Simple Summary
Adolescent and young adult (AYA) cancer patients, those who are diagnosed with cancer for the first time at the age of 15–39 years, are recognized as a distinct population within the oncology community due to the unique challenges they face throughout their disease trajectory. The number of AYAs who develop cancer per year has increased over the last decades, and >80% is expected to survive beyond 5 years. However, this rapidly growing AYA cancer survivor population is also at increased risk of cancer- and treatment-related long-term and late effects. There is a need for research efforts to inform the survivorship care of this unique population. The aims of this overview paper are to describe the epidemiology of the AYA cancer survivor population, the current knowledge on long-term and late effects, challenges and models of AYA survivorship care, and future opportunities within research as well as healthcare.
Abstract
Worldwide, more than 1.2 million adolescents and young adults (AYAs; those aged 15–39 years) are diagnosed with cancer each year. Although considerable variability exists according to cancer site and stage of disease, the 5-year relative survival at the time of diagnosis has been estimated at >80% for all AYA patients with cancer combined. Extensive survivorship research in recent decades has focused on patients diagnosed with cancer as children (<15 years) and older adults (>39 years), yet few studies to date have reported outcomes specifically for patients diagnosed as AYAs. With increasing incidence and improving survival for many tumor types, leading to the majority of AYA patients with cancer becoming long-term survivors, there is a critical need for research efforts to inform the survivorship care of this growing population. This article describes the population of AYA cancer survivors according to their epidemiology and late and long-term effects, the challenges and models of AYA survivorship care, as well as future opportunities for research and healthcare.
1. Introduction
Adolescent and young adult (AYA) [1,2,3] cancer patients, those who are diagnosed with cancer for the first time at the age of 15–39 years, are recognized as a distinct population within the oncology community due to the unique challenges they face throughout their disease trajectory: from symptom recognition, diagnosis and treatment to disease monitoring, advanced care planning, and survivorship [1,4,5]. Over the last decades, the incidence of cancer in AYAs has increased [6], and >80% is expected to survive beyond 5 years [7,8], meaning that there will be ever more AYA cancer survivors with the potential of many decades of life still to live. This rapidly growing AYA cancer survivor cohort is at increased risk of cancer- and treatment-related long-term and late effects [9]. There is, however, a paucity of evidence delineating these medical and psychosocial sequelae of AYA cancer survivors that are highly needed to underpin the design of AYA survivorship care programs and to ensure the quality of their survival. Currently, AYAs face several challenges in the transition from active cancer treatment to surveillance and early and late survivorship care [10]. The survivorship definition we use is depicted in Figure 1. The aims of this overview paper are to describe the epidemiology of the AYA cancer survivor population, the current knowledge on long-term and late effects, challenges and models of AYA survivorship care, and future opportunities within research as well as healthcare.
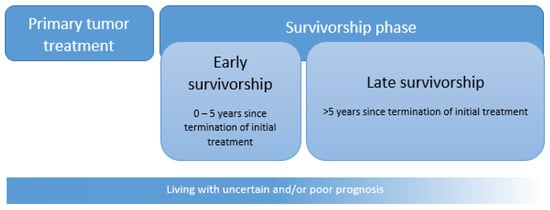
Figure 1.
Primary tumor treatment, early survivorship, and late survivorship, partially adapted from the American Cancer Society [11]. Survivorship starts at the termination of the initial treatment. In the case of long-lasting adjuvant hormonal treatment after initial adjuvant chemotherapy, survival time, in our definition, starts at the end of chemotherapy. During survivorship, survivors attempt to transition to the ‘new normal life’, which can be subdivided into early and late survivorship [12]. Early survivorship covers the first 5 years since the termination of initial treatment [excluding maintenance therapy] and late survivorship from 5 years onwards since the end of initial treatment. For patients with a poor prognosis at diagnosis, survivorship starts at diagnosis. They have to live with a life-limiting disease.
2. AYA Cancer Survivorship: Epidemiology
Approximately 1.2 million new cases of invasive cancer are diagnosed annually among AYAs [13], which is around 5% of all cancer diagnoses [7]. Over the last decades (2007–2016), cancer incidence increased slightly in all AYA age groups [14,15]. The distribution of different cancers varies substantially across the AYA age range. For example, pediatric malignancies, e.g., hematologic malignancies and brain tumors, are the most common cancers in adolescents (ages 15–19 years), while adult epithelial cancers, e.g., breast and colorectal cancers, are more common in older AYAs (ages 30–39 years), and there are also some cancers with the highest incidence among the AYA age group, e.g., germ cell tumors [7]. Triggered by the landmark report from the National Cancer Institute (NCI) in 2006, highlighting the dearth of improvements in survival rates for AYAs diagnosed with cancer compared with those diagnosed during childhood and adulthood—also called the “AYA gap”—the AYA group was defined as a high-priority group [1,16]. Fortunately, survival has improved more recently with a decline in overall cancer mortality during 2008 through 2017 by 1% annually [15]. The five- and ten-year relative survival (RS; defined as the ratio of observed survival in cancer survivors by the expected survival in a comparable general population matched on age and sex) of AYAs is now similar across age groups for all cancers combined [7,14,15]. Table 1 provides a brief overview of the age-standardized 5- and 10-year RS of AYA cancer survivors per cancer type, based on data from the American Surveillance, Epidemiology and End Results (SEER) 18 database of the NCI. Methods are described in Appendix A. Notable differences by tumor types and within some cancers are found in relative survival, reflecting differences in progress made, histologic subtype distribution, and available treatments. The distribution of tumor types among AYA cancer survivors has important consequences for the organization of AYA survivorship care, as risks for cancer recurrences, second primary malignancies, and treatment-related long-term and late effects will significantly impact supportive care needs, surveillance strategies, and follow-up care. In addition, despite the progress made for several cancers, there is also a group of patients living with an uncertain and/or poor cancer prognosis (UPCP). This includes patients undergoing established treatment with a life expectancy of 1 to 5 years (e.g., colorectal cancer stage IV), low-grade glioma survivors with a life expectancy of 5 to 10 years, and “new survivors” undergoing novel treatment(s) with uncertain prognosis (e.g., melanoma stage IV) [17]. Common challenges among AYAs with a UPCP include the loss of control of their life and disruption of developmental milestones, (unknown) effects of constant, aggressive, or novel treatments, a sense of social isolation (within the healthcare system and from peers), a constant balance between hope and risks, a lack of evidence-based prognostic information (e.g., due to a lack of focus on this specific patient group) and experiencing uncertainty about treatments (effectiveness and availability) [17]. These patients could potentially also benefit from AYA survivorship care programs.

Table 1.
Age-standardized 5- and 10-year relative survival (RS) by primary cancer type in adolescents and young adults diagnosed between 1998 and 2016 (SEER 18, Regs Research Data, Nov 2018 submission) in rank order of 10-year RS.
3. What Sets AYA Cancer Survivors Apart?
AYAs with cancer are distinct from the pediatric (<15 years) and the older adult (>40 years) cancer populations, not only with respect to their spectrum of cancer types, but also the biology of their cancers, their developmental status, their particular psychosocial needs, and the long-term complications of their cancer and treatment thereof [1,7,16,18]. The following aspects may all impact survivorship outcomes and have implications for the follow-up care of AYAs with cancer:
- The percentage of AYAs with cancer who carry pathogenic variants in genes that predispose to cancer is significant [18], with important consequences for an individuals’ surveillance strategy.
- There is insufficient awareness of cancer risk and symptoms among AYAs and healthcare professionals resulting in prolonged and complex diagnostic trajectories, negatively impacting quantity and quality of survival [19,20].
- Despite similar histological diagnoses, tumor genomics and biology can differ between AYAs and older adults, leading to different medical treatment strategies [21,22].
- The biology of the host may differ according to age, with potential distinct pharmacokinetics and pharmacodynamics between children and AYAs/adults and potential impact on therapy efficacy and toxicity profiles [18,23,24,25].
- In contrast to the emphasis put on late effects of treatment in pediatric cancer patients, for AYAs hardly any late effect clinics are in place, and the lasting (physical) impact of treatments at this age is only known to a limited degree [26].
- Adolescence, emerging, and young adulthood are complex, unique phases of life due to the many physical, emotional, cognitive, and social transitions [27]. A cancer diagnosis during the AYA life stage exacerbates typical developmental challenges and interferes with the attainment of important age-specific milestones [16], including identity-forming, establishing autonomy, responsibility and independence, finishing education and starting a career, obtaining romantic relationships and starting a family [27]. In addition, the type of informal caregivers may differ among cancer patients with varying ages and life courses [28]. The way in which AYA cancer patients adjust to their cancer experience might have life-long implications for the quality of their survival time [29,30,31,32,33].
4. What Do We Know about Long-Term and Late Effects among AYA Cancer Survivors?
Much of what we have learned about AYAs to date has been extrapolated from research on childhood and older adult cancer patient populations in disease- but not age-specific cohorts, e.g., breast cancer and Hodgkin’s [16,34,35,36]. However, there is still a dearth of literature on (long-term) adverse health outcomes in AYAs treated for other early-onset adult cancers, such as colorectal cancer, and AYA cancers, such as melanoma [37,38,39,40,41]. We summarize the most important literature according to the biopsychosocial model, which provides a comprehensive framework for the physical, psychological, and social issues caused by cancer (treatment) and the interactions between different issues.
4.1. Physical Issues
Which (long-term) physical issues develop mainly depends on the location of the primary tumor and the type of treatment received [42,43,44]. Compared to siblings and the general population with no history of cancer, AYAs are at greater risk of potentially life-threatening chronic medical conditions [43,44,45]. These long-term complications can lead to significant medical expenses and healthcare utilization [45].
4.1.1. Secondary Malignancies
AYA cancer survivors are at increased risk of developing secondary malignancies as a consequence of their first cancer diagnosis and its treatment [9,46,47,48,49]. For instance, survivors of Hodgkin’s lymphoma have, compared to the general population, a higher risk of developing secondary solid cancers due to treatments such as radiotherapy. This elevated risk can even persist for more than 25 years [50,51]. The observed increased risk of secondary cancers after Hodgkin’s lymphoma treatment has led to adaptations in treatment schedules, hence a reflection of the importance of survivorship research. Overall, well-known secondary malignancies for patients exposed to (high doses of) radiotherapy in certain areas or alkylating agents include, amongst others, breast, lung, stomach, and colorectal cancer, and leukemia. A better understanding of the exposure-related risk of other (newer) treatments, other concurrent risk factors, and appropriate screening measures are needed for secondary malignancies risk management among AYA cancer survivors [9,47,52].
4.1.2. Cardiovascular Disease
Cardiovascular disease (CVD; including, amongst others, vascular disease, coronary artery disease, congestive heart failure, and cerebrovascular disease) can present during treatment as well as during late survivorship and contributes to increased risks of both morbidity and mortality [9,53]. Multiple risk factors are involved in the development of CVD, including treatment-related factors (e.g., high-dose anthracycline chemotherapy, heart within radiotherapy field), preexisting risks factors as well as secondary factors, such as one’s behavior [9,53]. Long-term survivors of testicular cancer who are treated with chemotherapy (e.g., cisplatin-containing) are at increased risk for cardiac events and may develop an unfavorable cardiovascular risk profile, including but not limited to hypertension and overweight [54,55]. The risk of developing CVD is twofold higher among AYA cancer survivors compared to their matched healthy controls and increases with older age [9].
4.1.3. Endocrine Dysfunction
In AYAs, an increased risk of endocrine dysfunctions can occur due to changes in one’s metabolism or dysfunction of the gonads or thyroid, for example [9,56]. Thyroid dysfunction (e.g., hypo- and hyperthyroidism) is a common late effect for patients undergoing radiotherapy given the radio-sensitivity of the thyroid gland, of which the risk can persist even 20 years post-treatment [9]. Diabetes is another endocrine dysfunction, which frequently occurs as a treatment-related late effect and, besides thyroid dysfunction, is known as a leading cause for hospital visits [9,57]. Compared to the general population, AYAs have a 29% increased risk of developing diabetes. Besides lifestyle factors (e.g., poor nutrition, physical inactivity), treatments including abdominal radiotherapy and chemotherapy can contribute to an increased diabetes risk as well (e.g., insulin resistance, metabolic syndrome, changed pancreatic function) [9].
4.1.4. Neurocognitive Deficits
Cancer (treatment) related neurocognitive deficits include problems regarding attention, memory, and executive functions [9]. As the brains of many AYAs are still in development, they are especially vulnerable to cognitive dysfunction [58]. These cognitive issues may eventually lead to problems in everyday life, including completing education or maintaining employment. Subsequently, this may impact psychological well-being. AYA cancer survivors have, compared to the general population, higher rates of cognitive dysfunction [59]. Especially AYAs with primary brain tumors or metastases treated with cranial radiotherapy and chemotherapy directed at the central nervous system should be considered at risk and screened for impaired neurocognitive outcomes [9].
