Neoadjuvant Modified Short-Course Radiotherapy Followed by Delayed Surgery for Locally Advanced Rectal Cancer
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Treatment
2.3. Analysis
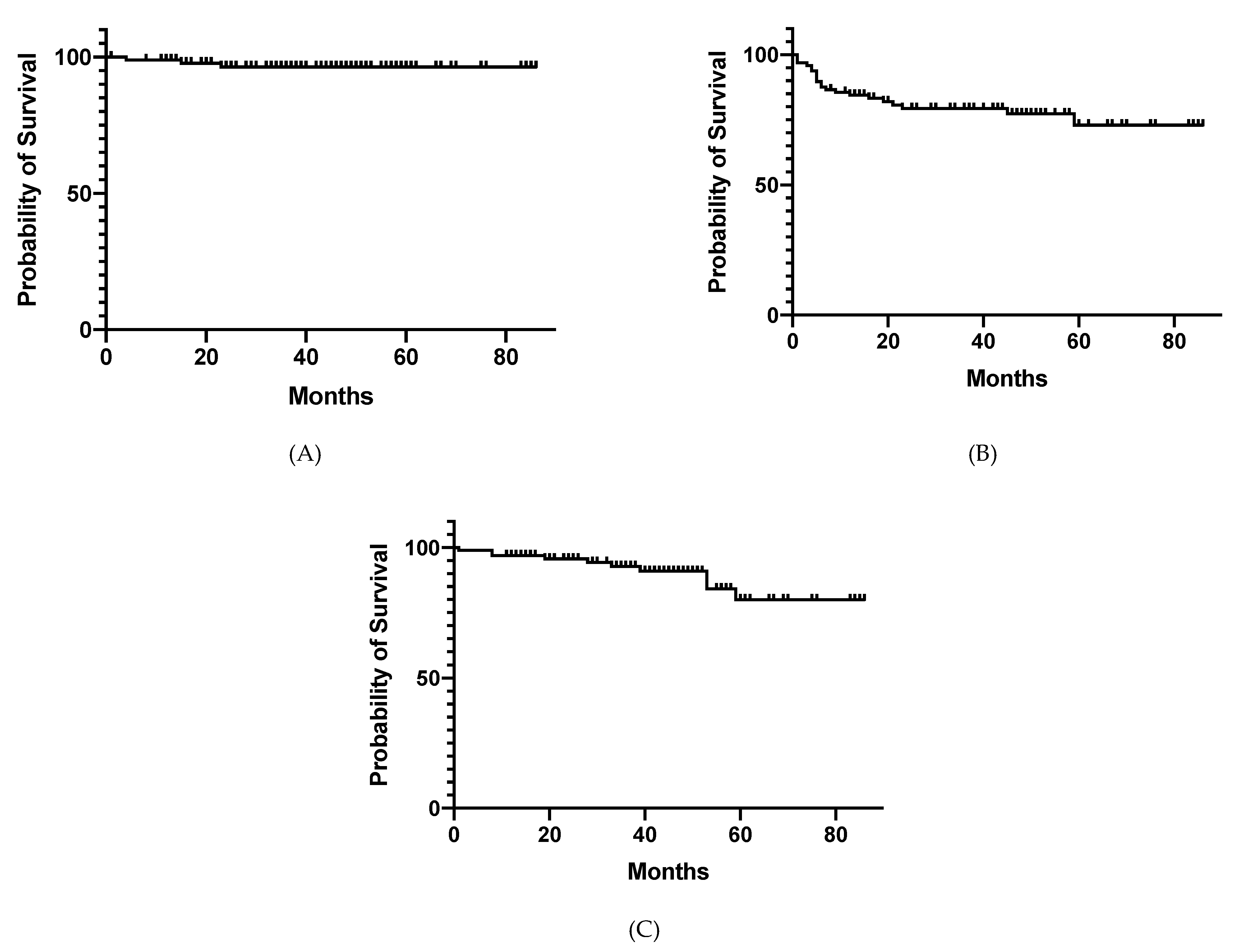
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Oronsky, B.; Reid, T.; Larson, C.; Knox, S.J. Locally Advanced Rectal Cancer: The Past, Present, and Future. Semin. Oncol. 2020, 47, 85–92. [Google Scholar] [CrossRef]
- Argilés, G.; Tabernero, J.; Labianca, R.; Hochhauser, D.; Salazar, R.; Iveson, T.; Laurent-Puig, P.; Quirke, P.; Yoshino, T.; Taieb, J.; et al. Localised colon cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann. Oncol. 2020, 31, 1291–1305. [Google Scholar] [CrossRef] [PubMed]
- Wo, J.Y.; Anker, C.J.; Ashman, J.B.; Bhadkamkar, N.A.; Bradfield, L.; Chang, D.T.; Dorth, J.; Garcia-Aguilar, J.; Goff, D.; Jacqmin, D.; et al. Radiation Therapy for Rectal Cancer: Executive Summary of an ASTRO Clinical Practice Guideline. Pract. Radiat. Oncol. 2021, 11, 13–25. [Google Scholar] [CrossRef] [PubMed]
- NCCN Clinical Practice Guidelines in Oncology (NCCN Guidelines®) Rectal Cancer Version 1. 2021. Available online: https://www.nccn.org/professionals/physician_gls/pdf/rectal.pdf (accessed on 6 July 2021).
