The BRAFV600E Mutation Is Not a Risk Factor for More Aggressive Tumor Behavior in Radiogenic and Sporadic Papillary Thyroid Carcinoma at a Young Age
Abstract
:Simple Summary
Abstract
1. Introduction
2. Results
2.1. Frequency of BRAFV600E in Radiogenic and Sporadic PTC
2.2. BRAFV600E Associations in Radiogenic PTC
2.3. BRAFV600E Associations in Sporadic PTC
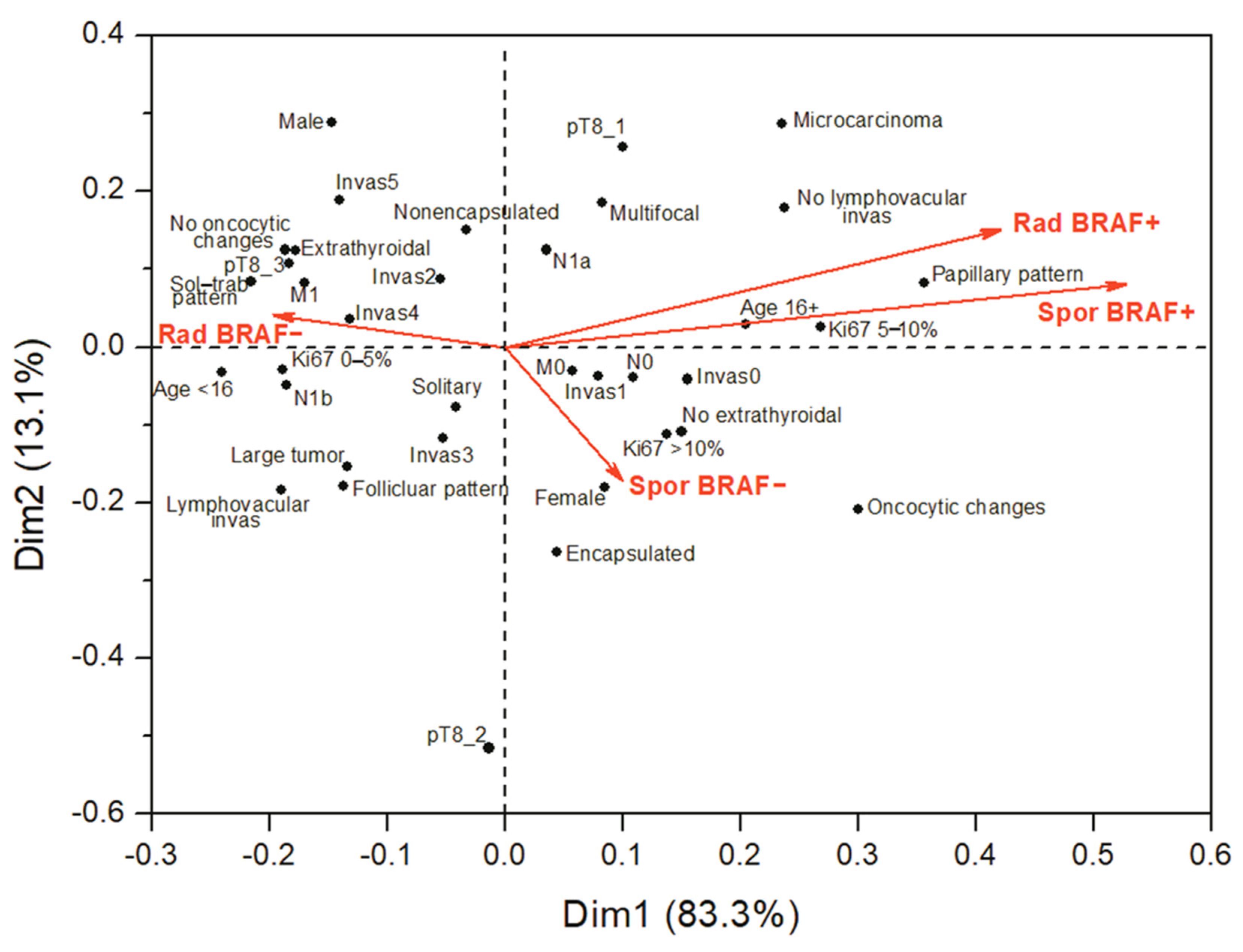
2.4. Comparison of the BRAFV600E-Positive or the BRAFV600E-Negative PTCs across the Radiogenic and Sporadic Series
3. Discussion
4. Materials and Methods
4.1. Patients
4.2. Histopathology
4.3. Immunohistochemistry
4.4. Thyroid Dosimetry
4.5. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tronko, M.D.; Bogdanova, T.I.; Komissarenko, I.V.; Epstein, O.V.; Oliynyk, V.; Kovalenko, A.; Likhtarev, I.A.; Kairo, I.; Peters, S.B.; LiVolsi, V.A. Thyroid carcinoma in children and adolescents in Ukraine after the Chernobyl nuclear accident: Statistical data and clinicomorphologic characteristics. Cancer 1999, 86, 149–156. [Google Scholar] [CrossRef]
- Williams, E.D.; Abrosimov, A.; Bogdanova, T.; Demidchik, E.P.; Ito, M.; LiVolsi, V.; Lushnikov, E.; Rosai, J.; Sidorov, Y.; Tronko, M.D.; et al. Thyroid carcinoma after Chernobyl latent period, morphology and aggressiveness. Br. J. Cancer 2004, 90, 2219–2224. [Google Scholar] [CrossRef] [Green Version]
- Bogdanova, T.; Zurnadzhy, L.; LiVolsi, V.A.; Williams, E.D.; Ito, M.; Nakashima, M.; Thomas, G.A. Thyroid cancer pathology in Ukraine after Chernobyl. In Thyroid Cancer in Ukraine after Chernobyl: Dosimetry, Epidemiology, Pathology, Molecular Biology; Tronko, M., Bogdanova, T., Saenko, V., Thomas, G.A., Likhtarov, I., Yamashita, S., Eds.; IN-TEX: Nagasaki, Japan, 2014; pp. 65–108. [Google Scholar]
- Bogdanova, T.I.; Saenko, V.A.; Brenner, A.V.; Zurnadzhy, L.Y.; Rogounovitch, T.I.; Likhtarov, I.A.; Masiuk, S.V.; Kovgan, L.M.; Shpak, V.M.; Thomas, G.A.; et al. Comparative Histopathologic Analysis of “Radiogenic” and “Sporadic” Papillary Thyroid Carcinoma: Patients Born Before and After the Chernobyl Accident. Thyroid 2018, 28, 880–890. [Google Scholar] [CrossRef]
- Nikiforov, Y.E.; Rowland, J.M.; Bove, K.E.; Monforte-Munoz, H.; Fagin, J.A. Distinct pattern of ret oncogene rearrangements in morphological variants of radiation-induced and sporadic thyroid papillary carcinomas in children. Cancer Res. 1997, 57, 1690–1694. [Google Scholar]
- Thomas, G.A.; Bunnell, H.; Cook, H.A.; Williams, E.D.; Nerovnya, A.; Cherstvoy, E.D.; Tronko, N.D.; Bogdanova, T.I.; Chiappetta, G.; Viglietto, G.; et al. High prevalence of RET/PTC rearrangements in Ukrainian and Belarussian post-Chernobyl thyroid papillary carcinomas: A strong correlation between RET/PTC3 and the solid-follicular variant. J. Clin. Endocrinol. Metab. 1999, 84, 4232–4238. [Google Scholar] [CrossRef] [PubMed]
- Santoro, M.; Thomas, G.A.; Vecchio, G.; Williams, G.H.; Fusco, A.; Chiappetta, G.; Pozcharskaya, V.; Bogdanova, T.I.; Demidchik, E.P.; Cherstvoy, E.D.; et al. Gene rearrangement and Chernobyl related thyroid cancers. Br. J. Cancer 2000, 82, 315–322. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rabes, H.M.; Demidchik, E.P.; Sidorow, J.D.; Lengfelder, E.; Beimfohr, C.; Hoelzel, D.; Klugbauer, S. Pattern of radiation-induced RET and NTRK1 rearrangements in 191 post-chernobyl papillary thyroid carcinomas: Biological, phenotypic, and clinical implications. Clin. Cancer Res. 2000, 6, 1093–1103. [Google Scholar] [PubMed]
- Leeman-Neill, R.J.; Brenner, A.V.; Little, M.P.; Bogdanova, T.I.; Hatch, M.; Zurnadzy, L.Y.; Mabuchi, K.; Tronko, M.D.; Nikiforov, Y.E. RET/PTC and PAX8/PPARgamma chromosomal rearrangements in post-Chernobyl thyroid cancer and their association with iodine-131 radiation dose and other characteristics. Cancer 2013, 119, 1792–1799. [Google Scholar] [CrossRef] [Green Version]
- Ricarte-Filho, J.C.; Li, S.; Garcia-Rendueles, M.E.; Montero-Conde, C.; Voza, F.; Knauf, J.A.; Heguy, A.; Viale, A.; Bogdanova, T.; Thomas, G.A.; et al. Identification of kinase fusion oncogenes in post-Chernobyl radiation-induced thyroid cancers. J. Clin. Investig. 2013, 123, 4935–4944. [Google Scholar] [CrossRef] [Green Version]
