Unravelling the Encapsulation of DNA and Other Biomolecules in HAp Microcalcifications of Human Breast Cancer Tissues by Raman Imaging
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Breast Tumor Samples
2.2. Raman Spectroscopy
2.3. Multivariate Analysis
2.4. Breast Tumor Sample Staining for Confocal Microscopy Imaging
2.5. In Vitro Synthesis of DNA-HAp Particles
3. Results
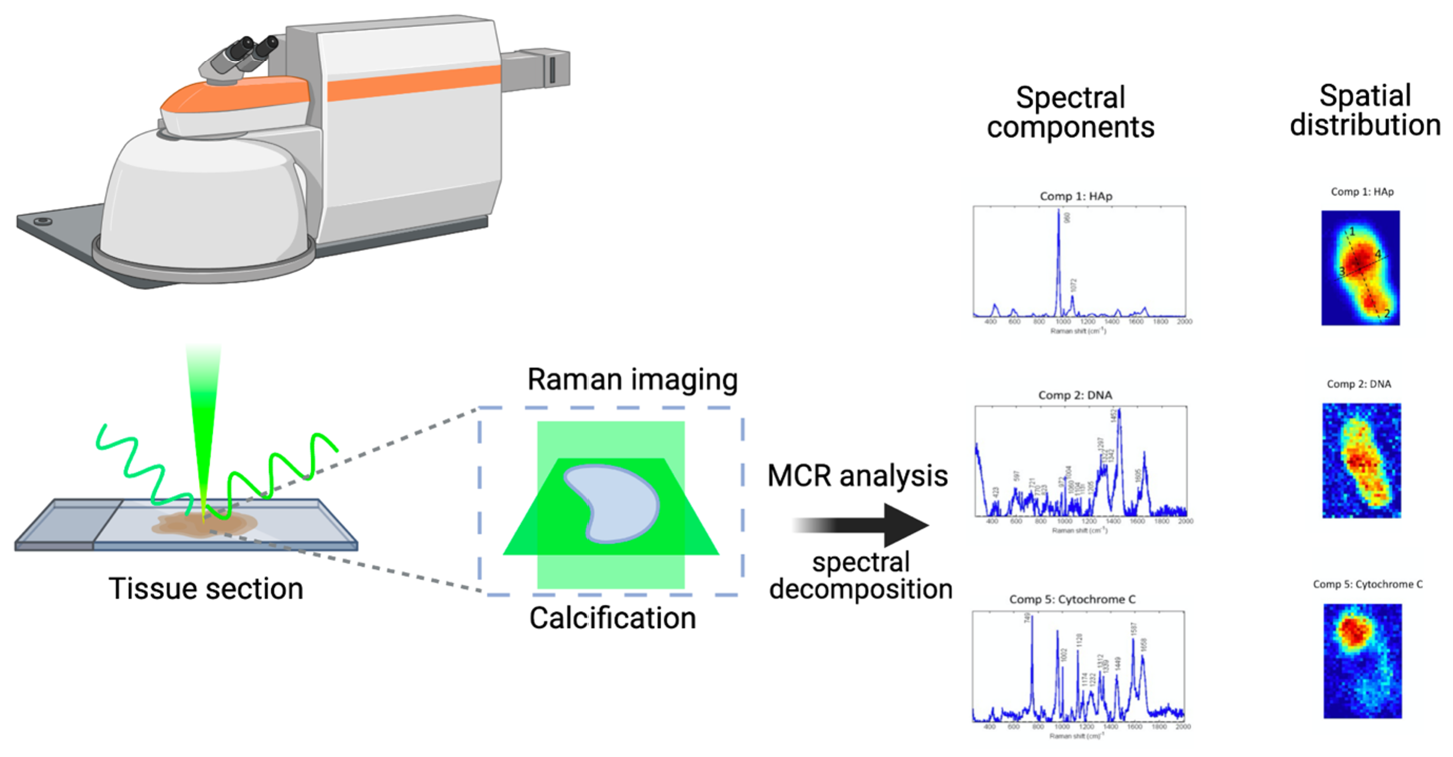
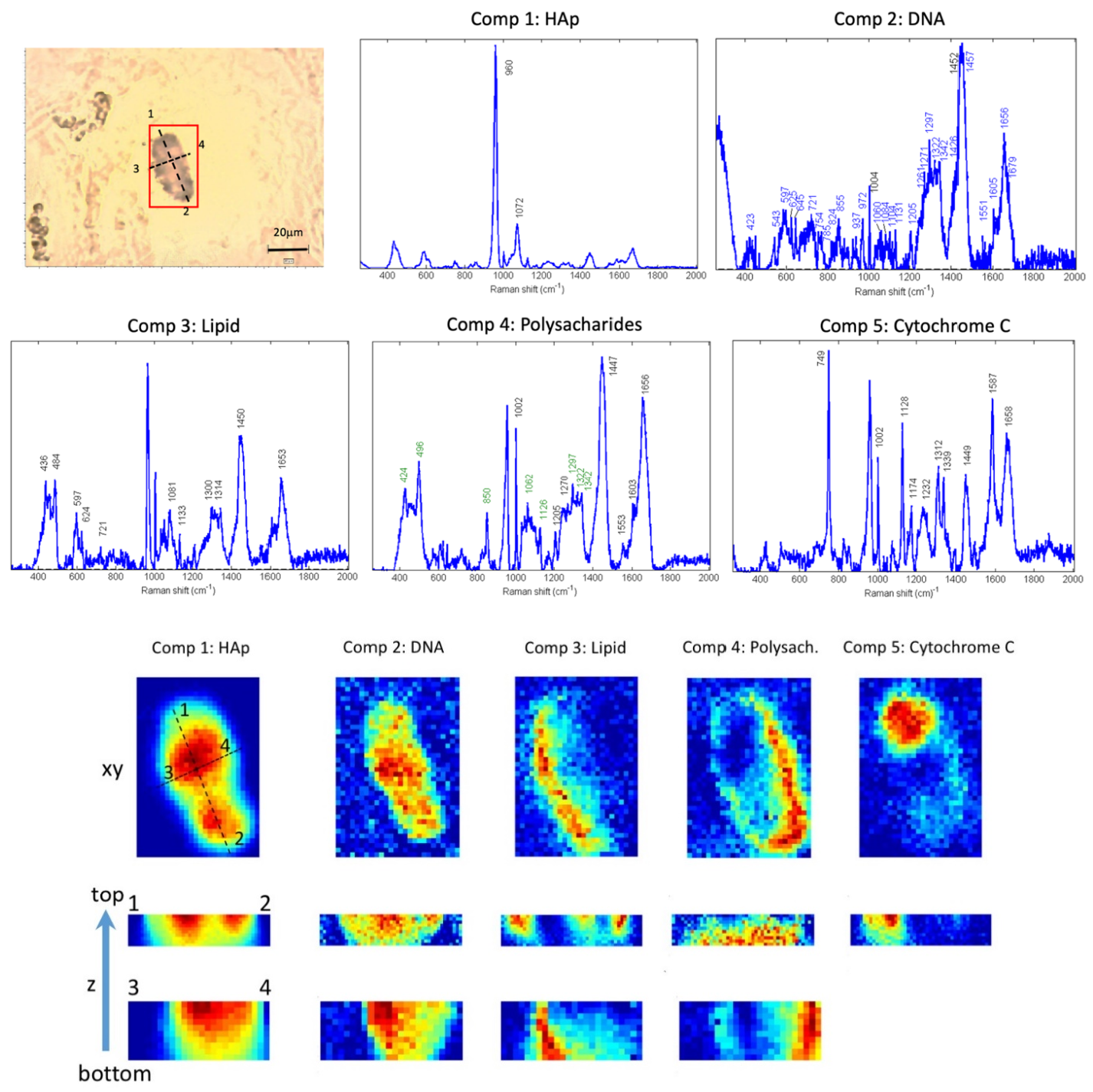
3.1. Molecular Raman Imaging and Advanced Multivariate Analysis of Tissue Calcifications
3.2. Study of DNA Mineralization in HAp Microcalcifications
3.2.1. Reference Raman Spectral Databases and Literature
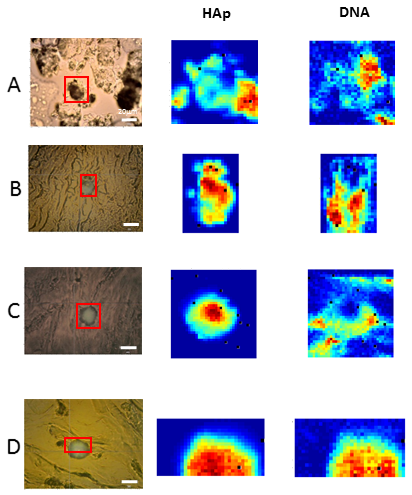
3.2.2. Identification of DNA in HAp Calcifications Found in Breast Cancer Tissues
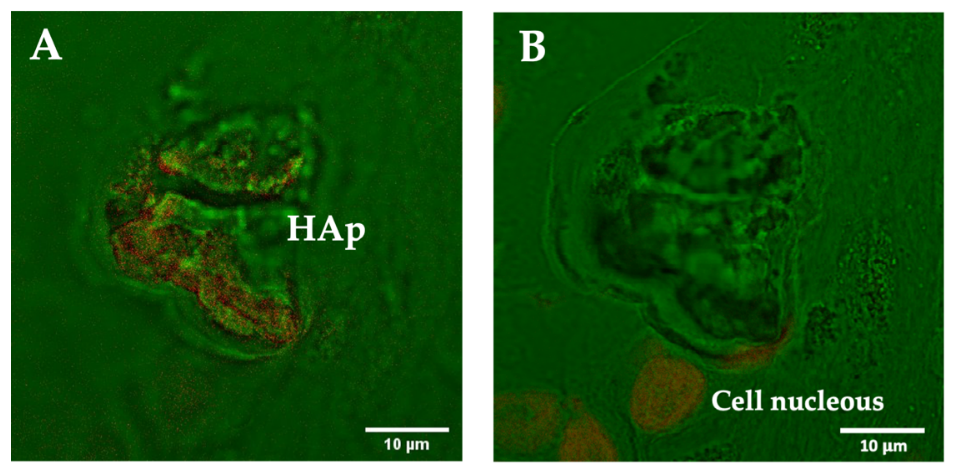
3.2.3. Fluorescence Staining of Microcalcifications with DAPI
3.3. Raman Images Reveal the Presence and Distribution of Biomolecules Inside HAp Calcifications
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global Cancer Statistics 2018: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Miller, K.D.; Siegel, R.L.; Lin, C.C.; Mariotto, A.B.; Kramer, J.L.; Rowland, J.H.; Stein, K.D.; Alteri, R.; Jemal, A. Cancer treatment and survivorship statistics, 2016. CA Cancer J. Clin. 2016, 66, 271–289. [Google Scholar] [CrossRef] [PubMed]
- DeSantis, C.E.; Lin, C.C.; Mariotto, A.B.; Siegel, R.L.; Stein, K.D.; Kramer, J.L.; Alteri, R.; Robbins, A.S.; Jemal, A. Cancer treatment and survivorship statistics, 2014. CA Cancer J. Clin. 2014, 64, 252–271. [Google Scholar] [CrossRef] [PubMed]
- DeSantis, C.E.; Ma, J.; Goding Sauer, A.; Newman, L.A.; Jemal, A. Breast cancer statistics, 2017, racial disparity in mortality by state. CA Cancer J. Clin. 2017, 67, 439–448. [Google Scholar] [CrossRef]
- Haka, A.S.; Shafer-Peltier, K.E.; Fitzmaurice, M.; Crowe, J.; Dasari, R.R.; Feld, M.S. Diagnosing breast cancer by using Raman spectroscopy. Proc. Natl. Acad. Sci. USA 2015, 102, 12371–12376. [Google Scholar] [CrossRef]
- Haka, A.S.; Shafer-Peltier, K.E.; Fitzmaurice, M.; Crowe, J.; Dasari, R.R.; Feld, M.S. Identifying microcalcifications in benign and malignant breast lesions by probing differences in their chemical composition using Raman spectroscopy. Cancer Res. 2002, 62, 5375–5380. [Google Scholar] [PubMed]
