Integrin αvβ6 as a Target for Tumor-Specific Imaging of Vulvar Squamous Cell Carcinoma and Adjacent Premalignant Lesions
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Tissue Samples
2.2. Immunohistochemistry
2.3. Digital Pathology Image Analysis
2.4. Marker Staining
2.5. FGS Criteria
- a median H-score in (pre)malignant tissue being at least twice as high as the median H-score in healthy control and stromal tissue [34];
- a minimum median H-score in (pre)malignant tissue of at least 25;
- homogeneous expression throughout the tumor;
- cell surface protein expression.
2.6. Statistics
3. Results
3.1. Tissue Characteristics
3.2. Immunohistochemical Marker Staining
3.2.1. αvβ6—Integrin Alphavbeta6
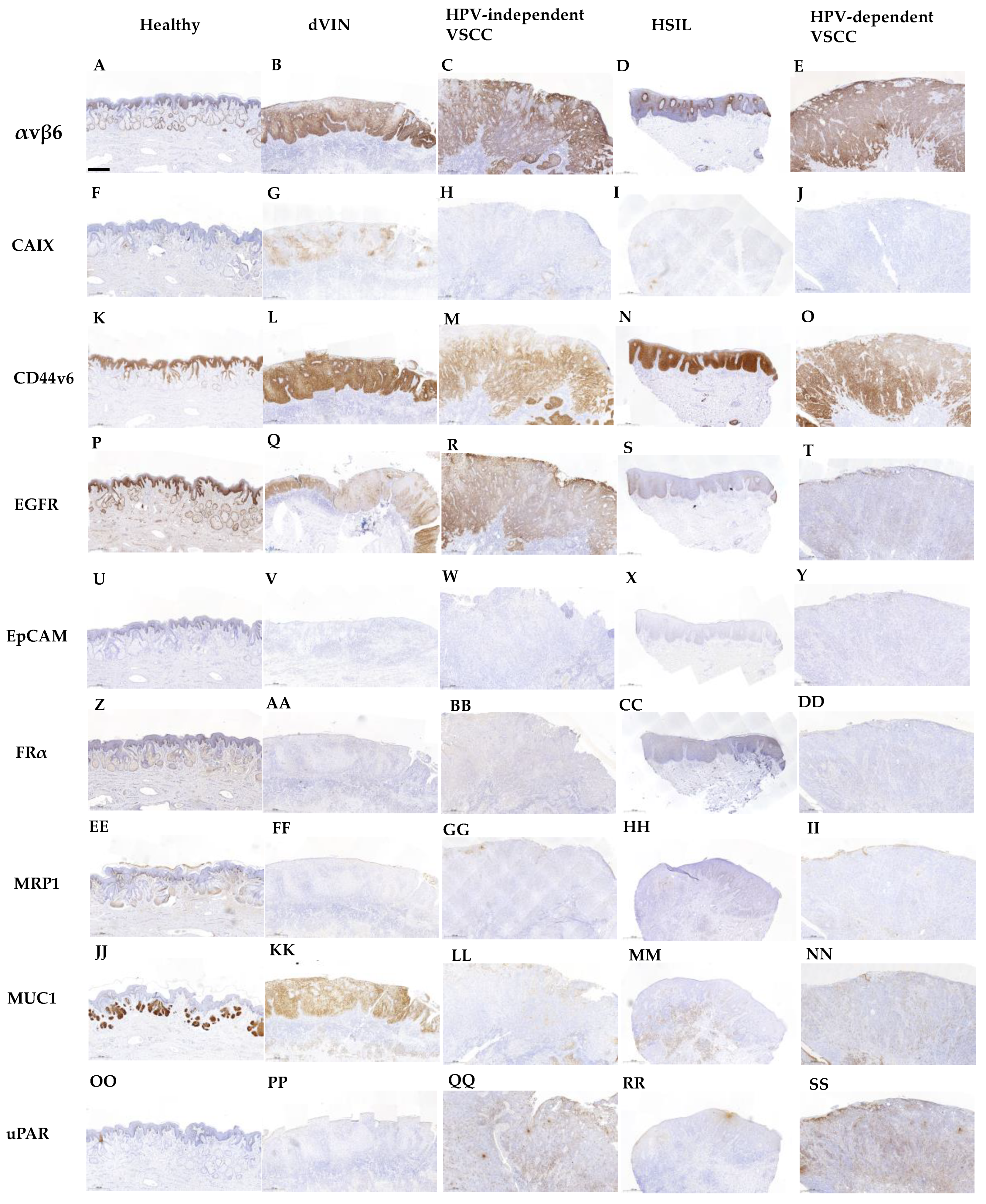
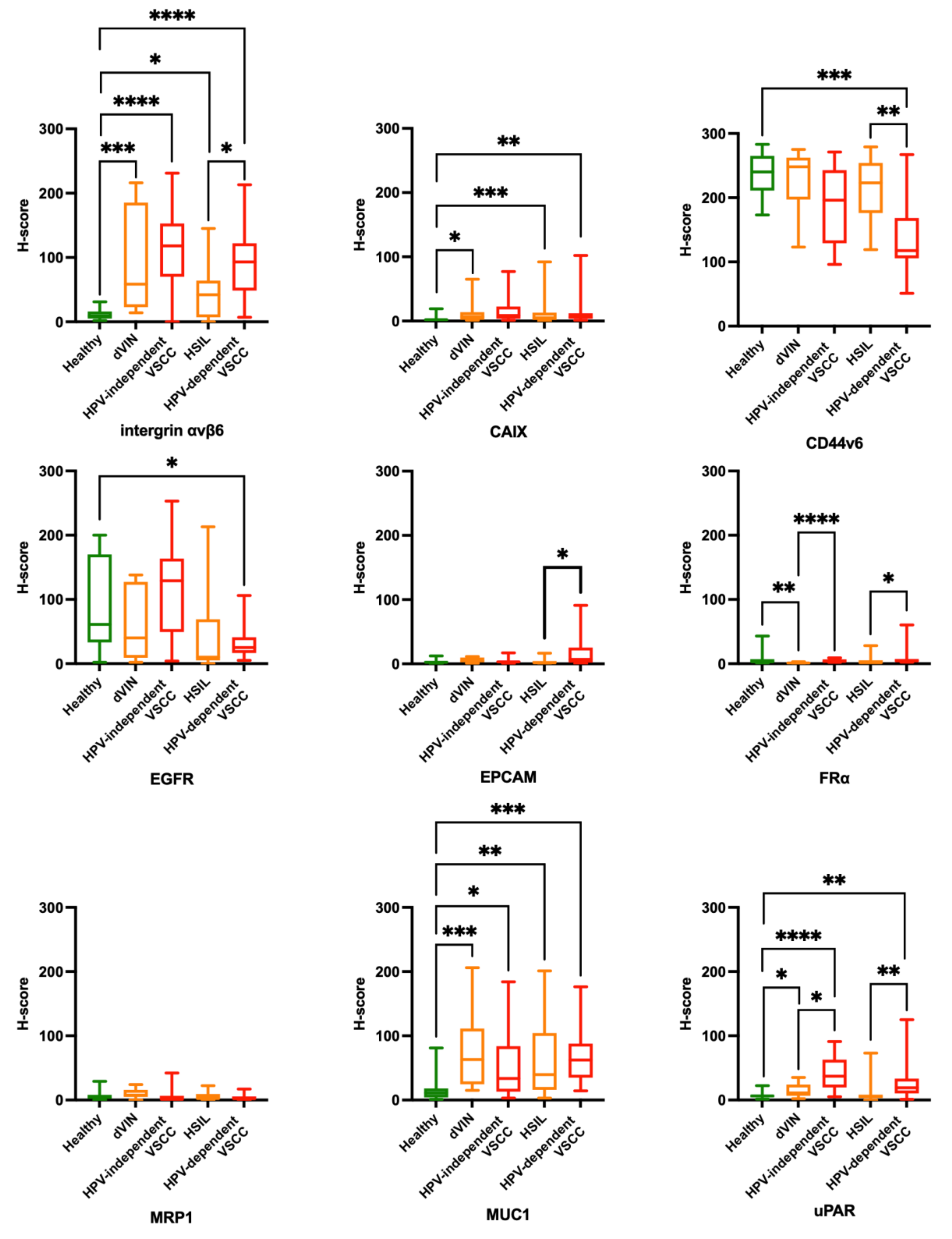
- Stromal tissue lacked αvβ6 expression. Healthy vulvar epithelium showed no or low expression of αvβ6. If αvβ6 was present in healthy vulvar tissue, it was mainly located in the spinosal and basal layer of the epithelium (Figure 1A). In addition, αvβ6 expression was higher in normal vulvar tissues wherein sebaceous glands were present (11/15 healthy vulvar tissues) compared with vulvar tissue sections that lacked those glands. αvβ6 staining within sebaceous glands was low to moderate (Figure 1A). The median H-score of healthy vulvar tissue was significantly lower compared with median H-scores of all vulvar (pre)malignant tissue types (Figure 2), resulting in TBRs > 2 (Table 2).
- Moderate αvβ6 expression was observed in 4/8 dVIN, 14/16 HPV-independent VSCC, 4/13 HSIL and 10/13 HPV-dependent VSCC tissues (Figure 1B–E respectively). αvβ6 expression lacked in 2/16 HPV-independent VSCC and 3/13 HPV-dependent VSCC tissues. The other premalignant tissues showed low expression. More intense αvβ6 staining was found in HSIL adjacent to HPV-dependent VSCC (average H-score 42) compared with isolated HSIL (average H-score 114). This difference was not observed for dVIN. Median H-scores per vulvar (pre)malignant tissues type were all above 25 (Table 2).
