Skin Toxicity as Predictor of Survival in Refractory Patients with RAS Wild-Type Metastatic Colorectal Cancer Treated with Cetuximab and Avelumab (CAVE) as Rechallenge Strategy
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Design and Patient Population
2.2. Patient Monitoring and Response Assessment
2.3. qPCR Analysis of Plasma Samples
2.4. Statistical Analysis
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Santini, D.; Vincenzi, B.; Addeo, R.; Garufi, C.; Masi, G.; Scartozzi, M.; Mancuso, A.; Frezza, A.M.; Venditti, O.; Imperatori, M.; et al. Cetuximab rechallenge in metastatic colorectal cancer patients: How to come away from acquired resistance? Ann. Oncol. 2017, 23, 2313–2318, Erratum in 2017, 28, 2906. [Google Scholar] [CrossRef]
- Cremolini, C.; Rossini, D.; Dell’Aquila, E.; Lonardi, S.; Conca, E.; Del Re, M.; Busico, A.; Pietrantonio, F.; Danesi, R.; Aprile, G.; et al. Rechallenge for Patients With RAS and BRAF Wild-Type Metastatic Colorectal Cancer With Acquired Resistance to First-line Cetuximab and Irinotecan: A Phase 2 Single-Arm Clinical Trial. JAMA Oncol. 2019, 5, 343–350. [Google Scholar] [CrossRef]
- Sunakawa, Y.; Nakamura, M.; Ishizaki, M.; Kataoka, M.; Satake, H.; Kitazono, M.; Yanagisawa, H.; Kawamoto, Y.; Kuramochi, H.; Ohori, H.; et al. RAS Mutations in Circulating Tumor DNA and Clinical Outcomes of Rechallenge Treatment With Anti-EGFR Antibodies in Patients With Metastatic Colorectal Cancer. JCO Precis. Oncol. 2020, 4, 898–911. [Google Scholar] [CrossRef]
- Masuishi, T.; Tsuji, A.; Kotaka, M.; Nakamura, M.; Kochi, M.; Takagane, A.; Shimada, K.; Denda, T.; Segawa, Y.; Tanioka, H.; et al. Phase 2 study of irinotecan plus cetuximab rechallenge as third-line treatment in KRAS wild-type metastatic colorectal cancer: JACCRO CC-08. Br. J. Cancer 2020, 123, 1490–1495. [Google Scholar] [CrossRef] [PubMed]
- Martinelli, E.; Martini, G.; Famiglietti, V.; Troiani, T.; Napolitano, S.; Pietrantonio, F.; Ciardiello, D.; Terminiello, M.; Borrelli, C.; Vitiello, P.P.; et al. Cetuximab Rechallenge Plus Avelumab in Pretreated Patients With RAS Wild-type Metastatic Colorectal Cancer: The Phase 2 Single-Arm Clinical CAVE Trial. JAMA Oncol. 2021, 7, 1529–1535. [Google Scholar] [CrossRef]
- Ciardiello, D.; Martini, G.; Famiglietti, V.; Napolitano, S.; De Falco, V.; Troiani, T.; Latiano, T.P.; Ros, J.; Elez Fernandez, E.; Vitiello, P.P.; et al. Biomarker-Guided Anti-Egfr Rechallenge Therapy in Metastatic Colorectal Cancer. Cancers 2021, 13, 1941. [Google Scholar] [CrossRef] [PubMed]