- Stone, N.; Baker, R.; Rogers, K.; Parker, A.W.; Matousek, P. Subsurface probing of calcifications with spatially offset Raman spectroscopy (SORS): Future possibilities for the diagnosis of breast cancer. Analyst 2007, 132, 899–905. [Google Scholar] [CrossRef] [PubMed]
- Gary, M.T.; Tan, P.H.; Cheung, H.S.; Chu, W.C.; Lam, W.W. Intermediate to highly suspicious calcification in breast lesions: A radio-pathologic correlation. Breast Cancer Res. Treat. 2008, 110, 1–7. [Google Scholar]
- Scott, R.; Kendall, C.; Stone, N.; Rogers, K. Elemental vs. phase composition of breast calcifications. Sci. Rep. 2017, 7, 136. [Google Scholar] [CrossRef]
- Frappart, L.; Boudeulle, M.; Boumendil, J.; Lin, H.C.; Martinon, I.; Palayer, C.; Mallet-Guy, Y.; Raudrant, D.; Bremond, A.; Feroldi, J. Structure and composition of microcalcifications in benign and malignant lesions of the breast: Study by light microscopy, transmission and scanning electron microscopy, microprobe analysis, and X-ray diffraction. Hum. Pathol. 1984, 15, 880–889. [Google Scholar] [CrossRef]
- Tornos, C.; Silva, E.; El-Naggar, A.; Pritzker, K.P.H. Calcium oxalate crystals in breast biopsies. Am. J. Surg. Pathol. 1990, 14, 961–968. [Google Scholar] [CrossRef]
- Radi, M.J. Calcium oxalate crystals in breast biopsies. An overlooked form of microcalcification associated with benign breast disease. Arch. Pathol. Lab. Med. 1989, 113, 1367–1369. [Google Scholar]
- Winston, J.S.; Yeh, I.T.; Evers, K.; Friedman, A.K. Calcium oxalate is associated with benign breast tissue: Can we avoid biopsy? Am. J. Clin. Pathol. 1993, 100, 488–492. [Google Scholar] [CrossRef]
- Yassin, N.I.R.; Omran, S.; El Houby, E.M.F.; Allam, H. Machine learning techniques for breast cancer computer aided diagnosis using different image modalities: A systematic review. Comput. Methods Programs Biomed. 2018, 156, 25–45. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Hauser, N.; Singer, G.; Trippel, M.; Kubik-Huch, R.A.; Schneider, C.W.; Stampanoni, M. Non-invasive classification of microcalcifications with phase-contrast X-ray mammography. Nat. Commun. 2014, 5, 3797. [Google Scholar] [CrossRef] [PubMed]
