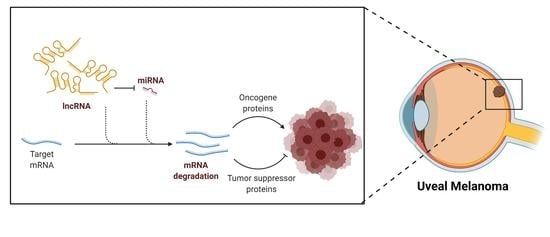
The Role of LncRNAs in Uveal Melanoma
Abstract
Simple Summary
Abstract
1. Introduction
- Intergenic, the lncRNAs transcribed from DNA strands between protein-coding genes. These lncRNAs act as master regulators of transcription and posttranscriptional and translation processes [30].
- Intronic, the lncRNAs transcribed from introns in the same orientation as the mRNA of protein-coding genes. Many of these lncRNAs are implicated in alternative splicing [31].
- Overlapping, the lncRNAs transcribed from overlapping mRNA of protein-coding genes. Many overlapping lncRNAs have implications in splicing, tissue specificity, and aging [32].
- Antisense, the lncRNAs transcribed from the opposite strand (antisense) of protein-coding genes. These lncRNAs can interfere with transcription or mRNA stability [33].
2. Uveal Melanoma
3. LncRNAs in Uveal Melanoma
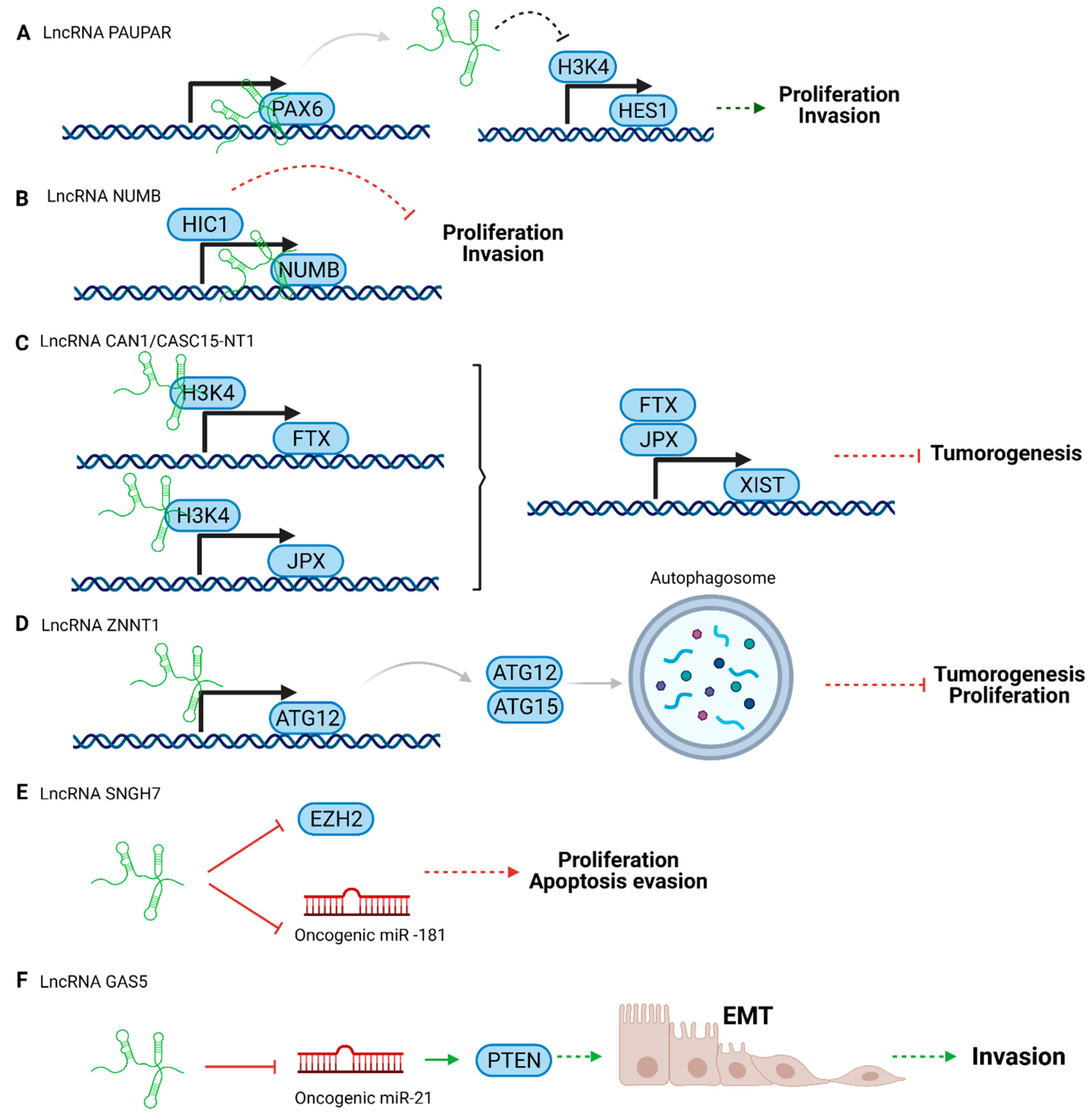
3.1. Tumor Suppressor LncRNAs in Uveal Melanoma
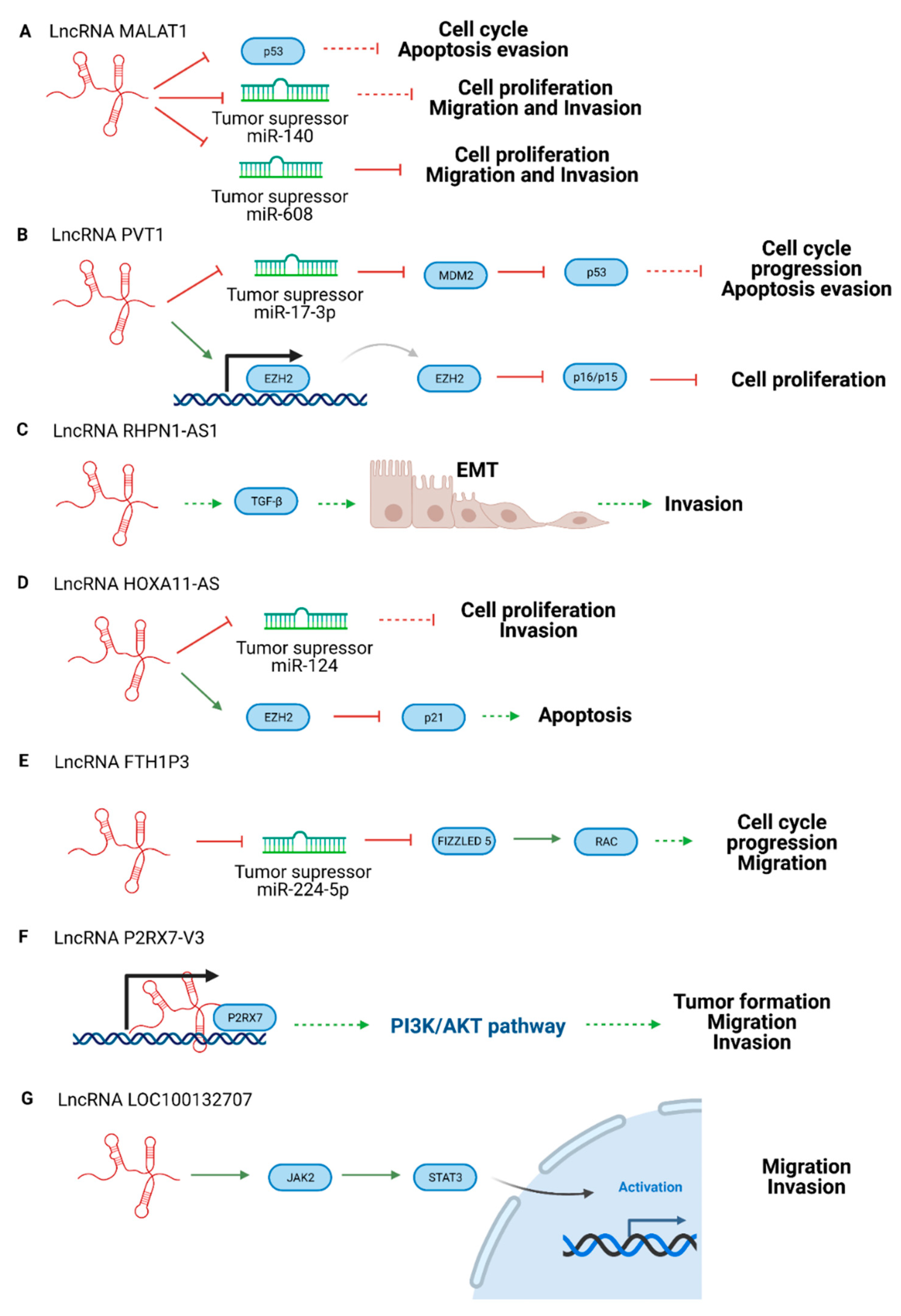
3.2. Oncogenic LncRNAs in Uveal Melanoma
4. LncRNAs as Therapeutic Agents
- Inhibit oncogenic lncRNA expression using specific siRNAs, antisense oligonucleotides (ASOs), gapmers, ribozymes, and Dnazymes, synthetic lncRNA mimics, or CRISPR systems.
