Molecular Response to Combined Molecular- and External Radiotherapy in Head and Neck Squamous Cell Carcinoma (HNSCC)
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
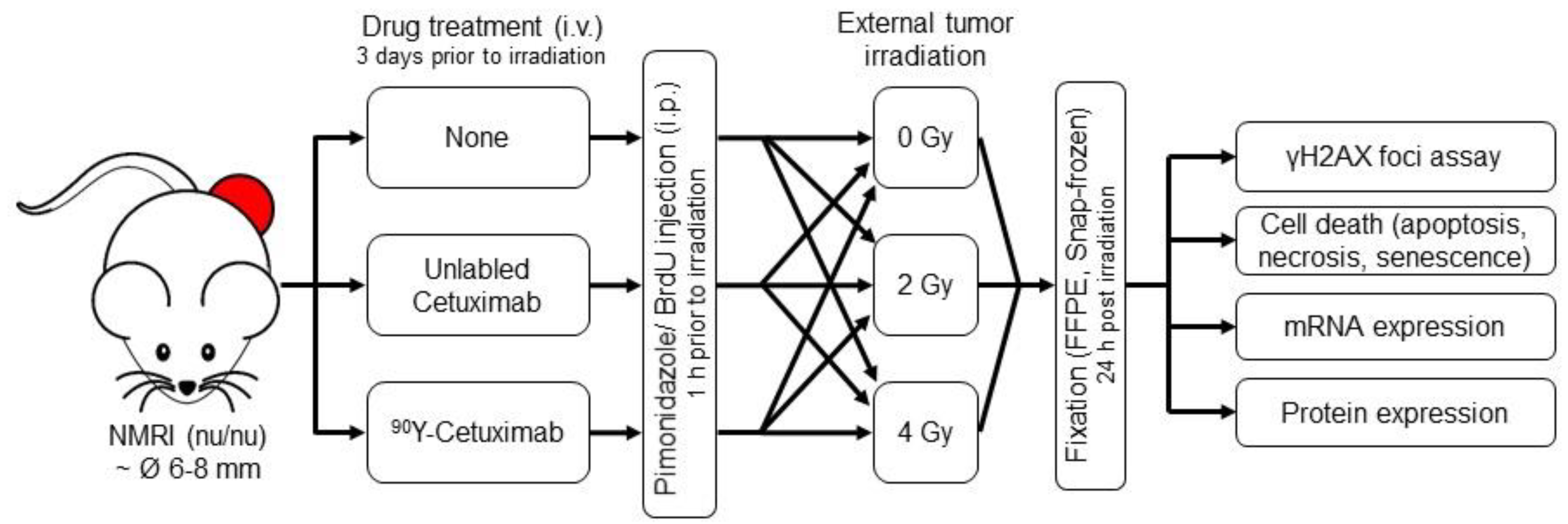
2.1. Animal Experiments
2.2. Radioactivity Measurement of 90Y in Tumors and Organs
2.3. Immunofluorescence, Immunohistochemistry and Histological Staining
2.4. Determination of RNA Expression
2.5. Immunoblotting
2.6. Image Acquisition and Analysis
2.7. Statistical Analysis
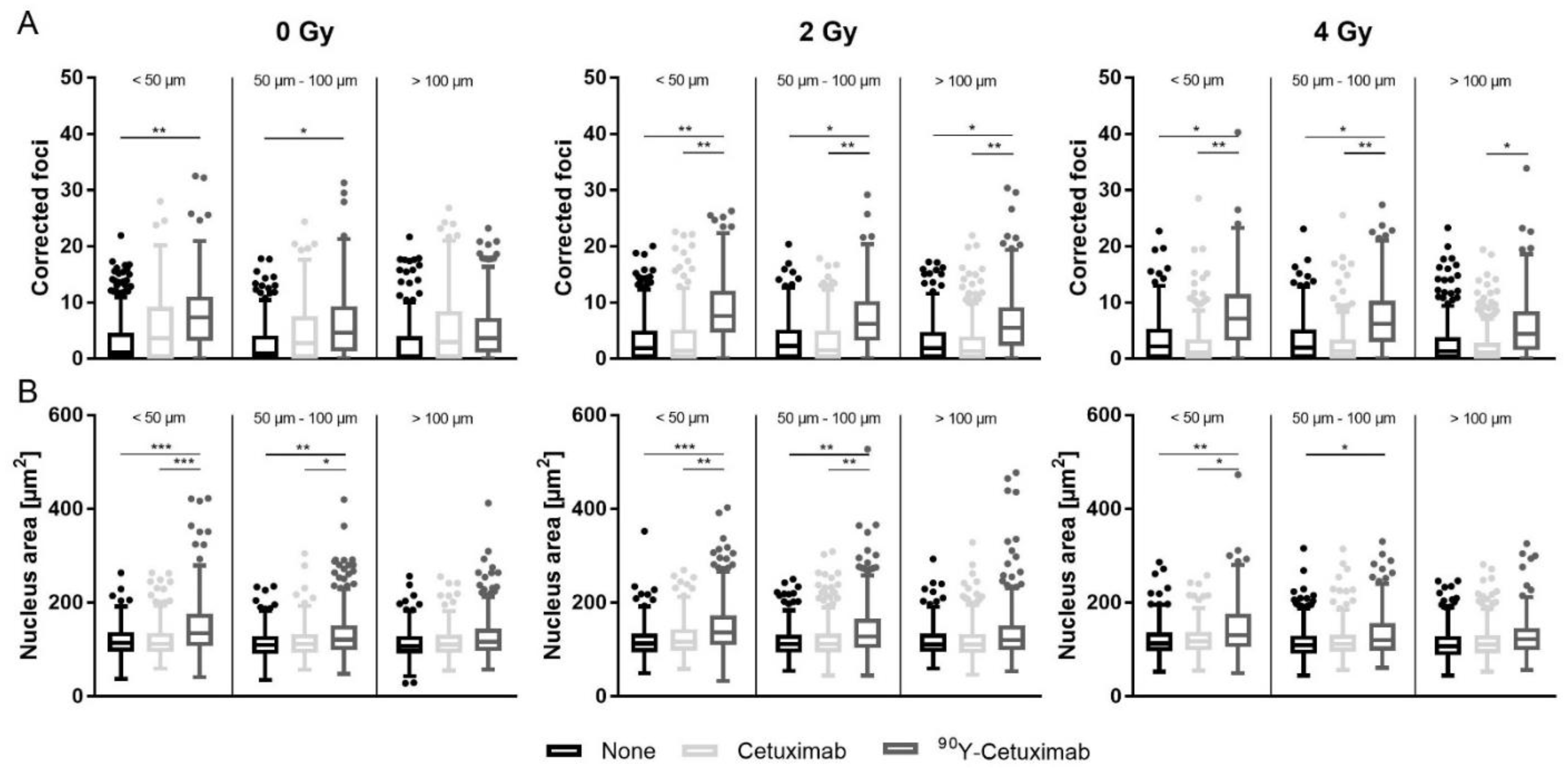
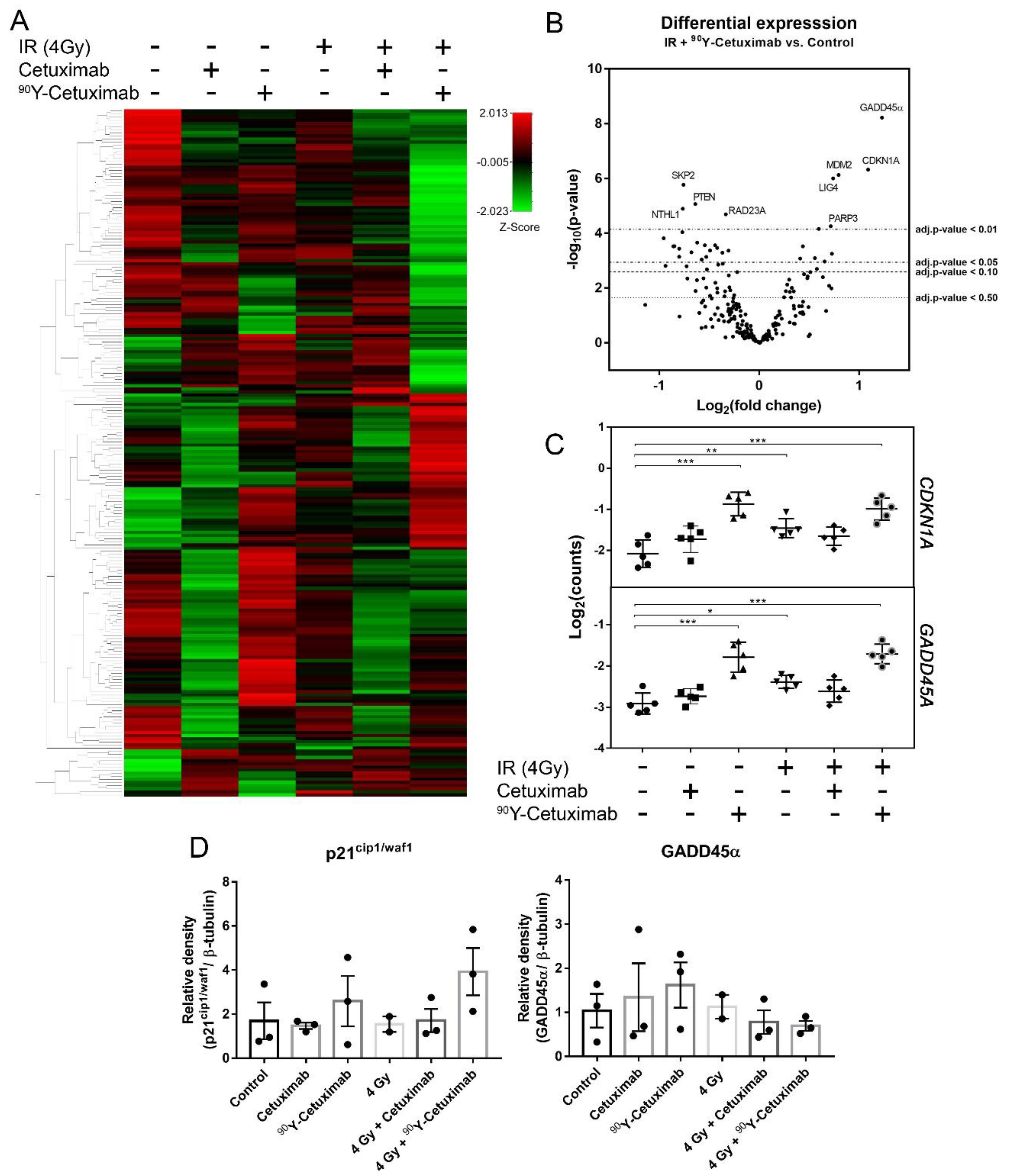
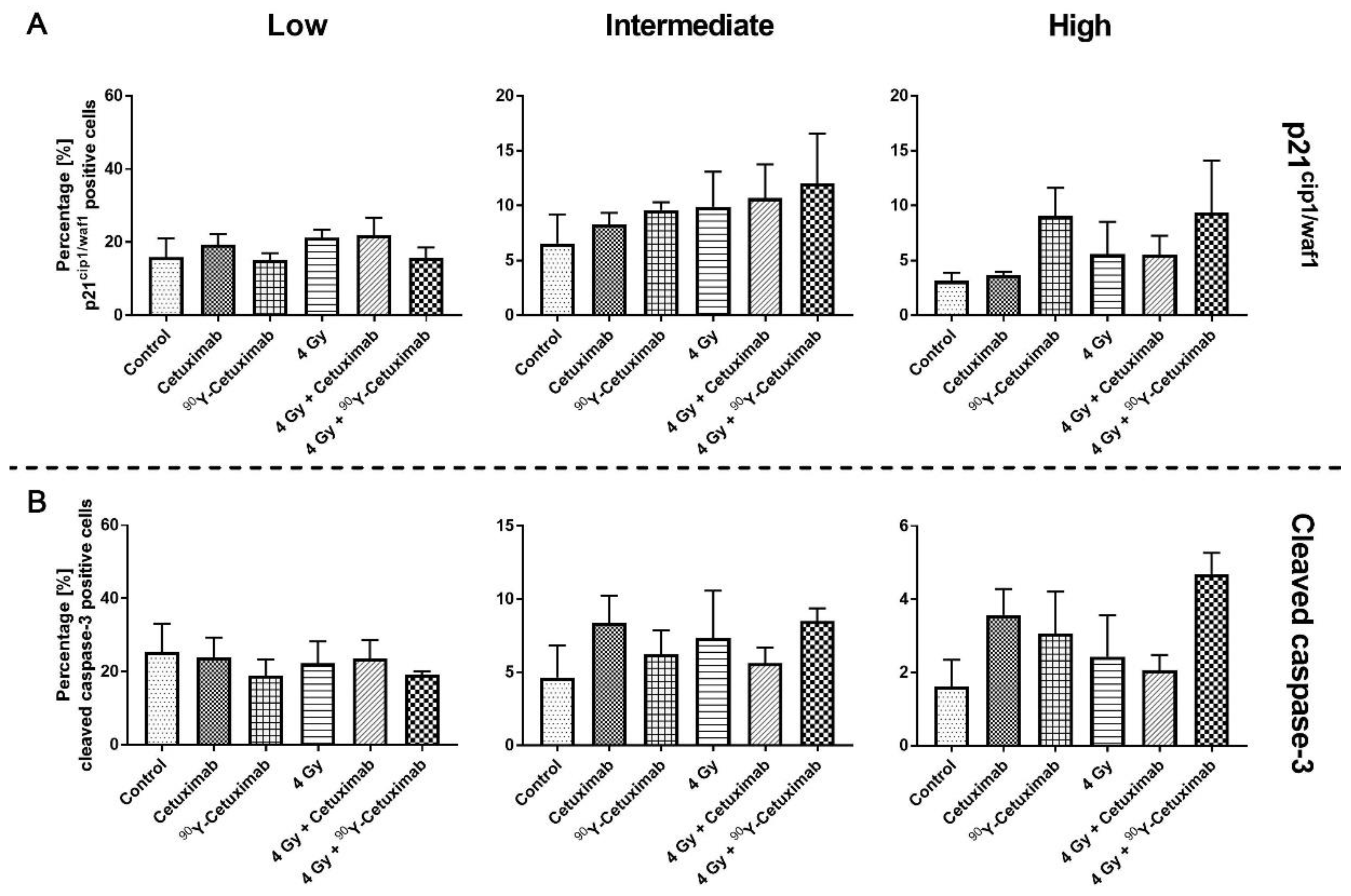
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Cramer, J.D.; Burtness, B.; Le, Q.T.; Ferris, R.L. The changing therapeutic landscape of head and neck cancer. Nat. Rev. Clin. Oncol. 2019, 16, 669–683. [Google Scholar] [CrossRef] [PubMed]
- Gudkov, S.V.; Shilyagina, N.Y.; Vodeneev, V.A.; Zvyagin, A.V. Targeted radionuclide therapy of human tumors. Int. J. Mol. Sci. 2016, 17, 33. [Google Scholar] [CrossRef] [PubMed]
- Dietrich, A.; Koi, L.; Zöphel, K.; Sihver, W.; Kotzerke, J.; Baumann, M.; Krause, M. Improving external beam radiotherapy by combination with internal irradiation. Br. J. Radiol. 2015, 88, 20150042. [Google Scholar] [CrossRef]
- Wieduwilt, M.J.; Moasser, M.M. The epidermal growth factor receptor family: Biology driving targeted therapeutics. Cell. Mol. Life Sci. 2008, 65, 1566–1584. [Google Scholar] [CrossRef]
- Sihver, W.; Pietzsch, J.; Krause, M.; Baumann, M.; Steinbach, J.; Pietzsch, H.J. Radiolabeled cetuximab conjugates for EGFR targeted cancer diagnostics and therapy. Pharmaceuticals 2014, 7, 311–338. [Google Scholar] [CrossRef]
- Song, I.H.; Lee, T.S.; Park, Y.S.; Lee, J.S.; Lee, B.C.; Moon, B.S.; An, G., II; Lee, H.W.; Kim, K., II; Lee, Y.J.; et al. Immuno-PET imaging and radioimmunotherapy of 64Cu-/177Lu-Labeled Anti-EGFR antibody in esophageal squamous cell carcinoma model. J. Nucl. Med. 2016, 57, 1105–1111. [Google Scholar] [CrossRef]
- Sgouros, G.; Bodei, L.; McDevitt, M.R.; Nedrow, J.R. Radiopharmaceutical therapy in cancer: Clinical advances and challenges. Nat. Rev. Drug Discov. 2020, 19, 589–608. [Google Scholar] [CrossRef]
