The Risk of Psychological Stress on Cancer Recurrence: A Systematic Review
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Search Strategy
2.2. Selection Criteria
2.3. Data Extraction and Quality Assessment
2.4. Review Process and Meta Analysis
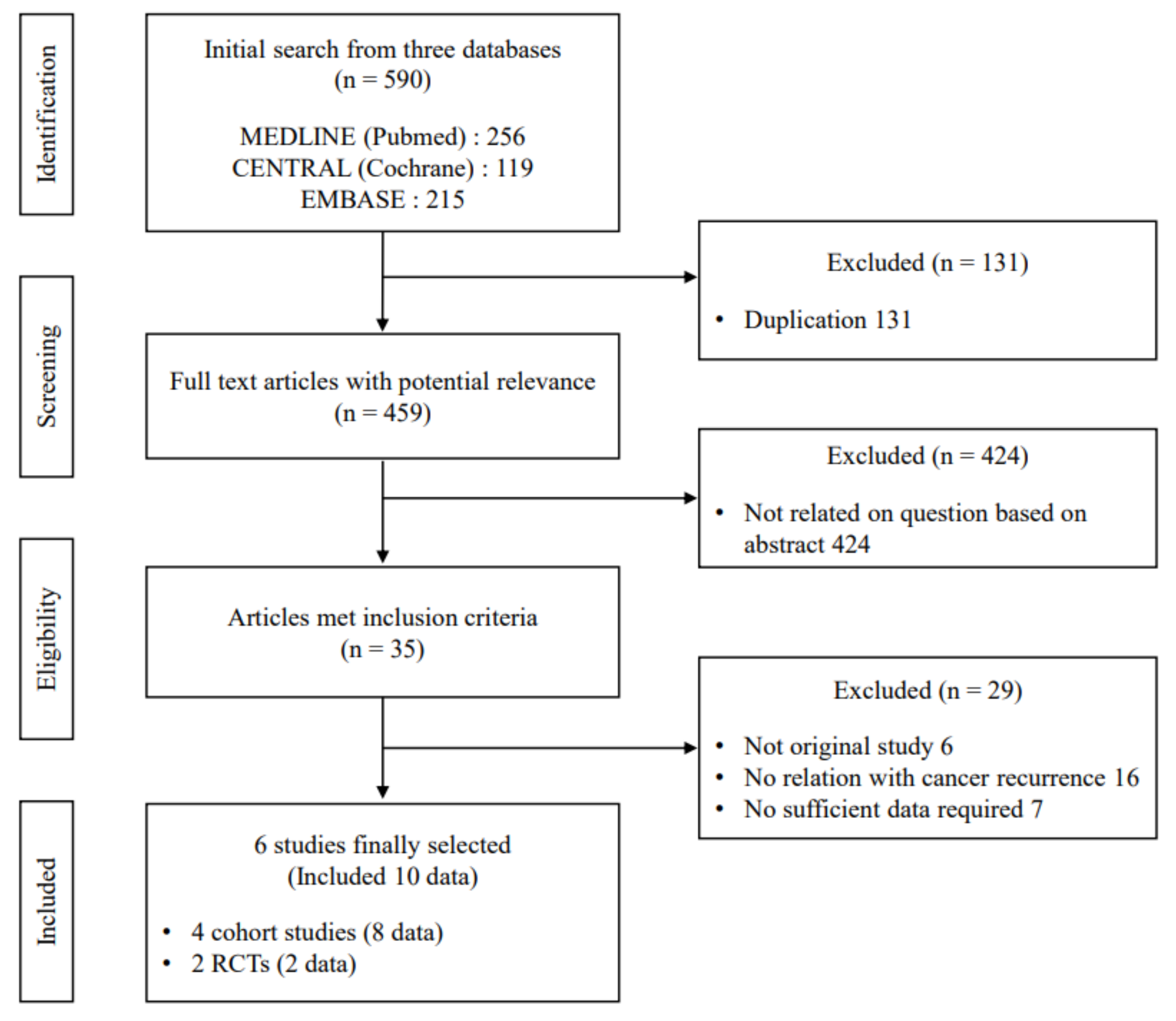
3. Results
3.1. Characteristics of the Included Studies
3.2. Features of Psychological Stress and Interventions in Cohort and RCT Studies
3.3. Psychological Stress and Risk of Tumor Recurrence from 4 Cohort Studies
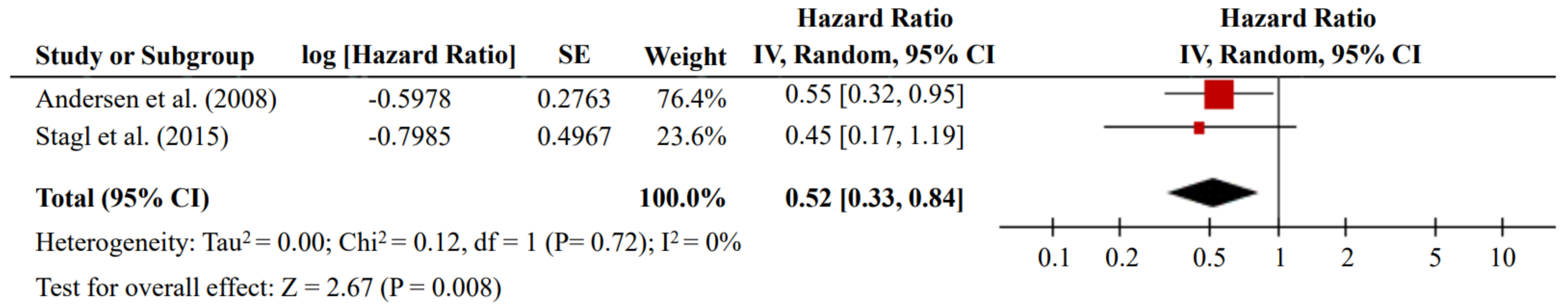
3.4. Psychological Stress and Risk of Tumor Recurrence from RCTs Using Adjusted Data
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA A Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Qian, C.-N.; Mei, Y.; Zhang, J. Cancer metastasis: Issues and challenges. Chin. J. Cancer 2017, 36, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Dillekås, H.; Rogers, M.S.; Straume, O. Are 90% of deaths from cancer caused by metastases? Cancer Med. 2019, 8, 5574–5576. [Google Scholar] [CrossRef] [PubMed]
- Wang, L. Early Diagnosis of Breast Cancer. Sensors 2017, 17, 1572. [Google Scholar] [CrossRef] [PubMed]
- Lebel, S.; Ozakinci, G.; Humphris, G.; Mutsaers, B.; Thewes, B.; Prins, J.; Dinkel, A.; Butow, P. From normal response to clinical problem: Definition and clinical features of fear of cancer recurrence. Support. Care Cancer 2016, 24, 3265–3268. [Google Scholar] [CrossRef]
- Brewster, A.M.; Hortobagyi, G.N.; Broglio, K.R.; Kau, S.-W.; Santa-Maria, C.A.; Arun, B.; Buzdar, A.U.; Booser, D.J.; Valero, V.; Bondy, M.; et al. Residual Risk of Breast Cancer Recurrence 5 Years After Adjuvant Therapy. J. Natl. Cancer Inst. 2008, 100, 1179–1183. [Google Scholar] [CrossRef]
- Tjandra, J.J.; Chan, M.K.Y. Follow-Up After Curative Resection of Colorectal Cancer: A Meta-Analysis. Dis. Colon Rectum 2007, 50, 1783–1799. [Google Scholar] [CrossRef] [PubMed]