- Efanov, A.A.; Brenner, A.V.; Bogdanova, T.I.; Kelly, L.M.; Liu, P.; Little, M.P.; Wald, A.I.; Hatch, M.; Zurnadzy, L.Y.; Nikiforova, M.N.; et al. Investigation of the Relationship Between Radiation Dose and Gene Mutations and Fusions in Post-Chernobyl Thyroid Cancer. JNCI J. Natl. Cancer. Inst. 2017, 110, 371–378. [Google Scholar] [CrossRef]
- Nikiforov, Y.E.; Nikiforova, M.N.; Gnepp, D.R.; Fagin, J.A. Prevalence of mutations of ras and p53 in benign and malignant thyroid tumors from children exposed to radiation after the Chernobyl nuclear accident. Oncogene 1996, 13, 687–693. [Google Scholar] [PubMed]
- Kumagai, A.; Namba, H.; Saenko, V.A.; Ashizawa, K.; Ohtsuru, A.; Ito, M.; Ishikawa, N.; Sugino, K.; Ito, K.; Jeremiah, S.; et al. Low frequency of BRAFT1796A mutations in childhood thyroid carcinomas. J. Clin. Endocrinol. Metab. 2004, 89, 4280–4284. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nikiforova, M.N.; Ciampi, R.; Salvatore, G.; Santoro, M.; Gandhi, M.; Knauf, J.A.; Thomas, G.A.; Jeremiah, S.; Bogdanova, T.I.; Tronko, M.D.; et al. Low prevalence of BRAF mutations in radiation-induced thyroid tumors in contrast to sporadic papillary carcinomas. Cancer Lett. 2004, 209, 1–6. [Google Scholar] [CrossRef]
- Powell, N.; Jeremiah, S.; Morishita, M.; Dudley, E.; Bethel, J.; Bogdanova, T.; Tronko, M.; Thomas, G. Frequency of BRAF T1796A mutation in papillary thyroid carcinoma relates to age of patient at diagnosis and not to radiation exposure. J. Pathol. 2005, 205, 558–564. [Google Scholar] [CrossRef]
- Morton, L.M.; Karyadi, D.M.; Stewart, C.; Bogdanova, T.I.; Dawson, E.T.; Steinberg, M.K.; Dai, J.; Hartley, S.W.; Schonfeld, S.J.; Sampson, J.N.; et al. Radiation-related genomic profile of papillary thyroid carcinoma after the Chernobyl accident. Science 2021, 372, 725–729. [Google Scholar] [CrossRef] [PubMed]
- Rogounovitch, T.I.; Mankovskaya, S.V.; Fridman, M.V.; Leonova, T.A.; Kondratovitch, V.A.; Konoplya, N.E.; Yamashita, S.; Mitsutake, N.; Saenko, V.A. Major Oncogenic Drivers and Their Clinicopathological Correlations in Sporadic Childhood Papillary Thyroid Carcinoma in Belarus. Cancers 2021, 13, 3374. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, H.; Yoshida, A.; Nakamura, Y.; Hayashi, H.; Miyagi, Y.; Wada, N.; Rino, Y.; Masuda, M.; Imada, T. Clinical significance of BRAF (V600E) mutation and Ki-67 labeling index in papillary thyroid carcinomas. Anticancer Res. 2007, 27, 3645–3649. [Google Scholar]
- Nikiforova, M.N.; Kimura, E.T.; Gandhi, M.; Biddinger, P.W.; Knauf, J.A.; Basolo, F.; Zhu, Z.; Giannini, R.; Salvatore, G.; Fusco, A.; et al. BRAF mutations in thyroid tumors are restricted to papillary carcinomas and anaplastic or poorly differentiated carcinomas arising from papillary carcinomas. J. Clin. Endocrinol. Metab. 2003, 88, 5399–5404. [Google Scholar] [CrossRef]
- Rosenbaum, E.; Hosler, G.; Zahurak, M.; Cohen, Y.; Sidransky, D.; Westra, W.H. Mutational activation of BRAF is not a major event in sporadic childhood papillary thyroid carcinoma. Mod. Pathol. 2005, 18, 898–902. [Google Scholar] [CrossRef]
- Trovisco, V.; Soares, P.; Preto, A.; de Castro, I.V.; Lima, J.; Castro, P.; Maximo, V.; Botelho, T.; Moreira, S.; Meireles, A.M.; et al. Type and prevalence of BRAF mutations are closely associated with papillary thyroid carcinoma histotype and patients’ age but not with tumour aggressiveness. Virchows Arch. 2005, 446, 589–595. [Google Scholar] [CrossRef]
- Fugazzola, L.; Puxeddu, E.; Avenia, N.; Romei, C.; Cirello, V.; Cavaliere, A.; Faviana, P.; Mannavola, D.; Moretti, S.; Rossi, S.; et al. Correlation between B-RAFV600E mutation and clinico-pathologic parameters in papillary thyroid carcinoma: Data from a multicentric Italian study and review of the literature. Endocr. Relat. Cancer 2006, 13, 455–464. [Google Scholar] [CrossRef]
- Kebebew, E.; Weng, J.; Bauer, J.; Ranvier, G.; Clark, O.H.; Duh, Q.Y.; Shibru, D.; Bastian, B.; Griffin, A. The prevalence and prognostic value of BRAF mutation in thyroid cancer. Ann. Surg. 2007, 246, 466–471. [Google Scholar] [CrossRef]