- Cox, R.F.; Hernandez-Santana, A.; Ramdass, S.; McMahon, G.; Harmey, J.H.; Morgan, M.P. Microcalcifications in breast cancer: Novel insights into the molecular mechanism and functional consequence of mammary mineralisation. Br. J. Cancer 2012, 106, 525–537. [Google Scholar] [CrossRef] [PubMed]
- Baker RRogers, K.D.; Shepherd, N.; Stone, N. New relationships between breast microcalcifications and cancer. Br. J. Cancer 2010, 103, 1034–1039. [Google Scholar] [CrossRef] [PubMed]
- Tabar, L.; Tony Chen, H.H.; Amy Yen, M.F.; Tot, T.; Tung, T.H.; Chen, L.S.; Duffy, S.W.; Smith, R.A. Mammographic tumor features can predict long-term outcomes reliably in women with 1–14-mm invasive breast carcinoma: Suggestions for the reconsideration of current therapeutic practice and the TNM classification system. Cancer Interdiscip. Int. J. Am. Cancer Soc. 2004, 101, 1745–1759. [Google Scholar] [CrossRef]
- Cox, R.F.; Morgan, M.P. Microcalcifications in breast cancer: Lessons from physiological mineralization. Bone 2013, 53, 437–450. [Google Scholar] [CrossRef]
- Scimeca, M.; Giannini, E.; Antonacci, C.; Pistolese, C.A.; Spagnoli, L.G.; Bonanno, E. Microcalcifications in breast cancer: An active phenomenon mediated by epithelial cells with mesenchymal characteristics. BMC Cancer 2014, 14, 286. [Google Scholar] [CrossRef]
- Sathyavathi, R.; Saha, A.; Soares, J.S.; Spegazzini, N.; McGee, S.; Dasari, R.R.; Fitzmaurice, M.; Barman, I. Raman spectroscopic sensing of carbonate intercalation in breast microcalcifications at stereotactic biopsy. Sci. Rep. 2015, 5, 9907. [Google Scholar] [CrossRef] [PubMed]
- Kerssens, M.M.; Matousek, P.; Rogers, K.; Stone, N. Towards a safe non-invasive method for evaluating the carbonate substitution levels of hydroxyapatite (HAP) in micro-calcifications found in breast tissue. Analyst 2010, 135, 3156–3161. [Google Scholar] [CrossRef] [PubMed]
- Castellaro, A.M.; Tonda, A.; Cejas, H.H.; Ferreyra, H.; Caputto, B.L.; Pucci, O.A.; Gil, G.A. Oxalate induces breast cancer. BMC Cancer 2015, 15, 761. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.Y.; Ouyang, J.M.; Xu, M. Synthesis, characterization, and cytotoxicity assay of calcium oxalate dihydrate crystals in various shapes. CrystEngComm 2016, 18, 5463–5473. [Google Scholar] [CrossRef]
- O’Grady, S.; Morgan, M.P. Microcalcifications in breast cancer: From pathophysiology to diagnosis and prognosis. BBA Rev. Cancer 2018, 1869, 310–320. [Google Scholar] [CrossRef]
