Racial Differences in Hepatocellular Carcinoma Incidence and Risk Factors among a Low Socioeconomic Population
Abstract
:Simple Summary
Abstract
1. Introduction
2. Materials and Methods
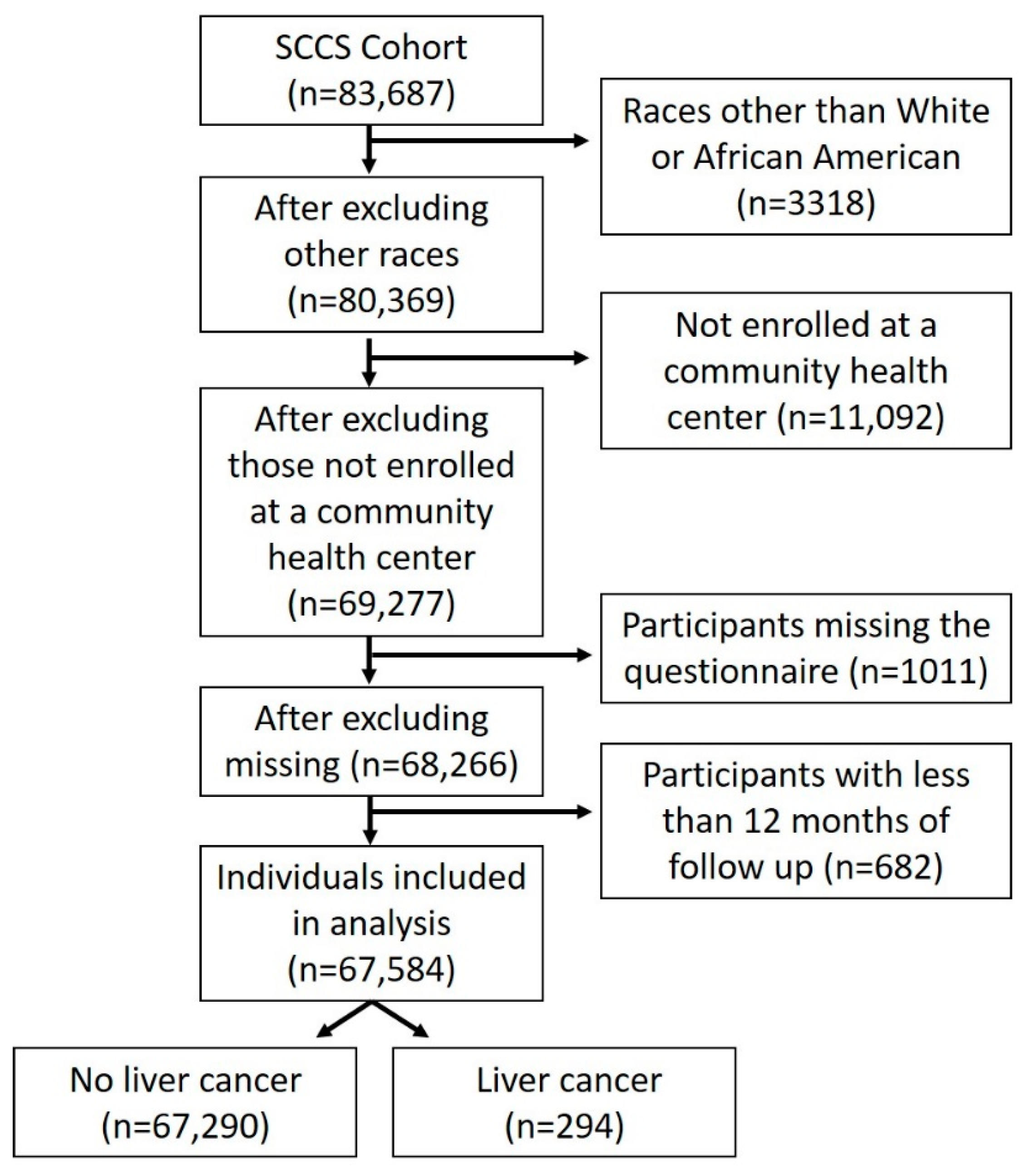
2.1. Study Population
2.2. Hepatocellular Carcinoma (HCC) Incidence
2.3. Population for Analysis
2.4. Statistical Analysis
3. Results
3.1. Study Population
3.2. Incidence Rates
3.3. Cox Regression Model
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Momin, B.R.; Pinheiro, P.S.; Carreira, H.; Li, C.; Weir, H.K. Liver cancer survival in the United States by race and stage (2001–2009): Findings from the CONCORD-2 study. Cancer 2017, 123 (Suppl. S24), 5059–5078. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Torre, L.A.; Siegel, R.L.; Ward, E.M.; Jemal, A. Global Cancer Incidence and Mortality Rates and Trends--An Update. Cancer Epidemiol. Biomark. Prev. 2016, 25, 16–27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rahib, L.; Smith, B.D.; Aizenberg, R.; Rosenzweig, A.B.; Fleshman, J.M.; Matrisian, L.M. Projecting cancer incidence and deaths to 2030: The unexpected burden of thyroid, liver, and pancreas cancers in the United States. Cancer Res. 2014, 74, 2913–2921. [Google Scholar] [CrossRef] [Green Version]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2019. CA Cancer J. Clin. 2019, 69, 7–34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smith, B.D.; Morgan, R.L.; Beckett, G.A.; Falck-Ytter, Y.; Holtzman, D.; Teo, C.G.; Jewett, A.; Baack, B.; Rein, D.B.; Patel, N.; et al. Recommendations for the identification of chronic hepatitis C virus infection among persons born during 1945–1965. Morb. Mortal. Wkly. Rep. Recomm. Rep. 2012, 61, 1–32. [Google Scholar]
- Denniston, M.M.; Jiles, R.B.; Drobeniuc, J.; Klevens, R.M.; Ward, J.W.; McQuillan, G.M.; Holmberg, S.D. Chronic hepatitis C virus infection in the United States, National Health and Nutrition Examination Survey 2003 to 2010. Ann. Intern. Med. 2014, 160, 293–300. [Google Scholar] [CrossRef] [Green Version]
- Islami, F.; Miller, K.D.; Siegel, R.L.; Fedewa, S.A.; Ward, E.M.; Jemal, A. Disparities in liver cancer occurrence in the United States by race/ethnicity and state. CA Cancer J. Clin. 2017, 67, 273–289. [Google Scholar] [CrossRef] [Green Version]
