Actual 10-Year Survival after Resection of Perihilar Cholangiocarcinoma: What Factors Preclude a Chance for Cure?
Abstract
Simple Summary
Abstract
1. Introduction
2. Methods
2.1. Patient Work-Up and Management
2.2. Definitions
2.3. Statistical Analysis
3. Results
3.1. Patient Characteristics
3.2. Overall Survival
3.3. Actual 10-Year Survivors
3.4. Observed Cure and Cure Model
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Lewis, H.L.; Rahnemai-Azar, A.A.; Dillhoff, M.; Schmidt, C.R.; Pawlik, T.M. Current management of perihilar cholangiocarcinoma and future perspectives. Chirurgia 2017, 112, 193–207. [Google Scholar] [CrossRef] [PubMed]
- Rassam, F.; Roos, E.; van Lienden, K.P.; van Hooft, J.E.; Klumpen, H.J.; van Tienhoven, G.; Bennink, R.J.; Engelbrecht, M.R.; Schoorlemmer, A.; Beuers, U.H.W.; et al. Modern work-up and extended resection in perihilar cholangiocarcinoma: The amc experience. Langenbecks Arch. Surg. 2018, 403, 289–307. [Google Scholar] [CrossRef]
- Jarnagin, W.R.; Fong, Y.; DeMatteo, R.P.; Gonen, M.; Burke, E.C.; Bodniewicz, B.J.; Youssef, B.M.; Klimstra, D.; Blumgart, L.H. Staging, resectability, and outcome in 225 patients with hilar cholangiocarcinoma. Ann. Surg. 2001, 234, 507–517; discussion 17–19. [Google Scholar] [CrossRef]
- Matsuo, K.; Rocha, F.G.; Ito, K.; D’Angelica, M.I.; Allen, P.J.; Fong, Y.; Dematteo, R.P.; Gonen, M.; Endo, I.; Jarnagin, W.R. The blumgart preoperative staging system for hilar cholangiocarcinoma: Analysis of resectability and outcomes in 380 patients. J. Am. Coll. Surg. 2012, 215, 343–355. [Google Scholar] [CrossRef]
- Liu, C.L.; Fan, S.T.; Lo, C.M.; Tso, W.K.; Lam, C.M.; Wong, J. Improved operative and survival outcomes of surgical treatment for hilar cholangiocarcinoma. Br. J. Surg. 2006, 93, 1488–1494. [Google Scholar] [CrossRef] [PubMed]
- Nagino, M.; Ebata, T.; Yokoyama, Y.; Igami, T.; Sugawara, G.; Takahashi, Y.; Nimura, Y. Evolution of surgical treatment for perihilar cholangiocarcinoma: A single-center 34-year review of 574 consecutive resections. Ann. Surg. 2013, 258, 129–140. [Google Scholar] [CrossRef]