- αvβ6 was homogeneously expressed in all HPV-independent VSCC tissues. 2/10 HPV-dependent VSCC tissues showed a patchy staining pattern throughout the tumor, for 2/10 expression was restricted to the spinosal and/or basal layers, the remainder showed homogeneous expression. To a greater or lesser extent in all dVIN and HSIL tissues, as for healthy vulvar tissue, αvβ6 expression was restricted to the spinosal and/or basal layers (Figure 1A,D).
- αvβ6 showed cell membrane staining.
3.2.2. CAIX—Carbonic Anhydrase IX
- Stromal and healthy vulvar epithelium lacked CAIX staining (Figure 1F). The median H-score of healthy vulvar tissues was significantly lower compared with median H-scores of dVIN, HSIL and HPV-dependent VSCC tissue groups (Figure 2), resulting in TBRs > 2 (Table 2). The median H-score of HPV-independent VSCC tissue group was not tested significantly higher compared with the median H-score of healthy vulvar tissue (TBR 4.5, Table 2).
- If CAIX staining was observed, it was positioned in the spinosal and/or basal layers of the vulvar epidermis in a heterogeneous and patchy pattern (Figure 1G–J).
- CAIX showed cell membrane staining.
3.2.3. CD44v6—CD44 Variant 6
- Stromal tissue lacked CD44v6 staining. Healthy vulvar epithelium showed in 7/15 tissues high CD44v6 expression, the remaining tissues showed moderate expression (Figure 1K). TBRs were inverse for all (pre)malignant vulvar tissue types, indicating downregulation of CD44v6 in (pre)malignant compared with healthy tissue (Figure 2). Consequently, TBRs were not in favor for FGS application at the surface of the vulva (Table 2).