- Kanwal, F.; Kramer, J.R.; Mapakshi, S.; Natarajan, Y.; Chayanupatkul, M.; Richardson, P.A.; Li, L.; Desiderio, R.; Thrift, A.P.; Asch, S.M.; et al. Risk of Hepatocellular Cancer in Patients With Non-Alcoholic Fatty Liver Disease. Gastroenterology 2018, 155, 1828–1837.e1822. [Google Scholar] [CrossRef] [Green Version]
- Ha, J.; Yan, M.; Aguilar, M.; Bhuket, T.; Tana, M.M.; Liu, B.; Gish, R.G.; Wong, R.J. Race/ethnicity-specific disparities in cancer incidence, burden of disease, and overall survival among patients with hepatocellular carcinoma in the United States. Cancer 2016, 122, 2512–2523. [Google Scholar] [CrossRef]
- Van Thiel, D.H.; Ramadori, G. Non-viral causes of hepatocellular carcinoma. J. Gastrointest. Cancer 2011, 42, 191–194. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Wu, F. Global burden of aflatoxin-induced hepatocellular carcinoma: A risk assessment. Environ. Health Perspect. 2010, 118, 818–824. [Google Scholar] [CrossRef] [Green Version]
- Mittal, S.; El-Serag, H.B. Epidemiology of hepatocellular carcinoma: Consider the population. J. Clin. Gastroenterol. 2013, 47, S2–S6. [Google Scholar] [CrossRef] [Green Version]
- Sahasrabuddhe, V.V.; Gunja, M.Z.; Graubard, B.I.; Trabert, B.; Schwartz, L.M.; Park, Y.; Hollenbeck, A.R.; Freedman, N.D.; McGlynn, K.A. Nonsteroidal anti-inflammatory drug use, chronic liver disease, and hepatocellular carcinoma. J. Natl. Cancer Inst. 2012, 104, 1808–1814. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bosch, F.X.; Ribes, J.; Diaz, M.; Cleries, R. Primary liver cancer: Worldwide incidence and trends. Gastroenterology 2004, 127, S5–S16. [Google Scholar] [CrossRef]
- Signorello, L.B.; Hargreaves, M.K.; Blot, W.J. The Southern Community Cohort Study: Investigating health disparities. J. Health Care Poor Underserved 2010, 21, 26–37. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Signorello, L.B.; Hargreaves, M.K.; Steinwandel, M.D.; Zheng, W.; Cai, Q.; Schlundt, D.G.; Buchowski, M.S.; Arnold, C.W.; McLaughlin, J.K.; Blot, W.J. Southern community cohort study: Establishing a cohort to investigate health disparities. J. Natl. Med. Assoc. 2005, 97, 972–979. [Google Scholar] [PubMed]
- US Department of Health Human Services. US Department of Agriculture. 2015–2020 Dietary Guidelines for Americans; 2015. Available online: https://health.gov/sites/default/files/2019-09/2015-2020_Dietary_Guidelines.pdf (accessed on 10 November 2019).