- Llovet, J.M.; Schwartz, M.; Mazzarerro, V. Resection and liver transplantation for hepatocellular carcinoma. Semin. Liver Dis. 2005, 25, 181–200. [Google Scholar] [CrossRef] [PubMed]
- Faisal, M.; Abu Bakar, M.; Sarwar, A.; Adeel, M.; Batool, F.; Malik, K.I.; Jamshed, A.; Hussain, R. Depth of invasion (DOI) as a predictor of cervical nodal metastasis and local recurrence in early stage squamous cell carcinoma of oral tongue (ESSCOT). PLoS ONE 2018, 13, e0202632. [Google Scholar] [CrossRef]
- Hoffmann, A.-C.; Warnecke-Eberz, U.; Luebke, T.; Prenzel, K.; Metzger, R.; Heitmann, M.; Neiss, S.; Vallbohmer, D.; Hoelscher, A.; Schneider, P. Survivin mRNA in peripheral blood is frequently detected and significantly decreased following resection of gastrointestinal cancers. J. Surg. Oncol. 2007, 95, 51–54. [Google Scholar] [CrossRef] [PubMed]
- Serrano, M.J.; Sanchez-Rovira, P.; Delgado-Rodriguez, M.; Gaforio, J.J. Detection of circulating tumor Cells in the context of treatment: Prognostic value in breast cancer patients. Cancer Biol. Ther. 2009, 8, 671–675. [Google Scholar] [CrossRef][Green Version]
- Abate, M.; Citro, M.; Caputo, M.; Pisanti, S.; Martinelli, R. Psychological Stress and Cancer: New Evidence of An Increasingly Strong Link. Transl. Med. UniSa 2020, 23, 53–57. [Google Scholar]
- Lutgendorf, S.K.; Sood, A.K.; Antoni, M.H. Host Factors and Cancer Progression: Biobehavioral Signaling Pathways and Interventions. J. Clin. Oncol. 2010, 28, 4094–4099. [Google Scholar] [CrossRef]
- Cole, S.W.; Nagaraja, A.; Lutgendorf, S.K.; Green, P.; Sood, A.K. Sympathetic nervous system regulation of the tumour microenvironment. Nat. Rev. Cancer 2015, 15, 563–572. [Google Scholar] [CrossRef]
- Phillips, K.M.; Antoni, M.H.; Lechner, S.C.; Blomberg, B.B.; Llabre, M.M.; Avisar, E.; Glück, S.; DerHagopian, R.; Carver, C.S. Stress Management Intervention Reduces Serum Cortisol and Increases Relaxation During Treatment for Nonmetastatic Breast Cancer. Psychosom. Med. 2008, 70, 1044–1049. [Google Scholar] [CrossRef]