- Nuzzo, G.; Giuliante, F.; Ardito, F.; Giovannini, I.; Aldrighetti, L.; Belli, G.; Bresadola, F.; Calise, F.; Dalla Valle, R.; D’Amico, D.F.; et al. Improvement in perioperative and long-term outcome after surgical treatment of hilar cholangiocarcinoma: Results of an italian multicenter analysis of 440 patients. Arch. Surg. 2012, 147, 26–34. [Google Scholar] [CrossRef]
- Popescu, I.; Dumitrascu, T. Curative-intent surgery for hilar cholangiocarcinoma: Prognostic factors for clinical decision making. Langenbecks Arch. Surg. 2014, 399, 693–705. [Google Scholar] [CrossRef]
- Burke, E.C.; Jarnagin, W.R.; Hochwald, S.N.; Pisters, P.W.; Fong, Y.; Blumgart, L.H. Hilar cholangiocarcinoma: Patterns of spread, the importance of hepatic resection for curative operation, and a presurgical clinical staging system. Ann. Surg. 1998, 228, 385–394. [Google Scholar] [CrossRef]
- Saxena, A.; Chua, T.C.; Chu, F.C.; Morris, D.L. Improved outcomes after aggressive surgical resection of hilar cholangiocarcinoma: A critical analysis of recurrence and survival. Am. J. Surg. 2011, 202, 310–320. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.J.; Jin, Y.W.; Shrestha, A.; Ma, W.J.; Wang, J.K.; Liu, F.; Zhu, Y.Y.; Zhou, R.X.; Regmi, P.; Cheng, N.S.; et al. Predictive factors of early recurrence after r0 resection of hilar cholangiocarcinoma: A single institution experience in china. Cancer Med. 2019, 8, 1567–1575. [Google Scholar] [CrossRef] [PubMed]
- Creasy, J.M.; Sadot, E.; Koerkamp, B.G.; Chou, J.F.; Gonen, M.; Kemeny, N.E.; Balachandran, V.P.; Kingham, T.P.; DeMatteo, R.P.; Allen, P.J.; et al. Actual 10-year survival after hepatic resection of colorectal liver metastases: What factors preclude cure? Surgery 2018, 163, 1238–1244. [Google Scholar] [CrossRef] [PubMed]
- Coelen, R.J.S.; Roos, E.; Wiggers, J.K.; Besselink, M.G.; Buis, C.I.; Busch, O.R.C.; Dejong, C.H.C.; van Delden, O.M.; van Eijck, C.H.J.; Fockens, P.; et al. Endoscopic versus percutaneous biliary drainage in patients with resectable perihilar cholangiocarcinoma: A multicentre, randomised controlled trial. Lancet Gastroenterol. Hepatol. 2018, 3, 681–690. [Google Scholar] [CrossRef]