- Revilla-López, G.; Rodríguez-Rivero, A.; Valle, L.; Puiggalí, J.; Turon, P.; Alemán, C. Biominerals Formed by DNA and Calcium Oxalate or Hydroxyapatite: A Comparative Study. Langmuir 2019, 3536, 11912–11922. [Google Scholar] [CrossRef]
- Kunitake, J.; Choi, S.; Nguyen, K.; Lee, M.; He, F.; Sudilovsky, D.; Morris, P.; Jochelson, M.; Hudis, C.; Muller, D.; et al. Correlative imaging reveals physiochemical heterogeneity of microcalcifications in human breast carcinomas. J. Struct. Biol. 2018, 202, 25–34. [Google Scholar] [CrossRef]
- Vanna, R.; Morasso, C.; Marcinno, B.; Piccotti, F.; Torti, E.; Altamura, D.; Albasini, S.; Agozzino, M.; Villani, L.; Sorrentino, L.; et al. Raman Spectroscopy Reveals That Biochemical Composition of Breast Microcalcifications Correlates with Histopathologic Features. Cancer Res. 2020, 80, 1762–1772. [Google Scholar] [CrossRef] [PubMed]
- Butler, H.J.; Ashton, L.; Bird, B.; Cinque, G.; Curtis, K.; Dorney, J. Using Raman spectroscopy to characterize biological materials. Nat. Protoc. 2016, 11, 664–687. [Google Scholar] [CrossRef]
- Marro, M.; Nieva, C.; Sanz-Pamplona, R.; Sierra, A. Molecular monitoring of epithelial-to-mesenchymal transition in breast cancer cells by means of Raman spectroscopy. Biochim. Biophys. Acta BBA Mol. Cell Res. 2014, 1843, 1785–1795. [Google Scholar] [CrossRef][Green Version]
- Marro, M.; Nieva, C.; de Juan, A.; Sierra, A. Unravelling the metabolic progression of breast cancer cells to bone metastasis by coupling Raman spectroscopy and a novel use of MCR-ALS algorithm. Anal. Chem. 2018, 90, 5594–5602. [Google Scholar] [CrossRef]
- Aschar-Sobbi, R.; Abramov, A.Y.; Diao, C.; Kargacin, M.E.; Kargacin, G.J.; French, R.J.; Pavlov, R. High Sensitivity, Quantitative Measurements of Polyphosphate Using a New DAPI-Based Approach. J. Fluoresc. 2008, 18, 859–866. [Google Scholar] [CrossRef] [PubMed]
- Gomes, F.M.; Ramos, I.B.; Wendt, C.; Girard-Dias, W.; De Souza, W.; Machado, E.A.; Miranda, E.A.K. New insights into the in situ microscopic visualization and quantification of inorganic polyphosphate stores by 4’, 6-diamidino-2-phenylindole (DAPI)-staining. Eur. J. Histochem. 2013, 57, e34. [Google Scholar]
