Duration of Reduced CA19-9 Levels Is a Better Prognostic Factor Than Its Rate of Reduction for Unresectable Locally Advanced Pancreatic Cancer
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Serum Levels of CA 19-9
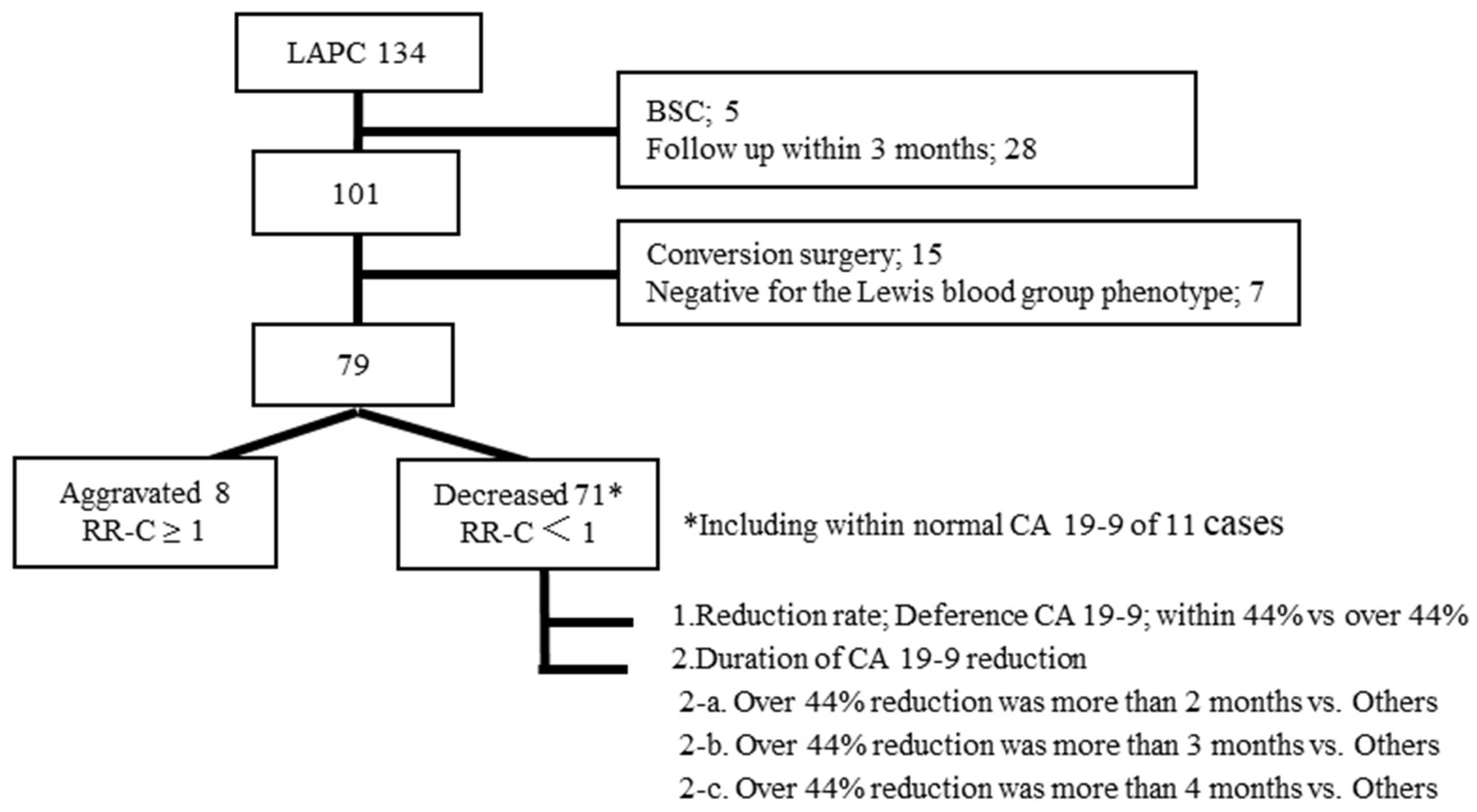
2.2. Patients
2.3. Resectability and Treatment
2.4. Statistical Analysis
3. Results
3.1. Patient Characteristics
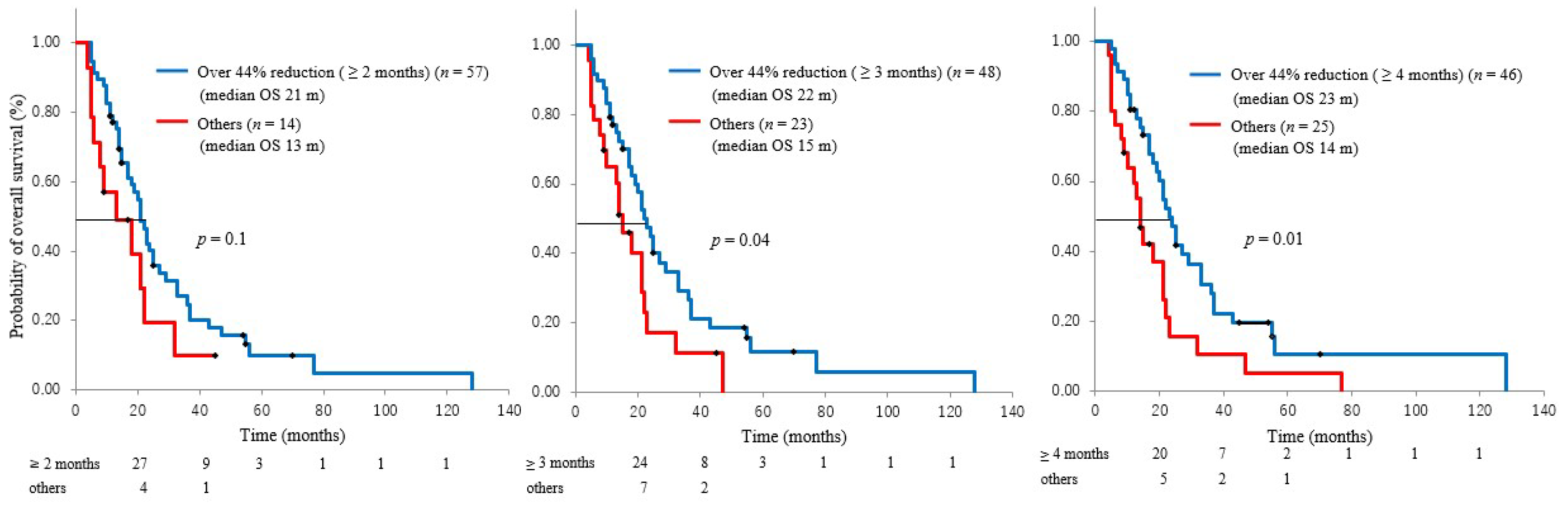
3.2. CA19-9 Reduction Rate and Survival
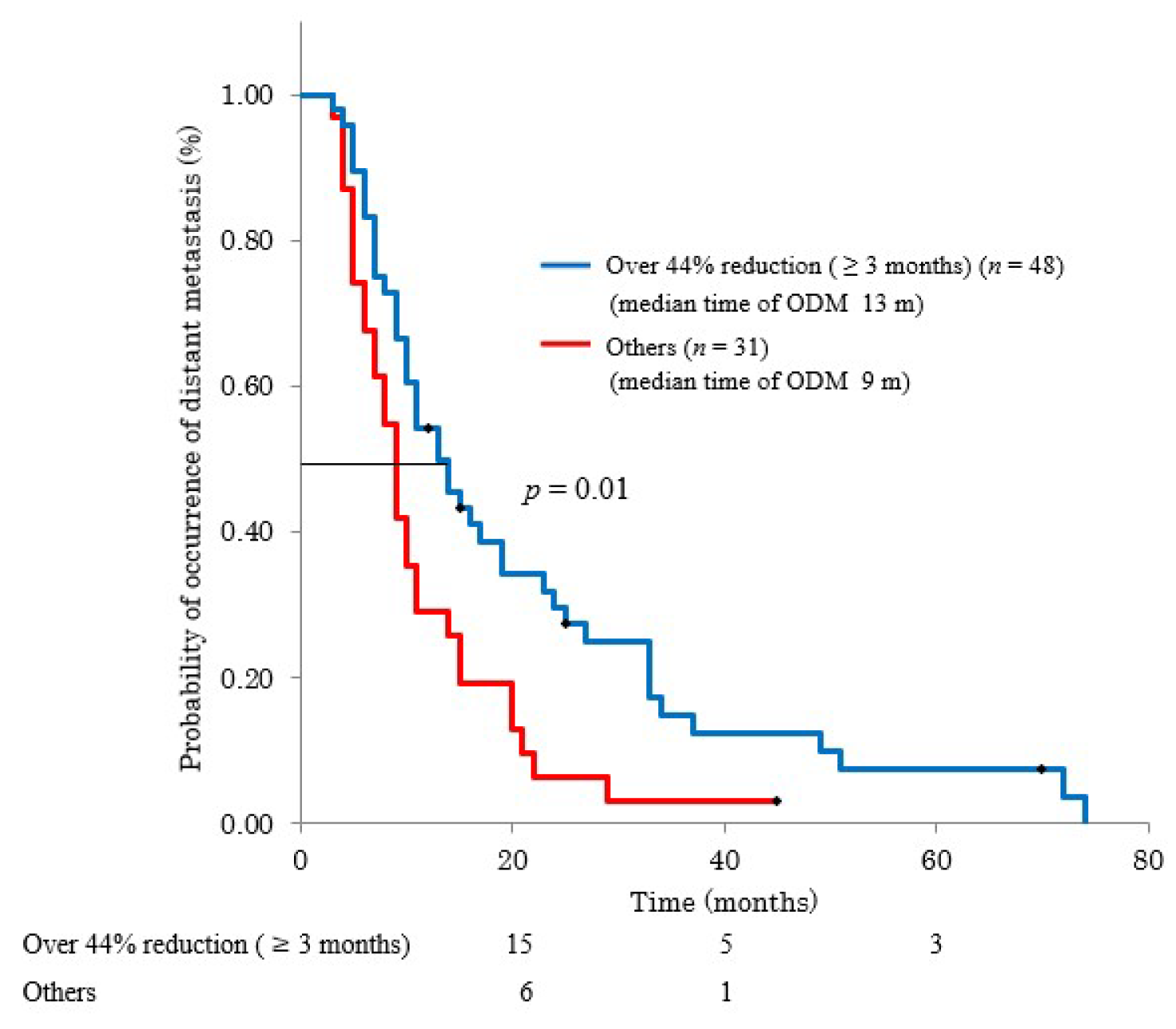
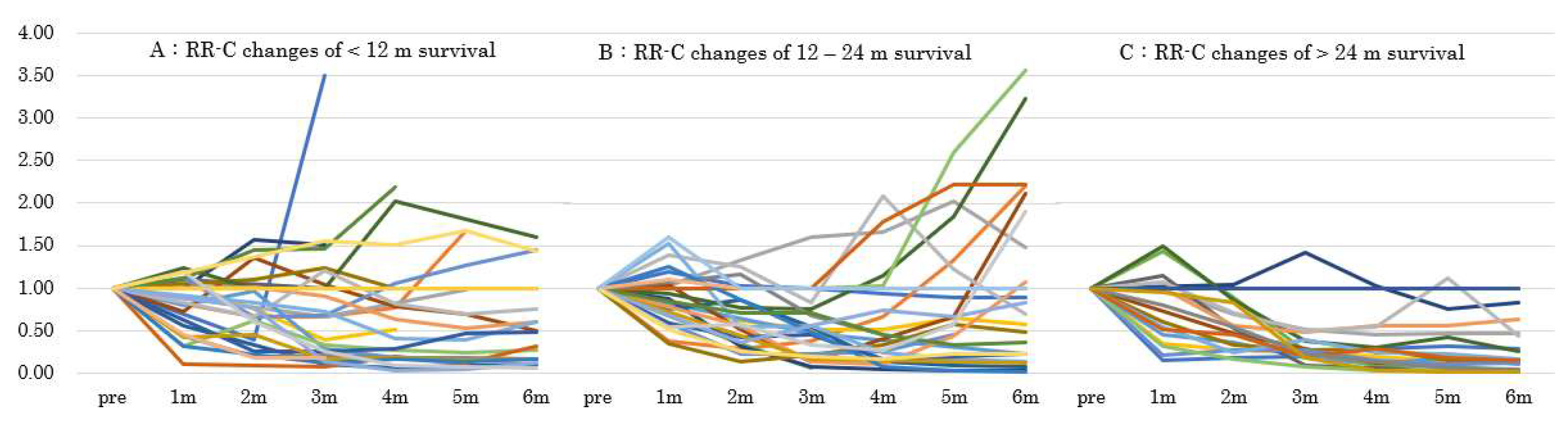
3.3. CA19-9 Reduction Duration and Overall Survival or Metastasis
3.4. Factors Related to Long-Term Survival
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Wolfgang, C.L.; Herman, J.M.; Laheru, D.A.; Klein, A.P.; Erdek, M.A.; Fishman, E.K.; Hruban, R.H. Recent progress in pancreatic cancer. CA Cancer J. Clin. 2013, 63, 318–348. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yoo, C.; Shin, S.H.; Kim, K.P.; Jeong, J.H.; Chang, H.M.; Kang, J.H.; Lee, S.S.; Park, D.H.; Song, T.J.; Seo, D.W.; et al. Clinical Outcomes of Conversion Surgery after Neoadjuvant Chemotherapy in Patients with Borderline Resectable and Locally Advanced Unresectable Pancreatic Cancer: A Single-Center, Retrospective Analysis. Cancers 2019, 11, 278. [Google Scholar] [CrossRef] [Green Version]
- Jang, J.Y.; Han, Y.; Lee, H.; Kim, S.W.; Kwon, W.; Lee, K.H.; Oh, D.Y.; Chie, E.K.; Lee, J.M.; Heo, J.S.; et al. Oncological Benefits of Neoadjuvant Chemoradiation With Gemcitabine Versus Upfront Surgery in Patients With Borderline Resectable Pancreatic Cancer: A Prospective, Randomized, Open-label, Multicenter Phase 2/3 Trial. Ann. Surg. 2018, 268, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Hackert, T.; Ulrich, A.; Büchler, M.W. Borderline resectable pancreatic cancer. Cancer Lett. 2016, 375, 231–237. [Google Scholar] [CrossRef]