- van der Gaag, N.A.; Rauws, E.A.; van Eijck, C.H.; Bruno, M.J.; van der Harst, E.; Kubben, F.J.; Gerritsen, J.J.; Greve, J.W.; Gerhards, M.F.; de Hingh, I.H.; et al. Preoperative biliary drainage for cancer of the head of the pancreas. N. Engl. J. Med. 2010, 362, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Edge, S.B.; Byrd, D.R.; Carducci, M.A.; Compton, C.C.; Fritz, A.; Greene, F. Ajcc Cancer Staging Manual; Springer: New York, NY, USA, 2010; Volume 649. [Google Scholar]
- Groot Koerkamp, B.; Wiggers, J.K.; Gonen, M.; Doussot, A.; Allen, P.J.; Besselink, M.G.H.; Blumgart, L.H.; Busch, O.R.C.; D’Angelica, M.I.; DeMatteo, R.P.; et al. Survival after resection of perihilar cholangiocarcinoma-development and external validation of a prognostic nomogram. Ann. Oncol. 2015, 26, 1930–1935. [Google Scholar] [CrossRef]
- Ebata, T.; Kosuge, T.; Hirano, S.; Unno, M.; Yamamoto, M.; Miyazaki, M.; Kokudo, N.; Miyagawa, S.; Takada, T.; Nagino, M. Proposal to modify the international union against cancer staging system for perihilar cholangiocarcinomas. Br. J. Surg. 2014, 101, 79–88. [Google Scholar] [CrossRef]
- Aoba, T.; Ebata, T.; Yokoyama, Y.; Igami, T.; Sugawara, G.; Takahashi, Y.; Nimura, Y.; Nagino, M. Assessment of nodal status for perihilar cholangiocarcinoma: Location, number, or ratio of involved nodes. Ann. Surg. 2013, 257, 718–725. [Google Scholar] [CrossRef]
- DeOliveira, M.L.; Cunningham, S.C.; Cameron, J.L.; Kamangar, F.; Winter, J.M.; Lillemoe, K.D.; Choti, M.A.; Yeo, C.J.; Schulick, R.D. Cholangiocarcinoma: Thirty-one-year experience with 564 patients at a single institution. Ann. Surg. 2007, 245, 755–762. [Google Scholar] [CrossRef]
- Castellano-Megias, V.M.; Ibarrola-de Andres, C.; Colina-Ruizdelgado, F. Pathological aspects of so called “hilar cholangiocarcinoma”. World J. Gastrointest. Oncol. 2013, 5, 159–170. [Google Scholar] [CrossRef]
- Kambakamba, P.; Linecker, M.; Slankamenac, K.; DeOliveira, M.L. Lymph node dissection in resectable perihilar cholangiocarcinoma: A systematic review. Am. J. Surg. 2015, 210, 694–701. [Google Scholar] [CrossRef]
