Treatment-Free Remission in Chronic Myeloid Leukemia: Can We Identify Prognostic Factors?
Simple Summary
Abstract
1. Introduction
2. The Concept of TFR
3. Clinical Trials of TFR
4. Mechanisms behind TFR
5. Predicting Factors for the Success or the Failure of TFR
5.1. Duration of TKI
5.2. Deep Molecular Response
5.3. BCRABL1 Transcript Type and Detection
5.4. Immunity Effect
5.5. TKI Resistance
5.6. Prognostic Scoring—Sokal, ELTS, and Eutos Score
5.7. Age
5.8. Prior Interferon-Alpha (IFN-a) Therapy
5.9. Re-Initiating TKI and Second Attempt TFR
5.10. Abrupt Discontinuation versus Gradual Discontinuation
6. Guidelines and Clinical Practice
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- O’Hare, T.; Zabriskie, M.S.; Eiring, A.M.; Deininger, M.W. Pushing the limits of targeted therapy in chronic myeloid leukaemia. Nat. Rev. Cancer 2012, 12, 513–526. [Google Scholar] [CrossRef]
- Nowell, P.C. The minute chromosome (Phl) in chronic granulocytic leukemia. Blut 1962, 8, 65–66. [Google Scholar] [CrossRef]
- Rowley, J.D. A New Consistent Chromosomal Abnormality in Chronic Myelogenous Leukaemia identified by Quinacrine Fluorescence and Giemsa Staining. Nature 1973, 243, 290–293. [Google Scholar] [CrossRef] [PubMed]
- Lugo, T.G.; Pendergast, A.M.; Muller, A.J.; Witte, O.N. Tyrosine kinase activity and transformation potency of bcr-abl oncogene products. Science 1990, 247, 1079–1082. [Google Scholar] [CrossRef] [PubMed]
- Granatowicz, A.; Piatek, C.I.; Moschiano, E.; El-Hemaidi, I.; Armitage, J.D.; Akhtari, M. An Overview and Update of Chronic Myeloid Leukemia for Primary Care Physicians. Korean J. Fam. Med. 2015, 36, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Kesarwani, M.; Kincaid, Z.; Gomaa, A.; Huber, E.; Rohrabaugh, S.; Siddiqui, Z.; Bouso, M.F.; Latif, T.; Xu, M.; Komurov, K.; et al. Targeting c-FOS and DUSP1 abrogates intrinsic resistance to tyrosine-kinase inhibitor therapy in BCR-ABL-induced leukemia. Nat. Med. 2017, 23, 472–482. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, A.; Helgason, G.V.; Schemionek, M.; Zhang, B.; Myssina, S.; Allan, E.K.; Nicolini, F.E.; Müller-Tidow, C.; Bhatia, R.; Brunton, V.G.; et al. Chronic myeloid leukemia stem cells are not dependent on Bcr-Abl kinase activity for their survival. Blood 2012, 119, 1501–1510. [Google Scholar] [CrossRef]
- Björkholm, M.; Ohm, L.; Eloranta, S.; Derolf, A.; Hultcrantz, M.; Sjöberg, J.; Andersson, T.; Höglund, M.; Richter, J.; Landgren, O.; et al. Success story of targeted therapy in chronic myeloid leukemia: A population-based study of patients diagnosed in Sweden from 1973 to 2008. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2011, 29, 2514–2520. [Google Scholar] [CrossRef]
- Kantarjian, H.; O’Brien, S.; Jabbour, E.; Garcia-Manero, G.; Quintas-Cardama, A.; Shan, J.; Rios, M.B.; Ravandi, F.; Faderl, S.; Kadia, T.; et al. Improved survival in chronic myeloid leukemia since the introduction of imatinib therapy: A single-institution historical experience. Blood 2012, 119, 1981–1987. [Google Scholar] [CrossRef] [PubMed]
- Bower, H.; Björkholm, M.; Dickman, P.W.; Höglund, M.; Lambert, P.C.; Andersson, T.M.-L. Life Expectancy of Patients With Chronic Myeloid Leukemia Approaches the Life Expectancy of the General Population. J. Clin. Oncol. 2016, 34, 2851–2857. [Google Scholar] [CrossRef] [PubMed]
- Gambacorti-Passerini, C.; Antolini, L.; Mahon, F.X.; Guilhot, F.; Deininger, M.; Fava, C.; Nagler, A.; Della Casa, C.M.; Morra, E.; Abruzzese, E.; et al. Multicenter independent assessment of outcomes in chronic myeloid leukemia patients treated with imatinib. J. Natl. Cancer Inst. 2011, 103, 553–561. [Google Scholar] [CrossRef]
- Viganò, I.; Di Giacomo, N.; Bozzani, S.; Antolini, L.; Piazza, R.; Gambacorti Passerini, C. First-line treatment of 102 chronic myeloid leukemia patients with imatinib: A long-term single institution analysis. Am. J. Hematol 2014, 89, E184–E187. [Google Scholar] [CrossRef]
- Baccarani, M.; Abruzzese, E.; Accurso, V.; Albano, F.; Annunziata, M.; Barulli, S.; Beltrami, G.; Bergamaschi, M.; Binotto, G.; Bocchia, M.; et al. Managing chronic myeloid leukemia for treatment-free remission: A proposal from the GIMEMA CML WP. Blood Adv. 2019, 3, 4280–4290. [Google Scholar] [CrossRef]
- Mahon, F.X. Treatment-free remission in CML: Who, how, and why? Hematol. Am. Soc. Hematol. Educ. Program. 2017, 2017, 102–109. [Google Scholar] [CrossRef]
- Etienne, G.; Dulucq, S.; Nicolini, F.-E.; Morisset, S.; Fort, M.-P.; Schmitt, A.; Etienne, M.; Hayette, S.; Lippert, E.; Bureau, C.; et al. Achieving deeper molecular response is associated with a better clinical outcome in chronic myeloid leukemia patients on imatinib front-line therapy. Haematologica 2014, 99, 458–464. [Google Scholar] [CrossRef]
- Falchi, L.; Kantarjian, H.M.; Wang, X.; Verma, D.; Quintás-Cardama, A.; O’Brien, S.; Jabbour, E.J.; Ravandi-Kashani, F.; Borthakur, G.; Garcia-Manero, G.; et al. Significance of deeper molecular responses in patients with chronic myeloid leukemia in early chronic phase treated with tyrosine kinase inhibitors. Am. J. Hematol. 2013, 88, 1024–1029. [Google Scholar] [CrossRef] [PubMed]
- Hehlmann, R.; Müller, M.C.; Lauseker, M.; Hanfstein, B.; Fabarius, A.; Schreiber, A.; Proetel, U.; Pletsch, N.; Pfirrmann, M.; Haferlach, C.; et al. Deep molecular response is reached by the majority of patients treated with imatinib, predicts survival, and is achieved more quickly by optimized high-dose imatinib: Results from the randomized CML-study IV. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2014, 32, 415–423. [Google Scholar] [CrossRef] [PubMed]
