Predictors of Sentinel Lymph Node Metastasis in Postoperatively Upgraded Invasive Breast Carcinoma Patients
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
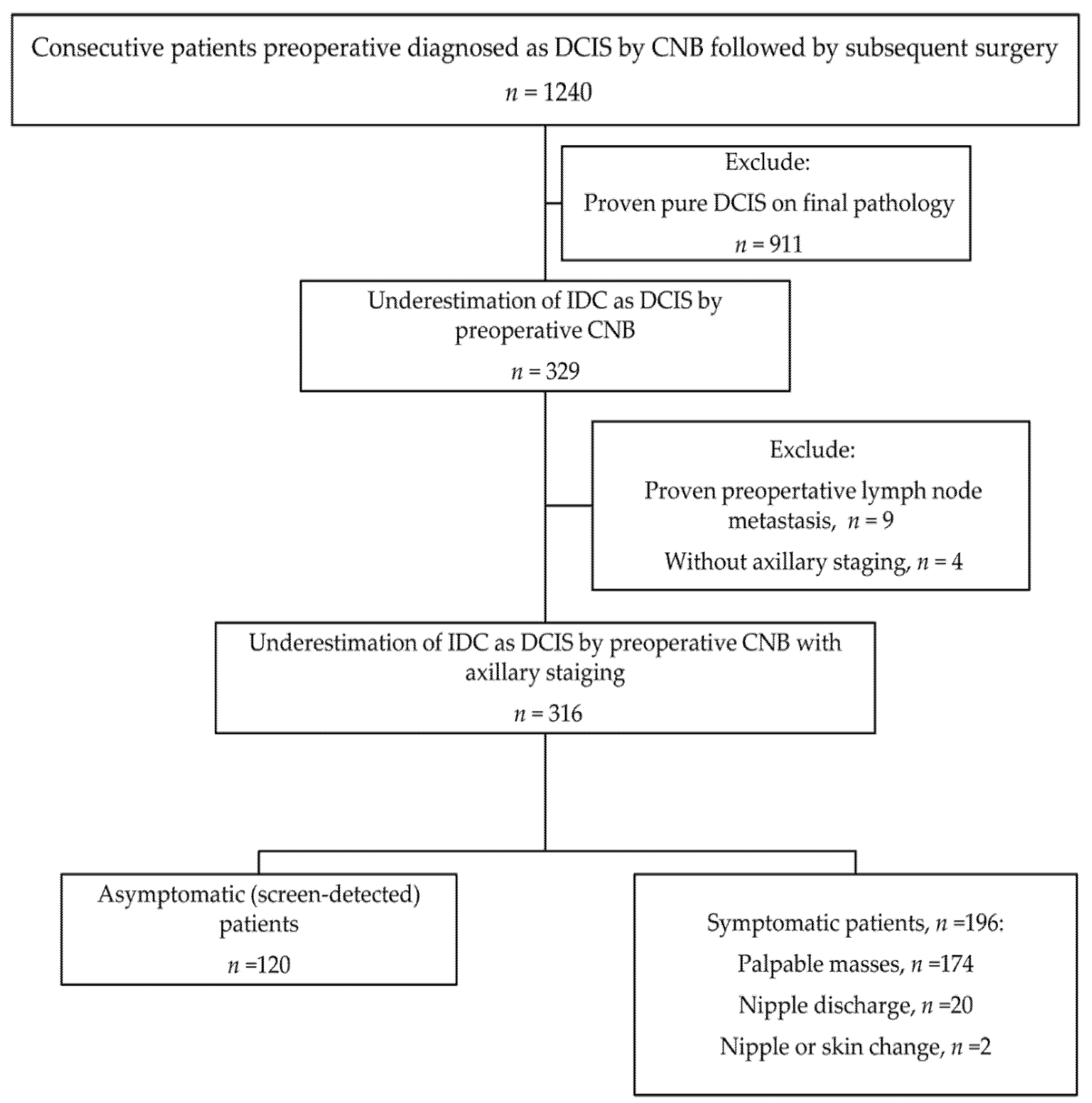
2.1. Patients
2.2. Image-Guided Core Needle Biopsy Procedure
2.3. Radiologic Features of Tumor
2.4. Surgical Procedure
2.5. Pathological Assessment
2.6. Statistical Analysis
3. Results
3.1. Clinical Data
3.2. Radiologic Features
3.3. Final Pathological Findings
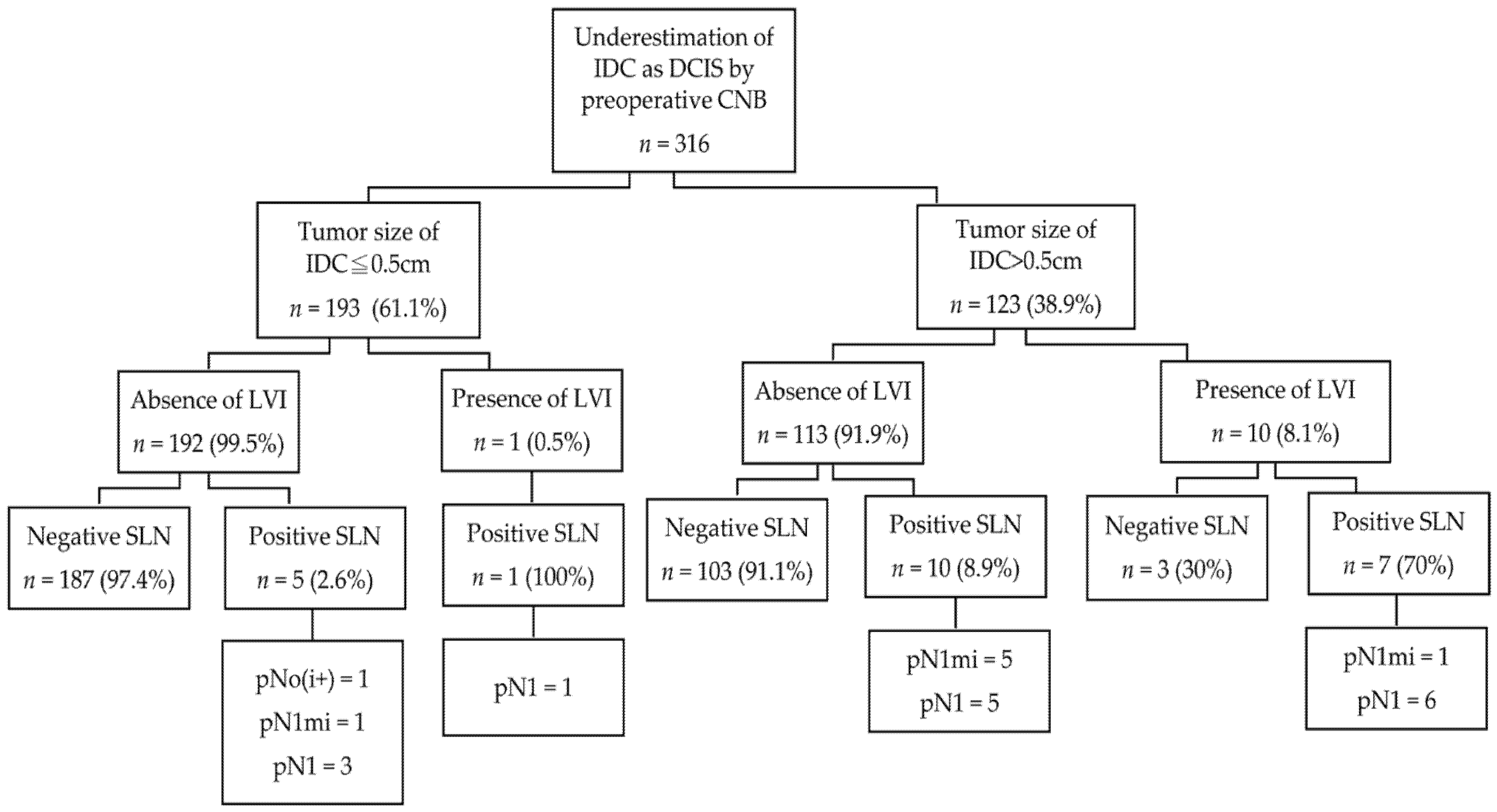
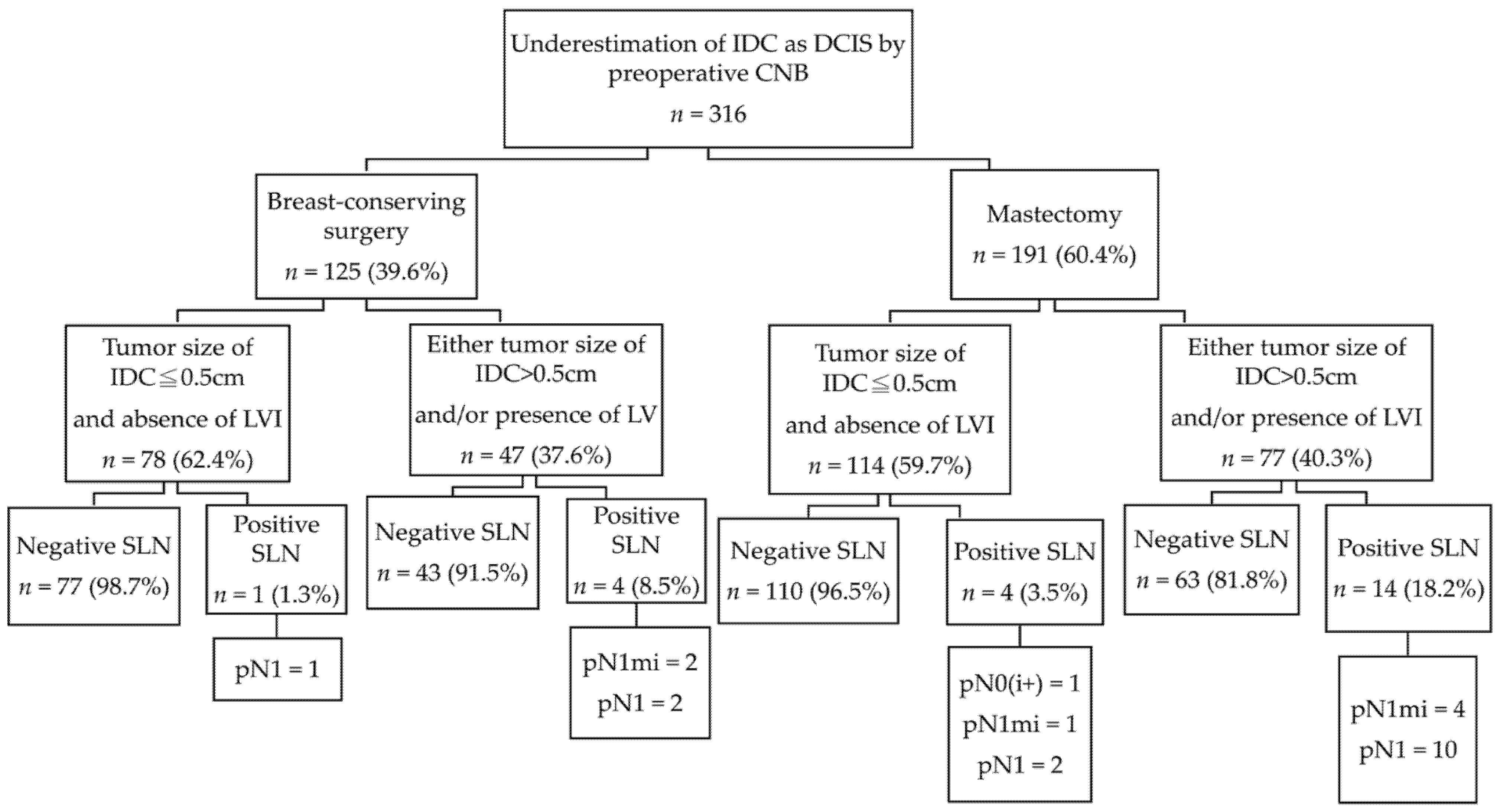
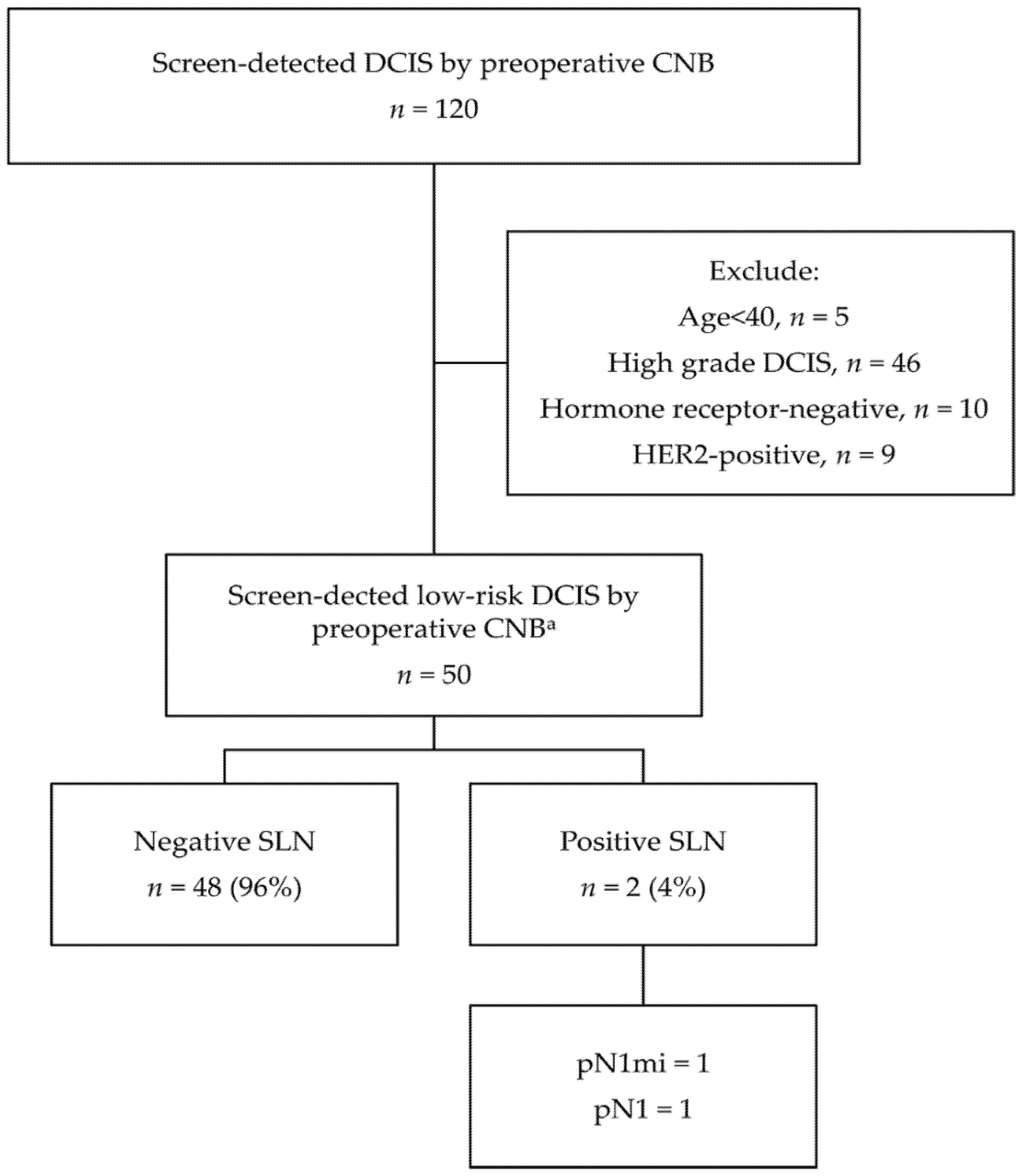
3.4. Evaluation of Axillary Lymph Node Status
3.5. Predictors of Sentinel Lymph Node Metastasis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gutwein, L.G.; Ang, D.N.; Liu, H.; Marshall, J.K.; Hochwald, S.N.; Copeland, E.M.; Grobmyer, S.R. Utilization of minimally invasive breast biopsy for the evaluation of suspicious breast lesions. Am. J. Surg. 2011, 202, 127–132. [Google Scholar] [CrossRef]
- Siziopikou, K.P. Ductal carcinoma in situ of the breast: Current concepts and future directions. Arch. Pathol. Lab. Med. 2013, 137, 462–466. [Google Scholar] [CrossRef]
- Brennan, M.E.; Turner, R.M.; Ciatto, S.; Marinovich, M.L.; French, J.R.; Macaskill, P.; Houssami, N. Ductal carcinoma in situ at core-needle biopsy: Meta-analysis of underestimation and predictors of invasive breast cancer. Radiology 2011, 260, 119–128. [Google Scholar] [CrossRef]
- Lyman, G.H.; Somerfield, M.R.; Bosserman, L.D.; Perkins, C.L.; Weaver, D.L.; Giuliano, A.E. Sentinel Lymph Node Biopsy for Patients With Early-Stage Breast Cancer: American Society of Clinical Oncology Clinical Practice Guideline Update. J. Clin. Oncol. 2017, 35, 561–564. [Google Scholar] [CrossRef] [Green Version]
- Veronesi, U.; Paganelli, G.; Viale, G.; Luini, A.; Zurrida, S.; Galimberti, V.; Intra, M.; Veronesi, P.; Robertson, C.; Maisonneuve, P.; et al. A randomized comparison of sentinel-node biopsy with routine axillary dissection in breast cancer. N. Engl. J. Med. 2003, 349, 546–553. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Crane-Okada, R.; Wascher, R.A.; Elashoff, D.; Giuliano, A.E. Long-term morbidity of sentinel node biopsy versus complete axillary dissection for unilateral breast cancer. Ann. Surg. Oncol. 2008, 15, 1996–2005. [Google Scholar] [CrossRef]
- Han, J.S.; Molberg, K.H.; Sarode, V. Predictors of invasion and axillary lymph node metastasis in patients with a core biopsy diagnosis of ductal carcinoma in situ: An analysis of 255 cases. Breast J. 2011, 17, 223–229. [Google Scholar] [CrossRef]
- Prendeville, S.; Ryan, C.; Feeley, L.; O’Connell, F.; Browne, T.J.; O’Sullivan, M.J.; Bennett, M.W. Sentinel lymph node biopsy is not warranted following a core needle biopsy diagnosis of ductal carcinoma in situ (DCIS) of the breast. Breast 2015, 24, 197–200. [Google Scholar] [CrossRef] [PubMed]
