Management of Cancer-Related Cognitive Impairment: A Systematic Review of Computerized Cognitive Stimulation and Computerized Physical Activity
Abstract
:Simple Summary
Abstract
1. Introduction
2. Objectives
- Are computerized physical activity, computerized cognitive stimulation or combined interventions efficient to improve cancer-related cognitive impairment?
- What are the risks of bias of the existing studies? Additionally, what is the level of quality of the evidence?
- -
- Population: Participants living with cancer (adults and children).
- -
- Intervention: computerized physical activity, computerized cognitive stimulation and combined intervention.
- -
- Comparisons: usual care, wait-list group or any other intervention other than computerized intervention.
- -
- Outcome: cognitive functioning.
3. Materials and Methods
3.1. Search Strategy
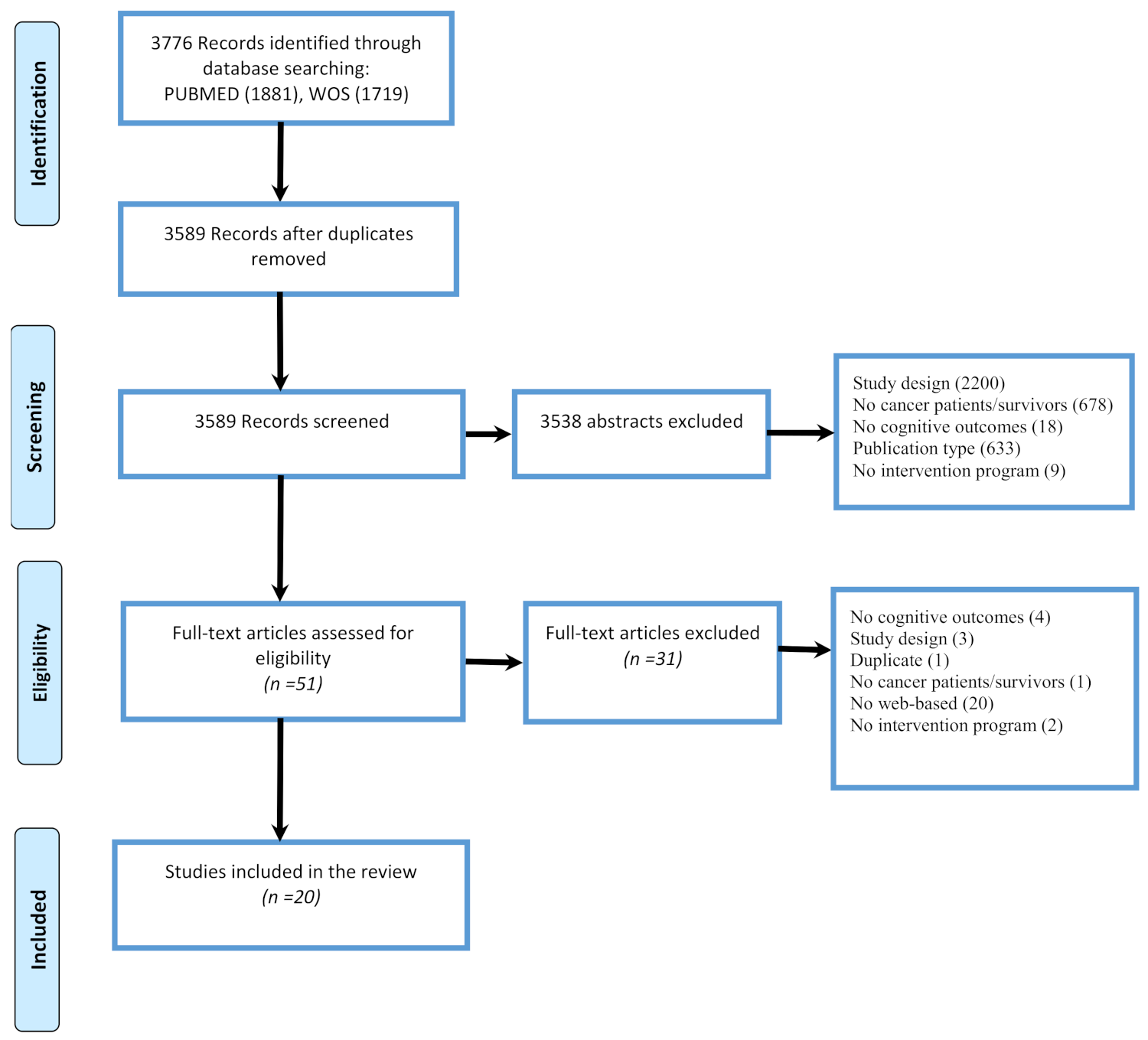
3.2. Selection Process
3.3. Data Extraction
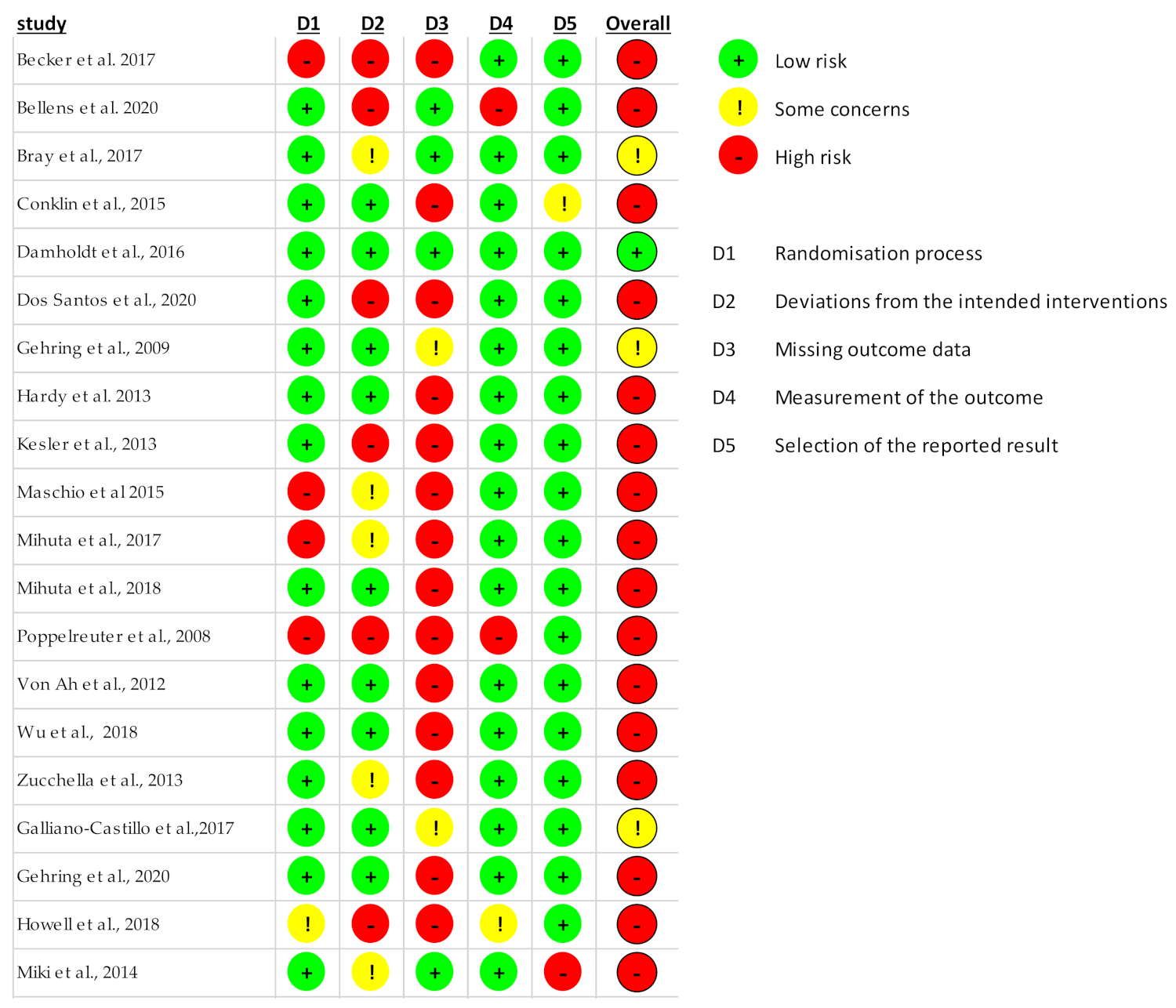
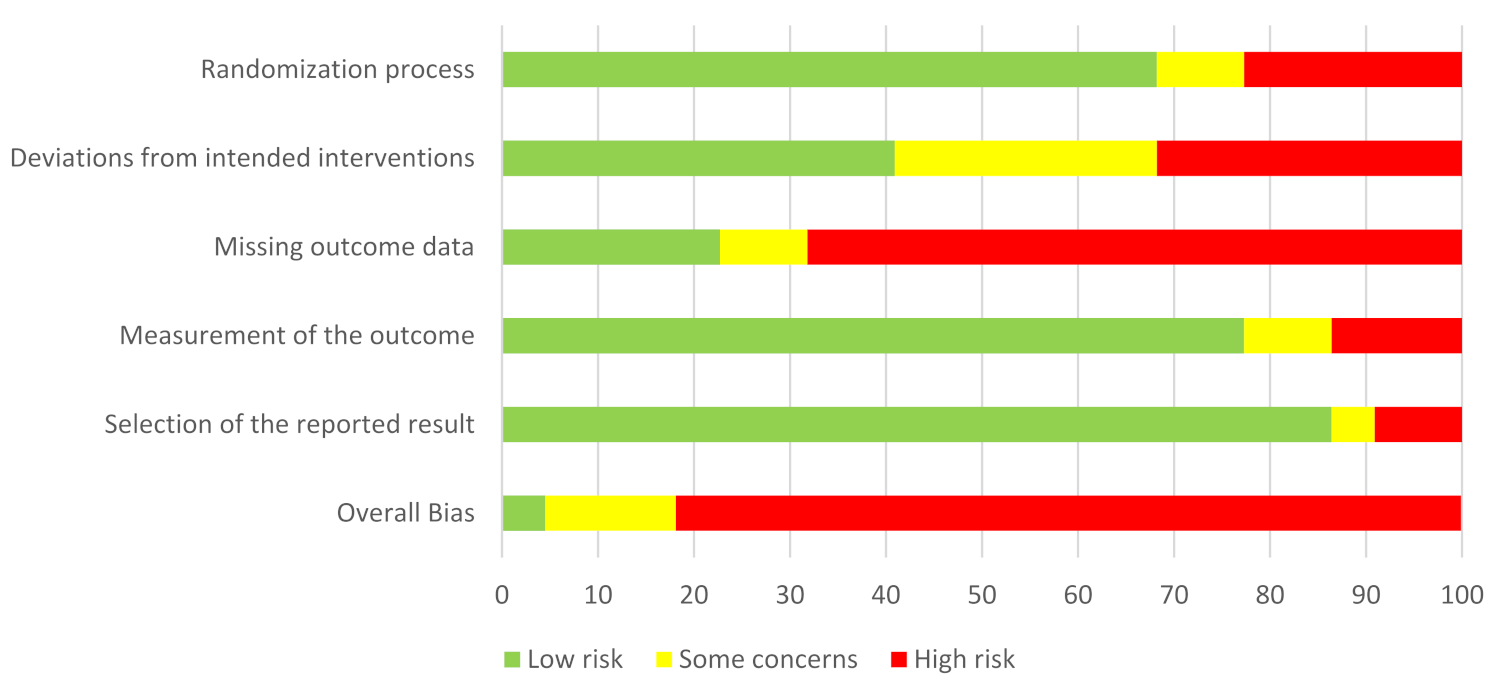
3.4. Quality Assessment and Risk of Bias
4. Results
4.1. Computerized Cognitive Stimulation
4.1.1. Characteristics of Studies
- Population: Five studies included breast-cancer patients [43,46,49,51,56], six did not focus on a specific type of cancer [44,47,50,53,54,55], four included patients with brain cancer [45,48,52,57] and one studied patients with prostate cancer [58]. The intervention was mainly proposed to adults, only two studies proposed the intervention to children [45,50] and none of them concerned older patients (>70 years old). In three studies, chemotherapy treatment was an inclusion criterion [44,47,51]. In one study, intervention was proposed after hematopoietic stem cell transplantation [55], after surgery [57] or to patients on androgen deprivation therapy [58]. The intervention was proposed mostly to cancer survivors [43,44,45,46,51,53,54] and not directly during or after treatment. When stated, the time since treatment completion was between 22 weeks [49] and 6 years [51]. Most of the studies had a control group (14/16), among which 9/16 were wait-list groups.