- Martinelli, E.; Ciardiello, D.; Martini, G.; Troiani, T.; Cardone, C.; Vitiello, P.P.; Normanno, N.; Rachiglio, A.M.; Maiello, E.; Latiano, T.; et al. Implementing anti-epidermal growth factor receptor (EGFR) therapy in metastatic colorectal cancer: Challenges and future perspectives. Ann. Oncol. 2020, 31, 30–40. [Google Scholar] [CrossRef]
- Goldberg, R.M.; Montagut, C.; Wainberg, Z.A.; Ronga, P.; Audhuy, F.; Taieb, J.; Stintzing, S.; Siena, S.; Santini, D. Optimising the use of cetuximab in the continuum of care for patients with metastatic colorectal cancer. ESMO Open 2018, 3, e000353. [Google Scholar] [CrossRef]
- Parseghian, C.M.; Loree, J.M.; Morris, V.K.; Liu, X.; Clifton, K.K.; Napolitano, S.; Henry, J.T.; Pereira, A.A.; Vilar, E.; Johnson, B. Anti-EGFR-resistant clones decay exponentially after progression: Implications for anti-EGFR re-challenge. Ann. Oncol. 2019, 30, 243–249. [Google Scholar] [CrossRef]
- Rossini, D.; Germani, M.M.; Pagani, F.; Pellino, A.; Dell’Aquila, E.; Bensi, M.; Liscia, N.; Moretto, R.; Boccaccino, A.; Prisciandaro, M.; et al. Retreatment With Anti-EGFR Antibodies in Metastatic Colorectal Cancer Patients: A Multi-institutional Analysis. Clin. Colorectal. Cancer 2020, 19, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Lacouture, M.E. Mechanisms of cutaneous toxicities to EGFR inhibitors. Nat. Rev. Cancer 2006, 6, 803–812. [Google Scholar] [CrossRef] [PubMed]
- Parmar, S.; Schumann, C.; Rüdiger, S.; Boeck, S.; Heinemann, V.; Kächele, V.; Seeringer, A.; Paul, T.; Seufferlein, T.; Stingl, J.C. Pharmacogenetic predictors for EGFR-inhibitor-associated skin toxicity. Pharm. J. 2013, 13, 181–188. [Google Scholar] [CrossRef][Green Version]
- Lacouture, M.E.; Sibaud, V.; Gerber, P.A.; Van den Hurk, C.; Fernández-Peñas, P.; Santini, D.; Jahn, F.; Jordan, K. Prevention and Management of Dermatological Toxicities Related to Anticancer Agents: ESMO Clinical Practice Guidelines†. Ann. Oncol. 2021, 32, 157–170. [Google Scholar] [CrossRef] [PubMed]
- Lupu, I.; Voiculescu, V.M.; Bacalbasa, N.; Prie, B.E.; Cojocaru, I.; Giurcaneanu, C. Cutaneous adverse reactions specific to epidermal growth factor receptor inhibitors. J. Med. Life 2015, 8, 57–61. [Google Scholar] [PubMed]