- Melzig, C.; Golestaneh, A.F.; Mier, W.; Schwager, C.; Das, S.; Schlegel, J.; Lasitschka, F.; Kauczor, H.-U.; Debus, J.; Haberkorn, U.; et al. Combined external beam radiotherapy with carbon ions and tumor targeting endoradiotherapy. Oncotarget 2018, 9, 29985–30004. [Google Scholar] [CrossRef]
- Liu, Z.; Liu, Y.; Jia, B.; Zhao, H.; Jin, X.; Li, F.; Chen, X.; Wang, F. Epidermal growth factor receptor-targeted radioimmunotherapy of human head and neck cancer xenografts using 90Y-labeled fully human antibody panitumumab. Mol. Cancer Ther. 2010, 9, 2297–2308. [Google Scholar] [CrossRef]
- Koi, L.; Bergmann, R.; Brüchner, K.; Pietzsch, J.; Pietzsch, H.J.; Krause, M.; Steinbach, J.; Zips, D.; Baumann, M. Radiolabeled anti-EGFR-antibody improves local tumor control after external beam radiotherapy and offers theragnostic potential. Radiother. Oncol. 2014, 110, 362–369. [Google Scholar] [CrossRef] [PubMed]
- Dietrich, A.; Andreeff, M.; Koi, L.; Bergmann, R.; Schubert, M.; Schreiner, L.; Löck, S.; Sihver, W.; Freudenberg, R.; Hering, S.; et al. Radiotherapy enhances uptake and efficacy of 90Y-cetuximab: A preclinical trial. Radiother. Oncol. 2021, 155, 285–292. [Google Scholar] [CrossRef]
- Bailey, K.E.; Costantini, D.L.; Cai, Z.; Scollard, D.A.; Chen, Z.; Reilly, R.M.; Vallis, K.A. Epidermal growth factor receptor inhibition modulates the nuclear localization and cytotoxicity of the Auger electron-emitting radiopharmaceutical 111In-DTPA-human epidermal growth factor. J. Nucl. Med. 2007, 48, 1562–1570. [Google Scholar] [CrossRef][Green Version]
- Ingargiola, M.; Runge, R.; Heldt, J.M.; Freudenberg, R.; Steinbach, J.; Cordes, N.; Baumann, M.; Kotzerke, J.; Brockhoff, G.; Kunz-Schughart, L.A. Potential of a Cetuximab-based radioimmunotherapy combined with external irradiation manifests in a 3-D cell assay. Int. J. Cancer 2014, 135, 968–980. [Google Scholar] [CrossRef] [PubMed]
- Kassis, A.I. Therapeutic radionuclides: Biophysical and radiobiologic principles. Semin. Nucl. Med. 2008, 38, 358–366. [Google Scholar] [CrossRef] [PubMed]
- Eke, I.; Ingargiola, M.; Förster, C.; Kunz-Schughart, L.A.; Baumann, M.; Runge, R.; Freudenberg, R.; Kotzerke, J.; Heldt, J.M.; Pietzsch, H.J.; et al. Cytotoxic properties of radionuclide-conjugated Cetuximab without and in combination with external irradiation in head and neck cancer cells in vitro. Int. J. Radiat. Biol. 2014, 90, 678–686. [Google Scholar] [CrossRef] [PubMed]
