Effects of Aerobic Exercise Training on MyomiRs Expression in Cachectic and Non-Cachectic Cancer Mice
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling
2.2. Training Protocol
2.3. Evaluation of the Maximal Physical Capacity
2.4. Grip Strength Meter
2.5. Ambulation Test
2.6. Tissue Preparation
2.7. Skeletal Muscle Oxidative Enzyme Activity
2.8. Analysis of MicroRNA
2.9. Immunohistochemistry
2.10. Immunoblotting
2.11. Statistical Analysis
3. Results
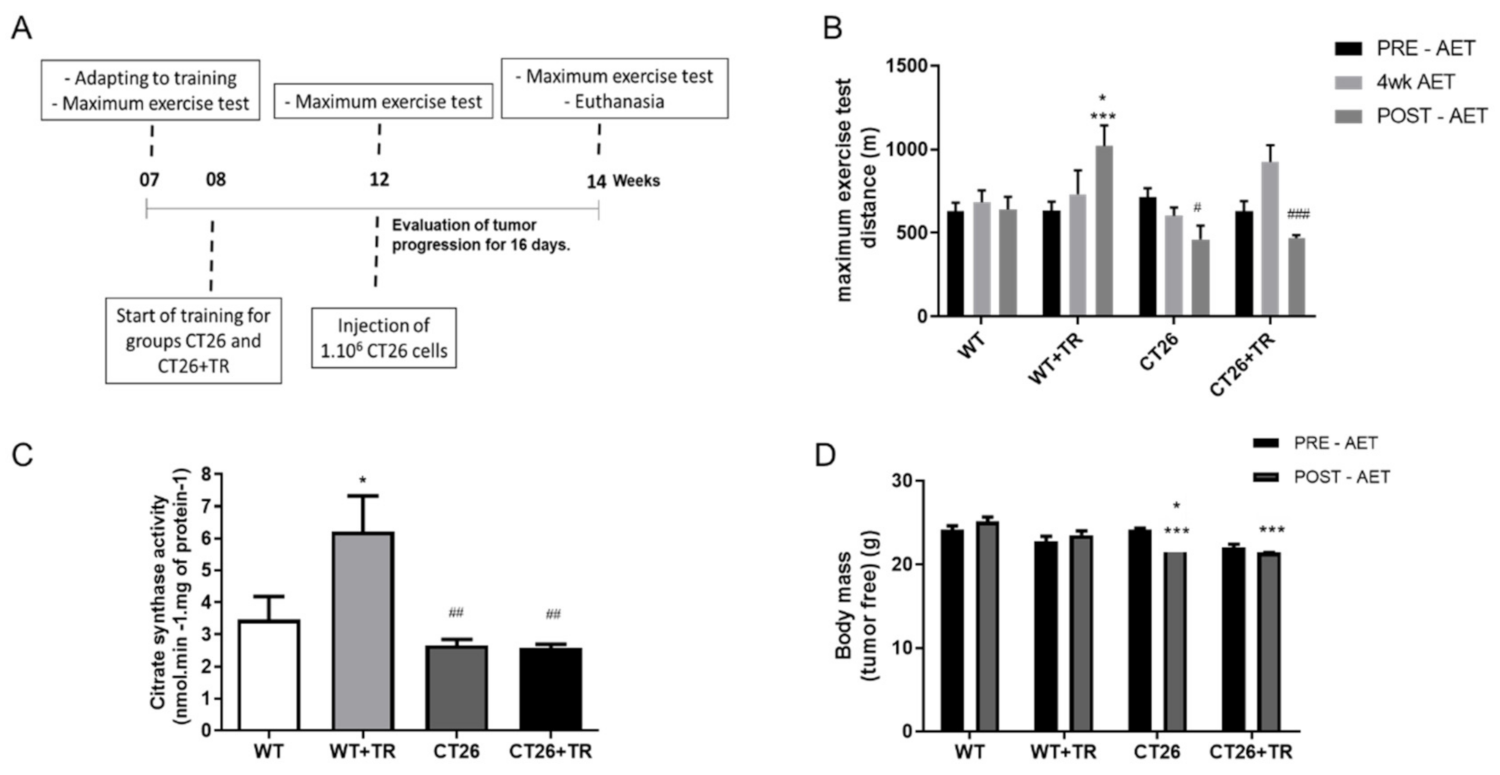
3.1. Effects of Aerobic Exercise Training on Functional Capacity in CT26 Cachectic Model
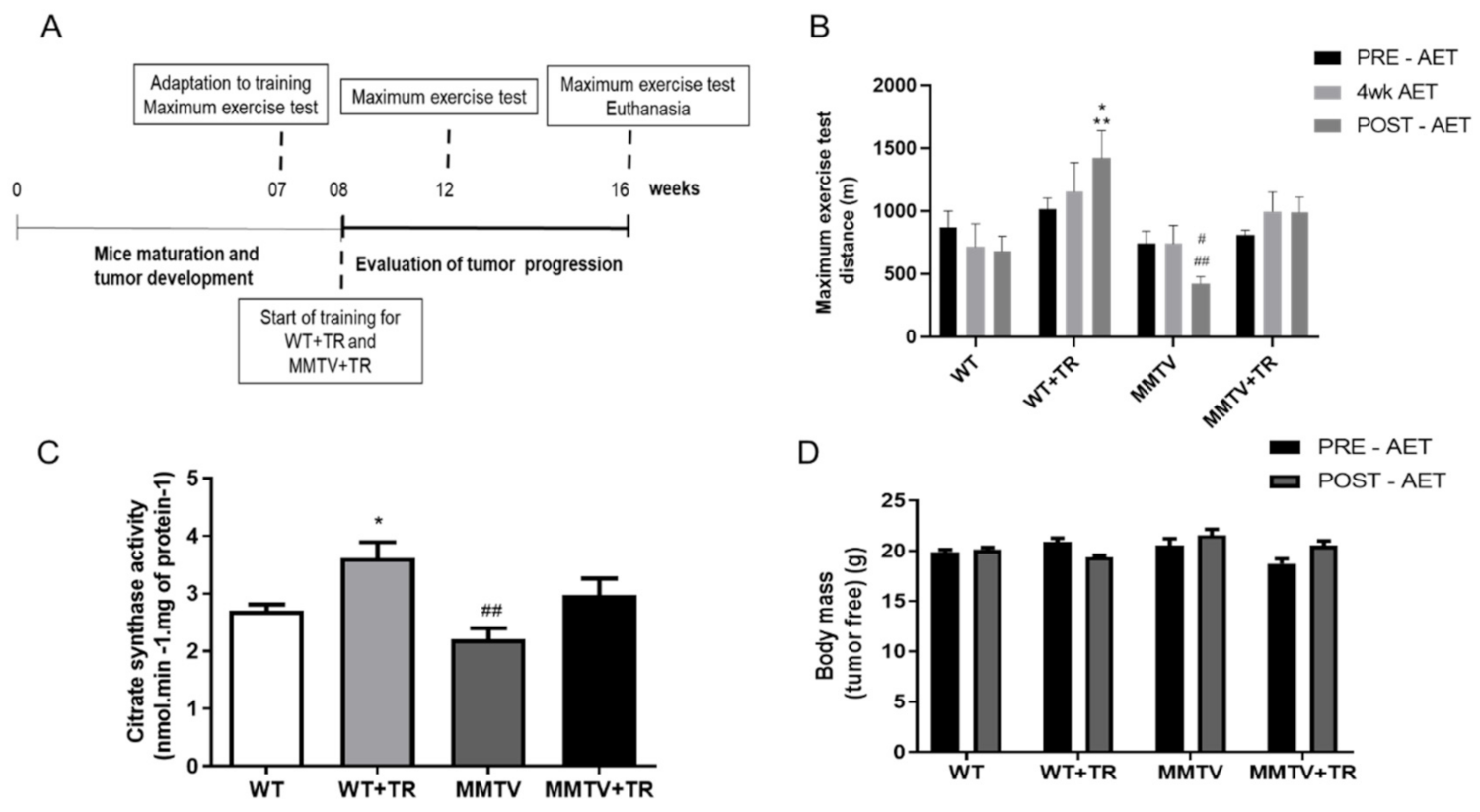
3.2. Effects of Aerobic Exercise Training on Functional Capacity in MMTV-PyMT Non-Cachectic Model
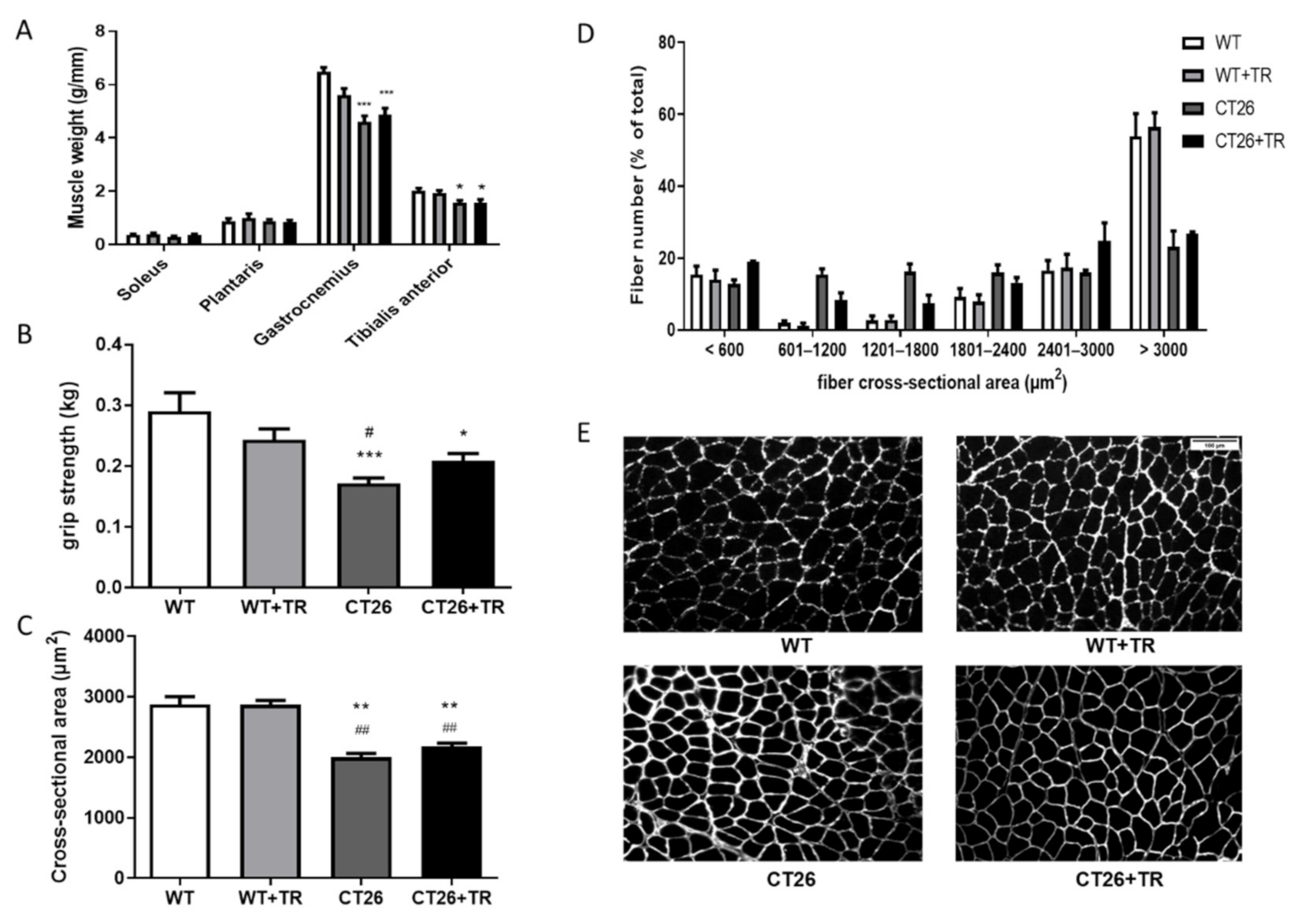
3.3. Effect of aerobic Exercise Training on Skeletal Muscle Mass, Cross-Sectional Area and Function in CT26 Cachectic and MMTV-PyMT Non-Cachectic Mice
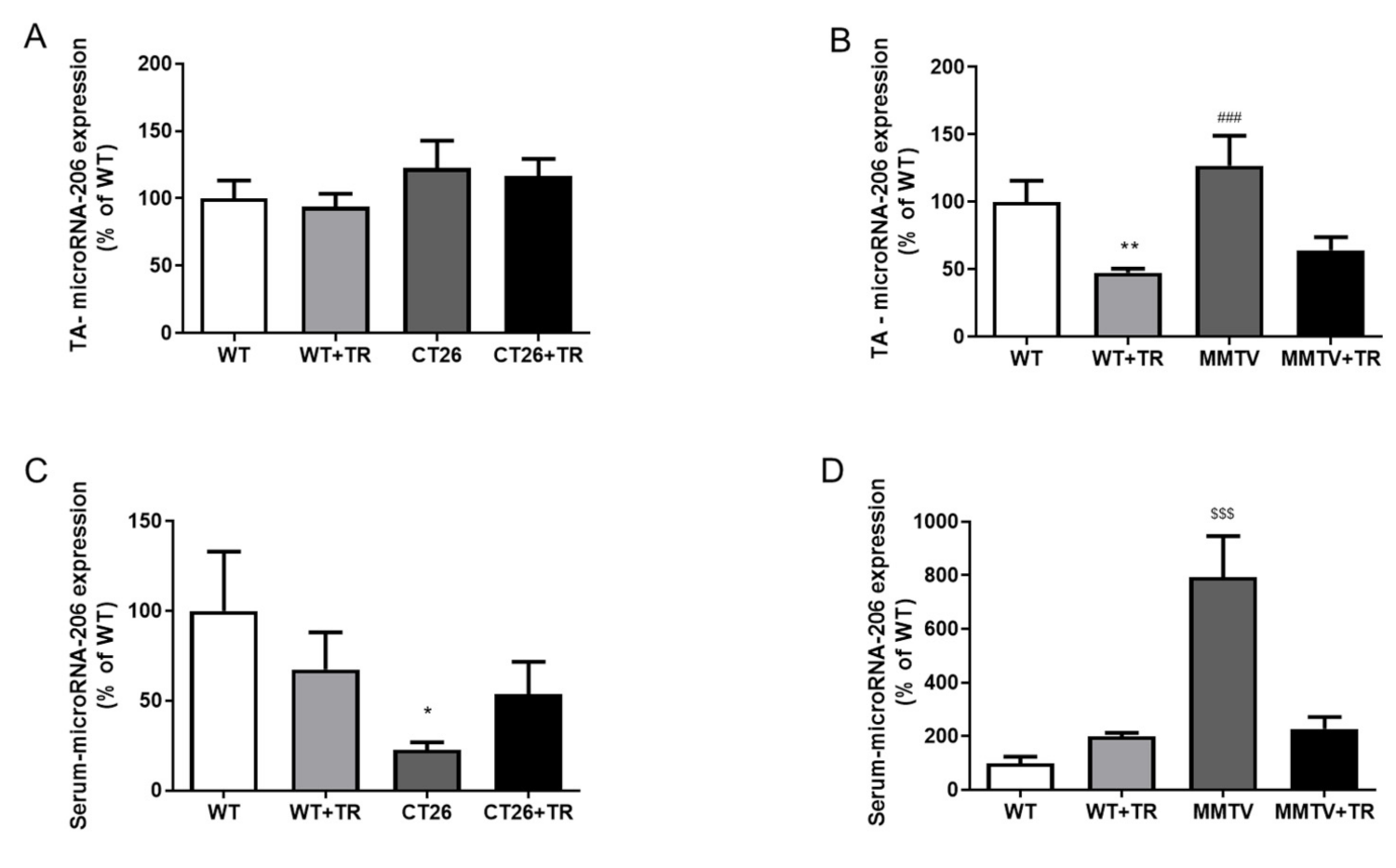
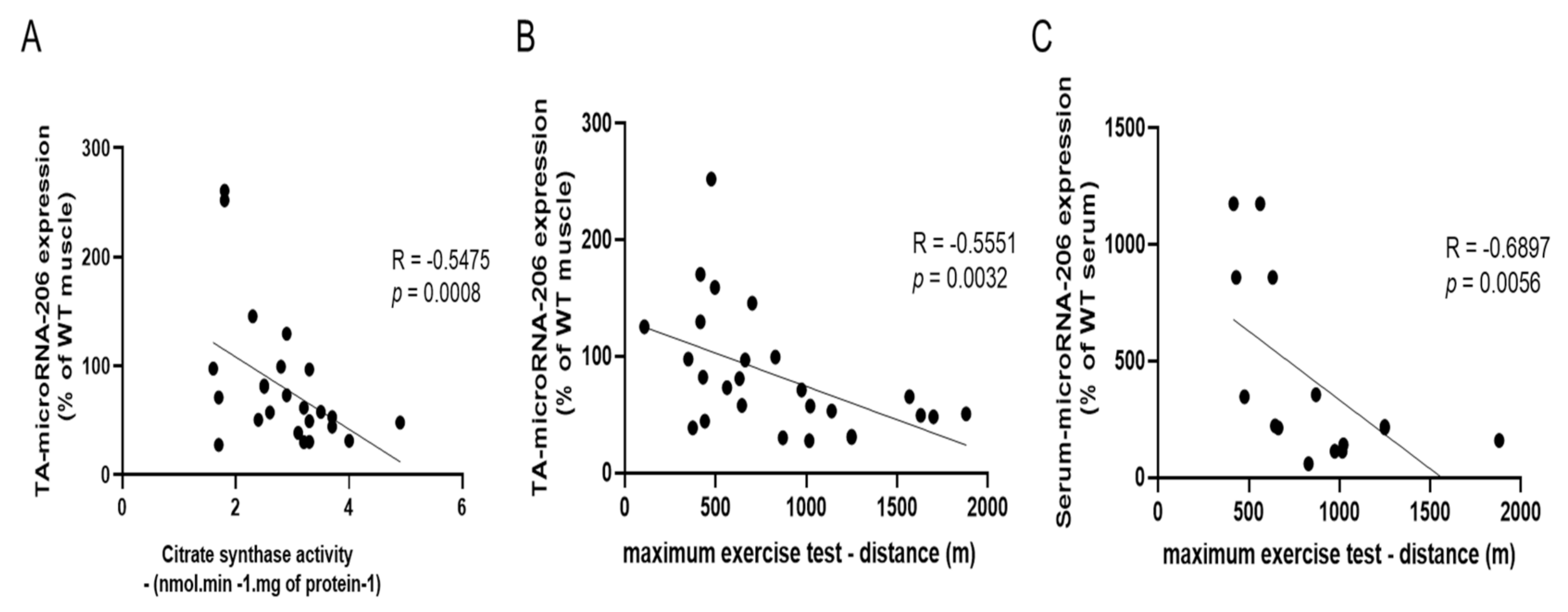
3.4. MyomiRs Expression Profile of CT26-Cachectic and MMTV-PyMT Non-Cachectic Mice
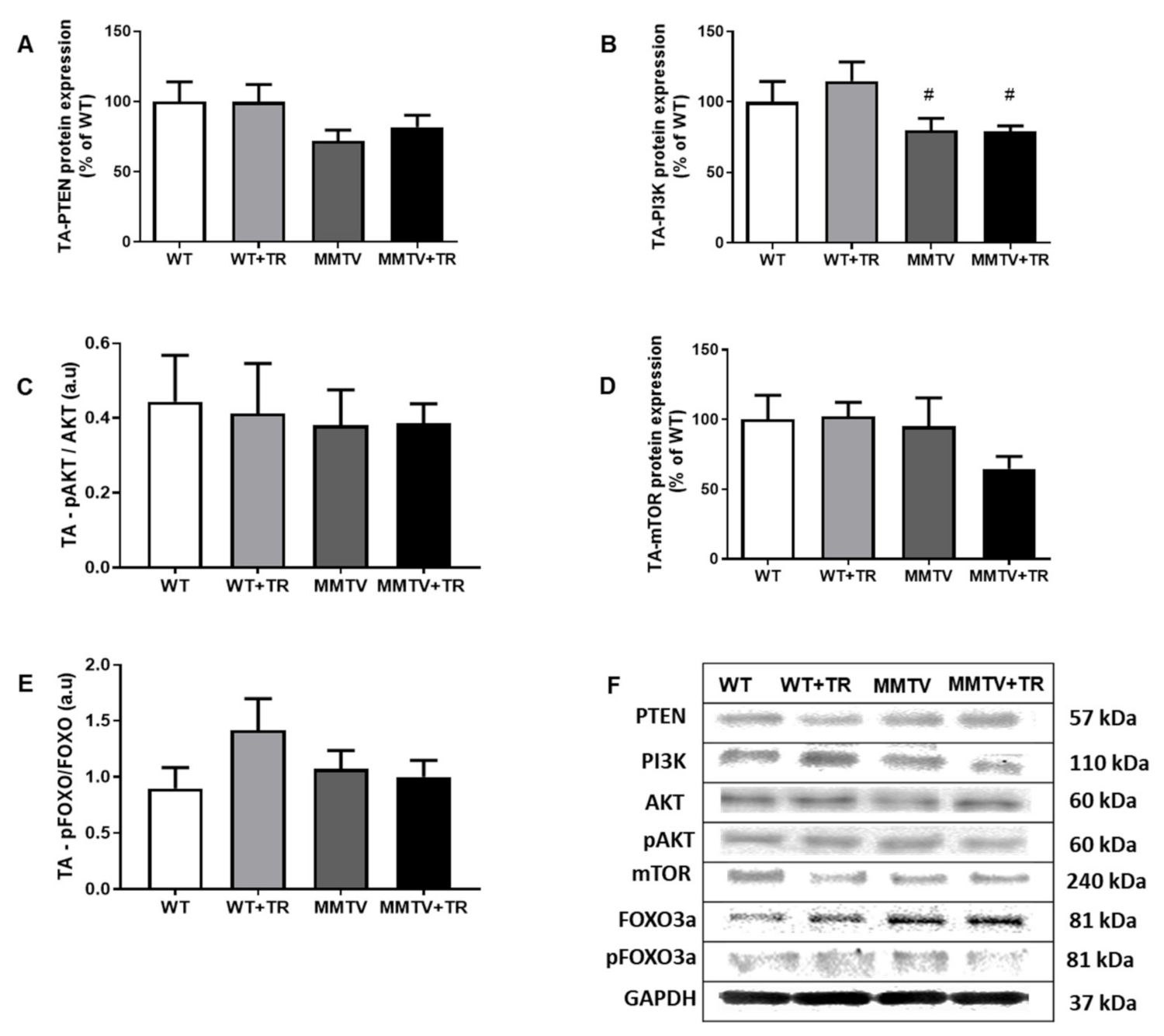
3.5. Effects of AET on MicroRNA-486/PTEN Axis in Skeletal Muscle of CT26-Cachectic and MMTV-PyMT Non-Cachectic Mice
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Conflicts of Interest
Abbreviations
References
- Fearon, K.; Strasser, F.; Anker, S.D.; Bosaeus, I.E.; Bruera, R.L.; Fainsinger, A.; Jatoi, C.; Loprinzi, N.; MacDonald, G.; Mantovani, M.; et al. Definition and classification of cancer cachexia: An international consensus. Lancet Oncol. 2011, 12, 489–495. [Google Scholar] [CrossRef]