- Francis, A.M.; Haugen, C.E.; Grimes, L.M.; Crow, J.R.; Yi, M.; Mittendorf, E.A.; Bedrosian, I.; Caudle, A.S.; Babiera, G.V.; Krishnamurthy, S.; et al. Is Sentinel Lymph Node Dissection Warranted for Patients with a Diagnosis of Ductal Carcinoma In Situ? Ann. Surg. Oncol. 2015, 22, 4270–4279. [Google Scholar] [CrossRef] [Green Version]
- van Roozendaal, L.M.; Goorts, B.; Klinkert, M.; Keymeulen, K.B.M.I.; De Vries, B.; Strobbe, L.J.A.; Wauters, C.A.P.; van Riet, Y.E.; Degreef, E.; Rutgers, E.J.T.; et al. Sentinel lymph node biopsy can be omitted in DCIS patients treated with breast conserving therapy. Breast Cancer Res. Treat. 2016, 156, 517–525. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heymans, C.; van Bastelaar, J.; Visschers, R.G.J.; Vissers, Y.L.J. Sentinel Node Procedure Obsolete in Lumpectomy for Ductal Carcinoma In Situ. Clin. Breast Cancer 2017, 17, e87–e93. [Google Scholar] [CrossRef] [PubMed]
- Sorrentino, L.; Sartani, A.; Bossi, D.; Amadori, R.; Nebuloni, M.; Truffi, M.; Bonzini, M.; Riggio, E.; Foschi, D.; Corsi, F. Sentinel node biopsy in ductal carcinoma in situ of the breast: Never justified? Breast J. 2018, 24, 325–333. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, Y.; Anan, K.; Saimura, M.; Koga, K.; Fujino, M.; Mine, M.; Tamiya, S.; Nishihara, K.; Nakano, T.; Mitsuyama, S. Upstaging to invasive ductal carcinoma after mastectomy for ductal carcinoma in situ: Predictive factors and role of sentinel lymph node biopsy. Breast Cancer. 2018, 25, 663–670. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Qian, L.; Zhu, Q.; Chang, C. Prediction of Sentinel Lymph Node Metastasis in Breast Ductal Carcinoma In Situ Diagnosed by Preoperative Core Needle Biopsy. Front. Oncol. 2020, 10, 590686. [Google Scholar] [CrossRef] [PubMed]
- Giuliano, A.E.; McCall, L.; Beitsch, P.; Whitworth, P.W.; Blumencranz, P.; Leitch, A.M.; Saha, S.; Hunt, K.K.; Morrow, M.; Ballman, K. Locoregional recurrence after sentinel lymph node dissection with or without axillary dissection in patients with sentinel lymph node metastases: The American College of Surgeons Oncology Group Z0011 randomized trial. Ann. Surg. 2010, 252, 426–432. [Google Scholar] [CrossRef] [Green Version]
- Galimberti, V.; Cole, B.F.; Zurrida, S.; Viale, G.; Luini, A.; Veronesi, P.; Baratella, P.; Chifu, C.; Sargenti, M.; Intra, M.; et al. Axillary dissection versus no axillary dissection in patients with sentinel-node micrometastases (IBCSG 23-01): A phase 3 randomised controlled trial. Lancet Oncol. 2013, 14, 297–305. [Google Scholar] [CrossRef] [Green Version]
- Gentilini, O.; Veronesi, U. Abandoning sentinel lymph node biopsy in early breast cancer? A new trial in progress at the European Institute of Oncology of Milan (SOUND: Sentinel node vs Observation after axillary UltraSouND). Breast 2012, 21, 678–681. [Google Scholar] [CrossRef]