- Saki, M.; Toulany, M.; Sihver, W.; Zenker, M.; Heldt, J.M.; Mosch, B.; Pietzsch, H.J.; Baumann, M.; Steinbach, J.; Rodemann, H.P. Cellular and molecular properties of 90Y-labeled cetuximab in combination with radiotherapy on human tumor cells in vitro. Strahlenther. Und Onkol. 2012, 188, 823–832. [Google Scholar] [CrossRef]
- Du Sert, N.P.; Hurst, V.; Ahluwalia, A.; Alam, S.; Avey, M.T.; Baker, M.; Browne, W.J.; Clark, A.; Cuthill, I.C.; Dirnagl, U.; et al. The arrive guidelines 2.0: Updated guidelines for reporting animal research. PLoS Biol. 2020, 18, e3000410. [Google Scholar] [CrossRef]
- Krause, M.; Hessel, F.; Zips, D.; Hilberg, F.; Baumann, M. Adjuvant inhibition of the epidermal growth factor receptor after fractionated irradiation of FaDu human squamous cell carcinoma. Radiother. Oncol. 2004, 72, 95–101. [Google Scholar] [CrossRef]
- Eicheler, W.; Zips, D.; Dörfler, A.; Grénman, R.; Baumann, M. Splicing mutations in TP53 in human squamous cell carcinoma lines influence immunohistochemical detection. J. Histochem. Cytochem. 2002, 50, 197–204. [Google Scholar] [CrossRef] [PubMed]
- Yaromina, A.; Zips, D.; Thames, H.D.; Eicheler, W.; Krause, M.; Rosner, A.; Haase, M.; Petersen, C.; Raleigh, J.A.; Quennet, V.; et al. Pimonidazole labelling and response to fractionated irradiation of five human squamous cell carcinoma (hSCC) lines in nude mice: The need for a multivariate approach in biomarker studies. Radiother. Oncol. 2006, 81, 122–129. [Google Scholar] [CrossRef]
- Baumann, M.; Liertz, C.; Baisch, H.; Wiegel, T.; Lorenzen, J.; Arps, H. Impact of overall treatment time of fractionated irradiation on local control of human FaDu squamous cell carcinoma in nude mice. Radiother. Oncol. 1994, 32, 137–143. [Google Scholar] [CrossRef]
- Rassamegevanon, T.; Löck, S.; Baumann, M.; Krause, M.; von Neubeck, C. Comparable radiation response of ex vivo and in vivo irradiated tumor samples determined by residual γH2AX. Radiother. Oncol. 2019, 139, 94–100. [Google Scholar] [CrossRef] [PubMed]
- Huang, D.W.; Sherman, B.T.; Lempicki, R.A. Systematic and integrative analysis of large gene lists using DAVID bioinformatics resources. Nat. Protoc. 2009, 4, 44–57. [Google Scholar] [CrossRef] [PubMed]
- Kanehisa, M.; Furumichi, M.; Sato, Y.; Ishiguro-Watanabe, M.; Tanabe, M. KEGG: Integrating viruses and cellular organisms. Nucleic Acids Res. 2021, 49, D545–D551. [Google Scholar] [CrossRef] [PubMed]