- Baccarani, M.; Cortes, J.; Pane, F.; Niederwieser, D.; Saglio, G.; Apperley, J.; Cervantes, F.; Deininger, M.; Gratwohl, A.; Guilhot, F.; et al. Chronic myeloid leukemia: An update of concepts and management recommendations of European LeukemiaNet. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2009, 27, 6041–6051. [Google Scholar] [CrossRef]
- Cross, N.C.P.; White, H.E.; Müller, M.C.; Saglio, G.; Hochhaus, A. Standardized definitions of molecular response in chronic myeloid leukemia. Leukemia 2012, 26, 2172–2175. [Google Scholar] [CrossRef] [PubMed]
- Saußele, S.; Richter, J.; Hochhaus, A.; Mahon, F.X. The concept of treatment-free remission in chronic myeloid leukemia. Leukemia 2016, 30, 1638–1647. [Google Scholar] [CrossRef]
- Goldman, J.; Gordon, M. Why do chronic myelogenous leukemia stem cells survive allogeneic stem cell transplantation or imatinib: Does it really matter? Leuk. Lymphoma 2006, 47, 1–7. [Google Scholar] [CrossRef]
- Ross, D.M.; Hughes, T.P. How I determine if and when to recommend stopping tyrosine kinase inhibitor treatment for chronic myeloid leukaemia. Br. J. Haematol. 2014, 166, 3–11. [Google Scholar] [CrossRef]
- Saglio, G.; Gale, R.P. Prospects for achieving treatment-free remission in chronic myeloid leukaemia. Br. J. Haematol. 2020, 190, 318–327. [Google Scholar] [CrossRef]
- Guérin, A.; Chen, L.; Ionescu-Ittu, R.; Marynchenko, M.; Nitulescu, R.; Hiscock, R.; Keir, C.; Wu, E.Q. Impact of low-grade adverse events on health-related quality of life in adult patients receiving imatinib or nilotinib for newly diagnosed Philadelphia chromosome positive chronic myelogenous leukemia in chronic phase. Curr. Med. Res. Opin. 2014, 30, 2317–2328. [Google Scholar] [CrossRef] [PubMed]
- Williams, L.A.; Garcia Gonzalez, A.G.; Ault, P.; Mendoza, T.R.; Sailors, M.L.; Williams, J.L.; Huang, F.; Nazha, A.; Kantarjian, H.M.; Cleeland, C.S.; et al. Measuring the symptom burden associated with the treatment of chronic myeloid leukemia. Blood 2013, 122, 641–647. [Google Scholar] [CrossRef]
- Efficace, F.; Baccarani, M.; Breccia, M.; Alimena, G.; Rosti, G.; Cottone, F.; Deliliers, G.L.; Baratè, C.; Rossi, A.R.; Fioritoni, G.; et al. Health-related quality of life in chronic myeloid leukemia patients receiving long-term therapy with imatinib compared with the general population. Blood 2011, 118, 4554–4560. [Google Scholar] [CrossRef] [PubMed]
- Lou, J.; Huang, J.; Wang, Z.; Wen, B.; Tu, C.; Huang, W.; Zhai, Z.; Du, X. Chronic myeloid leukemia patients and treatment-free remission attitudes: A multicenter survey. Patient Prefer. Adherence 2018, 12, 1025. [Google Scholar] [CrossRef]
- Experts in Chronic Myeloid Leukemia. The price of drugs for chronic myeloid leukemia (CML) is a reflection of the unsustainable prices of cancer drugs: From the perspective of a large group of CML experts. Blood 2013, 121, 4439–4442. [Google Scholar] [CrossRef]
- Mahon, F.X. Is going for cure in chronic myeloid leukemia possible and justifiable? Hematol. Am. Soc. Hematol. Educ. Program. 2012, 2012, 122–128. [Google Scholar] [CrossRef]
- Rousselot, P.; Huguet, F.; Rea, D.; Legros, L.; Cayuela, J.M.; Maarek, O.; Blanchet, O.; Marit, G.; Gluckman, E.; Reiffers, J.; et al. Imatinib mesylate discontinuation in patients with chronic myelogenous leukemia in complete molecular remission for more than 2 years. Blood 2007, 109, 58–60. [Google Scholar] [CrossRef] [PubMed]
- Thielen, N.; van der Holt, B.; Cornelissen, J.J.; Verhoef, G.E.; Gussinklo, T.; Biemond, B.J.; Daenen, S.M.; Deenik, W.; van Marwijk Kooy, R.; Petersen, E.; et al. Imatinib discontinuation in chronic phase myeloid leukaemia patients in sustained complete molecular response: A randomised trial of the Dutch-Belgian Cooperative Trial for Haemato-Oncology (HOVON). Eur. J. Cancer 2013, 49, 3242–3246. [Google Scholar] [CrossRef] [PubMed]
- Hannan, E.L. Randomized Clinical Trials and Observational Studies. Guidel. Assess. Respective Strengths Limit. 2008, 1, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Sharf, G.; Marin, C.; Bradley, J.A.; Pemberton-Whiteley, Z.; Bombaci, F.; Christensen, R.I.O.; Gouimi, B.; Deekes, N.B.; Daban, M.; Geissler, J. Treatment-free remission in chronic myeloid leukemia: The patient perspective and areas of unmet needs. Leukemia 2020, 34, 2102–2112. [Google Scholar] [CrossRef]
- Saussele, S.; Richter, J.; Guilhot, J.; Gruber, F.X.; Hjorth-Hansen, H.; Almeida, A.; Janssen, J.J.W.M.; Mayer, J.; Koskenvesa, P.; Panayiotidis, P.; et al. Discontinuation of tyrosine kinase inhibitor therapy in chronic myeloid leukaemia (EURO-SKI): A prespecified interim analysis of a prospective, multicentre, non-randomised, trial. Lancet Oncol. 2018, 19, 747–757. [Google Scholar] [CrossRef]
- Mahon, F.X.; Rea, D.; Guilhot, J.; Guilhot, F.; Huguet, F.; Nicolini, F.; Legros, L.; Charbonnier, A.; Guerci, A.; Varet, B.; et al. Discontinuation of imatinib in patients with chronic myeloid leukaemia who have maintained complete molecular remission for at least 2 years: The prospective, multicentre Stop Imatinib (STIM) trial. Lancet Oncol. 2010, 11, 1029–1035. [Google Scholar] [CrossRef]
- Etienne, G.; Guilhot, J.; Rea, D.; Rigal-Huguet, F.; Nicolini, F.; Charbonnier, A.; Guerci-Bresler, A.; Legros, L.; Varet, B.; Gardembas, M.; et al. Long-Term Follow-Up of the French Stop Imatinib (STIM1) Study in Patients With Chronic Myeloid Leukemia. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2017, 35, 298–305. [Google Scholar] [CrossRef]