- Block the interaction between lncRNAs and their target molecules (e.g., regulatory factors or promoters) or affect the lncRNA secondary structure with aptamers or small synthetic molecules.
5. LncRNAs as Diagnostic Agents
6. LncRNA-microRNA Interactions Related to Uveal Melanoma
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Slack, F.J.; Chinnaiyan, A.M. The Role of Non-coding RNAs in Oncology. Cell 2019, 179, 1033–1055. [Google Scholar] [CrossRef]
- Peng, W.X.; Koirala, P.; Mo, Y.Y. LncRNA-mediated regulation of cell signaling in cancer. Oncogene 2017, 36, 5661–5667. [Google Scholar] [CrossRef]
- Rinn, J.L.; Chang, H.Y. Genome Regulation by Long Noncoding RNAs. Annu. Rev. Biochem. 2012, 81, 145–166. [Google Scholar] [CrossRef]
- Qian, X.; Zhao, J.; Yeung, P.Y.; Zhang, Q.C.; Kwok, C.K. Revealing lncRNA Structures and Interactions by Sequencing-Based Approaches. Trends Biochem. Sci. 2019, 44, 33–52. [Google Scholar] [CrossRef] [PubMed]
- Anastasiadou, E.; Jacob, L.S.; Slack, F.J. Non-coding RNA networks in cancer. Nat. Rev. Cancer 2017, 18, 5–18. [Google Scholar] [CrossRef]
- Mongelli, A.; Martelli, F.; Farsetti, A.; Gaetano, C. The dark that matters: Long noncoding RNAs as master regulators of cellular metabolism in noncommunicable diseases. Front. Physiol. 2019, 10, 369. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.W.; Kim, H.W.; Nam, J.W. The small peptide world in long noncoding RNAs. Brief. Bioinform. 2019, 20, 1853–1864. [Google Scholar] [CrossRef] [PubMed]
- Statello, L.; Guo, C.J.; Chen, L.L.; Huarte, M. Gene regulation by long non-coding RNAs and its biological functions. Nat. Rev. Mol. Cell Biol. 2021, 22, 96–118. [Google Scholar] [CrossRef]
- Akhade, V.S.; Pal, D.; Kanduri, C. Advances in Experimental Medicine and Biology. In Long Noncoding RNA: Genome Organization and Mechanism of Action; Springer Nature Singapore Pte Ltd.: Singapore, 2017; Volume 1008, pp. 47–74. [Google Scholar]
- Willingham, A.T.; Orth, A.P.; Batalov, S.; Peters, E.C.; Wen, B.G.; Aza-Blanc, P.; Hogenesch, J.B.; Schultz, P.G. A Strategy for Probing the Function of Noncoding RNAs Finds a Repressor of NFAT. Science 2005, 309, 1570–1573. [Google Scholar] [CrossRef]
- Shamovsky, I.; Ivannikov, M.; Kandel, E.S.; Gershon, D.; Nudler, E. RNA-mediated response to heat shock in mammalian cells. Nature 2006, 440, 556–560. [Google Scholar] [CrossRef] [PubMed]
- Tripathi, V.; Ellis, J.D.; Shen, Z.; Song, D.Y.; Pan, Q.; Watt, A.T.; Freier, S.M.; Bennett, C.F.; Sharma, A.; Bubulya, P.A.; et al. The Nuclear-Retained Noncoding RNA MALAT1 Regulates Alternative Splicing by Modulating SR Splicing Factor Phosphorylation. Mol. Cell 2010, 39, 925–938. [Google Scholar] [CrossRef] [PubMed]
- Bardou, F.; Ariel, F.; Simpson, C.G.; Romero-Barrios, N.; Laporte, P.; Balzergue, S.; Brown, J.W.S.; Crespi, M. Long Noncoding RNA Modulates Alternative Splicing Regulators in Arabidopsis. Dev. Cell 2014, 30, 166–176. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Liu, X.; Wu, H.; Ni, P.; Gu, Z.; Qiao, Y.; Chen, N.; Sun, F.; Fan, Q. CREB up-regulates long non-coding RNA, HULC expression through interaction with microRNA-372 in liver cancer. Nucleic Acids Res. 2010, 38, 5366–5383. [Google Scholar] [CrossRef] [PubMed]
- Jeon, Y.; Lee, J.T. YY1 Tethers Xist RNA to the Inactive X Nucleation Center. Cell 2011, 146, 119–133. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Li, D.; Zhang, W.; Guo, M.; Zhan, Q. Long non-coding RNA gadd7 interacts with TDP-43 and regulates Cdk6 mRNA decay. EMBO J. 2012, 31, 4415–4427. [Google Scholar] [CrossRef]
- Tsai, M.-C.; Manor, O.; Wan, Y.; Mosammaparast, N.; Wang, J.K.; Lan, F.; Shi, Y.; Segal, E.; Chang, H.Y. Long Noncoding RNA as Modular Scaffold of Histone Modification Complexes. Science 2010, 329, 689–693. [Google Scholar] [CrossRef]
- Prasanth, K.V.; Prasanth, S.G.; Xuan, Z.; Hearn, S.; Freier, S.M.; Bennett, C.F.; Zhang, M.Q.; Spector, D.L. Regulating Gene Expression through RNA Nuclear Retention. Cell 2005, 123, 249–263. [Google Scholar] [CrossRef]
- Lee, S.; Kopp, F.; Chang, T.C.; Sataluri, A.; Chen, B.; Sivakumar, S.; Yu, H.; Xie, Y.; Mendell, J.T. Noncoding RNA NORAD Regulates Genomic Stability by Sequestering PUMILIO Proteins. Cell 2016, 164, 69–80. [Google Scholar] [CrossRef]
- Marchese, F.P.; Grossi, E.; Marín-Béjar, O.; Bharti, S.K.; Raimondi, I.; González, J.; Martínez-Herrera, D.J.; Athie, A.; Amadoz, A.; Brosh, R.M., Jr.; et al. A Long Noncoding RNA Regulates Sister Chromatid Cohesion. Mol. Cell 2016, 63, 397–407. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Schmidt, B.F.; Bruchez, M.P.; McManus, C.J. Structural analyses of NEAT1 lncRNAs suggest long-range RNA interactions that may contribute to paraspeckle architecture. Nucleic Acids Res. 2018, 46, 3742–3752. [Google Scholar] [CrossRef]
- Noh, J.H.; Kim, K.M.; Abdelmohsen, K.; Yoon, J.H.; Panda, A.C.; Munk, R.; Kim, J.; Curtis, J.; Moad, C.A.; Wohler, C.M.; et al. HuR and GRSF1 modulate the nuclear export and mitochondrial localization of the lncRNA RMRP. Genes Dev. 2016, 30, 1224–1239. [Google Scholar] [CrossRef]
