Involvement of Neutrophils in Metastatic Evolution of Pancreatic Neuroendocrine Tumors
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patient Population
2.2. NLR Calculation
2.3. Immunohistochemistry Neutrophils and Lymphocyte Assessment
2.4. RNA Sequencing Data and Bioinformatic Analysis
2.5. Statistical Analysis
3. Results
3.1. Patients’ Characteristics
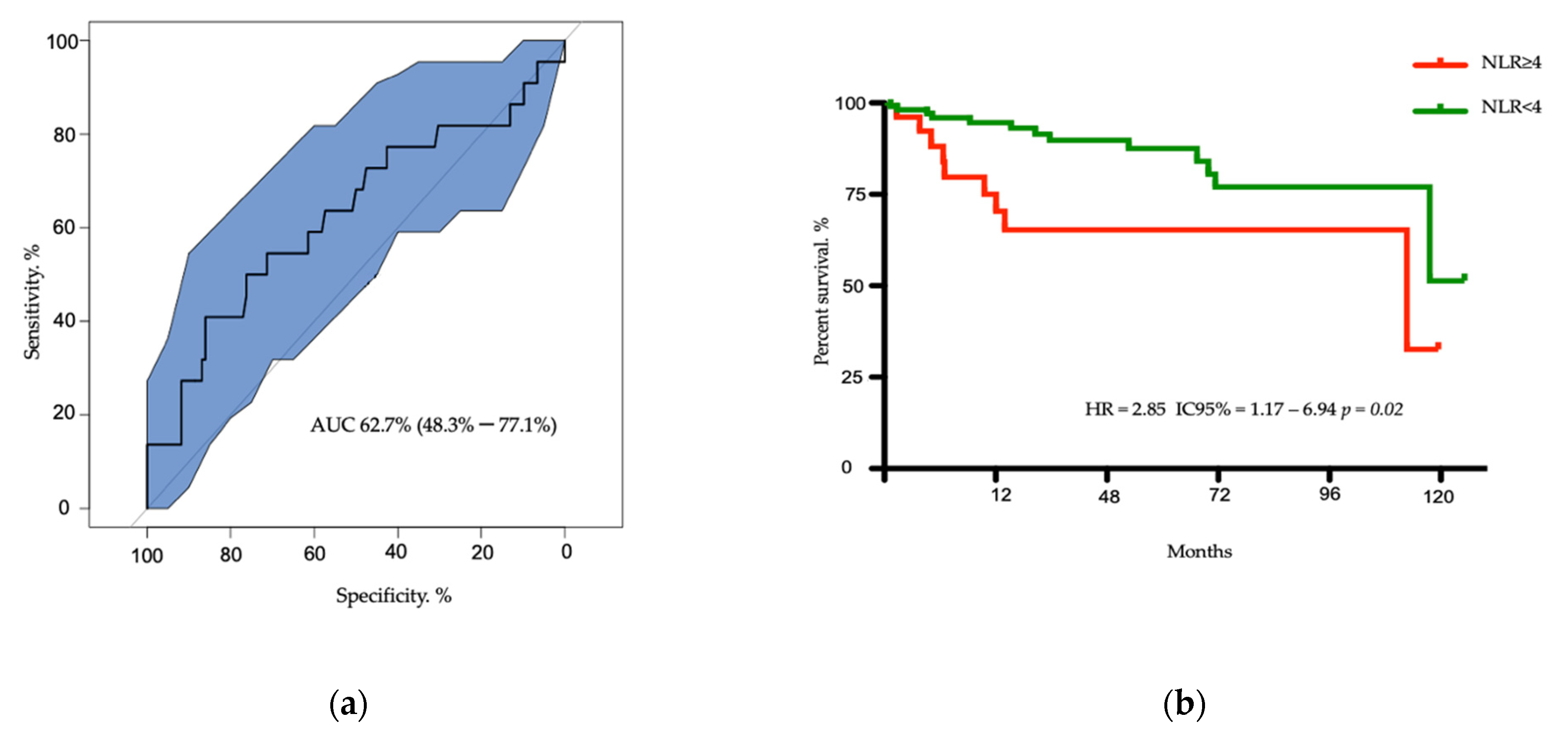
3.2. Survival of Patients According to NLR Ratio
3.3. Univariate and Multivariate Analysis for Overall Survival
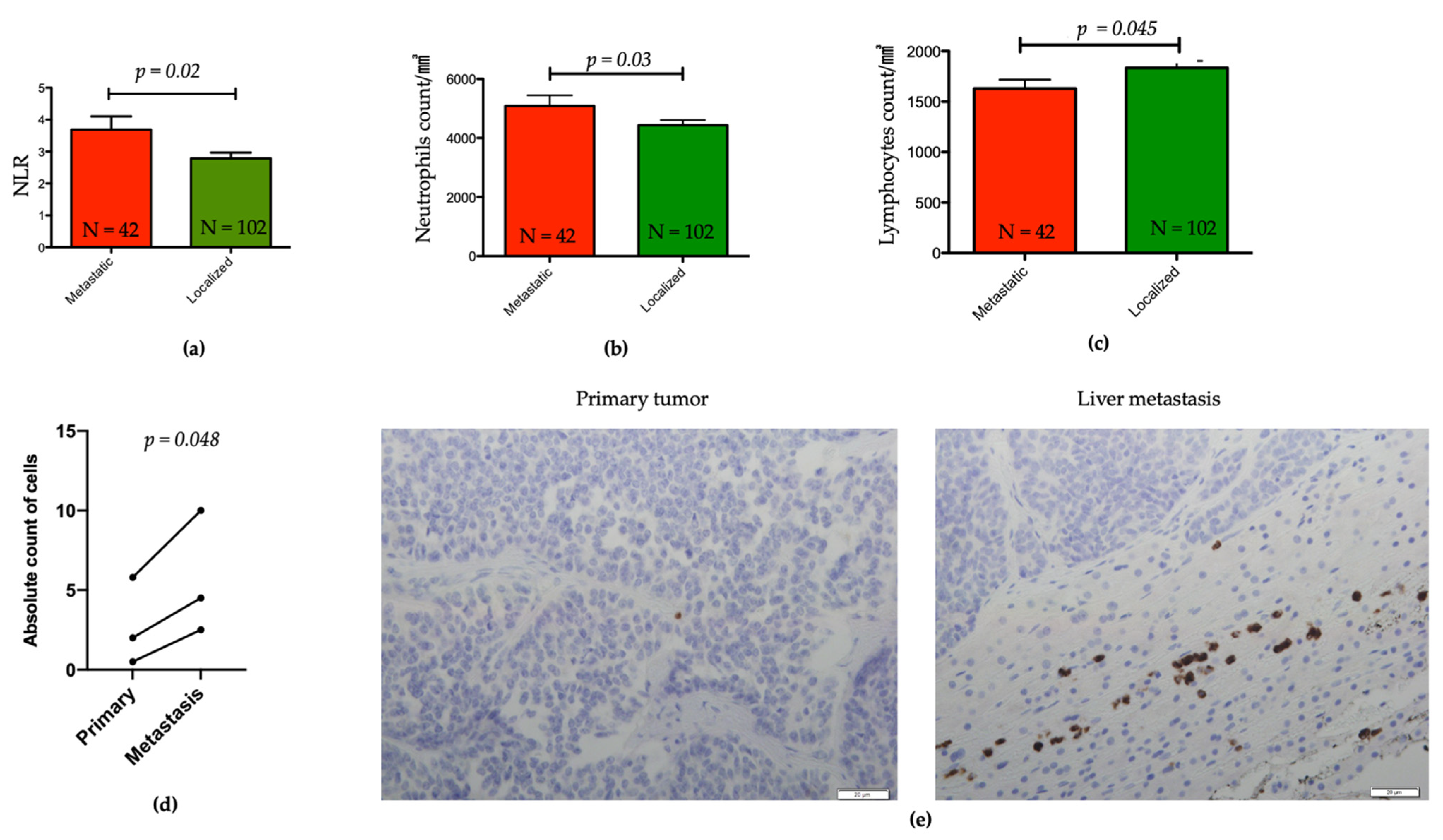
3.4. Association between NLR and Other Clinicopathological Features
3.5. Evaluation of Tumors Associated Neutrophils in Liver Metastasis and Matched Primary pNETs
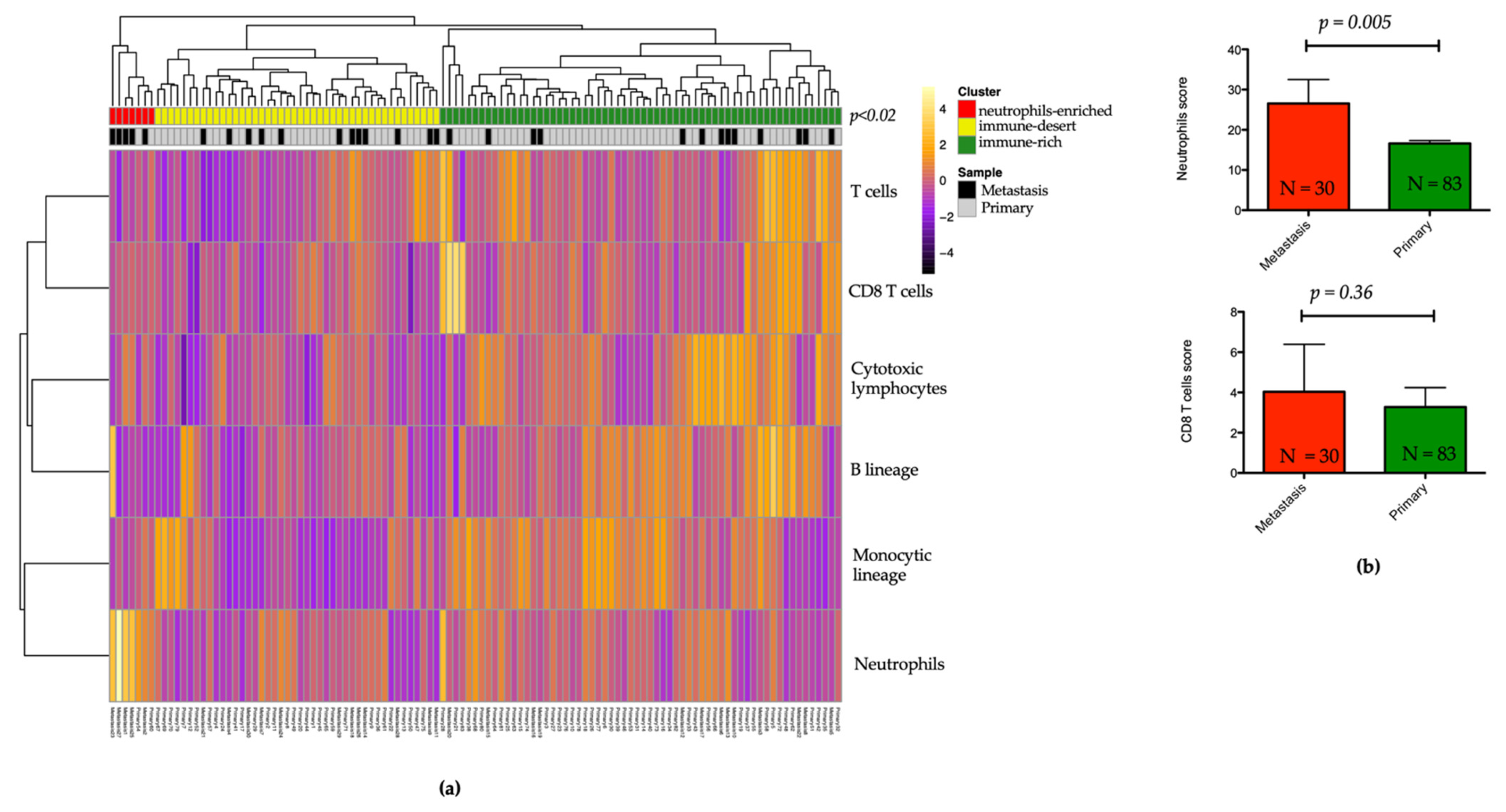
3.6. Landscape of the Microenvironment Phenotypes in pNET
3.7. Association between Neutrophils Infiltration, Complement Pathway Activation, and Metastatic Tumor Status
3.8. Subtypes Classification of pNETs Using Neutrophils and Complement Pathway Signature
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bosman, F.T.; Carneiro, F.; Hruban, R.H.; Theise, N.D. WHO Classification of Tumours of the Digestive System; WHO: Geneva, Switzerland, 2010. [Google Scholar]
- Dasari, A.; Shen, C.; Halperin, D.; Zhao, B.; Zhou, S.; Xu, Y.; Shih, T.; Yao, J.C. Trends in the Incidence, Prevalence, and Survival Outcomes in Patients with Neuroendocrine Tumours in the United States. JAMA Oncol. 2017, 3, 1335. [Google Scholar] [CrossRef] [PubMed]
- Falconi, M.; Eriksson, B.; Kaltsas, G.; Bartsch, D.K.; Capdevila, J.; Caplin, M.; Kos-Kudla, B.; Kwekkeboom, D.; Rindi, G.; Klöppel, G.; et al. ENETS Consensus Guidelines Update for the Management of Patients with Functional Pancreatic Neuroendocrine Tumours and Non-Functional Pancreatic Neuroendocrine Tumours. Neuroendocrinology 2016, 103, 153–171. [Google Scholar] [CrossRef] [PubMed]
- Klöppel, G.; Couvelard, A.; Hruban, R.; Klimstra, D.S.; Komminoth, P.; Osamura, R.Y.; Perren, A.; Rindi, G. Neoplasms of the Neuroendocrine Pancreas: Introduction. In WHO Classification of Tumours of Endocrine Organs; Lloyd, R.V., Osamura, R.Y., Klöppel, G., Rosai, J., Eds.; IARC: Lyon, France, 2017; Volume 10, ISBN 9789283244936. [Google Scholar]
- Genc, C.G.; Klümpen, H.J.; van Oijen, M.G.H.; van Eijck, C.H.J.; Nieveen van Dijkum, E.J.M. A Nationwide Population-Based Study on the Survival of Patients with Pancreatic Neuroendocrine Tumours in The Netherlands. World J. Surg. 2018, 42, 490–497. [Google Scholar] [CrossRef] [PubMed]