- Reimer, T.; Hartmann, S.; Stachs, A.; Gerber, B. Local treatment of the axilla in early breast cancer: Concepts from the national surgical adjuvant breast and bowel project B-04 to the planned intergroup sentinel mamma trial. Breast Care 2014, 9, 87–95. [Google Scholar] [CrossRef] [Green Version]
- Van Roozendaal, L.M.; Vane, M.L.G.; van Dalen, T.; van der Hage, J.A.; Strobbe, L.J.A.; Boersma, L.J.; Linn, S.C.; Lobbes, M.B.I.; Poortmans, P.M.P.; Tjan-Heijnen, V.C.G.; et al. Clinically node negative breast cancer patients undergoing breast conserving therapy, sentinel lymph node procedure versus follow-up: A Dutch randomized controlled multicentre trial (BOOG 2013-08). BMC Cancer 2017, 17, 459. [Google Scholar] [CrossRef]
- American College of Radiology. Breast Imaging Reporting and Data System, 5th ed.; American College of Radiology: Reston, VA, USA, 2013. [Google Scholar]
- Hoda, S.A.; Brogi, E.; Koerner, F.C.; Rosen, P.P. (Eds.) Rosen’s Breast Pathology, 5th ed.; Wolters Kluwer: Alphen aan den Rijn, The Netherlands, 2021; p. 478. [Google Scholar]
- Silverstein, M.J.; Poller, D.N.; Waisman, J.R.; Colburn, W.J.; Barth, A.; Gierson, E.D.; Lewinsky, B.; Gamagami, P.; Slamon, D.J. Prognostic classification of breast ductal carcinoma-in-situ. Lancet 1995, 345, 1154–1157. [Google Scholar]
- Allred, D.C. Ductal carcinoma in situ: Terminology, classification, and natural history. J. Natl. Cancer Inst. Monogr. 2010, 41, 134–138. [Google Scholar] [CrossRef]
- Harrison, B.T.; Hwang, E.S.; Partridge, A.H.; Thompson, A.M.; Schnitt, S.J. Variability in diagnostic threshold for comedo necrosis among breast pathologists: Implications for patient eligibility for active surveillance trials of ductal carcinoma in situ. Mod. Pathol. 2019, 32, 1257–1262. [Google Scholar] [CrossRef]
- Zhan, H.; Quinn, M.; Tian, W.; Yan, L.; Khoury, T. Significance of Removing Comedonecrosis as an Exclusion Criterion in Mammary Low-Risk Ductal Carcinoma In Situ Managed in an Active Surveillance Clinical Trial. Am. J. Clin. Pathol. 2021, 26, aqaa276. [Google Scholar] [CrossRef]
- Elston, C.W.; Ellis, I.O. Pathological prognostic factors in breast cancer. I. The value of histological grade in breast cancer: Experience from a large study with long-term follow-up. Histopathology 1991, 19, 403–410. [Google Scholar] [CrossRef]
- Hammond, M.E.; Hayes, D.F.; Dowsett, M.; Allred, D.C.; Hagerty, K.L.; Badve, S.; Fitzgibbons, P.L.; Francis, G.; Goldstein, N.S.; Hayes, M.; et al. American Society of Clinical Oncology/College of American Pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer (unabridged version). Arch. Pathol. Lab. Med. 2010, 134, e48–e72. [Google Scholar] [CrossRef]