- Wacker, B.; Nagrani, T.; Weinberg, J.; Witt, K.; Clark, G.; Cagnoni, P.J. Correlation between Development of Rash and Efficacy in Patients Treated with the Epidermal Growth Factor Receptor Tyrosine Kinase Inhibitor Erlotinib in Two Large Phase III Studies. Clin. Cancer Res. 2007, 13, 3913. [Google Scholar] [CrossRef]
- Herbst, R.S.; Arquette, M.; Shin, D.M.; Dicke, K.; Vokes, E.E.; Azarnia, N.; Hong, W.K.; Kies, M.S. Phase II multicenter study of the epidermal growth factor receptor antibody cetuximab and cisplatin for recurrent and refractory squamous cell carcinoma of the head and neck. J. Clin. Oncol. 2005, 23, 5578–5587. [Google Scholar] [CrossRef]
- Orditura, M.; De Vita, F.; Galizia, G.; Lieto, E.; Vecchione, L.; Vitiello, F.; Martinelli, E.; Ciardiello, F. Correlation between efficacy and skin rash occurrence following treatment with the epidermal growth factor receptor inhibitor cetuximab: A single institution retrospective analysis. Oncol. Rep. 2009, 21, 1023–1028. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Cunningham, D.; Humblet, Y.; Siena, S.; Khayat, D.; Bleiberg, H.; Santoro, A.; Bets, D.; Mueser, M.; Harstrick, A.; Verslype, C.; et al. Cetuximab Monotherapy and Cetuximab plus Irinotecan in Irinotecan-Refractory Metastatic Colorectal Cancer. N. Engl. J. Med. 2004, 351, 337–345. [Google Scholar] [CrossRef]
- Lenz, H.J.; Van Cutsem, E.; Khambata-Ford, S.; Mayer, R.J.; Gold, P.; Stella, P.; Mirtsching, B.; Cohn, A.L.; Pippas, A.W.; Azarnia, N.; et al. Multicenter phase II and translational study of cetuximab in metastatic colorectal carcinoma refractory to irinotecan, oxaliplatin, and fluoropyrimidines. J. Clin. Oncol. 2006, 24, 4914–4921. [Google Scholar] [CrossRef] [PubMed]
- Holch, J.W.; Held, S.; Stintzing, S.; Fischer von Weikersthal, L.; Decker, T.; Kiani, A.; Kaiser, F.; Heintges, T.; Kahl, C.; Kullmann, F.; et al. Relation of cetuximab-induced skin toxicity and early tumor shrinkage in metastatic colorectal cancer patients: Results of the randomized phase 3 trial FIRE-3 (AIO KRK0306). Ann. Oncol. 2020, 31, 72–78. [Google Scholar] [CrossRef]
- Vitiello, P.P.; De Falco, V.; Giunta, E.F.; Ciardiello, D.; Cardone, C.; Vitale, P.; Zanaletti, N.; Borrelli, C.; Poliero, L.; Terminiello, M.; et al. Clinical Practice Use of Liquid Biopsy to Identify RAS/BRAF Mutations in Patients with Metastatic Colorectal Cancer (mCRC): A Single Institution Experience. Cancers 2019, 11, 1504. [Google Scholar] [CrossRef] [PubMed]