- Genç, C.G.; Jilesen, A.P.; Partelli, S.; Falconi, M.; Muffatti, F.; van Kemenade, F.J.; van Eeden, S.; Verheij, J.; van Dieren, S.; van Eijck, C.H.J.; et al. A New Scoring System to Predict Recurrent Disease in Grade 1 and 2 Nonfunctional Pancreatic Neuroendocrine Tumours. Ann. Surg. 2018, 267, 1148–1154. [Google Scholar] [CrossRef]
- Pavel, M.; O’Toole, D.; Costa, F.; Capdevila, J.; Gross, D.; Kianmanesh, R.; Krenning, E.; Knigge, U.; Salazar, R.; Pape, U.-F.; et al. ENETS Consensus Guidelines Update for the Management of Distant Metastatic Disease of Intestinal, Pancreatic, Bronchial Neuroendocrine Neoplasms (NEN) and NEN of Unknown Primary Site. Neuroendocrinology 2016, 103, 172–185. [Google Scholar] [CrossRef]
- Gao, H.; Liu, L.; Wang, W.; Xu, H.; Jin, K.; Wu, C.; Qi, Z.; Zhang, S.; Liu, C.; Xu, J.; et al. Novel Recurrence Risk Stratification of Resected Pancreatic Neuroendocrine Tumour. Cancer Lett. 2018, 412, 188–193. [Google Scholar] [CrossRef]
- Marchegiani, G.; Landoni, L.; Andrianello, S.; Masini, G.; Cingarlini, S.; D’Onofrio, M.; De Robertis, R.; Davì, M.; Capelli, P.; Manfrin, E.; et al. Patterns of Recurrence after Resection for Pancreatic Neuroendocrine Tumours: Who, When, and Where? Neuroendocrinology 2018, 108, 161–171. [Google Scholar] [CrossRef]
- Jensen, R.T.; Bodei, L.; Capdevila, J.; Couvelard, A.; Falconi, M.; Glasberg, S.; Kloppel, G.; Lamberts, S.; Peeters, M.; Rindi, G.; et al. Unmet Needs in Functional and Nonfunctional Pancreatic Neuroendocrine Neoplasms. Neuroendocrinology 2019, 108, 26–36. [Google Scholar] [CrossRef]
- Hanahan, D.; Weinberg, R.A. Hallmarks of Cancer: The next Generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Coussens, L.M.; Werb, Z. Inflammation and Cancer. Nature 2002, 420, 860–867. [Google Scholar] [CrossRef]
- Liang, W.; Ferrara, N. The Complex Role of Neutrophils in Tumour Angiogenesis and Metastasis. Cancer Immunol. Res. 2016, 4, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Granot, Z.; Fridlender, Z.G. Plasticity beyond Cancer Cells and the “Immunosuppressive Switch”. Cancer Res. 2015, 75, 4441–4445. [Google Scholar] [CrossRef] [PubMed]
- Shaul, M.E.; Fridlender, Z.G. Tumour-Associated Neutrophils in Patients with Cancer. Nat. Rev. Clin. Oncol. 2019, 16, 601–620. [Google Scholar] [CrossRef]
- Haram, A.; Boland, M.R.; Kelly, M.E.; Bolger, J.C.; Waldron, R.M.; Kerin, M.J. The Prognostic Value of Neutrophil-to-Lymphocyte Ratio in Colorectal Cancer: A Systematic Review. J. Surg. Oncol. 2017, 115, 470–479. [Google Scholar] [CrossRef] [PubMed]