- McGregor, B.A.; Antoni, M.H. Psychological intervention and health outcomes among women treated for breast cancer: A review of stress pathways and biological mediators. Brain Behav. Immun. 2009, 23, 159–166. [Google Scholar] [CrossRef] [PubMed]
- MD Anderson Cancer Center. Psychiatric Oncology Center. Available online: https://www.mdanderson.org/patients-family/diagnosis-treatment/care-centers-clinics/psychiatric-oncology-center.html (accessed on 18 October 2021).
- Kruk, J.; Aboul-Enein, B.; Bernstein, J.; Gronostaj, M. Psychological Stress and Cellular Aging in Cancer: A Meta-Analysis. Oxidative Med. Cell. Longev. 2019, 2019, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Chida, Y.; Hamer, M.; Wardle, J.; Steptoe, A. Do stress-related psychosocial factors contribute to cancer incidence and survival? Nat. Clin. Pract. Oncol. 2008, 5, 466–475. [Google Scholar] [CrossRef] [PubMed]
- Thornton, L.; Andersen, B.L.; Carson, I.W.E. Immune, endocrine, and behavioral precursors to breast cancer recurrence: A case-control analysis. Cancer Immunol. Immunother. 2008, 57, 1471–1481. [Google Scholar] [CrossRef]
- Beswick, S.; Affleck, P.; Elliott, F.; Gerry, E.; Boon, A.; Bale, L.; Nolan, C.; Barrett, J.H.; Bertram, C.; Marsden, J.; et al. Environmental risk factors for relapse of melanoma. Eur. J. Cancer 2008, 44, 1717–1725. [Google Scholar] [CrossRef][Green Version]
- Schraub, S.; Sancho-Garnier, H.; Velten, M. Should psychological events be considered cancer risk factors? Rev. D’épidémiol. Sant Publique 2009, 57, e7–e16. [Google Scholar] [CrossRef]
- Melhem-Bertrandt, A.; Conzen, S.D. The Relationship Between Psychosocial Stressors and Breast Cancer Biology. Curr. Breast Cancer Rep. 2010, 2, 130–137. [Google Scholar] [CrossRef]
- Wells, G.A.; Shea, B.; O’connell, D.; Peterson, J.; Welch, V.; Losos, M.; Tugwell, P. The Newcastle-Ottawa Scale (NOS) for Assessing the Quality of Nonrandomised Studies in Meta-Analyses. Available online: http://www.ohri.ca/programs/clinical_epidemiology/oxford.htm (accessed on 18 October 2021).
