Metabolic Surgery and Cancer Risk: An Opportunity for Mechanistic Research
Abstract
:Simple Summary
Abstract
1. Introduction
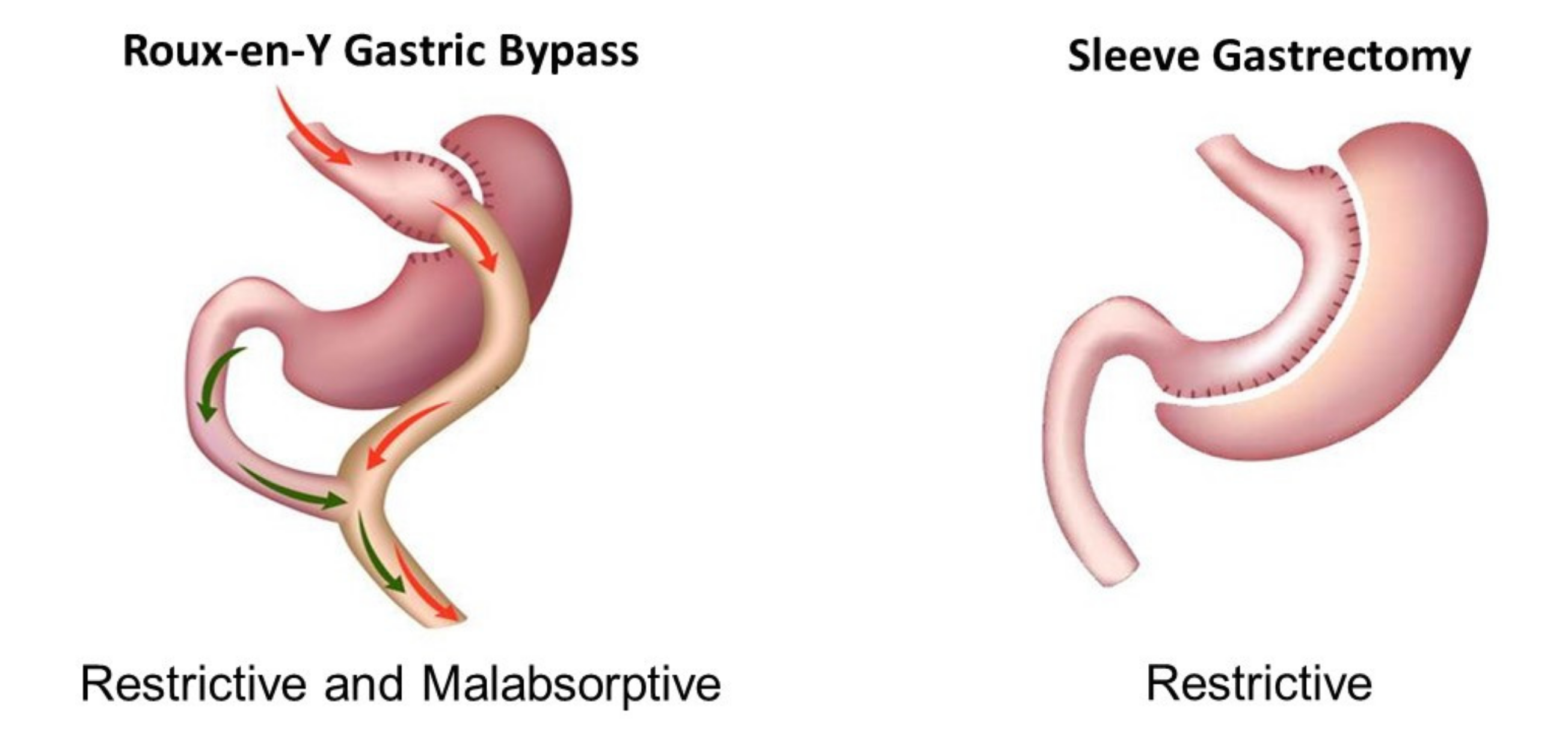
2. Animal Models of MBS
3. The National Institutes of Health (NIH) Focus on Obesity
3.1. Role of the NIH in MBS
- Patients seeking effective therapy for severe obesity for the first time should participate in a nonsurgical program, including a dietary regimen, appropriate exercise, and behavior modification. Transient success of very low-calorie diets, behavioral modification, and limited pharmacologic intervention was also noted.