- Mitsutake, N.; Fukushima, T.; Matsuse, M.; Rogounovitch, T.; Saenko, V.; Uchino, S.; Ito, M.; Suzuki, K.; Suzuki, S.; Yamashita, S. BRAF(V600E) mutation is highly prevalent in thyroid carcinomas in the young population in Fukushima: A different oncogenic profile from Chernobyl. Sci. Rep. 2015, 5, 16976. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nikita, M.E.; Jiang, W.; Cheng, S.M.; Hantash, F.M.; McPhaul, M.J.; Newbury, R.O.; Phillips, S.A.; Reitz, R.E.; Waldman, F.M.; Newfield, R.S. Mutational Analysis in Pediatric Thyroid Cancer and Correlations with Age, Ethnicity, and Clinical Presentation. Thyroid 2016, 26, 227–234. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oishi, N.; Kondo, T.; Nakazawa, T.; Mochizuki, K.; Inoue, T.; Kasai, K.; Tahara, I.; Yabuta, T.; Hirokawa, M.; Miyauchi, A.; et al. Frequent BRAF (V600E) and Absence of TERT Promoter Mutations Characterize Sporadic Pediatric Papillary Thyroid Carcinomas in Japan. Endocr. Pathol. 2017, 28, 103–111. [Google Scholar] [CrossRef]
- Yan, C.; Huang, M.; Li, X.; Wang, T.; Ling, R. Relationship between BRAF V600E and clinical features in papillary thyroid carcinoma. Endocr. Connect. 2019, 8, 988–996. [Google Scholar] [CrossRef] [Green Version]
- Iwadate, M.; Mitsutake, N.; Matsuse, M.; Fukushima, T.; Suzuki, S.; Matsumoto, Y.; Ookouchi, C.; Mizunuma, H.; Nakamura, I.; Nakano, K.; et al. The Clinicopathological Results of Thyroid Cancer with BRAFV600E Mutation in the Young Population of Fukushima. J. Clin. Endocrinol. Metab. 2020, 105, e4328–e4336. [Google Scholar] [CrossRef]
- Wang, Z.; Chen, J.Q.; Liu, J.L.; Qin, X.G. Clinical impact of BRAF mutation on the diagnosis and prognosis of papillary thyroid carcinoma: A systematic review and meta-analysis. Eur. J. Clin. Investig. 2016, 46, 146–157. [Google Scholar] [CrossRef] [PubMed]
- Ito, Y.; Miyauchi, A.; Kakudo, K.; Hirokawa, M.; Kobayashi, K.; Miya, A. Prognostic significance of ki-67 labeling index in papillary thyroid carcinoma. World J. Surg. 2010, 34, 3015–3021. [Google Scholar] [CrossRef]
- Lee, J.H.; Lee, E.S.; Kim, Y.S. Clinicopathologic significance of BRAF V600E mutation in papillary carcinomas of the thyroid: A meta-analysis. Cancer 2007, 110, 38–46. [Google Scholar] [CrossRef]
- Lupi, C.; Giannini, R.; Ugolini, C.; Proietti, A.; Berti, P.; Minuto, M.; Materazzi, G.; Elisei, R.; Santoro, M.; Miccoli, P.; et al. Association of BRAF V600E mutation with poor clinicopathological outcomes in 500 consecutive cases of papillary thyroid carcinoma. J. Clin. Endocrinol. Metab. 2007, 92, 4085–4090. [Google Scholar] [CrossRef] [Green Version]
- Kim, S.J.; Lee, K.E.; Myong, J.P.; Park, J.H.; Jeon, Y.K.; Min, H.S.; Park, S.Y.; Jung, K.C.; Koo, D.H.; Youn, Y.K. BRAF V600E mutation is associated with tumor aggressiveness in papillary thyroid cancer. World J. Surg. 2012, 36, 310–317. [Google Scholar] [CrossRef]
- Li, C.; Lee, K.C.; Schneider, E.B.; Zeiger, M.A. BRAF V600E mutation and its association with clinicopathological features of papillary thyroid cancer: A meta-analysis. J. Clin. Endocrinol. Metab. 2012, 97, 4559–4570. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, Y.J.; Deng, X.L.; Li, H.Q. BRAF(V600E) mutation and its association with clinicopathological features of papillary thyroid microcarcinoma: A meta-analysis. J. Huazhong Univ. Sci. Technolog. Med. Sci. 2015, 35, 591–599. [Google Scholar] [CrossRef] [PubMed]
- Tufano, R.P.; Teixeira, G.V.; Bishop, J.; Carson, K.A.; Xing, M. BRAF mutation in papillary thyroid cancer and its value in tailoring initial treatment: A systematic review and meta-analysis. Medicine 2012, 91, 274–286. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Sadow, P.M.; Suh, H.; Lee, K.E.; Choi, J.Y.; Suh, Y.J.; Wang, T.S.; Lubitz, C.C. BRAF(V600E) Is Correlated with Recurrence of Papillary Thyroid Microcarcinoma: A Systematic Review, Multi-Institutional Primary Data Analysis, and Meta-Analysis. Thyroid 2016, 26, 248–255. [Google Scholar] [CrossRef]
- Liu, C.; Chen, T.; Liu, Z. Associations between BRAF(V600E) and prognostic factors and poor outcomes in papillary thyroid carcinoma: A meta-analysis. World J. Surg. Oncol. 2016, 14, 241. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Q.; Liu, S.Z.; Zhang, Q.; Guan, Y.X.; Chen, Q.J.; Zhu, Q.Y. Meta-Analyses of Association Between BRAF(V600E) Mutation and Clinicopathological Features of Papillary Thyroid Carcinoma. Cell Physiol. Biochem. 2016, 38, 763–776. [Google Scholar] [CrossRef] [PubMed]