- Vendramin, R.; Verheyden, Y.; Ishikawa, H.; Goedert, L.; Nicolas, E.; Saraf, K.; Armaos, A.; Delli Ponti, R.; Izumikawa, K.; Mestdagh, P.; et al. SAMMSON fosters cancer cell fitness by concertedly enhancing mitochondrial and cytosolic translation. Nat. Struct. Mol. Biol. 2018, 25, 1035–1046. [Google Scholar] [CrossRef] [PubMed]
- Ferrè, F.; Colantoni, A.; Helmer-Citterich, M. Revealing protein-lncRNA interaction. Brief. Bioinform. 2016, 17, 106–116. [Google Scholar] [CrossRef] [PubMed]
- Charles Richard, J.L.; Eichhorn, P.J.A. Platforms for Investigating LncRNA Functions. SLAS Technol. 2018, 23, 493–506. [Google Scholar] [CrossRef]
- Chu, C.; Qu, K.; Zhong, F.L.; Artandi, S.E.; Chang, H.Y. Genomic Maps of Long Noncoding RNA Occupancy Reveal Principles of RNA-Chromatin Interactions. Mol. Cell 2011, 44, 667–678. [Google Scholar] [CrossRef]
- Kazimierczyk, M.; Kasprowicz, M.K.; Kasprzyk, M.E.; Wrzesinski, J. Human long noncoding RNA interactome: Detection, characterization and function. Int. J. Mol. Sci. 2020, 21, 1027. [Google Scholar] [CrossRef]
- Kopp, F.; Mendell, J.T. Functional Classification and Experimental Dissection of Long Noncoding RNAs. Cell 2018, 172, 393–407. [Google Scholar] [CrossRef] [PubMed]
- Mattick, J.S.; Rinn, J.L. Discovery and annotation of long noncoding RNAs. Nat. Struct. Mol. Biol. 2015, 22, 5–7. [Google Scholar] [CrossRef] [PubMed]
- Marques-Rocha, J.L.; Samblas, M.; Milagro, F.I.; Bressan, J.; Martínez, J.A.; Marti, A. Noncoding RNAs, cytokines, and inflammation-related diseases. FASEB J. 2015, 29, 3595–3611. [Google Scholar] [CrossRef] [PubMed]
- Matsui, M.; Corey, D.R. Non-coding RNAs as drug targets. Nat. Rev. Drug Discov. 2017, 16, 167–179. [Google Scholar] [CrossRef]
- Ning, Q.; Li, Y.; Wang, Z.; Zhou, S.; Sun, H.; Yu, G. The Evolution and Expression Pattern of Human Overlapping lncRNA and Protein-coding Gene Pairs. Sci. Rep. 2017, 7, 1–12. [Google Scholar] [CrossRef]
- Katayama, S.; Tomaru, Y.; Kasukawa, T.; Waki, K.; Nakanishi, M.; Nakamura, M.; Nishida, H.; Yap, C.C.; Suzuki, M.; Kawai, J.; et al. Molecular biology: Antisense transcription in the mammalian transcriptome. Science 2005, 309, 1564–1566. [Google Scholar] [CrossRef]
- Hua, J.T.; Ahmed, M.; Guo, H.; Zhang, Y.; Chen, S.; Soares, F.; Lu, J.; Zhou, S.; Wang, M.; Li, H.; et al. Risk SNP-Mediated Promoter-Enhancer Switching Drives Prostate Cancer through lncRNA PCAT19. Cell 2018, 174, 564–575.e18. [Google Scholar] [CrossRef]
- Ma, L.; Sun, X.; Kuai, W.; Hu, J.; Yuan, Y.; Feng, W.; Lu, X. LncRNA SOX2 overlapping transcript acts as a miRNA sponge to promote the proliferation and invasion of Ewing’s sarcoma. Am. J. Transl. Res. 2019, 11, 3841–3849. [Google Scholar]
- Wang, J.; Su, Z.; Lu, S.; Fu, W.; Liu, Z.; Jiang, X.; Tai, S. LncRNA HOXA-AS2 and its molecular mechanisms in human cancer. Clin. Chim. Acta 2018, 485, 229–233. [Google Scholar] [CrossRef] [PubMed]
- Ulitsky, I.; Bartel, D.P. XLincRNAs: Genomics, evolution, and mechanisms. Cell 2013, 154, 26. [Google Scholar] [CrossRef] [PubMed]
- Hanahan, D.; Weinberg, R.A.A. Hallmarks of Cancer: The Next Generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Bhan, A.; Mandal, S.S. Long noncoding RNAs: Emerging stars in gene regulation, epigenetics and human disease. ChemMedChem 2014, 9, 1932–1956. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, A.M.; Chang, H.Y. Long Noncoding RNAs in Cancer Pathways. Cancer Cell 2016, 29, 452–463. [Google Scholar] [CrossRef]
- Renganathan, A.; Felley-Bosco, E. Long noncoding RNAs in cancer and therapeutic potential. Adv. Exp. Med. Biol. 2017, 1008, 199–222. [Google Scholar] [CrossRef] [PubMed]
- Huarte, M. The emerging role of lncRNAs in cancer. Nat. Med. 2015, 21, 1253–1261. [Google Scholar] [CrossRef]
- Guzel, E. Tumor Suppressor and Oncogenic Role of Long Non-Coding RNAs in Cancer. North. Clin. Istanbul 2019, 7, 81–86. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Chen, Y.; Nie, X. Regulatory Mechanisms of lncRNAs and Their Target Gene Signaling Pathways in Laryngeal Squamous Cell Carcinoma. Front. Pharmacol. 2020, 11, 1140. [Google Scholar] [CrossRef] [PubMed]
- Dahl, M.; Kristensen, L.S.; Grønbæk, K. Long non-coding RNAs guide the fine-tuning of gene regulation in B-cell development and malignancy. Int. J. Mol. Sci. 2018, 19, 2475. [Google Scholar] [CrossRef] [PubMed]
- Shin, T.J.; Lee, K.H.; Cho, J.Y. Epigenetic mechanisms of lncrnas binding to protein in carcinogenesis. Cancers 2020, 12, 2925. [Google Scholar] [CrossRef]
- Sun, Y.; Ma, L. New Insights into Long Non-Coding RNA MALAT1 in Cancer and Metastasis. Cancers 2019, 11, 216. [Google Scholar] [CrossRef]
- Bhan, A.; Soleimani, M.; Mandal, S.S. Long noncoding RNA and cancer: A new paradigm. Cancer Res. 2017, 77, 3965–3981. [Google Scholar] [CrossRef]
- Carvajal, R.D.; Schwartz, G.K.; Tezel, T.; Marr, B.; Francis, J.H.; Nathan, P.D. Metastatic disease from uveal melanoma: Treatment options and future prospects. Br. J. Ophthalmol. 2017, 101, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Kaliki, S.; Shields, C.L. Uveal melanoma: Relatively rare but deadly cancer. Eye 2017, 31, 241–257. [Google Scholar] [CrossRef] [PubMed]