4.1.5. Fertility
Fortunately, many AYA cancer survivors were able to have children without the need for fertility preservation, and, over time, more attention has been paid to the potential impact of cancer (treatment) on fertility. However, cancer treatments (chemotherapy, surgery, or radiotherapy) still have the potential to destroy reproductive cells and can place both women and men at risk of fertility issues, depending on dosage and field, and age at exposure [9,36,41,46,60,61,62,63]. Issues include azoospermia among men and acute ovarian failure, premature ovarian insufficiency, and premature menopause among women [9,60,61]. Although some patients of younger ages may feel as if fertility is not relevant to them yet, it is important though to discuss the possibility of infertility, as it may have a huge impact later in life [16,64]. Unfortunately, despite the availability of fertility preservation options (e.g., embryo, oocyte, or ovarian tissue cryopreservation and hormonal therapies for women, or testicular tissue cryopreservation and sperm banking for men), the understanding of fertility risks may either not be complete or accurate, or the experience of discomfort among healthcare providers or patients may lead to the start of treatment without fertility preservation or fertility concerns among patients [16,31,46,65,66,67,68]. From the perspective of AYA survivors and their families, common barriers to fertility care include the lack of high-quality information regarding fertility risk and preservation, concerns regarding treatment delay, and concerns about the health of the AYA and their offspring. In addition, healthcare providers (HCPs) might make assumptions regarding the fertility interest of the patient that may be incongruent with the perspective of the AYA [68,69]. The use of a risk stratification model, including the risk for infertility in AYA cancer survivors and improving access to fertility preservation services, amongst others, may improve this process [9].
4.1.6. Sexual Dysfunction
Erectile dysfunction, ejaculation issues, vaginal atrophy, and changes in sexual desire and orgasm sensitivity may all be consequences of, for example, changes to blood vessels or nerves in the genital area due to treatments such as surgery and radiation. Chemotherapy might alter hormone levels leading to early menopause (including loss of elasticity, shrinking, and vaginal dryness). Altogether, sexual dysfunction can result in intercourse not being comfortable or interference with one’s interest or joy in sex [70]. Besides these direct effects, there may also be indirect treatment effects (e.g., fatigue) or changes in one’s physical appearance or self-esteem that may negatively impact one’s sexual functioning and (their partner’s) sex life [31,71]. A study among Swedish AYAs, aged 15–29 and at least 1-year post-treatment, showed less satisfaction with sexual functioning among AYA cancer survivors compared to their healthy peers [72]. Although AYAs express a need for information regarding sexual health, this is not always recognized by HCPs [16,73]. They may feel discomfort or not confident in providing appropriate sexual healthcare. Furthermore, they may have misconceptions regarding AYAs’ sexual behaviors or think AYAs do not want to talk about it [16,73]. Even though AYAs report positive outcomes when discussing sexual health with their HCP, there are still no standard sexual health practices addressing AYAs’ needs yet [16].
4.1.7. Body Disfigurement
Physical appearance is extremely important for AYAs, and related dissatisfaction is common [72,74]. AYAs are characterized as being very conscious of their body and appearance, which adds up to the vulnerability of this population. Especially young AYAs whose bodies are still in development may be more affected by treatments such as surgery or radiotherapy [41]. Disfigurement, the state of having one’s appearance deeply and persistently harmed medically due to cancer treatment, may include, among others, amputation (e.g., breast cancer or sarcoma survivors), scars, and chronic skin changes (e.g., head and neck cancer survivors), having an ostomy (e.g., colorectal cancer survivors) and having prosthesis [72,74,75,76]. Disfigurement negatively impacts one’s body image [72,74], which in turn may lead to feelings of insecurity and alienation from peers [31,71].
4.1.8. Physical Condition
Treatments including radiation and chemotherapy and, indirectly, treatment-related effects as shortness of breath and fatigue may influence survivors’ level of physical activity and lead to a poor physical condition [77]. A Swiss study among AYAs observed significantly lower physical health compared to the general population [30], and Tai and colleagues showed significantly more AYA cancer survivors being obese [56]. Furthermore, in an American study, Ketterl and colleagues showed increased odds of physical impairment of work-related tasks among patients of several cancers, including sarcoma and central nervous system cancers, treated with surgery [78].
4.2. Psychological Issues
Most important psychological survivorship issues include psychological distress, posttraumatic stress and fear of cancer recurrence [41,46,59,66,77,79,80,81]. Psychological distress is known to be common and significantly greater among AYAs than older adults: AYAs understand the severity of the situation but may lack sufficiently developed distress tolerance, emotion regulation, and/or interpersonal effectiveness skills [41,77,82,83,84]. This stresses the importance of delivering developmentally appropriate support, as AYAs try to accomplish key developmental tasks but at the same time may have different cognitive and emotional developmental levels than older adults [41]. Compared with healthy matched controls and siblings, female AYA cancer survivors have a higher risk of developing depression and anxiety [43,77]. Results of the AYA HOPE study indicate that more than 50% of AYAs who were in need of mental health services did not receive these [85]. Kwak and colleagues reported increased psychological distress levels among AYAs compared to population norms at the time of diagnosis and after 12 months of follow-up while transitioning into survivorship [86]. Unfortunately, AYAs may delay the reporting of symptoms of distress to HCPs, which may be due to their coping style or fear of stigmatization [31]. In a Canadian study among AYA cancer survivors, help for emotional concerns was not sought because survivors either did not want to ask for it, felt embarrassed or thought nothing could be changed because someone had told them it was normal [80]. A significant minority (40%) of the Canadian AYAs struggled to cope with everyday life challenges. A lack of age specific services and interventions may hinder AYAs in trying to access support services [80].
Furthermore, research has shown that fear of recurrence is more prevalent among AYAs compared to older adult survivors [87,88]. AYAs want to learn how to handle and manage the fear of recurrence, which is shown to be an unmet information need [88]. In general, cancer patients with high fear of recurrence also experience significant psychological distress and have increased use of health services, while others change their behavior by, for example, avoidance or excessive self-examination [89]. Self-examination behaviors may be a signal for a survivorship care need as the AYA needs to know which signs and symptoms to track and when to contact an HCP. Compared with healthy matched controls and siblings, AYA cancer survivors also have a higher risk of developing posttraumatic stress [43,77]. In the end, the health-related quality of life (HRQoL) of AYA cancer survivors may be significantly impacted [33,90]. In an Australian survey, both AYAs and parents self-reported elevated levels of general distress and posttraumatic stress symptoms: rates of emotional distress were comparable between parents and AYAs [91]. This underscores the importance of being aware of the impact a cancer diagnosis may have besides solely on the patient, such as, for example, on caregivers.
Despite the negative psychological survivorship issues possibly experienced after cancer (treatment), there are also AYAs experiencing positive outcomes, such as resilience, post-traumatic growth, and appreciation for life. Although its definition varies, resilience is often defined as the ability to cope with the negative effects of a stressful experience such as cancer [29,92]. Factors that can either contribute to or inhibit resilience include stress and coping, goals, purpose and planning, optimism, gratitude and meaning, and connection and belonging [92]. The balance of these factors can be shifted in case of specific experiences or moods, for example, but when in balance, resilience may empower AYAs and lead to improved psychosocial outcomes [29,92]. Resilient AYAs are less likely to report unsatisfied counseling needs compared to those with less resilience [79]. Post-traumatic growth (PTG) is described as benefit finding—a change in view of life, feeling stronger or more confident. In other words, there is a positive psychological change as a result of one’s cancer experience [29,93]. PTG can be seen in relationships, perspective on life, or one’s self, or in spiritual beliefs [29]. Enduring stress is negatively associated with PTG, while social support is positively associated with PTG [29]. Both resilience and PTG are positively associated with satisfaction with life and HRQoL [29]. In qualitative studies, most AYAs report at least some resilience or PTG, although exact prevalence rates are lacking due to the heterogeneity of studies, definitions, applied theoretical frameworks and used tools [29].
4.3. Social Issues
A patient’s social role (fulfilling a recognized position in society, e.g., parent, employee, student) may alter due to cancer (treatment): participation in social activities may be limited and social maturation may be disrupted [32]. Due to the long-term effects of treatment, AYAs may find it difficult to make or maintain social contacts and an active and independent life [32,41,94]. Social functioning has been shown to be more challenging for AYAs compared to an age-matched population [32]. Missing out on life experiences such as going to college, dating, becoming independent, or having children can all lead to feelings of isolation and alienation, which is common among AYAs as it sets them apart from their peers while becoming more dependent on parents or caregivers [41,71,95]. AYA cancer survivors indeed perceive themselves as being different from their peers who had no cancer diagnosis [32,96], and as their healthy peers may not be familiar with cancer, this effect may be amplified [97].
4.3.1. Education, Employment, and Financial Challenges
Although many AYAs continue or return to their education during or after cancer (treatment), as is the case for employment, educational trajectories may be disrupted due to being absent or unable to complete exams, amongst others [98,99], which may have a crucial impact on AYAs’ career development [100]. Although the impact of cancer (treatment) on education shows varying study results [56,101,102], AYAs indicate receiving inadequate educational support during their treatment and are in need of support with returning to school [31,103]. In a qualitative study among Danish AYAs, aged 15–25 at diagnosis, results indicate that AYAs experienced problems regarding their return to secondary or higher education due to the consequences of treatment, misunderstanding from their peers, and academic/municipal system barriers (e.g., complex navigation, miscommunication) [104]. Sources of support, such as parents, family members, and guidance counselors, were helpful in alleviating the burden of these problems [104].
In addition to educational challenges, returning to work after being absent due to treatment is also known to be a challenge for patients in general, while work addresses their financial needs, makes them feel productive, and suggests recovery and return to ‘normal life’ [73,97]. Compared to their peers, AYAs feel left behind in their employment trajectories [31]. Significantly more AYAs report being out of work or unable to work (24%) compared to their healthy peers (14%) [56]. In a study among Canadian AYA cancer survivors, respondents most often did not seek help for practical concerns such as returning to school or work because they did not want to ask for help or were either not aware of available services or where to go for these [80]. In some cases, AYAs experience discrimination from employers [31].
Interruptions in school or work may lead to significantly more distress [105] and eventually, in the long term, influence career opportunities and financial status [41,46], while this latter can already be impacted earlier on due to insurances and healthcare expenses [95]. Adding to this, AYA cancer survivors are known to face higher medical expenditures compared to their healthy peers [46,56,73] and are more likely to face financial problems than older cancer patients [101,106]. These problems may lead to loss of control and more (financial) reliance on others, including partners or parents [31,66,97,98]. Some AYAs may also avoid routine medical care due to these financial barriers [46,107]. However, given the nature of heath care provided (governmental primarily in Europe and Canada; private insurance and Medicaid in US), much of the access to care and financial toxicities will differ between continents and countries, although there will also be overlapping issues.
Important to note is that a change in education or employment status can be perceived both negatively and positively as some AYAs may deliberately chose for the change as their perspective on life has changed due to the cancer experience [108,109]. Future plans are re-evaluated due to the focused attention one has regarding purpose and reprioritized goals [99,109]. Vetsch and colleagues report that AYAs’ education or vocation can be of equal or even greater importance to them after their diagnosis than before. Some vocational goals of AYAs are directly informed by the experience of their own cancer diagnosis [109].
4.3.2. Relationships
A cancer diagnosis affects more people than only the patient him/herself, which can either create a distance or enhance a relationship [110]. AYAs report a need for information regarding communication with others if they felt cancer had impacted their close relationships [111]. Kirchhoff and colleagues reported fewer AYAs being married, and young adult cancer survivors were more likely to divorce or separate from their partner than those without a cancer history (a 77% higher risk of divorce or separation) [112]. For those who are married or in a relationship, a cancer diagnosis may not only affect the relationship in its entirety, the partner may also experience a change in role functioning and future plans may be disrupted, and feelings of distress may arise. Within a study among breast cancer survivors aged 40 years or younger, the partnership was suggested to be an important source of support, which was associated with greater HRQoL levels [113]. In addition, starting a new relationship with a partner was considered challenging, due to feeling ‘abnormal’, fertility issues, low self-esteem, or a negative body image [110,114,115].
While there may be a negative impact on relationships due to the cancer experience, some AYAs also report a positive change, including more meaningful interpersonal relationships [93,99]. The whole cancer experience was beneficial for the relationship as it strengthened the relationship. A study by Bellizzi and colleagues showed that most AYAs experienced a positive impact on relationships with their spouses, parents, and siblings [99].