- Naugler, W.E.; Sakurai, T.; Kim, S.; Maeda, S.; Kim, K.; Elsharkawy, A.M.; Karin, M. Gender disparity in liver cancer due to sex differences in MyD88-dependent IL-6 production. Science 2007, 317, 121–124. [Google Scholar] [CrossRef] [Green Version]
- Villa, E. Role of estrogen in liver cancer. Women’s Health 2008, 4, 41–50. [Google Scholar] [CrossRef] [Green Version]
- Baldissera, V.D.; Alves, A.F.; Almeida, S.; Porawski, M.; Giovenardi, M. Hepatocellular carcinoma and estrogen receptors: Polymorphisms and isoforms relations and implications. Med. Hypotheses 2016, 86, 67–70. [Google Scholar] [CrossRef]
- Davila, J.A.; Morgan, R.O.; Shaib, Y.; McGlynn, K.A.; El-Serag, H.B. Diabetes increases the risk of hepatocellular carcinoma in the United States: A population based case control study. Gut 2005, 54, 533–539. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.M.; Govindarajan, S.; Arakawa, K.; Yu, M.C. Synergism of alcohol, diabetes, and viral hepatitis on the risk of hepatocellular carcinoma in blacks and whites in the U.S. Cancer 2004, 101, 1009–1017. [Google Scholar] [CrossRef]
- Larsson, S.C.; Wolk, A. Overweight, obesity and risk of liver cancer: A meta-analysis of cohort studies. Br. J. Cancer 2007, 97, 1005–1008. [Google Scholar] [CrossRef] [PubMed]
- Calle, E.E.; Rodriguez, C.; Walker-Thurmond, K.; Thun, M.J. Overweight, obesity, and mortality from cancer in a prospectively studied cohort of U.S. adults. N. Engl. J. Med. 2003, 348, 1625–1638. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Setiawan, V.W.; Lim, U.; Lipworth, L.; Lu, S.C.; Shepherd, J.; Ernst, T.; Wilkens, L.R.; Henderson, B.E.; Le Marchand, L. Sex and Ethnic Differences in the Association of Obesity With Risk of Hepatocellular Carcinoma. Clin. Gastroenterol. Hepatol. 2016, 14, 309–316. [Google Scholar] [CrossRef] [Green Version]
- Ohki, T.; Tateishi, R.; Shiina, S.; Goto, E.; Sato, T.; Nakagawa, H.; Masuzaki, R.; Goto, T.; Hamamura, K.; Kanai, F.; et al. Visceral fat accumulation is an independent risk factor for hepatocellular carcinoma recurrence after curative treatment in patients with suspected NASH. Gut 2009, 58, 839–844. [Google Scholar] [CrossRef] [Green Version]
- Carroll, J.F.; Chiapa, A.L.; Rodriquez, M.; Phelps, D.R.; Cardarelli, K.M.; Vishwanatha, J.K.; Bae, S.; Cardarelli, R. Visceral fat, waist circumference, and BMI: Impact of race/ethnicity. Obesity 2008, 16, 600–607. [Google Scholar] [CrossRef] [PubMed]