- Ross, D.M.; Branford, S.; Seymour, J.F.; Schwarer, A.P.; Arthur, C.; Yeung, D.T.; Dang, P.; Goyne, J.M.; Slader, C.; Filshie, R.J. Safety and efficacy of imatinib cessation for CML patients with stable undetectable minimal residual disease: Results from the TWISTER study. Blood 2013, 122, 515–522. [Google Scholar] [CrossRef]
- Rousselot, P.; Charbonnier, A.; Cony-Makhoul, P.; Agape, P.; Nicolini, F.E.; Varet, B.R.; Gardembas, M.; Etienne, G.; Rea, D.; Roy, L.; et al. Loss of Major Molecular Response as a Trigger for Restarting Tyrosine Kinase Inhibitor Therapy in CP-CML Patients Who Have Stopped Imatinib after Durable Undetectable Minimal Residual Disease. Blood 2013, 122, 381. [Google Scholar] [CrossRef] [PubMed]
- Diral, E.; Mori, S.; Antolini, L.; Abruzzese, E.; Le Coutre, P.; Martino, B.; Pungolino, E.; Elena, C.; Bergamaschi, M.; Assouline, S.; et al. Increased tumor burden in patients with chronic myeloid leukemia after 36 months of imatinib discontinuation. Blood 2020, 136, 2237–2240. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.E.; Choi, S.Y.; Song, H.Y.; Kim, S.H.; Choi, M.Y.; Park, J.S.; Kim, H.J.; Kim, S.H.; Zang, D.Y.; Oh, S.; et al. Imatinib withdrawal syndrome and longer duration of imatinib have a close association with a lower molecular relapse after treatment discontinuation: The KID study. Haematologica 2016, 101, 717–723. [Google Scholar] [CrossRef]
- Imagawa, J.; Tanaka, H.; Okada, M.; Nakamae, H.; Hino, M.; Murai, K.; Ishida, Y.; Kumagai, T.; Sato, S.; Ohashi, K.; et al. Discontinuation of dasatinib in patients with chronic myeloid leukaemia who have maintained deep molecular response for longer than 1 year (DADI trial): A multicentre phase 2 trial. Lancet Haematol. 2015, 2, e528–e535. [Google Scholar] [CrossRef]
- Kimura, S.; Imagawa, J.; Murai, K.; Hino, M.; Kitawaki, T.; Okada, M.; Tanaka, H.; Shindo, M.; Kumagai, T.; Ikezoe, T.; et al. Treatment-free remission after first-line dasatinib discontinuation in patients with chronic myeloid leukaemia (first-line DADI trial): A single-arm, multicentre, phase 2 trial. Lancet Haematol. 2020, 7, e218–e225. [Google Scholar] [CrossRef]
- Hochhaus, A.; Masszi, T.; Giles, F.J.; Radich, J.P.; Ross, D.M.; Gómez Casares, M.T.; Hellmann, A.; Stentoft, J.; Conneally, E.; García-Gutiérrez, V.; et al. Treatment-free remission following frontline nilotinib in patients with chronic myeloid leukemia in chronic phase: Results from the ENESTfreedom study. Leukemia 2017, 31, 1525–1531. [Google Scholar] [CrossRef]
- Yhim, H.Y.; Lee, N.R.; Song, E.K.; Yim, C.Y.; Jeon, S.Y.; Shin, S.; Kim, J.A.; Kim, H.S.; Cho, E.H.; Kwak, J.Y. Imatinib mesylate discontinuation in patients with chronic myeloid leukemia who have received front-line imatinib mesylate therapy and achieved complete molecular response. Leuk. Res. 2012, 36, 689–693. [Google Scholar] [CrossRef] [PubMed]
- Fava, C.; Rege-Cambrin, G.; Dogliotti, I.; Cerrano, M.; Berchialla, P.; Dragani, M.; Rosti, G.; Castagnetti, F.; Gugliotta, G.; Martino, B.; et al. Observational study of chronic myeloid leukemia Italian patients who discontinued tyrosine kinase inhibitors in clinical practice. Haematologica 2019, 104, 1589–1596. [Google Scholar] [CrossRef] [PubMed]
- Mahon, F.-X.; Nicolini, F.E.; Noël, M.-P.; Escoffre, M.; Charbonnier, A.; Rea, D.; Dubruille, V.; Varet, B.R.; Legros, L.; Guerci, A.; et al. Preliminary Report Of The STIM2 Study: A Multicenter Stop Imatinib Trial For Chronic Phase Chronic Myeloid Leukemia De Novo Patients On Imatinib. Blood 2013, 122, 654. [Google Scholar] [CrossRef]
- Matsuki, E.; Ono, Y.; Tonegawa, K.; Sakurai, M.; Kunimoto, H.; Ishizawa, J.; Hashimoto, N.; Shimizu, T.; Yamane, A.; Matsushita, M.; et al. Detailed Investigation On Characteristics of Japanese Patients with Chronic Phase CML Who Achieved a Durable CMR After Discontinuation of Imatinib—An Updated Result of the Keio STIM Study. Blood 2012, 120, 2788. [Google Scholar] [CrossRef]
- Pagnano, K.B.; Seguro, F.S.; Miranda, E.C.; Lopes, A.B.P.; Abdo, A.; Delamain, M.T.; Pereira, T.; Duarte, G.B.; Santos, F.M.; Vianna, J.C.N.; et al. Duration of Major Molecular Response and Discontinuation in Deep Molecular Response (MR4.5) Were Associated with Longer Treatment-Free Survival after Imatinib Discontinuation—Results from Two Prospective Brazilian Trials. Blood 2019, 134, 1655. [Google Scholar] [CrossRef]
- Kim, D.D.H.; Bence-Bruckler, I.; Forrest, D.L.; Savoie, M.L.; Couban, S.; Busque, L.; Delage, R.; Laneuville, P.; Liew, E.; Xenocostas, A.; et al. Treatment-Free Remission Accomplished By Dasatinib (TRAD): Preliminary Results of the Pan-Canadian Tyrosine Kinase Inhibitor Discontinuation Trial. Blood 2016, 128, 1922. [Google Scholar] [CrossRef]
- Takahashi, N.; Tauchi, T.; Kitamura, K.; Miyamura, K.; Saburi, Y.; Hatta, Y.; Miyata, Y.; Kobayashi, S.; Usuki, K.; Matsumura, I.; et al. Deeper molecular response is a predictive factor for treatment-free remission after imatinib discontinuation in patients with chronic phase chronic myeloid leukemia: The JALSG-STIM213 study. Int. J. Hematol. 2018, 107, 185–193. [Google Scholar] [CrossRef]
- Rea, D.; Nicolini, F.E.; Tulliez, M.; Guilhot, F.; Guilhot, J.; Guerci-Bresler, A.; Gardembas, M.; Coiteux, V.; Guillerm, G.; Legros, L.; et al. Discontinuation of dasatinib or nilotinib in chronic myeloid leukemia: Interim analysis of the STOP 2G-TKI study. Blood 2017, 129, 846–854. [Google Scholar] [CrossRef] [PubMed]
- Kumagai, T.; Nakaseko, C.; Nishiwaki, K.; Yoshida, C.; Ohashi, K.; Takezako, N.; Takano, H.; Kouzai, Y.; Murase, T.; Matsue, K.; et al. Dasatinib cessation after deep molecular response exceeding 2 years and natural killer cell transition during dasatinib consolidation. Cancer Sci. 2018, 109, 182–192. [Google Scholar] [CrossRef]