- Sterne, J.A.C.; Savović, J.; Page, M.; Elbers, R.G.; Blencowe, N.; Boutron, I.; Cates, C.; Cheng, H.-Y.; Corbett, M.S.; Eldridge, S.M.; et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019, 366, l4898. [Google Scholar] [CrossRef]
- DerSimonian, R.; Laird, N. Meta-analysis in clinical trials. Control. Clin. Trials 1986, 7, 177–188. [Google Scholar] [CrossRef]
- Higgins, J.P.T.; Thompson, S.G. Quantifying heterogeneity in a meta-analysis. Stat. Med. 2002, 21, 1539–1558. [Google Scholar] [CrossRef] [PubMed]
- Groenvold, M.; Petersen, M.A.; Idler, E.; Bjorner, J.B.; Fayers, P.; Mouridsen, H.T. Psychological distress and fatigue predicted recurrence and survival in primary breast cancer patients. Breast Cancer Res. Treat. 2007, 105, 209–219. [Google Scholar] [CrossRef] [PubMed]
- Saquib, N.; Pierce, J.P.; Saquib, J.; Flatt, S.W.; Natarajan, L.; Bardwell, W.A.; Patterson, R.E.; Stefanick, M.L.; Thomson, C.A.; Rock, C.L.; et al. Poor physical health predicts time to additional breast cancer events and mortality in breast cancer survivors. Psycho-Oncology 2011, 20, 252–259. [Google Scholar] [CrossRef] [PubMed]
- Olsen, M.H.; Bidstrup, P.E.; Frederiksen, K.; Rod, N.H.; Grønbæk, M.K.; Dalton, S.O.; Johansen, C. Loss of partner and breast cancer prognosis—A population-based study, Denmark, 1994–2010. Br. J. Cancer 2012, 106, 1560–1563. [Google Scholar] [CrossRef][Green Version]
- Liu, J.; Zong, G.; Zhang, C.; Li, C.; Chen, X.; Zhang, Y. Anxiety and serum catecholamines as predictors of survival and recurrence in hepatocellular carcinoma. Psycho-Oncology 2016, 26, 1347–1353. [Google Scholar] [CrossRef] [PubMed]
- Andersen, B.L.; Yang, H.-C.; Farrar, W.B.; Golden-Kreutz, D.M.; Emery, C.F.; Thornton, L.; Young, D.C.; Carson, I.W.E. Psychologic intervention improves survival for breast cancer patients. Cancer 2008, 113, 3450–3458. [Google Scholar] [CrossRef]
- Stagl, J.M.; Lechner, S.C.; Carver, C.; Bouchard, L.C.; Gudenkauf, L.M.; Jutagir, D.R.; Diaz, A.; Yu, Q.; Blomberg, B.B.; Ironson, G.; et al. A randomized controlled trial of cognitive-behavioral stress management in breast cancer: Survival and recurrence at 11-year follow-up. Breast Cancer Res. Treat. 2015, 154, 319–328. [Google Scholar] [CrossRef]
- Caron, J.; Liu, A. Factors Associated with Psychological Distress in the Canadian Population: A Comparison of Low-Income and Non Low-Income Sub-Groups. Community Ment. Health J. 2010, 47, 318–330. [Google Scholar] [CrossRef]
- Cohen, S.; Janicki-Deverts, D.; Miller, G.E. Psychological Stress and Disease. JAMA 2007, 298, 1685–1687. [Google Scholar] [CrossRef] [PubMed]
- Miller, G.E.; Chen, E.; Parker, K.J. Psychological stress in childhood and susceptibility to the chronic diseases of aging: Moving toward a model of behavioral and biological mechanisms. Psychol. Bull. 2011, 137, 959–997. [Google Scholar] [CrossRef]
- Singer, S.; Das-Munshi, J.; Brähler, E. Prevalence of mental health conditions in cancer patients in acute care—A meta-analysis. Ann. Oncol. 2010, 21, 925–930. [Google Scholar] [CrossRef] [PubMed]
- Steel, Z.; Marnane, C.; Iranpour, C.; Chey, T.; Jackson, J.W.; Patel, V.; Silove, D. The global prevalence of common mental disorders: A systematic review and meta-analysis 1980–2013. Int. J. Epidemiol. 2014, 43, 476–493. [Google Scholar] [CrossRef]
- Todd, B.L.; Moskowitz, M.C.; Ottati, A.; Feuerstein, M. Stressors, Stress Response, and Cancer Recurrence. Cancer Nurs. 2014, 37, 114–125. [Google Scholar] [CrossRef] [PubMed]