- Satoi, S.; Yamaue, H.; Kato, K.; Takahashi, S.; Hirono, S.; Takeda, S.; Eguchi, H.; Sho, M.; Wada, K.; Shinchi, H.; et al. Role of adjuvant surgery for patients with initially unresectable pancreatic cancer with a long-term favorable response to non-surgical anti-cancer treatments: Results of a project study for pancreatic surgery by the Japanese Society of Hepato-Biliary-Pancreatic Surgery. J. Hepato-Biliary-Pancreat. Sci. 2013, 20, 590–600. [Google Scholar] [CrossRef]
- Rose, J.B.; Rocha, F.G.; Alseidi, A.; Biehl, T.; Moonka, R.; Ryan, J.A.; Lin, B.; Picozzi, V.; Helton, S. Extended neoadjuvant chemotherapy for borderline resectable pancreatic cancer demonstrates promising postoperative outcomes and survival. Ann. Surg. Oncol. 2014, 21, 1530–1537. [Google Scholar] [CrossRef]
- Wolfe, A.R.; Prabhakar, D.; Yildiz, V.O.; Cloyd, J.M.; Dillhoff, M.; Abushahin, L.; Alexandra Diaz, D.; Miller, E.D.; Chen, W.; Frankel, W.L.; et al. Neoadjuvant-modified FOLFIRINOX vs. nab-paclitaxel plus gemcitabine for borderline resectable or locally advanced pancreatic cancer patients who achieved surgical resection. Cancer Med. 2020, 9, 4711–4723. [Google Scholar] [CrossRef]
- Tsujimoto, A.; Sudo, K.; Nakamura, K.; Kita, E.; Hara, R.; Takayama, W.; Ishii, H.; Yamaguchi, T. Gemcitabine plus nab-paclitaxel for locally advanced or borderline resectable pancreatic cancer. Sci. Rep. 2019, 9, 16187. [Google Scholar] [CrossRef] [Green Version]
- Lee, W.; Park, Y.; Kwon, J.W.; Jun, E.; Song, K.B.; Lee, J.H.; Hwang, D.W.; Yoo, C.; Kim, K.P.; Jeong, J.H.; et al. Reduced and Normalized Carbohydrate Antigen 19-9 Concentrations after Neoadjuvant Chemotherapy Have Comparable Prognostic Performance in Patients with Borderline Resectable and Locally Advanced Pancreatic Cancer. J. Clin. Med. 2020, 9, 1477. [Google Scholar] [CrossRef] [PubMed]