- Ngan, S.Y.; Burmeister, B.; Fisher, R.J.; Solomon, M.; Goldstein, D.; Joseph, D.; Ackland, S.P.; Schache, D.; Mcclure, B.; Mclachlan, S.; et al. Randomized Trial of Short-Course Radiotherapy Versus Long-Course Chemoradiation Comparing Rates of Local Recurrence in Patients With T3 Rectal Cancer: Trans-Tasman Radiation Oncology Group Trial 01.04. J. Clin. Oncol. 2012, 30, 3827–3833. [Google Scholar] [CrossRef] [PubMed]
- Bujko, K.; Nowacki, M.P.; Nasierowska-Guttmejer, A.; Michalski, W.; Kryj, M. Long-term results of a randomized trial comparing preoperative short-course radiotherapy with preoperative conventionally fractionated chemoradiation for rectal cancer. Br. J. Surg. 2006, 93, 1215–1223. [Google Scholar] [CrossRef] [PubMed]
- Socha, J.; Kairevice, L.; Kepka, L.; Michalski, W.; Spałek, M.; Paciorek, K.; Bujko, K. Should Short-Course Neoadjuvant Radiation Therapy Be Applied for Low-Lying Rectal Cancer? A Systematic Review and Meta-Analysis of the Randomized Trials. Int. J. Radiat. Oncol. Biol. Phys. 2020, 108, 1257–1264. [Google Scholar] [CrossRef]
- Chen, C.; Sun, P.; Rong, J.; Weng, H.-W.; Dai, Q.; Ye, S. Short Course Radiation in the Treatment of Localized Rectal Cancer: A Systematic Review and Meta-Analysis. Sci. Rep. 2015, 5, 10953. [Google Scholar] [CrossRef] [Green Version]
- Ciseł, B.; Pietrzak, L.; Michalski, W.; Wyrwicz, L.; Rutkowski, A.; Kosakowska, E.; Cencelewicz, A.; Spałek, M.; Polkowski, W.; Jankiewicz, M.; et al. Long-course preoperative chemoradiation versus 5 × 5 Gy and consolidation chemotherapy for clinical T4 and fixed clinical T3 rectal cancer: Long-term results of the randomized Polish II study. Ann. Oncol. 2019, 30, 1298–1303. [Google Scholar] [CrossRef] [PubMed]
- Bahadoer, R.R.; Dijkstra, E.A.; van Etten, B.; Marijnen, C.A.M.; Putter, H.; Kranenbarg, E.M.-K.; Roodvoets, A.G.H.; Nagtegaal, I.D.; Beets-Tan, R.G.H.; Blomqvist, L.K.; et al. Short-course radiotherapy followed by chemotherapy before total mesorectal excision (TME) versus preoperative chemoradiotherapy, TME, and optional adjuvant chemotherapy in locally advanced rectal cancer (RAPIDO): A randomised, open-label, phase 3 trial. Lancet Oncol. 2021, 22, 29–42. [Google Scholar] [CrossRef]
- Pettersson, D.; Lörinc, E.; Holm, T.; Iversen, H.; Cedermark, B.; Glimelius, B.; Martling, A. Tumour regression in the randomized Stockholm III Trial of radiotherapy regimens for rectal cancer. Br. J. Surg. 2015, 102, 972–978. [Google Scholar] [CrossRef] [Green Version]
- Erlandsson, J.; Holm, T.; Pettersson, D.; Berglund, Å.; Cedermark, B.; Radu, C.; Johansson, H.; Machado, M.; Hjern, F.; Hallböök, O.; et al. Optimal fractionation of preoperative radiotherapy and timing to surgery for rectal cancer (Stockholm III): A multicentre, randomised, non-blinded, phase 3, non-inferiority trial. Lancet Oncol. 2017, 18, 336–346. [Google Scholar] [CrossRef]