- Martin, A.; Freyssenet, D. Phenotypic features of cancer cachexia-related loss of skeletal muscle mass and function: Lessons from human and animal studies. J. CachexSarcopenia Muscle 2021, 12, 252–273. [Google Scholar] [CrossRef]
- Groarke, J.D.; Cheng, S.; Jones, L.W.; Moslehi, J. Cancer cachexia: Getting to the heart of the matter. Eur. Hear. J. 2013, 40, e17–e19. [Google Scholar] [CrossRef] [PubMed]
- Sandri, M. Protein breakdown in cancer cachexia. Semin. Cell Dev. Biol. 2016, 54, 11–19. [Google Scholar] [CrossRef]
- Ambros, V. microRNAs: Tiny Regulators with Great Potential. Cell 2001, 107, 823–826. [Google Scholar] [CrossRef] [Green Version]
- Horak, M.; Novak, J.; Bienertova-Vasku, J. Muscle-specific microRNAs in skeletal muscle development. Dev. Biol. 2016, 410, 1–13. [Google Scholar] [CrossRef]
- Diniz, G.P.; Wang, D. Regulation of Skeletal Muscle by microRNAs. Compr. Physiol. 2016, 6, 1279–1294. [Google Scholar] [CrossRef]
- Lee, D.E.; Brown, J.L.; Rosa-Caldwell, M.E.; Blackwell, T.A.; Perry, R.A., Jr.; Brown, L.A.; Khatri, B.; Seo, D.; Bottje, W.G.; Washington, T.A.; et al. Cancer cachexia-induced muscle atrophy: Evidence for alterations in microRNAs important for muscle size. Physiol. Genom. 2017, 49, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Narasimhan, A.; Ghosh, S.; Stretch, C.; Greiner, R.; Bathe, O.F.O.F.; Baracos, V.; Damaraju, S. Small RNAome profiling from human skeletal muscle: Novel miRNAs and their targets associated with cancer cachexia. J. Cachex-Sarcopenia Muscle 2017, 8, 405–416. [Google Scholar] [CrossRef] [Green Version]