- Croce, M.; Ferrini, S.; Pfeffer, U.; Gangemi, R. Targeted therapy of uveal melanoma: Recent failures and new perspectives. Cancers 2019, 11, 846. [Google Scholar] [CrossRef]
- Louie, B.H.; Kurzrock, R. BAP1: Not just a BRCA1-associated protein. Cancer Treat. Rev. 2020, 90. [Google Scholar] [CrossRef]
- Li, Y.; Jia, R.; Ge, S. Role of epigenetics in uveal melanoma. Int. J. Biol. Sci. 2017, 13, 426–433. [Google Scholar] [CrossRef]
- Reichstein, D. New concepts in the molecular understanding of uveal melanoma. Curr. Opin. Ophthalmol. 2017, 28, 219–227. [Google Scholar] [CrossRef]
- Thornton, S.; Coupland, S.E.; Olohan, L.; Sibbring, J.S.; Kenny, J.G.; Hertz-Fowler, C.; Liu, X.; Haldenby, S.; Heimann, H.; Hussain, R.; et al. Targeted Next-Generation Sequencing of 117 Routine Clinical Samples Provides Further Insights into the Molecular Landscape of Uveal Melanoma. Cancers 2020, 12, 1039. [Google Scholar] [CrossRef] [PubMed]
- Bande, M.; Fernandez-Diaz, D.; Fernandez-Marta, B.; Rodriguez-Vidal, C.; Lago-Baameiro, N.; Silva-Rodríguez, P.; Paniagua, L.; Blanco-Teijeiro, M.J.; Pardo, M.; Piñeiro, A. The Role of Non-Coding RNAs in Uveal Melanoma. Cancers 2020, 12, 944. [Google Scholar] [CrossRef]
- Violanti, S.S.; Bononi, I.; Gallenga, C.E.; Martini, F.; Tognon, M.; Perri, P. New insights into molecular oncogenesis and therapy of uveal melanoma. Cancers 2019, 11, 694. [Google Scholar] [CrossRef] [PubMed]
- Rantala, E.S.; Hernberg, M.; Kivelä, T.T. Overall survival after treatment for metastatic uveal melanoma: A systematic review and meta-analysis. Melanoma Res. 2019, 29, 561–568. [Google Scholar] [CrossRef]
- Souto, E.B.; Zielinska, A.; Luis, M.; Carbone, C.; Martins-Gomes, C.; Souto, S.B.; Silva, A.M. Uveal melanoma: Physiopathology and new in situ-specific therapies. Cancer Chemother. Pharmacol. 2019, 84, 15–32. [Google Scholar] [CrossRef]
- Buder, K.; Gesierich, A.; Gelbrich, G.; Goebeler, M. Systemic treatment of metastatic uveal melanoma: Review of literature and future perspectives. Cancer Med. 2013, 2, 674–686. [Google Scholar] [CrossRef]
- Jager, M.J.; Shields, C.L.; Cebulla, C.M.; Abdel-Rahman, M.H.; Grossniklaus, H.E.; Stern, M.H.; Carvajal, R.D.; Belfort, R.N.; Jia, R.; Shields, J.A.; et al. Uveal melanoma. Nat. Rev. Dis. Prim. 2020, 6. [Google Scholar] [CrossRef]
- Zhang, L.; Dong, Y.; Wang, Y.; Gao, J.; Lv, J.; Sun, J.; Li, M.; Wang, M.; Zhao, Z.; Wang, J.; et al. Long non-coding RNAs in ocular diseases: New and potential therapeutic targets. FEBS J. 2019, 286, 2261–2272. [Google Scholar] [CrossRef]
- Pan, H.; Ni, H.; Zhang, L.L.; Xing, Y.; Fan, J.; Li, P.; Li, T.; Jia, R.; Ge, S.; Zhang, H.; et al. P2RX7-V3 is a novel oncogene that promotes tumorigenesis in uveal melanoma. Tumor Biol. 2016, 37, 13533–13543. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Sun, P.; Zhou, Q.Y.; Gao, X.; Han, Q. Long noncoding RNA MALAT1 promotes uveal melanoma cell growth and invasion by silencing of miR-140. Am. J. Transl. Res. 2016, 8, 3939–3946. [Google Scholar]
- Wu, S.; Chen, H.; Zuo, L.; Jiang, H.; Yan, H. Suppression of long noncoding RNA MALAT1 inhibits the development of uveal melanoma via microRNA-608-mediated inhibition of HOXC4. Am. J. Physiol. Physiol. 2020, 318, C903–C912. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.M.; Shi, S.S.; Jian, T.M.; Tang, D.R.; Wu, T.; Sun, F.Y. LncRNA PVT1 knockdown affects proliferation and apoptosis of uveal melanoma cells by inhibiting EZH2. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 2880–2887. [Google Scholar] [CrossRef]
- Wu, S.; Chen, H.; Han, N.; Zhang, C.; Yan, H. Long noncoding RNA PVT1 silencing prevents the development of uveal melanoma by impairing microRNA-17-3p–dependent MDM2 upregulation. Investig. Ophthalmol. Vis. Sci. 2019, 60, 4904–4914. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Yu, X.; Zhang, L.; Ding, X.; Pan, H.; Wen, X.; Xu, S.; Xing, Y.; Fan, J.; Ge, S.; et al. The long non-coding RNA RHPN1-AS1 promotes uveal melanoma progression. Int. J. Mol. Sci. 2017, 18, 226. [Google Scholar] [CrossRef]
- Lu, Q.; Zhao, N.; Zha, G.; Wang, H.; Tong, Q.; Xin, S. LncRNA HOXA11-AS Exerts Oncogenic Functions by Repressing p21 and miR-124 in Uveal Melanoma. DNA Cell Biol. 2017, 36, 837–844. [Google Scholar] [CrossRef] [PubMed]
- Zheng, X.; Tang, H.; Zhao, X.; Sun, Y.; Jiang, Y.; Liu, Y. Long non-coding RNA FTH1P3 facilitates uveal melanoma cell growth and invasion through miR-224-5p. PLoS ONE 2017, 12, e0184746. [Google Scholar] [CrossRef] [PubMed]
- Barbagallo, C.; Caltabiano, R.; Broggi, G.; Russo, A.; Puzzo, L.; Avitabile, T.; Longo, A.; Reibaldi, M.; Barbagallo, D.; Di Pietro, C.; et al. LncRNA LINC00518 Acts as an Oncogene in Uveal Melanoma by Regulating an RNA-Based Network. Cancers 2020, 12, 3867. [Google Scholar] [CrossRef]
- Qi, Y.; Yao, R.; Zhang, W.; Cui, Q.; Zhang, F. Knockdown of Long Non-Coding RNA LOC100132707 Inhibits the Migration of Uveal Melanoma Cells via Silencing JAK2. Onco. Targets. Ther. 2020, 13, 12955–12964. [Google Scholar] [CrossRef] [PubMed]