- Wiśniewski, J.R.; Gaugaz, F.Z. Fast and Sensitive Total Protein and Peptide Assays for Proteomic Analysis. Anal. Chem. 2015, 87, 4110–4116. [Google Scholar] [CrossRef]
- Schindelin, J.; Arganda-Carreras, I.; Frise, E.; Kaynig, V.; Longair, M.; Pietzsch, T.; Preibisch, S.; Rueden, C.; Saalfeld, S.; Schmid, B.; et al. Fiji: An open-source platform for biological-image analysis. Nat. Methods 2012, 9, 676–682. [Google Scholar] [CrossRef]
- Menegakis, A.; Eicheler, W.; Yaromina, A.; Thames, H.D.; Krause, M.; Baumann, M. Residual DNA double strand breaks in perfused but not in unperfused areas determine different radiosensitivity of tumours. Radiother. Oncol. 2011, 100, 137–144. [Google Scholar] [CrossRef] [PubMed]
- Meerz, A.; Deville, S.S.; Müller, J.; Cordes, N. Comparative Therapeutic Exploitability of Acute Adaptation Mechanisms to Photon and Proton Irradiation in 3D Head and Neck Squamous Cell Carcinoma Cell Cultures. Cancers 2021, 13, 1190. [Google Scholar] [CrossRef]
- Schmidt, U.; Weigert, M.; Broaddus, C.; Myers, G. Cell Detection with Star-Convex Polygons. In Medical Image Computing and Computer Assisted Intervention – MICCAI 2018, Proceedings of the 21st International Conference, Granada, Spain, 16-20 September 2018; Frangi, A., Schnabel, J., Davatzikos, C., Alberola-López, C., Fichtinger, G., Eds.; Springer International Publishing: Cham, Switzerland, 2018; pp. 265–273. [Google Scholar] [CrossRef]
- Rassamegevanon, T.; Löck, S.; Range, U.; Krause, M.; Baumann, M.; von Neubeck, C. Tumor heterogeneity determined with a γH2AX foci assay: A study in human head and neck squamous cell carcinoma (hHNSCC) models. Radiother. Oncol. 2017, 124, 379–385. [Google Scholar] [CrossRef]
- Bankhead, P.; Loughrey, M.B.; Fernández, J.A.; Dombrowski, Y.; McArt, D.G.; Dunne, P.D.; McQuaid, S.; Gray, R.T.; Murray, L.J.; Coleman, H.G.; et al. QuPath: Open source software for digital pathology image analysis. Sci. Rep. 2017, 7, 16878. [Google Scholar] [CrossRef] [PubMed]
- RStudio Team. RStudio: Integrated Development for R; RStudio, PBC: Boston, MA, USA, 2020. [Google Scholar]
- Overgaard, J. Hypoxic radiosensitization: Adored and ignored. J. Clin. Oncol. 2007, 25, 4066–4074. [Google Scholar] [CrossRef] [PubMed]