- LoConte, N.K.; Brewster, A.M.; Kaur, J.S.; Merrill, J.K.; Alberg, A.J. Alcohol and Cancer: A Statement of the American Society of Clinical Oncology. J. Clin. Oncol. 2018, 36, 83–93. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bagnardi, V.; Rota, M.; Botteri, E.; Tramacere, I.; Islami, F.; Fedirko, V.; Scotti, L.; Jenab, M.; Turati, F.; Pasquali, E.; et al. Alcohol consumption and site-specific cancer risk: A comprehensive dose-response meta-analysis. Br. J. Cancer 2015, 112, 580–593. [Google Scholar] [CrossRef]
- Lee, Y.C.; Cohet, C.; Yang, Y.C.; Stayner, L.; Hashibe, M.; Straif, K. Meta-analysis of epidemiologic studies on cigarette smoking and liver cancer. Int. J. Epidemiol. 2009, 38, 1497–1511. [Google Scholar] [CrossRef]
- Hall, E.W.; Rosenberg, E.S.; Sullivan, P.S. Estimates of state-level chronic hepatitis C virus infection, stratified by race and sex, United States, 2010. BMC Infect. Dis. 2018, 18, 224. [Google Scholar] [CrossRef] [PubMed]
- Moyer, V.A. Screening for hepatitis C virus infection in adults: U.S. Preventive Services Task Force recommendation statement. Ann. Intern. Med. 2013, 159, 349–357. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Centers for Disease Control and Prevention. CDC Fact Sheet: Viral Hepatitis and Liver Cancer. Available online: https://www.cdc.gov/nchhstp/newsroom/docs/factsheets/viral-hep-liver-cancer.pdf (accessed on 7 January 2021).
| Characteristics | White Americans | African Americans |
|---|---|---|
| Number at risk | 18,678 | 48,906 |
| Number of hepatocellular carcinoma cases | 57 | 237 |
| Follow-up time (months; median (IQR)) | 110 (89–129) | 126 (100–145) |
| Age at enrollment (years; median (IQR)) | 52 (46–59) | 50 (44–56) |
| 40–49 | 41.6 | 49.8 |
| 50–59 | 33.5 | 33.7 |
| 60–69 | 18.7 | 12.6 |
| 70–79 | 6.3 | 4.1 |
| Sex, % | ||
| Male | 34.3 | 41.3 |
| Female | 65.7 | 58.7 |
| BMI category, % | ||
| Normal | 25.8 | 24.5 |
| Overweight | 28.6 | 28.8 |
| Obese | 44.7 | 45.7 |
| Missing | 0.8 | 1.1 |
| Education, % | ||
| <High School | 29.0 | 33.2 |
| High School | 35.4 | 35.2 |
| >High School | 35.6 | 31.6 |
| Missing | 0.0 | 0.0 |
| Household income, % | ||
| <USD 15,000 | 56.0 | 61.8 |
| USD 15,000–49,999 | 34.3 | 33.5 |
| >USD 50,000 | 8.6 | 3.6 |
| Missing | 1.1 | 1.1 |