- Vuong, H.G.; Duong, U.N.; Altibi, A.M.; Ngo, H.T.; Pham, T.Q.; Tran, H.M.; Gandolfi, G.; Hassell, L. A meta-analysis of prognostic roles of molecular markers in papillary thyroid carcinoma. Endocr. Connect. 2017, 6, R8–R17. [Google Scholar] [CrossRef] [Green Version]
- Siironen, P.; Nordling, S.; Louhimo, J.; Haapiainen, R.; Haglund, C. Immunohistochemical expression of Bcl-2, Ki-67, and p21 in patients with papillary thyroid cancer. Tumour Biol. 2005, 26, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.; Wu, W.D.; Chen, G.M.; Chou, S.L.; Dai, X.M.; Xu, J.M.; Peng, Z.H. Could Tumor Size Be a Predictor for Papillary Thyroid Microcarcinoma: A Retrospective Cohort Study. Asian Pac. J. Cancer Prev. 2015, 16, 8625–8628. [Google Scholar] [CrossRef]
- Zhou, Y.; Jiang, H.G.; Lu, N.; Lu, B.H.; Chen, Z.H. Expression of ki67 in papillary thyroid microcarcinoma and its clinical significance. Asian Pac. J. Cancer Prev. 2015, 16, 1605–1608. [Google Scholar] [CrossRef] [Green Version]
- Avdalyan, A.M.; Ivanov, A.A.; Lushnikova, E.L.; Molodykh, O.P.; Vikhlyanov, I.V. The Relationship of Immunoexpression of Ki-67 and Hsp70 with Clinical and Morphological Parameters and Prognosis of Papillary Thyroid Cancer. Bull. Exp. Biol. Med. 2020, 168, 688–693. [Google Scholar] [CrossRef] [PubMed]
- Miyauchi, A.; Kudo, T.; Hirokawa, M.; Ito, Y.; Kihara, M.; Higashiyama, T.; Yabuta, T.; Masuoka, H.; Shindo, H.; Kobayashi, K.; et al. Ki-67 labeling index is a predictor of postoperative persistent disease and cancer growth and a prognostic indicator in papillary thyroid carcinoma. Eur. Thyroid J. 2013, 2, 57–64. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hirokawa, M.; Kudo, T.; Ota, H.; Suzuki, A.; Miyauchi, A. Pathological characteristics of low-risk papillary thyroid microcarcinoma with progression during active surveillance. Endocr. J. 2016, 63, 805–810. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tanaka, A.; Matsuse, M.; Saenko, V.; Nakao, T.; Yamanouchi, K.; Sakimura, C.; Yano, H.; Nishihara, E.; Hirokawa, M.; Suzuki, K.; et al. TERT mRNA Expression as a Novel Prognostic Marker in Papillary Thyroid Carcinomas. Thyroid 2019, 29, 1105–1114. [Google Scholar] [CrossRef]
- Ma, B.; Wang, Y.; Yang, S.; Ji, Q. Predictive factors for central lymph node metastasis in patients with cN0 papillary thyroid carcinoma: A systematic review and meta-analysis. Int. J. Surg. 2016, 28, 153–161. [Google Scholar] [CrossRef] [PubMed]
- Sassolas, G.; Hafdi-Nejjari, Z.; Ferraro, A.; Decaussin-Petrucci, M.; Rousset, B.; Borson-Chazot, F.; Borbone, E.; Berger, N.; Fusco, A. Oncogenic alterations in papillary thyroid cancers of young patients. Thyroid 2012, 22, 17–26. [Google Scholar] [CrossRef]
- Givens, D.J.; Buchmann, L.O.; Agarwal, A.M.; Grimmer, J.F.; Hunt, J.P. BRAF V600E does not predict aggressive features of pediatric papillary thyroid carcinoma. Laryngoscope 2014, 124, E389–E393. [Google Scholar] [CrossRef]
- Alzahrani, A.S.; Murugan, A.K.; Qasem, E.; Alswailem, M.; Al-Hindi, H.; Shi, Y. Single Point Mutations in Pediatric Differentiated Thyroid Cancer. Thyroid 2017, 27, 189–196. [Google Scholar] [CrossRef]
- Geng, J.; Wang, H.; Liu, Y.; Tai, J.; Jin, Y.; Zhang, J.; He, L.; Fu, L.; Qin, H.; Song, Y.; et al. Correlation between BRAF (V600E) mutation and clinicopathological features in pediatric papillary thyroid carcinoma. Sci. China Life Sci. 2017, 60, 729–738. [Google Scholar] [CrossRef]
- Mostoufi-Moab, S.; Labourier, E.; Sullivan, L.; LiVolsi, V.; Li, Y.; Xiao, R.; Beaudenon-Huibregtse, S.; Kazahaya, K.; Adzick, N.S.; Baloch, Z.; et al. Molecular Testing for Oncogenic Gene Alterations in Pediatric Thyroid Lesions. Thyroid 2018, 28, 60–67. [Google Scholar] [CrossRef]
- Midorikawa, S.; Ohtsuru, A.; Murakami, M.; Takahashi, H.; Suzuki, S.; Matsuzuka, T.; Shimura, H.; Ohira, T.; Suzuki, S.I.; Yasumura, S.; et al. Comparative Analysis of the Growth Pattern of Thyroid Cancer in Young Patients Screened by Ultrasonography in Japan After a Nuclear Accident: The Fukushima Health Management Survey. JAMA Otolaryngol. Head Neck Surg. 2018, 144, 57–63. [Google Scholar] [CrossRef]