- Ding, X.; Wang, X.; Lin, M.; Xing, Y.; Ge, S.; Jia, R.; Zhang, H.; Fan, X.; Li, J. PAUPAR lncRNA suppresses tumourigenesis by H3K4 demethylation in uveal melanoma. FEBS Lett. 2016, 1729–1738. [Google Scholar] [CrossRef]
- Cheng, G.; He, J.; Zhang, L.; Ge, S.; Zhang, H.; Fan, X. HIC1 modulates uveal melanoma progression by activating lncRNA-numb. Tumor Biol. 2016, 37, 12779–12789. [Google Scholar] [CrossRef] [PubMed]
- Xing, Y.; Wen, X.; Ding, X.; Fan, J.; Chai, P.; Jia, R.; Ge, S.; Qian, G.; Zhang, H.; Fan, X. CANT1 lncRNA Triggers Efficient Therapeutic Efficacy by Correcting Aberrant lncing Cascade in Malignant Uveal Melanoma. Mol. Ther. 2017, 25, 1209–1221. [Google Scholar] [CrossRef]
- Li, P.; He, J.; Yang, Z.; Ge, S.; Zhang, H.; Zhong, Q.; Fan, X. ZNNT1 long noncoding RNA induces autophagy to inhibit tumorigenesis of uveal melanoma by regulating key autophagy gene expression. Autophagy 2019, 3, 1–14. [Google Scholar] [CrossRef]
- Wu, X.; Yuan, Y.; Ma, R.; Xu, B.; Zhang, R. LncRNA SNHG7 affects malignant tumor behaviors through downregulation of EZH2 in uveal melanoma cell lines. Oncol. Lett. 2020, 19, 1505–1515. [Google Scholar] [CrossRef]
- Qi, Y.; Cui, Q.; Zhang, W.; Yao, R.; Xu, D.; Zhang, F. Long Non-Coding RNA GAS5 Targeting microRNA-21 to Suppress the Invasion and Epithelial–Mesenchymal Transition of Uveal Melanoma. Cancer Manag. Res. 2020, 12, 12259. [Google Scholar] [CrossRef] [PubMed]
- Mishra, S.; Maurya, S.K.; Srivastava, K.; Shukla, S.; Mishra, R. Pax6 influences expression patterns of genes involved in neuro-degeneration. Ann. Neurosci. 2015, 22, 226–231. [Google Scholar] [CrossRef]
- Singer, R.A.; Arnes, L.; Cui, Y.; Wang, J.; Gao, Y.; Guney, M.A.; Burnum-Johnson, K.E.; Rabadan, R.; Ansong, C.; Orr, G.; et al. The Long Noncoding RNA Paupar Modulates PAX6 Regulatory Activities to Promote Alpha Cell Development and Function. Cell Metab. 2019, 30, 1091–1106.e8. [Google Scholar] [CrossRef]
- Vance, K.W.; Sansom, S.N.; Lee, S.; Chalei, V.; Kong, L.; Cooper, S.E.; Oliver, P.L.; Ponting, C.P. The long non-coding RNA paupar regulates the expression of both local and distal genes. EMBO J. 2014, 33, 296–311. [Google Scholar] [CrossRef]
- Liu, H.; Lei, C.; Long, K.; Yang, X.; Zhu, Z.; Zhang, L.; Liu, J. Mutant GNAQ promotes cell viability and migration of uveal melanoma cells through the activation of Notch signaling. Oncol. Rep. 2015, 34, 295–301. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rood, B.R.; Leprince, D. Deciphering HIC1 control pathways to reveal new avenues in cancer therapeutics. Expert Opin. Ther. Targets 2013, 17, 811–827. [Google Scholar] [CrossRef] [PubMed]
- Lessard, L.; Liu, M.; Marzese, D.M.; Wang, H.; Chong, K.; Kawas, N.; Donovan, N.C.; Kiyohara, E.; Hsu, S.; Nelson, N.; et al. The CASC15 Long Intergenic Noncoding RNA Locus Is Involved in Melanoma Progression and Phenotype Switching. J. Invest. Dermatol. 2015, 135, 2464–2474. [Google Scholar] [CrossRef] [PubMed]
- McHugh, C.A.; Chen, C.K.; Chow, A.; Surka, C.F.; Tran, C.; McDonel, P.; Pandya-Jones, A.; Blanco, M.; Burghard, C.; Moradian, A.; et al. The Xist lncRNA interacts directly with SHARP to silence transcription through HDAC3. Nature 2015, 521, 232–236. [Google Scholar] [CrossRef] [PubMed]
- Luo, X.; Song, Y.; Tang, L.; Sun, D.-H.; Ji, D.-G. LncRNA SNHG7 promotes development of breast cancer by regulating microRNA-186. Eur. Rev. Med. Pharmacol. Sci. 2018, 22, 7788–7797. [Google Scholar] [CrossRef]
- Zhong, X.; Long, Z.; Wu, S.; Xiao, M.; Hu, W. LncRNA-SNHG7 regulates proliferation, apoptosis and invasion of bladder cancer cells. J. BU ON. 2018, 23, 776–781. [Google Scholar]
- Wang, M.W.; Liu, J.; Liu, Q.; Xu, Q.H.; Li, T.F.; Jin, S.; Xia, T.S. LncRNA SNHG7 promotes the proliferation and inhibits apoptosis of gastric cancer cells by repressing the P15 and P16 expression. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 4613–4622. [Google Scholar]
- Li, Y.; Zeng, C.; Hu, J.; Pan, Y.; Shan, Y.; Liu, B.; Jia, L. Long non-coding RNA-SNHG7 acts as a target of miR-34a to increase GALNT7 level and regulate PI3K/Akt/mTOR pathway in colorectal cancer progression. J. Hematol. Oncol. 2018, 11, 89. [Google Scholar] [CrossRef]
- Cheng, D.; Fan, J.; Ma, Y.; Zhou, Y.; Qin, K.; Shi, M.; Yang, J. LncRNA SNHG7 promotes pancreatic cancer proliferation through ID4 by sponging miR-342-3p. Cell Biosci. 2019, 9, 1–11. [Google Scholar] [CrossRef]
- Pei, Y.; He, Y.; Hu, L.; Zhou, B.; Xu, H.; Liu, X. The cross-talk between lncRNA-SNHG7/microRNA-181/cbx7 modulates malignant character in lung adenocarcinoma. Am. J. Pathol. 2020. [Google Scholar] [CrossRef]
- Tang, R.; Jiang, M.; Liang, L.; Xiong, D.; Dang, Y.; Chen, G. Long noncoding RNA MALAT-1 can predict poor prognosis: A meta-analysis. Med. Sci. Monit. 2016, 22, 302–309. [Google Scholar] [CrossRef]
- Tripathi, V.; Shen, Z.; Chakraborty, A.; Giri, S.; Freier, S.M.; Wu, X.; Zhang, Y.; Gorospe, M.; Prasanth, S.G.; Lal, A.; et al. Long Noncoding RNA MALAT1 Controls Cell Cycle Progression by Regulating the Expression of Oncogenic Transcription Factor B-MYB. PLoS Genet. 2013, 9. [Google Scholar] [CrossRef] [PubMed]