- Gastric restrictive or bypass procedures could be considered for well-informed and motivated patients with acceptable operative risks.
- Patients who were candidates for surgical procedures should undergo evaluation by a multidisciplinary team.
- The operation should be performed by an experienced surgeon in an appropriate clinical setting.
- Lifelong medical surveillance after surgical therapy is a necessity.
- Specific criteria for operative intervention were determined to be patients with a BMI > 40 kg/m2 as well as patients with a BMI 35–40 kg/m2 who have high risk comorbid conditions, such as cardiopulmonary disease, severe diabetes, and physical problems that interfered with their lifestyle (for example, employment, family functioning, and ambulation).
3.2. National Cancer Institute (NCI) Emphasis on Obesity
4. Impact of MBS on Cancer Risk and Mortality
4.1. Gender May Influence MBS Benefit
4.2. Does MBS Increase the Risk of CRC?
4.3. Do Race and Ethnicity Influence the Effect of MBS on Cancer Risk?
4.4. Does the Specific MBS Procedure Influence Benefit?
5. Proposed Mechanisms Leading to Cancer Risk Reduction after MBS
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
Disclaimer
References
- McCue, M. The History of Bariatric Sur-Gery. 2012. Available online: https://www.firsthealth.org/lifestyle/news-events/2012/09/the-history-of-bariatric-surgery (accessed on 24 June 2021).
- Surgery ASoMaB. Estimate of Bariatric Surgery Numbers, 2011–2018. Available online: https://asmbs.org/resources/estimate-of-bariatric-surgery-numbers (accessed on 24 June 2021).
- Aminian, S.A.; Kirwan, J.P.; Kashyap, S.R.; Burguera, B.; Schauer, P.R. How safe is metabolic/diabetes surgery? Diabetes Obes. Metab. 2015, 17, 198–201. [Google Scholar] [CrossRef]
- Gimeno, R.E.; Briere, D.A.; Seeley, R.J. Leveraging the Gut to Treat Metabolic Disease. Cell Metab. 2020, 31, 679–698. [Google Scholar] [CrossRef] [PubMed]
- Sjöström, L. Review of the key results from the Swedish Obese Subjects (SOS) trial—A prospective controlled intervention study of bariatric surgery. J. Intern. Med. 2013, 273, 219–234. [Google Scholar] [CrossRef]
- Borisenko, O.; Lukyanov, V.; Ahmed, A.R. Cost–utility analysis of bariatric surgery. BJS 2018, 105, 1328–1337. [Google Scholar] [CrossRef] [PubMed]
- Morton, J.M.; Wolfe, B.M.; Nguyen, N.T.; Brethauer, S.; Zundel, N.; Schirmer, B.; Higa, K. Advocacy in action: Bariatric and metabolic surgery. Bull. Am. Coll. Surg. 2019, 104, 51–61. [Google Scholar]
- Frikke-Schmidt, H.; O’Rourke, R.; Lumeng, C.N.; Sandoval, D.A.; Seeley, R.J. Does bariatric surgery improve adipose tissue function? Obes. Rev. 2016, 17, 795–809. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clemmensen, C.; Müller, T.D.; Woods, S.C.; Berthoud, H.-R.; Seeley, R.J.; Tschöp, M.H. Gut-Brain Cross-Talk in Metabolic Control. Cell 2017, 168, 758–774. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vily-Petit, J.; Soty-Roca, M.; Silva, M.; Raffin, M.; Gautier-Stein, A.; Rajas, F.; Mithieux, G. Intestinal gluconeogenesis prevents obesity-linked liver steatosis and non-alcoholic fatty liver disease. Gut 2020, 69, 2193–2202. [Google Scholar] [CrossRef] [PubMed]