- Krohne, H. Stress and Coping Theories. Int. Encycl. Soc. Behav. Sci. 2001, 22, 15163–15170. [Google Scholar] [CrossRef]
- Clayton, P.J. Bereavement. In Encyclopedia of Stress, 2nd ed.; Elsevier inc.: Amsterdam, The Netherlands, 2007; pp. 317–323. ISBN 9780123739476. [Google Scholar]
- Cano, A.; O’Leary, K.D.; Heinz, W. Short-Term Consequences of Severe Marital Stressors. J. Soc. Pers. Relatsh. 2004, 21, 419–430. [Google Scholar] [CrossRef]
- Ledermann, T.; Bodenmann, G.; Rudaz, M.; Bradbury, T.N. Stress, Communication, and Marital Quality in Couples. Fam. Relat. 2010, 59, 195–206. [Google Scholar] [CrossRef]
- Satheesan, S.C.; Satyanarayana, V.A. Quality of marital relationship, partner violence, psychological distress, and resilience in women with primary infertility. Int. J. Community Med. Public Health 2018, 5, 734–739. [Google Scholar] [CrossRef]
- Fisher, R.; Pusztai, L.; Swanton, C. Cancer heterogeneity: Implications for targeted therapeutics. Br. J. Cancer 2013, 108, 479–485. [Google Scholar] [CrossRef] [PubMed]
- Matud, M. Gender differences in stress and coping styles. Pers. Individ. Differ. 2004, 37, 1401–1415. [Google Scholar] [CrossRef]
- Morrow, B. An overview of cohort study designs and their advantages and disadvantages. Int. J. Ther. Rehabil. 2010, 17, 518–523. [Google Scholar] [CrossRef]
- Lengacher, C.A.; Johnson-Mallard, V.; Post-White, J.; Moscoso, M.S.; Jacobsen, P.B.; Klein, T.W.; Widen, R.H.; Fitzgerald, S.G.; Shelton, M.M.; Barta, M.; et al. Randomized controlled trial of mindfulness-based stress reduction (MBSR) for survivors of breast cancer. Psycho-Oncology 2009, 18, 1261–1272. [Google Scholar] [CrossRef] [PubMed]
- Lopez, C.; Antoni, M.; Penedo, F.; Weiss, D.; Cruess, S.; Segotas, M.-C.; Helder, L.; Siegel, S.; Klimas, N.; Fletcher, M.A. A pilot study of cognitive behavioral stress management effects on stress, quality of life, and symptoms in persons with chronic fatigue syndrome. J. Psychosom. Res. 2011, 70, 328–334. [Google Scholar] [CrossRef] [PubMed]
- Cruess, D.G.; Antoni, M.H.; McGregor, B.A.; Kilbourn, K.M.; Boyers, A.E.; Alferi, S.M.; Carver, C.S.; Kumar, M. Cognitive-Behavioral Stress Management Reduces Serum Cortisol By Enhancing Benefit Finding Among Women Being Treated for Early Stage Breast Cancer. Psychosom. Med. 2000, 62, 304–308. [Google Scholar] [CrossRef] [PubMed]
- Antoni, M.H.; Lechner, S.; Diaz, A.; Vargas, S.; Holley, H.; Phillips, K.; McGregor, B.; Carver, C.; Blomberg, B. Cognitive behavioral stress management effects on psychosocial and physiological adaptation in women undergoing treatment for breast cancer. Brain Behav. Immun. 2009, 23, 580–591. [Google Scholar] [CrossRef]
- Hosaka, T.; Tokuda, Y.; Sugiyama, Y.; Hirai, K.; Okuyama, T. Effects of a structured psychiatric intervention on immune function of cancer patients. Tokai J. Exp. Clin. Med. 2000, 25, 183–188. [Google Scholar]
- Hofmann, S.G.; Asnaani, A.; Vonk, I.J.J.; Sawyer, A.T.; Fang, A. The Efficacy of Cognitive Behavioral Therapy: A Review of Meta-analyses. Cogn. Ther. Res. 2012, 36, 427–440. [Google Scholar] [CrossRef]
- Siegel, R.L.; Mph, K.D.M.; Jemal, A. Cancer statistics, 2019. CA A Cancer J. Clin. 2018, 69, 7–34. [Google Scholar] [CrossRef] [PubMed]
| Author (Year), Country | Cancer Type N. of Participants Mean Age (SD) | Stress Measurement Tool | Psychological Stress /Intervention | Timing of Stress Measurement | Median Observation/Follow-Up Period (Years) | Stress High vs. Low /Intervention vs. Control | N. of Recurrence | Adjusted HR or RR (95% CI) | Adjustments |