- Boeck, S.; Stieber, P.; Holdenrieder, S.; Wilkowski, R.; Heinemann, V. Prognostic and therapeutic significance of carbohydrate antigen 19-9 as tumor marker in patients with pancreatic cancer. Oncology 2006, 70, 255–264. [Google Scholar] [CrossRef] [Green Version]
- Boone, B.A.; Steve, J.; Zenati, M.S.; Hogg, M.E.; Singhi, A.D.; Bartlett, D.L.; Zureikat, A.H.; Bahary, N.; Zeh, H.J., 3rd. Serum CA 19-9 response to neoadjuvant therapy is associated with outcome in pancreatic adenocarcinoma. Ann. Surg. Oncol. 2014, 21, 4351–4358. [Google Scholar] [CrossRef] [PubMed]
- Tzeng, C.W.; Balachandran, A.; Ahmad, M.; Lee, J.E.; Krishnan, S.; Wang, H.; Crane, C.H.; Wolff, R.A.; Varadhachary, G.R.; Pisters, P.W.; et al. Serum carbohydrate antigen 19-9 represents a marker of response to neoadjuvant therapy in patients with borderline resectable pancreatic cancer. HPB 2014, 16, 430–438. [Google Scholar] [CrossRef] [Green Version]
- Groot, V.P.; Gemenetzis, G.; Blair, A.B.; Rivero-Soto, R.J.; Yu, J.; Javed, A.A.; Burkhart, R.A.; Rinkes, I.; Molenaar, I.Q.; Cameron, J.L.; et al. Defining and Predicting Early Recurrence in 957 Patients With Resected Pancreatic Ductal Adenocarcinoma. Ann. Surg. 2019, 269, 1154–1162. [Google Scholar] [CrossRef]
- Klaiber, U.; Schnaidt, E.S.; Hinz, U.; Gaida, M.M.; Heger, U.; Hank, T.; Strobel, O.; Neoptolemos, J.P.; Mihaljevic, A.L.; Büchler, M.W.; et al. Prognostic Factors of Survival After Neoadjuvant Treatment and Resection for Initially Unresectable Pancreatic Cancer. Ann. Surg. 2021, 273, 154–162. [Google Scholar] [CrossRef]
- Chung, K.H.; Ryu, J.K.; Lee, B.S.; Jang, D.K.; Lee, S.H.; Kim, Y.T. Early decrement of serum carbohydrate antigen 19-9 predicts favorable outcome in advanced pancreatic cancer. J. Gastroenterol. Hepatol. 2016, 31, 506–512. [Google Scholar] [CrossRef]