- Peeters, K.C.M.J.; van de Velde, C.J.H.; Leer, J.W.H.; Martijn, H.; Junggeburt, J.M.C.; Kranenbarg, E.K.; Steup, W.H.; Wiggers, T.; Rutten, H.J.; Marijnen, C.A.M. Late side effects of short-course preoperative radiotherapy combined with total mesorectal excision for rectal cancer: Increased bowel dysfunction in irradiated patients--a Dutch colorectal cancer group study. J. Clin. Oncol. 2005, 23, 6199–6206. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Withers, H.R. Radiation biology and treatment options in radiation oncology. Cancer Res. 1999, 59, 1676s–1684s. [Google Scholar] [PubMed]
- Ceelen, W.; Fierens, K.; Nieuwenhove, Y.V.; Pattyn, P. Preoperative chemoradiation versus radiation alone for stage II and III resectable rectal cancer: A systematic review and meta-analysis. Int. J. Cancer 2009, 124, 2966–2972. [Google Scholar] [CrossRef]
- Beppu, N.; Matsubara, N.; Kakuno, A.; Doi, H.; Kamikonya, N.; Yamanaka, N.; Yanagi, H.; Tomita, N. Feasibility of modified short-course radiotherapy combined with a chemoradiosensitizer for T3 rectal cancer. Dis. Colon Rectum 2015, 58, 479–487. [Google Scholar] [CrossRef]
- Beppu, N.; Kimura, F.; Aihara, T.; Doi, H.; Tomita, N.; Yanagi, H.; Yamanaka, N. Patterns of Local Recurrence and Oncologic Outcomes in T3 Low Rectal Cancer (≤5 cm from the Anal Verge) Treated With Short-Course Radiotherapy With Delayed Surgery. Ann. Surg. Oncol. 2017, 24, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Beppu, N.; Kimura, F.; Okamoto, R.; Nakamoto, Y.; Tomita, N.; Yanagi, H.; Yamanaka, N. Early results of a phase-II study of modified short-course radiotherapy combined with capecitabine and delayed surgery for T3M0 lower rectal cancer. Acta Oncol. 2018, 57, 860–862. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Divitiis, C.D.; Nasti, G.; Montano, M.; Fisichella, R.; Iaffaioli, R.V.; Berretta, M. Prognostic and predictive response factors in colorectal cancer patients: Between hope and reality. World J. Gastroenterol. 2014, 20, 15049–15059. [Google Scholar] [CrossRef]
- Diakos, C.I.; Charles, K.A.; McMillan, D.C.; Clarke, S.J. Cancer-Related Inflammation and Treatment Effectiveness. Lancet Oncol. 2014, 15, e493–e503. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, X.; Xu, M.; Chen, K.; Li, S.; Guan, G. Prognostic value of pretreatment systemic inflammatory markers in patients with locally advanced rectal cancer following neoadjuvant chemoradiotherapy. Sci. Rep. 2020, 10, 8017. [Google Scholar] [CrossRef]
- Shibutani, M.; Maeda, K.; Nagahara, H.; Iseki, Y.; Ikeya, T.; Hirakawa, K. Prognostic Significance of the Preoperative Ratio of C-Reactive Protein to Albumin in Patients with Colorectal Cancer. Anticancer Res. 2016, 3, 995–1001. [Google Scholar]
- Classification of Malignant Tumours, 8th ed.; Brierley, J.D.; Gospodarowicz, M.K.; Christian Wittekind, C. (Eds.) International Union Against Cancer: Geneva, Switzerland; Wiley-Blackwell: Hoboken, NJ, USA, 2016. [Google Scholar]
- Common Terminology Criteria for Adverse Events (CTCAE) v5.0. Available online: https://ctep.cancer.gov/protocoldevelopment/electronic_applications/docs/ctcae_v5_quick_reference_5x7.pdf (accessed on 17 April 2021).