- Groebe, K.; Vaupel, P. Evaluation of oxygen diffusion distances in human breast cancer xenografts using tumor-specific in vivo data: Role of various mechanisms in the development of tumor hypoxia. Int. J. Radiat. Oncol. Biol. Phys. 1988, 15, 691–697. [Google Scholar] [CrossRef]
- Grimes, D.R.; Kannan, P.; Warren, D.R.; Markelc, B.; Bates, R.; Muschel, R.; Partridge, M. Estimating oxygen distribution from vasculature in three-dimensional tumour tissue. J. R. Soc. Interface 2016, 13, 20160070. [Google Scholar] [CrossRef]
- Dittmann, K.; Mayer, C.; Fehrenbacher, B.; Schaller, M.; Kehlbach, R.; Rodemann, H.P. Nuclear EGFR shuttling induced by ionizing radiation is regulated by phosphorylation at residue Thr654. FEBS Lett. 2010, 584, 3878–3884. [Google Scholar] [CrossRef] [PubMed]
- Dittmann, K.; Mayer, C.; Fehrenbacher, B.; Schaller, M.; Raju, U.; Milas, L.; Chen, D.J.; Kehlbach, R.; Rodemann, H.P. Radiation-induced epidermal growth factor receptor nuclear import is linked to activation of DNA-dependent protein kinase. J. Biol. Chem. 2005, 280, 31182–31189. [Google Scholar] [CrossRef]
- Cornelissen, B.; A Vallis, K. Targeting the Nucleus: An Overview of Auger-Electron Radionuclide Therapy. Curr. Drug Discov. Technol. 2010, 7, 263–279. [Google Scholar] [CrossRef]
- Kis, E.; Szatmári, T.; Keszei, M.; Farkas, R.; Ésik, O.; Lumniczky, K.; Falus, A.; Sáfrány, G. Microarray analysis of radiation response genes in primary human fibroblasts. Int. J. Radiat. Oncol. Biol. Phys. 2006, 66, 1506–1514. [Google Scholar] [CrossRef] [PubMed]
- Nickoloff, J.A.; Sharma, N.; Taylor, L. Clustered DNA double-strand breaks: Biological effects and relevance to cancer radiotherapy. Genes (Basel) 2020, 11, 99. [Google Scholar] [CrossRef]
- Caron, M.C.; Sharma, A.K.; O’Sullivan, J.; Myler, L.R.; Ferreira, M.T.; Rodrigue, A.; Coulombe, Y.; Ethier, C.; Gagné, J.P.; Langelier, M.F.; et al. Poly(ADP-ribose) polymerase-1 antagonizes DNA resection at double-strand breaks. Nat. Commun. 2019, 10, 2954. [Google Scholar] [CrossRef] [PubMed]
- Schultz, N.; Lopez, E.; Saleh-Gohari, N.; Helleday, T. Poly(ADP-ribose) polymerase (PARP-1) has a controlling role in homologous recombination. Nucleic Acids Res. 2003, 31, 4959. [Google Scholar] [CrossRef]