- Cognitive evaluation: In most studies [43,44,46,47,48,49,50,51,53,54,55,56,58,64], the efficacy of the intervention was evaluated at the end of the intervention by both objective and subjective cognitive assessment, while in two studies it was evaluated only by objective cognitive assessment [52,57] Twelve studies also investigated the maintenance of the efficacy of the intervention on cognition [43,44,46,48,49,50,52,53,54,55,56,58].
4.1.2. Characteristics of Cognitive Stimulation Programs
4.1.3. Efficacy of Cognitive Stimulation Programs
4.2. Computerized Physical Activity
4.2.1. Characteristics of Studies
- Population: Two studies proposed the intervention to adults between 18 and 70 years old [59,60] while one study focused on the elderly (>70 years old) [62] and another on patients younger than 18 years old [61]. One study was proposed to patients with breast cancer [59], two studies did not focus on any specific type of cancer [60,61] and the intervention was proposed to both breast and prostate cancer patients in the last one [62]. Three studies provided treatment history, which showed that most patients were treated with chemotherapy, hormone therapy and/or radiotherapy [59,60,62]. None of them reported information on the time of completion of treatment. All studies had an active control group. Sample size: mean sample size was 66 participants (minimum: 32 [60]; maximum: 78 [61,62]).
- Cognitive evaluation: all studies assessed the efficacy of the physical activity intervention on objective cognitive functioning, and two studies also on cognitive complaints [60,62]. Only one study evaluated the cognitive effect after the post-intervention assessment with another assessment at 6 months [59].