- Bogdanova, T.I.; Zurnadzhy, L.Y.; Nikiforov, Y.E.; Leeman-Neill, R.J.; Tronko, M.D.; Chanock, S.; Mabuchi, K.; Likhtarov, I.A.; Kovgan, L.M.; Drozdovitch, V.; et al. Histopathological features of papillary thyroid carcinomas detected during four screening examinations of a Ukrainian-American cohort. Br. J. Cancer 2015, 113, 1556–1564. [Google Scholar] [CrossRef] [Green Version]
- Smida, J.; Salassidis, K.; Hieber, L.; Zitzelsberger, H.; Kellerer, A.M.; Demidchik, E.P.; Negele, T.; Spelsberg, F.; Lengfelder, E.; Werner, M.; et al. Distinct frequency of ret rearrangements in papillary thyroid carcinomas of children and adults from Belarus. Int. J. Cancer 1999, 80, 32–38. [Google Scholar] [CrossRef]
- Wang, Y.L.; Zhang, R.M.; Luo, Z.W.; Wu, Y.; Du, X.; Wang, Z.Y.; Zhu, Y.X.; Li, D.S.; Ji, Q.H. High frequency of level II-V lymph node involvement in RET/PTC positive papillary thyroid carcinoma. Eur. J. Surg. Oncol. 2008, 34, 77–81. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, S.; Saenko, V. Mechanisms of Disease: Molecular genetics of childhood thyroid cancers. Nat. Clin. Pract. Endocrinol. Metab. 2007, 3, 422–429. [Google Scholar] [CrossRef] [PubMed]
- Tronko, M.; Shpak, V.; Bogdanova, T.; Saenko, V.; Yamashita, S. Epidemiology of thyroid cancer in Ukraine after Chernobyl. In Thyroid Cancer in Ukraine after Chernobyl: Dosimetry, Epidemiology, Pathology, Molecular Biology; Tronko, M., Bogdanova, T., Saenko, V., Thomas, G.A., Likhtarov, I., Yamashita, S., Eds.; IN-TEX: Nagasaki, Japan, 2014; pp. 39–64. [Google Scholar]
- Bogdanova, T.I.; Saenko, V.A.; Hashimoto, Y.; Hirokawa, M.; Zurnadzhy, L.Y.; Hayashi, T.; Ito, M.; Iwadate, M.; Mitsutake, N.; Rogounovitch, T.I.; et al. Papillary Thyroid Carcinoma in Ukraine After Chernobyl and in Japan After Fukushima: Different Histopathological Scenarios. Thyroid 2021, 31, 1322–1334. [Google Scholar] [CrossRef]
- Likhtarov, I.; Thomas, G.; Kovgan, L.; Masiuk, S.; Chepurny, M.; Ivanova, O.; Gerasymenko, V.; Tronko, M.; Bogdanova, T.; Bouville, A. Reconstruction of individual thyroid doses to the Ukrainian subjects enrolled in the Chernobyl Tissue Bank. Radiat. Prot. Dosim. 2013, 156, 407–423. [Google Scholar] [CrossRef] [PubMed]
- Lloyd, R.V.; Osamura, R.Y.; Kloppel, G.; Rosai, J. WHO Classification of Tumours of Endocrine Organs, 4th ed.; IARC Press: Lyon, France, 2017. [Google Scholar]
- Thomas, G.A.; Williams, E.D.; Becker, D.V.; Bogdanova, T.I.; Demidchik, E.P.; Lushnikov, E.; Nagataki, S.; Ostapenko, V.; Pinchera, A.; Souchkevitch, G.; et al. Chernobyl tumor bank. Thyroid 2000, 10, 1126–1127. [Google Scholar] [CrossRef]
- Thomas, G.A. The Chernobyl Tissue Bank: Integrating research on radiation-induced thyroid cancer. J. Radiol. Prot. 2012, 32, N77–N80. [Google Scholar] [CrossRef] [PubMed]
- Brierley, J.D.; Gospodarowich, M.K.; Wittekind, C. TNM Classification of Malignant Tumours, 8th ed.; Wiley-Blackwell: Oxford, UK, 2017. [Google Scholar]
- Parker, K.G.; White, M.G.; Cipriani, N.A. Comparison of Molecular Methods and BRAF Immunohistochemistry (VE1 Clone) for the Detection of BRAF V600E Mutation in Papillary Thyroid Carcinoma: A Meta-Analysis. Head Neck Pathol. 2020, 14, 1067–1079. [Google Scholar] [CrossRef]
- Nakao, T.; Matsuse, M.; Saenko, V.; Rogounovitch, T.; Tanaka, A.; Suzuki, K.; Higuchi, M.; Sasai, H.; Sano, T.; Hirokawa, M.; et al. Preoperative detection of the TERT promoter mutations in papillary thyroid carcinomas. Clin. Endocrinol. 2021, 95, 790–799. [Google Scholar] [CrossRef] [PubMed]
- Thomas, L.; Reyes, E.M. Tutorial: Survival Estimation for Cox Regression Models with Time-Varying Coefficients Using SAS and R. J. Stat. Softw. 2014, 61, 1–23. [Google Scholar] [CrossRef] [Green Version]
- Nenadic, O.; Greenacre, M. Correspondence Analysis in R, with two- and three-dimensional graphics: The ca package. J. Stat. Softw. 2007, 20, 1–13. [Google Scholar]

| Age Groups | Radiogenic PTC (n = 247) | Sporadic PTC (n = 138) | p-Value 1 | ||
|---|---|---|---|---|---|
| Number | % | Number | % | ||
| Children aged ≤14 years | 1/104 | 1.0 | 5/39 | 12.8 | 0.006 |
| Adolescents aged 15–18 years | 6/52 | 11.5 | 12/37 | 32.4 | 0.030 |
| Young adults aged 19–28 years | 19/91 | 20.9 | 25/62 | 40.3 | 0.011 |
| Total | 26/247 | 10.5 | 42/138 | 30.4 | 2.67 × 10−6 |