- Japanese Society for Cancer of the Colon and Rectum. Japanese Classification of Colorectal, Appendiceal and Anal Carcinoma: The 3d English Edition [Secondary Publication]. J. Anus Rectum Colon 2019, 3, 175–195. [Google Scholar] [CrossRef] [Green Version]
- Viani, G.A.; Stefano, E.J.; Soares, F.V.; Afonso, S.L. Evaluation of Biologic Effective Dose and Schedule of Fractionation for Preoperative Radiotherapy for Rectal Cancer: Meta-Analyses and Meta-Regression. Int. J. Radiat. Oncol. Biol. Phys. 2011, 80, 985–991. [Google Scholar] [CrossRef] [PubMed]
- Jin, F.; Luo, H.; Zhou, J.; Wu, Y.; Sun, H.; Liu, H.; Zheng, X.; Wang, Y. Dose-time fractionation schedules of preoperative radiotherapy and timing to surgery for rectal cancer. Ther. Adv. Med. Oncol. 2020, 12, 1758835920907537. [Google Scholar] [CrossRef] [Green Version]
- Feeney, G.; Sehgal, R.; Sheehan, M.; Hogan, A.; Regan, M.; Joyce, M.; Kerin, M. Neoadjuvant radiotherapy for rectal cancer management. World J. Gastroenterol. 2019, 25, 4850–4869. [Google Scholar] [CrossRef]
- Ansari, N.; Solomon, M.J.; Fisher, R.J.; Mackay, J.; Burmeister, B.; Ackland, S.; Heriot, A.; Joseph, D.; Mclachlan, S.; Mcclure, B.; et al. Acute Adverse Events and Postoperative Complications in a Randomized Trial of Preoperative Short-course Radiotherapy Versus Long-course Chemoradiotherapy for T3 Adenocarcinoma of the Rectum: Trans-Tasman Radiation Oncology Group Trial (TROG 01.04). Ann. Surg. 2017, 265, 882–888. [Google Scholar] [CrossRef]
- Bujko, K.; Nowacki, M.P.; Nasierowska-Guttmejer, A.; Michalski, W.; Bebenek, M.; Pudełko, M.; Kryj, M.; Olędzki, J.; Szmeja, J.; Słuszniak, J.; et al. Sphincter preservation following preoperative radiotherapy for rectal cancer: Report of a randomised trial comparing short-term radiotherapy vs. conventionally fractionated radiochemotherapy. Radiother. Oncol. 2004, 72, 15–24. [Google Scholar] [CrossRef] [PubMed]
- Bromham, N.; Kallioinen, M.; Hoskin, P.; Davies, R.J.; Guideline Committee. Colorectal cancer: Summary of NICE guidance. BMJ 2020, 368, m461. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Allegra, C.J.; Yothers, G.; O’Connell, M.J.; Beart, R.W.; Wozniak, T.F.; Pitot, H.C.; Shields, A.F.; Landry, J.C.; Ryan, D.P.; Arora, A.; et al. Neoadjuvant 5-FU or Capecitabine Plus Radiation With or Without Oxaliplatin in Rectal Cancer Patients: A Phase III Randomized Clinical Trial. J. Natl. Cancer Inst. 2015, 107, djv248. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kwakman, J.J.M.; Simkens, L.H.J.; van Rooijen, J.M.; van de Wouw, A.J.; ten Tije, A.J.; Creemers, G.J.M.; Hendriks, M.P.; Los, M.; van Alphen, R.J.; Polée, M.B.; et al. Randomized phase III trial of S-1 versus capecitabine in the first-line treatment of metastatic colorectal cancer: SALTO study by the Dutch Colorectal Cancer Group. Ann. Oncol. 2017, 28, 1288–1293. [Google Scholar] [CrossRef]