Altogether, the findings of these studies highlight the urgent need to better understand, screen for, and risk stratify, prevent and/or treat the negative long-term and late effects [16,41,43,64,73,116,117]. Many cancer-related sequelae experienced by AYAs have complex etiologies involving multiple overlapping mechanisms, making them difficult to prevent and treat [16,66,118]. This complexity, however, together with AYAs’ overall long-term survival [119], creates a strong rationale for exploring multidisciplinary (e.g., sexologist, occupational physician, (neuro) psychologist) AYA survivorship care, which includes prevention and treatment strategies for late effects.
5. Challenges and Models of AYA Survivorship Care
In many parts of the world, AYAs with cancer faced disparities of care as they were poorly served (not patient-centric) by the traditional dichotomy of pediatric and adult oncology services [90]. Fortunately, this gap in unmet care needs and poor outcomes of AYAs has been recognized, which led to the development of a new medical discipline called ‘AYA oncology’ [26] and the start of dedicated AYA care programs in some countries [12,25,73,120,121,122,123,124,125]. Overall, several essential components of AYA care programs are described in the literature, including (1) patient focus, (2) multidisciplinary approach, (3) cooperating pediatric and adult expertise, (4) staff education and training, (5) dedicated AYA space, (6) patient and family advocacy, and (7) research and clinical trial availability [12,25,123,126,127,128,129]. Although countries handle different age ranges for their AYA programs (varying from 15–25 years in Australia to 15–39 years in the United States and 18–39 years in the Netherlands), which may have consequences for the type of care provided, the patient focus remains an essential part in AYA care [12,123]. The multidisciplinary team (MDT) approach, including clinicians, clinical nurse specialists, and psychosocial/allied healthcare professionals, benefits AYAs due to the complexity and variety of care (needs) [125]. As referral pathways and services may not be directly available or visible to AYAs, who are often inexperienced in navigating the healthcare system, the help of healthcare teams are of great importance to guide AYAs through the system. Facilitating the cooperation between pediatric and adult medical oncologists for relevant tumor types is of importance as the best treatment regimen (pediatric or adult) needs to be chosen [12,123,125,127,130,131,132]. The benefits of such collaborative effort have been demonstrated for young AYA acute lymphoblastic leukemia patients, which showed superior survival outcomes when treated in a pediatric cancer setting compared to an adult cancer setting [133]. In addition, educating staff is of importance as HCP may lack AYA specific experience, which may affect the provision of care to this unique population [12,123,128]. Up till now, there is no optimal survivorship care model, and many differences (between and within countries) may exist, leaving much room for improvement and still a lot can be learned from each other (e.g., by benchmarking). Despite the emergence of AYA care programs, there is a limited number of programs with survivorship elements [1,25,73,77,134,135,136,137].
5.1. How Is Survivorship Care Organized for Children and Older Adults?
5.1.1. Survivorship Care for Children
Pediatric cancer care tends to be family-centered, in which the child, parents, and physician are involved in the decision-making process [25]. Holistic, multidisciplinary care for pediatric cancer patients is often provided centralized in pediatric cancer centers, where both treatment and follow-up care are provided [25]. In general, pediatric cancer care is relatively well resourced, with overall a large amount of time available per patient and with a high staff/patient ratio [126]. As most pediatric cancer patients are treated intensively, they are at risk for early and/or late adverse medical and psychosocial effects. The Childhood Cancer Survivor Study showed that 62% of childhood cancer survivors had any mild/moderate chronic condition 5 to 14 years after diagnosis, which increased to 81% at 25 to 36 years after diagnosis. In addition, 27% of childhood cancer survivors had any severe/life-threatening/disabling chronic condition 5 to 14 years after diagnosis, which increased to 42% at 25 to 36 years after diagnosis [138]. Most pressing problems include treatment-related second cancers, endocrine disorders (hypothalamic-pituitary dysfunction, primary thyroid dysfunction, metabolic syndrome, decreased bone mineral density) [139], and other coexisting medical conditions typically depending on treatment-specific factors [34,140,141,142]. The large body of knowledge about cancer (treatment) related effects among childhood cancer survivors has led to exposure-related and risk-based long-term follow-up guidelines specific for this group of survivors, made available by the Children’s Oncology Group [143,144]. Different models of survivorship care are available for providing long-term follow-up care for childhood cancer survivors: cancer center-delivered care (high-risk survivors), shared care between treatment hospital and local hospital or primary care, and primary care physician-led/community-based follow-up (low risk survivors) [145].
5.1.2. Survivorship Care for Older Adults (>40 Years)
In contrast to childhood cancer care, adult models of cancer care are more patient- and disease-focused, in which the patient typically makes the decisions in consultation with the physician. Psychosocial support is not always part of standard care, thus being offered most often when problems have already arisen [12]. The staff/patient ratio is much lower compared to pediatric oncology, and care is dispersed among many departments and centers [12,126].
Cancer survivorship care for late effects in older adults is tumor-specific. Which long-term and late effects arise and how often depends on patient and cancer (treatment) characteristics [146]. Older adult cancer survivors have extensive guidelines available, just as pediatric survivors, through the National Comprehensive Cancer Network (NCCN) [147,148]. The guidelines for survivorship provide recommendations regarding screening, evaluation, and treatment, as well as preventive health (e.g., healthy lifestyles) and late and long-term effects (e.g., anxiety, lymphedema).
The Institute of Medicine (IOM) introduced survivorship care plans (SCPs) in 2006 as a possible solution to the lack of coordination of care between active treatment and long-term survivorship. According to the IOM, survivorship should be recognized as a distinct phase in cancer care in which survivors’ concerns should be addressed [118]. SCPs are aimed to inform, support communication, and facilitate self-care of the cancer survivor regarding their cancer diagnosis, treatment, and follow-up [149,150]. It provides an overview of the survivor’s diagnosis and treatment, including follow-up schedules, recommendations for early detection, and management of treatment-related effects and other health problems [118,151]. Despite widespread effort, the use of SCPs in practice remains suboptimal, and evidence regarding their impact on survivors’ outcomes is limited [150,151,152,153]. Hill and colleagues performed a systematic review and meta-analysis of care plan outcomes suggesting that SCPs seem feasible and can have a positive impact on HCPs’ knowledge (e.g., regarding late effects, survivorship care), but there is a lack of evidence that SCPs have an impact on the patient-reported outcomes of survivors [151]. However, whether this is due to SCP ineffectiveness or issues regarding implementation, for example, remains yet unclear. Nevertheless, the American College of Surgeons, for example, no longer requires SCPs to maintain institutional accreditation.
5.2. Challenges for AYA Survivorship Care
With regard to survivorship care, AYAs again fall in between the dichotomy of risk-based age-centered follow-up for childhood cancer survivors and disease-centered follow-up for adult cancer survivors, both with thorough survivorship guidelines. As a small sub-set of the overall adult cancer survivorship population, AYAs have unique needs relating to their age, developmental stage, and potential years of life lost to disability or sub-optimal health and well-being. AYA cancer survivors have many unaddressed concerns when they transition out of active cancer treatment, including the already described long-term and late effects from treatment, ongoing psychosocial issues, and navigating follow-up care [118,137]. Factors hindering the reception of survivorship care on a patient level include, among others, the patient’s poor knowledge of cancer history and risk of late effects, reluctance to return to the hospital, and logistical reasons [12,25]. When medical treatment is over, the support delivered during treatment may diminish as there will be less contact between the healthcare team and the patient. Some patients may not have access to specialized services, as they remain with their treating specialist to obtain their follow-up, focusing primarily on relapse detection [46]. In addition, many AYA survivorship issues are still poorly understood and possibly neglected [16,66,73,118,137,154]. Because both HCPs in hospitals and general practitioners (GPs) only see a limited number of AYAs in their practice, they are often unfamiliar with AYA-specific issues and have limited expertise in AYA specific care [12,46,118,136]. Being unfamiliar with this patient population may lead to the provision of suboptimal follow-up care. All things considered, at this moment in time it is questionable which survivorship model is best suited for use within the AYA cancer survivorship population and who should be responsible for this follow-up care.
5.3. Models of AYA Survivorship Care and Services Needed
A recent report from CanTeen Australia described the transition into survivorship when treatment is over as a complex process, which should ideally be supported by multiple services addressing the care needs of AYAs [77]. These services should focus on: (1) education about cancer (treatment) and related effects, (2) surveillance, screening, and treatment for medical, long-term, and late effects, (3) psychosocial support for reintegration, appropriate for the developmental phase of the AYA, (4) peer support (5) and having access to legal and/or financial support. They are currently integrating these elements in their AYA programs.
Nevertheless, maintaining the provision of care may be challenged by the wide variety of hospitals where AYAs are treated and the high mobility of this population [10]. However, as AYA survivors may have a long life ahead of them while potentially suffering from the biopsychosocial issues described earlier, it is of utmost importance to establish long-term AYA survivorship care for AYA survivors, regardless of where initial treatment took place. The NCCN developed clinical practice guidelines for AYA oncology addressing AYA specific issues and management considerations, recommended interventions, and education for HCPs [10,41]. In addition, different risk-based long-term survivorship models of care are available, which involve a screening, surveillance, and prevention plan based on the characteristics of the AYA survivor. Three AYA survivorship models of care highlighted by the NCCN include: (1) cancer center follow-up, led by an oncology team or a center of expertise, (2) primary care follow-up provided by the primary care provider, nurse practitioner or family practice physician, and (3) shared-care follow-up, led by a team of HCPs including primary care provider and oncologist. Which survivorship model is best to apply depends on several factors such as available resources [10,41].
5.4. Examples of AYA Survivorship Care
Fortunately, there are some great examples of AYA survivorship care. The Princess Margaret AYA program at the Princess Margaret Cancer Centre in Ontario, Canada, is one of the programs that integrated survivorship care as a core element [73,155]. Within their AYA program (those aged <39), concerns related to both cancer as well as ‘regular life’ are addressed by a clinical nurse specialist (CNS) who connects the AYA with the right services and resources needed. AYAs can meet the CNS either through self-referral or referral by an HCP: the latter can directly refer to services or to the AYA program. After gaining insight into the patient’s needs, the CNS provides teaching and resources to address these needs, also on a longitudinal basis when needed. Resources focus on but are not limited to financial support, fertility, exercise, nutrition, psychosocial support, sexual health, and advanced cancer. Both the comprehensive assessment as well as the survivorship treatment plan are tailored to the individual. The survivorship strategy includes an MDT, disease site-specific care plans, and the provision of both information and education by the CNS. Survivorship care is built on self-management principles and provides comprehensive, ongoing care across the age spectrum [73]. Recently, the dedicated AYA program was evaluated, after which the incremental added benefit of the program was declared in several key domains, including information provision regarding fertility, physical appearance, work, and school [156]. The essence of the program was about support, education, triage, and navigation. AYAs were satisfied with the offered supports, and even when they were very satisfied with their primary team, the program could still be of added value. Over time, more AYA consultations took place, showing an increased awareness and use of the program. This all may be the result of the program having identified key gaps in resources and care in AYA specific domains and having time invested in identifying AYA specialists, developing peer support networks, and creating resources addressing fertility issues, for example.
6. Future Directions
6.1. AYA Cancer Survivorship Research
It is key to (1) pool different types of data (patient-reported outcomes (PROs), clinical and biological data) across organizations and/or countries and (2) to create representative cohort studies, including all AYA ages and diagnoses, in order to make improvements in the AYA oncology field [3,10,46,157,158]. The results of these big data approaches, possibly linked to national registry databases, provide insight in subgroups of AYAs who are at risk of impaired outcomes and why, and may form the basis for further development and promotion of AYA cancer survivorship care, including widely available evidence-based AYA healthcare programs and guidelines. Eventually, it may evolve national policies to improve AYAs’ outcomes [77,157].
ESMO/SIOPE’s AYA Working Group and Salsman and colleagues both expressed several research barriers that need to be addressed first in order to compare and improve AYAs’ outcomes, including, amongst others, a lack of AYA-specific outcome measures [18,159]. Salsman and colleagues highlight, for example, the need for AYA-specific measurement tools that are valid, reliable, and cross the full AYA age spectrum in order to perform research and gain insight into the needs, issues, and health status of AYA cancer survivors [159]. Defining a standardized core outcome set with PROs and clinical outcomes, specifically for AYAs, may lead to the collection and report of AYA-specific standardized outcomes, which will enable the comparison of outcomes across different levels (e.g., patients, studies, countries). The input of AYAs themselves is of importance to compose core outcome sets and develop AYA-specific outcome measures that are relevant to them, take into account their developmental phase, and target their needs [137,160]. Table 2 provides a summarizing overview of the key points of AYA-specific survivorship research.

Table 2.
Key points of AYA-specific survivorship.