- Siracusa, J.; Koulmann, N.; Banzet, S. Circulating myomiRs: A new class of biomarkers to monitor skeletal muscle in physiology and medicine. J. Cachex-Sarcopenia Muscle 2017, 9, 20–27. [Google Scholar] [CrossRef]
- Kondo, N.; Toyama, T.; Sugiura, H.; Fujii, Y.; Yamashita, H. miR-206 Expression Is Down-regulated in Estrogen Receptor α–Positive Human Breast Cancer. Cancer Res. 2008, 68, 5004–5008. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Souza, R.W.A.; Fernandez, G.J.; Cunha, J.P.Q.; Piedade, W.P.; Soares, L.C.; Souza, P.A.T.; de Campos, D.H.S.; Okoshi, K.; Cicogna, A.C.; Dal-Pai-Silva, M.; et al. Regulation of cardiac microRNAs induced by aerobic exercise training during heart failure. Am. J. Physiol. Circ. Physiol. 2015, 309, H1629–H1641. [Google Scholar] [CrossRef] [PubMed]
- Cunha, T.F.; Moreira, J.B.N.; Paixão, N.A.; Campos, J.C.; Monteiro, A.W.A.; Bacurau, A.V.N.; Bueno, C.R.; Ferreira, J.C.B.; Brum, P.C. Aerobic exercise training upregulates skeletal muscle calpain and ubiquitin-proteasome systems in healthy mice. J. Appl. Physiol. 2012, 112, 1839–1846. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Antunes-Correa, L.M.; Trevizan, P.F.; Bacurau, A.V.; Ferreira-Santos, L.; Gomes, J.L.; Urias, U.; Oliveira, P.A.; Alves, M.J.N.; de Almeida, D.R.; Brum, P.C.; et al. Effects of aerobic and inspiratory training on skeletal muscle microRNA-1 and downstream-associated pathways in patients with heart failure. J. Cachex-Sarcopenia Muscle 2020, 11, 89–102. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gomes, J.L.P.; Fernandes, T.; Soci, U.P.R.; Silveira, A.C.; Barretti, D.L.M.; Negrão, C.E.; Oliveira, E.M. Obesity Downregulates MicroRNA-126 Inducing Capillary Rarefaction in Skeletal Muscle: Effects of Aerobic Exercise Training. Oxidative Med. Cell. Longev. 