- Hwang, E.S.; Hyslop, T.; Lynch, T.; Frank, E.; Pinto, D.; Basila, D.; Collyar, D.; Bennett, A.; Kaplan, C.; Rosenberg, S.; et al. The COMET (Comparison of Operative versus Monitoring and Endocrine Therapy) trial: A phase III randomised controlled clinical trial for low-risk ductal carcinoma in situ (DCIS). BMJ Open 2019, 12, e026797. [Google Scholar] [CrossRef] [Green Version]
- Schwartz, G.F.; Patchefsky, A.S.; Finklestein, S.D.; Sohn, S.H.; Prestipino, A.; Feig, S.A.; Singer, J.S. Nonpalpable in situ ductal carcinoma of the breast. Predictors of multicentricity and microinvasion and implications for treatment. Arch. Surg. 1989, 124, 29–32. [Google Scholar] [CrossRef]
- Silverstein, M.J.; Waisman, J.R.; Gamagami, P.; Gierson, E.D.; Colburn, W.J.; Rosser, R.J.; Gordon, P.S.; Lewinsky, B.S.; Fingerhut, A. Intraductal carcinoma of the breast (208 cases). Clinical factors influencing treatment choice. Cancer 1990, 66, 102–108. [Google Scholar] [CrossRef]
- Virnig, B.A.; Tuttle, T.M.; Shamliyan, T.; Kane, R.L. Ductal carcinoma in situ of the breast: A systematic review of incidence, treatment, and outcomes. J. Natl. Cancer Inst. 2010, 102, 170–178. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boyages, J.; Delaney, G.; Taylor, R. Predictors of local recurrence after treatment of ductal carcinoma in situ: A meta-analysis. Cancer 1999, 85, 616–628. [Google Scholar] [CrossRef]
- Giuliano, A.E.; Barth, A.M.; Spivack, B.; Beitsch, P.D.; Evans, S.W. Incidence and predictors of axillary metastasis in T1 carcinoma of the breast. J. Am. Coll. Surg. 1996, 183, 185–189. [Google Scholar]
- Barth, A.; Craig, P.H.; Silverstein, M.J. Predictors of axillary lymph node metastases in patients with T1 breast carcinoma. Cancer 1997, 79, 1918–1922. [Google Scholar] [CrossRef]
- Markopoulos, C.; Kouskos, E.; Gogas, H.; Mandas, D.; Kakisis, J.; Gogas, J. Factors affecting axillary lymph node metastases in patients with T1 breast carcinoma. Am. Surg. 2000, 66, 1011–1013. [Google Scholar] [PubMed]
- Ran, S.; Volk, L.; Hall, K.; Flister, M.J. Lymphangiogenesis and lymphatic metastasis in breast cancer. Pathophysiology 2010, 17, 229–251. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sleeman, J.P.; Thiele, W. Tumor metastasis and the lymphatic vasculature. Int. J. Cancer 2009, 125, 2747–2756. [Google Scholar] [CrossRef] [PubMed]
- Podgrabinska, S.; Skobe, M. Role of lymphatic vasculature in regional and distant metastases. Microvasc. Res. 2014, 95, 46–52. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lauria, R.; Perrone, F.; Carlomagno, C.; De Laurentiis, M.; Morabito, A.; Gallo, C.; Varriale, E.; Pettinato, G.; Panico, L.; Petrella, G.; et al. The prognostic value of lymphatic and blood vessel invasion in operable breast cancer. Cancer 1995, 76, 1772–1778. [Google Scholar] [CrossRef]
- Bader, A.A.; Tio, J.; Petru, E.; Bühner, M.; Pfahlberg, A.; Volkholz, H.; Tulusan, A.H. T1 breast cancer: Identification of patients at low risk of axillary lymph node metastases. Breast Cancer Res. Treat. 2002, 76, 11–17. [Google Scholar] [CrossRef]