- Shah, N.P.; García-Gutiérrez, V.; Jiménez-Velasco, A.; Larson, S.; Saussele, S.; Rea, D.; Mahon, F.-X.; Levy, M.Y.; Gómez-Casares, M.T.; Pane, F.; et al. Dasatinib discontinuation in patients with chronic-phase chronic myeloid leukemia and stable deep molecular response: The DASFREE study. Leuk. Lymphoma 2020, 61, 650–659. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, N.; Nishiwaki, K.; Nakaseko, C.; Aotsuka, N.; Sano, K.; Ohwada, C.; Kuroki, J.; Kimura, H.; Tokuhira, M.; Mitani, K.; et al. Treatment-free remission after two-year consolidation therapy with nilotinib in patients with chronic myeloid leukemia: STAT2 trial in Japan. Haematologica 2018, 103, 1835–1842. [Google Scholar] [CrossRef]
- Fujisawa, S.; Ueda, Y.; Usuki, K.; Kobayashi, H.; Kondo, E.; Doki, N.; Nakao, T.; Kanda, Y.; Kosugi, N.; Kosugi, H.; et al. Feasibility of the imatinib stop study in the Japanese clinical setting: Delightedly overcome CML expert stop TKI trial (DOMEST Trial). Int. J. Clin. Oncol. 2019, 24, 445–453. [Google Scholar] [CrossRef] [PubMed]
- Corbin, A.S.; Agarwal, A.; Loriaux, M.; Cortes, J.; Deininger, M.W.; Druker, B.J. Human chronic myeloid leukemia stem cells are insensitive to imatinib despite inhibition of BCR–ABL activity. J. Clin. Investig. 2011, 121, 396–409. [Google Scholar] [CrossRef]
- Chomel, J.C.; Bonnet, M.L.; Sorel, N.; Bertrand, A.; Meunier, M.C.; Fichelson, S.; Melkus, M.; Bennaceur-Griscelli, A.; Guilhot, F.; Turhan, A.G. Leukemic stem cell persistence in chronic myeloid leukemia patients with sustained undetectable molecular residual disease. Blood 2011, 118, 3657–3660. [Google Scholar] [CrossRef]
- Chu, S.; McDonald, T.; Lin, A.; Chakraborty, S.; Huang, Q.; Snyder, D.S.; Bhatia, R. Persistence of leukemia stem cells in chronic myelogenous leukemia patients in prolonged remission with imatinib treatment. Blood 2011, 118, 5565–5572. [Google Scholar] [CrossRef]
- Roeder, I.; Horn, M.; Glauche, I.; Hochhaus, A.; Mueller, M.C.; Loeffler, M. Dynamic modeling of imatinib-treated chronic myeloid leukemia: Functional insights and clinical implications. Nat. Med. 2006, 12, 1181–1184. [Google Scholar] [CrossRef]
- Kanodia, S.; Wieder, E.; Lu, S.; Talpaz, M.; Alatrash, G.; Clise-Dwyer, K.; Molldrem, J.J. PR1-specific T cells are associated with unmaintained cytogenetic remission of chronic myelogenous leukemia after interferon withdrawal. PLoS ONE 2010, 5, e11770. [Google Scholar] [CrossRef]
- Talpaz, M.; Mercer, J.; Hehlmann, R. The interferon-alpha revival in CML. Ann. Hematol. 2015, 94 (Suppl. 2), S195–S207. [Google Scholar] [CrossRef]
- Burchert, A.; Inselmann, S.; Saussele, S.; Dietz, C.T.; Müller, M.C.; Eigendorff, E.; Brümmendorf, T.H.; Waller, C.; Dengler, J.; Goebeler, M.E.; et al. Frequency of CTLA-4 Receptor Ligand (CD86, B7.2) -Positive Plasmacytoid Dendritic Cells Predicts Risk of Disease Recurrence after Tyrosine-Kinase Inhibitor Discontinuation in Chronic Myeloid Leukemia: Results from a Prospective Substudy of the Euroski Trial. Blood 2015, 126, 599. [Google Scholar] [CrossRef]
- Hughes, A.; Tang, C.; Clarson, J.; Vidovic, L.; Hughes, T.P.; Yong, A.S. Chronic Myeloid Leukemia Patients with Deep Molecular Responses to Tyrosine Kinase Inhibitors Have Increased Effector Natural Killer and Cytotoxic T Cell Immune Responses to Leukaemia-Associated Antigens and Concomitant Reduced Immune Suppressors. Blood 2015, 126, 18. [Google Scholar] [CrossRef]
- Ilander, M.M.; Olsson-Strömberg, U.; Lähteenmäki, H.; Kasanen, T.; Koskenvesa, P.; Söderlund, S.; Hoglund, M.; Markevärn, B.; Själander, A.; Lofti, K.; et al. Disease Relapse After TKI Discontinuation in CML Is Related Both To Low Number and Impaired Function Of NK-Cells: Data From Euro-SKI. Blood 2013, 122, 379. [Google Scholar] [CrossRef]
- Gale, R.P.; Opelz, G. Is there immune surveillance against chronic myeloid leukaemia? Possibly, but not much. Leuk. Res. 2017, 57, 109–111. [Google Scholar] [CrossRef] [PubMed]
- Folley, J.H.; Borges, W.; Yamawaki, T. Incidence of leukemia in survivors of the atomic bomb in Hiroshima and Nagasaki, Japan. Am. J. Med. 1952, 13, 311–321. [Google Scholar] [CrossRef]
- Hsu, W.L.; Preston, D.L.; Soda, M.; Sugiyama, H.; Funamoto, S.; Kodama, K.; Kimura, A.; Kamada, N.; Dohy, H.; Tomonaga, M.; et al. The incidence of leukemia, lymphoma and multiple myeloma among atomic bomb survivors: 1950–2001. Radiat. Res. 2013, 179, 361–382. [Google Scholar] [CrossRef] [PubMed]
- Gale, R.P.; Hochhaus, A. Therapy-free remission in chronic myeloid leukemia: Possible mechanism. Expert Rev. Hematol. 2018, 11, 269–272. [Google Scholar] [CrossRef] [PubMed]
- Cortes, J.; O’Brien, S.; Kantarjian, H. Discontinuation of imatinib therapy after achieving a molecular response. Blood 2004, 104, 2204–2205. [Google Scholar] [CrossRef] [PubMed]
- Mauro, M.J.; Druker, B.J.; Maziarz, R.T. Divergent clinical outcome in two CML patients who discontinued imatinib therapy after achieving a molecular remission. Leuk. Res. 2004, 28, 71–73. [Google Scholar] [CrossRef]
- Merante, S.; Orlandi, E.; Bernasconi, P.; Calatroni, S.; Boni, M.; Lazzarino, M. Outcome of four patients with chronic myeloid leukemia after imatinib mesylate discontinuation. Haematologica 2005, 90, 979. [Google Scholar] [PubMed]
- Takahashi, N.; Kyo, T.; Maeda, Y.; Sugihara, T.; Usuki, K.; Kawaguchi, T.; Usui, N.; Okamoto, S.; Ohe, Y.; Ohtake, S.; et al. Discontinuation of imatinib in Japanese patients with chronic myeloid leukemia. Haematologica 2012, 97, 903–906. [Google Scholar] [CrossRef] [PubMed]
- Deininger, M.W.; Goldman, J.M.; Melo, J.V. The molecular biology of chronic myeloid leukemia. Blood 2000, 96, 3343–3356. [Google Scholar] [CrossRef] [PubMed]