6.2. AYA Cancer Survivorship Care
Research findings can subsequently provide evidence-based input for improving AYA cancer survivorship care, including:
- Leading to improved information provision and communication among AYA cancer survivors and HCPs. Although the use of SCPs among cancer survivors in general was limited, they may serve their purpose among AYA cancer survivors, as Hydeman and colleagues highlight the desire of AYAs for more information regarding late and long-term biopsychosocial issues with which they are dealing [118]. Many AYA cancer survivors do not adhere to recommended tumor-specific survivorship care, which may be due to poor knowledge of the possible late effects, treatment, and health maintenance, underinsurance, costs, or reluctance to go back to the hospital [12,25,118]. AYAs should be empowered to assume ownership for their care and to have support for personal transitions (e.g., school, employment, and family planning) and practical issues (e.g., insurances and finances) [43]. As there is a lack of communication around AYA-specific issues, the SCP as part of standard care may facilitate key information provision [118]. An SCP may be beneficial for AYA cancer survivors to transition back to ‘normal life,’ e.g., by providing education and information regarding their treatments’ long-term and late effects and lifestyle recommendations [12,126]. It is, however, important to tailor SCPs to the developmental phase of AYAs [118,161].
- Gaining knowledge about specific (sub) groups at risk may lead to the development of risk-stratified AYA survivorship programs, guidelines, and surveillance strategies. Within pediatric oncology, survivorship care has been associated with improvements on both the patient level (e.g., improved patients’ knowledge regarding treatment and risks of late effects) as well as on a system level (e.g., fewer emergency room visits, hospitalizations, and better surveillance practices) [149]. The potential to mitigate the severity of late effects with early detection and management through survivorship care programs offers a great financial opportunity for healthcare systems [155]. A core outcome set, as described previously, can promote the development of quality care standards and guidelines and improves AYA-specific healthcare. Performance indicators, which can be part of an AYA-specific core outcome set, can improve quality of care but can also serve as a benchmark to achieve outcome-improvement goals and provide the information needed to make policy-related decisions and resources allocation. In Canada, besides an AYA-specific survivorship program, AYA-specific system performance indicators have been established by a national, multidisciplinary panel of AYA oncology experts [73,155]. Identifying system performance indicators specifically for AYAs may be applied internationally to monitor, evaluate, and benchmark the progress of (inter)national cancer care programs and institutions. Adequately resourced AYA care programs are, on a system level, more sustainable when ensured by a governmental policy [12,123,137], with a clear model of care with key performance indicators [124,126,162]. Table 2 provides a summarizing overview of the key points of AYA-specific survivorship care.
7. Conclusions
There are significant costs of being an AYA cancer survivor related to the individual and society. We are at the beginning of a journey to understand how to provide the best surveillance, and follow-up care that will enable HCPs and empowers AYA cancer survivors to (1) minimize the risk of adverse long-term and late effects and (2) mitigate the multiple factors that potentially negatively affect subsequent morbidity and mortality. Special attention should be paid to the unmet needs of AYAs living with an uncertain and poor prognosis [17]. Although lessons can be learned from the published evidence in pediatric and adult oncology survivors, not all are applicable to AYAs. We need to gain insight into which specific subgroups of AYAs are at risk of certain survivorship issues with the help of AYA specific research, improve the information provision and communication to create awareness among AYAs and HCPs, and empower them to act upon it. For this, international collaborative research efforts are needed, wherein diverse data are pooled, to inform the development of AYA cancer survivorship care programs, come to specific AYA cancer survivorship guidelines, and develop performance indicators to evaluate (future) care programs. All these efforts are needed to ensure the quality of survival for AYAs with cancer.
Author Contributions
Conceptualization, S.H.M.J., O.H. and W.T.A.v.d.G.; methodology, S.H.M.J., O.H. and W.T.A.v.d.G.; software, S.H.M.J. and D.J.v.d.M.; validation, S.H.M.J. and D.J.v.d.M.; formal analysis, S.H.M.J. and D.J.v.d.M.; investigation, S.H.M.J. and O.H.; resources, S.H.M.J. and O.H.; data curation, S.H.M.J. and D.J.v.d.M.; writing—original draft preparation, S.H.M.J. and O.H.; writing—review and editing, S.H.M.J., O.H., D.J.v.d.M., E.M.-H. and W.T.A.v.d.G.; visualization, S.H.M.J.; supervision, O.H. and W.T.A.v.d.G.; project administration, S.H.M.J., O.H. and W.T.A.v.d.G.; funding acquisition, O.H. All authors have read and agreed to the published version of the manuscript.
Funding
O.H. and S.H.M.J. are supported by a VIDI grant from the Netherlands Organization for Scientific Research (VIDI198.007).
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
Appendix A. Methods Used to Calculate the Age-Standardized 5- and 10-Year Relative Survival
SEER*Stat version 8.3.8 and STATA version 16.1 were used to collect and analyze the data. Relative survival estimates were determined with the –STRS– command in STATA and using the Ederer II method [163]. The United States Annual Life Tables: 1970–2017 were used for general population expected survival probabilities [164]. Outcomes were age-adjusted to the International Cancer Survival Standard 3 [165]. AYAs aged 15–39 years at their first malignant cancer diagnosis between 1998 and 2016 were included. Cancer types were classified based on the site recode International Classification of Diseases for Oncology-3 (ICD-O-3)/WHO 2008 (see Appendix B for exceptions), including local, regional, and distant staging based on the SEER Summary Stage 2000 (1998+). Patients with more than one cancer diagnosis were not included. Those with missing follow-up data were removed from the database (n = 175). Year of death, emigration, or the year 2016 determined end of follow-up, whichever came first.
Appendix B. Tumor Type Categorization of Table 1 Explained, Based on Site Recode ICD-O-3/WHO 2008 and ICD-O-3 Histology/Behavior, Malignant
a Includes: ICD-O-3: 8240, 8241, 8244, 8249.
b Includes: ICD-O-3: 8240, 8241, 8249.
c Includes: Hodgkin Extranodal and Nodal.
d Includes: Sexcord stromal tumors (ICD-O-3: 8590, 8593, 8600, 8620, 8622, 8631, 8632, 8634, 8640, 8650, 8670), Sarcoma (ICD-O-3: 8800, 8803, 8805, 8806, 8810, 8851, 8854, 8890, 8896, 8900, 8901, 8910, 8920, 8931, 8933, 8935, 8950, 8951, 9044, 9120, 9260), Germ cell tumors (ICD-O-3: 8052, 8070, 8071, 8240, 8243, 9060, 9064, 9065, 9070, 9071, 9073, 9080-9085, 9090, 9091, 9100, 9101).
e Includes: Corpus Uteri and Uterus, NOS.
f Includes: Vulva and Vagina.
g Includes: ICD-O-3: 8150-8153, 8155, 8156, 8240, 8249, 8452, 8680.
h Includes: Kidney and Renal pelvis.
i Includes: Non-Hodgkin Lymphoma Extranodal and Nodal.
j Includes: ICD-O-3: 8000, 8010, 8140, 8210,8220, 8243, 8245, 8246, 8255, 8261, 8263, 8440, 8470, 8480, 8481, 8490, 8574, 8936, 9015.
k Includes: ICD-O-3: 8010, 8012, 8013, 8020, 8021, 8033, 8041, 8045, 8046, 8050, 8120, 8140, 8144, 8230, 8246, 8255, 8290, 8310, 8313, 8320 8323, 8344, 8380-8382, 8401, 8410, 8441, 8442, 8450, 8460-8463, 8470-8472, 8480-8482, 8504, 8560, 8570, 8574, 8980, 9000, 9014, 9015.
l Includes: Soft tissue including heart.
m Includes Acute lymphocytic leukemia, Acute myeloid leukemia, Chronic lymphocytic leukemia, and Chronic myeloid leukemia.
n Includes: Anus, Anal canal, and Anorectum.
o Includes: Splenic flexure, Hepatic flexure, Ascending colon, Transverse colon, Descending colon, and Cecum.
p Includes: Sigmoid colon.
q Includes: ICD-O-3: 8000, 8001, 8010, 8012, 8013, 8020, 8021, 8032, 8035, 8041, 8046, 8050, 8070, 8140, 8154, 8160, 8230, 8246, 8255, 8260, 8290, 8440, 8450, 8470, 8471, 8480, 8481, 8490, 8500, 8504, 8550, 8551, 8560, 8574, 8890, 8936, 8963, 8971, 9120, 9260, 9364, 9473.
r Includes: ICD-O-3: 8000, 8001, 8010, 8012, 8013, 8020-8022, 8031-8033, 8041, 8042, 8044–8046, 8050, 8051, 8070-8072, 8076, 8082, 8083, 8123, 8140, 8144, 8200, 8230, 8246, 8250–8255, 8260, 8310, 8323, 8333, 8345, 8370, 8430, 8471, 8480, 8481, 8490, 8507, 8550, 8560, 8574, 8576, 8580, 8680, 8711, 8800-8803, 8805, 8810, 8815, 8824, 8830, 8854, 8855, 8890, 8972, 8980, 9040, 9041, 9043, 9061, 9065, 9071, 9084, 9085, 9100, 9120, 9130, 9133, 9150, 9220, 9231, 9250, 9260, 9364, 9473, 9540, 9560, 9581.
References
- Adolescent; Young Adult Oncology Progress Review Group. Closing the Gap: Research and Care Imperatives for Adolescents and Young Adults with Cancer. 2006. Available online: https://www.livestrong.org/content/closing-gap-research-and-care-imperatives-adolescents-and-young-adults-cancer (accessed on 21 April 2020).
- Barr, R.D.J.C.T.R. Common cancers in adolescents. Cancer Treat. Rev. 2007, 33, 597–602. [Google Scholar] [CrossRef]
- Smith, A.W.; Seibel, N.L.; Lewis, D.R.; Albritton, K.H.; Blair, D.F.; Blanke, C.D.; Bleyer, W.A.; Freyer, D.R.; Geiger, A.M.; Hayes-Lattin, B.; et al. Next steps for adolescent and young adult oncology workshop: An update on progress and recommendations for the future. Cancer 2016, 122, 988–999. [Google Scholar] [CrossRef]
- Lewis, D.R.; Seibel, N.L.; Smith, A.W.; Stedman, M.R. Adolescent and Young Adult Cancer Survival. J. Natl. Cancer Inst. Monogr. 2014, 2014, 228–235. [Google Scholar] [CrossRef]
- Meeneghan, M.R.; Wood, W.A. Challenges for cancer care delivery to adolescents and young adults: Present and future. Acta. Haematol. 2014, 132, 414–422. [Google Scholar] [CrossRef]
- Surveillance, Epidemiology and End Results Program. All Cancer Sites Combined Recent Trends in SEER Age-Adjusted Incidence Rates. Available online: https://seer.cancer.gov/explorer/application.html?site=1&data_type=1&graph_type=2&compareBy=age_range&chk_age_range_62=62&sex=1&race=1&hdn_stage=101&rate_type=1&advopt_precision=1&advopt_display=2#graphArea (accessed on 14 July 2020).
- Close, A.G.; Dreyzin, A.; Mph, K.D.M.; Seynnaeve, B.K.; Rapkin, L.B. Adolescent and young adult oncology—Past, present, and future. CA Cancer J. Clin. 2019, 69, 485–496. [Google Scholar] [CrossRef]
- Van Der Meer, D.J.; Karim-Kos, H.E.; Van Der Mark, M.; Aben, K.K.H.; Bijlsma, R.M.; Rijneveld, A.W.; Van Der Graaf, W.T.A.; Husson, O. Incidence, Survival, and Mortality Trends of Cancers Diagnosed in Adolescents and Young Adults (15–39 Years): A Population-Based Study in The Netherlands. Cancers 2020, 12, 3421. [Google Scholar] [CrossRef]
- Adams, S.C.; Herman, J.; Lega, I.C.; Mitchell, L.; Hodgson, D.; Edelstein, K.; Travis, L.B.; Sabiston, C.M.; Thavendiranathan, P.; Gupta, A.A. Young Adult Cancer Survivorship: Recommendations for Patient Follow-up, Exercise Therapy, and Research. JNCI Cancer Spectr. 2021, 5, pkaa099. [Google Scholar] [CrossRef]
- Kinahan, K.E.; Sanford, S.; Sadak, K.T.; Salsman, J.M.; Danner-Koptik, K.; Didwania, A. Models of Cancer Survivorship Care for Adolescents and Young Adults. Semin. Oncol. Nurs. 2015, 31, 251–259. [Google Scholar] [CrossRef][Green Version]
- ACS. Cancer Treatment and Survivorship Facts & Figures 2012-American Cancer Society. 2012. Available online: https://www.cancer.org/content/dam/cancer-org/research/cancer-facts-and-statistics/cancer-treatment-and-survivorship-facts-and-figures/cancer-treatment-and-survivorship-facts-and-figures-2012-2013.pdf (accessed on 9 December 2020).