2017, 2017, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Fernandes, T.; Baraúna, V.G.; Negrão, C.E.; Phillips, M.I.; Oliveira, E.M. Aerobic exercise training promotes physiological cardiac remodeling involving a set of microRNAs. Am. J. Physiol. Circ. Physiol. 2015, 309, H543–H552. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, J.; Lanza, D.G.; Guest, I.; Uk-Lim, C.; Glinskii, A.; Glinsky, G.; Sell, S. Characterization of mammary cancer stem cells in the MMTV-PyMT mouse model. Tumor Biol. 2012, 33, 1983–1996. [Google Scholar] [CrossRef] [PubMed]
- Ferreira, J.C.B.; Rolim, N.P.L.; Bartholomeu, J.B.; Gobatto, C.; Kokubun, E.; Brum, P.C. Maximal lactate steady state in running mice: Effect of exercise training. Clin. Exp. Pharmacol. Physiol. 2007, 34, 760–765. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]
- Zammit, V.A.; Newsholme, E.A. The maximum activities of hexokinase, phosphorylase, phosphofructokinase, glycerol phosphate dehydrogenases, lactate dehydrogenase, octopine dehydrogenase, phosphoenolpyruvate carboxykinase, nucleoside diphosphatekinase, glutamate-oxaloacetate transaminase and arginine kinase in relation to carbohydrate utilization in muscles from marine invertebrates. Biochem. J. 1976, 160, 447–462. [Google Scholar] [CrossRef] [Green Version]
- Fernandes, T.; Magalhães, F.C.; Roque, F.R.; Phillips, M.I.; Oliveira, E.M. Exercise Training Prevents the Microvascular Rarefaction in Hypertension Balancing Angiogenic and Apoptotic Factors. Hypertension 2012, 59, 513–520. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Small, E.M.; O’Rourke, J.R.; Moresi, V.; Sutherland, L.B.; McAnally, J.; Gerard, R.D.; Richardson, J.A.; Olson, E.N. Regulation of PI3-kinase/Akt signaling by muscle-enriched microRNA-486. Proc. Natl. Acad. Sci. USA 2010, 107, 4218–4223. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alexander, M.S.; Casar, J.C.; Motohashi, N.; Vieira, N.M.; Eisenberg, I.; Marshall, J.L.; Gasperini, M.J.; Lek, A.; Myers, J.A.; Estrella, E.A.; et al. MicroRNA-486–de-pendent modulation of DOCK3/PTEN/AKT signaling pathways improves muscular dystrophy-associated symptoms. J. Clin. Investig. 2014, 124, 2651–2667. [Google Scholar] [CrossRef] [PubMed]
- Chen, D.; Goswami, C.P.; Burnett, R.M.; Anjanappa, M.; Bhat-Nakshatri, P.; Muller, W.; Nakshatri, H. Cancer Affects microRNA Expression, Release, and Function in Cardiac and Skeletal Muscle. Cancer Res. 2014, 74, 4270–4281. [Google Scholar] [CrossRef] [Green Version]
- McCarthy, J.J. MicroRNA-206: The skeletal muscle-specific myomiR. Biochim. Biophys. Acta Bioenerg. 2008, 1779, 682–691. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xing, H.; Guo, S.; Zhang, Y.; Zheng, Z.; Wang, H. Upregulation of microRNA-206 enhances lipopolysaccharide-induced inflammation and release of amyloid-β by targeting insulin-like growth factor 1 in microglia. Mol. Med. Rep. 2016, 14, 1357–1364. [Google Scholar] [CrossRef] [Green Version]
- Wu, H.-Y.; Wang, X.-H.; Liu, K.; Zhang, J.-L. LncRNA MALAT1 regulates trophoblast cells migration and invasion via miR-206/IGF-1 axis. Cell Cycle 2020, 19, 39–52. [Google Scholar] [CrossRef] [PubMed]