- Morris, S.W.; Daniel, L.; Ahmed, C.M.; Elias, A.; Lebowitz, P. Relationship of bcr breakpoint to chronic phase duration, survival, and blast crisis lineage in chronic myelogenous leukemia patients presenting in early chronic phase. Blood 1990, 75, 2035–2041. [Google Scholar] [CrossRef]
- Polampalli, S.; Choughule, A.; Negi, N.; Shinde, S.; Baisane, C.; Amre, P.; Subramanian, P.G.; Gujral, S.; Prabhash, K.; Parikh, P. Analysis and comparison of clinicohematological parameters and molecular and cytogenetic response of two Bcr/Abl fusion transcripts. Genet. Mol. Res. 2008, 7, 1138–1149. [Google Scholar] [CrossRef] [PubMed]
- Rosas-Cabral, A.; Martínez-Mancilla, M.; Ayala-Sánchez, M.; Vela-Ojeda, J.; Bahena-Reséndiz, P.; Vadillo-Buenfil, M.; Aviña-Zubieta, J.; Salazar-Exaire, D.; Miranda-Peralta, E.; Marroquín, A.; et al. Analysis of Bcr–abl type transcript and its relationship with platelet count in Mexican patients with chronic myeloid leukemia. Gac. Méd. México 2003, 139, 553–559. [Google Scholar]
- Meissner, R.D.V.; Dias, P.M.B.; Covas, D.T.; Job, F.; Leite, M.; Nardi, N.B. A polymorphism in exon b2 of the major breakpoint cluster region (M-bcr) identified in chronic myeloid leukaemia patients. Br. J. Haematol. 1998, 103, 224–226. [Google Scholar] [CrossRef]
- Perego, R.A.; Costantini, M.; Cornacchini, G.; Gargantini, L.; Bianchi, C.; Pungolino, E.; Rovida, E.; Morra, E. The possible influences of B2A2 and B3A2 BCR/ABL protein structure on thrombopoiesis in chronic myeloid leukaemia. Eur. J. Cancer 2000, 36, 1395–1401. [Google Scholar] [CrossRef]
- Shepherd, P.; Suffolk, R.; Halsey, J.; Allan, N. Analysis of molecular breakpoInt. and m-RNA transcripts in a prospective randomized trial of interferon in chronic myeloid leukaemia: No correlation with clinical features, cytogenetic response, duration of chronic phase, or survival. Br. J. Haematol. 1995, 89, 546–554. [Google Scholar] [CrossRef]
- Mills, K.I.; Benn, P.; Birnie, G.D. Does the breakpoint within the major breakpoint cluster region (M-bcr) influence the duration of the chronic phase in chronic myeloid leukemia? An analytical comparison of current literature. Blood 1991, 78, 1155–1161. [Google Scholar] [CrossRef]
- Lucas, C.M.; Harris, R.J.; Giannoudis, A.; Davies, A.; Knight, K.; Watmough, S.J.; Wang, L.; Clark, R.E. Chronic myeloid leukemia patients with the e13a2 BCR-ABL fusion transcript have inferior responses to imatinib compared to patients with the e14a2 transcript. Haematologica 2009, 94, 1362–1367. [Google Scholar] [CrossRef] [PubMed]
- Hanfstein, B.; Lauseker, M.; Hehlmann, R.; Saussele, S.; Erben, P.; Dietz, C.; Fabarius, A.; Proetel, U.; Schnittger, S.; Haferlach, C.; et al. Distinct characteristics of e13a2 versus e14a2 BCR-ABL1 driven chronic myeloid leukemia under first-line therapy with imatinib. Haematologica 2014, 99, 1441–1447. [Google Scholar] [CrossRef]
- D’Adda, M.; Farina, M.; Schieppati, F.; Borlenghi, E.; Bottelli, C.; Cerqui, E.; Ferrari, S.; Gramegna, D.; Pagani, C.; Passi, A.; et al. The e13a2 BCR-ABL transcript negatively affects sustained deep molecular response and the achievement of treatment-free remission in patients with chronic myeloid leukemia who receive tyrosine kinase inhibitors. Cancer 2019, 125, 1674–1682. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, K.; Kantarjian, H.; O’Brien, S.; Ravandi, F.; Konopleva, M.; Borthakur, G.; Garcia-Manero, G.; Wierda, W.; Daver, N.; Ferrajoli, A.; et al. Prediction for sustained deep molecular response of BCR-ABL1 levels in patients with chronic myeloid leukemia in chronic phase. Cancer 2018, 124, 1160–1168. [Google Scholar] [CrossRef] [PubMed]
- Castagnetti, F.; Gugliotta, G.; Breccia, M.; Iurlo, A.; Levato, L.; Albano, F.; Vigneri, P.; Abruzzese, E.; Rossi, G.; Rupoli, S.; et al. The BCR-ABL1 transcript type influences response and outcome in Philadelphia chromosome-positive chronic myeloid leukemia patients treated frontline with imatinib. Am. J. Hematol. 2017, 92, 797–805. [Google Scholar] [CrossRef]
- Baccarani, M.; Rosti, G.; Soverini, S. Chronic myeloid leukemia: The concepts of resistance and persistence and the relationship with the BCR-ABL1 transcript type. Leukemia 2019, 33, 2358–2364. [Google Scholar] [CrossRef]
- Clark, R.E.; Polydoros, F.; Apperley, J.F.; Milojkovic, D.; Pocock, C.; Smith, G.; Byrne, J.L.; de Lavallade, H.; O’Brien, S.G.; Coffey, T.; et al. De-escalation of tyrosine kinase inhibitor dose in patients with chronic myeloid leukaemia with stable major molecular response (DESTINY): An interim analysis of a non-randomised, phase 2 trial. Lancet Haematol. 2017, 4, e310–e316. [Google Scholar] [CrossRef]
- Shanmuganathan, N.; Branford, S.; Yong, A.S.; Hiwase, D.K.; Yeung, D.T.; Ross, D.M.; Hughes, T.P. The e13a2 BCR-ABL1 transcript is associated with higher rates of molecular recurrence after treatment-free remission attempts: Retrospective analysis of the Adelaide cohort. Blood 2018, 132, 1731. [Google Scholar] [CrossRef]
- Nicolini, F.E.; Dulucq, S.; Boureau, L.; Cony-Makhoul, P.; Charbonnier, A.; Escoffre-Barbe, M.; Rigal-Huguet, F.; Coiteux, V.; Varet, B.; Dubruille, V.; et al. Evaluation of Residual Disease and TKI Duration Are Critical Predictive Factors for Molecular Recurrence after Stopping Imatinib First-line in Chronic Phase CML Patients. Clin. Cancer Res. 2019, 25, 6606–6613. [Google Scholar] [CrossRef]
- Graham, S.M.; Jørgensen, H.G.; Allan, E.; Pearson, C.; Alcorn, M.J.; Richmond, L.; Holyoake, T.L. Primitive, quiescent, Philadelphia-positive stem cells from patients with chronic myeloid leukemia are insensitive to STI571 in vitro. Blood J. Am. Soc. Hematol. 2002, 99, 319–325. [Google Scholar] [CrossRef] [PubMed]