| Self-reported hepatitis B virus infection, % | ||
| No | 95.7 | 97.2 |
| Yes | 2.0 | 1.5 |
| Missing | 2.4 | 1.3 |
| Self-reported hepatitis C virus infection, % | ||
| No | 92.6 | 95.6 |
| Yes | 5.0 | 3.1 |
| Missing | 2.4 | 1.3 |
| Diabetes, % | ||
| No | 79.0 | 77.8 |
| Yes | 20.7 | 22.0 |
| Missing | 0.3 | 1.3 |
| Among those with diabetes, use of prescription diabetes medication, % | ||
| No | 19.4 | 13.3 |
| Yes | 80.6 | 86.6 |
| Missing | 0.0 | 0.01 |
| Among those with diabetes, number of years with diabetes (years, median (IQR)) | 6 (2–12) | 7 (2–14) |
| Insurance coverage, % | ||
| No | 43.3 | 43.2 |
| Yes | 56.0 | 56.3 |
| Missing | 0.7 | 0.6 |
| Use of aspirin, % a | ||
| No | 85.3 | 88.2 |
| Yes | 14.3 | 11.5 |
| Missing | 0.4 | 0.4 |
| Use of over-the-counter pain-relievers, a % | ||
| No | 78.0 | 84.0 |
| Yes | 21.6 | 15.6 |
| Missing | 0.4 | 0.4 |
| Use of prescription pain-relievers, a % | ||
| No | 93.5 | 96.6 |
| Yes | 6.1 | 3.1 |
| Missing | 0.4 | 0.3 |
| Cigarette smoking status, % | ||
| Never | 30.8 | 36.4 |
| Former | 25.6 | 19.2 |
| Current | 43.4 | 44.2 |
| Missing | 0.2 | 0.2 |
| Heavy alcohol drinkers, b % | ||
| No | 87.1 | 78.6 |
| Yes | 11.7 | 20.4 |
| Missing | 1.2 | 1.0 |
| Currently employed, % | ||
| No | 66.8 | 61.9 |
| Yes | 32.5 | 37.5 |
| Missing | 0.8 | 0.7 |
| Laborer, including construction worker, c % | ||
| No | 88.9 | 87.5 |
| Yes | 10.4 | 11.8 |
| Missing | 0.8 | 0.7 |
| Dry cleaning, d % | ||
| No | 98.7 | 98.3 |
| Yes | 0.6 | 1.0 |
| Missing | 0.8 | 0.7 |
| Farming, d % | ||
| No | 93.3 | 94.8 |
| Yes | 5.9 | 4.6 |
| Missing | 0.8 | 0.6 |
| Chemical production or use, d % | ||
| No | 96.0 | 97.2 |
| Yes | 3.2 | 2.1 |
| Missing | 0.8 | 0.7 |
| No Incident Cancer | Incident HCC | Univariate | Multivariable a | |
|---|---|---|---|---|
| HR (95%CI) | HR (95%CI) | |||
| Age at enrollment b | ||||
| 40 | 2644 | 6 | 0.19 (0.10–0.38) | 0.23 (0.12–0.46) |
| 46 | 2348 | 10 | 0.72 (0.63–0.84) | 0.71 (0.61–0.82) |
| 50 (Median) | 2203 | 15 | 1.00 (Ref.) | 1.00 (Ref.) |
| 53 | 1911 | 11 | 1.04 (0.90–1.20) | 1.12 (0.97–1.31) |
| 68 | 422 | 0 | 0.86 (0.58–1.28) | 1.75 (1.14–2.68) |
| Sex | ||||
| Female | 28,624 | 67 | 1.00 (Ref.) | 1.00 (Ref.) |
| Male | 20,045 | 170 | 3.81 (2.87–5.06) | 2.05 (1.50–2.80) |
| BMI category | ||||
| Normal | 11,888 | 96 | 1.00 (Ref.) | 1.00 (Ref.) |