- Price, T.J.; Peeters, M.; Kim, T.W.; Li, J.; Cascinu, S.; Ruff, P.; Suresh, A.S.; Thomas, A.; Tjulandin, S.; Zhang, K.; et al. Panitumumab versus cetuximab in patients with chemotherapy-refractory wild-type KRAS exon 2 metastatic colorectal cancer (ASPECCT): A randomised, multicentre, open-label, non-inferiority phase 3 study. Lancet Oncol. 2014, 15, 569–579. [Google Scholar] [CrossRef]
- Van Cutsem, E.; Tejpar, S.; Vanbeckevoort, D.; Peeters, M.; Humblet, Y.; Gelderblom, H.; Vermorken, J.B.; Viret, F.; Glimelius, B.; Gallerani, E.; et al. Intrapatient cetuximab dose escalation in metastatic colorectal cancer according to the grade of early skin reactions: The randomized EVEREST study. J. Clin. Oncol. 2012, 30, 2861–2868. [Google Scholar] [CrossRef] [PubMed]
- Grothey, A.; Van Cutsem, E.; Sobrero, A.; Siena, S.; Falcone, A.; Ychou, M.; Humblet, Y.; Bouché, O.; Mineur, L.; Barone, C.; et al. Regorafenib monotherapy for previously treated metastatic colorectal cancer (CORRECT): An international, multicentre, randomised, placebo-controlled, phase 3 trial. Lancet 2013, 381, 303–312. [Google Scholar] [CrossRef]
- Mayer, R.J.; Van Cutsem, E.; Falcone, A.; Yoshino, T.; Garcia-Carbonero, R.; Mizunuma, N.; Yamazaki, K.; Shimada, Y.; Tabernero, J.; Komatsu, Y.; et al. Randomized trial of TAS-102 for refractory metastatic colorectal cancer. N. Engl. J. Med. 2015, 372, 1909–1919. [Google Scholar] [CrossRef]
- Ciardiello, D.; Vitiello, P.P.; Cardone, C.; Martini, G.; Troiani, T.; Martinelli, E.; Ciardiello, F. Immunotherapy of colorectal cancer: Challenges for therapeutic efficacy. Cancer Treat. Rev. 2019, 76, 22–32. [Google Scholar] [CrossRef] [PubMed]
- Bourhis, J.; Stein, A.; Paul de Boer, J.; Van Den Eynde, M.; Gold, K.A.; Stintzing, S.; Becker, J.C.; Moran, M.; Schroeder, A.; Pennock; et al. Avelumab and cetuximab as a therapeutic combination: An overview of scientific rationale and current clinical trials in cancer. Cancer Treat. Rev. 2021, 97, 102172. [Google Scholar] [CrossRef]
- Ferris, R.L.; Lenz, H.J.; Trotta, A.M.; García-Foncillas, J.; Schulten, J.; Audhuy, F.; Merlano, M.; Milano, G. Rationale for combination of therapeutic antibodies targeting tumor cells and immune checkpoint receptors: Harnessing innate and adaptive immunity through IgG1 isotype immune effector stimulation. Cancer Treat. Rev. 2018, 63, 48–60. [Google Scholar] [CrossRef]
- Fasano, M.; Della Corte, C.M.; Di Liello, R.; Barra, G.; Sparano, F.; Viscardi, G.; Iacovino, M.L.; Paragliola, F.; Famiglietti, V.; Ciaramella, E.; et al. Induction of natural killer antibody-dependent cell cytotoxicity and of clinical activity of cetuximab plus avelumab in non-small cell lung cancer. ESMO Open 2020, 5, e000753. [Google Scholar] [CrossRef]