- Hu, J.; Kong, M.; Ye, Y.; Hong, S.; Cheng, L.; Jiang, L. Serum miR-206 and other muscle-specific microRNAs as non-invasive biomarkers for Duchenne muscular dystrophy. J. Neurochem. 2014, 129, 877–883. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.-F.; Mandel, E.M.; Thomson, J.M.; Wu, Q.E.; Callis, T.; Hammond, S.M.; Conlon, F.L.; Wang, D.-Z. The Role of MicroRNA-1 and MicroRNA-133 in Skeletal Muscle Proliferation and Differentiation. Nat. Genet. 2005, 38, 228–233. [Google Scholar] [CrossRef]
- Chen, J.-F.; Tao, Y.; Li, J.; Deng, Z.; Yan, Z.; Xiao, X.; Wang, D.-Z. microRNA-1 and microRNA-206 regulate skeletal muscle satellite cell proliferation and differentiation by repressing Pax7. J. Cell Biol. 2010, 190, 867–879. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abreu, P.; Kowaltowski, A.J. Satellite cell self-renewal in endurance exercise is mediated by inhibition of mitochondrial oxygen consumption. J. Cachex- Sarcopenia Muscle 2020, 11, 1661–1676. [Google Scholar] [CrossRef] [PubMed]
- Kodama, S.; Saito, K.; Tanaka, S.; Maki, M.; Yachi, Y.; Asumi, M.; Sugawara, A.; Totsuka, K.; Shimano, H.; Ohashi, Y.; et al. Cardiorespiratory Fitness as a Quantitative Predictor of All-Cause Mortality and Cardiovascular Events in Healthy Men and Women. JAMA 2009, 301, 2024–2035. [Google Scholar] [CrossRef] [Green Version]
- Neil-Sztramko, S.E.; Kirkham, A.; Hung, S.H.; Niksirat, N.; Nishikawa, K.; Campbell, K.L. Aerobic capacity and upper limb strength are reduced in women diagnosed with breast cancer: A systematic review. J. Physiother. 2014, 60, 189–200. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alves, C.; da Cunha, T.F.; da Paixão, N.A.; Brum, P.C. Aerobic exercise training as therapy for cardiac and cancer cachexia. Life Sci. 2015, 125, 9–14. [Google Scholar] [CrossRef]