| Overweight | 13,994 | 85 | 0.72 (0.54–0.96) | 0.98 (0.72–1.32) |
| Obese | 22,272 | 56 | 0.30 (0.21–0.41) | 0.65 (0.45–0.95) |
| Education | ||||
| <High School | 16,136 | 91 | 1.00 (Ref.) | 1.00 (Ref.) |
| High School | 17,128 | 91 | 0.92 (0.69–1.23) | 1.06 (0.78–1.43) |
| >High School | 15,395 | 55 | 0.59 (0.42–0.83) | 0.78 (0.55–1.11) |
| Household income | ||||
| <USD 15,000 | 30,061 | 177 | 1.00 (Ref.) | 1.00 (Ref.) |
| USD 15,000–49,999 | 16,325 | 56 | 0.55 (0.40–0.74) | 0.84 (0.61–1.17) |
| >USD 50,000 | 1746 | 3 | 0.27 (0.09–0.85) | 0.53 (0.17–1.70) |
| Hepatitis B | ||||
| No | 47,335 | 223 | 1.00 (Ref.) | 1.00 (Ref.) |
| Yes | 714 | 9 | 2.78 (1.43–5.42) | 1.82 (0.93–3.56) |
| Hepatitis C | ||||
| No | 46,578 | 168 | 1.00 (Ref.) | 1.00 (Ref.) |
| Yes | 1471 | 64 | 13.07 (9.79–17.44) | 7.73 (5.71–10.47) |
| Diabetes | ||||
| No | 37,850 | 182 | 1.00 (Ref.) | 1.00 (Ref.) |
| Yes | 10,720 | 55 | 1.17 (0.87–1.59) | 1.48 (1.06–2.06) |
| Cigarette smoking status | ||||
| Never | 17,792 | 28 | 1.00 (Ref.) | 1.00 (Ref.) |
| Former | 9364 | 35 | 2.44 (1.48–4.01) | 1.45 (0.87–2.42) |
| Current | 21,424 | 174 | 5.37 (3.61–8.01) | 2.91 (1.87–4.52) |
| Heavy alcohol drinker c | ||||
| No | 38,275 | 146 | 1.00 (Ref.) | 1.00 (Ref.) |
| Yes | 9891 | 91 | 2.49 (1.92–3.24) | 1.59 (1.19–2.11) |
| Currently employed | ||||
| No | 30,084 | 182 | 1.00 (Ref.) | 1.00 (Ref.) |
| Yes | 18,264 | 55 | 0.45 (0.33–0.61) | 0.73 (0.53–0.99) |
| No Incident Cancer | Incident HCC | Univariate | Multivariable a | |
|---|---|---|---|---|
| HR (95%CI) | HR (95%CI) | |||
| Age at enrollment b | ||||
| 42 | 763 | 0 | 0.52 (0.26–1.05) | 0.50 (0.24–1.04) |
| 52 (Median) | 665 | 6 | 1.00 (Ref.) | 1.00 (Ref.) |
| 66 | 313 | 1 | 0.64 (0.32–1.30) | 1.44 (0.69–3.01) |
| Sex | ||||
| Female | 12,248 | 19 | 1.00 (Ref.) | 1.00 (Ref.) |
| Male | 6373 | 38 | 4.24 (2.44–7.35) | 2.83 (1.58–5.07) |
| BMI category | ||||
| Normal | 4806 | 21 | 1.00 (Ref.) | 1.00 (Ref.) |
| Overweight | 5336 | 12 | 0.51 (0.25–1.03) | 0.52 (0.25–1.07) |
| Obese | 8325 | 24 | 0.65 (0.36–1.16) | 0.79 (0.41–1.53) |
| Education | ||||
| < High School | 5395 | 20 | 1.00 (Ref.) | 1.00 (Ref.) |
| High School | 6593 | 16 | 0.66 (0.34–1.27) | 0.67 (0.35–1.31) |
| > High School | 6632 | 21 | 0.87 (0.47–0) | 0.82 (0.42–1.59) |
| Household income | ||||
| <USD 15,000 | 10,423 | 40 | 1.00 (Ref.) | 1.00 (Ref.) |