- Nakai, Y.; Kawabe, T.; Isayama, H.; Sasaki, T.; Yagioka, H.; Yashima, Y.; Kogure, H.; Arizumi, T.; Togawa, O.; Ito, Y.; et al. CA 19-9 response as an early indicator of the effectiveness of gemcitabine in patients with advanced pancreatic cancer. Oncology 2008, 75, 120–126. [Google Scholar] [CrossRef]
- Hess, V.; Glimelius, B.; Grawe, P.; Dietrich, D.; Bodoky, G.; Ruhstaller, T.; Bajetta, E.; Saletti, P.; Figer, A.; Scheithauer, W.; et al. CA 19-9 tumour-marker response to chemotherapy in patients with advanced pancreatic cancer enrolled in a randomised controlled trial. Lancet. Oncol. 2008, 9, 132–138. [Google Scholar] [CrossRef]
- Reni, M.; Peretti, U.; Zanon, S.; Macchini, M.; Balzano, G.; Mazza, E.; Tamburrino, D.; Orsi, G.; Arcidiacono, P.G.; Falconi, M.; et al. Time to CA19-9 nadir: A clue for defining optimal treatment duration in patients with resectable pancreatic ductal adenocarcinoma. Cancer Chemother. Pharmacol. 2020, 85, 641–650. [Google Scholar] [CrossRef]
- Tempero, M.A.; Uchida, E.; Takasaki, H.; Burnett, D.A.; Steplewski, Z.; Pour, P.M. Relationship of carbohydrate antigen 19-9 and Lewis antigens in pancreatic cancer. Cancer Res. 1987, 47, 5501–5503. [Google Scholar]
- Tempero, M.A.; Malafa, M.P.; Al-Hawary, M.; Asbun, H.; Bain, A.; Behrman, S.W.; Benson, A.B., 3rd; Binder, E.; Cardin, D.B.; Cha, C.; et al. Pancreatic Adenocarcinoma, Version 2.2017, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Canc. Netw. 2017, 15, 1028–1061. [Google Scholar] [CrossRef] [PubMed]
- Eisenhauer, E.A.; Therasse, P.; Bogaerts, J.; Schwartz, L.H.; Sargent, D.; Ford, R.; Dancey, J.; Arbuck, S.; Gwyther, S.; Mooney, M.; et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur. J. Cancer 2009, 45, 228–247. [Google Scholar] [CrossRef] [PubMed]