- Predominantly moderate CD44v6 staining was observed in vulvar (pre)malignant tissues (Figure 1L–O), in 5/10 dVIN, 3/16 HPV-independent VSCC, 5/15 HSIL and 1/12 HPV-dependent VSCC tissues high CD44v6 expression was observed. Median CD44v6 H-scores per vulvar (pre)malignant tissues type were all above 25 (Table 2).
- CD44v6 showed homogenous expression.
- CD44v6 showed cell membrane staining.
3.2.4. EGFR—Epithelial Cell Adhesion Molecule
- EGFR staining was observed in glands, blood vessels and adnexa. Healthy vulvar epithelium showed moderate EGFR expression in 10/15 tissues (Figure 1P) and low expression in 5/15 tissues. TBRs were inverse for all (pre)malignant vulvar tissue types, indicating downregulation of EGFR in (pre)malignant tissue compared with healthy (Figure 2) Consequently, TBRs were not in favor for FGS application at the surface of the vulva (Table 2).
- EGFR was moderately expressed in 5/10 dVIN, 11/16 HPV-independent VSCC, 5/15 HSIL and 2/13 HPV-dependent VSCC tissues (Figure 1Q,R), the expression in the remaining samples was low (Figure 1S,T, except 1 HPV-independent VSCC with high expression). HSIL showed a median H-score below 25, the H-scores for other vulvar (pre)malignant tissue types were at least 25 (Table 2).
- EGFR was gradually expressed in healthy vulvar epithelium, being more strongly expressed in the stratum basal compared with the stratum corneum. For (pre)malignant tissues the expression patterns were diverse. Homogenous (Figure 1R), patchy (Figure 1Q,S) and on/off expression patterns (Figure 1T) were observed in these tissues.
- EGFR showed cell membrane staining.
3.2.5. EpCAM—Epithelial Cell Adhesion Molecule
- EpCAM staining was not observed in stromal tissue, except for the endothelial lining of blood vessels. Healthy vulvar epithelium lacked EpCAM expression (Figure 1U). The median H-score of healthy vulvar tissue was not significantly different compared with any vulvar (pre)malignant tissue group (Figure 2), resulting in TBRs < 2, except for dVIN with an TBR of 2.5 (Table 2).
- No pattern could be recognized due to the low expression of EpCAM in vulvar tissues.
- EpCAM showed cell membrane staining on the endothelial lining of blood vessels.
3.2.6. FRα—Folate Receptor α
- No pattern could be recognized due to the low expression of FRα in all vulvar tissues.
- Cell membrane staining for FRα was observed in lung tumor tissue (control).
3.2.7. MRP1—Multidrug Resistance-Associated Protein
- Low to moderate MRP1 staining was observed in stromal cells and several sebaceous glands of a few healthy and (pre) malignant tissues. No MRP1 expression was observed in healthy vulvar epithelium (Figure 1EE). The median H-score of healthy vulvar tissue was not significantly lower compared with any median H-score of (pre)malignant tissues (Figure 2), resulting in TBRs < 2 (Table 2).
- No expression pattern could be recognized due to the overall low expression of MRP1.
- In both stromal vulvar tissue as in placental tissue (control), cytoplasmatic and membranous presence of MRP1 was observed on cells.
3.2.8. MUC1—Mucin 1
- Stromal tissue lacked MUC1 staining, except for sebaceous glands positioned in the dermis, which showed moderate or high MUC1 expression (Figure 1JJ). Half of the healthy vulvar epithelial tissues lacked MUC1 expression (Figure 1JJ), others showed low expression restricted to the stratum spinosum. The median H-score of healthy vulvar tissue was significantly lower compared with median H-scores of all vulvar (pre)malignant tissue types (Figure 2), resulting in TBRs > 2 (Table 2).
- Moderate MUC1 expression was observed in 5/10 dVIN (Figure 1 KK), 6/16 HPV-dependent VSCC, 6/14 HSIL and 7/13 HPV-dependent VSCC tissues, the remaining tissues showed low expression (Figure 1LL–NN). Median H-scores for MUC1 expression per vulvar (pre)malignant tissues type were all above 25 (Table 2).
- The expression pattern was heterogenous and patchy throughout all tissue samples.
- MUC1 showed cell membrane staining.
3.2.9. uPAR—Urokinase Plasminogen Activator Receptor
- Low stromal expression of uPAR was observed in healthy and (pre)malignant tissues. Healthy vulvar epithelium lacked uPAR staining(Figure 1OO). The median H-score of healthy vulvar tissues was significantly lower compared with median H-scores of dVIN, HPV-dependent and independent VSCC tissue groups (Figure 2), resulting in TBRs > 2 (Table 2). For the HSIL group, the TBR < 2.
- Moderate uPAR expression was observed in 4/12 HPV-independent VSCC (Figure 1QQ), 1/12 HSIL and 2/12 HPV-dependent VSCC tissues (Figure 1SS), the remaining vulvar (pre)malignant tissues showed low or absent expression (Figure 1PP,RR). Only the median H-score for uPAR expression in the HPV-independent VSCC tissue group was above 25 (Table 2).
- uPAR was heterogeneously expressed throughout (pre)malignant vulvar tissue.
- uPAR showed cell membrane staining and sometimes cytoplasmatic staining in cells.