- Aguilar-Quesada, R.; Muñoz-Gámez, J.A.; Martín-Oliva, D.; Peralta, A.; Valenzuela, M.T.; Matínez-Romero, R.; Quiles-Pérez, R.; Menissier-de Murcia, J.; de Murcia, G.; Ruiz de Almodóvar, M.; et al. Interaction between ATM and PARP-1 in response to DNA damage and sensitization of ATM deficient cells through PARP inhibition. BMC Mol. Biol. 2007, 8, 29. [Google Scholar] [CrossRef] [PubMed]
- Jin, S.; Mazzacurati, L.; Zhu, X.; Tong, T.; Song, Y.; Shujuan, S.; Petrik, K.L.; Rajasekaran, B.; Wu, M.; Zhan, Q. Gadd45a contributes to p53 stabilization in response to DNA damage. Oncogene 2003, 22, 8536–8540. [Google Scholar] [CrossRef]
- Abbas, T.; Dutta, A. P21 in cancer: Intricate networks and multiple activities. Nat. Rev. Cancer 2009, 9, 400–414. [Google Scholar] [CrossRef] [PubMed]
- Gartel, A.L. The conflicting roles of the cdk inhibitor p21(CIP1/WAF1) in apoptosis. Leuk. Res. 2005, 29, 1237–1238. [Google Scholar] [CrossRef]
- Yosef, R.; Pilpel, N.; Papismadov, N.; Gal, H.; Ovadya, Y.; Vadai, E.; Miller, S.; Porat, Z.; Ben-Dor, S.; Krizhanovsky, V. p21 maintains senescent cell viability under persistent DNA damage response by restraining JNK and caspase signaling. EMBO J. 2017, 36, 2280–2295. [Google Scholar] [CrossRef]
- Cazzalini, O.; Scovassi, A.I.; Savio, M.; Stivala, L.A.; Prosperi, E. Multiple roles of the cell cycle inhibitor p21CDKN1A in the DNA damage response. Mutat. Res.-Rev. Mutat. Res. 2010, 704, 12–20. [Google Scholar] [CrossRef] [PubMed]
- Jeong, J.H.; Kang, S.S.; Park, K.K.; Chang, H.W.; Magae, J.; Chang, Y.C. p53-independent induction of G1 arrest and p21WAF1/CIP1 expression by ascofuranone, an isoprenoid antibiotic, through downregulation of c-Myc. Mol. Cancer Ther. 2010, 9, 2102–2113. [Google Scholar] [CrossRef]
- Bonner, J.A.; Harari, P.M.; Giralt, J.; Azarnia, N.; Shin, D.M.; Cohen, R.B.; Jones, C.U.; Sur, R.; Raben, D.; Jassem, J.; et al. Radiotherapy plus Cetuximab for Squamous-Cell Carcinoma of the Head and Neck. N. Engl. J. Med. 2006, 354, 567–578. [Google Scholar] [CrossRef] [PubMed]
- Bonner, J.A.; Harari, P.M.; Giralt, J.; Cohen, R.B.; Jones, C.U.; Sur, R.K.; Raben, D.; Baselga, J.; Spencer, S.A.; Zhu, J.; et al. Radiotherapy plus cetuximab for locoregionally advanced head and neck cancer: 5-year survival data from a phase 3 randomised trial, and relation between cetuximab-induced rash and survival. Lancet Oncol. 2010, 11, 21–28. [Google Scholar] [CrossRef]