- Guglielmi, A.; Ruzzenente, A.; Campagnaro, T.; Pachera, S.; Conci, S.; Valdegamberi, A.; Sandri, M.; Iacono, C. Prognostic significance of lymph node ratio after resection of peri-hilar cholangiocarcinoma. HPB 2011, 13, 240–245. [Google Scholar] [CrossRef]
- Olthof, P.B.; Aldrighetti, L.; Alikhanov, R.; Cescon, M.; Groot Koerkamp, B.; Jarnagin, W.R.; Nadalin, S.; Pratschke, J.; Schmelze, M.; Sparrelid, E.; et al. Portal vein embolization is associated with reduced liver failure and mortality in high-risk resections for perihilar cholangiocarcinoma. Ann. Surg. Oncol. 2020, 27, 2311–2318. [Google Scholar] [CrossRef]
- Rassam, F.; Olthof, P.B.; van Lienden, K.P.; Bennink, R.J.; Erdmann, J.I.; Swijnenburg, R.J.; Busch, O.R.; Besselink, M.G.; van Gulik, T.M. Comparison of functional and volumetric increase of the future remnant liver and postoperative outcomes after portal vein embolization and complete or partial associating liver partition and portal vein ligation for staged hepatectomy (alpps). Ann. Transl. Med. 2020, 8, 436. [Google Scholar] [CrossRef] [PubMed]
- Ripamonti, L.; De Carlis, R.; Lauterio, A.; Mangoni, I.; Frassoni, S.; Bagnardi, V.; Centonze, L.; Poli, C.; Buscemi, V.; Ferla, F.; et al. Major hepatectomy for perihilar cholangiocarcinoma in elderly patients: Is it reasonable? Updates Surg. 2021. [Google Scholar] [CrossRef] [PubMed]
- Akashi, K.; Ebata, T.; Mizuno, T.; Yokoyama, Y.; Igami, T.; Yamaguchi, J.; Onoe, S.; Nagino, M. Surgery for perihilar cholangiocarcinoma from a viewpoint of age: Is it beneficial to octogenarians in an aging society? Surgery 2018, 164, 1023–1029. [Google Scholar] [CrossRef] [PubMed]
- Schmucker, D.L.; Sanchez, H. Liver regeneration and aging: A current perspective. Curr. Gerontol. Geriatr. Res. 2011, 2011, 526379. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Kim, M.H.; Kim, K.P.; Park, D.H.; Moon, S.H.; Song, T.J.; Eum, J.; Lee, S.S.; Seo, D.W.; Lee, S.K. Natural history and prognostic factors of advanced cholangiocarcinoma without surgery, chemotherapy, or radiotherapy: A large-scale observational study. Gut Liver 2009, 3, 298–305. [Google Scholar] [CrossRef]