- Templeton, A.J.; McNamara, M.G.; Šeruga, B.; Vera-Badillo, F.E.; Aneja, P.; Ocaña, A.; Leibowitz-Amit, R.; Sonpavde, G.; Knox, J.J.; Tran, B.; et al. Prognostic Role of Neutrophil-to-Lymphocyte Ratio in Solid Tumours: A Systematic Review and Meta-Analysis. JNCI J. Natl. Cancer Inst. 2014, 106, dju124. [Google Scholar] [CrossRef] [PubMed]
- Luo, G.; Liu, C.; Cheng, H.; Jin, K.; Guo, M.; Lu, Y.; Long, J.; Xu, J.; Ni, Q.; Chen, J.; et al. Neutrophil-Lymphocyte Ratio Predicts Survival in Pancreatic Neuroendocrine Tumours. Oncol. Lett. 2017, 13, 2454–2458. [Google Scholar] [CrossRef]
- Arima, K.; Okabe, H.; Hashimoto, D.; Chikamoto, A.; Nitta, H.; Higashi, T.; Kaida, T.; Yamamura, K.; Kitano, Y.; Komohara, Y.; et al. Neutrophil-to-Lymphocyte Ratio Predicts Metachronous Liver Metastasis of Pancreatic Neuroendocrine Tumours. Int. J. Clin. Oncol. 2017, 22, 734–739. [Google Scholar] [CrossRef]
- Panni, R.Z.; Lopez-Aguiar, A.G.; Liu, J.; Poultsides, G.A.; Rocha, F.G.; Hawkins, W.G.; Strasberg, S.M.; Trikalinos, N.A.; Maithel, S.; Fields, R.C.; et al. Association of Preoperative Monocyte-to-lymphocyte and Neutrophil-to-lymphocyte Ratio with Recurrence-free and Overall Survival after Resection of Pancreatic Neuroendocrine Tumours (US-NETSG). J. Surg. Oncol. 2019, 22, 734–739. [Google Scholar] [CrossRef]
- Tong, Z.; Liu, L.; Zheng, Y.; Jiang, W.; Zhao, P.; Fang, W.; Wang, W. Predictive Value of Preoperative Peripheral Blood Neutrophil/Lymphocyte Ratio for Lymph Node Metastasis in Patients of Resectable Pancreatic Neuroendocrine Tumours: A Nomogram-Based Study. World J. Surg. Oncol. 2017, 15, 1–9. [Google Scholar] [CrossRef]
- Harimoto, N.; Hoshino, K.; Muranushi, R.; Hagiwara, K.; Yamanaka, T.; Ishii, N.; Tsukagoshi, M.; Igarashi, T.; Tanaka, H.; Watanabe, A.; et al. Prognostic Significance of Neutrophil-Lymphocyte Ratio in Resectable Pancreatic Neuroendocrine Tumours with Special Reference to Tumour-Associated Macrophages. Pancreantology 2019, 19, 897–902. [Google Scholar] [CrossRef]
- Gaitanidis, A.; Patel, D.; Nilubol, N.; Tirosh, A.; Sadowski, S.; Kebebew, E. Markers of Systemic Inflammatory Response Are Prognostic Factors in Patients with Pancreatic Neuroendocrine Tumours (PNETs): A Prospective Analysis. Ann. Surg. Oncol. 2018, 25, 122–130. [Google Scholar] [CrossRef] [PubMed]
- Milione, M.; Miceli, R.; Barretta, F.; Pellegrinelli, A.; Spaggiari, P.; Tagliabue, G.; Centonze, G.; Paolino, C.; Mangogna, A.; Kankava, K.; et al. Microenvironment and Tumour Inflammatory Features Improve Prognostic Prediction in Gastro-Entero-Pancreatic Neuroendocrine Neoplasms. J. Pathol. Clin. Res. 2019, 5, 217–226. [Google Scholar] [CrossRef] [PubMed]