- Aldakkak, M.; Christians, K.K.; Krepline, A.N.; George, B.; Ritch, P.S.; Erickson, B.A.; Johnston, F.M.; Evans, D.B.; Tsai, S. Pre-treatment carbohydrate antigen 19-9 does not predict the response to neoadjuvant therapy in patients with localized pancreatic cancer. HPB 2015, 17, 942–952. [Google Scholar] [CrossRef] [Green Version]
- Truty, M.J.; Kendrick, M.L.; Nagorney, D.M.; Smoot, R.L.; Cleary, S.P.; Graham, R.P.; Goenka, A.H.; Hallemeier, C.L.; Haddock, M.G.; Harmsen, W.S.; et al. Factors Predicting Response, Perioperative Outcomes, and Survival Following Total Neoadjuvant Therapy for Borderline/Locally Advanced Pancreatic Cancer. Ann. Surg. 2021, 273, 341–349. [Google Scholar] [CrossRef]
- Rose, J.B.; Edwards, A.M.; Rocha, F.G.; Clark, C.; Alseidi, A.A.; Biehl, T.R.; Lin, B.S.; Picozzi, V.J.; Helton, W.S. Sustained Carbohydrate Antigen 19-9 Response to Neoadjuvant Chemotherapy in Borderline Resectable Pancreatic Cancer Predicts Progression and Survival. Oncologist 2020, 25, 859–866. [Google Scholar] [CrossRef] [Green Version]
- Lopez, N.E.; Prendergast, C.; Lowy, A.M. Borderline resectable pancreatic cancer: Definitions and management. World J. Gastroenterol. 2014, 20, 10740–10751. [Google Scholar] [CrossRef] [PubMed]
- Hamada, C.; Okusaka, T.; Ikari, T.; Isayama, H.; Furuse, J.; Ishii, H.; Nakai, Y.; Imai, S.; Okamura, S. Efficacy and safety of gemcitabine plus S-1 in pancreatic cancer: A pooled analysis of individual patient data. Br. J. Cancer 2017, 116, 1544–1550. [Google Scholar] [CrossRef]
- Chen, Y.; Sun, X.J.; Jiang, T.H.; Mao, A.W. Combined radiochemotherapy in patients with locally advanced pancreatic cancer: A meta-analysis. World J. Gastroenterol. 2013, 19, 7461–7471. [Google Scholar] [CrossRef]