- Feng, F.Y.; Lopez, C.A.; Normolle, D.P.; Varambally, S.; Li, X.; Chun, P.Y.; Davis, M.A.; Lawrence, T.S.; Nyati, M.K. Effect of epidermal growth factor receptor inhibitor class in the treatment of head and neck cancer with concurrent radiochemotherapy in vivo. Clin. Cancer Res. 2007, 13, 2512–2518. [Google Scholar] [CrossRef]
- Harari, P.M.; Huang, S.M. Head and neck cancer as a clinical model for molecular targeting of therapy: Combining EGFR blockade with radiation. Int. J. Radiat. Oncol. Biol. Phys. 2001, 49, 427–433. [Google Scholar] [CrossRef]
- Balaban, N.; Moni, J.; Shannon, M.; Dang, L.; Murphy, E.; Goldkorn, T. The effect of ionizing radiation on signal transduction: Antibodies to EGF receptor sensitize A431 cells to radiation. Biochim. Biophys. Acta-Mol. Cell Res. 1996, 1314, 147–156. [Google Scholar] [CrossRef]
- Petersen, C.; Zips, D.; Krause, M.; Schöne, K.; Eicheler, W.; Hoinkis, C.; Thames, H.D.; Baumann, M. Repopulation of FaDu human squamous cell carcinoma during fractionated radiotherapy correlateswith reoxygenation. Int. J. Radiat. Oncol. Biol. Phys. 2001, 51, 483–493. [Google Scholar] [CrossRef]
- Krause, M.; Ostermann, G.; Petersen, C.; Yaromina, A.; Hessel, F.; Harstrick, A.; van der Kogel, A.J.; Thames, H.D.; Baumann, M. Decreased repopulation as well as increased reoxygenation contribute to the improvement in local control after targeting of the EGFR by C225 during fractionated irradiation. Radiother. Oncol. 2005, 76, 162–167. [Google Scholar] [CrossRef]
- Liccardi, G.; Hartley, J.A.; Hochhauser, D. EGFR nuclear translocation modulates DNA repair following cisplatin and ionizing radiation treatment. Cancer Res. 2011, 71, 1103–1114. [Google Scholar] [CrossRef]
- Milas, L.; Mason, K.; Hunter, N.; Petersen, S.; Yamakawa, M.; Ang, K.; Mendelsohn, J.; Fan, Z. In vivo enhancement of tumor radioresponse by C225 antiepidermal growth factor receptor antibody. Clin. Cancer Res. 2000, 6, 701–708. [Google Scholar] [PubMed]
- Vassileva, V.; Rajkumar, V.; Mazzantini, M.; Robson, M.; Badar, A.; Sharma, S.; Årstad, E.; Hochhauser, D.; Lythgoe, M.F.; Boxer, G.M.; et al. Significant Therapeutic Efficacy with Combined Radioimmunotherapy and Cetuximab in Preclinical Models of Colorectal Cancer. J. Nucl. Med. 2015, 56, 1239–1245. [Google Scholar] [CrossRef] [PubMed]