3.3. Evaluation of FGS Criteria
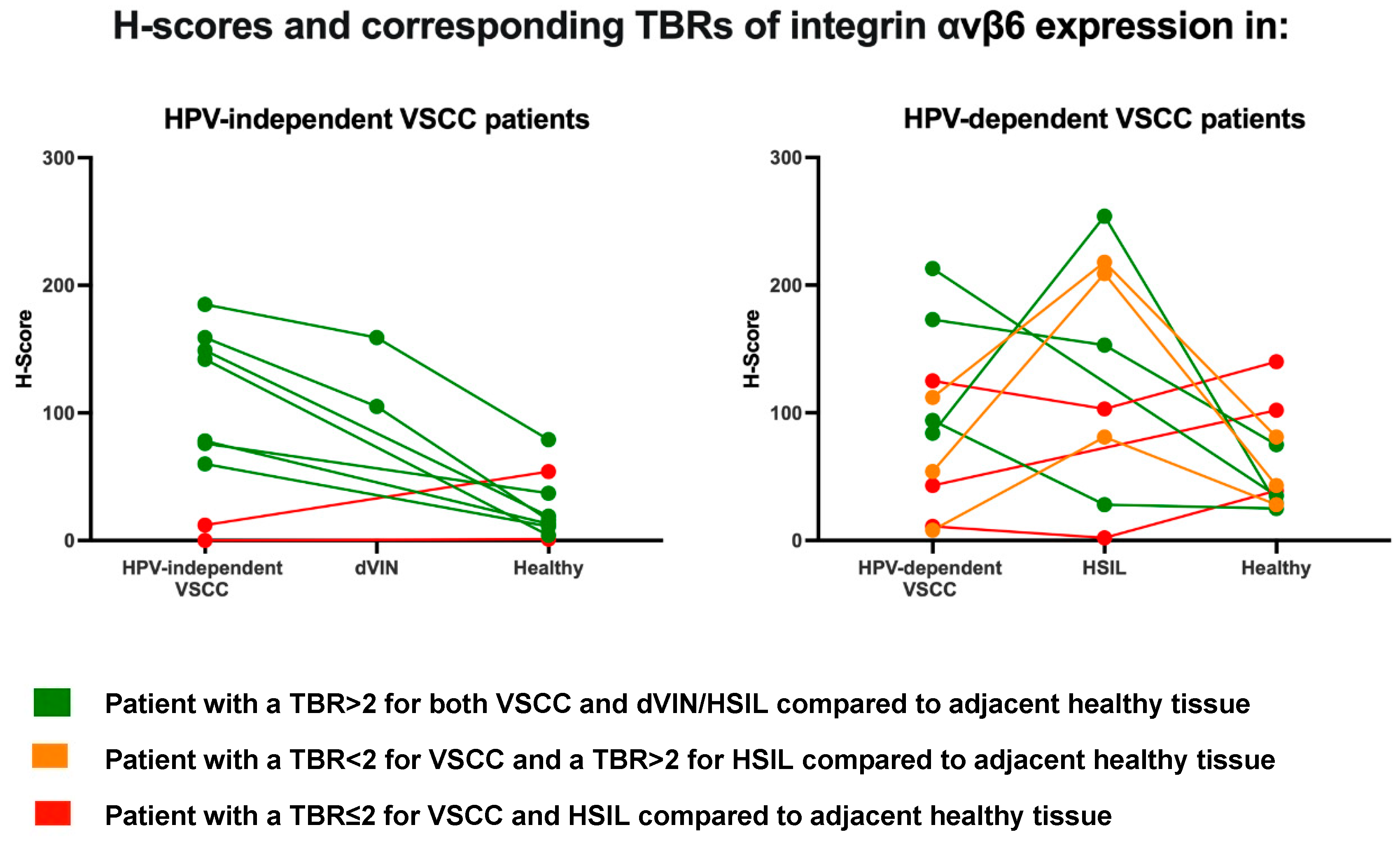
3.4. αvβ6 Expression in Individual VSCC Tissue Sections
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Eva, L.J.; Sadler, L.; Fong, K.L.; Sahota, S.; Jones, R.W.; Bigby, S.M. Trends in HPV-dependent and HPV-independent vulvar cancers: The changing face of vulvar squamous cell carcinoma. Gynecol. Oncol. 2020, 157, 450–455. [Google Scholar] [CrossRef]
- Tan, A.; Bieber, A.K.; Stein, J.; Pomeranz, M.K. Diagnosis and management of vulvar cancer: A review. J. Am. Acad. Dermatol. 2019, 81, 1387–1396. [Google Scholar] [CrossRef]
- Schuurman, M.; Einden, L.V.D.; Massuger, L.; Kiemeney, L.; van der Aa, M.; de Hullu, J. Trends in incidence and survival of Dutch women with vulvar squamous cell carcinoma. Eur. J. Cancer 2013, 49, 3872–3880. [Google Scholar] [CrossRef]
- Thuijs, N.B.; Van Beurden, M.; Bruggink, A.H.; Steenbergen, R.D.M.; Berkhof, J.; Bleeker, M.C.G. Vulvar intraepithelial neoplasia: Incidence and long-term risk of vulvar squamous cell carcinoma. Int. J. Cancer 2021, 148, 90–98. [Google Scholar] [CrossRef]
- Hinten, F.; Molijn, A.; Eckhardt, L.; Massuger, L.; Quint, W.; Bult, P.; Bulten, J.; Melchers, W.; de Hullu, J. Vulvar cancer: Two pathways with different localization and prognosis. Gynecol. Oncol. 2018, 149, 310–317. [Google Scholar] [CrossRef] [PubMed]
- McAlpine, J.N.; Kim, S.Y.; Akbari, A.; Eshragh, S.; Reuschenbach, M.; Doeberitz, M.V.K.; Prigge, E.S.; Jordan, S.; Singh, N.; Miller, D.M.; et al. HPV-independent Differentiated Vulvar Intraepithelial Neoplasia (dVIN) is Associated With an Aggressive Clinical Course. Int. J. Gynecol. Pathol. 2017, 36, 507–516. [Google Scholar] [CrossRef] [PubMed]
- Oonk, M.H.; Planchamp, F.; Baldwin, P.; Bidzinski, M.; Brännström, M.; Landoni, F.; Mahner, S.; Mahantshetty, U.; Mirza, M.; Petersen, C.; et al. European Society of Gynaecological Oncology Guidelines for the Management of Patients With Vulvar Cancer. Int. J. Gynecol. Cancer 2017, 27, 832–837. [Google Scholar] [CrossRef] [PubMed]
- Dellinger, T.H.; Hakim, A.A.; Lee, S.J.; Wakabayashi, M.T.; Morgan, R.J.; Han, E.S. Surgical Management of Vulvar Cancer. J. Natl. Compr. Cancer Netw. 2016, 15, 121–128. [Google Scholar] [CrossRef]
- Nooij, L.; Brand, F.; Gaarenstroom, K.; Creutzberg, C.; de Hullu, J.; van Poelgeest, M. Risk factors and treatment for recurrent vulvar squamous cell carcinoma. Crit. Rev. Oncol. 2016, 106, 1–13. [Google Scholar] [CrossRef] [Green Version]
- Gaarenstroom, K.N.; Kenter, G.G.; Trimbos, J.B.; Agous, I.; Amant, F.; Peters, A.A.W.; Vergote, I. Postoperative complications after vulvectomy and inguinofemoral lymphadenectomy using separate groin incisions. Int. J. Gynecol. Cancer 2003, 13, 522–527. [Google Scholar] [CrossRef]
- Muigai, J.; Jacob, L.; Dinas, K.; Kostev, K.; Kalder, M. Potential delay in the diagnosis of vulvar cancer and associated risk factors in women treated in German gynecological practices. Oncotarget 2018, 9, 8725–8730. [Google Scholar] [CrossRef] [Green Version]
- Micheletti, L.; Preti, M.; Cintolesi, V.; Corvetto, E.; Privitera, S.; Palmese, E.; Benedetto, C. Prognostic impact of reduced tumor-free margin distance on long-term survival in FIGO stage IB/II vulvar squamous cell carcinoma. J. Gynecol. Oncol. 2018, 29, e61. [Google Scholar] [CrossRef] [Green Version]
- Nooij, L.S.; van der Slot, M.A.; Dekkers, O.M.; Stijnen, T.; Gaarenstroom, K.N.; Creutzberg, C.L.; Smit, V.T.H.B.M.; Bosse, T.; Van Poelgeest, M.I.E. Tumour-free margins in vulvar squamous cell carcinoma: Does distance really matter? Eur. J. Cancer 2016, 65, 139–149. [Google Scholar] [CrossRef]