- Seggewiss, R.; Price, D.; Purbhoo, M. Immunomodulatory effects of imatinib and second-generation tyrosine kinase inhibitors on T cells and dendritic cells: An update. Cytotherapy 2008, 10, 633–641. [Google Scholar] [CrossRef]
- Rea, D.; Dulphy, N.; Henry, G.; Guilhot, J.; Guilhot, F.; Nicolini, F.E.; Legros, L.; Rousselot, P.; Mahon, F.-X.; Toubert, A.; et al. Low Natural Killer (NK) Cell Counts and Functionality Are Associated With Molecular Relapse After Imatinib Discontinuation in Patients (pts) With Chronic Phase (CP)-Chronic Myeloid Leukemia (CML) With Undetectable BCR-ABL Transcripts For At Least 2 Years: Preliminary Results From Immunostim, On Behalf Of STIM Investigators. Blood 2013, 122, 856. [Google Scholar] [CrossRef]
- Ilander, M.; Olsson-Strömberg, U.; Schlums, H.; Guilhot, J.; Brück, O.; Lähteenmäki, H.; Kasanen, T.; Koskenvesa, P.; Söderlund, S.; Höglund, M.; et al. Increased proportion of mature NK cells is associated with successful imatinib discontinuation in chronic myeloid leukemia. Leukemia 2017, 31, 1108–1116. [Google Scholar] [CrossRef]
- Hughes, A.; Clarson, J.; White, D.L.; Ross, D.M.; Hughes, T.P.; Yong, A.S. Enhanced Natural Killer and Cytotoxic T Lymphocyte Responses, with Decreased Monocytic Myeloid Derived Suppressor Cells May Promote Treatment Free Remission in Chronic Myeloid Leukaemia Patients Following Tyrosine Kinase Inhibitor Cessation. Blood 2016, 128, 1122. [Google Scholar] [CrossRef]
- Yoshida, C.; Iriyama, N.; Najima, Y.; Fujisawa, S.; Wakita, H.; Chiba, S.; Okamoto, S.; Kawakami, K.; Takezako, N.; Kumagai, T.; et al. Association of Peripheral Regulatory T Cells with Achievement of Deep Molecular Response in Newly Diagnosed Chronic Phase Chronic Myeloid Leukemia Treated with Dasatinib—The Final Results of D-First Study. Blood 2016, 128, 1916. [Google Scholar] [CrossRef]
- Hughes, A.; Yong, A.S.M. Immune Effector Recovery in Chronic Myeloid Leukemia and Treatment-Free Remission. Front. Immunol. 2017, 8, 469. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.; Savani, B.N. Impact of risk score calculations in choosing front-line tyrosine kinase inhibitors for patients with newly diagnosed chronic myeloid leukemia in the chronic phase. Eur. J. Haematol. 2014, 93, 179–186. [Google Scholar] [CrossRef] [PubMed]
- Sokal, J.E.; Cox, E.B.; Baccarani, M.; Tura, S.; Gomez, G.A.; Robertson, J.E.; Tso, C.Y.; Braun, T.J.; Clarkson, B.D.; Cervantes, F.; et al. Prognostic discrimination in “good-risk” chronic granulocytic leukemia. Blood 1984, 63, 789–799. [Google Scholar] [CrossRef] [PubMed]
- Chhikara, S.; Sazawal, S.; Singh, K.; Chaubey, R.; Pati, H.; Tyagi, S.; Mahapatra, M.; Saxena, R. Comparative analysis of the Sokal, Euro and European Treatment and Outcome Study score in prognostication of Indian chronic myeloid leukemia-chronic phase patients on imatinib. South Asian J. Cancer 2018, 7, 258–262. [Google Scholar] [CrossRef] [PubMed]
- Kuntegowdanahalli, L.C.; Kanakasetty, G.B.; Thanky, A.H.; Dasappa, L.; Jacob, L.A.; Mallekavu, S.B.; Lakkavalli, R.K.; Kadabur, L.N.; Haleshappa, R.A. Prognostic and predictive implications of Sokal, Euro and EUTOS scores in chronic myeloid leukaemia in the imatinib era-experience from a tertiary oncology centre in Southern India. Ecancermedicalscience 2016, 10, 679. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, V.S.; Baccarani, M.; Hasford, J.; Lindoerfer, D.; Burgstaller, S.; Sertic, D.; Costeas, P.; Mayer, J.; Indrak, K.; Everaus, H.; et al. The EUTOS population-based registry: Incidence and clinical characteristics of 2904 CML patients in 20 European Countries. Leukemia 2015, 29, 1336–1343. [Google Scholar] [CrossRef]
- Hoffmann, V.S.; Baccarani, M.; Lindoerfer, D.; Castagnetti, F.; Turkina, A.; Zaritsky, A.; Hellmann, A.; Prejzner, W.; Steegmann, J.L.; Mayer, J.; et al. The EUTOS prognostic score: Review and validation in 1288 patients with CML treated frontline with imatinib. Leukemia 2013, 27, 2016–2022. [Google Scholar] [CrossRef]
- Hasford, J.; Baccarani, M.; Hoffmann, V.; Guilhot, J.; Saussele, S.; Rosti, G.; Guilhot, F.; Porkka, K.; Ossenkoppele, G.; Lindoerfer, D.; et al. Predicting complete cytogenetic response and subsequent progression-free survival in 2060 patients with CML on imatinib treatment: The EUTOS score. Blood 2011, 118, 686–692. [Google Scholar] [CrossRef]
- Pfirrmann, M.; Baccarani, M.; Saussele, S.; Guilhot, J.; Cervantes, F.; Ossenkoppele, G.; Hoffmann, V.S.; Castagnetti, F.; Hasford, J.; Hehlmann, R.; et al. Prognosis of long-term survival considering disease-specific death in patients with chronic myeloid leukemia. Leukemia 2016, 30, 48–56. [Google Scholar] [CrossRef]
- Mori, S.; Vagge, E.; le Coutre, P.; Abruzzese, E.; Martino, B.; Pungolino, E.; Elena, C.; Pierri, I.; Assouline, S.; D’Emilio, A.; et al. Age and dPCR can predict relapse in CML patients who discontinued imatinib: The ISAV study. Am. J. Hematol. 2015, 90, 910–914. [Google Scholar] [CrossRef]
- Castagnetti, F.; Gugliotta, G.; Baccarani, M.; Breccia, M.; Specchia, G.; Levato, L.; Abruzzese, E.; Rossi, G.; Iurlo, A.; Martino, B.; et al. Differences among young adults, adults and elderly chronic myeloid leukemia patients. Ann. Oncol. 2015, 26, 185–192. [Google Scholar] [CrossRef]
- Kim, M.J.; Kim, M.H.; Kim, S.A.; Chang, J.S. Age-related Deterioration of Hematopoietic Stem Cells. Int. J. Stem Cells 2008, 1, 55–63. [Google Scholar] [CrossRef] [PubMed]