- Ozmen, V.; Karanlik, H.; Cabioglu, N.; Igci, A.; Kecer, M.; Asoglu, O.; Tuzlali, S.; Mudun, A. Factors predicting the sentinel and non-sentinel lymph node metastases in breast cancer. Breast Cancer Res. Treat. 2006, 95, 1–6. [Google Scholar] [CrossRef]
- Gajdos, C.; Tartter, P.I.; Bleiweiss, I.J. Lymphatic invasion, tumor size, and age are independent predictors of axillary lymph node metastases in women with T1 breast cancers. Ann. Surg. 1999, 230, 692–696. [Google Scholar] [CrossRef]
- Wong, H.; Lau, S.; Yau, T.; Cheung, P.; Epstein, R.J. Presence of an in situ component is associated with reduced biological aggressiveness of size-matched invasive breast cancer. Br. J. Cancer. 2010, 102, 1391–1396. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dieterich, M.; Hartwig, F.; Stubert, J.; Klöcking, S.; Kundt, G.; Stengel, B.; Reimer, T.; Gerber, B. Accompanying DCIS in breast cancer patients with invasive ductal carcinoma is predictive of improved local recurrence-free survival. Breast 2014, 23, 346–351. [Google Scholar] [CrossRef] [PubMed]
- Goh, C.W.; Wu, J.; Ding, S.; Lin, C.; Chen, X.; Huang, O.; Chen, W.; Li, Y.; Shen, K.; Zhu, L. Invasive ductal carcinoma with coexisting ductal carcinoma in situ (IDC/DCIS) versus pure invasive ductal carcinoma (IDC): A comparison of clinicopathological characteristics, molecular subtypes, and clinical outcomes. J. Cancer Res. Clin. Oncol. 2019, 145, 1877–1886. [Google Scholar] [CrossRef] [PubMed]
- Lyons, J.M., 3rd; Stempel, M.; Van Zee, K.J.; Cody, H.S., 3rd. Axillary node staging for microinvasive breast cancer: Is it justified? Ann. Surg. Oncol. 2012, 19, 3416–3421. [Google Scholar] [CrossRef]
| Variable | No. of Cases | % |
|---|---|---|
| Age (years), median (IQR) | 53 (16) | |
| Lesion location | ||
| Left breast | 180 | 57.0 |
| Right breast | 136 | 43.0 |
| First-degree family history of breast cancer | ||
| Yes | 31 | 9.8 |
| No | 285 | 90.2 |
| Palpability | ||
| Palpable | 174 | 55.1 |
| Non-palpable | 142 | 44.9 |
| Lesion-detecting imaging modality | ||
| Ultrasonography only | 44 | 13.9 |
| Mammography only | 36 | 11.4 |
| Detected by both ultrasonography and mammography | 236 | 74.7 |
| Radiologic morphology | ||
| Mass with calcifications | 164 | 51.9 |
| Mass without calcifications | 85 | 26.9 |
| Non-mass | 67 | 21.2 |
| Parenchymal density | ||
| Entirely fatty | 11 | 3.5 |
| Scattered fibroglandular | 52 | 16.5 |
| Heterogeneously dense | 179 | 56.6 |
| Extremely dense | 74 | 23.4 |
| MMG-BI-RADS category | ||
| 0 | 49 | 15.5 |
| 1 | 6 | 1.9 |
| 2 | 26 | 8.2 |
| 4a | 50 | 15.8 |
| 4b | 71 | 22.5 |
| 4c | 78 | 24.7 |
| 5 | 36 | 11.4 |
| Ultrasound-BI-RADS category | ||
| 1 | 15 | 4.7 |
| 2 | 10 | 3.2 |
| 3 | 10 | 3.2 |
| 4a | 82 | 25.9 |
| 4b | 72 | 22.8 |
| 4c | 75 | 23.7 |
| 5 | 52 | 16.5 |
| Image-guided procedure | ||
| Ultrasound-guided | 275 | 87.0 |