- Qi, L.; Guo, Y.; Liu, C.Q.; Huang, Z.P.; Sheng, Y.; Zou, D.J. Effects of bariatric surgery on glycemic and lipid metabolism, sur-gical complication and quality of life in adolescents with obesity: A systematic review and meta-analysis. Surg. Obes. Relat. Dis. 2017, 13, 2037–2055. [Google Scholar] [CrossRef]
- Ashrafian, H.; Bueter, M.; Ahmed, K.; Suliman, A.; Bloom, S.R.; Darzi, A.; Athanasiou, T. Metabolic surgery: An evolution through bariatric animal models. Obes. Rev. 2010, 11, 907–920. [Google Scholar] [CrossRef]
- Troy, S.; Soty, M.; Ribeiro, L.; Laval, L.; Migrenne, S.; Fioramonti, X.; Pillot, B.; Fauveau, V.; Aubert, R.; Viollet, B.; et al. Intestinal Gluconeogenesis Is a Key Factor for Early Metabolic Changes after Gastric Bypass but Not after Gastric Lap-Band in Mice. Cell Metab. 2008, 8, 201–211. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, H.; Hu, C.; Zhang, X.; Jia, W. Role of gut microbiota, bile acids and their cross-talk in the effects of bariatric surgery on obesity and type 2 diabetes. J. Diabetes Investig. 2017, 9, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Kornyushin, O.V.; Sonin, D.L.; Toropova, Y.G.; Pochkaeva, E.I.; Semikova, G.V.; Berko, O.M.; Zelinskaya, I.A.; Todosenko, N.M.; Litvinova, L.S.; Neimark, A.E.; et al. Effects of Bariatric Surgeries on the Size of Myocardial Infarction and Ghrelin Level in Rats with Experimental Decompensated Type 2 Diabetes Mellitus. Bull. Exp. Biol. Med. 2019, 168, 210–213. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, B.M.; Walker, E.; Sarwer, D.; Nguyen, N.T.; Mitchell, J.; Blackstone, R.; Kaplan, L.M.; Buchwald, H.; Pories, W.J. The role of the NIH in the development of metabolic and bariatric surgery. J. Am. Coll. Surg. 2019, 104, 42–50. [Google Scholar]
- Van Itallie, T.B.; Burton, B.T. National Institutes of Health consensus development conference on surgical treatment of morbid obesity. Ann. Surg. 1979, 189, 455–457. [Google Scholar]
- Gastrointestinal surgery for severe obesity. In Proceedings of the National Institutes of Health Consensus Development Conference, Bethesda, MD, USA; Volume 55, pp. 487S–619S.
- National Institutes of Health. Clinical Guidelines on the Identification, Evaluation, and Treatment of Overweight and Obesity in Adults—The Evidence Report. Obes. Res. 1998, 6, 51S–209S. [Google Scholar]
- NIH: Managing Overweight and Obesity in Adults: Systematic Evidence Review from the Obesity Expert Panel. 2013. Available online: https://www.nhlbi.nih.gov/health-topics/managing-overweight-obesity-in-adults (accessed on 24 June 2021).