- Bruni, D.; Angell, H.K.; Galon, J. The Immune Contexture and Immunoscore in Cancer Prognosis and Therapeutic Efficacy. Nat. Rev. Cancer 2020, 20, 662–680. [Google Scholar] [CrossRef]
- Petitprez, F.; de Reyniès, A.; Keung, E.Z.; Chen, T.W.-W.; Sun, C.-M.; Calderaro, J.; Jeng, Y.-M.; Hsiao, L.-P.; Lacroix, L.; Bougoüin, A.; et al. B Cells Are Associated with Survival and Immunotherapy Response in Sarcoma. Nature 2020, 577, 556–560. [Google Scholar] [CrossRef]
- Sobin, L.H.; Gospodarowicz, M.K.; Wittekind, C. TNM Classification of Malignant Tumours; John Wiley & Sons: Hoboken, NJ, USA, 2011; ISBN 9781444358964. [Google Scholar]
- Inzani, F.; Petrone, G.; Rindi, G. The New World Health Organization Classification for Pancreatic Neuroendocrine Neoplasia. Endocrinol. Metab. Clin. North Am. 2018, 47, 463–470. [Google Scholar] [CrossRef]
- Alvarez, M.J.; Subramaniam, P.S.; Tang, L.H.; Grunn, A.; Aburi, M.; Rieckhof, G.; Komissarova, E.V.; Hagan, E.A.; Bodei, L.; Clemons, P.A.; et al. A Precision Oncology Approach to the Pharmacological Targeting of Mechanistic Dependencies in Neuroendocrine Tumours. Nat. Genet. 2018, 50, 979. [Google Scholar] [CrossRef] [PubMed]
- Becht, E.; Giraldo, N.A.; Lacroix, L.; Buttard, B.; Elarouci, N.; Petitprez, F.; Selves, J.; Laurent-Puig, P.; Sautès-Fridman, C.; Fridman, W.H.; et al. Estimating the Population Abundance of Tissue-Infiltrating Immune and Stromal Cell Populations Using Gene Expression. Genome Biol. 2016, 17, 1–20. [Google Scholar] [CrossRef]
- Love, M.I.; Huber, W.; Anders, S. Moderated Estimation of Fold Change and Dispersion for RNA-Seq Data with DESeq2. Genome Biol. 2014, 15, 550. [Google Scholar] [CrossRef]
- Benjamini, Y.; Hochberg, Y. Controlling the False Discovery Rate: A Practical and Powerful Approach to Multiple Testing. J. R. Stat. Soc. Ser. B Methodol. 1995, 57, 289–300. [Google Scholar] [CrossRef]
- Subramanian, A.; Tamayo, P.; Mootha, V.K.; Mukherjee, S.; Ebert, B.L.; Gillette, M.A.; Paulovich, A.; Pomeroy, S.L.; Golub, T.R.; Lander, E.S.; et al. Gene Set Enrichment Analysis: A Knowledge-Based Approach for Interpreting Genome-Wide Expression Profiles. Proc. Natl. Acad. Sci. USA 2005, 102, 15545–15550. [Google Scholar] [CrossRef]
- Mootha, V.K.; Lindgren, C.M.; Eriksson, K.-F.; Subramanian, A.; Sihag, S.; Lehar, J.; Puigserver, P.; Carlsson, E.; Ridderstråle, M.; Laurila, E.; et al. PGC-1alpha-Responsive Genes Involved in Oxidative Phosphorylation Are Coordinately Downregulated in Human Diabetes. Nat. Genet. 2003, 34, 267–273. [Google Scholar] [CrossRef]