- Xu, H.; Gong, J.; Liu, H. High expression of lncRNA PVT1 independently predicts poor overall survival in patients with primary uveal melanoma. PLoS ONE 2017, 12, e0189675. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.; Liu, H.W.; Chen, J.Q.; Wang, S.H.; Hao, L.Q.; Liu, M.; Wang, B. The long noncoding RNA PVT1 functions as a competing endogenous RNA by sponging miR-186 in gastric cancer. Biomed. Pharmacother. 2017, 88, 302–308. [Google Scholar] [CrossRef]
- Wang, B.J.; Ding, H.W.; Ma, G.A. Long noncoding RNA PVT1 promotes melanoma progression via endogenous sponging miR-26b. Oncol. Res. 2018, 26, 675–681. [Google Scholar] [CrossRef]
- Zhao, L.; Kong, H.; Sun, H.; Chen, Z.; Chen, B.; Zhou, M. LncRNA-PVT1 promotes pancreatic cancer cells proliferation and migration through acting as a molecular sponge to regulate miR-448. J. Cell. Physiol. 2018, 233, 4044–4055. [Google Scholar] [CrossRef]
- Li, P.D.; Hu, J.L.; Ma, C.; Ma, H.; Yao, J.; Chen, L.L.; Chen, J.; Cheng, T.T.; Yang, K.Y.; Wu, G.; et al. Upregulation of the long non-coding RNA PVT1 promotes esophageal squamous cell carcinoma progression by acting as a molecular sponge of miR-203 and LASP1. Oncotarget 2017, 8, 34164–34176. [Google Scholar] [CrossRef] [PubMed]
- Duan, H.; Li, X.; Chen, Y.; Wang, Y.; Li, Z. LncRNA RHPN1-AS1 promoted cell proliferation, invasion and migration in cervical cancer via the modulation of miR-299–3p/FGF2 axis. Life Sci. 2019, 239, 116856. [Google Scholar] [CrossRef] [PubMed]
- Cui, P.; Su, J.; Li, Q.; Xu, G.; Zhu, N. LncRNA RHPN1-AS1 targeting miR-625/REG3A promotes cell proliferation and invasion of glioma cells. Onco. Targets. Ther. 2019, 12, 7911–7921. [Google Scholar] [CrossRef] [PubMed]
- Liang, D.; Liu, H.; Yang, Q.; He, Y.; Yan, Y.; Li, N.; You, W. Long noncoding RNA RHPN1-AS1, induced by KDM5B, is involved in breast cancer via sponging miR-6884-5p. J. Cell. Biochem. 2020, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.; Yan, T.; Chai, Y.; Jiang, Y.; Zhu, X. Long noncoding RNA HOTTIP as an independent prognostic marker in cancer. Clin. Chim. Acta 2018, 482, 224–230. [Google Scholar] [CrossRef]
- Li, L.; Wang, Y.; Song, G.; Zhang, X.; Gao, S.; Liu, H. HOX cluster-embedded antisense long non-coding RNAs in lung cancer. Cancer Lett. 2019, 450, 14–21. [Google Scholar] [CrossRef]
- Lu , C.-W.; Zhou, D.-D.; Xie, T.; Hao, G.L.; Pant, O.P.; Lu, C.B.; Liu, X.F. HOXA11 antisense long noncoding RNA (HOXA11-AS): A promising lncRNA in human cancers. Cancer Med. 2018, 7, 3792–3799. [Google Scholar] [CrossRef] [PubMed]
- Su, J.C.; Hu, X.F. Long non-coding RNA HOXA11-AS promotes cell proliferation and metastasis in human breast cancer. Mol. Med. Rep. 2017, 16, 4887–4894. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Li, Z.; Wang, Y. Long non-coding RNA FTH1P3 promotes the metastasis and aggressiveness of non-small cell lung carcinoma by inducing epithelial-mesenchymal transition. Int. J. Clin. Exp. Pathol. 2019, 12, 3782–3790. [Google Scholar] [PubMed]
- Lv, R.; Zhang, Q.W. The long noncoding RNA FTH1P3 promotes the proliferation and metastasis of cervical cancer through microRNA-145. Oncol. Rep. 2020, 43, 31–40. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Ying, L.I.; Wang, J.; Lei, P. Long non-coding RNA ferritin heavy polypeptide 1 pseudogene 3 controls glioma cell proliferation and apoptosis via regulation of the microRNA-224-5p/tumor protein D52 axis. Mol. Med. Rep. 2018, 18, 4239–4246. [Google Scholar] [CrossRef]
- Yang, L.; Sun, K.; Chu, J.; Qu, Y.; Zhao, X.; Yin, H.; Ming, L.; Wan, J.; He, F. Long non-coding RNA FTH1P3 regulated metastasis and invasion of esophageal squamous cell carcinoma through SP1/NF-kB pathway. Biomed. Pharmacother. 2018, 106, 1570–1577. [Google Scholar] [CrossRef]
- Wang, R.; Zhang, T.; Yang, Z.; Jiang, C.; Seng, J. Long non-coding RNA FTH1P3 activates paclitaxel resistance in breast cancer through miR-206/ABCB1. J. Cell. Mol. Med. 2018, 22, 4068–4075. [Google Scholar] [CrossRef]
- Young, C.N.J.; Górecki, D.C. P2RX7 purinoceptor as a therapeutic target-The second coming? Front. Chem. 2018, 6. [Google Scholar] [CrossRef]
- Lavorgna, G.; Vago, R.; Sarmini, M.; Montorsi, F.; Salonia, A.; Bellone, M. Long non-coding RNAs as novel therapeutic targets in cancer. Pharmacol. Res. 2016, 110, 131–138. [Google Scholar] [CrossRef] [PubMed]
- Ling, H.; Fabbri, M.; Calin, G.A. MicroRNAs and other non-coding RNAs as targets for anticancer drug development. Nat. Rev. Drug Discov. 2013, 12, 847–865. [Google Scholar] [CrossRef]
- Wahlestedt, C. Targeting long non-coding RNA to therapeutically upregulate gene expression. Nat. Rev. Drug Discov. 2013, 12, 433–446. [Google Scholar] [CrossRef] [PubMed]
- Mercer, T.R.; Mattick, J.S. Structure and function of long noncoding RNAs in epigenetic regulation. Nat. Struct. Mol. Biol. 2013, 20, 300–307. [Google Scholar] [CrossRef] [PubMed]
- Bregoli, L.; Movia, D.; Gavigan-Imedio, J.D.; Lysaght, J.; Reynolds, J.; Prina-Mello, A. Nanomedicine applied to translational oncology: A future perspective on cancer treatment. Nanomed. Nanotechnol. Biol. Med. 2016, 12, 81–103. [Google Scholar] [CrossRef] [PubMed]