- Vahrmeijer, A.L.; Hutteman, M.; Van Der Vorst, J.R.; Van De Velde, C.J.H.; Frangioni, J.V. Image-guided cancer surgery using near-infrared fluorescence. Nat. Rev. Clin. Oncol. 2013, 10, 507–518. [Google Scholar] [CrossRef] [Green Version]
- Rosenthal, E.L.; Warram, J.M.; de Boer, E.; Basilion, J.P.; Biel, M.A.; Bogyo, M.; Bouvet, M.; Brigman, B.E.; Colson, Y.L.; DeMeester, S.R.; et al. Successful Translation of Fluorescence Navigation During Oncologic Surgery: A Consensus Report. J. Nucl. Med. 2016, 57, 144–150. [Google Scholar] [CrossRef] [Green Version]
- Boonstra, M.C.; De Geus, S.W.; Prevoo, H.A.; Hawinkels, L.; Van De Velde, C.J.; Kuppen, P.J.; Vahrmeijer, A.L.; Sier, C.F. Selecting Targets for Tumor Imaging: An Overview of Cancer-Associated Membrane Proteins. Biomarkers Cancer 2016, 8, BIC–S38542. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hernot, S.; van Manen, L.; Debie, P.; Mieog, J.S.D.; Vahrmeijer, A.L. Latest developments in molecular tracers for fluorescence image-guided cancer surgery. Lancet Oncol. 2019, 20, e354–e367. [Google Scholar] [CrossRef]
- De Muynck, L.D.A.N.; Gaarenstroom, K.N.; Sier, C.F.M.; Van Duijvenvoorde, M.; Bosse, T.; Mieog, J.S.D.; De Kroon, C.D.; Vahrmeijer, A.L.; Peters, I.T.A. Novel Molecular Targets for Tumor-Specific Imaging of Epithelial Ovarian Cancer Metastases. Cancers 2020, 12. [Google Scholar] [CrossRef] [PubMed]
- Hollandsworth, H.M.; Turner, M.A.; Hoffman, R.M.; Bouvet, M. A review of tumor-specific fluorescence-guided surgery for colorectal cancer. Surg. Oncol. 2021, 36, 84–90. [Google Scholar] [CrossRef]
- Baart, V.M.; Van Duijn, C.; Van Egmond, S.L.; Dijckmeester, W.A.; Jansen, J.C.; Vahrmeijer, A.L.; Sier, C.F.M.; Cohen, D. EGFR and αvβ6 as Promising Targets for Molecular Imaging of Cutaneous and Mucosal Squamous Cell Carcinoma of the Head and Neck Region. Cancers 2020, 12, 1474. [Google Scholar] [CrossRef]
- Odenthal, J.; Rijpkema, M.; Bos, D.; Wagena, E.; Croes, H.; Grenman, R.; Boerman, O.; Takes, R.; Friedl, P. Targeting CD44v6 for fluorescence-guided surgery in head and neck squamous cell carcinoma. Sci. Rep. 2018, 8, 1–11. [Google Scholar] [CrossRef]
- Huisman, B.; Burggraaf, J.; Vahrmeijer, A.; Schoones, J.; Rissmann, R.; Sier, C.; Van Poelgeest, M. Potential targets for tumor-specific imaging of vulvar squamous cell carcinoma: A systematic review of candidate biomarkers. Gynecol. Oncol. 2020, 156, 734–743. [Google Scholar] [CrossRef]
- Christensen, A.; Juhl, K.; Persson, M.; Charabi, B.W.; Mortensen, J.; Kiss, K.; Lelkaitis, G.; Rubek, N.; Von Buchwald, C.; Kjær, A. uPAR-targeted optical near-infrared (NIR) fluorescence imaging and PET for image-guided surgery in head and neck cancer: Proof-of-concept in orthotopic xenograft model. Oncotarget 2016, 8, 15407–15419. [Google Scholar] [CrossRef] [Green Version]
- Boonstra, M.; Van Driel, P.; Keereweer, S.; Prevoo, H.; Stammes, M.; Baart, V.; Löwik, C.; Mazar, A.; van de Velde, C.; Vahrmeijer, A.; et al. Preclinical uPAR-targeted multimodal imaging of locoregional oral cancer. Oral Oncol. 2017, 66, 1–8. [Google Scholar] [CrossRef]
- Cheng, A.S.; Karnezis, A.N.; Jordan, S.; Singh, N.; McAlpine, J.N.; Gilks, C.B. p16 Immunostaining Allows for Accurate Subclassification of Vulvar Squamous Cell Carcinoma Into HPV-Associated and HPV-Independent Cases. Int. J. Gynecol. Pathol. 2016, 35, 385–393. [Google Scholar] [CrossRef]
- Kortekaas, K.E.; Bastiaannet, E.; van Doorn, H.C.; de Vos van Steenwijk, P.J.; Ewing-Graham, P.C.; Creutzberg, C.L.; Akdeniz, K.; Nooij, L.S.; van der Burg, S.H.; Bosse, T.; et al. Vulvar cancer subclassification by HPV and p53 status results in three clinically distinct subtypes. Gynecol. Oncol. 2020, 159, 649–656. [Google Scholar] [CrossRef]