4.2.2. Characteristics of Programs
4.2.3. Efficacy of Programs
5. Discussion
6. Implication for Practice
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
References
- Janelsins, M.C.; Heckler, C.E.; Peppone, L.J.; Kamen, C.; Mustian, K.M.; Mohile, S.G.; Magnuson, A.; Kleckner, I.R.; Guido, J.J.; Young, K.L. Cognitive Complaints in Survivors of Breast Cancer after Chemotherapy Compared with Age-Matched Controls: An Analysis from a Nationwide, Multicenter, Prospective Longitudinal Study. J. Clin. Oncol. 2017, 35, 506. [Google Scholar] [CrossRef]
- Lange, M.; Joly, F.; Vardy, J.; Ahles, T.; Dubois, M.; Tron, L.; Winocur, G.; De Ruiter, M.; Castel, H. Cancer-Related Cognitive Impairment: An Update on State of the Art, Detection, and Management Strategies in Cancer Survivors. Ann. Oncol. 2019, 30, 1925–1940. [Google Scholar] [CrossRef] [Green Version]
- Bower, J.E. Behavioral Symptoms in Breast Cancer Patients and Survivors: Fatigue, Insomnia, Depression, and Cognitive Disturbance. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2008, 26, 768. [Google Scholar] [CrossRef] [Green Version]
- Harrington, C.B.; Hansen, J.A.; Moskowitz, M.; Todd, B.L.; Feuerstein, M. It’s Not over When It’s over: Long-Term Symptoms in Cancer Survivors—A Systematic Review. Int. J. Psychiatry Med. 2010, 40, 163–181. [Google Scholar] [CrossRef]
- Stein, K.D.; Syrjala, K.L.; Andrykowski, M.A. Physical and Psychological Long-term and Late Effects of Cancer. Cancer 2008, 112, 2577–2592. [Google Scholar] [CrossRef]
- Bolton, G.; Isaacs, A. Women’s Experiences of Cancer-Related Cognitive Impairment, Its Impact on Daily Life and Care Received for It Following Treatment for Breast Cancer. Psychol. Health Med. 2018, 23, 1261–1274. [Google Scholar] [CrossRef] [PubMed]
- Selamat, M.H.; Loh, S.Y.; Mackenzie, L.; Vardy, J. Chemobrain Experienced by Breast Cancer Survivors: A Meta-Ethnography Study Investigating Research and Care Implications. PLoS ONE 2014, 9, e108002. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koppelmans, V.; Breteler, M.; Boogerd, W.; Seynaeve, C.; Gundy, C.; Schagen, S. Neuropsychological Performance in Survivors of Breast Cancer More than 20 Years after Adjuvant Chemotherapy. J. Clin. Oncol. 2012, 30, 1080–1086. [Google Scholar] [CrossRef]
- Padgett, L.S.; Van Dyk, K.; Kelly, N.C.; Newman, R.; Hite, S.; Asher, A. Addressing Cancer-Related Cognitive Impairment in Cancer Survivorship. Oncol. Issues 2020, 35, 52–57. [Google Scholar] [CrossRef]
- Olsson Möller, U.; Beck, I.; Rydén, L.; Malmström, M. A Comprehensive Approach to Rehabilitation Interventions Following Breast Cancer Treatment—A Systematic Review of Systematic Reviews. BMC Cancer 2019, 19, 472. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fernandes, H.A.; Richard, N.M.; Edelstein, K. Cognitive Rehabilitation for Cancer-Related Cognitive Dysfunction: A Systematic Review. Support. Care Cancer 2019, 27, 3253–3279. [Google Scholar] [CrossRef]
- Vance, D.E.; Frank, J.S.; Bail, J.; Triebel, K.L.; Niccolai, L.M.; Gerstenecker, A.; Meneses, K. Interventions for Cognitive Deficits in Breast Cancer Survivors Treated With Chemotherapy. Cancer Nurs. 2017, 40, E11. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Y.; Cheng, A.S.; Chan, C.C. Meta-Analysis of the Effects of Neuropsychological Interventions on Cognitive Function in Non–Central Nervous System Cancer Survivors. Integr. Cancer Ther. 2016, 15, 424–434. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clare, L.; Woods, R.T. Cognitive Training and Cognitive Rehabilitation for People with Early-Stage Alzheimer’s Disease: A Review. Neuropsychol. Rehabil. 2004, 14, 385–401. [Google Scholar] [CrossRef]
- Tremblay, M.S.; Aubert, S.; Barnes, J.D.; Saunders, T.J.; Carson, V.; Latimer-Cheung, A.E.; Chastin, S.F.M.; Altenburg, T.M.; Chinapaw, M.J.M.; Altenburg, T.M.; et al. Sedentary Behavior Research Network (SBRN)—Terminology Consensus Project Process and Outcome. Int. J. Behav. Nutr. Phys. Act. 2017, 14, 75. [Google Scholar] [CrossRef] [Green Version]
- Fissler, P.; Küster, O.; Schlee, W.; Kolassa, I.-T. Novelty Interventions to Enhance Broad Cognitive Abilities and Prevent Dementia. In Progress in Brain Research; Elsevier: Amsterdam, The Netherlands, 2013; Volume 207, pp. 403–434. ISBN 978-0-444-63327-9. [Google Scholar]
- Law, L.L.F.; Barnett, F.; Yau, M.K.; Gray, M.A. Effects of Combined Cognitive and Exercise Interventions on Cognition in Older Adults with and without Cognitive Impairment: A Systematic Review. Ageing Res. Rev. 2014, 15, 61–75. [Google Scholar] [CrossRef]
- Curlik, D.M.; Shors, T.J. Training Your Brain: Do Mental and Physical (MAP) Training Enhance Cognition through the Process of Neurogenesis in the Hippocampus? Neuropharmacology 2013, 64, 506–514. [Google Scholar] [CrossRef]
- Hötting, K.; Röder, B. Beneficial Effects of Physical Exercise on Neuroplasticity and Cognition. Neurosci. Biobehav. Rev. 2013, 37, 2243–2257. [Google Scholar] [CrossRef]
- Gheysen, F.; Poppe, L.; DeSmet, A.; Swinnen, S.; Cardon, G.; De Bourdeaudhuij, I.; Chastin, S.; Fias, W. Physical Activity to Improve Cognition in Older Adults: Can Physical Activity Programs Enriched with Cognitive Challenges Enhance the Effects? A Systematic Review and Meta-Analysis. Int. J. Behav. Nutr. Phys. Act. 2018, 15, 63. [Google Scholar] [CrossRef] [Green Version]
- Park, H.; Park, J.H.; Na, H.R.; Hiroyuki, S.; Kim, G.M.; Jung, M.K.; Kim, W.K.; Park, K.W. Combined Intervention of Physical Activity, Aerobic Exercise, and Cognitive Exercise Intervention to Prevent Cognitive Decline for Patients with Mild Cognitive Impairment: A Randomized Controlled Clinical Study. J. Clin. Med. 2019, 8, 940. [Google Scholar] [CrossRef] [Green Version]
- Lauenroth, A.; Ioannidis, A.E.; Teichmann, B. Influence of Combined Physical and Cognitive Training on Cognition: A Systematic Review. BMC Geriatr. 2016, 16, 141. [Google Scholar] [CrossRef] [Green Version]
- Medrano, J.; Brown, J.; Shackelford, D.; Beebe, C.; Brennecke, A. The Effects of a Twelve-Week Aerobic and Cognitive Training Intervention on Cognitive Function in Cancer Survivors. Ursidae Undergrad. Res. J. Univ. North. Colo. 2014, 4, 1. [Google Scholar]