- Osborn, M.; Johnson, R.; Thompson, K.; Anazodo, A.; Albritton, K.; Ferrari, A.; Stark, D. Models of care for adolescent and young adult cancer programs. Pediatr. Blood Cancer 2019, 66, e27991. [Google Scholar] [CrossRef]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Gupta, S.; Harper, A.; Ruan, Y.; Barr, R.; Frazier, A.L.; Ferlay, J.; Steliarova-Foucher, E.; Fidler-Benaoudia, M.M. International Trends in the Incidence of Cancer among Adolescents and Young Adults. J. Natl. Cancer Inst. 2020, 112, 1105–1117. [Google Scholar] [CrossRef]
- Miller, K.D.; Fidler-Benaoudia, M.; Keegan, T.H.; Hipp, H.S.; Jemal, A.; Siegel, R.L. Cancer statistics for adolescents and young adults. CA Cancer J. Clin. 2020, 70, 443–459. [Google Scholar] [CrossRef]
- Perez, G.K.; Salsman, J.M.; Fladeboe, K.; Kirchhoff, A.C.; Park, E.R.; Rosenberg, A.R. Taboo Topics in Adolescent and Young Adult Oncology: Strategies for Managing Challenging but Important Conversations Central to Adolescent and Young Adult Cancer Survivorship. Am. Soc. Clin. Oncol. Educ. Book 2020, 40, e171–e185. [Google Scholar] [CrossRef]
- Burgers, V.W.; van der Graaf, W.T.; van der Meer, D.J.; McCabe, M.G.; Rijneveld, A.W.; Bent, M.J.V.D.; Husson, O. Adolescents and Young Adults Living With an Uncertain or Poor Cancer Prognosis: The “New” Lost Tribe. J. Natl. Compr. Cancer Netw. 2021, 19, 240–246. [Google Scholar] [CrossRef]
- Ferrari, A.; Stark, D.; Peccatori, F.; Fern, L.; Laurence, V.; Gaspar, N.; Bozovic-Spasojevic, I.; Smith, O.; De Munter, J.; Derwich, K.; et al. Adolescents and young adults (AYA) with cancer: A position paper from the AYA Working Group of the European Society for Medical Oncology (ESMO) and the European Society for Paediatric Oncology (SIOPE). ESMO Open 2021, 6, 100096. [Google Scholar] [CrossRef]
- Dommett, R.M.; Redaniel, T.; Stevens, M.C.G.; Hamilton, W.; Martin, R.M. Features of cancer in teenagers and young adults in primary care: A population-based nested case–control study. Br. J. Cancer 2013, 108, 2329–2333. [Google Scholar] [CrossRef]
- Fern, L.A.; Birch, R.; Whelan, J.; Cooke, M.; Sutton, S.; Neal, R.D.; Gerrand, C.; Hubbard, G.; Smith, S.; Lethaby, C. Why can’t we improve the timeliness of cancer diagnosis in children, teenagers, and young adults? BMJ 2013, 347, f6493. [Google Scholar] [CrossRef]
- Tricoli, J.V.; Bleyer, A. Adolescent and Young Adult Cancer Biology. Cancer J. 2018, 24, 267–274. [Google Scholar] [CrossRef]
- Sender, L.; Zabokrtsky, K.B. Adolescent and young adult patients with cancer: A milieu of unique features. Nat. Rev. Clin. Oncol. 2015, 12, 465–480. [Google Scholar] [CrossRef]
- Bleyer, A.; on behalf of the Biology and Clinical Trials Subgroups of the US National Cancer Institute Progress Review Group in Adolescent and Young Adult Oncology; Barr, R.; Hayes-Lattin, B.; Thomas, D.; Ellis, C.; Anderson, B. The distinctive biology of cancer in adolescents and young adults. Nat. Rev. Cancer 2008, 8, 288–298. [Google Scholar] [CrossRef]
- Potosky, A.L.; Harlan, L.C.; Albritton, K.; Cress, R.D.; Friedman, D.L.; Hamilton, A.S.; Kato, I.; Keegan, T.H.; Keel, G.; Schwartz, S.M.; et al. Use of Appropriate Initial Treatment Among Adolescents and Young Adults With Cancer. J. Natl. Cancer Inst. 2014, 106, 300. [Google Scholar] [CrossRef]
- Fardell, J.E.; Patterson, P.; Wakefield, C.E.; Signorelli, C.; Cohn, R.; Anazodo, A.; Zebrack, B.; Sansom-Daly, U. A Narrative Review of Models of Care for Adolescents and Young Adults with Cancer: Barriers and Recommendations. J. Adolesc. Young-Adult Oncol. 2018, 7, 148–152. [Google Scholar] [CrossRef]
- Stoneham, S.J. AYA survivorship: The next challenge. Cancer 2020, 126, 2116–2119. [Google Scholar] [CrossRef] [PubMed]
- Zebrack, B.J. Psychological, social, and behavioral issues for young adults with cancer. Cancer 2011, 117, 2289–2294. [Google Scholar] [CrossRef] [PubMed]
- Kent, E.E.; Mollica, M.A.; Buckenmaier, S.; Smith, A.W. The Characteristics of Informal Cancer Caregivers in the United States. Semin. Oncol. Nurs. 2019, 35, 328–332. [Google Scholar] [CrossRef]
- Greup, S.R.; Kaal, S.E.J.; Jansen, R.; Manten-Horst, E.; Thong, M.S.Y.; Van Der Graaf, W.T.A.; Prins, J.B.; Husson, O. Post-Traumatic Growth and Resilience in Adolescent and Young Adult Cancer Patients: An Overview. J. Adolesc. Young-Adult Oncol. 2018, 7, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Harju, E.; Roser, K.; Dehler, S.; Michel, G. Health-related quality of life in adolescent and young adult cancer survivors. Support. Care Cancer 2018, 26, 3099–3110. [Google Scholar] [CrossRef]
- Husson, O.; Huijgens, P.C.; Van Der Graaf, W.T.A. Psychosocial challenges and health-related quality of life of adolescents and young adults with hematologic malignancies. Blood 2018, 132, 385–392. [Google Scholar] [CrossRef]
- Husson, O.; Zebrack, B.J.; Aguilar, C.; Hayes-Lattin, B.; Cole, S. Cancer in adolescents and young adults: Who remains at risk of poor social functioning over time? Cancer 2017, 123, 2743–2751. [Google Scholar] [CrossRef]
- Schulte, F.S.M.; Chalifour, K.; Eaton, G.; Garland, S.N. Quality of life among survivors of adolescent and young adult cancer in Canada: A Young Adults with Cancer in Their Prime (YACPRIME) study. Cancer 2021, 127, 1325–1333. [Google Scholar] [CrossRef]
- Robison, L.L.; Hudson, M.M. Survivors of childhood and adolescent cancer: Life-long risks and responsibilities. Nat. Rev. Cancer 2014, 14, 61–70. [Google Scholar] [CrossRef]
- Robison, L.L.; Mertens, A.C.; Boice, J.D.; Breslow, N.E.; Donaldson, S.S.; Green, D.M.; Li, F.P.; Meadows, A.T.; Mulvihill, J.J.; Neglia, J.; et al. Study design and cohort characteristics of the childhood cancer survivor study: A multi-institutional collaborative project. Med. Pediatr. Oncol. 2002, 38, 229–239. [Google Scholar] [CrossRef]
- Travis, L.B.; Beard, C.; Allan, J.; Dahl, A.A.; Feldman, D.; Oldenburg, J.; Daugaard, G.; Kelly, J.L.; Dolan, M.E.; Hannigan, R.; et al. Testicular Cancer Survivorship: Research Strategies and Recommendations. J. Natl. Cancer Inst. 2010, 102, 1114–1130. [Google Scholar] [CrossRef]
- Steininger, J.; Gellrich, F.; Schulz, A.; Westphal, D.; Beissert, S.; Meier, F. Systemic Therapy of Metastatic Melanoma: On the Road to Cure. Cancers 2021, 13, 1430. [Google Scholar] [CrossRef]
- Eggermont, A.M.M.; Blank, C.U.; Mandalà, M.; Long, G.V.; Atkinson, V.G.; Dalle, S.; Haydon, A.M.; Meshcheryakov, A.; Khattak, A.; Carlino, M.S.; et al. Adjuvant pembrolizumab versus placebo in resected stage III melanoma (EORTC 1325-MG/KEYNOTE-054): Distant metastasis-free survival results from a double-blind, randomised, controlled, phase 3 trial. Lancet Oncol. 2021, 22, 643–654. [Google Scholar] [CrossRef]
- Bottomley, A.; Coens, C.; Mierzynska, J.; Blank, C.U.; Mandalà, M.; Long, G.V.; Atkinson, V.G.; Dalle, S.; Haydon, A.M.; Meshcheryakov, A. Adjuvant pembrolizumab versus placebo in resected stage III melanoma (EORTC 1325-MG/KEYNOTE-054): Health-related quality-of-life results from a double-blind, randomised, controlled, phase 3 trial. Lancet Oncol. 2021, 22, 655–664. [Google Scholar] [CrossRef]
- Larkin, J.; Chiarion-Sileni, V.; Gonzalez, R.; Grob, J.-J.; Rutkowski, P.; Lao, C.D.; Cowey, C.L.; Schadendorf, D.; Wagstaff, J.; Dummer, R.; et al. Five-Year Survival with Combined Nivolumab and Ipilimumab in Advanced Melanoma. N. Engl. J. Med. 2019, 381, 1535–1546. [Google Scholar] [CrossRef] [PubMed]
- Coccia, P.F.; Pappo, A.S.; Beaupin, L.; Borges, V.F.; Borinstein, S.C.; Chugh, R.; Dinner, S.; Folbrecht, J.; Frazier, A.L.; Goldsby, R.; et al. Adolescent and Young Adult Oncology, Version 2.2018, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Cancer Netw. 2018, 16, 66–97. [Google Scholar] [CrossRef]
- Bleyer, W.A.; Barr, R.D.; Ries, L.; Whelan, J.; Ferrari, A. Cancer in Adolescents and Young Adults; Springer: Berlin, Germany, 2007. [Google Scholar]
- Patterson, P.; McDonald, F.E.; Zebrack, B.; Medlow, S. Emerging Issues Among Adolescent and Young Adult Cancer Survivors. Semin. Oncol. Nurs. 2015, 31, 53–59. [Google Scholar] [CrossRef] [PubMed]
- Chao, C.; Bhatia, S.; Xu, L.; Cannavale, K.L.; Wong, F.L.; Huang, P.-Y.S.; Cooper, R.; Armenian, S.H. Chronic Comorbidities Among Survivors of Adolescent and Young Adult Cancer. J. Clin. Oncol. 2020, 38, 3161–3174. [Google Scholar] [CrossRef]
- Abdelhadi, O.A.; Joseph, J.; Pollock, B.H.; Keegan, T.H.M. Additional medical costs of chronic conditions among adolescent and young adult cancer survivors. J. Cancer Surviv. 2021, 1–10. [Google Scholar] [CrossRef]