| MMG-guided | 41 | 13.0 |
| Needle gauge | ||
| 7/10 | 38 | 12.0 |
| 14/16/18 | 278 | 88.0 |
| Operation type | ||
| Mastectomy | 191 | 60.4 |
| BCS | 125 | 39.6 |
| DCIS tumor size (cm), median (IQR) | 3.3 (2.6) | |
| Tumor necrosis area | ||
| None | 36 | 11.4 |
| Focal | 178 | 56.3 |
| Large | 102 | 32.3 |
| DCIS tumor grading | ||
| Low | 34 | 10.7 |
| Intermediate | 144 | 45.6 |
| High | 138 | 43.7 |
| DCIS architecture pattern | ||
| Comedo | 122 | 38.6 |
| Non-comedo | 194 | 61.4 |
| Lymphovascular invasion | ||
| Present | 11 | 3.5 |
| Absent | 305 | 96.5 |
| IDC area | ||
| Multifocal/multicentric | 142 | 44.9 |
| Unifocal | 174 | 55.1 |
| IDC a | ||
| T1mi | 101 | 32.0 |
| T1a | 92 | 29.1 |
| T1b | 50 | 15.8 |
| T1c | 47 | 14.9 |
| T2 | 26 | 8.2 |
| IDC tumor grading | ||
| 1 | 81 | 25.6 |
| 2 | 133 | 42.1 |
| 3 | 44 | 13.9 |
| Unknown | 58 | 18.4 |
| Estrogen receptor status | ||
| Positive | 197 | 62.3 |
| Negative | 119 | 37.7 |
| Progesterone receptor status | ||
| Positive | 176 | 55.7 |
| Negative | 140 | 44.3 |
| HER2 status | ||
| Positive | 110 | 34.8 |
| Negative | 206 | 65.2 |
| Ki-67 | ||
| <20 | 221 | 69.9 |
| ≥20 | 70 | 22.2 |
| Unknown | 25 | 7.9 |
| Age (yrs) | Radiologic Morphology | Surgery Type | Final Pathology | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| LVI | DCIS (mm) | IDC (mm) | Molecular Subtype | No. of SLNs a | pN by SLNB b | No. of LN by ALND c | Final TNM Stage b | |||
| 50 | non-mass | Mast. | present | 26 | 9 | HER2 positive | 1/1 | pN1 | 1/17 | pT1bN1aM0 |
| 38 | mass without calcifications | Mast. | absent | 37 | 9 | Luminal A | 1/1 | pN1mi | 0/39 | pT1bN1miM0 |
| 54 | mass without calcifications | BCS | absent | 11 | 7 | Luminal A | 1/1 | pN1 | 0/20 | pT1bN1M0 |
| 55 | mass with calcifications | Mast. | absent | 70 | <1 | HER2 positive | 1/2 | pN1 | 0/12 | pT1miN1aM0 |
| 42 | mass with calcifications | Mast. | present | 56 | 12 | Luminal B/HER2 positive | 1/2 | pN1mi | 0/20 | pT1cN1miM0 |
| 69 | mass with calcifications | Mast. | present | 10 | 35 | Luminal A | 1/2 | pN1 | 0/10 | pT2N1aM0 |
| 41 | mass with calcifications | Mast. | present | 42 | 32 | Luminal A | 1/1 | pN1 | 1/16 | pT2N1aM0 |
| 41 | mass with calcifications | Mast. | absent | 6 | 8 | Triple negative | 1/1 | pN1mi | NP | pT1bN1miM0 |
| 68 | mass without calcifications | Mast. | absent | 52 | <1 | Luminal A | 1/5 | pN0(i+) | NP | pT1miN0(i+)M0 |
| 59 | mass without calcifications | BCS | absent | 47 | 2.5 | Triple negative | 1/1 | pN1 | 0/20 | pT1aN1aM0 |
| 53 | mass without calcifications | Mast. | absent | 46 | 12 | HER2 positive | 1/1 | pN1 | 0/44 | pT1cN1aM0 |
| 42 | mass without calcifications | Mast. | absent | 60 | 19 | Luminal A | 1/1 | pN1 | NP | pT1cN1aM0 |
| 65 | mass without calcifications | Mast. | absent | 48 | 20 | Luminal A | 2/2 | pN1 | 0/14 | pT1cN1aM0 |