- Peterson, B.M.; Johnson, C.; Case, K.R.; Shackelford, D.Y.; Brown, J.M.; Lalonde, T.L.; Hayward, R. Feasibility of a Combined Aerobic and Cognitive Training Intervention on Cognitive Function in Cancer Survivors: A Pilot Investigation. Pilot Feasibility Stud. 2018, 4, 50. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Runowicz, C.D.; Leach, C.R.; Henry, N.L.; Henry, K.S.; Mackey, H.T.; Cowens-Alvarado, R.L.; Cannady, R.S.; Pratt-Chapman, M.L.; Edge, S.B.; Jacobs, L.A.; et al. American Cancer Society/American Society of Clinical Oncology Breast Cancer Survivorship Care Guideline: ACS/ASCO Breast Cancer Survivorship Guideline. CA. Cancer J. Clin. 2016, 66, 43–73. [Google Scholar] [CrossRef] [PubMed]
- Smidt, K.; Mackenzie, L.; Dhillon, H.; Vardy, J.; Lewis, J.; Loh, S.Y. The Perceptions of Australian Oncologists about Cognitive Changes in Cancer Survivors. Support. Care Cancer 2016, 24, 4679–4687. [Google Scholar] [CrossRef] [PubMed]
- Lange, M.; Heutte, N.; Morel, N.; Eustache, F.; Joly, F.; Giffard, B. Cognitive Complaints in Cancer: The French Version of the Functional Assessment of Cancer Therapy–Cognitive Function (FACT-Cog), Normative Data from a Healthy Population. Neuropsychol. Rehabil. 2016, 26, 392–409. [Google Scholar] [CrossRef]
- Haberlin, C.; O’Dwyer, T.; Mockler, D.; Moran, J.; O’Donnell, D.M.; Broderick, J. The Use of EHealth to Promote Physical Activity in Cancer Survivors: A Systematic Review. Support. Care Cancer 2018, 26, 3323–3336. [Google Scholar] [CrossRef]
- Eysenbach, G. What Is E-Health? J. Med. Internet Res. 2001, 3, e20. [Google Scholar] [CrossRef]
- Buntin, M.B.; Burke, M.F.; Hoaglin, M.C.; Blumenthal, D. The Benefits Of Health Information Technology: A Review Of The Recent Literature Shows Predominantly Positive Results. Health Aff. 2011, 30, 464–471. [Google Scholar] [CrossRef]
- Carey, M.; Noble, N.; Mansfield, E.; Waller, A.; Henskens, F.; Sanson-Fisher, R. The Role of EHealth in Optimizing Preventive Care in the Primary Care Setting. J. Med. Internet Res. 2015, 17, e126. [Google Scholar] [CrossRef]
- Triberti, S.; Savioni, L.; Sebri, V.; Pravettoni, G. EHealth for Improving Quality of Life in Breast Cancer Patients: A Systematic Review. Cancer Treat. Rev. 2019, 74, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Barello, S.; Triberti, S.; Graffigna, G.; Libreri, C.; Serino, S.; Hibbard, J.; Riva, G. EHealth for Patient Engagement: A Systematic Review. Front. Psychol. 2016, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luxton, D.D. Chapter 1—An Introduction to Artificial Intelligence in Behavioral and Mental Health Care. In Artificial Intelligence in Behavioral and Mental Health Care; Luxton, D.D., Ed.; Academic Press: San Diego, CA, USA, 2016; pp. 1–26. ISBN 978-0-12-420248-1. [Google Scholar]
- Barak, A.; Hen, L.; Boniel-Nissim, M.; Shapira, N. A Comprehensive Review and a Meta-Analysis of the Effectiveness of Internet-Based Psychotherapeutic Interventions. J. Technol. Hum. Serv. 2008, 26, 109–160. [Google Scholar] [CrossRef]
- Marks, I.M.; Cavanagh, K.; Gega, L. Hands-on Help: Computer-Aided Psychotherapy; Psychology Press: East Sussex, UK, 2007; ISBN 1-135-43045-4. [Google Scholar]
- Murray, E.; Burns, J.; Tai, S.S.; Lai, R.; Nazareth, I. Interactive Health Communication Applications for People with Chronic Disease. Cochrane Database Syst. Rev. 2004, 4. [Google Scholar] [CrossRef]
- Griffiths, F.; Lindenmeyer, A.; Powell, J.; Lowe, P.; Thorogood, M. Why Are Health Care Interventions Delivered over the Internet? A Systematic Review of the Published Literature. J. Med. Internet Res. 2006, 8, e10. [Google Scholar] [CrossRef]
- Liberati, A.; Altman, D.G.; Tetzlaff, J.; Mulrow, C.; Gøtzsche, P.C.; Ioannidis, J.P.; Clarke, M.; Devereaux, P.J.; Kleijnen, J.; Moher, D. The PRISMA Statement for Reporting Systematic Reviews and Meta-Analyses of Studies That Evaluate Health Care Interventions: Explanation and Elaboration. J. Clin. Epidemiol. 2009, 62, e1–e34. [Google Scholar] [CrossRef] [Green Version]
- Ouzzani, M.; Hammady, H.; Fedorowicz, Z.; Elmagarmid, A. Rayyan—A Web and Mobile App for Systematic Reviews. Syst. Rev. 2016, 5, 210. [Google Scholar] [CrossRef] [Green Version]
- Sterne, J.A.; Savović, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.-Y.; Corbett, M.S.; Eldridge, S.M. RoB 2: A Revised Tool for Assessing Risk of Bias in Randomised Trials. BMJ 2019, 366. [Google Scholar] [CrossRef] [Green Version]
- Berkman, N.D.; Lohr, K.N.; Ansari, M.T.; Balk, E.M.; Kane, R.; McDonagh, M.; Morton, S.C.; Viswanathan, M.; Bass, E.B.; Butler, M.; et al. Grading the Strength of a Body of Evidence When Assessing Health Care Interventions: An EPC Update. J. Clin. Epidemiol. 2015, 68, 1312–1324. [Google Scholar] [CrossRef]
- Bellens, A.; Roelant, E.; Sabbe, B.; Peeters, M.; van Dam, P.A. A Video-Game Based Cognitive Training for Breast Cancer Survivors with Cognitive Impairment: A Prospective Randomized Pilot Trial. Breast 2020, 53, 23–32. [Google Scholar] [CrossRef]
- Bray, V.; Dhillon, H.; Bell, M.; Kabourakis, M.; Fiero, M.; Yip, D.; Boyle, F.; Price, M.; Vardy, J.; Bray, V.J.; et al. Evaluation of a Web-Based Cognitive Rehabilitation Program in Cancer Survivors Reporting Cognitive Symptoms After Chemotherapy. J. Clin. Oncol. 2017, 35, 217. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Conklin, H.; Ogg, R.; Ashford, J.; Scoggins, M.; Zou, P.; Clark, K.; Martin-Elbahesh, K.; Hardy, K.; Merchant, T.; Jeha, S.; et al. Computerized Cognitive Training for Amelioration of Cognitive Late Effects Among Childhood Cancer Survivors: A Randomized Controlled Trial. J. Clin. Oncol. 2015, 33, 3894. [Google Scholar] [CrossRef] [Green Version]
- Damholdt, M.; Mehlsen, M.; O’Toole, M.; Andreasen, R.; Pedersen, A.; Zachariae, R. Web-Based Cognitive Training for Breast Cancer Survivors with Cognitive Complaints-a Randomized Controlled Trial: Web-Based Cognitive Training for Breast Cancer Survivors. Psychooncology 2016, 25, 1293–1300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Santos, M.D.; Hardy-Léger, I.; Rigal, O.; Licaj, I.; Dauchy, S.; Levy, C.; Noal, S.; Segura, C.; Delcambre, C.; Allouache, D.; et al. Cognitive Rehabilitation Program to Improve Cognition of Cancer Patients Treated with Chemotherapy: A 3-arm Randomized Trial. Cancer 2020, 126, 5328–5336. [Google Scholar] [CrossRef]