- Fidler, M.M.; Frobisher, C.; Hawkins, M.M.; Nathan, P.C. Challenges and opportunities in the care of survivors of adolescent and young adult cancers. Pediatr. Blood Cancer 2019, 66, e27668. [Google Scholar] [CrossRef] [PubMed]
- Bright, C.J.; Reulen, R.C.; Winter, D.L.; Stark, D.P.; McCabe, M.G.; Edgar, A.B.; Frobisher, C.; Hawkins, M.M. Risk of subsequent primary neoplasms in survivors of adolescent and young adult cancer (Teenage and Young Adult Cancer Survivor Study): A population-based, cohort study. Lancet Oncol. 2019, 20, 531–545. [Google Scholar] [CrossRef]
- Lee, J.S.; DuBois, S.G.; Coccia, P.F.; Bleyer, A.; Olin, R.L.; Goldsby, R.E. Increased risk of second malignant neoplasms in adolescents and young adults with cancer. Cancer 2016, 122, 116–123. [Google Scholar] [CrossRef]
- Chao, C.; Bhatia, S.; Xu, L.; Cannavale, K.L.; Wong, F.L.; Huang, P.-Y.S.; Cooper, R.; Armenian, S.H. Incidence, Risk Factors, and Mortality Associated With Second Malignant Neoplasms Among Survivors of Adolescent and Young Adult Cancer. JAMA Netw. Open 2019, 2, e195536. [Google Scholar] [CrossRef]
- Van Leeuwen, F.E.; Ng, A.K. Long-term risk of second malignancy and cardiovascular disease after Hodgkin lymphoma treatment. Hematology Am. Soc. Hematol. Educ. Program 2016, 2016, 323–330. [Google Scholar] [CrossRef]
- Schaapveld, M.; Aleman, B.M.P.; Van Eggermond, A.M.; Janus, C.P.M.; Krol, S.; Van Der Maazen, R.W.M.; Roesink, J.M.; Raemaekers, J.M.M.; De Boer, J.P.; Zijlstra, J.M.; et al. Second Cancer Risk Up to 40 Years after Treatment for Hodgkin’s Lymphoma. N. Engl. J. Med. 2015, 373, 2499–2511. [Google Scholar] [CrossRef]
- Hauptmann, M.; Fossa, S.D.; Stovall, M.; Van Leeuwen, F.E.; Johannesen, T.B.; Rajaraman, P.; Gilbert, E.S.; Smith, S.A.; Weathers, R.E.; Aleman, B.M.P.; et al. Increased stomach cancer risk following radiotherapy for testicular cancer. Br. J. Cancer 2014, 112, 44–51. [Google Scholar] [CrossRef]
- Keegan, T.H.M.; Kushi, L.H.; Li, Q.; Brunson, A.; Chawla, X.; Chew, H.K.; Malogolowkin, M.; Wun, T. Cardiovascular disease incidence in adolescent and young adult cancer survivors: A retrospective cohort study. J. Cancer Surviv. 2018, 12, 388–397. [Google Scholar] [CrossRef]
- Meinardi, M.; Gietema, J.; Van Der Graaf, W.A.; Van Veldhuisen, D.; Runne, M.; Sluiter, W.; de Vries, E.; Willemse, P.H.; Mulder, N.; Berg, M.V.D.; et al. Cardiovascular Morbidity in Long-Term Survivors of Metastatic Testicular Cancer. J. Clin. Oncol. 2000, 18, 1725–1732. [Google Scholar] [CrossRef]
- Belt-Dusebout, A.W.V.D.; de Wit, R.; Gietema, J.A.; Horenblas, S.; Louwman, M.W.; Ribot, J.G.; Hoekstra, H.J.; Ouwens, G.M.; Aleman, B.M.; van Leeuwen, F.E. Treatment-Specific Risks of Second Malignancies and Cardiovascular Disease in 5-Year Survivors of Testicular Cancer. J. Clin. Oncol. 2007, 25, 4370–4378. [Google Scholar] [CrossRef] [PubMed]
- Tai, E.; Buchanan, N.; Ms, J.T.; Fairley, T.; Moore, A.; Richardson, L.C. Health status of adolescent and young adult cancer survivors. Cancer 2012, 118, 4884–4891. [Google Scholar] [CrossRef] [PubMed]
- Jensen, M.V.; Rugbjerg, K.; de Fine Licht, S.; Johansen, C.; Schmiegelow, K.; Andersen, K.K.; Winther, J.F. Endocrine late effects in survivors of cancer in adolescence and young adulthood: A Danish population-based cohort study. JAMA Netw. Open 2018, 1, e180349. [Google Scholar] [CrossRef] [PubMed]
- Jim, H.S.; Jennewein, S.L.; Quinn, G.; Reed, D.R.; Small, B.J. Cognition in Adolescent and Young Adults Diagnosed With Cancer: An Understudied Problem. J. Clin. Oncol. 2018, 36, 2752–2754. [Google Scholar] [CrossRef] [PubMed]
- Dewar, E.O.; Ahn, C.; Eraj, S.; Mahal, B.A.; Sanford, N.N. Psychological distress and cognition among long-term survivors of adolescent and young adult cancer in the USA. J. Cancer Surviv. 2021, 15, 776–784. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.J.; Schover, L.R.; Partridge, A.H.; Patrizio, P.; Wallace, W.H.; Hagerty, K.; Beck, L.N.; Brennan, L.V.; Oktay, K. American Society of Clinical Oncology Recommendations on Fertility Preservation in Cancer Patients. J. Clin. Oncol. 2006, 24, 2917–2931. [Google Scholar] [CrossRef]
- Murphy, D.; Orgel, E.; Termuhlen, A.; Shannon, S.; Warren, K.; Quinn, G.P. Why Healthcare Providers Should Focus on the Fertility of AYA Cancer Survivors: It’s Not Too Late! Front. Oncol. 2013, 3, 248. [Google Scholar] [CrossRef]
- Haugnes, H.S.; Bosl, G.; Boer, H.; Gietema, J.; Brydøy, M.; Oldenburg, J.; Dahl, A.A.; Bremnes, R.M.; Fosså, S.D. Long-Term and Late Effects of Germ Cell Testicular Cancer Treatment and Implications for Follow-Up. J. Clin. Oncol. 2012, 30, 3752–3763. [Google Scholar] [CrossRef]
- Teenage Cancer Trust. A Blueprint of Care for Teenagers and Young Adults with Cancer. 2012. Available online: https://www.teenagecancertrust.org/sites/default/files/Blueprint-of-Care.pdf (accessed on 3 April 2020).
- Barr, R.D.; Ferrari, A.; Ries, L.; Whelan, J.; Bleyer, W.A. Cancer in adolescents and young adults: A narrative review of the current status and a view of the future. JAMA Pediatr. 2016, 170, 495–501. [Google Scholar] [CrossRef]
- Loren, A.W. Fertility issues in patients with hematologic malignancies. Hematology 2015, 2015, 138–145. [Google Scholar] [CrossRef]
- Barnett, M.; McDonnell, G.; DeRosa, A.; Schuler, T.; Philip, E.; Peterson, L.; Touza, K.; Jhanwar, S.; Atkinson, T.M.; Ford, J.S. Psychosocial outcomes and interventions among cancer survivors diagnosed during adolescence and young adulthood (AYA): A systematic review. J. Cancer Surviv. 2016, 10, 814–831. [Google Scholar] [CrossRef]
- Benedict, C.; Thom, B.; Kelvin, J. Fertility preservation and cancer: Challenges for adolescent and young adult patients. Curr. Opin. Support. Palliat. Care. 2016, 10, 87. [Google Scholar] [CrossRef]
- Russell, A.M.; Galvin, K.M.; Harper, M.M.; Clayman, M.L. A comparison of heterosexual and LGBTQ cancer survivors’ outlooks on relationships, family building, possible infertility, and patient-doctor fertility risk communication. J. Cancer Surviv. 2016, 10, 935–942. [Google Scholar] [CrossRef]
- Su, H.I.; Lee, Y.T.; Barr, R. Oncofertility: Meeting the fertility goals of adolescents and young adults with cancer. Cancer J. 2018, 24, 328. [Google Scholar]
- Zhou, E.S.; Falk, S.J.; Bober, S.L. Managing premature menopause and sexual dysfunction. Curr. Opin. Support. Palliat. Care 2015, 9, 294–300. [Google Scholar] [CrossRef]
- Bolte, S.; Zebrack, B. Sexual Issues in Special Populations: Adolescents and Young Adults. Semin. Oncol. Nurs. 2008, 24, 115–119. [Google Scholar] [CrossRef]
- Olsson, M.; Enskär, K.; Steineck, G.; Wilderäng, U.; Jarfelt, M. Self-Perceived Physical Attractiveness in Relation to Scars Among Adolescent and Young Adult Cancer Survivors: A Population-Based Study. J. Adolesc. Young-Adult Oncol. 2018, 7, 358–366. [Google Scholar] [CrossRef]
- Pt, M.L.M.; Med, J.K.P.; Jones, J.M.; Amin, L.; Chang, E.; Korenblum, C.; Mina, D.S.; McCabe, L.; Mitchell, L.; Giuliani, M. Reimagining care for adolescent and young adult cancer programs: Moving with the times. Cancer 2016, 122, 1038–1046. [Google Scholar] [CrossRef]
- Brierley, M.E.E.; Sansom-Daly, U.M.; Baenziger, J.; McGill, B.; Wakefield, C.E. Impact of physical appearance changes reported by adolescent and young adult cancer survivors: A qualitative analysis. Eur. J. Cancer Care 2019, 28, e13052. [Google Scholar] [CrossRef] [PubMed]
- Sun, V.; Grant, M.; Ms, C.S.W.; McMullen, C.K.; Bulkley, J.E.; Altschuler, A.; Ramirez, M.; Baldwin, C.; Herrinton, L.J.; Hornbrook, M.C.; et al. Dietary and Behavioral Adjustments to Manage Bowel Dysfunction After Surgery in Long-Term Colorectal Cancer Survivors. Ann. Surg. Oncol. 2015, 22, 4317–4324. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, S.N.; Arshad, O.; Kwok, J.; Tran, E.; Howard, A.F.; Serrano, I.; Goddard, K. Documentation and incidence of late effects and screening recommendations for adolescent and young adult head and neck cancer survivors treated with radiotherapy. Support. Care Cancer 2018, 27, 2609–2616. [Google Scholar] [CrossRef]
- CanTeen. Exploring Survivorship Care for Adolescent and Young Adult Cancer Survivors in Australia. Sydney, Australia: CanTeen Australia. 2015. Available online: https://www.canteen.org.au/wp-content/uploads/2016/09/Adolescent-and-Young-Adult-Cancer-Survivorship-Report.pdf (accessed on 4 July 2021).