- Gou, Y.; Miao, D.; Zhou, M.; Wang, L.; Zhou, H.; Su, G. Bio-Inspired Protein-Based Nanoformulations for Cancer Theranostics. Front. Pharmacol. 2018, 9, 421–462. [Google Scholar] [CrossRef] [PubMed]
- Mishra, D.K.; Shandilya, R.; Mishra, P.K. Lipid based nanocarriers: A translational perspective. Nanomed. Nanotechnol. Biol. Med. 2018, 14, 2023–2050. [Google Scholar] [CrossRef]
- Weidle, U.H.; Birzele, F.; Kollmorgen, G.; Rüger, R. Long non-coding RNAs and their role in metastasis. Cancer Genom. Proteom. 2017, 14, 143–160. [Google Scholar] [CrossRef]
- Winkle, M.; El-Daly, S.M.; Fabbri, M.; Calin, G.A. Noncoding RNA therapeutics — challenges and potential solutions. Nat. Rev. Drug Discov. 2021, 20, 629–651. [Google Scholar] [CrossRef]
- Mahajan, S.; Patharkar, A.; Kuche, K.; Maheshwari, R.; Deb, P.K.; Kalia, K.; Tekade, R.K. Functionalized carbon nanotubes as emerging delivery system for the treatment of cancer. Int. J. Pharm. 2018, 548, 540–558. [Google Scholar] [CrossRef]
- Singh, P.; Pandit, S.; Mokkapati, V.R.S.S.; Garg, A.; Ravikumar, V.; Mijakovic, I. Gold Nanoparticles in Diagnostics and Therapeutics for Human Cancer. Int. J. Mol. Sci. 2018, 19, 1979. [Google Scholar] [CrossRef]
- Núñez, C.; Estévez, S.V.; del Pilar Chantada, M. Inorganic nanoparticles in diagnosis and treatment of breast cancer. JBIC J. Biol. Inorg. Chem. 2018, 23, 331–345. [Google Scholar] [CrossRef]
- Manzano, M.; Vallet-Regí, M. Mesoporous silica nanoparticles in nanomedicine applications. J. Mater. Sci. Mater. Med. 2018, 29, 65. [Google Scholar] [CrossRef]
- Pattni, B.S.; Chupin, V.V.; Torchilin, V.P. New Developments in Liposomal Drug Delivery. Chem. Rev. 2015, 115, 10938–10966. [Google Scholar] [CrossRef]
- Steinmetz, N.F. Viral Nanoparticles in Drug Delivery and Imaging. Mol. Pharm. 2013, 10, 1–2. [Google Scholar] [CrossRef]
- Ren, Y.; Li, R.Q.; Cai, Y.R.; Xia, T.; Yang, M.; Xu, F.J. Effective Codelivery of lncRNA and pDNA by Pullulan-Based Nanovectors for Promising Therapy of Hepatocellular Carcinoma. Adv. Funct. Mater. 2016, 26, 7314–7325. [Google Scholar] [CrossRef]
- Gong, N.; Teng, X.; Li, J.; Liang, X.J. Antisense Oligonucleotide-Conjugated Nanostructure-Targeting lncRNA MALAT1 Inhibits Cancer Metastasis. ACS Appl. Mater. Interfaces 2019, 11, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Vaidya, A.M.; Sun, Z.; Ayat, N.; Schilb, A.; Liu, X.; Jiang, H.; Sun, D.; Scheidt, J.; Qian, V.; He, S.; et al. Systemic Delivery of Tumor-Targeting siRNA Nanoparticles against an Oncogenic LncRNA Facilitates Effective Triple-Negative Breast Cancer Therapy. Bioconjug. Chem. 2019, 30, 907–919. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Li, Z.; Chen, X.; Zhang, S. Long non-coding RNAs: From disease code to drug role. Acta Pharm. Sin. B 2021, 11, 340–354. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, S.; Yang, F.; Meng, Z.; Liu, Y. LncRNA ROR1-AS1 high expression and its prognostic significance in liver cancer. Oncol. Rep. 2020, 43, 55–74. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Huo, X.; Yang, X.-R.; He, J.; Cheng, L.; Wang, N.; Deng, X.; Jin, H.; Wang, N.; Wang, C.; et al. STAT3-mediated upregulation of lncRNA HOXD-AS1 as a ceRNA facilitates liver cancer metastasis by regulating SOX4. Mol. Cancer 2017, 16, 136. [Google Scholar] [CrossRef] [PubMed]
- Chandra Gupta, S.; Nandan Tripathi, Y. Potential of long non-coding RNAs in cancer patients: From biomarkers to therapeutic targets. Int. J. Cancer 2017, 140, 1955–1967. [Google Scholar] [CrossRef] [PubMed]
- Jiang, N.; Pan, J.; Fang, S.; Zhou, C.; Han, Y.; Chen, J.; Meng, X.; Jin, X.; Gong, Z. Liquid biopsy: Circulating exosomal long noncoding RNAs in cancer. Clin. Chim. Acta 2019, 495, 331–337. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez, M.F.B.; Marta, B.F.; Baameiro, N.L.; Santiago-Varela, M.; Silva-Rodríguez, P.; Blanco-Teijeiro, M.J.; Perez, M.P.; Ces, A.P.; Bande Rodríguez, M.F.; Fernandez Marta, B.; et al. Blood Biomarkers of Uveal Melanoma: Current Perspectives. Clin. Ophthalmol. 2020, 14, 157–169. [Google Scholar] [CrossRef] [PubMed]
- Barak, V.; Frenkel, S.; Kalickman, I.; Maniotis, A.J.; Folberg, R.; Pe’er, J. Serum markers to detect metastatic uveal melanoma. Anticancer Res. 2007, 27, 1897–1900. [Google Scholar]
- Anfossi, S.; Babayan, A.; Pantel, K.; Calin, G.A. Clinical utility of circulating non-coding RNAs — an update. Nat. Rev. Clin. Oncol. 2018, 15, 541–563. [Google Scholar] [CrossRef]
- Wang, M.; Gu, H.; Qian, H.; Zhu, W.; Zhao, C.; Zhang, X.; Tao, Y.; Zhang, L.; Xu, W. MiR-17-5p/20a are important markers for gastric cancer and murine double minute 2 participates in their functional regulation. Eur. J. Cancer 2013, 49, 2010–2021. [Google Scholar] [CrossRef]
- HOXC4 Homeobox C4 [Homo sapiens (Human)]-Gene-NCBI. Available online: https://www.ncbi.nlm.nih.gov/gene/3221 (accessed on 26 October 2020).
- AKT2 AKT Serine/Threonine Kinase 2 [Homo sapiens (Human)]-Gene-NCBI. Available online: https://www.ncbi.nlm.nih.gov/gene/208 (accessed on 26 October 2020).