- Gaspersz, M.P.; Buettner, S.; van Vugt, J.L.A.; Roos, E.; Coelen, R.J.S.; Vugts, J.; Belt, E.J.; de Jonge, J.; Polak, W.G.; Willemssen, F.; et al. Conditional survival in patients with unresectable perihilar cholangiocarcinoma. HPB 2017, 19, 966–971. [Google Scholar] [CrossRef][Green Version]
- Wynter-Blyth, V.; Moorthy, K. Prehabilitation: Preparing patients for surgery. BMJ 2017, 358, j3702. [Google Scholar] [CrossRef]
- Moran, J.; Guinan, E.; McCormick, P.; Larkin, J.; Mockler, D.; Hussey, J.; Moriarty, J.; Wilson, F. The ability of prehabilitation to influence postoperative outcome after intra-abdominal operation: A systematic review and meta-analysis. Surgery 2016, 160, 1189–1201. [Google Scholar] [CrossRef]
- Dewulf, M.; Verrips, M.; Coolsen, M.M.E.; Olde Damink, S.W.M.; Den Dulk, M.; Bongers, B.C.; Dejong, K.; Bouwense, S.A.W. The effect of prehabilitation on postoperative complications and postoperative hospital stay in hepatopancreatobiliary surgery a systematic review. HPB 2021, 23, 1299–1310. [Google Scholar] [CrossRef] [PubMed]
- Snowden, C.P.; Prentis, J.; Jacques, B.; Anderson, H.; Manas, D.; Jones, D.; Trenell, M. Cardiorespiratory fitness predicts mortality and hospital length of stay after major elective surgery in older people. Ann. Surg. 2013, 257, 999–1004. [Google Scholar] [CrossRef] [PubMed]
- Groot Koerkamp, B.; Wiggers, J.K.; Allen, P.J.; Besselink, M.G.; Blumgart, L.H.; Busch, O.R.; Coelen, R.J.; D’Angelica, M.I.; DeMatteo, R.P.; Gouma, D.J.; et al. Recurrence rate and pattern of perihilar cholangiocarcinoma after curative intent resection. J. Am. Coll. Surg. 2015, 221, 1041–1049. [Google Scholar] [CrossRef] [PubMed]
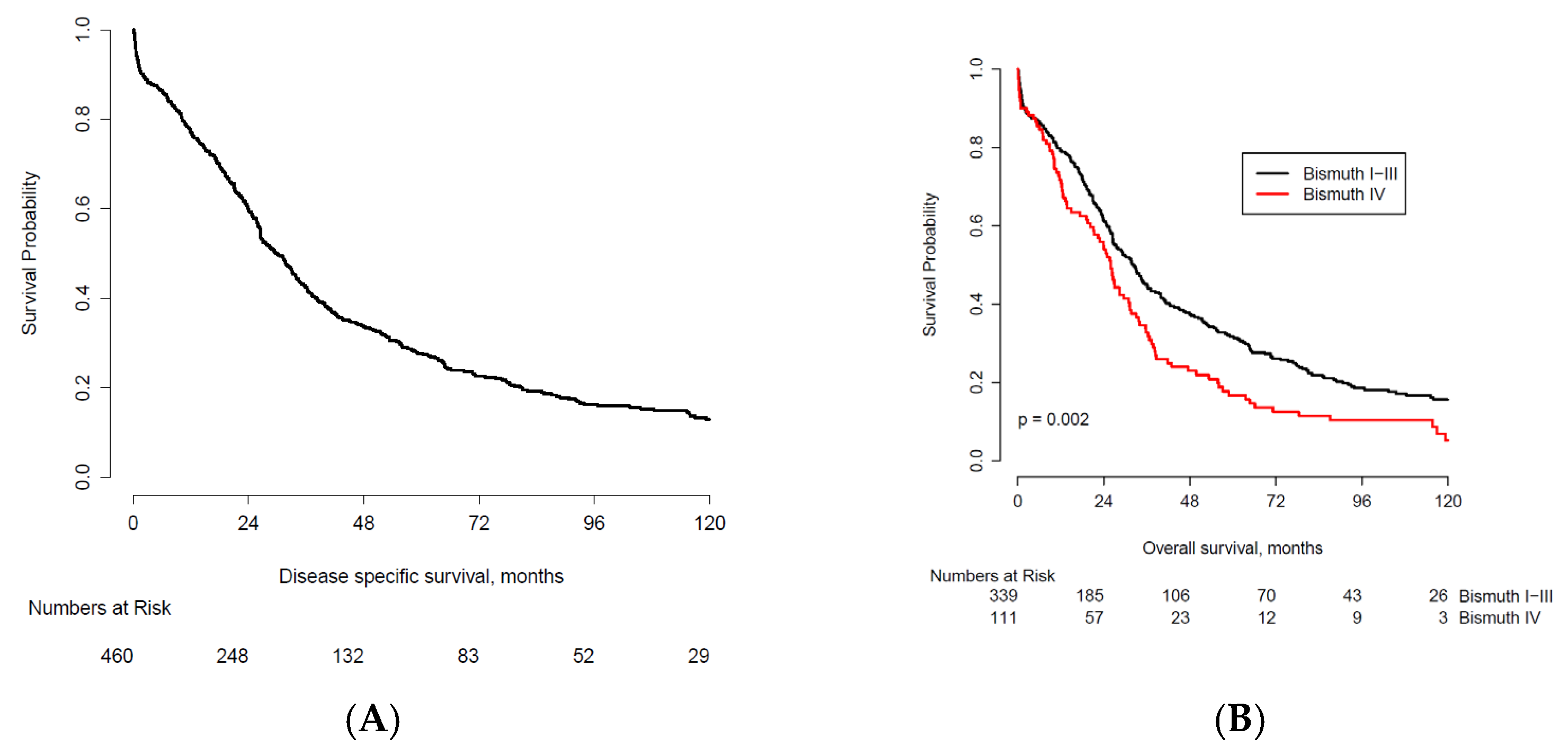
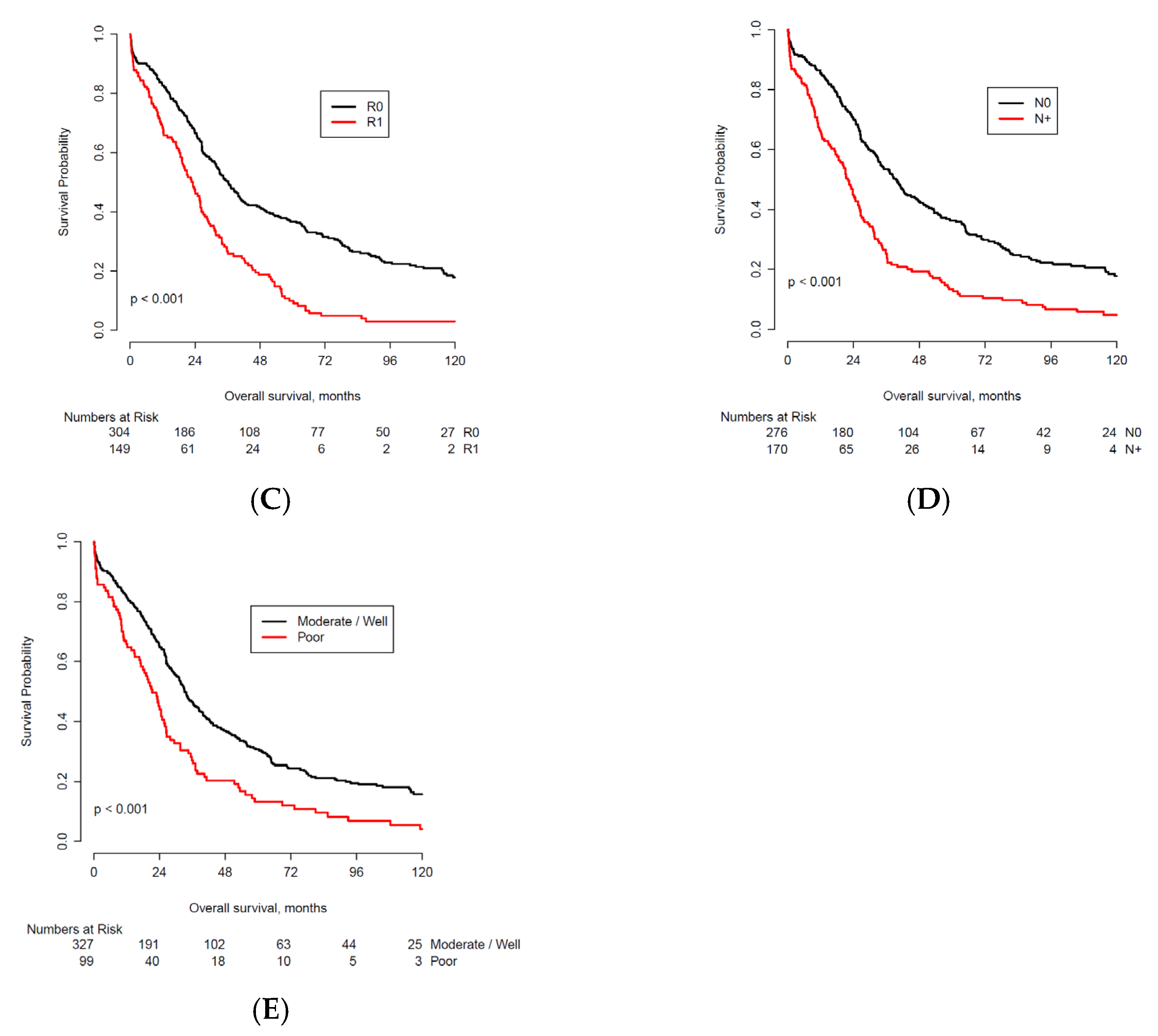
| Characteristic | n | <2 Years * | % | 2-Year OS Probability † | 2–5 Years * | % | 5-Year OS Probability † | 5–10 Years * | % | 10-Year OS Probability † | >10 Years | % | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| n | 460 | 177 | 38.5 | 129 | 28.0 | 50 | 10.9 | 29 | 6.3 | ||||
| Preoperative Factors | |||||||||||||
