T.; King, M.-C.; Fowler, D.M.; Pritchard, C.C.; Shirts, B.H.; Pritchard, C.C. Systematic misclassification of missense variants in BRCA1 and BRCA2 “coldspots”. Genet. Med. 2020, 22, 825–830. [Google Scholar] [CrossRef]
- Enginler, S.O.; Akış, I.; Toydemir, T.S.F.; Oztabak, K.; Haktanir, D.; Gündüz, M.C.; Kırşan, I.; Fırat, I. Genetic variations of BRCA1 and BRCA2 genes in dogs with mammary tumours. Veter-Res. Commun. 2014, 38, 21–27. [Google Scholar] [CrossRef]
- Warren, M.; Smith, A.; Partridge, N.; Masabanda, J.; Griffin, D.; Ashworth, A. Structural analysis of the chicken BRCA2 gene facilitates identification of functional domains and disease causing mutations. Hum. Mol. Genet. 2002, 11, 841–851. [Google Scholar] [CrossRef] [PubMed]
| Reference | Cancer Type, [Odds Ratio] | p-Value | Population (Number) | Recommendation |
|---|---|---|---|---|
| [42,43] | Breast (1.28) | 5.9 × 10−6, 3.8 × 10−3, 3.4 × 10−5, 4.1 × 10−5 | 70,000 cases | Expanded counseling |
| Invasive ovarian (1.26) | 80,000 controls | |||
| Serous ovarian (1.46) | - | |||
| ER-negative breast (1.5) | - | |||
| [44] | Early onset | N/A | Small (case study) | Further evaluation |
| [46] | Ovarian cancer (4.95) | <0.01 | 48 | Treat as low risk pathogenic |
| [47] | Familial pancreatic (4.84) | <0.01 | 114 familial pancreatic cancer | None |
| 115 environmental control | ||||
| 125 controls | ||||
| [48] | Pancreatic ductal adenocarcinoma (1.78) | 0.0012 | 2935 cases | None |
| 5626 controls | ||||
| [49] | Small cell lung (2.06) | 9 × 10−4, 4.2 × 10−4, 1.2 × 10−4, 1.6 × 10−5 | 43,641 cases 370,971 controls | Vulnerability to environmental cancers |
| Squamous skin (1.69) | ||||
| Lung cancer (1.54) | ||||
| All cancers (1.23) | ||||
| [50] | Upper aero digestive tract (2.53) | 3 × 10−10 | 5942 cases | Warrants further investigation |
| 8086 controls | ||||
| [51] | Cutaneous Squamous cell carcinoma (2.29) | 1.0 × 10−6 | Meta-analysis of 19,149 cases, 680,049 controls | Variant likely implicated in skin cancer development |
| [52] | Esophageal squamous cell carcinoma (3.38) | 0.0002 | 190 cases | None |
| 1373 controls | ||||
| [53] | Esophageal cancer | N/A | 2276 cases | Rare variant in Henan and Hong Kong ESCC patients |
| 2058 controls | ||||
| [54] | Lung Cancer (1.72) | 0.0075 | 1529 cases | Low penetrance alleles contribute to risk |
| 2707 controls | ||||
| [55] | Breast Cancer (1.53) | 0.047 | 2634 cases | Variant is not neutral, should be included in SNP panels for evaluating risk |
| 1996 controls | ||||
| [56] | Squamous lung cancer (2.47) | 4.7 × 10−20 | 10,246 cases | K3326X may have a direct effect on lung cancer development |
| 38,295 controls |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Baughan, S.; Tainsky, M.A. K3326X and Other C-Terminal BRCA2 Variants Implicated in Hereditary Cancer Syndromes: A Review. Cancers 2021, 13, 447. https://doi.org/10.3390/cancers13030447
Baughan S, Tainsky MA. K3326X and Other C-Terminal BRCA2 Variants Implicated in Hereditary Cancer Syndromes: A Review. Cancers. 2021; 13(3):447. https://doi.org/10.3390/cancers13030447
Chicago/Turabian StyleBaughan, Scott, and Michael A. Tainsky. 2021. "K3326X and Other C-Terminal BRCA2 Variants Implicated in Hereditary Cancer Syndromes: A Review" Cancers 13, no. 3: 447. https://doi.org/10.3390/cancers13030447
APA StyleBaughan, S., & Tainsky, M. A. (2021). K3326X and Other C-Terminal BRCA2 Variants Implicated in Hereditary Cancer Syndromes: A Review. Cancers, 13(3), 447. https://doi.org/10.3390/cancers13030447