- Dasgupta, S.; Ewing-Graham, P.C.; Swagemakers, S.M.; van der Spek, P.J.; van Doorn, H.C.; Hegt, V.N.; Koljenović, S.; van Kemenade, F.J. Precursor lesions of vulvar squamous cell carcinoma – histology and biomarkers: A systematic review. Crit. Rev. Oncol. 2020, 147, 102866. [Google Scholar] [CrossRef]
- Rakislova, N.; Alemany, L.; Clavero, O.; Saco, A.; Torné, A.; Del Pino, M.; Munmany, M.; Rodrigo-Calvo, M.; Guerrero, J.; Marimon, L.; et al. p53 Immunohistochemical Patterns in HPV-Independent Squamous Cell Carcinomas of the Vulva and the Associated Skin Lesions: A Study of 779 Cases. Int. J. Mol. Sci. 2020, 21, 8091. [Google Scholar] [CrossRef] [PubMed]
- Bankhead, P.; Loughrey, M.B.; Fernández, J.A.; Dombrowski, Y.; McArt, D.G.; Dunne, P.D.; McQuaid, S.; Gray, R.T.; Murray, L.J.; Coleman, H.G.; et al. QuPath: Open source software for digital pathology image analysis. Sci. Rep. 2017, 7, 1–7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Berben, L.; Wildiers, H.; Marcelis, L.; Antoranz, A.; Bosisio, F.; Hatse, S.; Floris, G. Computerised scoring protocol for identification and quantification of different immune cell populations in breast tumour regions by the use of QuPath software. Histopathology 2020, 77, 79–91. [Google Scholar] [CrossRef] [PubMed]
- Greene, C.; O’Doherty, E.; Sidi, F.A.; Bingham, V.; Fisher, N.C.; Humphries, M.P.; Craig, S.G.; Harewood, L.; McQuaid, S.; Lewis, C.; et al. The Potential of Digital Image Analysis to Determine Tumor Cell Content in Biobanked Formalin-Fixed, Paraffin-Embedded Tissue Samples. Biopreservation Biobanking 2021, 19, 324–331. [Google Scholar] [CrossRef] [PubMed]
- Thike, A.A.; Chng, M.J.; Tan, P.H.; Fook-Chong, S. Immunohistochemical expression of hormone receptors in invasive breast carcinoma: Correlation of results of H-score with pathological parameters. Pathology 2001, 33, 21–25. [Google Scholar] [CrossRef]
- Van Oosten, M.; Crane, L.M.; Bart, J.; van Leeuwen, F.W.; van Dam, G.M. Selecting Potential Targetable Biomarkers for Imaging Purposes in Colorectal Cancer Using TArget Selection Criteria (TASC): A Novel Target Identification Tool. Transl. Oncol. 2011, 4, 71–82. [Google Scholar] [CrossRef] [Green Version]
- Hoogstins, C.E.; Weixler, B.; Boogerd, L.S.; Hoppener, D.J.; Prevoo, H.A.; Sier, C.; Burger, J.W.; Verhoef, C.; Bhairosingh, S.; Sarasqueta, A.F.; et al. In Search for Optimal Targets for Intraoperative Fluorescence Imaging of Peritoneal Metastasis From Colorectal Cancer. Biomarkers Cancer 2017, 9, 1179299–1772825. [Google Scholar] [CrossRef] [Green Version]
- Aleksandrov, A.; Meshulam, M.; Smith, A.V.; Chauvet, P.; Canis, M.; Bourdel, N. Fluorescence-guided management of deep endometriosis. Fertil. Steril. 2020, 114, 1116–1118. [Google Scholar] [CrossRef]
- Chantima, W.; Thepthai, C.; Cheunsuchon, P.; Dharakul, T. EpCAM expression in squamous cell carcinoma of the uterine cervix detected by monoclonal antibody to the membrane-proximal part of EpCAM. BMC Cancer 2017, 17, 811. [Google Scholar] [CrossRef] [Green Version]
- Bolognia, J.L. Aging skin. Am. J. Med. 1995, 98, S99–S103. [Google Scholar] [CrossRef]
- Koivisto, L.; Bi, J.; Häkkinen, L.; Larjava, H. Integrin αvβ6: Structure, function and role in health and disease. Int. J. Biochem. Cell Biol. 2018, 99, 186–196. [Google Scholar] [CrossRef]
- Desgrosellier, J.S.; Cheresh, D.A. Integrins in cancer: Biological implications and therapeutic opportunities. Nat. Rev. Cancer 2010, 10, 9–22. [Google Scholar] [CrossRef] [Green Version]