- Bertran, O.; del Valle, L.J.; Revilla-López, G.; Chaves, G.; Cardús, L.; Casas, M.T.; Casanovas, J.; Turon, P.; Puiggalí JAlemán, C. Mineralization of DNA into nanoparticles of hydroxyapatite. Dalton Trans. 2014, 43, 317–327. [Google Scholar] [CrossRef] [PubMed]
- Revilla-López, G.; Casanovas, J.; Bertran, O.; Turon, P.; Puiggalí, J.; Alemán, C. Modeling biominerals formed by apatites and DNA. Biointerphases 2013, 8, 10. [Google Scholar] [CrossRef]
- Turon, P.; Puiggalí, J.; Bertrán, O.; Alemán, C. Surviving mass extinctions through biomineralized DNA. Chem. Eur. J. 2015, 21, 18892–18898. [Google Scholar] [CrossRef]
- Del Valle, L.J.; Bertran, O.; Chaves, G.; Revilla-López, G.; Rivas, M.; Casas, M.T.; Casanovas, J.; Turon, P.; Puiggalí, J.; Alemán, C. DNA adsorbed on hydroxyapatite surfaces. J. Mater. Chem. B 2014, 2, 6953–6966. [Google Scholar] [CrossRef]
- Graham, F.L.; van der Eb, A.J. A new technique for the assay of infectivity of human adenovirus 5 DNA. Virology 1973, 52, 456–467. [Google Scholar] [CrossRef]
- Eilers, P.H.; Boelens, H.F. Baseline correction with asymmetric least squares smoothing. Leiden Univ. Med. Cent. Rep. 2005, 1, 1–5. [Google Scholar]
- Talari, A.; Movasaghi, Z.; Rehman, S.; Rehman, I. Raman Spectroscopy of Biological Tissues. Appl. Spectrosc. Rev. 2015, 50, 46–111. [Google Scholar] [CrossRef]
- Czamara, K.; Majzner, K.; Pacia, M.Z.; Kochan, K.; Kaczora, A.; Baranskaa, M. Raman spectroscopy of lipids: A review. J. Raman Spectrosc. 2014, 46, 4–20. [Google Scholar] [CrossRef]
- Brazhe, N.; Evlyukhin, A.; Goodilin, E.; Semenova, A.; Novikov, S.; Bozhevolnyi, S.; Chichkov, B.; Sarycheva, A.; Baizhumano, A.; Nikelshparg, E.; et al. Probing Cytochrome C in living mitochondria with surface enhanced Raman spectroscopy. Sci. Rep. 2015, 5, 13793. [Google Scholar] [CrossRef] [PubMed]
- Gelder, J.; Gussem, K.; Vandenabeele, P.; Moens, L. Reference database of Raman spectra of biological molecules. J. Raman Spectrosc. 2007, 38, 1133–1147. [Google Scholar] [CrossRef]
- Sokolova, V.; Epple, M. Inorganic Nanoparticles as Carriers of Nucleic Acids into Cells. Angew. Chem. Int. Ed. 2008, 47, 1382–1395. [Google Scholar] [CrossRef]
- Bellahcene, A.; Castronovo, V. Increased expression of osteonectin and osteopontin, two bonematrix proteins, in human breast cancer. Am. J. Pathol. 1995, 146, 95–100. [Google Scholar] [PubMed]