|---|---|---|---|---|---|---|---|---|---|
| Cohort studies | |||||||||
| Groenvold et al. (2007), Denmark [28] | Breast cancer 1,588 52.4 ± 10.3 | EORTC QLQ-C30 HADS HADS | Emotional health Anxiety Depression | 7 weeks after surgery | 12.9 | EORTC QLQ-C30 score: 83–100 vs. 0–75 HADS score: 8–21 vs. 0–7 HADS score: 8–21 vs. 0–7 | 761 (No information on each group) | RR = 0.80 (0.69–0.93) RR = 1.19 (1.02–1.39) RR = 1.19 (0.95–1.50) | age, adjuvant treatment regimen, grading of anaplasia, histopathological diagnosis, local radiotherapy, menopausal status, number of tumor positive axillary nodes, social class type of operation, tumor receptor status, tumor size |
| Saquib et al. (2011), USA [29] | Breast cancer 2,967 53.3 ± 8.9 | SF-36 CMHS | Mental health Hostility | At first clinic visit | 7.3 | SF-36 score: 90.6–100 vs. 0–63.3 CMHS score: 6–13 vs. 0 | 492 (No information on each group) | HR = 1.21 (0.85–1.72) HR = 1.24 (0.92–1.68) | age, anti-estrogen use, hot flashes, menopausal status, physical activity, race/ethnicity, clinical site, time between cancer diagnosis and study entry, randomization group, tumor type, tumor grade, tumor stage |
| Olsen et al. (2012), Denmark [30] | Breast cancer 21,213 (Alive 19,312 vs. Before 762, After 1,139) 59.3 ± 4.11 | - | Loss of partner | - | 7.7 | Loss of partner vs. partner alive | 2,779 (Alive 2,635 vs. Before diagnosis 59 After diagnosis 85) | HR = 0.82 (0.61–1.09) HR = 0.98 (0.73–1.18) | age, disposable income, comorbidity, highest-attained educational level, hormone receptor status, number of tumor-positive lymph-nodes, period of diagnosis, tumor size, tumor grade |
| Liu et al. (2016), China [31] | HCC 110 (High 62 vs. Low 48) 54.5, range 36-76 | HAMA | Anxiety | 3 months after surgery | 4.0 | HAMA score: 17–56 vs 0–16 | 44 (High 31 vs. Low 13) | HR = 1.04 (1.00–1.10) | No information |
| RCTs | |||||||||
| Andersen et al. (2008), USA [32] | Breast cancer 211 (Inter. 103 vs. Cont. 109) 55.5 ± 11.9/52.2 ± 10.8 | Structured group meeting (48 weeks) | 11 | 48 weeks of structured group meeting vs. Assessment only | 62 (Inter. 29 vs. Cont. 33) * | HR = 0.55 (0.32–0.96) | age, chemotherapy, hormonal therapies, lymph node status, hormone receptor status, histologic grade, histologic type, Karnofsky performance status, menopausal status, POMS score, radiotherapy, surgery type, tumor size | ||
| Stagl et al. (2015), USA [33] | Breast cancer 240 (Inter. 120 vs. Cont. 120) 49.7 ± 9.0/51.0 ± 9.1 | CBSM (10 weeks) | 11 | 10 weeks of CBSM vs. 1-day psychoeducation | 47 (Inter. 24 vs. Cont. 23) * | HR = 0.45 (0.17–1.18) | age, disease stage, Her2/neu status, hormonal therapies, tumor size | ||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Oh, H.-M.; Son, C.-G. The Risk of Psychological Stress on Cancer Recurrence: A Systematic Review. Cancers 2021, 13, 5816. https://doi.org/10.3390/cancers13225816
Oh H-M, Son C-G. The Risk of Psychological Stress on Cancer Recurrence: A Systematic Review. Cancers. 2021; 13(22):5816. https://doi.org/10.3390/cancers13225816
Chicago/Turabian StyleOh, Hyeon-Muk, and Chang-Gue Son. 2021. "The Risk of Psychological Stress on Cancer Recurrence: A Systematic Review" Cancers 13, no. 22: 5816. https://doi.org/10.3390/cancers13225816
APA StyleOh, H.-M., & Son, C.-G. (2021). The Risk of Psychological Stress on Cancer Recurrence: A Systematic Review. Cancers, 13(22), 5816. https://doi.org/10.3390/cancers13225816