- Panzuto, F.; Boninsegna, L.; Fazio, N.; Campana, D.; Pia Brizzi, M.; Capurso, G.; Scarpa, A.; De Braud, F.; Dogliotti, L.; Tomassetti, P.; et al. Metastatic and Locally Advanced Pancreatic Endocrine Carcinomas: Analysis of Factors Associated with Disease Progression. J. Clin. Oncol. 2011, 29, 2372–2377. [Google Scholar] [CrossRef]
- Scarpa, A.; Mantovani, W.; Capelli, P.; Beghelli, S.; Boninsegna, L.; Bettini, R.; Panzuto, F.; Pederzoli, P.; delle Fave, G.; Falconi, M. Pancreatic Endocrine Tumours: Improved TNM Staging and Histopathological Grading Permit a Clinically Efficient Prognostic Stratification of Patients. Mod. Pathol. 2010, 23, 824–833. [Google Scholar] [CrossRef] [PubMed]
- Boninsegna, L.; Panzuto, F.; Partelli, S.; Capelli, P.; Fave, G.D.; Bettini, R.; Pederzoli, P.; Scarpa, A.; Falconi, M. Malignant Pancreatic Neuroendocrine Tumour: Lymph Node Ratio and Ki67 Are Predictors of Recurrence after Curative Resections. Eur. J. Cancer 2012, 48, 1608–1615. [Google Scholar] [CrossRef] [PubMed]
- Ausania, F.; Senra Del Rio, P.; Gomez-Bravo, M.A.; Martin-Perez, E.; Pérez-Daga, J.A.; Dorcaratto, D.; González-Nicolás, T.; Sanchez-Cabus, S.; Tardio-Baiges, A. Can We Predict Recurrence in WHO G1-G2 Pancreatic Neuroendocrine Neoplasms? Results from a Multi-Institutional Spanish Study. Pancreatology 2019, 19, 367–371. [Google Scholar] [CrossRef] [PubMed]
- Carmona-Bayonas, A.; Jiménez-Fonseca, P.; Lamarca, Á.; Barriuso, J.; Castaño, Á.; Benavent, M.; Alonso, V.; Riesco-Martínez, M.D.C.; Alonso-Gordoa, T.; Custodio, A.; et al. Prediction of Progression-Free Survival in Patients With Advanced, Well-Differentiated, Neuroendocrine Tumours Being Treated With a Somatostatin Analog: The GETNE-TRASGU Study. J. Clin. Oncol. 2019, 37, 2571–2580. [Google Scholar] [CrossRef]
- Zhou, Y.; Li, D.; Lin, Y.; Yu, M.; Lu, X.; Jian, Z.; Na, N.; Hou, B. Pretreatment Hematologic Markers as Prognostic Predictors of Gastroenteropancreatic Neuroendocrine Tumours: A Systematic Review and Meta-Analysis. OncoTargets Ther. 2018, 11, 2489–2496. [Google Scholar] [CrossRef] [PubMed]
- Salman, T.; Kazaz, S.N.; Varol, U.; Oflazoglu, U.; Unek, I.T.; Kucukzeybek, Y.; Alacacioglu, A.; Atag, E.; Semiz, H.S.; Cengiz, H.; et al. Prognostic Value of the Pretreatment Neutrophil-to-Lymphocyte Ratio and Platelet-to-Lymphocyte Ratio for Patients with Neuroendocrine Tumours: An Izmir Oncology Group Study. Chemotherapy 2016, 61, 281–286. [Google Scholar] [CrossRef]
- Chan, D.L.; Yao, J.C.; Carnaghi, C.; Buzzoni, R.; Herbst, F.; Ridolfi, A.; Strosberg, J.; Kulke, M.H.; Pavel, M.; Singh, S. Markers of Systemic Inflammation in Neuroendocrine Tumours: A Pooled Analysis of the RADIANT-3 and RADIANT-4 Studies. Pancreas 2021, 50, 130–137. [Google Scholar] [CrossRef]