- Toiyama, Y.; Okugawa, Y.; Fleshman, J.; Boland, C.R.; Goel, A. MicroRNAs as potential liquid biopsy biomarkers in colorectal cancer: A systematic review. Biochim. et Biophys. Acta (BBA)-Bioenerg. 2018, 1870, 274–282. [Google Scholar] [CrossRef] [PubMed]
- Brase, J.C.; Wuttig, D.; Kuner, R.; Sültmann, H. Serum microRNAs as non-invasive biomarkers for cancer. Mol. Cancer 2010, 9, 306. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, Y.; Zhang, D.; Zhang, C.; Sun, Y. The Diagnostic and Prognostic Value of Serum lncRNA NEAT1 in Colorectal Cancer. Cancer Manag. Res. 2020, ume 12, 10985–10992. [Google Scholar] [CrossRef]
- Yan, B.; Zhao, L.; Guo, J.-T.; Zhao, J.-L. miR-206 regulates the growth of the teleost tilapia (Oreochromis niloticus) through the modulation of IGF-1 gene expression. J. Exp. Biol. 2013, 216, 1265–1269. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Y.J.; Xu, F.; Zhang, Y.J.; Li, H.B.; Han, J.C.; Li, L. MiR-206 inhibits non small cell lung cancer cell proliferation and inva-sion by targeting SOX9. Int. J. Clin. Exp. Med. 2015, 8, 9107–9113. [Google Scholar] [PubMed]
- Zhou, Y.; Wang, M.; Tong, Y.; Liu, X.; Zhang, L.; Dong, D.; Shao, J.; Zhou, Y. miR-206 Promotes Cancer Progression by Targeting Full-Length Neurokinin-1 Receptor in Breast Cancer. Technol. Cancer Res. Treat. 2019, 18. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, J.; Tian, Y.; Li, J.; Lu, B.; Sun, M.; Zou, Y.; Kong, R.; Luo, Y.; Shi, Y.; Wang, K.; et al. miR-206 is down-regulated in breast cancer and inhibits cell proliferation through the up-regulation of cyclinD2. Biochem. Biophys. Res. Commun. 2013, 433, 207–212. [Google Scholar] [CrossRef] [PubMed]