- Lulli, D.; Carbone, M.L.; Pastore, S. Epidermal growth factor receptor inhibitors trigger a type I interferon response in human skin. Oncotarget 2016, 7, 47777–47793. [Google Scholar] [CrossRef] [PubMed]
- Gurule, N.J.; Heasley, L.E. Linking tyrosine kinase inhibitor-mediated inflammation with normal epithelial cell homeostasis and tumor therapeutic responses. Cancer Drug Resist. 2018, 1, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Mascia, F.; Lam, G.; Keith, C.; Garber, C.; Steinberg, S.M.; Kohn, E.; Yuspa, S.H. Genetic ablation of epidermal EGFR reveals the dynamic origin of adverse effects of anti-EGFR therapy. Sci. Transl. Med. 2013, 5, 199ra110. [Google Scholar] [CrossRef] [PubMed]
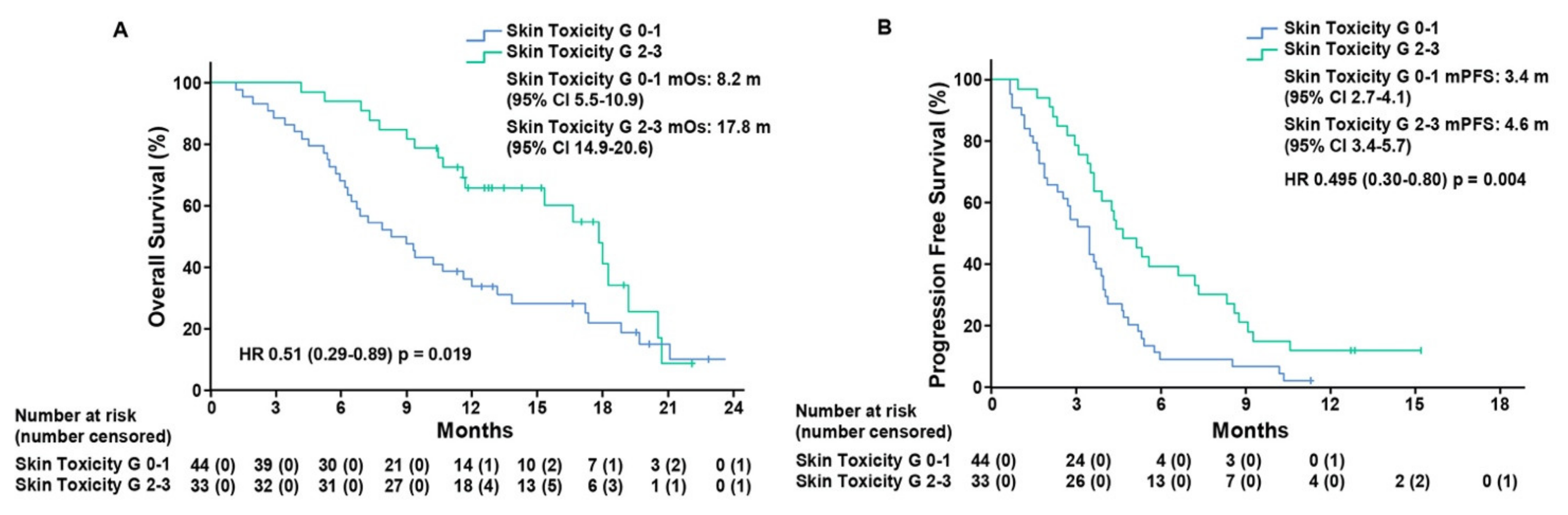

| Univariate Analysis | Multivariate Analysis | ||||
|---|---|---|---|---|---|
| Variable | Number of Patients | Hazard Ratio (CI 95%) | p-Value | Hazard Ratio (CI 95%) | p-Value |
| Skin Rash | |||||
| Grade 0–1 | 44 (57.1%) | 1.00 (ref) | 1.00 (ref) | ||
| Grade 2–3 | 33 (42.9%) | 0.495 (0.30–0.80) | 0.004 | 0.71 (0.39–1.27) | 0.25 |
| Sex | |||||
| Female | 35 (45.5%) | 0.89 (0.56–1.42) | 0.64 | ||
| Male | 42 (54.5%) | 1.00 (ref) | |||
| ECOG | |||||
| 0 | 52 (67.5%) | 0.78 (0.48–1.31) | 0.37 | ||
| 1 | 25 (32.5%) | 1.00 (ref) | |||
| Lines of treatment | |||||
| III | 56 (72.7%) | 1.00 (ref) | |||
| >III | 21 (27.3%) | 0.87 (0.51–1.47) | 0.61 | ||
| Number of metastatic sites | |||||
| ≤II | 45 (58.4%) | 0.54 (0.33–0.87) | 0.013 | 0.72 (0.41–1.25) | 0.24 |
| >II | 32 (41.6%) | 1.00 (ref) | 1.00 (ref) | ||
| Surgery of the primary tumor | |||||
| Yes | 48 (62.3%) | 0.58 (0.36–0.94) | 0.028 | 0.70 (0.41–1.20) | 0.20 |
| No | 29 (37.7%) | 1.00 (ref) | 1.00 (ref) | ||
| Microsatellites Instability | |||||
| MSI | 3 (5.41%) | 0.59 (0.18–1.91) | 0.59 | ||
| MSS | 71 (95.9%) | 1.00 (ref) | |||
| Sidedness | |||||
| Left and rectum | 72 (93.5%) | 0.45 (0.17–1.15) | 0.096 | ||
| Right | 5 (6.5%) | 1.00 (ref) | |||
| Synchronous metastases | |||||
| Yes | 56 (72.7%) | 1.00 (ref) | |||
| No | 21 (27.3%) | 0.64 (0.37–1.10) | 0.11 | ||
| RAS/BRAF/EGFR mutational status | |||||
| WT | 48 (71.6%) | 0.41 (0.23–0.75) | 0.004 | 0.58 (0.3–1.08) | 0.10 |
| MT | 19 (28.4%) | 1.00 (ref) | 1.00 (ref) | ||
| Univariate Analysis | Multivariate Analysis | ||||