- Davis, T.A.; Kaminski, M.S.; Leonard, J.P.; Hsu, F.J.; Wilkinson, M.; Zelenetz, A.; Wahl, R.L.; Kroll, S.; Coleman, M.; Goris, M.; et al. The Radioisotope Contributes Significantly to the Activity of Radioimmunotherapy. Clin. Cancer Res. 2004, 10, 7792–7798. [Google Scholar] [CrossRef] [PubMed]
- Aerts, H.J.W.L.; Dubois, L.; Perk, L.; Vermaelen, P.; Van Dongen, G.A.M.S.; Wouters, B.G.; Lambin, P. Disparity between in vivo EGFR expression and 89Zr-labeled cetuximab uptake assessed with PET. J. Nucl. Med. 2009, 50, 123–131. [Google Scholar] [CrossRef] [PubMed]
- Van Helden, E.J.; Elias, S.G.; Gerritse, S.L.; van Es, S.C.; Boon, E.; Huisman, M.C.; van Grieken, N.C.T.; Dekker, H.; van Dongen, G.A.M.S.; Vugts, D.J.; et al. [89Zr]Zr-cetuximab PET/CT as biomarker for cetuximab monotherapy in patients with RAS wild-type advanced colorectal cancer. Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 849–859. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rassamegevanon, T.; Feindt, L.; Koi, L.; Müller, J.; Freudenberg, R.; Löck, S.; Sihver, W.; Çevik, E.; Kühn, A.C.; von Neubeck, C.; et al. Molecular Response to Combined Molecular- and External Radiotherapy in Head and Neck Squamous Cell Carcinoma (HNSCC). Cancers 2021, 13, 5595. https://doi.org/10.3390/cancers13225595
Rassamegevanon T, Feindt L, Koi L, Müller J, Freudenberg R, Löck S, Sihver W, Çevik E, Kühn AC, von Neubeck C, et al. Molecular Response to Combined Molecular- and External Radiotherapy in Head and Neck Squamous Cell Carcinoma (HNSCC). Cancers. 2021; 13(22):5595. https://doi.org/10.3390/cancers13225595
Chicago/Turabian StyleRassamegevanon, Treewut, Louis Feindt, Lydia Koi, Johannes Müller, Robert Freudenberg, Steffen Löck, Wiebke Sihver, Enes Çevik, Ariane Christel Kühn, Cläre von Neubeck, and et al. 2021. "Molecular Response to Combined Molecular- and External Radiotherapy in Head and Neck Squamous Cell Carcinoma (HNSCC)" Cancers 13, no. 22: 5595. https://doi.org/10.3390/cancers13225595
APA StyleRassamegevanon, T., Feindt, L., Koi, L., Müller, J., Freudenberg, R., Löck, S., Sihver, W., Çevik, E., Kühn, A. C., von Neubeck, C., Linge, A., Pietzsch, H.-J., Kotzerke, J., Baumann, M., Krause, M., & Dietrich, A. (2021). Molecular Response to Combined Molecular- and External Radiotherapy in Head and Neck Squamous Cell Carcinoma (HNSCC). Cancers, 13(22), 5595. https://doi.org/10.3390/cancers13225595