- Ketterl, T.G.; Syrjala, K.L.; Casillas, J.; Jacobs, L.A.; Palmer, S.C.; McCabe, M.S.; Ganz, P.A.; Overholser, L.; Partridge, A.; Rajotte, E.J.; et al. Lasting effects of cancer and its treatment on employment and finances in adolescent and young adult cancer survivors. Cancer 2019, 125, 1908–1917. [Google Scholar] [CrossRef]
- Zebrack, B.J.; Corbett, V.; Embry, L.; Aguilar, C.; Meeske, K.A.; Hayes-Lattin, B.; Block, R.; Zeman, D.T.; Cole, S. Psychological distress and unsatisfied need for psychosocial support in adolescent and young adult cancer patients during the first year following diagnosis. Psycho-Oncology 2014, 23, 1267–1275. [Google Scholar] [CrossRef]
- Jones, J.M.; Fitch, M.; Bongard, J.; Maganti, M.; Gupta, A.; D’Agostino, N.; Korenblum, C. The Needs and Experiences of Post-Treatment Adolescent and Young Adult Cancer Survivors. J. Clin. Med. 2020, 9, 1444. [Google Scholar] [CrossRef]
- De, R.; Sutradhar, R.; Kurdyak, P.; Aktar, S.; Pole, J.D.; Baxter, N.; Nathan, P.C.; Gupta, S. Incidence and Predictors of Mental Health Outcomes Among Survivors of Adolescent and Young Adult Cancer: A Population-Based Study Using the IMPACT Cohort. J. Clin. Oncol. 2021, 39, 1010–1019. [Google Scholar] [CrossRef] [PubMed]
- Zebrack, B.; Isaacson, S. Psychosocial care of adolescent and young adult patients with cancer and survivors. J. Clin. Oncol. 2012, 30, 1221–1226. [Google Scholar] [CrossRef]
- Kahl, K.G.; Winter, L.; Schweiger, U. The third wave of cognitive behavioural therapies: What is new and what is effective? Current opinion in psychiatry. Curr. Opin. Psychiatry 2012, 25, 522–528. [Google Scholar] [CrossRef]
- Hulbert-Williams, N.J.; Storey, L.; Wilson, K.G. Psychological interventions for patients with cancer: Psychological flexibility and the potential utility of Acceptance and Commitment Therapy. Eur. J. Cancer Care 2014, 24, 15–27. [Google Scholar] [CrossRef]
- Keegan, T.H.M.; Lichtensztajn, D.; Kato, I.; Kent, E.E.; Wu, X.-C.; West, M.M.; Hamilton, A.S.; Zebrack, B.; Bellizzi, K.M.; et al.; the AYA HOPE Study Collaborative Group Unmet adolescent and young adult cancer survivors information and service needs: A population-based cancer registry study. J. Cancer Surviv. 2012, 6, 239–250. [Google Scholar] [CrossRef] [PubMed]
- Kwak, M.; Zebrack, B.J.; Meeske, K.A.; Embry, L.; Aguilar, C.; Block, R.; Hayes-Lattin, B.; Li, Y.; Butler, M.; Cole, S. Trajectories of Psychological Distress in Adolescent and Young Adult Patients with Cancer: A 1-Year Longitudinal Study. J. Clin. Oncol. 2013, 31, 2160–2166. [Google Scholar] [CrossRef] [PubMed]
- Thewes, B.; Kaal, S.E.J.; Custers, J.A.E.; Manten-Horst, E.; Jansen, R.; Servaes, P.; Van Der Graaf, W.T.A.; Prins, J.B.; Husson, O. Prevalence and correlates of high fear of cancer recurrence in late adolescents and young adults consulting a specialist adolescent and young adult (AYA) cancer service. Support. Care Cancer 2017, 26, 1479–1487. [Google Scholar] [CrossRef]
- Shay, L.A.; Carpentier, M.Y.; Vernon, S.W. Prevalence and correlates of fear of recurrence among adolescent and young adult versus older adult post-treatment cancer survivors. Support. Care Cancer 2016, 24, 4689–4696. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Li, W.; Wen, Y.; Wang, H.; Sun, H.; Liang, W.; Zhang, B.; Humphris, G. Fear of cancer recurrence in adolescent and young adult cancer survivors: A systematic review of the literature. Psycho-Oncology 2019, 28, 675–686. [Google Scholar] [CrossRef] [PubMed]
- Zebrack, B.; Santacroce, S.J.; Patterson, P.; Gubin, A. Adolescents and Young Adults with Cancer: A Biopsychosocial Approach. In Pediatric Psychosocial Oncology: Textbook for Multidisciplinary Care; Springer Science and Business Media LLC: Berlin, Germany, 2016; pp. 199–217. [Google Scholar]
- McCarthy, M.C.; McNeil, R.; Drew, S.; Dunt, D.; Kosola, S.; Orme, L.; Sawyer, S.M. Psychological Distress and Posttraumatic Stress Symptoms in Adolescents and Young Adults with Cancer and Their Parents. J. Adolesc. Young-Adult Oncol. 2016, 5, 322–329. [Google Scholar] [CrossRef] [PubMed]
- Rosenberg, A.R.; Yi-Frazier, J.P.; Wharton, C.; Gordon, K.; Jones, B. Contributors and Inhibitors of Resilience Among Adolescents and Young Adults with Cancer. J. Adolesc. Young-Adult Oncol. 2014, 3, 185–193. [Google Scholar] [CrossRef] [PubMed]
- McDonnell, G.A.; Pope, A.W.; Schuler, T.A.; Ford, J.S. The relationship between cancer-related worry and posttraumatic growth in adolescent and young adult cancer survivors. Psycho-Oncology 2018, 27, 2155–2164. [Google Scholar] [CrossRef]
- Lidington, E.; Vlooswijk, C.; Stallard, K.; Travis, E.; Younger, E.; Edwards, P.; Nandhabalan, M.; Hunter, N.; Sarpal, N.; Flett, D.; et al. ‘This is not part of my life plan’: A qualitative study on the psychosocial experiences and practical challenges in young adults with cancer age 25 to 39 years at diagnosis. Eur. J. Cancer Care 2021, 30, e13458. [Google Scholar] [CrossRef]
- D’Agostino, N.M.; Penney, A.; Zebrack, B. Providing developmentally appropriate psychosocial care to adolescent and young adult cancer survivors. Cancer 2011, 117, 2329–2334. [Google Scholar] [CrossRef]
- Carpentier, M.Y.; Fortenberry, J.D.; Ott, M.; Brames, M.J.; Einhorn, L.H. Perceptions of masculinity and self-image in adolescent and young adult testicular cancer survivors: Implications for romantic and sexual relationships. Psycho-Oncology 2010, 20, 738–745. [Google Scholar] [CrossRef]
- Warner, E.L.; Kent, E.E.; Trevino, K.M.; Parsons, H.M.; Zebrack, B.J.; Kirchhoff, A.C. Social well-being among adolescents and young adults with cancer: A systematic review. Cancer 2016, 122, 1029–1037. [Google Scholar] [CrossRef]
- Grinyer, A. The biographical impact of teenage and adolescent cancer. Chronic Illn. 2007, 3, 265–277. [Google Scholar] [CrossRef]
- Bellizzi, K.M.; Smith, A.; Schmidt, S.; Keegan, T.H.M.; Zebrack, B.; Lynch, C.F.; Deapen, D.; Shnorhavorian, M.; Tompkins, B.J.; Simon, M.; et al. Positive and negative psychosocial impact of being diagnosed with cancer as an adolescent or young adult. Cancer 2012, 118, 5155–5162. [Google Scholar] [CrossRef] [PubMed]
- Herbertson, R.; Hancock, B. Hodgkin Lymphoma in adolescents. Cancer Treat. Rev. 2005, 31, 339–360. [Google Scholar] [CrossRef]
- Guy, G.P.; Yabroff, K.R.; Ekwueme, D.U.; Smith, A.W.; Dowling, E.C.; Rechis, R.; Nutt, S.; Richardson, L.C. Estimating The Health And Economic Burden Of Cancer Among Those Diagnosed As Adolescents And Young Adults. Health Aff. 2014, 33, 1024–1031. [Google Scholar] [CrossRef] [PubMed]
- Husson, O.; Prins, J.B.; Kaal, S.E.J.; Oerlemans, S.; Stevens, W.B.; Zebrack, B.; Van Der Graaf, W.T.A.; Van De Poll-Franse, L.V. Adolescent and young adult (AYA) lymphoma survivors report lower health-related quality of life compared to a normative population: Results from the PROFILES registry. Acta Oncol. 2017, 56, 288–294. [Google Scholar] [CrossRef] [PubMed]
- Goodall, S.; King, M.; Ewing, J.; Smith, N.; Kenny, P. Preferences for support services among adolescents and young adults with cancer or a blood disorder: A discrete choice experiment. Heal. Policy 2012, 107, 304–311. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Elsbernd, A.; Pedersen, K.J.; Boisen, K.A.; Midtgaard, J.; Larsen, H.B. “On Your Own”: Adolescent and Young Adult Cancer Survivors’ Experience of Managing Return to Secondary or Higher Education in Denmark. J. Adolesc. Young-Adult Oncol. 2018, 7, 618–625. [Google Scholar] [CrossRef]
- Yanez, B.; Garcia, S.F.; Victorson, D.; Salsman, J.M. Distress among young adult cancer survivors: A cohort study. Support. Care Cancer 2013, 21, 2403–2408. [Google Scholar] [CrossRef]
- Kent, E.E.; Forsythe, L.P.; Yabroff, K.R.; Weaver, K.E.; De Moor, J.S.; Rodriguez, J.L.; Rowland, J.H. Are survivors who report cancer-related financial problems more likely to forgo or delay medical care? Cancer 2013, 119, 3710–3717. [Google Scholar] [CrossRef]
- Kirchhoff, A.C.; Lyles, C.R.; Fluchel, M.; Wright, J.; Leisenring, W. Limitations in health care access and utilization among long-term survivors of adolescent and young adult cancer. Cancer 2012, 118, 5964–5972. [Google Scholar] [CrossRef]
- Leuteritz, K.; Friedrich, M.; Sender, A.; Richter, D.; Mehnert-Theuerkauf, A.; Sauter, S.; Geue, K. Return to Work and Employment Situation of Young Adult Cancer Survivors: Results from the Adolescent and Young Adult-Leipzig Study. J. Adolesc. Young-Adult Oncol. 2021, 10, 226–233. [Google Scholar] [CrossRef]
- Vetsch, J.; Wakefield, C.E.; McGill, B.C.; Cohn, R.; Ellis, S.J.; Stefanic, N.; Sawyer, S.M.; Zebrack, B.; Sansom-Daly, U.M. Educational and vocational goal disruption in adolescent and young adult cancer survivors. Psycho-Oncology 2018, 27, 532–538. [Google Scholar] [CrossRef]
- Kent, E.E.; Parry, C.; Montoya, M.J.; Sender, L.S.; Morris, R.A.; Anton-Culver, H. “You’re too young for this”: Adolescent and young adults’ perspectives on cancer survivorship. J. Psychosoc. Oncol. 2012, 30, 260–279. [Google Scholar] [CrossRef]
- Kent, E.E.; Smith, A.W.; Keegan, T.H.M.; Lynch, C.F.; Wu, X.-C.; Hamilton, A.S.; Kato, I.; Schwartz, S. Talking About Cancer and Meeting Peer Survivors: Social Information Needs of Adolescents and Young Adults Diagnosed with Cancer. J. Adolesc. Young-Adult Oncol. 2013, 2, 44–52. [Google Scholar] [CrossRef]
- Kirchhoff, A.C.; Yi, J.; Wright, J.; Warner, E.L.; Smith, K.R. Marriage and divorce among young adult cancer survivors. J. Cancer Surviv. 2012, 6, 441–450. [Google Scholar] [CrossRef] [PubMed]
- Borstelmann, N.A.; Rosenberg, S.M.; Ruddy, K.J.; Tamimi, R.M.; Gelber, S.; Schapira, L.; Come, S.; Borges, V.; Morgan, E.; Partridge, A.H. Partner support and anxiety in young women with breast cancer. Psycho-Oncology 2015, 24, 1679–1685. [Google Scholar] [CrossRef] [PubMed]
- Fosså, S.D.; Dahl, A.A. Fertility and Sexuality in Young Cancer Survivors Who Have Adult-Onset Malignancies. Hematol. Clin. N. Am. 2008, 22, 291–303. [Google Scholar] [CrossRef] [PubMed]
- Olsen, P.R.; Lorenzo, R. Supportive Care. Prog. Tumor Res. 2016, 43, 16–26. [Google Scholar] [CrossRef]
- Mellblom, A.V.; Kiserud, C.E.; Rueegg, C.S.; Ruud, E.; Loge, J.H.; Fosså, S.D.; Lie, H.C. Self-reported late effects and long-term follow-up care among 1889 long-term Norwegian Childhood, Adolescent, and Young Adult Cancer Survivors (the NOR-CAYACS study). Support. Care Cancer 2021, 29, 2947–2957. [Google Scholar] [CrossRef] [PubMed]
- Hauken, M.A.; Larsen, T.M.B.; Holsen, I. Meeting reality: Young adult cancer survivors’ experiences of reentering everyday life after cancer treatment. Cancer Nurs. 2013, 36, E17–E26. [Google Scholar] [CrossRef]
- Hydeman, J.A.; Uwazurike, O.C.; Adeyemi, E.I.; Beaupin, L.K. Survivorship needs of adolescent and young adult cancer survivors: A concept mapping analysis. J. Cancer Surviv. 2019, 13, 34–42. [Google Scholar] [CrossRef] [PubMed]
- Anderson, C.; Smitherman, A.; Nichols, H.B. Conditional relative survival among long-term survivors of adolescent and young adult cancers. Cancer 2018, 124, 3037–3043. [Google Scholar] [CrossRef]
- Adolescent and Young Adult Oncology Review Group. Closing the Gap: Research and Care Imperaives for Adolescents and Young Adults with Cancer; National Institute of Health, National Cancer Institute, and Livestrong Young Ault Alliance: Bethesda, MD, USA, 2006. [Google Scholar]
- Albritton, K.; Bleyer, W. The management of cancer in the older adolescent. Eur. J. Cancer 2003, 39, 2584–2599. [Google Scholar] [CrossRef] [PubMed]
- Lewis, I.; Morgan, S. Models of Care and Specialized Units. In Cancer in Adolescents and Young Adults; Springer Science: Berlin/Heidelberg, Germany, 2007; pp. 341–352. [Google Scholar]
- Husson, O.; Manten-Horst, E.; van der Graaf, W. Collaboration and Networking. Prog. Tumor Res. 2016, 43, 50–63. [Google Scholar] [CrossRef] [PubMed]
- Cancer Australia; CanTeen. National Service Delivery Framework for Adolescents and Young Adults with Cancer. Cranberra Australia. 2008. Available online: https://www.canceraustralia.gov.au/publications-and-resources/cancer-australia-publications/national-service-delivery-framework-adolescents-and-young-adults-cancer (accessed on 3 April 2020).