- Sudo, K.; Yamaguchi, T.; Ishihara, T.; Nakamura, K.; Hara, T.; Denda, T.; Tawada, K.; Imagumbai, T.; Araki, H.; Sakai, M.; et al. Phase II study of oral S-1 and concurrent radiotherapy in patients with unresectable locally advanced pancreatic cancer. Int. J. Radiat. Oncol. Biol. Phys. 2011, 80, 119–125. [Google Scholar] [CrossRef]
- Okusaka, T.; Ito, Y.; Ueno, H.; Ikeda, M.; Takezako, Y.; Morizane, C.; Kagami, Y.; Ikeda, H. Phase II study of radiotherapy combined with gemcitabine for locally advanced pancreatic cancer. Br. J. Cancer 2004, 91, 673–677. [Google Scholar] [CrossRef] [PubMed] [Green Version]

| Factors | n (%) | Median (Range) |
|---|---|---|
| Age, years | 68 (36–87) | |
| Sex, female/male | 32 (41)/47 (59) | |
| PS, 0/1,2 | 63 (80)/16 (20) | |
| Pre-CA19-9, U/mL, | 182 (10–21084) | |
| Location, Head/Body-tail | 41 (52)/38 (48) | |
| Tumor size, mm | 32 (12–64) | |
| Resectability, BR/LA | 22 (28)/57 (72) | |
| Invasion, (CA/SMA/CHA/PV) | 35 (44)/36 (45)/3 (4)/5 (7) | |
| 1st Treatment, CTx/CRTx | 63 (80)/16 (20) | |
| CTx regimen | GnP 41, Gem 13, GS 4, S1 3, FFX 2 | |
| CRTx regimen | Gem + S1 + RT | |
| PFS, month | 8 (3–74) | |
| OS, month | 17 (4–128) |
| Variable | Univariate Analysis | Multivariate Analysis | |||||
|---|---|---|---|---|---|---|---|
| LS (n = 22) | not LS (n = 57) | p Value | Odds Ratio | 95% CI | p Value | ||
| Age (year) | 0.80 | ||||||
| ≥65/<65 | 14/8 | 38/19 | |||||
| Gender | 0.30 | ||||||
| Male/Female | 15/7 | 32/25 | |||||
| Performance status | 0.07 | 0.35 | 0.07–1.81 | 0.21 | |||
| ≥1/0 | 2/20 | 16/41 | |||||
| Location | 0.80 | ||||||
| Head/Body-Tail | 12/10 | 29/28 | |||||
| Tumor Size (cm) | 0.80 | ||||||
| ≥3/<3 | 14/8 | 35/22 | |||||
| Initial Resectability | 0.9 | ||||||
| BR/LA | 6/16 | 16/41 | |||||
| Treatment | 0.03 | 2.63 | 0.83–8.26 | 0.09 | |||
| CRTx/CTx | 10/12 | 12/45 | |||||