- Mejia-Ramirez, E.; Florian, M.C. Understanding intrinsic hematopoietic stem cell aging. Haematologica 2020, 105, 22–37. [Google Scholar] [CrossRef] [PubMed]
- Hughes, T.P.; Ross, D.M. Moving treatment-free remission into mainstream clinical practice in CML. Blood 2016, 128, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Essers, M.A.G.; Offner, S.; Blanco-Bose, W.E.; Waibler, Z.; Kalinke, U.; Duchosal, M.A.; Trumpp, A. IFNα activates dormant haematopoietic stem cells in vivo. Nature 2009, 458, 904–908. [Google Scholar] [CrossRef] [PubMed]
- Talpaz, M.; Hehlmann, R.; Quintás-Cardama, A.; Mercer, J.; Cortes, J. Re-emergence of interferon-α in the treatment of chronic myeloid leukemia. Leukemia 2013, 27, 803–812. [Google Scholar] [CrossRef] [PubMed]
- Burchert, A.; Müller, M.C.; Kostrewa, P.; Erben, P.; Bostel, T.; Liebler, S.; Hehlmann, R.; Neubauer, A.; Hochhaus, A. Sustained molecular response with interferon alfa maintenance after induction therapy with imatinib plus interferon alfa in patients with chronic myeloid leukemia. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2010, 28, 1429–1435. [Google Scholar] [CrossRef] [PubMed]
- Jabbour, E.; Kantarjian, H.; O’Brien, S.; Rios, M.B.; Abruzzo, L.; Verstovsek, S.; Garcia-Manero, G.; Cortes, J. Sudden blastic transformation in patients with chronic myeloid leukemia treated with imatinib mesylate. Blood 2006, 107, 480–482. [Google Scholar] [CrossRef] [PubMed]
- Yong, A.S.; Goldman, J.M. Relapse of chronic myeloid leukaemia 14 years after allogeneic bone marrow transplantation. Bone Marrow Transplant. 1999, 23, 827–828. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Baccarani, M.; Deininger, M.W.; Rosti, G.; Hochhaus, A.; Soverini, S.; Apperley, J.F.; Cervantes, F.; Clark, R.E.; Cortes, J.E.; Guilhot, F.; et al. European LeukemiaNet recommendations for the management of chronic myeloid leukemia: 2013. Blood 2013, 122, 872–884. [Google Scholar] [CrossRef]
- Palani, R.; Milojkovic, D.; Apperley, J.F. Managing pregnancy in chronic myeloid leukaemia. Ann. Hematol. 2015, 94 (Suppl. 2), S167–S176. [Google Scholar] [CrossRef]
- Legros, L.; Nicolini, F.E.; Etienne, G.; Rousselot, P.; Rea, D.; Giraudier, S.; Guerci-Bresler, A.; Huguet, F.; Gardembas, M.; Escoffre, M.; et al. Second tyrosine kinase inhibitor discontinuation attempt in patients with chronic myeloid leukemia. Cancer 2017, 123, 4403–4410. [Google Scholar] [CrossRef]
- Kim, D.D.H.; Busque, L.; Forrest, D.L.; Savoie, L.; Bence-Bruckler, I.; Couban, S.; Delage, R.; Xenocostas, A.; Liew, E.; Laneuville, P.; et al. Second Attempt of TKI Discontinuation with Dasatinib for Treatment-Free Remission after Failing First Attempt with Imatinib: Treatment-Free Remission Accomplished By Dasatinib (TRAD) Trial. Blood 2018, 132, 787. [Google Scholar] [CrossRef]
- Faber, E.; Divoká, M.; Skoumalová, I.; Novák, M.; Marešová, I.; Mičová, K.; Friedecký, D.; Adam, T.; Jarošová, M.; Indrák, K. A lower dosage of imatinib is sufficient to maintain undetectable disease in patients with chronic myeloid leukemia with long-term low-grade toxicity of the treatment. Leuk. Lymphoma 2016, 57, 370–375. [Google Scholar] [CrossRef] [PubMed]
- Fassoni, A.C.; Baldow, C.; Roeder, I.; Glauche, I. Reduced tyrosine kinase inhibitor dose is predicted to be as effective as standard dose in chronic myeloid leukemia: A simulation study based on phase III trial data. Haematologica 2018, 103, 1825–1834. [Google Scholar] [CrossRef]
- Clark, R.; Polydoros, F.; Apperley, J.; Milojkovic, D.; Rothwell, K.; Pocock, C.; Byrne, J.; de Lavallade, H.; Osborne, W.; Robinson, L.; et al. Initial reduction of therapy prior to complete treatment discontinuation in chronic myeloid leukaemia: Final results of the British DESTINY Study. Lancet Haematol. 2019, 6, e375–e383. [Google Scholar] [CrossRef]
- Hochhaus, A.; Saussele, S.; Rosti, G.; Mahon, F.-X.; Janssen, J.J.; Hjorth-Hansen, H.; Richter, J.; Buske, C.; Committee, E.G. Chronic myeloid leukaemia: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2017, 28, iv41–iv51. [Google Scholar] [CrossRef]
- Radich, J.P.; Deininger, M.; Abboud, C.N.; Altman, J.K.; Berman, E.; Bhatia, R.; Bhatnagar, B.; Curtin, P.; DeAngelo, D.J.; Gotlib, J.; et al. Chronic Myeloid Leukemia, Version 1.2019, NCCN Clinical Practice Guidelines in Oncology. J. Natl. Compr. Canc. Netw. 2018, 16, 1108–1135. [Google Scholar] [CrossRef] [PubMed]
- Clark, R.E. Another set of guidelines for treating chronic myeloid leukaemia. Br. J. Haematol. 2020, 191, 147–149. [Google Scholar] [CrossRef]
- Rea, D.; Ame, S.; Berger, M.; Cayuela, J.-M.; Charbonnier, A.; Coiteux, V.; Cony-Makhoul, P.; Dubruille, V.; Dulucq, S.; Etienne, G.; et al. Discontinuation of tyrosine kinase inhibitors in chronic myeloid leukemia: Recommendations for clinical practice from the French Chronic Myeloid Leukemia Study Group. Cancer 2018, 124, 2956–2963. [Google Scholar] [CrossRef] [PubMed]
- Berger, M.G.; Pereira, B.; Oris, C.; Saugues, S.; Cony-Makhoul, P.; Gardembas, M.; Legros, L.; Escoffre-Barbe, M.; Nicolini, F.E.; Rousselot, P.; et al. Osteoarticular Pain after Discontinuation of Tyrosine Kinase Inhibitors (TKI): A French Cohort. Blood 2015, 126, 137. [Google Scholar] [CrossRef]
- Richter, J.; Söderlund, S.; Lübking, A.; Dreimane, A.; Lotfi, K.; Markevärn, B.; Själander, A.; Saussele, S.; Olsson-Strömberg, U.; Stenke, L. Musculoskeletal Pain in Patients With Chronic Myeloid Leukemia After Discontinuation of Imatinib: A Tyrosine Kinase Inhibitor Withdrawal Syndrome? J. Clin. Oncol. 2014, 32, 2821–2823. [Google Scholar] [CrossRef]