- Tinzl, M.; Marberger, M.; Horvath, S.; Chypre, C. DD3PCA3 RNA analysis in urine - A new perspective for detecting prostate cancer. Eur. Urol. 2004, 46, 182–187. [Google Scholar] [CrossRef]
- Barbagallo, C.; Di Maria, A.; Alecci, A.; Barbagallo, D.; Alaimo, S.; Colarossi, L.; Ferro, A.; Di Pietro, C.; Purrello, M.; Pulvirenti, A.; et al. VECTOR: An Integrated Correlation Network Database for the Identification of CeRNA Axes in Uveal Melanoma. Genes 2021, 12, 1004. [Google Scholar] [CrossRef]
- Shohet, J.; Kim, E. Targeted molecular therapy for neuroblastoma: The ARF/MDM2/p53 Axis. J. Natl. Cancer Inst. 2009, 101, 1527–1529. [Google Scholar] [CrossRef]
- Amodio, N.; Raimondi, L.; Juli, G.; Stamato, M.A.; Caracciolo, D.; Tagliaferri, P.; Tassone, P. MALAT1: A druggable long non-coding RNA for targeted anti-cancer approaches. J. Hematol. Oncol. 2018, 11, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Duan, R.; Du, W.; Guo, W. EZH2: A novel target for cancer treatment. J. Hematol. Oncol. 2020, 13. [Google Scholar] [CrossRef] [PubMed]
- Peixoto, P.; Grandvallet, C.; Feugeas, J.P.; Guittaut, M.; Hervouet, E. Epigenetic Control of Autophagy in Cancer Cells: A Key Process for Cancer-Related Phenotypes. Cells 2019, 8, 1656. [Google Scholar] [CrossRef] [PubMed]
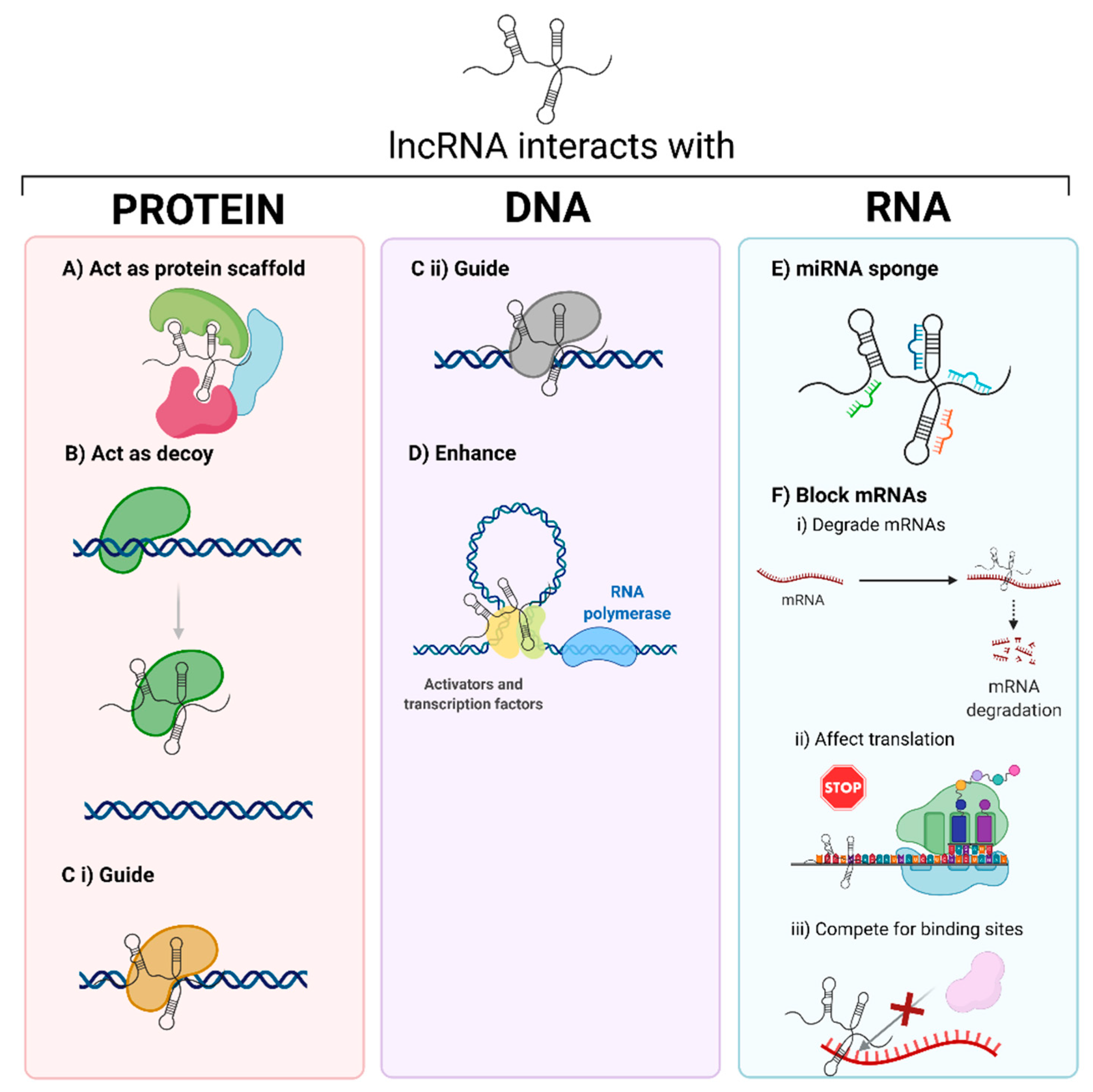
| Biological Process | LncRNA | Citation |
|---|---|---|
| Transcription | lncRNA NRON, lncRNA HSR1 | [10,11] |
| Splicing | lncRNA MALAT, lncRNA ASCO | [12,13] |
| Translation | lncRNA HULC | [14] |
| RNA localization | lncRNA XIST | [15] |
| RNA decay | lncRNA gadd7 | [16] |
| RNA editing | lncRNA CTN | [17] |
| Epigenetic remodeling | lncRNA HOTAIR | [18] |
| Genome integrity | lncRNA NORAD, lncRNA CONCR | [19,20] |
| Structural functions | lncRNA NEAT1, lncRNA FIRRE | [21] |
| Cellular organelle functions | lncRNA RMRP, lncRNA SAMMSON | [22,23] |
| LncRNA Name | Mechanism | Role | Reference |
|---|---|---|---|
| lncRNA CASC15 | Switches tumor phenotype through MITF and/or SOX10 | Oncogenic | [56] |
| lncRNA P2RX7-V3 | Affects PI3K/AKT pathway | Oncogenic | [63] |
| lncRNA MALAT1 | Suppresses miR-608. Promotes miR-140 expression and suppresses Slug and ADAM10 expression. | Oncogenic | [64,65] |
| lncRNA PVT1 | Regulates the expression of EZH2 and blocks miR-17-3p | Oncogenic | [66,67] |
| lncRNA RHPN1-AS1 | Participates in TGF-β pathway | Oncogenic | [68] |
| lncRNA HOXA11-AS | Suppresses p21 and acts as a sponge of miRNA -124 | Oncogenic | [69] |
| lncRNA FTH1P3 | Suppresses miR-NA 224-5p expression and promotes the expression of Rac1 and Fizzled 5 | Oncogenic | [70] |
| lncRNA LINC00518 | Participates in the metastatic process | Oncogenic | [71] |
| lncRNA LOC100132707 | Promotes migration via JAK2 | Oncogenic | [72] |
| lncRNA PAUPAR | Modulates HES1 expression | Tumor suppressor | [73] |
| lncRNA NUMB | Restores the expression of HIC1 (Hypermethylated in cancer 1) | Tumor suppressor | [74] |
| lncRNA CANT1 | Modulates JPX or FTX by methylation at their promoters | Tumor suppressor | [75] |
| lncRNA ZNNT1 | Promotes autophagy | Tumor suppressor | [76] |
| lncRNA SNHG7 | Regulates EZH2 pathway | Tumor suppressor | [77] |
| lncRNA GAS5 | Induces PTEN expression | Tumor suppressor | [78] |
| LncRNA | Expression Level | Source | Association | Reference |
|---|---|---|---|---|
| lncRNA PVT1 | Up | Tumor, gastric juice, serum | Poor overall survival | [94] |
| lncRNA HOXA11AS | Up | Tumor | Poor overall survival | [104] |
| lncRNA SNHG7 | Down | Tumor | Higher tumor-node-metastasis stage (TNM) and poor histological type | [77,79,97] |
| lncRNA MALAT1 | Up | Tumor, urine, serum | Melanoma progression and metastasis | [133] |
| lncRNA CASC15 | Up | Tumor | Cancer recurrence | [133] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Milán-Rois, P.; Quan, A.; Slack, F.J.; Somoza, Á. The Role of LncRNAs in Uveal Melanoma. Cancers 2021, 13, 4041. https://doi.org/10.3390/cancers13164041
Milán-Rois P, Quan A, Slack FJ, Somoza Á. The Role of LncRNAs in Uveal Melanoma. Cancers. 2021; 13(16):4041. https://doi.org/10.3390/cancers13164041
Chicago/Turabian StyleMilán-Rois, Paula, Anan Quan, Frank J. Slack, and Álvaro Somoza. 2021. "The Role of LncRNAs in Uveal Melanoma" Cancers 13, no. 16: 4041. https://doi.org/10.3390/cancers13164041
APA StyleMilán-Rois, P., Quan, A., Slack, F. J., & Somoza, Á. (2021). The Role of LncRNAs in Uveal Melanoma. Cancers, 13(16), 4041. https://doi.org/10.3390/cancers13164041