- Jensen, M.D.; Ryan, D.H.; Apovian, C.M.; Ard, J.D.; Comuzzie, A.G.; Donato, K.A.; Hu, F.B.; Hubbard, V.S.; Jakicic, J.M.; Kushner, R.F.; et al. 2013 AHA/ACC/TOS guideline for the management of overweight and obesity in adults: A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines and The Obesity Society. J. Am. Coll. Cardiol. 2014, 63, 2985–3023. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Belle, S.H.; Berk, P.D.; Courcoulas, A.P.; Flum, D.R.; Miles, C.W.; Mitchell, J.E.; Pories, W.J.; Wolfe, B.M.; Yanovski, S.Z.; Writing, G. Safety and efficacy of bariatric surgery: Longitudinal Assessment of Bariatric Surgery. Surg. Obes. Relat. Dis. 2007, 3, 116–126. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Q.; Li, J.; Wang, Q. Reply to letter to the editor: The effects of bariatric surgery on fracture risk: Is there a significant higher risk? Obes. Rev. 2018, 20, 631–632. [Google Scholar] [CrossRef]
- Schauer, D.P.; Feigelson, H.S.; Koebnick, C.; Caan, B.; Weinmann, S.; Leonard, A.C.; Powers, J.D.; Yenumula, P.R.; Arterburn, D.E. Bariatric Surgery and the Risk of Cancer in a Large Multisite Cohort. Ann. Surg. 2019, 269, 95–101. [Google Scholar] [CrossRef]
- Carlsson, L.M.; Sjöholm, K.; Jacobson, P.; Andersson-Assarsson, J.C.; Svensson, P.-A.; Taube, M.; Carlsson, B.; Peltonen, M. Life Expectancy after Bariatric Surgery in the Swedish Obese Subjects Study. N. Engl. J. Med. 2020, 383, 1535–1543. [Google Scholar] [CrossRef]
- Sjöström, L.; Gummesson, A.; Sjöström, C.D.; Narbro, K.; Peltonen, M.; Wedel, H.; Bengtsson, C.; Bouchard, C.; Carlsson, B.; Dahlgren, S.; et al. Effects of bariatric surgery on cancer incidence in obese patients in Sweden (Swedish Obese Subjects Study): A prospective, controlled intervention trial. Lancet Oncol. 2009, 10, 653–662. [Google Scholar] [CrossRef]
- Mackenzie, H.; Markar, S.R.; Askari, A.; Faiz, O.; Hull, M.; Purkayastha, S.; Møller, H.; Lagergren, J. Obesity surgery and risk of cancer. BJS 2018, 105, 1650–1657. [Google Scholar] [CrossRef] [PubMed]
- Aravani, A.; Downing, A.; Thomas, J.D.; Lagergren, J.; Morris, E.J.; Hull, M.A. Obesity surgery and risk of colorectal and other obesity-related cancers: An English population-based cohort study. Cancer Epidemiol. 2018, 53, 99–104. [Google Scholar] [CrossRef] [PubMed]
- Östlund, M.P.; Lu, Y.; Lagergren, J. Risk of Obesity-Related Cancer After Obesity Surgery in a Population-Based Cohort Study. Ann. Surg. 2010, 252, 972–976. [Google Scholar] [CrossRef] [PubMed]
- Bailly, L.; Fabre, R.; Pradier, C.; Iannelli, A. Colorectal Cancer Risk Following Bariatric Surgery in a Nationwide Study of French Individuals With Obesity. JAMA Surg. 2020, 155, 395–402. [Google Scholar] [CrossRef]
- Tee, M.C.; Cao, Y.; Warnock, G.L.; Hu, F.B.; Chavarro, J.E. Effect of bariatric surgery on oncologic outcomes: A systematic review and meta-analysis. Surg. Endosc. 2013, 27, 4449–4456. [Google Scholar] [CrossRef] [Green Version]
- Williamson, D.F.; Pamuk, E.; Thun, M.; Flanders, D.; Byers, T.; Heath, C. Prospective Study of Intentional Weight Loss and Mortality in Never-Smoking Overweight US White Women Aged 40–64 Years. Am. J. Epidemiol. 1995, 141, 1128–1141. [Google Scholar] [CrossRef]