- Lev-Toaff, A.S.; Feig, S.A.; Saitas, V.L.; Finkel, G.C.; Schwartz, G.F. Stability of malignant breast microcalcifications. Radiology 1994, 192, 153–156. [Google Scholar] [CrossRef]
- Eleftheriadis, T.; Pissas, G.; Liakopoulos, V.; Stefanidis, I. Cytochrome c as a Potentially Clinical Useful Marker of Mitochondrial and Cellular Damage. Front. Immunol. 2016, 7, 279. [Google Scholar] [CrossRef]

| Diagnosis | Oxalate | HAp | DNA | Size a | Shape b | Localisation c | |
|---|---|---|---|---|---|---|---|
| 1 | Infiltrating ductal carcinoma (IDC) | No | Yes | Yes | m, D = 20 µm | S | t |
| 2 | Infiltrating ductal carcinoma (IDC) | No | Yes | Yes | m, D = 20 µm | R | t |
| 3 | Infiltrating ductal carcinoma (IDC) | No | Yes | No | m, D = 10 µm | S | t |
| 3 | Infiltrating ductal carcinoma (IDC) | No | Yes | Yes | m | R | t |
| 4 | Infiltrating ductal carcinoma (IDC) | No | Yes | Yes | m, D = 20 µm | R | d |
| 5 | Infiltrating ductal carcinoma (IDC) | No | Yes | Yes | m, D = 20 µm | S | t |
| 5 | Infiltrating ductal carcinoma (IDC) | No | Yes | Yes | m, 30 × 20 µm | R | t |
| 6 | Infiltrating ductal carcinoma (IDC) | No | No | No | - | - | - |
| 7 | Infiltrating ductal carcinoma (IDC) | No | Yes | Yes | m, D = 10 µm | R | t |
| 8 | Infiltrating ductal carcinoma (IDC) | No | No | No | - | - | - |
| 9 | Infiltrating ductal carcinoma (IDC) | No | Yes | Yes | m, 25 × 15 µm | R | t |
| 9 | Infiltrating ductal carcinoma (IDC) | No | Yes | Yes | m, 20 µm | S | t |
| 10 | Infiltrating ductal carcinoma (IDC), multifocal | No | Yes | Yes | m, 30 µm | R | t |
| 11 | Infiltrating ductal carcinoma (IDC) | No | Yes | Yes | m | R | t |
| 12 | Infiltrating ductal carcinoma (IDC) | No | No | - | - | - | - |
| 13 | Infiltrating ductal carcinoma (IDC) | No | Yes | No | b | R | t |
| 14 | Infiltrating ductal carcinoma (IDC) | No | Yes | Yes (nc) | S | R | t |
| 15 | Infiltrating ductal carcinoma (IDC) | No | Yes | Yes | b | R | t |
| 16 | Infiltrating ductal carcinoma (IDC) | No | Yes | Yes | m, 100 µm | R | d |
| 17 | Infiltrating ductal carcinoma (IDC) | No | Yes | Yes | m, 40 × 50 µm | R | t |
| 18 | Infiltrating ductal carcinoma (IDC) | No | Yes | Yes | s, D = 7 µm | S | t |
| 19 | Infiltrating ductal carcinoma (IDC) | No | Yes | Yes | m | R | t |