- Clark, R.E. Tyrosine Kinase Inhibitor Therapy Discontinuation for Patients with Chronic Myeloid Leukaemia in Clinical Practice. Curr. Hematol. Malig. Rep. 2019, 14, 507–514. [Google Scholar] [CrossRef]
- Ross, D.M.; Hughes, T.P. Treatment-free remission in patients with chronic myeloid leukaemia. Nat. Rev. Clin. Oncol. 2020, 17, 493–503. [Google Scholar] [CrossRef]
- Bernardi, S.; Malagola, M.; Zanaglio, C.; Polverelli, N.; Dereli Eke, E.; D’Adda, M.; Farina, M.; Bucelli, C.; Scaffidi, L.; Toffoletti, E.; et al. Digital PCR improves the quantitation of DMR and the selection of CML candidates to TKIs discontinuation. Cancer Med. 2019, 8, 2041–2055. [Google Scholar] [CrossRef] [PubMed]
- Claudiani, S.; Metelli, S.; Kamvar, R.; Szydlo, R.; Khan, A.; Byrne, J.; Gallipoli, P.; Bulley, S.J.; Horne, G.A.; Rothwell, K.; et al. Introducing a Predictive Score for Successful Treatment Free Remission in Chronic Myeloid Leukemia (CML). Blood 2019, 134, 26. [Google Scholar] [CrossRef]
- Rea, D.; Nicolini, F.E.; Tulliez, M.; Rousselot, P.; Gardembas, M.; Etienne, G.; Guilhot, F.; Guilhot, J.; Guerci, A.; Escoffre-Barbe, M.; et al. Prognostication of Molecular Relapses after Dasatinib or Nilotinib Discontinuation in Chronic Myeloid Leukemia (CML): A FI-LMC STOP 2G-TKI Study Update. Blood 2019, 134, 30. [Google Scholar] [CrossRef]
- Legros, L.; Nicolini, F.E.; Etienne, G.; Rousselot, P.; Rea, D.; Giraudier, S.; Guerci, A.; Huguet, F.; Gardembas, M.; Ianotto, J.-C. The Tki-Free Duration after A First Discontinuation Attempt That Failed in Cp Cml Patients Is a Predictive Factor of Tki-Free Remission after a Second Attempt; American Society of Hematology: Washington, DC, USA, 2019. [Google Scholar]
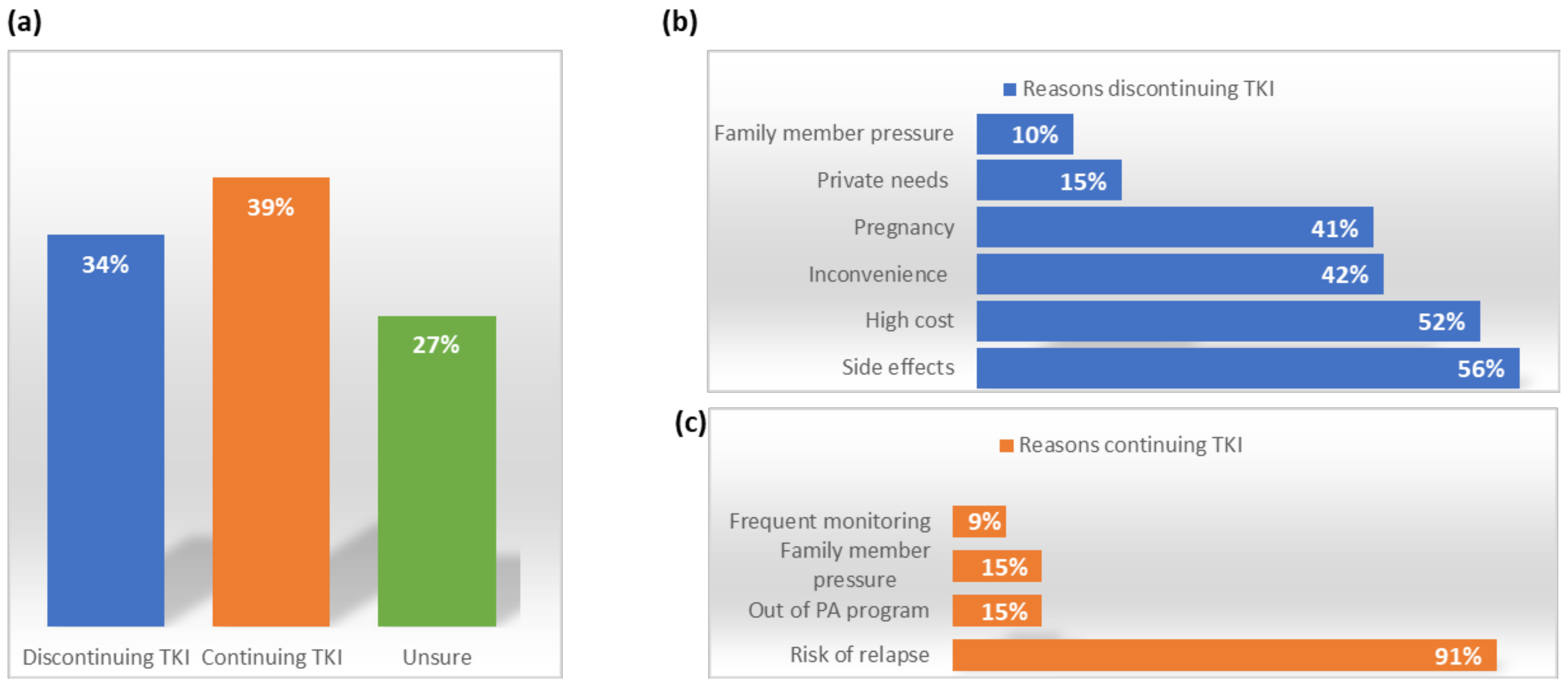
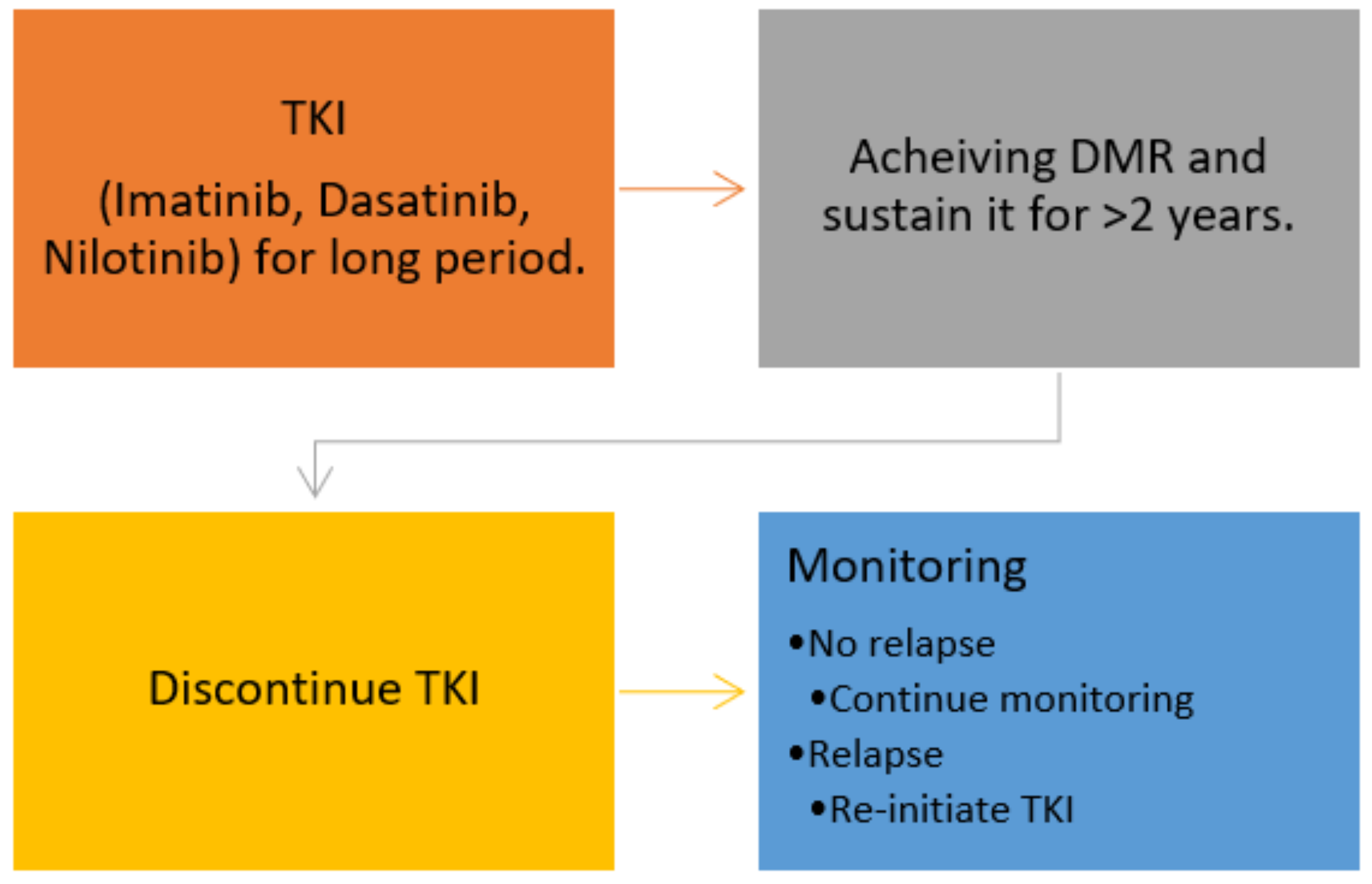
| Author | N | Minimum TKI Duration (y) | Type of TKI | Minimum DMR Duration (y) | Median Follow Up (m) | The Median Time to MR (m) | TFR Rate | Treatment Reinitiation |
|---|---|---|---|---|---|---|---|---|
| STIM [35,36] | 100 | 3 | IM | UMRD ≥ 2 | 77 | 2.5 | 43% at 24 months and 38% at 60 months. | Loss of UMRD |
| TWISTER [37] | 40 | 3 | IM | UMRD ≥ 2 | 43 | 3 | 47.1% at 24 months. | Loss of UMRD |
| ASTIM [38] | 80 | 3 | IM | MR4 ≥ 2 | 31 | 4 | 64% at 24 months and 61% at 36 months. | Loss of MMR, UMRD |
| HOVON [31] | 15 | 2 | IM | MR4.5 > 2 | 36 | 3 | 33% at 24 months. | Loss of MR4.5 |
| ISAV [39] | 108 | 2 | IM | UMRD ≥ 1 | 21.6 | NR | 48% at 36 months. | Loss of MMR |
| KID [40] | 90 | 2 | IM | MR4.5 > 2 | 26.6 | 3.3 | 62.2% at 12 months and 58.5% at 24 months. | Loss of MMR |
| EURO-SKI [34] | 755 | 3 | IM & NL & DA | MR4 ≥ 1 | 27 | 6 | 61% at 6 months and 50% at 24 months. | Loss of MRR |
| DADI (2015) [41] | 63 | 1 | DA | DMR ≥ 2 | 20 | 3 | 49% at six months and 48% at 12 months. | Loss of MR4 |
| DADI (2020) [42] | 58 | 3 | DA | DMR ≥ 2 | 23.3 | 2 | 55% at 6 months. | Loss of MR4 |
| ENESTfreedom [43] | 190 | 3 | NL | MR4.5 ≥ 2 | 12 | NR | 51.6 at 48 weeks. | Loss of MMR |
| Ho-Young Yhim [44] | 14 | 2 | IM | DMR ≥ 2 | 23 | NR | 28.6% at 12 months. | Loss of MMR |
| GIMEMA [45] | 293 | 7 | IM, NL, DA, BO | DMR ≥ 3 | 34 | 3–4 | 69% at 12 months and 62% at 34 months. | Loss of MMR |
| STIM2 [46] | 124 | 3 | IM | DMR ≥ 2 | 12 | 6 | 61.2% at 12 months. | Loss of MMR |
| Kieo STIM [47] | 40 | 2 | IM | DMR ≥ 2 | 15.5 | NR | 55.4% at 12 months. | Loss of MMR |
| Pagnano KB [48] | 48 | 3 | IM | DMR ≥ 2 | NR | NR | 61% at 20 months. | Loss of MMR |
| TRAD [49] | 67 | 3 | IM | MR4.5 ≥ 2 | NR | NR | 64.7% at 6 months. | Loss of MMR |
| STIM231 [50] | 68 | 3 | IM | MR4 ≥ 2 | NR | NR | 67.6% at 12 months and 65% at 36 months. | Loss of MMR |
| STOP2G [51] | 60 | 3 | NL, DA | MR4.5 ≥ 2 | 47 | 4 | 63.3% at 12 months and 53.7% at 48 months. | Loss of MMR |
| D-STOP [52] | 54 | 2 | DA | DMR ≥ 2 | 16 | NR | 62.9% at 1 year. | Loss of MMR |
| DASFREE [53] | 84 | 2 | DA | MR4.5 ≥ 1 | NR | 5 | 48% at 12 months and 46% at 24 months. | Loss of MMR |
| STATS [54] | 78 | 2 | NL | MR4.5 ≥ 2 | 35.4 | 6 | 67.9% at 12 months and 62.8% at 24 months. | Loss of MR4.5 |
| DOMEST [55] | 99 | 2 | IM | MR4 ≥ 2 | 12 | NR | 70% at 6 months, 68% at 12 months, and 64% at 24 months. | Loss of MR4 |
| Criteria | FCMLSG [125] | NCCN | ESMO [122] |
|---|---|---|---|
| Age | >18 | >18 | >18 |
| Phase | CP only | CP only | CP only |
| Sokal score | not defined | Not defined | Not a high score |
| BCR–ABL1 transcript | e13a2, e14a2, or e13a2 + e14a2 | Quantifiable typical transcript | e13a2, e14a2 |
| TKI duration (years) | >5 | >3 | >5 |
| DMR type | MR4.5 | MR4 | MR4.5 |
| DMR duration (years) | >2 | >2 | >2 |
| Re-treatment | Loss of MMR | Loss of MMR or < MR4 | Not defined |
| Prior history | No HSCT, no progression, no resistance, suboptimal response | No prior history of progression or treatment resistance | Optimal response |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Saifullah, H.H.; Lucas, C.M. Treatment-Free Remission in Chronic Myeloid Leukemia: Can We Identify Prognostic Factors? Cancers 2021, 13, 4175. https://doi.org/10.3390/cancers13164175
Saifullah HH, Lucas CM. Treatment-Free Remission in Chronic Myeloid Leukemia: Can We Identify Prognostic Factors? Cancers. 2021; 13(16):4175. https://doi.org/10.3390/cancers13164175
Chicago/Turabian StyleSaifullah, Hilbeen Hisham, and Claire Marie Lucas. 2021. "Treatment-Free Remission in Chronic Myeloid Leukemia: Can We Identify Prognostic Factors?" Cancers 13, no. 16: 4175. https://doi.org/10.3390/cancers13164175
APA StyleSaifullah, H. H., & Lucas, C. M. (2021). Treatment-Free Remission in Chronic Myeloid Leukemia: Can We Identify Prognostic Factors? Cancers, 13(16), 4175. https://doi.org/10.3390/cancers13164175