| Age trend, p-value 2 | 5.97 × 10−6 | 0.004 | |||
| Age association 3 | 1.179 (1.092–1.273); 2.40 × 10−5 | 1.118 (1.031–1.212); 0.007 | |||
| Parameters | BRAFV600E(+) (n = 26) | BRAFV600E(−) (n = 221) | p-Value | OR, b or HR (95%CI) | p-Value |
|---|---|---|---|---|---|
| Number or Value (% or IQR) | Number or Value (% or IQR) | Univariate | Multivariate 1 | ||
| Sex (F/M, %M, F:M ratio; ref = F) | 20/6 (23.1%; 3.3:1) | 141/80 (36.2%; 1.8:1) | 0.276 | 0.706 (0.260–1.914) 2 | 0.494 |
| Age at operation, years | 24.1 (18.7–27.3) | 15.4 (12.0–21.3) | 2.03 × 10−6 | 6.012 (3.552–8.472)3 | 3.00 × 10−6 |
| Age at exposure, years | 2.0 (1.0–3.0) | 2.0 (1.0–3.0) | 0.924 | −0.019 (−0.519–0.481) 3 | 0.941 |
| Period of latency, years | 22.1 (17.5–24.9) | 13.1 (9.3–18.5) | 1.24 × 10−6 | 6.133 (3.679–8.588)3 | 2.00 × 10−6 |
| Radiation dose to the thyroid, mGy | 200 (134–390) | 350 (173–825) | 0.019 | −0.295 (−0.563–0.027)3 | 0.031 |
| Tumor size, mm, median | 10 (6–20) | 16 (12–30) | 4.57 × 10−4 | −0.528 (−0.943–0.113) | 0.013 |
| ≤10 mm (microcarcinoma) | 14 (53.8%) | 36 (16.3%) | 5.44 × 10−5 | 3.475 (1.380–8.751) | 0.008 |
| Complete tumor capsule | 5 (19.2%) | 32 (14.5%) | 0.560 | 0.721 (0.239–2.174) | 0.561 |
| Dominant growth pattern | 1.98 × 10−6 | 0.334 (0.180–0.622) | 0.001 | ||
| papillary | 17 (65.4%) | 40 (18.1%) | 1.12 × 10−6 | 5.735 (2.289–14.366) | 1.93 × 10−4 |
| follicular | 5 (19.2%) | 75 (33.9%) | 0.183 | 0.410 (0.141–1.190) | 0.101 |
| solid-trabecular | 4 (15.4%) | 106 (48.0%) | 0.001 | 0.289 (0.093–0.901) | 0.032 |
| Oncocytic changes | 8 (30.8%) | 31 (14.0%) | 0.042 | 1.671 (0.629–4.441) | 0.303 |
| Multifocality | 6 (23.1%) | 30 (13.6%) | 0.235 | 1.124 (0.395–3.194) | 0.827 |
| Lymphatic/vascular invasion | 9 (34.6%) | 159 (71.9%) | 1.91 × 10−4 | 0.417 (0.165–1.052) | 0.064 |
| Extrathyroidal extension (any) | 7 (26.9%) | 119 (53.8%) | 0.012 | 0.565 (0.216–1.473) | 0.243 |
| N category (N1) | 9 (34.6%) | 122 (55.2%) | 0.061 | 0.649 (0.264–1.592) | 0.345 |
| N1a | 6 (23.1%) | 43 (19.5%) | 0.611 | 1.210 (0.434–3.376) | 0.715 |
| N1b | 3 (11.5%) | 79 (35.7%) | 0.014 | 0.392 (0.109–1.406) | 0.151 |
| M category (M1) | 1 (3.8%) | 38 (17.2%) | 0.091 | 0.340 (0.042–2.723) | 0.309 |
| pT | 0.078 | 0.605 (0.310–1.176) | 0.139 | ||
| pT1 | 21 (80.8%) | 126 (57.0%) | 0.021 | 2.437 (0.854–6.959) | 0.096 |
| pT1a | 14 (53.8%) | 36 (16.3%) | 5.40 × 10−5 | 3.475 (1.380–8.751) | 0.008 |
| pT1b | 7 (26.9%) | 90 (40.7%) | 0.206 | 0.658 (0.255–1.700) | 0.387 |
| pT2 | 2 (7.7%) | 42 (19.0%) | 0.185 | 0.338 (0.074–1.542) | 0.161 |
| pT3 | 3 (11.5%) | 53 (24.0%) | 0.216 | 0.640 (0.174–2.350) | 0.501 |
| pT3a | 1 (3.8%) | 17 (7.7%) | 0.703 | 0.634 (0.075–5.385) | 0.676 |
| pT3b | 2 (7.7%) | 36 (16.3%) | 0.389 | 0.678 (0.144–3.201) | 0.623 |
| Invasiveness score | 1 (0–2) | 2 (1–3) | 0.072 | 0.749 (0.531–1.056) | 0.100 |
| 0 | 10 (38.5%) | 39 (17.6%) | 0.018 | 1.836 (0.737–4.575) | 0.192 |
| 1 | 4 (15.4%) | 37 (16.7%) | 1.000 | 0.603 (0.187–1.941) | 0.396 |
| 2 | 8 (30.8%) | 60 (27.1%) | 0.651 | 1.306 (0.511–3.339) | 0.577 |
| 3 | 4 (15.4%) | 44 (19.9%) | 0.794 | 1.321 (0.394–4.423) | 0.652 |
| 4 | 0 | 29 (13.1%) | 0.052 | 0.148 (0.008–2.724) | 0.199 |
| 5 | 0 | 12 (5.4%) | 0.621 | 0.444 (0.017–11.596) | 0.626 |
| Ki-67 labeling index, median | 4.7 (3.8–6.3) | n = 210; 2.3 (1.3–3.9) | 7.64 × 10−6 | 1.647 (0.467–2.827) | 0.006 |
| 0–5% | 13 (50.0%) | 173 (78.3%) | 0.003 | 0.268 (0.109–0.659) | 0.004 |
| >5–10% | 11 (42.3%) | 29 (23.1%) | 7.00 × 10−4 | 3.974 (1.565–10.095) | 0.004 |
| >10% | 2 (7.7%) | 8 (3.6%) | 0.284 | 1.351 (0.252–7.253) | 0.726 |
| Follow-up, years | 9.9 (8.3–12.2) | 13.5 (8.5–18.7) | 0.016 | 0.469 (−1.978–2.916) | 0.706 |
| LN recurrence (reoperated after 6 mo) | 0 | n = 210; 8 (3.8%) | 0.574 4 | 0.523 (0.004–5.410) 5 | 0.708 |
| Parameters | BRAFV600E(+) (n = 42) | BRAFV600E(−) (n = 96) | p-Value | OR, b or HR (95%CI) | p-Value |
|---|---|---|---|---|---|
| Number or Value (% or IQR) | Number or Value (% or IQR)1 | Univariate | Multivariate 1 | ||
| Sex (F/M, %M, F:M ratio; ref = F) | 34/8 (19.0%; 4.3:1) | 81/15 (15.6%; 5.4:1) | 0.626 | 1.378 (0.518–3.670) 2 | 0.521 |
| Age at operation, years | 21.0 (16.4–24.3) | 17.1 (14.0–21.7) | 0.006 | 2.516 (0.748–4.284)3 | 0.006 |