- Sadahiro, S.; Suzuki, T.; Tanaka, A.; Okada, K.; Kamijo, A.; Murayama, C.; Akiba, T.; Nakayama, Y. Phase I/II study of preoperative concurrent chemoradiotherapy with S-1 for locally advanced, resectable rectal adenocarcinoma. Oncology 2012, 81, 306–311. [Google Scholar] [CrossRef]
- Sacdalan, D.B.; Lucero, J.A.; Sacdalan, D.L. Prognostic utility of baseline neutrophil-to-lymphocyte ratio in patients receiving immune checkpoint inhibitors: A review and meta-analysis. Onco Targ. Ther. 2018, 11, 955–965. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gavriilidis, P.; Wheeler, J.; Spinelli, A.; de‘Angelis, N.; Simopoulos, C.; Saverio, S.D. Robotic vs laparoscopic total mesorectal excision for rectal cancers: Has a paradigm change occurred? A systematic review by updated meta-analysis. Colorectal Dis. 2020, 22, 1506–1517. [Google Scholar] [CrossRef] [PubMed]
- Oikonomou, C.; Gourgiotis, S.; Cirocchi, R.; Piagkou, M.; Protogerou, V.; Troupis, T.; Biondi, A.; Sileri, P.; Filippou, D.; Pelvic-Neuroanatomy Collaborating Group; et al. Re-exploring the pelvic neuroanatomy from a new perspective and a potential guidance for TaTME: A “bottom-up” approach. Updates Surg. 2021, 73, 503–512. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | n = 97 | (%) | |
| Age, years (range) | 66 (34–87) | ||
| Sex | Male | 52 | (53.6) |
| Female | 45 | (46.4) | |
| ECOG-PS | 0 | 80 | (82.5) |
| 1 | 15 | (15.5) | |
| 2 | 1 | (1.0) | |
| 3 | 1 | (1.0) | |
| Localization | Ra | 23 | (23.7) |
| Rb | 74 | (76.3) | |
| Pathological diagnosis | |||
| Tubular adenocarcinoma | 92 | (94.8) | |
| Papillary adenocarcinoma | 3 | (3.1) | |
| Poorly differentiated adenocarcinoma | 2 | (2.1) | |
| Clinical stage at the diagnosis | |||
| T | 1 | 3 | (3.1) |
| 2 | 31 | (32.0) | |
| 3 | 50 | (51.5) | |
| 4 | 13 | (13.4) | |
| N | 0 | 8 | (8.2) |
| 1 | 53 | (54.6) | |
| 2 | 36 | (37.1) | |
| Stage | II | 9 | (9.3) |
| III | 88 | (90.7) | |
| Previous chemotherapy before radiotherapy † | |||
| Any | 34 | (35.1) | |
| None | 63 | (64.9) | |
| Combined chemotherapy | |||
| Capecitabine | 67 | (69.1) | |
| S-1 | 22 | (22.7) | |
| UFT | 5 | (5.2) | |