| Sex | Female | 192 | 70 | 36.5 | 61.7 | 56 | 29.2 | 27.9 | 19 | 9.9 | 14.1 | 14 | 7.3 |
| Male | 268 | 107 | 39.9 | 58.4 | 73 | 27.2 | 27.4 | 31 | 11.6 | 11.9 | 15 | 5.6 | |
| Age ≥ 70 years | No | 327 | 125 | 38.2 | 60.2 | 99 | 30.3 | 25.9 | 33 | 10.1 | 12.7 | 23 | 7.0 |
| Yes | 131 | 52 | 39.7 | 58.4 | 30 | 22.9 | 31.8 | 17 | 13.0 | 13.2 | 6 | 4.6 | |
| Jaundice at presentation | No | 81 | 21 | 25.9 | 72.5 | 21 | 25.9 | 43.1 | 15 | 18.5 | 17.3 | 5 | 6.2 |
| Yes | 318 | 136 | 42.8 | 55.4 | 89 | 28.0 | 23.4 | 27 | 8.5 | 12 | 20 | 6.3 | |
| ASA classification ≥ 3 | No | 251 | 89 | 35.5 | 62.3 | 68 | 27.1 | 30.5 | 28 | 11.2 | 15.1 | 20 | 8.0 |
| Yes | 170 | 75 | 44.1 | 54.5 | 43 | 25.3 | 25.3 | 19 | 11.2 | 10 | 7 | 4.1 | |
| Preoperative biliary drainage | No | 79 | 26 | 32.9 | 64.7 | 19 | 24.1 | 38.5 | 17 | 21.5 | 7.6 | 3 | 3.8 |
| Yes | 381 | 151 | 39.6 | 58.5 | 110 | 28.9 | 25.4 | 33 | 8.7 | 13.8 | 26 | 6.8 | |
| Preoperative cholangitis | No | 344 | 133 | 38.7 | 59.4 | 92 | 26.7 | 27.9 | 38 | 11.0 | 12.4 | 20 | 5.8 |
| Yes | 77 | 31 | 40.3 | 57.9 | 23 | 29.9 | 24.9 | 6 | 7.8 | 14.9 | 7 | 9.1 | |
| Bismuth- Corlette IV | No | 339 | 124 | 36.6 | 61.4 | 86 | 25.4 | 31.7 | 40 | 11.8 | 15.7 | 26 | 7.7 |
| Yes | 111 | 49 | 44.1 | 54.9 | 39 | 35.1 | 16.7 | 9 | 8.1 | 5.2 | 3 | 2.7 | |
| PVE performed | No | 379 | 138 | 36.4 | 62.1 | 111 | 29.3 | 28.8 | 47 | 12.4 | 12.4 | 24 | 6.3 |
| Yes | 80 | 39 | 48.8 | 48.4 | 18 | 22.5 | 21.8 | 3 | 3.8 | 15.3 | 5 | 6.3 | |
| Operative Factors | |||||||||||||
| Major resection | No | 54 | 21 | 38.9 | 59.7 | 17 | 31.5 | 26 | 5 | 9.3 | 14.9 | 5 | 9.3 |
| Yes | 406 | 156 | 38.4 | 59.6 | 112 | 27.6 | 27.9 | 45 | 11.1 | 12.5 | 24 | 5.9 | |
| PV reconstruction | No | 322 | 115 | 35.7 | 62.5 | 89 | 27.6 | 31.3 | 41 | 12.7 | 13.7 | 22 | 6.8 |
| Yes | 138 | 62 | 44.9 | 52.4 | 40 | 29.0 | 19.2 | 9 | 6.5 | 10.7 | 7 | 5.1 | |
| HA reconstruction | No | 394 | 147 | 37.3 | 60.5 | 113 | 28.7 | 27.1 | 44 | 11.2 | 11.1 | 19 | 4.8 |
| Yes | 10 | 6 | 60.0 | 40 | 3 | 30.0 | 10 | 1 | 10.0 | 0 | 0 | 0.0 | |
| Pancreatoduodenectomy | No | 375 | 138 | 36.8 | 60.9 | 109 | 29.1 | 26.8 | 40 | 10.7 | 11.3 | 18 | 4.8 |