- Williamson, D.F.; Pamuk, E.; Thun, M.; Flanders, D.; Byers, T.; Heath, C. Prospective Study of Intentional Weight Loss and Mortality in Overweight White Men Aged 40-64 Years. Am. J. Epidemiol. 1999, 149, 491–503. [Google Scholar] [CrossRef] [Green Version]
- Fuchs, H.F.; Broderick, R.C.; Harnsberger, C.R.; Chang, D.C.; Sandler, B.J.; Jacobsen, G.R.; Horgan, S. Benefits of Bariatric Surgery Do Not Reach Obese Men. J. Laparoendosc. Adv. Surg. Technol. 2015, 25, 196–201. [Google Scholar] [CrossRef]
- Karp, I.; Behlouli, H.; LeLorier, J.; Pilote, L. Statins and Cancer Risk. Am. J. Med. 2008, 121, 302–309. [Google Scholar] [CrossRef] [PubMed]
- MacKintosh, M.L.; Derbyshire, A.E.; McVey, R.J.; Bolton, J.; Nickkho-Amiry, M.; Higgins, C.L.; Kamieniorz, M.; Pemberton, P.W.; Kirmani, B.H.; Ahmed, B.; et al. The impact of obesity and bariatric surgery on circulating and tissue biomarkers of en-dometrial cancer risk. Int. J. Cancer 2019, 144, 641–650. [Google Scholar] [CrossRef] [PubMed]
- Coussens, L.M.; Werb, Z. Inflammation and cancer. Nature 2002, 420, 860–867. [Google Scholar] [CrossRef]
- Marseglia, L.; Manti, S.; D’Angelo, G.; Nicotera, A.G.; Parisi, E.; Di Rosa, G.; Gitto, E.; Arrigo, T. Oxidative Stress in Obesity: A Critical Component in Human Diseases. Int. J. Mol. Sci. 2014, 16, 378–400. [Google Scholar] [CrossRef] [Green Version]
- Djiogue, S.; Kamdje, A.H.N.; Vecchio, L.; Kipanyula, M.J.; Farahna, M.; Aldebasi, Y.; Etet, P.F.S. Insulin resistance and cancer: The role of insulin and IGFs. Endocr. Relat. Cancer 2012, 20, R1–R17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Barataud, A.; Vily-Petit, J.; Goncalves, D.; Zitoun, C.; Duchampt, A.; Philippe, E.; Gautier-Stein, A.; Mithieux, G. Metabolic benefits of gastric bypass surgery in the mouse: The role of fecal losses. Mol. Metab. 2020, 31, 14–23. [Google Scholar] [CrossRef]
- Booth, A.; Magnuson, A.; Fouts, J.; Foster, M. Adipose tissue, obesity and adipokines: Role in cancer promotion. Horm. Mol. Biol. Clin. Investig. 2015, 21, 57–74. [Google Scholar] [CrossRef]
- Izquierdo, A.G.; Crujeiras, A.B. Obesity-Related Epigenetic Changes After Bariatric Surgery. Front. Endocrinol. 2019, 10, 232. [Google Scholar] [CrossRef]
- Al-Assal, K.; Martinez, A.C.; Torrinhas, R.S.; Cardinelli, C.; Waitzberg, D. Gut microbiota and obesity. Clin. Nutr. Exp. 2018, 20, 60–64. [Google Scholar] [CrossRef] [Green Version]
- Cornejo-Pareja, I.; Clemente-Postigo, M.; Tinahones, F.J. Metabolic and Endocrine Consequences of Bariatric Surgery. Front. Endocrinol. 2019, 10, 626. [Google Scholar] [CrossRef] [Green Version]
- Sainsbury, A.; Goodlad, R.A.; Perry, S.L.; Pollard, S.G.; Robins, G.G.; Hull, M.A. Increased Colorectal Epithelial Cell Proliferation and Crypt Fission Associated with Obesity and Roux-en-Y Gastric Bypass. Cancer Epidemiol. Biomark. Prev. 2008, 17, 1401–1410. [Google Scholar] [CrossRef] [Green Version]
- Li, J.V.; Ashrafian, H.; Bueter, M.; Kinross, J.; Sands, C.; Le Roux, C.; Bloom, S.R.; Darzi, A.; Athanasiou, T.; Marchesi, J.R.; et al. Metabolic surgery profoundly influences gut microbial-host metabolic cross-talk. Gut 2011, 60, 1214–1223. [Google Scholar] [CrossRef] [PubMed]