- Takakura, K.; Ito, Z.; Suka, M.; Kanai, T.; Matsumoto, Y.; Odahara, S.; Matsudaira, H.; Haruki, K.; Fujiwara, Y.; Saito, R.; et al. Comprehensive Assessment of the Prognosis of Pancreatic Cancer: Peripheral Blood Neutrophil-Lymphocyte Ratio and Immunohistochemical Analyses of the Tumour Site. Scand. J. Gastroenterol. 2016, 51, 610–617. [Google Scholar] [CrossRef]
- Coffelt, S.B.; Kersten, K.; Doornebal, C.W.; Weiden, J.; Vrijland, K.; Hau, C.-S.; Verstegen, N.J.M.; Ciampricotti, M.; Hawinkels, L.J.A.C.; Jonkers, J.; et al. IL-17-Producing Γδ T Cells and Neutrophils Conspire to Promote Breast Cancer Metastasis. Nature 2015, 522, 345–348. [Google Scholar] [CrossRef]
- Masucci, M.T.; Minopoli, M.; Carriero, M.V. Tumor Associated Neutrophils. Their Role in Tumorigenesis, Metastasis, Prognosis and Therapy. Front. Oncol. 2019, 9, 1146. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.-H.; Wang, W.-Q.; Gao, H.-L.; Xu, S.-S.; Li, S.; Li, T.-J.; Han, X.; Xu, H.-X.; Li, H.; Jiang, W.; et al. Tumour-Infiltrating Neutrophils Predict Poor Survival of Non-Functional Pancreatic Neuroendocrine Tumour. J. Clin. Endocrinol. Metab. 2020, 105. [Google Scholar] [CrossRef]
- da Silva, A.; Bowden, M.; Zhang, S.; Masugi, Y.; Thorner, A.R.; Herbert, Z.T.; Zhou, C.W.; Brais, L.; Chan, J.A.; Hodi, F.S.; et al. Characterisation of the Neuroendocrine Tumour Immune Microenvironment. Pancreas 2018, 47, 1123–1129. [Google Scholar] [CrossRef]
- de Reuver, P.R.; Mehta, S.; Gill, P.; Andrici, J.; D’Urso, L.; Clarkson, A.; Mittal, A.; Hugh, T.J.; Samra, J.S.; Gill, A.J. Immunoregulatory Forkhead Box Protein P3-Positive Lymphocytes Are Associated with Overall Survival in Patients with Pancreatic Neuroendocrine Tumours. J. Am. Coll. Surg. 2016, 222, 281–287. [Google Scholar] [CrossRef] [PubMed]
- Katz, S.C.; Donkor, C.; Glasgow, K.; Pillarisetty, V.G.; Gönen, M.; Espat, N.J.; Klimstra, D.S.; D’Angelica, M.I.; Allen, P.J.; Jarnagin, W.; et al. T Cell Infiltrate and Outcome Following Resection of Intermediate-Grade Primary Neuroendocrine Tumours and Liver Metastases. HPB 2010, 12, 674–683. [Google Scholar] [CrossRef] [PubMed]
- Cai, L.; Michelakos, T.; Deshpande, V.; Arora, K.S.; Yamada, T.; Ting, D.T.; Taylor, M.S.; Castillo, C.F.-D.; Warshaw, A.L.; Lillemoe, K.D.; et al. Role of Tumour-Associated Macrophages in the Clinical Course of Pancreatic Neuroendocrine Tumours (PanNETs). Clin. Cancer Res. 2019, 25, 2644–2655. [Google Scholar] [CrossRef]