- Gehring, K.; Sitskoorn, M.; Gundy, C.; Sikkes, S.; Klein, M.; Postma, T.; van den Bent, M.; Beute, G.; Enting, R.; Kappelle, A.; et al. Cognitive Rehabilitation in Patients With Gliomas: A Randomized, Controlled Trial. J. Clin. Oncol. 2009, 27, 3712–3722. [Google Scholar] [CrossRef] [PubMed]
- Becker, H.; Henneghan, A.M.; Volker, D.L.; Mikan, S.Q. A Pilot Study of a Cognitive-Behavioral Intervention for Breast Cancer Survivors. Oncol. Nurs. Forum 2017, 44, 255. [Google Scholar]
- Hardy, K.K.; Willard, V.W.; Allen, T.M.; Bonner, M.J. Working Memory Training in Survivors of Pediatric Cancer: A Randomized Pilot Study: Working Memory Training in Survivors. Psychooncology 2013, 22, 1856–1865. [Google Scholar] [CrossRef] [Green Version]
- Maschio, M.; Dinapoli, L.; Fabi, A.; Giannarelli, D.; Cantelmi, T. Cognitive Rehabilitation Training in Patients with Brain Tumor-Related Epilepsy and Cognitive Deficits: A Pilot Study. J. Neurooncol. 2015, 125, 419–426. [Google Scholar] [CrossRef] [PubMed]
- Mihuta, M.; Green, H.; Shum, D.; Mihuta, M.E.; Green, H.J.; Shum, D.H.K. Web-Based Cognitive Rehabilitation for Survivors of Adult Cancer: A Randomised Controlled Trial. Psychooncology 2018, 27, 1172–1179. [Google Scholar] [CrossRef] [Green Version]
- Mihuta, M.E.; Green, H.J.; Shum, D.H.K. Efficacy of a Web-Based Cognitive Rehabilitation Intervention for Adult Cancer Survivors: A Pilot Study. Eur. J. Cancer Care 2018, 27, e12805. [Google Scholar] [CrossRef] [PubMed]
- Poppelreuter, M.; Weis, J.; Mumm, A.; Orth, H.B.; Bartsch, H.H. Rehabilitation of Therapy-Related Cognitive Deficits in Patients after Hematopoietic Stem Cell Transplantation. Bone Marrow Transplant. 2008, 41, 79–90. [Google Scholar] [CrossRef]
- Ah, D.V.; Carpenter, J.S.; Saykin, A.; Monahan, P.; Wu, J.; Yu, M.; Rebok, G.; Ball, K.; Schneider, B.; Weaver, M.; et al. Advanced Cognitive Training for Breast Cancer Survivors: A Randomized Controlled Trial. Breast Cancer Res. Treat. 2012, 135, 799–809. [Google Scholar] [CrossRef] [Green Version]
- Zucchella, C.; Capone, A.; Codella, V.; De Nunzio, A.M.; Vecchione, C.; Sandrini, G.; Pace, A.; Pierelli, F.; Bartolo, M. Cognitive Rehabilitation for Early Post-Surgery Inpatients Affected by Primary Brain Tumor: A Randomized, Controlled Trial. J. Neurooncol. 2013, 114, 93–100. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.M.; Amidi, A.; Tanenbaum, M.L.; Winkel, G.; Gordon, W.A.; Hall, S.J.; Bovbjerg, K.; Diefenbach, M.A. Computerized Cognitive Training in Prostate Cancer Patients on Androgen Deprivation Therapy: A Pilot Study. Support. Care Cancer 2018, 26, 1917–1926. [Google Scholar] [CrossRef] [PubMed]
- Galiano-Castillo, N.; Cantarero-Villanueva, I.; Fernández-Lao, C.; Ariza-García, A.; Díaz-Rodríguez, L.; Del-Moral-Ávila, R.; Arroyo-Morales, M. Telehealth System: A Randomized Controlled Trial Evaluating the Impact of an Internet-Based Exercise Intervention on Quality of Life, Pain, Muscle Strength, and Fatigue in Breast Cancer Survivors: Telehealth System in Breast Cancer. Cancer 2016, 122, 3166–3174. [Google Scholar] [CrossRef]
- Gehring, K.; Stuiver, M.M.; Visser, E.; Kloek, C.; van den Bent, M.; Hanse, M.; Tijssen, C.; Rutten, G.-J.; Taphoorn, M.J.B.; Aaronson, N.K.; et al. A Pilot Randomized Controlled Trial of Exercise to Improve Cognitive Performance in Patients with Stable Glioma: A Proof of Concept. Neuro-Oncol. 2020, 22, 103–115. [Google Scholar] [CrossRef] [PubMed]
- Howell, C.R.; Krull, K.R.; Partin, R.E.; Kadan-Lottick, N.S.; Robison, L.L.; Hudson, M.M.; Ness, K.K. Randomized Web-Based Physical Activity Intervention in Adolescent Survivors of Childhood Cancer. Pediatr. Blood Cancer 2018, 65, e27216. [Google Scholar] [CrossRef] [PubMed]
- Miki, E.; Kataoka, T.; Okamura, H. Feasibility and Efficacy of Speed-Feedback Therapy with a Bicycle Ergometer on Cognitive Function in Elderly Cancer Patients in Japan: Speed-Feedback Therapy in Elderly Cancer Patients. Psychooncology 2014, 23, 906–913. [Google Scholar] [CrossRef]
- Galiano-Castillo, N.; Arroyo-Morales, M.; Lozano-Lozano, M.; Fernández-Lao, C.; Martín-Martín, L.; Del-Moral-Ávila, R.; Cantarero-Villanueva, I. Effect of an Internet-Based Telehealth System on Functional Capacity and Cognition in Breast Cancer Survivors: A Secondary Analysis of a Randomized Controlled Trial. Support. Care Cancer 2017, 25, 3551–3559. [Google Scholar] [CrossRef]
- Kesler, S.; Hadi Hosseini, S.M.; Heckler, C.; Janelsins, M.; Palesh, O.; Mustian, K.; Morrow, G. Cognitive Training for Improving Executive Function in Chemotherapy-Treated Breast Cancer Survivors. Clin. Breast Cancer 2013, 13, 299–306. [Google Scholar] [CrossRef] [Green Version]
- Chan, R.J.; McCarthy, A.L.; Devenish, J.; Sullivan, K.A.; Chan, A. Systematic Review of Pharmacologic and Non-Pharmacologic Interventions to Manage Cognitive Alterations after Chemotherapy for Breast Cancer. Eur. J. Cancer 2015, 51, 437–450. [Google Scholar] [CrossRef] [Green Version]
- Wirtz, P.; Baumann, F.T. Physical Activity, Exercise and Breast Cancer—What Is the Evidence for Rehabilitation, Aftercare, and Survival A Review. Breast Care 2018, 13, 92–100. [Google Scholar] [CrossRef]
- Bamidis, P.D.; Vivas, A.B.; Styliadis, C.; Frantzidis, C.; Klados, M.; Schlee, W.; Siountas, A.; Papageorgiou, S.G. A Review of Physical and Cognitive Interventions in Aging. Neurosci. Biobehav. Rev. 2014, 44, 206–220. [Google Scholar] [CrossRef]
- Lampit, A.; Hallock, H.; Valenzuela, M. Computerized Cognitive Training in Cognitively Healthy Older Adults: A Systematic Review and Meta-Analysis of Effect Modifiers. Available online: https://click.endnote.com/viewer?doi=10.1371%2Fjournal.pmed.1001756&token=WzIwOTAxNTgsIjEwLjEzNzEvam91cm5hbC5wbWVkLjEwMDE3NTYiXQ.fp5WgKTML8touTpqADfQ871XKFw (accessed on 4 October 2021).