- Istfan, N.; Anderson, W.A.; Apovian, C.; Ruth, M.; Carmine, B.; Hess, D. Racial differences in weight loss, hemoglobin A1C, and blood lipid profiles after Roux-en-Y gastric bypass surgery. Surg. Obes. Relat. Dis. 2016, 12, 1329–1336. [Google Scholar] [CrossRef] [PubMed]
- Wood, M.H.; Carlin, A.M.; Ghaferi, A.A.; Varban, O.A.; Hawasli, A.; Bonham, A.J.; Birkmeyer, N.J.; Finks, J.F. Association of Race With Bariatric Surgery Outcomes. JAMA Surg. 2019, 154, e190029. [Google Scholar] [CrossRef]
- McTigue, K.M.; Wellman, R.; Nauman, E.; Anau, J.; Coley, R.Y.; Odor, A.; Tice, J.; Coleman, K.J.; Courcoulas, A.; Pardee, R.E.; et al. Comparing the 5-Year Diabetes Outcomes of Sleeve Gastrectomy and Gastric Bypass: The National Patient-Centered Clinical Research Network (PCORNet) Bariatric Study. JAMA Surg. 2020, 155, e200087. [Google Scholar] [CrossRef] [Green Version]
- Sjöström, L.; Narbro, K.; Sjöström, C.D.; Karason, K.; Larsson, B.; Wedel, H.; Ted Lystig, T.; Sullivan, M.; Bouchard, C.; Carlsson, B.; et al. Effects of bariatric surgery on mortality in obese swedish subjects. N. Engl. J. Med. 2007, 357, 741–752. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stroud, A.M.; Dewey, E.N.; Husain, F.A.; Fischer, J.M.; Courcoulas, A.P.; Flum, D.R.; Mitchell, J.E.; Pories, W.J.; Purnell, J.Q.; Wolfe, B.M. Association between weight loss and serum biomarkers with risk of incident cancer in the Longitudinal Assessment of Bariatric Surgery cohort. Surg. Obes. Relat. Dis. 2020, 16, 1086–1094. [Google Scholar] [CrossRef]
- Hopkins, B.D.; Goncalves, M.; Cantley, L.C. Obesity and Cancer Mechanisms: Cancer Metabolism. J. Clin. Oncol. 2016, 34, 4277–4283. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Title | Dates | Institute(s) | Purpose | Focus of Surgical Procedures Discussed | Outcomes |
|---|---|---|---|---|---|
| Consensus Development Conference on Surgical Treatment of Morbid Obesity | 12.4–5.1978 | NIDDK | Establish agreement among experts on use of emerging technologies for morbid obesity | GB, JI bypass | 1. Improvement in CV and T2DM with weight loss after BS; 2. GB has fewer side effects than JI bypass; 3. surgery should be limited to patients with morbid obesity; 4. the mechanism(s) of the effect of MBS needs more attention |
| Consensus Conference on the Health Implications of Obesity | 2.11–13.1985 | NIDDK, NHLBI | What is obesity; what is the evidence that obesity adversely affects health; for what medical indications can weight loss be recommended; what is the evidence that obesity affects longevity; what should be the directions of future research in the area | Primary conference focus not on BS | Increased risk of CV disease, dyslipidemia, T2DM, gallbladder disease, increased prevalence of and mortality from cancer |
| Gastrointestinal surgery for severe obesity. Proceedings of an NIH Consensus Development Conference | 3.25–27.1991 | NIDDK | Assess the MBS treatments for obesity, the criteria for selection of MBS, the efficacy and risks of MBS, and the need for further research on MBS therapies | VBG, RYGB | (1) MBS restrictive or bypass (malabsorptive) procedures could be considered for candidates who are good surgical risks; (2) MBS surgical candidates should be evaluated by multiple disciplines with medical, surgical, psychiatric and nutritional expertise; (3) the MBS procedure should be performed in a setting with adequate support for all aspects of MBS care; (4) specific BMI criteria for surgery; (5) lifelong medical surveillance after MBS in needed |