|---|---|---|---|---|---|
| Variable | Number of Patients | Hazard Ratio (CI 95%) | p-Value | Hazard Ratio (CI 95%) | p-Value |
| Skin Rash | |||||
| Grade 0–1 | 44 (57.1%) | 1.00 (ref) | 1.00 (ref) | ||
| Grade 2–3 | 33 (42.9%) | 0.51 (0.29–0.89) | 0.019 | 0.54 (0.29–0.1.01) | 0.054 |
| Sex | |||||
| Female | 35 (45.5%) | 1.03 (0.60–1.76) | 0.90 | ||
| Male | 42 (54.5%) | 1.00 (ref) | |||
| ECOG | |||||
| 0 | 52 (67.5%) | 0.64 (0.36–1.11) | 0.11 | ||
| 1 | 25 (32.5%) | 1.00 (ref) | |||
| Lines of treatment | |||||
| III | 56 (72.7%) | 1.00 (ref) | |||
| >III | 21 (27.3%) | 1.12 (0.63–1.99) | 0.69 | ||
| Number of metastatic sites | |||||
| ≤II | 45 (58.4%) | 0.74 (0.43–1.27) | 0.28 | ||
| >II | 32 (41.6%) | 1.00 (ref) | |||
| Surgery of the primary tumor | |||||
| Yes | 48 (62.3%) | 0.65 (0.38–1.11) | 0.12 | ||
| No | 29 (37.7%) | 1.00 (ref) | |||
| Microsatellites Instability | |||||
| MSI | 3 (5.41%) | 1.25 (0.30–5.1) | 0.75 | ||
| MSS | 71 (95.9%) | 1.00 (ref) | |||
| Sidedness | |||||
| Left and rectum | 72 (93.5%) | 1.25 (0.44–3.47) | 0.67 | ||
| Right | 5 (6.5%) | 1.00 (ref) | |||
| Synchronous metastases | |||||
| Yes | 56 (72.7%) | 1.00 (ref) | |||
| No | 21 (27.3%) | 0.73 (0.39–1.37) | 0.34 | ||
| RAS/BRAF/EGFR mutational status | |||||
| WT | 48 (71.6%) | 0.50 (0.27–0.90) | 0.022 | 0.49 (0.27–0.90) | 0.023 |
| MT | 19 (28.4%) | 1.00 (ref) | 1.00 (ref) | ||
| Variable | Number of Patients | CR | PR | SD | PD |
|---|---|---|---|---|---|
| Skin toxicity | |||||
| Grade 0–1 | 44 (57.1%) | 0 (0%) | 3 (6.8%) | 20 (45.5%) | 21 (47.7%) |
| Grade 2–3 | 33 (42.9%) | 1 (3%) | 2 (6.1%) | 24 (72.7%) | 6 (18.2%) |
| Sex | |||||
| Female | 35 (45.5%) | 1 (2.9%) | 3 (8.6%) | 21 (60%) | 10 (28.6%) |
| Male | 42 (54.5%) | 0 (0%) | 2 (4.8%) | 23 (54.8%) | 17 (40.5%) |
| ECOG | |||||
| 0 | 52 (67.5%) | 1 (1.9%) | 4 (7.7%) | 29 (55.8%) | 18 (34.6%) |
| 1 | 25 (32.5%) | 0 (0%) | 1 (4.0%) | 15 (60%) | 9 (36.0%) |
| Lines of treatment | |||||
| III | 56 (72.7%) | 1 (1.8%) | 3 (5.4%) | 31 (55.4%) | 21 (37.5%) |
| >III | 21 (27.3%) | 0 (0%) | 2 (9.5%) | 13 (61.9%) | 6 (28.6%) |
| Number of metastatic sites | |||||
| ≤II | 45 (58.4%) | 1 (2.2%) | 2 (4.4%) | 29 (64.4%) | 13 (28.9%) |
| >II | 32 (41.6%) | 0 (0%) | 3 (9.4%) | 15 (46.9%) | 14 (43.8) |
| Surgery of the primary tumor | |||||
| Yes | 48 (62.3%) | 1 (2.1%) | 4 (8.3%) | 31 (64.6%) | 12 (25%) |
| No | 29 (37.7%) | 0 (0%) | 1 (3.4%) | 13 (44.8%) | 15 (51.7%) |