| Polysaccharide-K | 1 | (1.0) | |
| none | 2 | (2.1) | |
| Adjuvant chemotherapy after surgery | |||
| Any | 37 | (38.1) | |
| None | 60 | (61.9) | |
| Adverse events per patient | n = 97 | Incidence (%) | |
| Any | 16 | (16.5) | |
| Worst grade per patient | n = 97 | Incidence (%) | |
| Grade | 1 | 13 | (13.4) |
| 2 | 3 | (3.1) | |
| None | 81 | (83.5) | |
| Adverse events | Grade | n = 97 | Incidence (%) |
| Nausea | 1 | 4 | (4.1) |
| 2 | 1 | (1.0) | |
| Diarrhea | 1 | 5 | (5.2) |
| Enterocolitis | 1 | 2 | (2.1) |
| 2 | 1 | (1.0) | |
| Fatigue | 1 | 3 | (3.1) |
| Dermatitis | 2 | 1 | (1.0) |
| Anorexia | 1 | 1 | (1.0) |
| Urinary frequency | 1 | 1 | (1.0) |
| Cystitis noninfective | 1 | 1 | (1.0) |
| Pathological tumor response | n = 92 * | (%) | |
| Grade | 0 | 1 | (1.1) |
| 1a | 21 | (22.8) | |
| 1b | 22 | (23.9) | |
| 2 | 35 | (38.0) | |
| 3 | 13 | (14.1) | |
| Pathologic stage | |||
| T | 0 | 13 | (13.4) |
| Tis | 3 | (3.1) | |
| 1 | 14 | (14.4) | |
| 2 | 27 | (27.8) | |
| 3 | 33 | (34.0) | |
| 4 | 7 | (7.2) | |
| N | 0 | 62 | (63.9) |
| 1 | 25 | (25.8) | |
| 2 | 10 | (10.3) | |
| Stage | 0 | 15 | (15.5) |
| I | 32 | (33.0) | |
| II | 15 | (15.5) | |
| III | 33 | (34.0) | |
| IV | 2 | (2.1) | |
| Adverse events per patient | n = 97 | Incidence (%) | |
| Any | 50 | (51.5) | |
| Worst grade per patient | n = 97 | Incidence (%) | |
| Grade | 1 | 5 | (5.2) |
| 2 | 34 | (35.1) | |
| 3 | 10 | (10.3) | |
| 4 | 0 | (0.0) | |
| 5 | 1 | (1.0) | |
| None | 47 | (48.5) | |
| Adverse events | Grade | n = 97 | Incidence (%) |
| Ileus | 1 | 1 | (1.0) |
| 2 | 11 | (11.3) | |
| 3 | 4 | (4.1) | |
| Dysuria | 1 | 1 | (1.0) |
| 2 | 5 | (5.2) | |
| Fever | 1 | 6 | (6.2) |
| Pelvic infection | 2 | 4 | (4.1) |
| 3 | 1 | (1.0) | |
| Rectal anastomotic leak | 2 | 2 | (2.1) |
| 3 | 2 | (2.1) | |
| Colonic fistula | 2 | 1 | (1.0) |
| 3 | 2 | (2.1) | |
| Colonic obstruction | 2 | 3 | (3.1) |
| Intestinal stoma obstruction | 2 | 3 | (3.1) |
| Dehydration | 2 | 2 | (2.1) |
| Nausea | 2 | 2 | (2.1) |
| Wound infection | 2 | 2 | (2.1) |
| Abdominal infection | 2 | 2 | (2.1) |
| Fatigue | 2 | 1 | (1.0) |
| 3 | 1 | (1.0) | |
| Rectal stenosis | 1 | 1 | (1.0) |
| 3 | 1 | (1.0) | |
| Aspiration | 5 | 1 | (1.0) |
| Prolapse of intestinal stoma | 3 | 1 | (1.0) |
| Postoperative hemorrhage | 3 | 1 | (1.0) |
| Enterocolitis | 2 | 1 | (1.0) |
| Enterocolitis infectious | 2 | 1 | (1.0) |
| Anal pain | 2 | 1 | (1.0) |
| Stoma site infection | 2 | 1 | (1.0) |
| Intestinal stoma leak | 2 | 1 | (1.0) |