| Expert Panel on the Identification, Evaluation, and Treatment of Overweight and Obesity in Adults | Panel convened May 1995 | NHLBI, NIDDK | To evaluate published randomized clinical trials published between 1980 and 1997 to determine optimal treatment for overweight and obesity | VBG, RYGB | MBS is an option for subjects with BMI > 40 or > 35 kg/m2 with comorbid conditions when medical management has failed |
| Managing Overweight and Obesity in Adults: Systematic Evidence Review from the Obesity Expert Panel | 2008 | NHLBI | Expert panels were convened to update the existing clinical guidelines on cholesterol, blood pressure, and overweight/obesity, by conducting rigorous systematic evidence reviews. | VBG, adjustable gastric banding, RYGB, SG | MBS produces greater initial and maintained weight loss than other weight loss approaches. In obese adults, MBS generally results in a more favorable impact on obesity-related comorbid conditions than that produced by usual care, conventional medical treatment, lifestyle intervention, or medically supervised weight loss. MBS results in greater improvement in obesity-related comorbid conditions than nonsurgical approaches. Complications, weight loss and remission-related comorbidities following MBS vary by procedure. |
| Study Design | Study Groups/Numbers | Treatment Procedure(s) | Cancer Incidence +/- Mortality | Cancer Effect | Reference | |||
|---|---|---|---|---|---|---|---|---|
| Overall | Cancer | Females | Males | |||||
| Prospective matched cohort | T: 2010; C: 2037; F: 2867; M: 1180 | GB, VBG, RYGB | Cancer incidence, mortality | Incidence HR = 0.67 (p < 0.01); mortality HR = 0.77, p < 0.01 | Melanoma HR = ND, p < 0.01; Hematologic HR = 0.16, p = 0.015; Other cancers HR = 0.40; p = 0.04 | GB HR = 0.54, p = 0.026; VBG = 0.60, p < 0.01; RYGB HR = 0.54, p = 0.11 | HR = 0.97, NS | [25,26] |
| Retrospective matched cohort | T: 22,198; C: 66,427; F: >80% | Various MBS procedures | Cancer incidence | HR = 0.67, p < 0.01; obesity related cancers HR: 0.59, p < 0.01 | Postmenopausal breast HR = 0.58, p < 0.01; colon HR = 0.59; p = 0.04; endometrial HR = 0.50, p < 0.01; pancreatic cancer HR = 0.46, p = 0.04 | HR = 0.64, p < 0.01), obesity associated cancers (HR = 0.58, p < 0.01), not obesity associated (HR 0.74, p < 0.01) | No significant cancer reduction for any cancer type | [24] |
| Meta analysis | T: 304,516; C: 8,492,408; both M/F | Various MBS procedures | Cancer incidence, mortality | Incidence OR = 0.56, p < 0.01; mortality OR = 0.56, p < 0.01 | Breast HR = 0.49, p < 0.01; CRC 0.82, p,0.01; Endometrial HR = 0.43, p = 0.01 | Not studied | [23] | |
| Retrospective matched cohort | T: 8794; C: 8794 | Bypass (including RYGB), GB, SG | Cancer incidence | Focus on obesity related cancers | Hormone related cancers OR = 0.23 (RYGB OR = 0.16), breast OR = 0.25; endometrium OR:0.21, prostate OR:0.37; CRC OR = 2.63 (RYGB) | Hormone related cancers OR = 0.22; CRC OR = 3.01 (RYGB) | Hormone related cancers OR = 0.37; CRC OR = 2.01 (RYGB) | [27] |