- Knapova, L.; Klocek, A.; Elavsky, S. The Role of Psychological Factors in Older Adults’ Readiness to Use EHealth Technology: Cross-Sectional Questionnaire Study. J. Med. Internet Res. 2020, 22, e14670. [Google Scholar] [CrossRef]
- Kueider, A.M.; Parisi, J.M.; Gross, A.L.; Rebok, G.W. Computerized Cognitive Training with Older Adults: A Systematic Review. PLoS ONE 2012, 7, e40588. [Google Scholar] [CrossRef] [Green Version]
- Holmes, J.; Gathercole, S.E.; Dunning, D.L. Adaptive Training Leads to Sustained Enhancement of Poor Working Memory in Children. Dev. Sci. 2009, 12, F9–F15. [Google Scholar] [CrossRef]
- Klingberg, T.; Johnson, M.; Gillberg, C.G.; Westerberg, H. Computerized Training of Working Memory in Children With ADHD—A Randomized, Controlled Trial. J. Am. Acad. Child Adolesc. Psychiatry 2005, 44, 177–186. [Google Scholar] [CrossRef] [Green Version]
- Grunewaldt, K.H.; Lohaugen, G.C.C.; Austeng, D.; Brubakk, A.-M.; Skranes, J. Working Memory Training Improves Cognitive Function in VLBW Preschoolers. Pediatrics 2013, 131, e747–e754. [Google Scholar] [CrossRef] [Green Version]
- Mezzacappa, E.; Buckner, J. Working Memory Training for Children with Attention Problems or Hyperactivity: A School-Based Pilot Study. Sch. Ment. Health 2010, 2, 202–208. [Google Scholar] [CrossRef]
- Beck, S.J.; Hanson, C.A.; Puffenberger, S.S.; Benninger, K.L.; Benninger, W.B. A Controlled Trial of Working Memory Training for Children and Adolescents with ADHD. J. Clin. Child Adolesc. Psychol. 2010, 39, 825–836. [Google Scholar] [CrossRef] [PubMed]
- Lonkhuizen, P.J.C.; Klaver, K.M.; Wefel, J.S.; Sitskoorn, M.M.; Schagen, S.B.; Gehring, K. Interventions for Cognitive Problems in Adults with Brain Cancer: A Narrative Review. Eur. J. Cancer Care 2019, 28, e13088. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chodzko-Zajko, W.J.; Proctor, D.N.; Fiatarone Singh, M.A.; Minson, C.T.; Nigg, C.R.; Salem, G.J.; Skinner, J.S. Exercise and Physical Activity for Older Adults. Med. Sci. Sports Exerc. 2009, 41, 1510–1530. [Google Scholar] [CrossRef] [PubMed]
- Colcombe, S.; Kramer, A.F. Fitness Effects on the Cognitive Function of Older Adults: A Meta-Analytic Study. Psychol. Sci. 2003, 14, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Hartman, S.J.; Natarajan, L.; Palmer, B.W.; Parker, B.; Patterson, R.E.; Sears, D.D. Impact of Increasing Physical Activity on Cognitive Functioning in Breast Cancer Survivors: Rationale and Study Design of Memory & Motion. Contemp. Clin. Trials 2015, 45, 371–376. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Myers, J.S.; Mitchell, M.; Krigel, S.; Steinhoff, A.; Boyce-White, A.; Van Goethem, K.; Valla, M.; Dai, J.; He, J.; Liu, W.; et al. Qigong Intervention for Breast Cancer Survivors with Complaints of Decreased Cognitive Function. Support. Care Cancer 2019, 27, 1395–1403. [Google Scholar] [CrossRef] [PubMed]
- Derry, H.M.; Jaremka, L.M.; Bennett, J.M.; Peng, J.; Andridge, R.; Shapiro, C.; Malarkey, W.B.; Emery, C.F.; Layman, R.; Mrozek, E.; et al. Yoga and Self-Reported Cognitive Problems in Breast Cancer Survivors: A Randomized Controlled Trial: Yoga and Self-Reported Cognitive Problems in Breast Cancer Survivors. Psychooncology 2015, 24, 958–966. [Google Scholar] [CrossRef]
- Oh, B.; Butow, P.N.; Mullan, B.A.; Clarke, S.J.; Beale, P.J.; Pavlakis, N.; Lee, M.S.; Rosenthal, D.S.; Larkey, L.; Vardy, J. Effect of Medical Qigong on Cognitive Function, Quality of Life, and a Biomarker of Inflammation in Cancer Patients: A Randomized Controlled Trial. Support. Care Cancer 2012, 20, 1235–1242. [Google Scholar] [CrossRef]
- Vadiraja, H.; Rao, M.R.; Nagarathna, R.; Nagendra, H.; Rekha, M.; Vanitha, N.; Gopinath, K.; Srinath, B.; Vishweshwara, M.; Madhavi, Y. Effects of Yoga Program on Quality of Life and Affect in Early Breast Cancer Patients Undergoing Adjuvant Radiotherapy: A Randomized Controlled Trial. Complement. Ther. Med. 2009, 17, 274–280. [Google Scholar] [CrossRef]
- Janelsins, M.C.; Peppone, L.J.; Heckler, C.E.; Kesler, S.R.; Sprod, L.K.; Atkins, J.; Melnik, M.; Kamen, C.; Giguere, J.; Messino, M.J. YOCAS©® Yoga Reduces Self-Reported Memory Difficulty in Cancer Survivors in a Nationwide Randomized Clinical Trial: Investigating Relationships between Memory and Sleep. Integr. Cancer Ther. 2016, 15, 263–271. [Google Scholar] [CrossRef] [Green Version]
- Campbell, K.L.; Zadravec, K.; Bland, K.A.; Chesley, E.; Wolf, F.; Janelsins, M.C. The Effect of Exercise on Cancer-Related Cognitive Impairment and Applications for Physical Therapy: Systematic Review of Randomized Controlled Trials. Phys. Ther. 2020, 100, 523–542. [Google Scholar] [CrossRef]
- Kim, Y.; Kang, S.J. Computerized Programs for Cancer Survivors with Cognitive Problems: A Systematic Review. J. Cancer Surviv. 2019, 13, 911–920. [Google Scholar] [CrossRef]
- Von Ah, D.; Crouch, A. Cognitive Rehabilitation for Cognitive Dysfunction after Cancer and Cancer Treatment: Implications for Nursing Practice. Semin. Oncol. Nurs. 2020, 36, 150977. [Google Scholar] [CrossRef]
| Intervention | N of Randomized Studies/N of Pilot Studies | Strength of Evidence | Risk of Bias | Directness | Consistency | Precision |
|---|---|---|---|---|---|---|
| Computerized cognitive stimulation | 10/6 | Low | High | Direct | Inconsistent | Imprecise |
| Computerized physical activity | 3/1 | Low | High | Direct | Unknown | Imprecise |
| Publication | Study Design and Assessment | Participants | Intervention | Supervision | Outcomes and Tools | Results | Conclusion |
|---|---|---|---|---|---|---|---|
| Becker et al., 2017 [49] | Pilot study Pre-intervention Post-intervention Two-weeks after intervention | Breast cancer survivors N = 20 | 3–4 times/week of 45-min cognitive training (home-based) + 6 90-min group classes supervised by a nurse (BrainHQ) 6 weeks of the program | Home-based cognitive training without supervision On-site group intervention supervised by a nurse | Efficacy: cognition (CVLT; COWAT; SDMT; PROMIS; MMQ) and quality of life (FACT-G) | Efficacy: no significant results | Program feasible but no significant cognitive improvement |
| Bellens et al., 2020 [43] | Pilot RCTBefore intervention Cognitive assessment every 4 weeks For other measures: after 3 and 6 months | Breast cancer survivors early intervention group: n = 23 Delayed intervention group: n = 23 | 3×/week of 60-min video-game over 6 months (Aquasnap) | Home-based intervention with 3 monthly meetings with the principal investigator. Contacts by email or telephone of study team if problems | Primary outcome: MyCQ (cognitive tests) Secondary outcomes: Anxiety (HADS), self-reflectiveness (BCIS), cognitive complaints (CFQ), quality of sleep (PSQI) | Primary outcome: non-significant change in overall MyCQ score between groups for baseline to 3 months. Secondary outcomes: Significant improvement in CFQ scores (p = 0.029) | Improvement of cognitive complaints |
| Bray et al., 2017 [44] | RCT T1: before intervention T2: after intervention T3: 6 months later | Cancer survivors Intervention group: n = 121 Control group: n = 121 | 40-min sessions/week 15 weeks intervention (Insight) or standard care | Home-based intervention without supervision or support | Primary outcome: FACT-COG PCI (cognitive complaints) Secondary outcome: cognitive tests (Cog-State), anxiety/depression (General Health Questionnaire); QoL (FACT-G); fatigue (FACT-F) and stress (Perceived Stress scale) | Primary outcome: Difference between the groups on the FACT-COG PCI was statistically significant, with less PCI in the intervention group at T2 (p < 0.001) sustained at T3 (p < 0.001) Secondary outcomes: No significant difference between groups in cognitive tests at T2 and T3 but significant improvement at T2 on anxiety/depression, fatigue and stress. | Improvement of cognitive complaints |