| USD 15,000–49,999 | 6393 | 10 | 0.39 (0.20–0.79) | 0.54 (0.26–1.12) |
| >USD 50,000 | 1601 | 7 | 1.14 (0.51–2.54) | 1.88 (0.74–4.75) |
| Hepatitis B | ||||
| No | 17,817 | 53 | 1.00 (Ref.) | 1.00 (Ref.) |
| Yes | 361 | 4 | 3.83 (1.39–10.58) | 1.66 (0.58–4.73) |
| Hepatitis C | ||||
| No | 17,275 | 28 | 1.00 (Ref.) | 1.00 (Ref.) |
| Yes | 903 | 29 | 22.41 (13.33–37.67) | 19.24 (10.58–35.00) |
| Diabetes | ||||
| No | 14,714 | 35 | 1.00 (Ref.) | 1.00 (Ref.) |
| Yes | 3849 | 22 | 2.61 (1.53–4.45) | 3.55 (1.96–6.43) |
| Cigarette smoking status | ||||
| Never | 5739 | 11 | 1.00 (Ref.) | 1.00 (Ref.) |
| Former | 4770 | 12 | 1.37 (0.60–3.10) | 0.87 (0.38–2.00) |
| Current | 8069 | 34 | 2.35 (1.19–4.65) | 1.23 (0.56–2.71) |
| Heavy alcohol drinker c | ||||
| No | 16,234 | 42 | 1.00 (Ref.) | 1.00 (Ref.) |
| Yes | 2164 | 15 | 2.53 (1.40–4.56) | 1.80 (0.95–3.42) |
| Currently employed | ||||
| No | 12,438 | 38 | 1.00 (Ref.) | 1.00 (Ref.) |
| Yes | 6043 | 19 | 0.96 (0.55–1.67) | 1.68 (0.90–3.14) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muhimpundu, S.; Conway, R.B.N.; Warren Andersen, S.; Lipworth, L.; Steinwandel, M.D.; Blot, W.J.; Shu, X.-O.; Sudenga, S.L. Racial Differences in Hepatocellular Carcinoma Incidence and Risk Factors among a Low Socioeconomic Population. Cancers 2021, 13, 3710. https://doi.org/10.3390/cancers13153710
Muhimpundu S, Conway RBN, Warren Andersen S, Lipworth L, Steinwandel MD, Blot WJ, Shu X-O, Sudenga SL. Racial Differences in Hepatocellular Carcinoma Incidence and Risk Factors among a Low Socioeconomic Population. Cancers. 2021; 13(15):3710. https://doi.org/10.3390/cancers13153710
Chicago/Turabian StyleMuhimpundu, Sylvie, Rebecca Baqiyyah N. Conway, Shaneda Warren Andersen, Loren Lipworth, Mark D. Steinwandel, William J. Blot, Xiao-Ou Shu, and Staci L. Sudenga. 2021. "Racial Differences in Hepatocellular Carcinoma Incidence and Risk Factors among a Low Socioeconomic Population" Cancers 13, no. 15: 3710. https://doi.org/10.3390/cancers13153710
APA StyleMuhimpundu, S., Conway, R. B. N., Warren Andersen, S., Lipworth, L., Steinwandel, M. D., Blot, W. J., Shu, X.-O., & Sudenga, S. L. (2021). Racial Differences in Hepatocellular Carcinoma Incidence and Risk Factors among a Low Socioeconomic Population. Cancers, 13(15), 3710. https://doi.org/10.3390/cancers13153710