| 64 | mass without calcifications | Mast. | present | 25 | 45 | Luminal A | 1/2 | pN1 | 1/28 | pT2N1aM0 |
| 47 | non-mass | Mast. | present | 109 | 12 | HER2 positive | 1/2 | pN1 | 0/20 | pT1cN1aM0 |
| 74 | mass with calcifications | Mast. | present | 38 | <1 | HER2 positive | 1/2 | pN1 | 1/18 | pT1miN1aM0 |
| 57 | mass with calcifications | Mast. | absent | 50 | 17 | Luminal B HER2 negative | 1/2 | pN1mi | NP | pT1cN1miM0 |
| 37 | mass with calcifications | BCS | absent | 18 | 18 | Luminal B/HER2 negative | 1/2 | pN1mi | NP | pT1cN1miM0 |
| 39 | mass with calcifications | Mast. | present | 47 | 39 | Luminal B/HER2 negative | 3/3 | pN1 | 3/12 | pT2N2aM0 |
| 42 | mass with calcifications | BCS | absent | 40 | 31 | Luminal B/HER2 negative | 2/3 | pN1 | 11/27 | pT2N3aM0 |
| 67 | mass without calcifications | Mast. | absent | 45 | 5 | Luminal A | 1/2 | pN1mi | 0/11 | pT1aN1miM0 |
| 44 | non-mass | Mast. | absent | 47 | 1.6 | Luminal B/HER2 negative | 1/2 | pN1 | 0/17 | pT1aN1aM0 |
| 55 | non-mass | BCS | absent | 34 | 32 | Luminal A | 1/1 | pN1mi | NP | pT2N1miM0 |
| Variable | Lymph Node | p Value | |
|---|---|---|---|
| Negative (n = 293) | Positive (n = 23) | ||
| Age (years), median (IQR) | 53.0 (16.0) | 53.0 (22.0) | |
| Lesion location | 0.965 | ||
| Left breast | 167 (57.0) | 13 (56.5) | |
| Right breast | 126 (43.0) | 10 (43.5) | |
| 1st degree family history of breast cancer | 0.146 | ||
| Yes | 31 (10.6) | 0 | |
| No | 262 (89.4) | 23 (100.0) | |
| Palpability | 0.059 | ||
| Palpable | 157 (53.6) | 17 (73.9) | |
| Non-palpable | 136 (46.4) | 6 (26.1) | |
| Lesion-detecting imaging modality | 0.652 | ||
| Ultrasonography only | 42 (14.3) | 2 (8.7) | |
| Mammography only | 34 (11.6) | 2 (8.7) | |
| Detected by both ultrasonography and mammography | 217 (74.1) | 19 (82.6) | |
| Radiologic morphology | 0.389 | ||
| Mass with calcifications | 154 (52.6) | 10 (43.5) | |
| Mass without calcifications | 76 (25.9) | 9 (39.1) | |
| Non-mass with calcifications | 63 (21.5) | 4 (17.4) | |
| Parenchymal density | 0.070 | ||
| Entirely fatty | 8 (2.7) | 3 (13.0) | |
| Scattered fibroglandular | 49 (16.7) | 3 (13.0) | |
| Heterogeneously dense | 166 (56.7) | 13 (56.5) | |
| Extremely dense | 70 (23.9) | 4 (17.4) | |
| MMG-BI-RADS category | 0.492 | ||
| 0 | 43 (14.7) | 6 (26.1) | |
| 1 | 5 (1.7) | 1 (4.3) | |
| 2 | 25 (8.5) | 1 (4.3) | |
| 4a | 49 (16.7) | 1 (4.3) | |
| 4b | 65 (22.2) | 6 (26.1) | |
| 4c | 72 (24.6) | 6 (26.1) | |
| 5 | 34 (11.6) | 2 (8.7) | |
| Ultrasound-BI-RADS category | 0.080 | ||
| 1 | 14 (4.8) | 1 (4.3) | |
| 2 | 10 (3.4) | 0 | |
| 3 | 10 (3.4) | 0 | |
| 4a | 80 (27.3) | 2 (8.7) | |
| 4b | 62 (21.2) | 10 (43.5) | |
| 4c | 67 (22.9) | 8 (34.8) | |
| 5 | 50 (17.1) | 2 (8.7) | |
| Image-guided procedure | 0.751 | ||
| Ultrasound-guided | 254 (86.7) | 21 (91.3) | |
| MMG-guided | 39 (13.3) | 2 (8.7) | |
| Needle gauge | >0.999 | ||