| Large intestinal anastomotic leak | 2 | 1 | (1.0) |
| Lymphedema | 2 | 1 | (1.0) |
| Stomal ulcer | 2 | 1 | (1.0) |
| Diarrhea | 2 | 1 | (1.0) |
| Chylous ascites | 1 | 1 | (1.0) |
| Univariate Analysis | Multivariate Analysis | ||||
|---|---|---|---|---|---|
| Factors | n = 97 | Hazard Ratio (95% CI) | p-Value | Hazard Ratio (95% CI) | p-Value |
| Age (y) | |||||
| < 65 | 48 | 1 | 0.191 | 1 | 0.865 |
| ≥ 65 | 49 | 2.177 (0.684–8.168) | 1.137 (0.256–5.358) | ||
| Sex | |||||
| Male | 52 | 1 | 0.309 | ||
| Female | 45 | 1.805 (0.576–6.103) | |||
| ECOG-PS | |||||
| 0 | 80 | 1 | 0.378 | ||
| 1- | 17 | 0.440 (0.024–2.264) | |||
| Location of primary tumor | |||||
| Ra | 23 | 1 | 0.456 | ||
| Rb | 74 | 1.725 (0.454–11.226) | |||
| Primary tumor stage | |||||
| cT1-3 | 84 | 1 | 0.303 | ||
| cT4 | 13 | 2.083 (0.462–7.000) | |||
| cStage | |||||
| 2 | 9 | 1 | 0.093 | 1 | 0.306 |
| 3 | 88 | not applicable | not applicable | ||
| Previous chemotherapy before neoadjuvant radiotherapy | |||||
| Yes | 34 | 1 | 0.466 | ||
| No | 63 | 0.647 (0.206–2.193) | |||
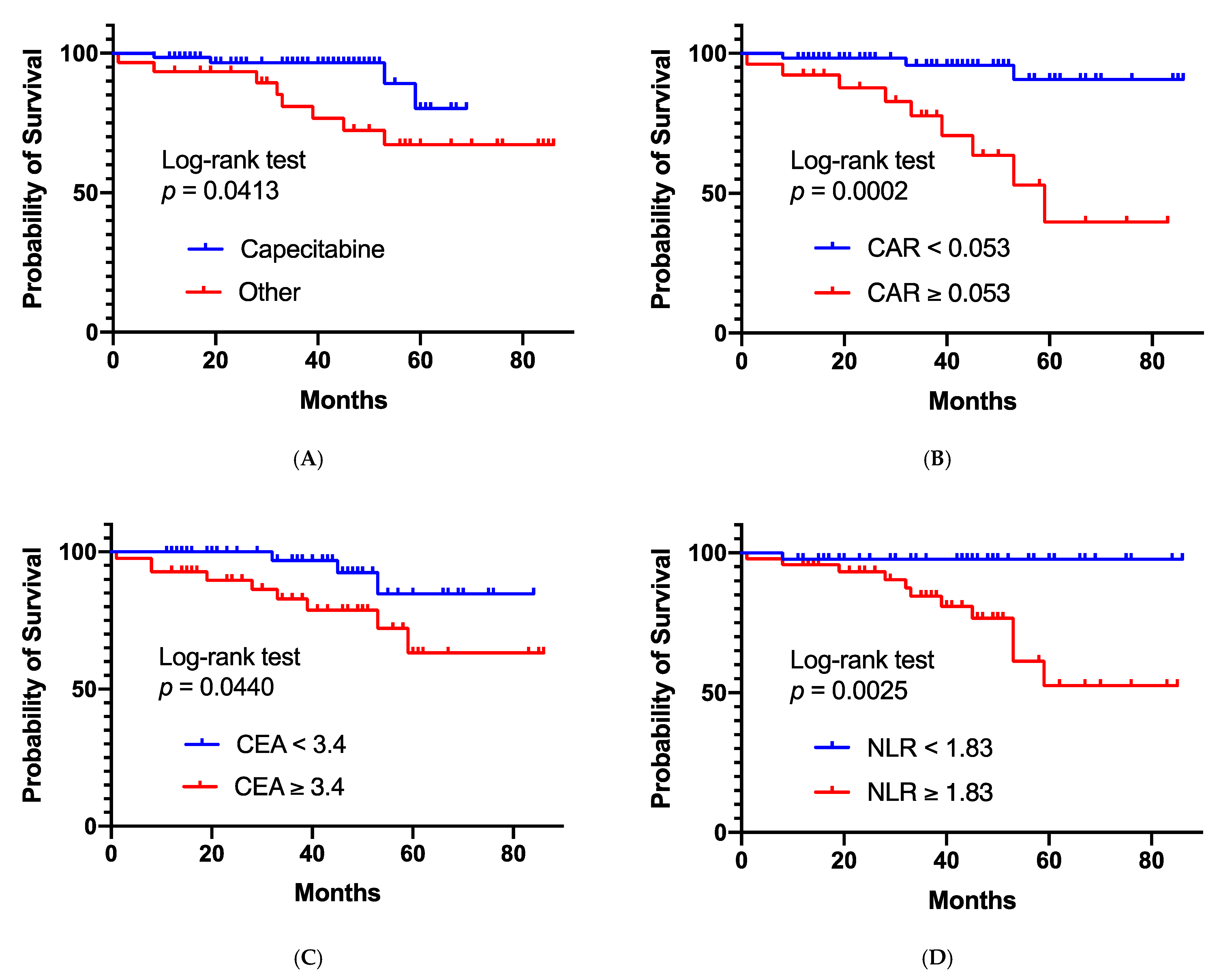
| Chemotherapy administered with radiotherapy | |||||
| Capecitabine | 67 | 1 | 0.045 | 1 | 0.223 |
| Other | 30 | 3.310 (1.027–12.538) | 2.598 (0.574–14.103) | ||
| Capecitabine | 67 | 1 | 0.132 | ||
| S-1 | 23 | 2.667 (0.741–10.655) | |||
| Adjuvant chemotherapy | |||||
| Yes | 37 | 1 | 0.777 | ||
| No | 60 | 1.189 (0.373–4.469) | |||
| Residual tumor condition after surgery | |||||
| R0 resection | 93 | 1 | 0.084 | 1 | 0.443 |
| R1 or presence of dissemination | 4 | 4.988 (0.765–19.004) | 2.212 (0.249–14.846) | ||
| Sphincter preservation surgery | |||||
| Yes | 90 | 1 | 0.230 | ||
| No | 7 | not applicable | |||
| Interval between neoadjuvant radiotherapy and surgery | |||||
| < 6 weeks | 36 | 1 | 0.165 | 1 | 0.378 |
| ≥ 6 weeks | 61 | 0.432 (0.113–1.404) | 2.082 (0.396–10.681) | ||
| Pathological response to the preoperative treatment | n = 92 | ||||
| Responder | 48 | 1 | 0.806 | ||
| Non-responder | 44 | 1.153 (0.360–3.696) | |||
| Pathological findings | |||||
| Complete response | 13 | 1 | 0.447 | ||
| Other | 84 | 2.054 (0.398–37.583) | |||
| C- reactive-protein-to-albumin ratio | n = 85 | ||||
| <0.053 | 59 | 1 | 0.001 | 1 | 0.157 |
| ≥0.053 | 26 | 8.103 (2.407–36.632) | 2.801 (0.681–14.356) | ||
| CEA | n = 87 | ||||
| <3.4 ng/mL | 46 | 1 | 0.041 | 1 | 0.400 |
| ≥3.4 ng/mL | 41 | 3.522 (1.050–15.885) | 1.819 (0.461–8.924) | ||
| Neutrophil-to-lymphocyte ratio | n = 90 | ||||
| <1.83 | 43 | 1 | 0.001 | 1 | 0.018 |