- Takahashi, D.; Kojima, M.; Suzuki, T.; Sugimoto, M.; Kobayashi, S.; Takahashi, S.; Konishi, M.; Gotohda, N.; Ikeda, M.; Nakatsura, T.; et al. Profiling the Tumour Immune Microenvironment in Pancreatic Neuroendocrine Neoplasms with Multispectral Imaging Indicates Distinct Subpopulation Characteristics Concordant with WHO 2017 Classification. Sci. Rep. 2018, 8, 1–10. [Google Scholar] [CrossRef]
- Camous, L.; Roumenina, L.; Bigot, S.; Brachemi, S.; Frémeaux-Bacchi, V.; Lesavre, P.; Halbwachs-Mecarelli, L. Complement Alternative Pathway Acts as a Positive Feedback Amplification of Neutrophil Activation. Blood 2011, 117, 1340–1349. [Google Scholar] [CrossRef]
- Roumenina, L.T.; Daugan, M.V.; Noé, R.; Petitprez, F.; Vano, Y.A.; Sanchez-Salas, R.; Becht, E.; Meilleroux, J.; Clec’h, B.L.; Giraldo, N.A.; et al. Tumour Cells Hijack Macrophage-Produced Complement C1q to Promote Tumour Growth. Cancer Immunol. Res. 2019, 7, 1091–1105. [Google Scholar] [CrossRef]
- Yang, J.; Lin, P.; Yang, M.; Liu, W.; Fu, X.; Liu, D.; Tao, L.; Huo, Y.; Zhang, J.; Hua, R.; et al. Integrated Genomic and Transcriptomic Analysis Reveals Unique Characteristics of Hepatic Metastases and Pro-Metastatic Role of Complement C1q in Pancreatic Ductal Adenocarcinoma. Genome Biol. 2021, 22, 4. [Google Scholar] [CrossRef] [PubMed]
- Mehra, N.; Sharp, A.; Lorente, D.; Dolling, D.; Sumanasuriya, S.; Johnson, B.; Dearnaley, D.; Parker, C.; de Bono, J. Neutrophil to Lymphocyte Ratio in Castration-Resistant Prostate Cancer Patients Treated With Daily Oral Corticosteroids. Clin. Genitourin. Cancer 2017, 15, 678–684.e1. [Google Scholar] [CrossRef] [PubMed]
- Hsu, B.E.; Tabariès, S.; Johnson, R.M.; Andrzejewski, S.; Senecal, J.; Lehuédé, C.; Annis, M.G.; Ma, E.H.; Völs, S.; Ramsay, L.; et al. Immature Low-Density Neutrophils Exhibit Metabolic Flexibility That Facilitates Breast Cancer Liver Metastasis. Cell Rep. 2019, 27, 3902–3915.e6. [Google Scholar] [CrossRef] [PubMed]
- Gordon-Weeks, A.N.; Lim, S.Y.; Yuzhalin, A.E.; Jones, K.; Markelc, B.; Kim, K.J.; Buzzelli, J.N.; Fokas, E.; Cao, Y.; Smart, S.; et al. Neutrophils Promote Hepatic Metastasis Growth through Fibroblast Growth Factor 2-Dependent Angiogenesis in Mice. Hepatol. Baltim. Md 2017, 65, 1920–1935. [Google Scholar] [CrossRef] [PubMed]
- Mizuno, R.; Kawada, K.; Itatani, Y.; Ogawa, R.; Kiyasu, Y.; Sakai, Y. The Role of Tumour-Associated Neutrophils in Colorectal Cancer. Int. J. Mol. Sci. 2019, 20, 529. [Google Scholar] [CrossRef] [PubMed]


| Variable | n = 144 |
|---|---|
| Median age, years (range) | 56 (20–81) |
| Gender, male | 82 (57%) |
| BMI, kg/m2 | 25,7 (16.5–46.3) |
| Missing data | 19 |