- Ferrari, A.; Thomas, D.; Franklin, A.R.; Hayes-Lattin, B.M.; Mascarin, M.; van der Graaf, W.; Albritton, K.H. Starting an Adolescent and Young Adult Program: Some Success Stories and Some Obstacles to Overcome. J. Clin. Oncol. 2010, 28, 4850–4857. [Google Scholar] [CrossRef]
- Ferrari, A.; Albritton, K.; Osborn, M.; Barr, R.; Johnson, R.H.; Stark, D.; Whelan, J. Access and Models of Care. In Pediatric Oncology; Springer Science and Business Media LLC: Berlin, Germany, 2017; pp. 509–547. [Google Scholar]
- Reed, D.; Block, R.G.; Johnson, R. Creating an Adolescent and Young Adult Cancer Program: Lessons Learned From Pediatric and Adult Oncology Practice Bases. J. Natl. Compr. Cancer Netw. 2014, 12, 1409–1415. [Google Scholar] [CrossRef]
- Bradford, N.K.; Greenslade, R.; Edwards, R.M.; Orford, R.; Roach, J.; Henney, R. Educational Needs of Health Professionals Caring for Adolescents and Young Adults with Cancer. J. Adolesc. Young-Adult Oncol. 2018, 7, 298–305. [Google Scholar] [CrossRef]
- Linendoll, N.; Murphy-Banks, R.; Barthel, E.; Bartucca, L.; Boehm, L.; Welch, M.; Weidner, R.A.; Parsons, S.K. The Creation of a Comprehensive Adolescent and Young Adult Cancer Survivorship Program: “Lost in Transition” No More. J. Adolesc. Young-Adult Oncol. 2020, 10, 397–403. [Google Scholar] [CrossRef]
- Gupta, S.; Pole, J.D.; Baxter, N.N.; Sutradhar, R.; Lau, C.; Nagamuthu, C.; Nathan, P.C. The effect of adopting pediatric protocols in adolescents and young adults with acute lymphoblastic leukemia in pediatric vs. adult centers: An IMPACT Cohort study. Cancer Med. 2019, 8, 2095–2103. [Google Scholar] [CrossRef]
- Ramphal, R.; Meyer, R.; Schacter, B.; Rogers, P.; Pinkerton, R. Active therapy and models of care for adolescents and young adults with cancer. Cancer 2011, 117, 2316–2322. [Google Scholar] [CrossRef]
- Stark, D.; Bielack, S.; Brugieres, L.; Dirksen, U.; Duarte, X.; Dunn, S.; Erdelyi, D.; Grew, T.; Hjorth, L.; Jazbec, J.; et al. Teenagers and young adults with cancer in Europe: From national programmes to a European integrated coordinated project. Eur. J. Cancer Care 2015, 25, 419–427. [Google Scholar] [CrossRef] [PubMed]
- Muffly, L.; Alvarez, E.; Lichtensztajn, D.; Abrahão, R.; Gomez, S.L.; Keegan, T. Patterns of care and outcomes in adolescent and young adult acute lymphoblastic leukemia: A population-based study. Blood Adv. 2018, 2, 895–903. [Google Scholar] [CrossRef] [PubMed]
- Saloustros, E.; Stark, D.P.; Michailidou, K.; Mountzios, G.; Brugieres, L.; Peccatori, F.; Jezdic, S.; Essiaf, S.; Douillard, J.-Y.; Bielack, S. The care of adolescents and young adults with cancer: Results of the ESMO/SIOPE survey. ESMO Open 2017, 2, e000252. [Google Scholar] [CrossRef] [PubMed]
- Li, C.K.; Dalvi, R.; Yonemori, K.; Ariffin, H.; Lyu, C.J.; Farid, M.; Gonzales-Santos, J.R.N.; Zhou, Q.; Bielack, S.; Brugieres, L.; et al. Care of adolescents and young adults with cancer in Asia: Results of an ESMO/SIOPE/SIOP Asia survey. ESMO Open 2019, 4, e000467. [Google Scholar] [CrossRef]
- Guirguis, S.; Fitch, M.; Maganti, M.; Gupta, A.; D’Agostino, N.; Korenblum, C.; Jones, J. Biopsychosocial Factors Associated with Supportive Care Needs in Canadian Adolescent and Young Adult Cancer Survivors. J. Clin. Med. 2021, 10, 2628. [Google Scholar] [CrossRef]
- Baird, H.; Patterson, P.; Medlow, S.; Allison, K.R. Understanding and Improving Survivorship Care for Adolescents and Young Adults with Cancer. J. Adolesc. Young-Adult Oncol. 2019, 8, 581–586. [Google Scholar] [CrossRef]
- Phillips, S.M.; Padgett, L.S.; Leisenring, W.; Stratton, K.K.; Bishop, K.; Krull, K.R.; Alfano, C.M.; Gibson, T.M.; De Moor, J.S.; Hartigan, D.B.; et al. Survivors of Childhood Cancer in the United States: Prevalence and Burden of Morbidity. Cancer Epidemiol. Biomark. Prev. 2015, 24, 653–663. [Google Scholar] [CrossRef]
- Chemaitilly, W.; Cohen, L.E.; Mostoufi-Moab, S.; Patterson, B.; Simmons, J.; Meacham, L.R.; van Santen, H.M.; Sklar, C.A. Endocrine Late Effects in Childhood Cancer Survivors. J. Clin. Oncol. 2018, 36, 2153–2159. [Google Scholar] [CrossRef]
- Shapiro, C.L. Cancer Survivorship. N. Engl. J. Med. 2018, 379, 2438–2450. [Google Scholar] [CrossRef]
- Oeffinger, K.C.; Mertens, A.C.; Sklar, C.A.; Kawashima, T.; Hudson, M.M.; Meadows, A.T.; Friedman, D.L.; Marina, N.; Hobbie, W.; Kadan-Lottick, N.; et al. Chronic Health Conditions in Adult Survivors of Childhood Cancer. N. Engl. J. Med. 2006, 355, 1572–1582. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.; DeSantis, C.; Virgo, K.; Stein, K.; Mariotto, A.; Smith, T.; Cooper, D.; Gansler, T.; Lerro, C.; Fedewa, S.; et al. Cancer treatment and survivorship statistics. CA Cancer J. Clin. 2012, 62, 220–241. [Google Scholar] [CrossRef]
- Landier, W.; Armenian, S.; Bhatia, S. Late Effects of Childhood Cancer and Its Treatment. Pediatr. Clin. N. Am. 2015, 62, 275–300. [Google Scholar] [CrossRef]
- Children’s Oncology Group. Long-Term Follow-Up Guidelines, Version 5.0. Available online: http://www.survivorshipguidelines.org/pdf/2018/COG_LTFU_Guidelines_v5.pdf (accessed on 9 July 2020).
- Michel, G.; Mulder, R.L.; Van Der Pal, H.J.H.; Skinner, R.; Bárdi, E.; Brown, M.C.; Vetsch, J.; Frey, E.; Windsor, R.; Kremer, L.C.M.; et al. Evidence-based recommendations for the organization of long-term follow-up care for childhood and adolescent cancer survivors: A report from the PanCareSurFup Guidelines Working Group. J. Cancer Surviv. 2019, 13, 759–772. [Google Scholar] [CrossRef]
- Miller, K.D.; Siegel, R.L.; Lin, C.C.; Mariotto, A.B.; Kramer, J.L.; Rowland, J.H.; Stein, K.D.; Alteri, R.; Jemal, A. Cancer treatment and survivorship statistics. CA Cancer J. Clin. 2016, 66, 271–289. [Google Scholar] [CrossRef]
- Denlinger, C.S.; Sanft, T.; Baker, K.S.; Broderick, G.; Demark-Wahnefried, W.; Friedman, D.L.; Goldman, M.; Hudson, M.; Khakpour, N.; King, A.; et al. Survivorship, Version 2.2018, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Cancer Netw. 2018, 16, 1216–1247. [Google Scholar] [CrossRef]
- National Comprehensive Cancer Network. NCCN Clinical Practical Guidelines in Oncology. Breast Cancer, Version 2. Available online: https://www.nccn.org/professionals/physician_gls/default.aspx (accessed on 9 December 2020).
- Tonorezos, E.S.; Barnea, D.; Cohn, R.; Cypriano, M.S.; Fresneau, B.C.; Haupt, R.; Hjorth, L.; Ishida, Y.; Kruseova, J.; Kuehni, C.E.; et al. Models of Care for Survivors of Childhood Cancer from Across the Globe: Advancing Survivorship Care in the Next Decade. J. Clin. Oncol. 2018, 36, 2223–2230. [Google Scholar] [CrossRef]
- Tawfik, B.; Jaffe, S.A.; Mohler, L.; Oomen-Hajagos, J.; Gil, I.S.; Chamberlain, R.; Gagnon, S.; Kano, M.; Gundelach, A.; Ryan, S.R.; et al. Developing a survivorship care plan (SCP) delivery process for patients and primary care providers serving poor, rural, and minority patients with cancer. Support. Care Cancer 2021, 29, 5021–5028. [Google Scholar] [CrossRef]
- Hill, R.E.; Wakefield, C.E.; Cohn, R.J.; Fardell, J.E.; Brierley, M.E.; Kothe, E.; Jacobsen, P.B.; Hetherington, K.; Mercieca-Bebber, R. Survivorship Care Plans in Cancer: A Meta-Analysis and Systematic Review of Care Plan Outcomes. Oncology 2019, 25, 351. [Google Scholar] [CrossRef] [PubMed]
- Haggstrom, D.A.; Kahn, K.L.; Klabunde, C.N.; Gray, S.W.; Keating, N.L. Oncologists’ perceptions of the usefulness of cancer survivorship care plan components. Support. Care Cancer 2021, 29, 945–954. [Google Scholar] [CrossRef] [PubMed]
- Jacobsen, P.; DeRosa, A.P.; Henderson, T.O.; Mayer, D.K.; Moskowitz, C.S.; Paskett, E.D.; Rowland, J.H. Systematic Review of the Impact of Cancer Survivorship Care Plans on Health Outcomes and Health Care Delivery. J. Clin. Oncol. 2018, 36, 2088–2100. [Google Scholar] [CrossRef] [PubMed]
- Nass, S.J.; Beaupin, L.K.; Demark-Wahnefried, W.; Fasciano, K.; Ganz, P.A.; Hayes-Lattin, B.; Hudson, M.M.; Nevidjon, B.; Oeffinger, K.C.; Rechis, R.; et al. Identifying and Addressing the Needs of Adolescents and Young Adults with Cancer: Summary of an Institute of Medicine Workshop. Oncologist 2015, 20, 186–195. [Google Scholar] [CrossRef] [PubMed]
- Marjerrison, S.; Barr, R.D. Unmet survivorship care needs of adolescent and young adult cancer survivors. JAMA Netw. Open. 2018, 1, e180350. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, L.; Tam, S.; Lewin, J.; Srikanthan, A.; Heck, C.; Hodgson, D.; Vakeesan, B.; Sim, H.-W.; Gupta, A. Measuring the Impact of an Adolescent and Young Adult Program on Addressing Patient Care Needs. J. Adolesc. Young-Adult Oncol. 2018, 7, 612–617. [Google Scholar] [CrossRef] [PubMed]
- Pollock, B.H. What’s missing in the assessment of adolescent and young adult (AYA) cancer outcomes? J. Natl. Cancer Inst. 2020, 112, 975–976. [Google Scholar] [CrossRef] [PubMed]
- Husson, O.; Ligtenberg, M.; van de Poll-Franse, L.; Prins, J.; Bent, M.V.D.; van Eenbergen, M.; Fles, R.; Manten-Horst, E.; Gietema, J.; van der Graaf, W. Comprehensive Assessment of Incidence, Risk Factors, and Mechanisms of Impaired Medical and Psychosocial Health Outcomes among Adolescents and Young Adults with Cancer: Protocol of the Prospective Observational COMPRAYA Cohort Study. Cancers 2021, 13, 2348. [Google Scholar] [CrossRef] [PubMed]
- Salsman, J.M.; Danhauer, S.C.; Moore, J.B.; Canzona, M.R.; Victorson, D.E.; Zebrack, B.J.; Reeve, B.B. Optimizing the measurement of health-related quality of life in adolescents and young adults with cancer. Cancer 2020, 126, 4818–4824. [Google Scholar] [CrossRef]
- Kaal, S.; Lidington, E.; Prins, J.; Jansen, R.; Manten-Horst, E.; Servaes, P.; van der Graaf, W.; Husson, O. Health-Related Quality of Life Issues in Adolescents and Young Adults with Cancer: Discrepancies with the Perceptions of Health Care Professionals. J. Clin. Med. 2021, 10, 1833. [Google Scholar] [CrossRef]
- Siembida, E.J.; Reeve, B.B.; Zebrack, B.J.; Snyder, M.A.; Salsman, J.M. Measuring health-related quality of life in adolescent and young adult cancer survivors with the National Institutes of Health Patient-Reported Outcomes Measurement Information System®: Comparing adolescent, emerging adult, and young adult survivor perspectives. Psycho-Oncology 2021, 30, 303–311. [Google Scholar] [CrossRef]
- CanTeen Australia. Australian Youth Cancer Framework for Adolescents and Young Adults with Cancer. Available online: https://www.canteen.org.au/wp-content/uploads/2017/09/Australian_Youth_Cancer_Framework_2017.pdf (accessed on 4 December 2020).
- Dickman, P.W.; Coviello, E. Estimating and Modeling Relative Survival. Stata J. Promot. Commun. Stat. Stata 2015, 15, 186–215. [Google Scholar] [CrossRef]
- Surveillance Epidemiology and End Results (SEER) Program. Expected Survival Life Tables. Available online: https://seer.cancer.gov/expsurvival/ (accessed on 9 December 2020).
- Surveillance Epidemiology and End Results (SEER) Program. Age Standards for Survival. Available online: https://seer.cancer.gov/stdpopulations/survival.html (accessed on 9 December 2020).
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).