| Retrospective observational cohort | T: 39,747 (F: 76.6%); C: 962,860 (F: 62.9%) | 52% restrictive, 48% restrictive & malabsorptive | Cancer incidence | Focus on obesity related cancers | Breast SIR = 0.76; Uterus SIR = 2.98; Kidney SIR = 3.06; Lung SIR = 0.70; CRC SIR = 1.26; restrictive CRC SIR = 1.41; restrictive & malabsorptive CRC SIR = 1.05 | CRC SIR = 1.19 | CRC SIR = 1.41 | [28] |
| Retrospective observational cohort | T: 13,123 (F: 77%); C: Swedish national registry | GB, VBG, Bypass (including RYGB) | Cancer incidence | Focus on obesity related cancers | Obesity related cancers SIR = 1.04; CRC SIR = 1.52; Kidney SIR = 2.68; Bypass SIR = 1.01; VBG SIR = 1.05; GB SIR = 1.05 | Breast SIR = 0.54; Endometrial SIR = 2.15; CRC SIR = 1.28 | Prostate SIR = 0.84; CRC SIR = 2.34 | [29] |
| Retrospective observational cohort | T: 74,131(F: 77.9%); C: 971,217(F: 49.4%) | GB, SG, Bypass (including RYGB) | Cancer incidence | Focus on CRC | CRC SIR = 0.75 | CRC SIR = 0.79 | CRC SIR = 0.77 | [30] |
| Proposed Mechanism | How the Mechanism Influences Obesity Related Cancer | Reference |
|---|---|---|
| ↓ Cell Proliferation | Lower cell proliferation→lower cell division→lower mutation rate | [36] |
| ↓ Chronic Inflammation | ↓ growth factors and cytokines→↓cell proliferation, cell survival and migration | [36,37] |
| ↓ Oxidative Stress | ↓chronic inflammation→↓oxidative stress→↑antioxidant enzymes, ↓OS byproducts, ↓ cancer risk | [38] |
| ↓ Insulin Resistance | Decreased cell proliferation and increased apoptosis | [36,39] |
| ↑ IGN | IGN improves insulin sensitivity, energy homoeostasis and exerts anti-obesity effects | [10,40] |
| ↓ IGF-1 | ↓growth factor expression→↓cell proliferation, cell survival and migration↓ | [37] |
| ↓ Adipocyte Size | Normalizes adipocyte function | [41] |
| Epigenetic ∆ in Adipocytes | Favorable changes in obesity epigenome after MBS | [42] |
| ∆ Adipokine Expression | ↓ growth-promoting adipokines, ↑ adiponectin decreases IGF-1 | [41] |
| ∆ Gut Microbiome | Gut microbiota types influence obesity; levels change after bariatric surgery | [14,43] |
| ↓ Aromatase | Fewer adipocytes→lower aromatase levels Lower hormone receptor levels leads to lower cell proliferation from hormone signaling | [44] |
| ↓ ER, PR ↑ Bile Acid Concentration ∆ Concentrations of Gut Hormones | May increase colonic proliferation Influences brain-gut crosstalk | [45] [9] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sauter, E.R.; Heckman-Stoddard, B. Metabolic Surgery and Cancer Risk: An Opportunity for Mechanistic Research. Cancers 2021, 13, 3183. https://doi.org/10.3390/cancers13133183
Sauter ER, Heckman-Stoddard B. Metabolic Surgery and Cancer Risk: An Opportunity for Mechanistic Research. Cancers. 2021; 13(13):3183. https://doi.org/10.3390/cancers13133183
Chicago/Turabian StyleSauter, Edward R., and Brandy Heckman-Stoddard. 2021. "Metabolic Surgery and Cancer Risk: An Opportunity for Mechanistic Research" Cancers 13, no. 13: 3183. https://doi.org/10.3390/cancers13133183
APA StyleSauter, E. R., & Heckman-Stoddard, B. (2021). Metabolic Surgery and Cancer Risk: An Opportunity for Mechanistic Research. Cancers, 13(13), 3183. https://doi.org/10.3390/cancers13133183