- Binmadi, N.; Elsissi, A.; Elsissi, N. Expression of cell adhesion molecule CD44 in mucoepidermoid carcinoma and its association with the tumor behavior. Head Face Med. 2016, 12, 1–5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, J.; Ln, H. A Review of Prognostic Factors in Squamous Cell Carcinoma of the Vulva: Evidence from the Last Decade. Semin. Diagn. Pathol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Tummers, W.S.; Kimura, R.H.; Abou-Elkacem, L.; Beinat, C.; Vahrmeijer, A.L.; Swijnenburg, R.-J.; Willmann, J.K.; Gambhir, S.S. Development and Preclinical Validation of a Cysteine Knottin Peptide Targeting Integrin αvβ6 for Near-infrared Fluorescent-guided Surgery in Pancreatic Cancer. Clin. Cancer Res. 2018, 24, 1667–1676. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hausner, S.H.; DiCara, D.; Marik, J.; Marshall, J.F.; Sutcliffe, J.L. Use of a peptide derived from foot-and-mouth disease virus for the noninvasive imaging of human Cancer: Generation and evaluation of 4-[18F]fluorobenzoyl A20FMDV2 for in vivo imaging of integrin αvβ6 expression with positron emission tomography. Cancer Res. 2007, 67, 7833–7840. [Google Scholar] [CrossRef] [Green Version]
- De Valk, K.S.; Deken, M.M.; Handgraaf, H.J.M.; Bhairosingh, S.S.; Bijlstra, O.D.; Van Esdonk, M.J.; Van Scheltinga, A.G.T.; Valentijn, A.R.P.; March, T.L.; Vuijk, J.; et al. First-in-Human Assessment of cRGD-ZW800-1, a Zwitterionic, Integrin-Targeted, Near-Infrared Fluorescent Peptide in Colon Carcinoma. Clin. Cancer Res. 2020, 26, 3990–3998. [Google Scholar] [CrossRef]
- Saleem, A.; Helo, Y.; Win, Z.; Dale, R.; Cook, J.; Searle, G.E.; Wells, P. Integrin αvβ6 Positron Emission Tomography Imaging in Lung Cancer Patients Treated With Pulmonary Radiation Therapy. Int. J. Radiat. Oncol. 2020, 107, 370–376. [Google Scholar] [CrossRef]
- Sachindra, S.; Hellberg, T.; Exner, S.; Prasad, S.; Beindorff, N.; Rogalla, S.; Kimura, R.; Gambhir, S.S.; Wiedenmann, B.; Grötzinger, C. SPECT/CT Imaging, Biodistribution and Radiation Dosimetry of a 177Lu-DOTA-Integrin αvβ6 Cystine Knot Peptide in a Pancreatic Cancer Xenograft Model. Front. Oncol. 2021, 11. [Google Scholar] [CrossRef]
- Liang, B.; Shahbaz, M.; Wang, Y.; Gao, H.; Fang, R.; Niu, Z.; Liu, S.; Wang, B.; Sun, Q.; Niu, W.; et al. Integrinβ6-Targeted Immunoliposomes Mediate Tumor-Specific Drug Delivery and Enhance Therapeutic Efficacy in Colon Carcinoma. Clin. Cancer Res. 2015, 21, 1183–1195. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Niehues, H.; Bouwstra, J.A.; El Ghalbzouri, A.; Brandner, J.M.; Zeeuwen, P.L.J.M.; Van Den Bogaard, E.H. 3D skin models for 3R research: The potential of 3D reconstructed skin models to study skin barrier function. Exp. Dermatol. 2018, 27, 501–511. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mathes, S.H.; Ruffner, H.; Graf-Hausner, U. The use of skin models in drug development. Adv. Drug Deliv. Rev. 2014, 69–70, 81–102. [Google Scholar] [CrossRef]
| Biomarker | Source | Clone Number | Catalogue Number | Stock | Dilution | Antigen Retrieval | Positive Control |
|---|---|---|---|---|---|---|---|
| αvβ6 | Biogen, Inc., Cambridge, MA, USA | 6.2A1 | 62A1CEO2 | 50 µg/mL | 1/100 | 0.4% pepsin (S3002 Agilent) 37 °C for 15 min. | Normal colon |
| CA IX | Santa Cruz Biotechnology, Inc., Danvers, MA, USA | H-11 | Sc-365900 | 200 µg/mL | 1/2500 | Target retrieval solution, pH 6.1 (K8005 Agilent) | Normal stomach |
| CD44v6 | Abcam, Cambridge, UK | VFF7 | ab30436 | 1 mg/mL | 1/3200 | Target retrieval solution, pH 6.1 (K8005 Agilent) | Normal skin |
| EGFR | Dako, Glostrup, Denmark | E30 | M7239 | 286 µg/mL | 1/600 | 0.4% pepsin (S3002 Agilent) 37 °C for 10 min. | Normal placenta |
| EpCAM | LUMC, department of pathology 1 | 323/A3 | - | 0.4 mg/mL | 1/1600 | 0.1% trypsin (T7409 Sigma Aldrich) 37 °C for 30 min. | Colon tumor |
| FRα | BioCare Medical, Pacheco, CA, USA | 26B3.F2 | BRI 4006K AA (kit) | Assay kit | N.A. | Ready-to-use | Lung tumor |