| Microsatellites Instability | |||||
| MSI | 3 (5.41%) | 0 (0%) | 0 (0%) | 3 (100%) | 0 (0%) |
| MSS | 71 (95.9%) | 1 (1.4%) | 5 (7%) | 40 (56.3%) | 25 (35.2%) |
| Sidedness | |||||
| Left and rectum | 72 (93.5%) | 1 (1.4%) | 4 (5.6%) | 43 (59.7%) | 24 (33.3%) |
| Right | 5 (6.5%) | 0 (0%) | 1 (20%) | 1 (20%) | 3 (60%) |
| Synchronous metastases | |||||
| Yes | 56 (72.7%) | 1 (1.8%) | 3 (5.4%) | 30 (53.6%) | 22 (39.3%) |
| No | 21 (27.3%) | 0 (0%) | 2 (9.5%) | 14 (66.7%) | 5 (23.8%) |
| RAS/BRAF/EGFR mutational status | |||||
| WT | 48 (71.6%) | 1 (2.1%) | 3 (6.3%) | 31 (64.6%) | 13 (27.1%) |
| MT | 19 (28.4%) | 0 (0%) | 1 (5.3%) | 8 (42.1%) | 10 (52.6%) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ciardiello, D.; Famiglietti, V.; Napolitano, S.; Esposito, L.; Normanno, N.; Avallone, A.; Latiano, T.; Maiello, E.; Pietrantonio, F.; Cremolini, C.; et al. Skin Toxicity as Predictor of Survival in Refractory Patients with RAS Wild-Type Metastatic Colorectal Cancer Treated with Cetuximab and Avelumab (CAVE) as Rechallenge Strategy. Cancers 2021, 13, 5715. https://doi.org/10.3390/cancers13225715
Ciardiello D, Famiglietti V, Napolitano S, Esposito L, Normanno N, Avallone A, Latiano T, Maiello E, Pietrantonio F, Cremolini C, et al. Skin Toxicity as Predictor of Survival in Refractory Patients with RAS Wild-Type Metastatic Colorectal Cancer Treated with Cetuximab and Avelumab (CAVE) as Rechallenge Strategy. Cancers. 2021; 13(22):5715. https://doi.org/10.3390/cancers13225715
Chicago/Turabian StyleCiardiello, Davide, Vincenzo Famiglietti, Stefania Napolitano, Lucia Esposito, Nicola Normanno, Antonio Avallone, Tiziana Latiano, Evaristo Maiello, Filippo Pietrantonio, Chiara Cremolini, and et al. 2021. "Skin Toxicity as Predictor of Survival in Refractory Patients with RAS Wild-Type Metastatic Colorectal Cancer Treated with Cetuximab and Avelumab (CAVE) as Rechallenge Strategy" Cancers 13, no. 22: 5715. https://doi.org/10.3390/cancers13225715
APA StyleCiardiello, D., Famiglietti, V., Napolitano, S., Esposito, L., Normanno, N., Avallone, A., Latiano, T., Maiello, E., Pietrantonio, F., Cremolini, C., Santabarbara, G., Pinto, C., Troiani, T., Martinelli, E., Ciardiello, F., & Martini, G. (2021). Skin Toxicity as Predictor of Survival in Refractory Patients with RAS Wild-Type Metastatic Colorectal Cancer Treated with Cetuximab and Avelumab (CAVE) as Rechallenge Strategy. Cancers, 13(22), 5715. https://doi.org/10.3390/cancers13225715