| 20 | Infiltrating ductal carcinoma (IDC) | No | Yes | Yes | m | R | d |
| 21 | Infiltrating ductal carcinoma (IDC) | No | Yes | Yes | m | R | d |
| 22 | Infiltrating ductal carcinoma (IDC) | No | Yes | Yes | m, 20 × 10 µm | R | t |
| 23 | Infiltrating ductal carcinoma (IDC) | No | Yes | Yes | b | R | t |
| 24 | Infiltrating lobular carcinoma (ILC) | No | Yes | Yes | m, D = 20 µm | R | t |
| 25 | Infiltrating lobular carcinoma (ILC) | Yes | No | No | s | S | t |
| 25 | Infiltrating lobular carcinoma (ILC) | No | Yes | Yes | m | R | t |
| 26 | Infiltrating lobular carcinoma (ILC) | No | Yes | Yes | m | R | t |
| Component 2: DNA | |||
|---|---|---|---|
| Band/cm−1 | Assignment | Band/cm−1 | Assignment |
| 1679 | T, DNA base | 1084 | Phosphodiester groups in nucleic acids |
| 1656 | T, G, C (ring breathing modes of the DNA/RNA bases); amide I (protein); C=C lipids | 1060 | PO2-stretching of DNA |
| 1605 | Cytosine (NH2) | 1004 | Phenylalanine |
| 1551 | Guanine | 972 | T, ribose, Phosphate monoester groups of phosphorylated proteins and cellular nucleic acids |
| 1457 | A, C, T DNA bases | 937 | G, A DNA bases |
| 1452 | CH2 CH3 deformation | 855 | Phosphate group |
| 1426 | Deoxyribose (B, Z marker) | 824 | O-P-O stretch DNA |
| 1342 | A, G DNA bases | 785 | U, T, C, O-P-O |
| 1322 | G, DNA base | 754 | T, DNA |
| 1297 | C, palmitic acid, CH2 | 721 | A, DNA base |
| 1271 | C, G DNA bases | 645 | G DNA base |
| 1261 | A, G, C DNA bases | 625 | A, T DNA bases |
| 1205 | A, T, ring breathing mode of DNA bases | 597 | C DNA base |
| 1131 | A DNA base | 543 | C DNA base |
| 1104 | O-P-O backbone stretch DNA, U | 423 | T, U DNA bases |
| Component 3: Lipid | Component 4: Polysaccharides and Proteins | Component 5: Cytochrome C | |||
|---|---|---|---|---|---|
| Band/cm−1 | Assignment | Band/cm−1 | Assignment | Band/cm−1 | Assignment |
| 436 | Cholesterol, cholesterol ester | 424 | Polysaccharide | 749 | Cytochrome C |
| 597 | Phosphatidylinositol | 496 | Glycogen | 1002 | Phenylalanine |
| 624 | Cholesterol ester | 850 | Polysaccharide | 1128 | Cytochrome C |
| 721 | Phosphatidylcholine, Sphingomyelin | 1002 | Phenylalanine | 1174 | CH tyrosine phenylalanine |
| 1081 | Phosphatidylcholine, membrane lipids | 1062 | C-C carbohydrates | 1232 | Amide III |