| Tumor size, mm | 11 (8–15) | 21 (13–31) | 2.64 × 10−5 | −8.706 (−13.122–4.290) | 1.52 × 10−4 |
| ≤10 mm (microcarcinoma) | 19 (45.2%) | 18 (18.8%) | 0.002 | 3.499 (1.531–7.966) | 0.003 |
| Complete tumor capsule | 6 (14.3%) | 32 (33.3%) | 0.023 | 0.231 (0.083–0.648) | 0.005 |
| Dominant growth pattern | 1.72 × 10−4 | 0.427 (0.248–0.736) | 0.002 | ||
| papillary | 31 (73.8%) | 35 (36.5%) | 7.91 × 10−5 | 5.462 (2.355–12.666) | 7.60 × 10−5 |
| follicular | 4 (9.5%) | 33 (34.4%) | 0.003 | 0.143 (0.045–0.460) | 0.001 |
| solid-trabecular | 7 (16.7%) | 28 (29.2%) | 0.141 | 0.565 (0.219–1.460) | 0.238 |
| Oncocytic changes | 26 (61.9%) | 36 (37.5%) | 0.010 | 2.488 (1.155–5.358) | 0.020 |
| Multifocality | 10 (23.8%) | 11 (11.5%) | 0.075 | 1.905 (0.712–5.095) | 0.199 |
| Lymphatic/vascular invasion | 13 (31.0%) | 62 (64.6%) | 7.46 × 10−6 | 0.304 (0.134–0.688) | 0.004 |
| Extrathyroidal extension (any) | 11 (26.2%) | 33 (34.4%) | 0.543 | 0.897 (0.382–2.106) | 0.803 |
| N category (N1) | 15 (35.7%) | 42 (43.8%) | 0.454 | 0.793 (0.362–1.739) | 0.562 |
| N1a | 11 (26.2%) | 16 (16.7%) | 0.244 | 1.696 (0.682–4.214) | 0.255 |
| N1b | 4 (9.5%) | 26 (27.1%) | 0.025 | 0.343 (0.109–1.084) | 0.068 |
| M category (M1) | 1 (2.4%) | 5 (5.2%) | 0.407 | 0.445 (0.049–3.998) | 0.469 |
| pT | 0.078 | 0.259 (0.115–0.580) | 0.001 | ||
| pT1 | 35 (83.3%) | 48 (50.0%) | 2.72 × 10−4 | 5.098 (2.017–12.883) | 0.001 |
| pT1a | 19 (45.2%) | 18 (18.8%) | 0.002 | 3.499 (1.531–7.996) | 0.003 |
| pT1b | 16 (38.1%) | 30 (31.3%) | 0.440 | 1.427 (0.651–3.130) | 0.374 |
| pT2 | 6 (14.3%) | 36 (37.5%) | 0.008 | 0.269 (0.101–0.719) | 0.009 |
| pT3 | 1 (2.4%) | 12 (12.5%) | 0.109 | 0.175 (0.021–1.422) | 0.103 |
| pT3a | 1 (2.4%) | 9 (9.4%) | 0.282 | 0.227 (0.027–1.904) | 0.172 |
| pT3b | 0 | 3 (3.1%) | 0.553 | 0.336 (0.009–12.891) | 0.558 |
| Invasiveness score | 1 (0–2) | 2 (0–3) | 0.424 | 0.835 (0.607–1.147) | 0.266 |
| 0 | 15 (35.7%) | 27 (28.1%) | 0.423 | 1.215 (0.543–2.719) | 0.635 |
| 1 | 13 (31.0%) | 20 (20.8%) | 0.203 | 1.601 (0.688–3.728) | 0.275 |
| 2 | 7 (16.7%) | 21 (21.9%) | 0.646 | 0.705 (0.267–1.866) | 0.482 |
| 3 | 5 (11.9%) | 21 (21.9%) | 0.237 | 0.506 (0.171–1.491) | 0.217 |
| 4 | 2 (4.8%) | 7 (7.3%) | 0.722 | 1.175 (0.205–6.750) | 0.856 |
| 5 | 0 | 0 | ND | ND | ND |
| Ki-67 labeling index | n = 40; 5.9 (4.3–8.7) | 4.1 (2.5–6.9) | 0.003 | 2.221 (0.842–3.600) | 0.002 |
| 0–5% | 14 (35.0%) | 62 (64.6%) | 0.002 | 0.231 (0.101–0.531) | 5.49 × 10−4 |
| >5–10% | 20 (50.0%) | 25 (26.0%) | 0.009 | 3.109 (1.395–6.930) | 0.006 |
| >10% | 6 (15.0%) | 9 (9.4%) | 0.374 | 2.322 (0.716–7.534) | 0.161 |
| Follow-up, years | 4.9 (2.4–8.6) | 5.5 (2.6–9.3) | 0.343 | −0.144 (−1.787–1.499) | 0.863 |
| LN recurrence (reoperated after 6 mo) | n = 39; 3 (7.7%) | n = 88; 1 (1.1%) | 0.0074 | 19.042 (1.299–279.067)5 | 0.031 |
| Characteristics | BRAFV600E(+) (n = 26/42) 1 | BRAFV600E(−) (n = 221/96) 1 | ||||
|---|---|---|---|---|---|---|
| Univariate | OR, b or HR (95%CI) 2 | Multivariate | Univariate | OR, b or HR (95%CI) 2 | Multivariate | |
| p-Value | p-Value | p-Value | p-Value | |||
| Sex (ref = F) | 0.762 | 1.357 (0.389–4.731) 3 | 0.632 | 1.80 × 10−4 | 2.952 (1.590–5.482)3 | 0.001 |
| Age at operation, years | 0.015 | 2.838 (0.496–5.180)4 | 0.018 | 0.070 | −0.593 (−2.015–0.829) 4 | 0.412 |
| Tumor size, mm | 0.519 | 0.264 (−4.730–5.258) | 0.916 | 0.224 | −0.865 (−4.149–2.419) | 0.605 |
| ≤10 mm (microcarcinoma) | 0.619 | 1.262 (0.447–3.561) | 0.661 | 0.627 | 0.781 (0.404–1.508) | 0.461 |
| Complete tumor capsule | 0.737 | 1.516 (0.383–5.998) | 0.553 | 2.22 × 10−4 | 0.315 (0.171–0.581) | 2.15 × 10−4 |
| Dominant growth pattern | 0.539 | 0.955 (0.303–3.004) | 0.937 | 5.88 × 10−4 | 2.340 (1.472–3.721) | 3.25 × 10−4 |
| papillary | 0.585 | 0.938 (0.290–3.034) | 0.914 | 0.001 | 0.416 (0.239–0.724) | 0.002 |
| follicular | 0.287 | 1.779 (0.388–8.147) | 0.458 | 1 | 0.882 (0.522–1.493) | 0.641 |
| solid-trabecular | 1 | 0.622 (0.140–2.755) | 0.531 | 0.002 | 2.350 (1.370–4.032) | 0.002 |
| Oncocytic changes | 0.024 | 0.242 (0.080–0.733) | 0.012 | 9.00 × 10−6 | 0.298 (0.167–0.531) | 4.00 × 10−5 |
| Multifocality | 1 | 0.612 (0.170–2.209) | 0.454 | 0.717 | 1.281 (0.600–2.737) | 0.523 |