| MRP1 | Santa Cruz Biotechnology, Inc., Danvers, MA, USA | QCRL-1 | Sc-18835 | 200 µg /mL | 1/400 | Target retrieval solution, pH 6.1 (K8005 Agilent) | Normal placenta |
| MUC1 | Invitrogen, Waltham, MA, USA | E29 | MA5-14077 | 0.2 mg/mL | 1/4800 | Target retrieval solution, pH 9.0 (K8004 Agilent) | Normal colon |
| uPAR | Monopar 2 | ATN617 | - | 0.48 mg/mL | 1/200 | Target retrieval solution, pH 6.1 (K8005 Agilent) | Colon tumor |
| p16 | Roche, Almere, The Netherlands | E6H4 | 06695248001 | Ready-to-use | 1/25 | TRIS/EDTA | Normal cervix |
| p53 | DAKO, Santa Clara, CA, USA | DO-7 | GA61661-2 | Ready to use | 1/2000 | TRIS/EDTA | Normal cervix |
| αvβ6 | CAIX | CD44v6 | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Median | Min | Max | TBR | Median | Min | Max | TBR | Median | Min | Max | TBR | |
| Healthy (n = 15) | 9 | 2 | 31 | - | 2 | 0 | 19 | - | 240 | 173 | 283 | - |
| dVIN (n = 10) | 59 b | 20 | 216 | 6.6 | 7 b | 1 | 65 | 3.5 | 248 | 123 | 275 | 1.0 |
| HPV− VSCC (n = 16) | 118 | 0 | 231 | 13.1 | 9 a | 1 | 77 | 4.5 | 196 | 96 | 271 | 0.8 |
| HSIL (n = 15) | 42 b | 3 | 145 | 4.7 | 5 | 1 | 92 | 2.5 | 223 | 119 | 279 | 0.9 |
| HPV + VSCC (n = 13) | 93 | 7 | 213 | 10.3 | 6 | 1 | 102 | 3.0 | 118 a | 51 | 267 | 0.5 |
| EGFR | EpCAM | FRα | ||||||||||
| Median | Min | Max | TBR | Median | Min | Max | TBR | Median | Min | Max | TBR | |
| Healthy (n = 15) | 61 | 2 | 200 | - | 2 | 0 | 12 | - | 3 | 1 | 43 | - |
| dVIN (n = 10) | 40 | 2 | 138 | 0.7 | 5 | 0 | 11 | 2.5 | 1 | 1 | 3 | 0.3 |
| HPV− VSCC (n = 16) | 129 | 4 | 253 | 2.2 | 3 | 0 | 17 | 1.5 | 4 a | 2 | 7 | 1.3 |
| HSIL (n = 15) | 10 | 0 | 213 | 0.2 | 2 | 0 | 17 | 1.0 | 2 | 0 | 28 | 0.7 |
| HPV + VSCC (n = 13) | 25 | 5 | 106 | 0.4 | 7 a | 0 | 91 | 3.5 | 4 | 2 | 60 | 0.3 |
| MRP1 | MUC1 | uPAR | ||||||||||
| Median | Min | Max | TBR | Median | Min | Max | TBR | Median | Min | Max | TBR | |
| Healthy (n = 15) | 4 | 0 | 29 | - | 11 | 1 | 81 | - | 6 | 1 | 22 | - |
| dVIN (n = 10) | 8 | 0 | 24 | 2.0 | 63 | 20 | 206 | 5.7 | 12 b | 2 | 35 | 2.0 |
| HPV− VSCC (n = 16) | 13 c | 0 | 42 | 0.3 | 34 a | 3 | 184 | 3.1 | 37 c | 5 | 91 | 6.2 |
| HSIL (n = 15) | 2 | 0 | 22 | 0.5 | 40 | 3 | 201 | 3.6 | 6 c | 0 | 73 | 1.0 |
| HPV + VSCC (n = 13) | 2 | 0 | 17 | 0.5 | 62 | 14 | 176 | 5.6 | 19 a | 1 | 125 | 3.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huisman, B.W.; Cankat, M.; Bosse, T.; Vahrmeijer, A.L.; Rissmann, R.; Burggraaf, J.; Sier, C.F.M.; van Poelgeest, M.I.E. Integrin αvβ6 as a Target for Tumor-Specific Imaging of Vulvar Squamous Cell Carcinoma and Adjacent Premalignant Lesions. Cancers 2021, 13, 6006. https://doi.org/10.3390/cancers13236006
Huisman BW, Cankat M, Bosse T, Vahrmeijer AL, Rissmann R, Burggraaf J, Sier CFM, van Poelgeest MIE. Integrin αvβ6 as a Target for Tumor-Specific Imaging of Vulvar Squamous Cell Carcinoma and Adjacent Premalignant Lesions. Cancers. 2021; 13(23):6006. https://doi.org/10.3390/cancers13236006
Chicago/Turabian StyleHuisman, Bertine W., Merve Cankat, Tjalling Bosse, Alexander L. Vahrmeijer, Robert Rissmann, Jacobus Burggraaf, Cornelis F. M. Sier, and Mariette I. E. van Poelgeest. 2021. "Integrin αvβ6 as a Target for Tumor-Specific Imaging of Vulvar Squamous Cell Carcinoma and Adjacent Premalignant Lesions" Cancers 13, no. 23: 6006. https://doi.org/10.3390/cancers13236006
APA StyleHuisman, B. W., Cankat, M., Bosse, T., Vahrmeijer, A. L., Rissmann, R., Burggraaf, J., Sier, C. F. M., & van Poelgeest, M. I. E. (2021). Integrin αvβ6 as a Target for Tumor-Specific Imaging of Vulvar Squamous Cell Carcinoma and Adjacent Premalignant Lesions. Cancers, 13(23), 6006. https://doi.org/10.3390/cancers13236006