| Conklin et al., 2015 [45] | RCT pre-intervention post-intervention | Survivors of childhood acute lymphoblastic leukaemia (ALL) or brain tumour (BT) Intervention group: n = 34 Waitlist group: n = 34 | 25 training 30–45 min sessions (Cogmed)+ weekly telephone-based coaching over 5 to 9 weeks | Home-based intervention with weekly coaching telephone calls | Primary outcome: spatial span backwards (WISC-IV) Secondary outcome: Other cognitive tests (WISC-IV, CPT-II, WJ-III) and parent-reported measures (CPRS-3, BRIEF) | Primary outcome: Greater improvement of spatial span backward in the intervention group than the control group (p = 0.002) Secondary outcomes: Greater improvement of intervention vs. control group in WM, attention and processing speed (p = 0.01). Improvement in reported-attention and executive dysfunctions (p < 0.01) | Intervention feasible and efficacious for childhood cancer survivors |
| Damholdt et al., 2016 [46] | RCT pre-intervention post-intervention 5-month follow-up | Breast cancer survivors Intervention group: n = 94 Waitlist group: n = 63 | 30 training 30 min-session over 6 weeks (HappyNeuron Pro) with phone support | Home-based intervention with telephone and email support in case of difficulties. 2 phone calls: one at the beginning of the program and one at the end. | Primary outcome: PASAT (attention and working memory) Secondary outcomes: Cognitive complaints (CFQ), verbal learning (RAVLT), WM (WAIS-IV), EF (Multilingual aphasia examination; D-KEFS, Cognitive estimation task) | Primary outcome: no significant time x group interaction for PASAT Secondary outcomes: post-intervention and 5-month follow-up significant increase in verbal learning (p = 0.043) and digit span backwards (p = 0.040) for the intervention group compared to a waiting list | Improvements in verbal learning and working memory including at 5-month follow-up |
| Dos Santos et al., 2020 [47] | RCT pre-intervention post-intervention | Cancer patients The experimental group (A): N = 55 Control group B: N = 56 Control group C: N = 56 | Experimental group: computer-assisted cognitive rehabilitation (Rehacom). 9 sessions (45–60 min) over 3 months Group control B: cognitive exercises at home (booklet) 9 sessions (30–60 min) over 3 months Group control C: a phone call. 9 over 3 months | On-site intervention supervised by a neuropsychologist | Primary outcome: PCI FACT-COG Secondary outcomes: episodic memory (Grober and Buschke), attention (d2), executive functions and processing speed (Verbal fluency test, TMT), working memory and short-term memory (digit span WAIS-IV) | Primary outcome: no significant difference between groups on PCI improvement Secondary outcomes: compared to control groups significant improvement of PCI (p = 0.02), perceived cognitive abilities (p > 0.01) and working memory (p = 0.03) for group A | Improvement of cognitive complaints and working memory |
| Gehring et al., 2009 [48] | RCT Pre-intervention post-intervention 6-month follow-up | Adult patients with gliomasIntervention group: n = 66 Waitlist group: n = 69 | 6 weekly 2h-sessions of attention program (C-Car) and psychoeducation | On-site intervention with the supervision of a neuropsychologist | Attention (SCWT, DS, LDST, MST, TEA), verbal memory (WLT), executive function (CST, LF, BADS) and cognitive complaints (MOS CFS, CFQ), fatigue (MFI) | Cognitive tests: significant group differences for attention (p = 0.028) and verbal memory (p = 0.015). Cognitive complaints: significant group difference overtime for CFS total score, burden, and CFQ total score (p = 0.003) and mental aspects of fatigue (p = 0.049) | Improvement of cognitive complaints, attention and verbal memory |
| Hardy et al., 2013 [50] | Pilot study Pre-intervention Post-intervention 3 month follow-up | Survivors of childhood cancer Intervention group n = 13 Control group n = 7 | 5–8 weeks 25 sessions (Cogmed RM) | Home-based intervention with phone-based coaching support | Preliminary efficacy of the program (WASI, WRAML2, Conner’s rating scale, SERS) | Efficacy: significant group differences for working memory (p = 0.05) and parent-reported learning problems (p = 0.05) | Program feasible and Improvement of working memory and learning abilities |
| Kesler et al., 2013 [51] | Feasibility study pre-intervention post-intervention | Breast-cancer survivors Intervention group: n = 21 Wait list: n = 20 | 48 session (20-30 min) of EF program (Lumosity) over 12 weeks | Home-based intervention without supervision and reminder | Primary outcome: WCST (flexibility) Secondary outcome: EF (letter fluency test, BRIEF), verbal memory (HVLT-R), working memory (digit span), processing speed (symbol search) and depression (CAD) | Primary outcome: significant improvement of flexibility in the intervention vs. control group (p = 0.008) Secondary outcomes: Improvement in letter fluency and symbol search (p < 0.01) | Improvement of flexibility, letter fluency and symbol search |
| Maschio et al., 2015 [52] | Pilot study Pre-intervention After intervention 6-month follow-up | Brain-tumor patients Intervention group n = 16 | 1h/week 10 weeks RehabTr | On-site intervention With the supervision of a neuropsychologist | Cognitive improvement (MMS; TMT; frontal assessment battery; Raven Matrices; ROCF-Copy and recall; Clock Drawing test; Span forward and backward; 15 Rey-Osterrieth Word list, fluency test | Primary outcome: significant improvement of memory (p = 0.0017; p = 0.036) and fluency (p = 0.043) | significant improvement of memory and fluency after intervention and at 6-month follow up |
| Mihuta et al., 2017 [54] | Pilot study pre-intervention post-intervention 3-month follow-up | Cancer survivor Cancer intervention group: n = 13 Non-cancer intervention group: n = 21 Non cancer wait-list: n =17 | 4-week 2-h session (eRECog program) | Home-based intervention with reminder emails | Primary outcome: PCI FACT-Cog (cognitive complaints) Secondary outcome: other cognitive complaints questionnaires (BAPM, BADL, EORTC QLQ-C30, IADL), cognitive tests (WebNeuro), distress (K10), illness perception (BIPQ) and program satisfaction | Primary outcome: No significant interaction for PCI Secondary outcome: Significant improvement of cognitive complaints (BADL) and attention in the intervention group | High participant satisfaction and some improvements in subjective and objective cognitive functioning |
| Mihuta et al., 2018 [53] | RCT pre-intervention post intervention 3-month follow-up | Cancer patients Intervention group: n = 40 Waitlist group:n = 36 | 30–60 min sessions/week 4 weeks (eRECog program) | Home-based intervention with emails reminder 5 days after non-completion of the session and phone call after 3 mails reminder without answer. Encouragement mail after completion of the first module | Primary outcome: PCI FACT-Cog (cognitive complaints) Secondary outcome: other cognitive complaints questionnaires (BAPM, BADL, EORTC QLQ-C30, IADL), cognitive tests (WebNeuro), distress (K10), illness perception (BIPQ), fatigue (EORTC QLQ-C30) and program satisfaction | Primary outcome: No significant interaction for PCI Secondary outcome: Significant improvement of the prospective memory IADL score in the intervention vs. control group. No significant interaction for other variables | No significant group effect on cognition |