| 1133 | Membrane lipids (PC, PE, SM) | 1126 | Disaccharides, glucose, sucrose, C-O stretching carbohydrates | 1312 | Cytochrome C |
| 1300 | Membrane lipids (PC, PE) | 1205 | Amide III proteins | 1339 | tryptophan |
| 1314 | CH3CH2 twisting of lipid | 1270 | Amide III proteins | 1449 | C-H proteins |
| 1450 | CH (lipids and proteins) | 1297 | CH2 deformation | 1587 | Cytochrome C |
| 1653 | C=C lipids, proteins | 1322 | CH deformation proteins | 1658 | Amide I proteins |
| 1342 | CH deformation protein and carbohydrates | ||||
| 1447 | CH2 deformation proteins | ||||
| 1553 | Tryptophan, Amide II | ||||
| 1603 | Phenylalanine | ||||
| 1656 | Amide I proteins | ||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Marro, M.; Rodríguez-Rivero, A.M.; Araujo-Andrade, C.; Fernández-Figueras, M.T.; Pérez-Roca, L.; Castellà, E.; Navinés, J.; Mariscal, A.; Julián, J.F.; Turon, P.; et al. Unravelling the Encapsulation of DNA and Other Biomolecules in HAp Microcalcifications of Human Breast Cancer Tissues by Raman Imaging. Cancers 2021, 13, 2658. https://doi.org/10.3390/cancers13112658
Marro M, Rodríguez-Rivero AM, Araujo-Andrade C, Fernández-Figueras MT, Pérez-Roca L, Castellà E, Navinés J, Mariscal A, Julián JF, Turon P, et al. Unravelling the Encapsulation of DNA and Other Biomolecules in HAp Microcalcifications of Human Breast Cancer Tissues by Raman Imaging. Cancers. 2021; 13(11):2658. https://doi.org/10.3390/cancers13112658
Chicago/Turabian StyleMarro, Monica, Anna M. Rodríguez-Rivero, Cuauhtémoc Araujo-Andrade, Maria Teresa Fernández-Figueras, Laia Pérez-Roca, Eva Castellà, Jordi Navinés, Antonio Mariscal, Joan Francesc Julián, Pau Turon, and et al. 2021. "Unravelling the Encapsulation of DNA and Other Biomolecules in HAp Microcalcifications of Human Breast Cancer Tissues by Raman Imaging" Cancers 13, no. 11: 2658. https://doi.org/10.3390/cancers13112658
APA StyleMarro, M., Rodríguez-Rivero, A. M., Araujo-Andrade, C., Fernández-Figueras, M. T., Pérez-Roca, L., Castellà, E., Navinés, J., Mariscal, A., Julián, J. F., Turon, P., & Loza-Alvarez, P. (2021). Unravelling the Encapsulation of DNA and Other Biomolecules in HAp Microcalcifications of Human Breast Cancer Tissues by Raman Imaging. Cancers, 13(11), 2658. https://doi.org/10.3390/cancers13112658