| Yes | 5 | 4 | 80.0 | 20 | 1 | 20.0 | 0 | 0 | 0.0 | 0 | 0 | 0.0 | |
| Postoperative Factors | |||||||||||||
| Positive resection margins | No | 304 | 98 | 32.2 | 66.5 | 80 | 26.3 | 36.7 | 42 | 13.8 | 17.9 | 27 | 8.9 |
| Yes | 149 | 73 | 49.0 | 47.6 | 48 | 32.2 | 10.1 | 8 | 5.4 | 3 | 2 | 1.3 | |
| AJCC T3 or T4 | No | 229 | 66 | 28.8 | 70.5 | 69 | 30.1 | 37.1 | 32 | 14.0 | 17.6 | 20 | 8.7 |
| Yes | 214 | 101 | 47.2 | 48.8 | 56 | 26.2 | 18.1 | 17 | 7.9 | 7.9 | 8 | 3.7 | |
| Positive lymph nodes | No | 276 | 80 | 29.0 | 69.8 | 84 | 30.4 | 36.3 | 38 | 13.8 | 17.8 | 24 | 8.7 |
| Yes | 170 | 87 | 51.2 | 44.8 | 44 | 25.9 | 13.4 | 11 | 6.5 | 4.8 | 4 | 2.4 | |
| Poor tumor differentiation | No | 327 | 109 | 33.3 | 64.7 | 97 | 29.7 | 30.7 | 36 | 11.0 | 15.8 | 25 | 7.6 |
| Yes | 99 | 52 | 52.5 | 45.1 | 28 | 28.3 | 13.2 | 7 | 7.1 | 4.1 | 3 | 3.0 | |
| Perineural invasion | No | 96 | 22 | 22.9 | 75.9 | 22 | 22.9 | 42.6 | 11 | 11.5 | 21.5 | 9 | 9.4 |
| Yes | 241 | 115 | 47.7 | 50.1 | 71 | 29.5 | 19.2 | 17 | 7.1 | 10.9 | 17 | 7.1 | |
| Characteristics | Full Cohort | Observed Cure * | % | Probability of Cure † | |
|---|---|---|---|---|---|
| n | 460 | 25 | 5.4 | ||
| Preoperative Factors | |||||
| Sex | Female | 192 | 10 | 5.2 | 5.2 |
| Male | 268 | 15 | 5.6 | 11.6 | |
| Age ≥ 70 years | No | 327 | 21 | 6.4 | 10.7 |
| Yes | 131 | 4 | 3.1 | 0.0 | |
| Jaundice at presentation | No | 81 | 5 | 6.2 | 13.8 |
| Yes | 318 | 17 | 5.3 | 8.1 | |
| ASA classification ≥ 3 | No | 251 | 19 | 7.6 | 10.6 |
| Yes | 170 | 6 | 3.5 | 7.3 | |
| Preoperative biliary drainage | No | 79 | 3 | 3.8 | 8.6 |
| Yes | 381 | 22 | 5.8 | 9.1 | |
| Preoperative cholangitis | No | 344 | 19 | 5.5 | 7.7 |
| Yes | 77 | 6 | 7.8 | 13.7 | |
| Bismuth-Corlette IV | No | 339 | 22 | 6.5 | 12.9 |
| Yes | 111 | 3 | 2.7 | 0.0 | |
| PVE performed | No | 379 | 21 | 5.5 | 9.0 |
| Yes | 80 | 4 | 5.0 | 10.9 | |
| Operative Factors | |||||
| Major resection | No | 54 | 5 | 9.3 | 8.2 |
| Yes | 406 | 20 | 4.9 | 8.8 | |
| PV reconstruction | No | 322 | 18 | 5.6 | 11.9 |
| Yes | 138 | 7 | 5.1 | 4.6 | |
| HA reconstruction | No | 394 | 15 | 3.8 | 9.3 |