| Poppelreuter et al., 2008 [55] | RCT Pre-intervention Post intervention 6-month follow-up | Patients after HSCT Intervention group (NPT) n = 21 Intervention group (PC) n = 26 Control group n = 28 | 1 h/week 3–5 weeks Different training software | On-site intervention NPT = neuropsychological training group (max 8 participants) supervised by an occupational therapist PC = individualized computer-based training + individual coaching | Attention, memory (battery of standardized tests) and cognitive complaints questionnaires (EORTC; MFI; FEDA) | No significant results | No significant improvement |
| Von Ah et al., 2012 [56] | RCT Pre-intervention Post-intervention 2-month follow-up | Breast cancer survivors Memory training group n = 29 Speed of processing intervention group n = 30 Wait-list control group n = 29 | 10 1 h sessions over 6–8 weeks (Insight program) | On-site group intervention supervised | Primary outcomes: Objective memory (AVLT; Rivermead Behaviourall Paragraph Recall Test) and speed of processing (UFOV) Secondary outcome: Perceived cognitive functioning (FACT-COG); symptoms distress (CES-D; STAI-S; FACT-F) and quality of life (QOL-CS; SF-36) | Primary outcomes: significant improvement of immediate and delayed memory (p = 0.036, p = 0.013) at the 2-month follow-up in the intervention group vs. control group. Significant improvement of processing speed after the intervention (p = 0.007) and at the 2-month follow-up (p = 0.004) Secondary outcomes: significant improvement of perceived cognition (p ≤ 0.005) | Improvement of objective and perceived cognition |
| Wu et al., 2018 [58] | Pilot study Pre-intervention Post-intervention 8-weeks follow-up | Prostate cancer patients Intervention group (CCT) n = 40 Wait-list group: 20 | 1 h/day, 5 days/week for 8 weeks (BrainHQ) | Home-based intervention with e-mail reminders and weekly phone calls | Efficacy: Objective cognitive functioning (CNS Vital Signs); Self-reported cognitive functioning (PAOFI); Neurobehavioral functioning (FrSBe) | Significant improvement of reaction time in the intervention group vs. control group | Program feasible with some effects on reaction time |
| Zucchella et al., 2013 [57] | RCT Pre-intervention Post-intervention | Patients with brain tumor Rehabilitation group n = 30 Wait-list group n = 32 | 4 weeks 4 1 h sessions/week (training di riabilitatione cognitive; una palestra per la mente) | On-site intervention with direct training and metacognitive training supervised by two neuropsychologist | Cognitive functioning (MMS, digit span, Corsi′s test, RAVLT, PM47, FAB, TMT, ENPA) | Significant improvement in all the neuropsychological measures in the intervention vs. control group. | Significant cognitive improvement |
| Interventions | Memory | Attention | Executive Functions | Processing Speed | Subjective Cognition |
|---|---|---|---|---|---|
| Computerized-Cognitive Stimulation | |||||
| Becker et al., 2017 [49] | |||||
| Bellens et al., 2020 [43] | ✓ | ||||
| Bray et al., 2017 [44] | ✓ | ||||
| Conklin et al., 2015 [45] | ✓ | ✓ | ✓ | ✓ | |
| Damholdt et al., 2016 [46] | ✓ | ||||
| Dos Santos et al., 2020 [47] | ✓ | ✓ | |||
| Gehring et al., 2009 [48] | ✓ | ✓ | ✓ | ||
| Hardy et al., 2013 [50] | ✓ | NA | NA | NA | |
| Kesler et al., 2013 [51] | ✓ | ✓ | |||
| Maschio et al., 2015 [52] | ✓ | NA | |||
| Mihuta et al., 2017 [54] | ✓ | ||||
| Mihuta et al., 2018 [53] | |||||
| Poppelreuter et al., 2008 [55] | |||||
| Von Ah et al., 2012 [56] | ✓ | NA | NA | ✓ | ✓ |
| Wu et al., 2018 [58] | |||||
| Zucchella et al., 2013 [57] | ✓ | ✓ | NA | ||
| Computerized-Physical Activity | |||||
| Galliano-Castillo et al.,2017 [63] | NA | NA | NA | ||
| Gehring et al., 2020 [60] | ✓ | ✓ | ✓ | ✓ | ✓ |
| Howell et al., 2018 [61] | NA | NA | NA | NA | |
| Miki et al., 2014 [62] | NA | NA | ✓ | NA | NA |
| NA = not assessed |
| Publication | Study Design/Assessment | Participants | Intervention | Supervision | Outcomes/Tools | Results | Conclusion |
|---|---|---|---|---|---|---|---|
| Galliano-Castillo et al., 2017 [59] | RCT Pre-intervention Post-intervention 6-month follow-up | Breast cancer survivors Intervention group: n = 39 Control group: n = 37 | 3 sessions/week (90 min) over 8-week internet-based tailored exercise program | Home-based intervention with individual supervision through instant messages, video-conference sessions and phone calls | 6-min walk test (functional capacity) and ACT + TMT (cognitive tests) | Functional capacity: significant improvement in the intervention vs. control group for the 2 follow-up assessments (p = 0.001) Cognitive function: significant improvement on 1/5 of the ACT score in the intervention vs. control group for the 2 follow-up assessments (p < 0.05). No effect on TMT scores | Some improvement in functional performance and cognition |
| Gehring et al., 2020 [60] | RCT Baseline Post-intervention | Stable patients with grades II/III glioma Exercise group: n = 21 Control group: n = 11 | 6-month intervention with 3 aerobic sessions/week (20–45 min) | Home-based remotely coached intervention | Attention (SCWT-int; LDST; WAIS-R digit span; test of everyday attention), memory (VVLT; WMS-III verbal paired associates); executive function (CST-Shift; GIT letter fluency, GIT category fluency); cognitive complaints (CFS cognitive functioning scale; CFQ); fatigue, sleep, mood and QoL (MFI; PSQI; POMS; QLQ-BN20; SF-36) | Better post-intervention scores of the exercise group: attention, processing speed, verbal memory, executive function and cognitive complaints | Improvement in several domains of cognition and cognitive complaints |
| Howell et al., 2018 [61] | RCT Pre-intervention Post-intervention | Adolescent cancer survivors Intervention group, n = 53 Control group, n = 25 | 24 weeks web-delivered physical activity intervention | Home-based intervention without supervision | Physical activity (wGT3X-BT, ACTi Graph); fitness; general intelligence (vocabulary and visual-spatial construction WASI) and flexibility (Delis-Kaplan Executive Function System); quality of life (PedsQL) | No statistical difference between groups for mean change in weekly MVPA. No significant improvement in intervention vs. control group for cognitive scores | No significant difference between groups on cognition |
| Miki et al., 2014 [62] | Feasibility study pre-intervention post-intervention | Breast and prostate elderly cancer patients Intervention group: n = 38 Control group: n = 40 | 4-week 1/week on-site intervention (5min) with a bicycle ergometer | On-site intervention supervised by a therapist | FAB (executive function), BI+IADL (activities of daily living), FACT-G (QoL) | Significant effect of group, time and time x group for FAB score | Feasible intervention to improve cognition |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Binarelli, G.; Joly, F.; Tron, L.; Lefevre Arbogast, S.; Lange, M. Management of Cancer-Related Cognitive Impairment: A Systematic Review of Computerized Cognitive Stimulation and Computerized Physical Activity. Cancers 2021, 13, 5161. https://doi.org/10.3390/cancers13205161
Binarelli G, Joly F, Tron L, Lefevre Arbogast S, Lange M. Management of Cancer-Related Cognitive Impairment: A Systematic Review of Computerized Cognitive Stimulation and Computerized Physical Activity. Cancers. 2021; 13(20):5161. https://doi.org/10.3390/cancers13205161
Chicago/Turabian StyleBinarelli, Giulia, Florence Joly, Laure Tron, Sophie Lefevre Arbogast, and Marie Lange. 2021. "Management of Cancer-Related Cognitive Impairment: A Systematic Review of Computerized Cognitive Stimulation and Computerized Physical Activity" Cancers 13, no. 20: 5161. https://doi.org/10.3390/cancers13205161
APA StyleBinarelli, G., Joly, F., Tron, L., Lefevre Arbogast, S., & Lange, M. (2021). Management of Cancer-Related Cognitive Impairment: A Systematic Review of Computerized Cognitive Stimulation and Computerized Physical Activity. Cancers, 13(20), 5161. https://doi.org/10.3390/cancers13205161