| ≥1.83 | 47 | 12.054 (2.328–220.775) | 8.682 (1.376–175.625) | ||
| Adverse events per patient | n = 97 | Incidence (%) | |
| Any | 45 | (46.4) | |
| Worst grade per patient | n = 97 | Incidence (%) | |
| Grade | 1 | 12 | (12.4) |
| 2 | 23 | (23.7) | |
| 3 | 10 | (10.3) | |
| None | 47 | (48.5) | |
| Adverse events | Grade | n = 97 | Incidence (%) |
| Diarrhea | 1 | 13 | (13.4) |
| 2 | 10 | (10.3) | |
| Ileus | 1 | 1 | (1.0) |
| 2 | 3 | (3.1) | |
| Colonic obstruction | 2 | 2 | (2.1) |
| 3 | 1 | (1.0) | |
| Wound dehiscence | 2 | 1 | (1.0) |
| 3 | 2 | (2.1) | |
| Pelvic infection | 2 | 1 | (1.0) |
| 3 | 2 | (2.1) | |
| Constipation | 1 | 2 | (2.1) |
| 2 | 1 | (1.0) | |
| Gastrointestinal fistula | 3 | 2 | (2.1) |
| Vascular access complication | 3 | 1 | (1.0) |
| Gastrointestinal disorders—other, specify (mucosal prolapse) | 3 | 1 | (1.0) |
| Anal pain | 3 | 1 | (1.0) |
| Enterocolitis | 2 | 1 | (1.0) |
| Rectal anastomotic leak | 2 | 1 | (1.0) |
| Erectile dysfunction | 2 | 1 | (1.0) |
| Cystitis noninfective | 2 | 1 | (1.0) |
| Rectal stenosis | 2 | 1 | (1.0) |
| Colonic fistula | 1 | 1 | (1.0) |
| Fatigue | 1 | 1 | (1.0) |
| Anal pain | 1 | 1 | (1.0) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Doi, H.; Yokoyama, H.; Beppu, N.; Fujiwara, M.; Harui, S.; Kakuno, A.; Yanagi, H.; Hishikawa, Y.; Yamanaka, N.; Kamikonya, N. Neoadjuvant Modified Short-Course Radiotherapy Followed by Delayed Surgery for Locally Advanced Rectal Cancer. Cancers 2021, 13, 4112. https://doi.org/10.3390/cancers13164112
Doi H, Yokoyama H, Beppu N, Fujiwara M, Harui S, Kakuno A, Yanagi H, Hishikawa Y, Yamanaka N, Kamikonya N. Neoadjuvant Modified Short-Course Radiotherapy Followed by Delayed Surgery for Locally Advanced Rectal Cancer. Cancers. 2021; 13(16):4112. https://doi.org/10.3390/cancers13164112
Chicago/Turabian StyleDoi, Hiroshi, Hiroyuki Yokoyama, Naohito Beppu, Masayuki Fujiwara, Shogo Harui, Ayako Kakuno, Hidenori Yanagi, Yoshio Hishikawa, Naoki Yamanaka, and Norihiko Kamikonya. 2021. "Neoadjuvant Modified Short-Course Radiotherapy Followed by Delayed Surgery for Locally Advanced Rectal Cancer" Cancers 13, no. 16: 4112. https://doi.org/10.3390/cancers13164112
APA StyleDoi, H., Yokoyama, H., Beppu, N., Fujiwara, M., Harui, S., Kakuno, A., Yanagi, H., Hishikawa, Y., Yamanaka, N., & Kamikonya, N. (2021). Neoadjuvant Modified Short-Course Radiotherapy Followed by Delayed Surgery for Locally Advanced Rectal Cancer. Cancers, 13(16), 4112. https://doi.org/10.3390/cancers13164112