| Yes | 10 | 0 | 0.0 | 0.0 | |
| Pancreatoduodenectomy | No | 375 | 14 | 3.7 | - |
| Yes | 5 | 0 | 0.0 | - | |
| Postoperative Factors | |||||
| Positive resection margin | No | 304 | 23 | 7.6 | 14.6 |
| Yes | 149 | 2 | 1.3 | 0.9 | |
| AJCC T3 or T4 | No | 229 | 18 | 7.9 | 11.6 |
| Yes | 214 | 6 | 2.8 | 7.0 | |
| Positive lymph nodes | No | 276 | 21 | 7.6 | 13.9 |
| Yes | 170 | 3 | 1.8 | 2.5 | |
| Poor tumor differentiation | No | 327 | 21 | 6.4 | 11.7 |
| Yes | 99 | 3 | 3.0 | 3.0 | |
| Perineural invasion | No | 96 | 8 | 8.3 | 12.7 |
| Yes | 241 | 14 | 5.8 | 8.1 | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
van Keulen, A.-M.; Olthof, P.B.; Cescon, M.; Guglielmi, A.; Jarnagin, W.R.; Nadalin, S.; Pratschke, J.; Ratti, F.; Troisi, R.I.; Groot Koerkamp, B.; et al. Actual 10-Year Survival after Resection of Perihilar Cholangiocarcinoma: What Factors Preclude a Chance for Cure? Cancers 2021, 13, 6260. https://doi.org/10.3390/cancers13246260
van Keulen A-M, Olthof PB, Cescon M, Guglielmi A, Jarnagin WR, Nadalin S, Pratschke J, Ratti F, Troisi RI, Groot Koerkamp B, et al. Actual 10-Year Survival after Resection of Perihilar Cholangiocarcinoma: What Factors Preclude a Chance for Cure? Cancers. 2021; 13(24):6260. https://doi.org/10.3390/cancers13246260
Chicago/Turabian Stylevan Keulen, Anne-Marleen, Pim B. Olthof, Matteo Cescon, Alfredo Guglielmi, William R. Jarnagin, Silvio Nadalin, Johann Pratschke, Francesca Ratti, Roberto I. Troisi, Bas Groot Koerkamp, and et al. 2021. "Actual 10-Year Survival after Resection of Perihilar Cholangiocarcinoma: What Factors Preclude a Chance for Cure?" Cancers 13, no. 24: 6260. https://doi.org/10.3390/cancers13246260
APA Stylevan Keulen, A.-M., Olthof, P. B., Cescon, M., Guglielmi, A., Jarnagin, W. R., Nadalin, S., Pratschke, J., Ratti, F., Troisi, R. I., Groot Koerkamp, B., Buettner, S., Erdmann, J. I., & on behalf of the ‘Perihilar Cholangiocarcinoma Collaboration Group’. (2021). Actual 10-Year Survival after Resection of Perihilar Cholangiocarcinoma: What Factors Preclude a Chance for Cure? Cancers, 13(24), 6260. https://doi.org/10.3390/cancers13246260