- Calzia, D.; Ottaggio, L.; Cora, A.; Chiappori, G.; Cuccarolo, P.; Cappelli, E.; Izzotti, A.; Tavella, S.; Degan, P. Characterization of C2C12 cells in simulated microgravity: Possible use for myoblast regeneration. J. Cell Physiol. 2020, 235, 3508–3518. [Google Scholar] [CrossRef] [PubMed]




Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gomes, J.L.P.; Tobias, G.C.; Fernandes, T.; Silveira, A.C.; Negrão, C.E.; Chammas, R.; Brum, P.C.; Oliveira, E.M. Effects of Aerobic Exercise Training on MyomiRs Expression in Cachectic and Non-Cachectic Cancer Mice. Cancers 2021, 13, 5728. https://doi.org/10.3390/cancers13225728
Gomes JLP, Tobias GC, Fernandes T, Silveira AC, Negrão CE, Chammas R, Brum PC, Oliveira EM. Effects of Aerobic Exercise Training on MyomiRs Expression in Cachectic and Non-Cachectic Cancer Mice. Cancers. 2021; 13(22):5728. https://doi.org/10.3390/cancers13225728
Chicago/Turabian StyleGomes, João Lucas Penteado, Gabriel Cardial Tobias, Tiago Fernandes, André Casanova Silveira, Carlos Eduardo Negrão, Roger Chammas, Patrícia Chakur Brum, and Edilamar Menezes Oliveira. 2021. "Effects of Aerobic Exercise Training on MyomiRs Expression in Cachectic and Non-Cachectic Cancer Mice" Cancers 13, no. 22: 5728. https://doi.org/10.3390/cancers13225728
APA StyleGomes, J. L. P., Tobias, G. C., Fernandes, T., Silveira, A. C., Negrão, C. E., Chammas, R., Brum, P. C., & Oliveira, E. M. (2021). Effects of Aerobic Exercise Training on MyomiRs Expression in Cachectic and Non-Cachectic Cancer Mice. Cancers, 13(22), 5728. https://doi.org/10.3390/cancers13225728