| 7/10 | 36 (12.3) | 2 (8.7) | |
| 14/16/18 | 257 (87.7) | 21 (91.3) | |
| Operation type | 0.070 | ||
| Mastectomy | 173 (59.0) | 18 (78.3) | |
| BCS | 120 (41.0) | 5 (21.7) | |
| DCIS tumor size (cm), median (IQR) | 3.3 (2.6) | 4.5 (2.4) | 0.117 |
| DCIS tumor grading | 0.797 | ||
| Low | 32 (10.9) | 2 (8.7) | |
| Intermediate | 132 (45.1) | 12 (52.2) | |
| High | 129 (44.0) | 9 (39.1) | |
| DCIS architecture pattern | 0.084 | ||
| Comedo | 117 (39.9) | 5 (21.7) | |
| Non-comedo | 176 (60.1) | 18 (78.3) | |
| Tumor necrosis area | 0.912 | ||
| None | 33 (11.3) | 3 (13.0) | |
| Focal | 166 (56.7) | 12 (52.2) | |
| Large | 94 (32.1) | 8 (34.8) | |
| IDC area | 0.884 | ||
| Multiple | 132 (45.1) | 10 (43.5) | |
| Single | 161 (54.9) | 13 (56.5) | |
| IDC tumor size (cm) | <0.001 | ||
| ≦0.5 | 187 (63.8) | 6 (26.1) | |
| >0.5 | 106 (36.2) | 17 (73.9) | |
| IDC tumor grading | 0.165 | ||
| 1 | 78 (26.6) | 3 (13.0) | |
| 2 | 119 (40.6) | 14 (60.9) | |
| 3 | 40 (13.7) | 4 (17.4) | |
| Unknown | 56 (19.1) | 2 (8.7) | |
| Lymphovascular invasion | <0.0001 | ||
| Present | 3 (1.0) | 8 (34.8) | |
| Absent | 290 (99.0) | 15 (65.2) | |
| Estrogen receptor status | 0.458 | ||
| Positive | 181 (61.8) | 16 (69.6) | |
| Negative | 112 (38.2) | 7 (30.4) | |
| Progesterone receptor status | 0.340 | ||
| Positive | 161 (54.9) | 15 (65.2) | |
| Negative | 132 (45.1) | 8 (34.8) | |
| HER2 status | 0.647 | ||
| Positive | 103 (35.2) | 7 (30.4) | |
| Negative | 190 (64.8) | 16 (69.6) | |
| Ki-67 | 0.873 | ||
| <20 | 206 (70.3) | 15 (65.2) | |
| ≧20 | 64 (21.8) | 6 (26.1) | |
| Unknown | 23 (7.8) | 2 (8.7) | |
| Variable | Odds Ratio | 95% Confidence Interval of Odds | p Value |
|---|---|---|---|
| IDC tumor size (cm) | |||
| ≤0.5 | 1 | ||
| >0.5 | 3.11 | 1.09–8.81 | 0.033 |
| Lymphovascular invasion | |||
| Present | 32.85 | 7.56–142.80 | <0.0001 |
| Absent | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, C.-C.; Cheung, Y.-C.; Hsueh, C.; Chen, S.-C. Predictors of Sentinel Lymph Node Metastasis in Postoperatively Upgraded Invasive Breast Carcinoma Patients. Cancers 2021, 13, 4099. https://doi.org/10.3390/cancers13164099
Yu C-C, Cheung Y-C, Hsueh C, Chen S-C. Predictors of Sentinel Lymph Node Metastasis in Postoperatively Upgraded Invasive Breast Carcinoma Patients. Cancers. 2021; 13(16):4099. https://doi.org/10.3390/cancers13164099
Chicago/Turabian StyleYu, Chi-Chang, Yun-Chung Cheung, Chuen Hsueh, and Shin-Cheh Chen. 2021. "Predictors of Sentinel Lymph Node Metastasis in Postoperatively Upgraded Invasive Breast Carcinoma Patients" Cancers 13, no. 16: 4099. https://doi.org/10.3390/cancers13164099
APA StyleYu, C.-C., Cheung, Y.-C., Hsueh, C., & Chen, S.-C. (2021). Predictors of Sentinel Lymph Node Metastasis in Postoperatively Upgraded Invasive Breast Carcinoma Patients. Cancers, 13(16), 4099. https://doi.org/10.3390/cancers13164099