| Lymphatic/vascular invasion | 0.794 | 1.915 (0.594–6.172) | 0.277 | 0.231 | 1.364 (0.787–2.366) | 0.269 |
| Extrathyroidal extension | 1 | 0.946 (0.294–3.043) | 0.925 | 0.001 | 2.115 (1.244–3.595) | 0.006 |
| minimal | 1 | 0.804 (0.242–2.667) | 0.721 | 0.253 | 1.255 (0.737–2.138) | 0.403 |
| gross | 0.382 | 3.224 (0.137–60.094) | 0.433 | 0.001 | 5.219 (1.532–17.781) | 0.008 |
| N category (N1) | 1 | 0.791 (0.264–2.372) | 0.675 | 0.067 | 1.438 (0.871–2.374) | 0.155 |
| N1a | 1 | 0.683 (0.201–2.321) | 0.541 | 0.639 | 1.187 (0.623–2.265) | 0.602 |
| N1b | 1 | 1.198 (0.229–6.262) | 0.831 | 0.154 | 1.327 (0.765–2.302) | 0.315 |
| M category (M1) | 1 | 2.504 (0.128–49.133) | 0.545 | 0.055 | 2.139 (0.929–4.924) | 0.074 |
| pT | 0.306 | 1.462 (0.389–5.503) | 0.574 | 0.001 | 0.976 (0.608–1.567) | 0.920 |
| pT1 | 1 | 0.770 (0.200–2.957) | 0.703 | 0.270 | 1.361 (0.830–2.231) | 0.222 |
| pT1a | 0.619 | 1.262 (0.447–3.581) | 0.661 | 0.627 | 0.781 (0.404–1.508) | 0.461 |
| pT1b | 0.433 | 0.649 (0.214–1.974) | 0.447 | 0.131 | 1.555 (0.923–2.621) | 0.097 |
| pT2 | 0.701 | 0.563 (0.099–3.211) | 0.517 | 6.39 × 10−4 | 0.385 (0.223–0.666) | 6.49 × 10−4 |
| pT3 | 0.152 | 5.074 (0.465–55.415) | 0.183 | 0.023 | 2.155 (1.072–4.335) | 0.031 |
| pT3a | 1 | 2.504 (0.128–49.133) | 0.545 | 0.658 | 0.842 (0.354–2.006) | 0.698 |
| pT3b | 0.143 | 1.476 (0.036–60.523) | 0.837 | 6.30 × 10−4 | 5.599 (1.651–18.985) | 0.006 |
| Invasiveness score (any Ex) | 0.401 | 1.063 (0.422–2.678) | 0.897 | 0.024 | 1.644 (1.059–2.552) | 0.027 |
| 0 | 1 | 1.105 (0.379–3.222) | 0.854 | 0.050 | 0.549 (0.305–0.990) | 0.046 |
| 1 | 0.249 | 0.482 (0.133–1.754) | 0.268 | 0.427 | 0.816 (0.436–1.526) | 0.524 |
| 2 | 0.231 | 1.940 (0.574–6.558) | 0.286 | 0.401 | 1.378 (0.770–2.464) | 0.28 |
| 3 | 0.723 | 1.036 (0.228–4.714) | 0.964 | 0.762 | 0.713 (0.384–1.323) | 0.283 |
| 4 | 0.521 | 0.495 (0.027–8.990) | 0.634 | 0.177 | 1.918 (0.792–4.644) | 0.149 |
| 5 | ND 5 | ND | ND | 0.021 | 10.119 (0.618–165.760) | 0.105 |
| Ki-67 labeling index | 0.112 6 | −1.114 (−3.135–0.907) | 0.275 | 3.84 × 10−9 7 | −1.714 (−2.455–0.973) | 8.00 × 10−6 |
| 0–5% | 0.306 | 2.057 (0.702–6.031) | 0.189 | 0.001 | 2.283 (1.304–3.995) | 0.004 |
| >5–10% | 0.618 | 0.560 (0.191–1.645) | 0.292 | 0.015 | 0.488 (0.264–0.903) | 0.022 |
| >10% | 0.464 | 0.710 (0.117–4.312) | 0.71 | 0.061 | 0.473 (0.174–1.283) | 0.142 |
| Follow-up, years (n = 26/39) | 3.39 × 10−5 | 13.324 (8.434–18.213) | 8.82 × 10−7 | 1.50 × 10−14 | 6.413 (5.048–7.777) | 3.81 × 10−18 |
| LN recurrences (reoperated after 6 mo) | 0.0278 | 0.031 (0.000–0.870) 9 | 0.147 | 0.872 8 | 0.694 (0.074–6.535) 10 | 0.750 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zurnadzhy, L.; Bogdanova, T.; Rogounovitch, T.I.; Ito, M.; Tronko, M.; Yamashita, S.; Mitsutake, N.; Chernyshov, S.; Masiuk, S.; Saenko, V.A. The BRAFV600E Mutation Is Not a Risk Factor for More Aggressive Tumor Behavior in Radiogenic and Sporadic Papillary Thyroid Carcinoma at a Young Age. Cancers 2021, 13, 6038. https://doi.org/10.3390/cancers13236038
Zurnadzhy L, Bogdanova T, Rogounovitch TI, Ito M, Tronko M, Yamashita S, Mitsutake N, Chernyshov S, Masiuk S, Saenko VA. The BRAFV600E Mutation Is Not a Risk Factor for More Aggressive Tumor Behavior in Radiogenic and Sporadic Papillary Thyroid Carcinoma at a Young Age. Cancers. 2021; 13(23):6038. https://doi.org/10.3390/cancers13236038
Chicago/Turabian StyleZurnadzhy, Liudmyla, Tetiana Bogdanova, Tatiana I. Rogounovitch, Masahiro Ito, Mykola Tronko, Shunichi Yamashita, Norisato Mitsutake, Serhii Chernyshov, Sergii Masiuk, and Vladimir A. Saenko. 2021. "The BRAFV600E Mutation Is Not a Risk Factor for More Aggressive Tumor Behavior in Radiogenic and Sporadic Papillary Thyroid Carcinoma at a Young Age" Cancers 13, no. 23: 6038. https://doi.org/10.3390/cancers13236038
APA StyleZurnadzhy, L., Bogdanova, T., Rogounovitch, T. I., Ito, M., Tronko, M., Yamashita, S., Mitsutake, N., Chernyshov, S., Masiuk, S., & Saenko, V. A. (2021). The BRAFV600E Mutation Is Not a Risk Factor for More Aggressive Tumor Behavior in Radiogenic and Sporadic Papillary Thyroid Carcinoma at a Young Age. Cancers, 13(23), 6038. https://doi.org/10.3390/cancers13236038