| CA19-9 | |||||||
| Initial level | 1.00 | ||||||
| ≥37/<37 | 18/4 | 45/12 | |||||
| Reduction cases | 0.09 | ||||||
| ≥1/<1 | 0/22 | 8/49 | |||||
| Duration of 44% reduction | <0.01 | 5.75 | 1.47–22.36 | 0.01 | |||
| ≥3 m/Others | 19/3 | 29/28 | |||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tomishima, K.; Ishii, S.; Fujisawa, T.; Ikemura, M.; Ota, H.; Kabemura, D.; Ushio, M.; Fukuma, T.; Takahashi, S.; Yamagata, W.; et al. Duration of Reduced CA19-9 Levels Is a Better Prognostic Factor Than Its Rate of Reduction for Unresectable Locally Advanced Pancreatic Cancer. Cancers 2021, 13, 4224. https://doi.org/10.3390/cancers13164224
Tomishima K, Ishii S, Fujisawa T, Ikemura M, Ota H, Kabemura D, Ushio M, Fukuma T, Takahashi S, Yamagata W, et al. Duration of Reduced CA19-9 Levels Is a Better Prognostic Factor Than Its Rate of Reduction for Unresectable Locally Advanced Pancreatic Cancer. Cancers. 2021; 13(16):4224. https://doi.org/10.3390/cancers13164224
Chicago/Turabian StyleTomishima, Ko, Shigeto Ishii, Toshio Fujisawa, Muneo Ikemura, Hiroto Ota, Daishi Kabemura, Mako Ushio, Taito Fukuma, Sho Takahashi, Wataru Yamagata, and et al. 2021. "Duration of Reduced CA19-9 Levels Is a Better Prognostic Factor Than Its Rate of Reduction for Unresectable Locally Advanced Pancreatic Cancer" Cancers 13, no. 16: 4224. https://doi.org/10.3390/cancers13164224
APA StyleTomishima, K., Ishii, S., Fujisawa, T., Ikemura, M., Ota, H., Kabemura, D., Ushio, M., Fukuma, T., Takahashi, S., Yamagata, W., Takasaki, Y., Suzuki, A., Ito, K., Saito, H., Nagahara, A., & Isayama, H. (2021). Duration of Reduced CA19-9 Levels Is a Better Prognostic Factor Than Its Rate of Reduction for Unresectable Locally Advanced Pancreatic Cancer. Cancers, 13(16), 4224. https://doi.org/10.3390/cancers13164224