| Symptoms at diagnosis, Yes | 80 (55.6%) |
| Functional tumor | 28 (19.4%) |
| MEN1 | 10 (6.9%) |
| Size, median in mm, range | 25 (5–120) |
| Missing data | 9 |
| Metastasis at diagnosis | 42 (29.2%) |
| Ki67, median % | 3 (1–20) |
| Grade 2 Grade 1 | 69 (48%) 75 (52%) |
| Size, T from AJCC 2017 | |
| T1 | 58 (40.3%) |
| T2 | 33 (22.9%) |
| T3 | 48 (33.3%) |
| T4 | 4 (2.8%) |
| Lymph node status, N1 | 50 (34.7%) |
| Missing data | 9 |
| Surgery | 129 (89.6%) |
| Surgical margins, R1 | 17 (11.8%) |
| Median NLR | 2,31 (0.99–14.05) |
| Median neutrophils count,/mm3 | 4245 (1370–14470) |
| Median lymphocytes count,/mm3 | 1695 (420–4040) |
| n = 144 | Univariate | Multivariate | |||||
|---|---|---|---|---|---|---|---|
| Variable | n | HR | CI 95% | p Value | HR | CI 95% | p Value |
| Age, >50 years | 98 | 1.88 | 0.693–5.106 | 0.22 | |||
| Sex, male | 82 | 2.46 | 0.908–6.676 | 0.08 | |||
| Ki67, continuous value | 144 | 1.03 | 0.958–1.111 | 0.41 | |||
| Tumor size, T3-T4 | 52 | 1.17 | 0.494–2.783 | 0.72 | |||
| Lymph node involvement | 50 | 3.27 | 1.315–8.117 | 0.01 | |||
| Metastasis | 42 | 3.32 | 1.417–7.766 | 0.006 | 3.35 | 1.1411–7.973 | 0.006 |
| NLR ≥ 4 | 27 | 3.53 | 1.502–8.313 | 0.004 | 2.57 | 1.061–6.216 | 0.036 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Debien, V.; Davidson, G.; Baltzinger, P.; Kurtz, J.-E.; Séverac, F.; Imperiale, A.; Pessaux, P.; Addeo, P.; Bachellier, P.; Su, X.; et al. Involvement of Neutrophils in Metastatic Evolution of Pancreatic Neuroendocrine Tumors. Cancers 2021, 13, 2771. https://doi.org/10.3390/cancers13112771
Debien V, Davidson G, Baltzinger P, Kurtz J-E, Séverac F, Imperiale A, Pessaux P, Addeo P, Bachellier P, Su X, et al. Involvement of Neutrophils in Metastatic Evolution of Pancreatic Neuroendocrine Tumors. Cancers. 2021; 13(11):2771. https://doi.org/10.3390/cancers13112771
Chicago/Turabian StyleDebien, Véronique, Guillaume Davidson, Philippe Baltzinger, Jean-Emmanuel Kurtz, François Séverac, Alessio Imperiale, Patrick Pessaux, Pietro Addeo, Philippe Bachellier, Xiaoping Su, and et al. 2021. "Involvement of Neutrophils in Metastatic Evolution of Pancreatic Neuroendocrine Tumors" Cancers 13, no. 11: 2771. https://doi.org/10.3390/cancers13112771
APA StyleDebien, V., Davidson, G., Baltzinger, P., Kurtz, J.-E., Séverac, F., Imperiale, A., Pessaux, P., Addeo, P., Bachellier, P., Su, X., Davidson, I., Chenard, M.-P., Goichot, B., & Malouf, G. G. (2021). Involvement of Neutrophils in Metastatic Evolution of Pancreatic Neuroendocrine Tumors. Cancers, 13(11), 2771. https://doi.org/10.3390/cancers13112771








