Molecular and Clinical Features of Hospital Admissions in Patients with Thoracic Malignancies on Immune Checkpoint Inhibitors
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Clinical and Molecular Information Collection
2.3. Statistical Analysis
3. Results
3.1. Patients Characteristics
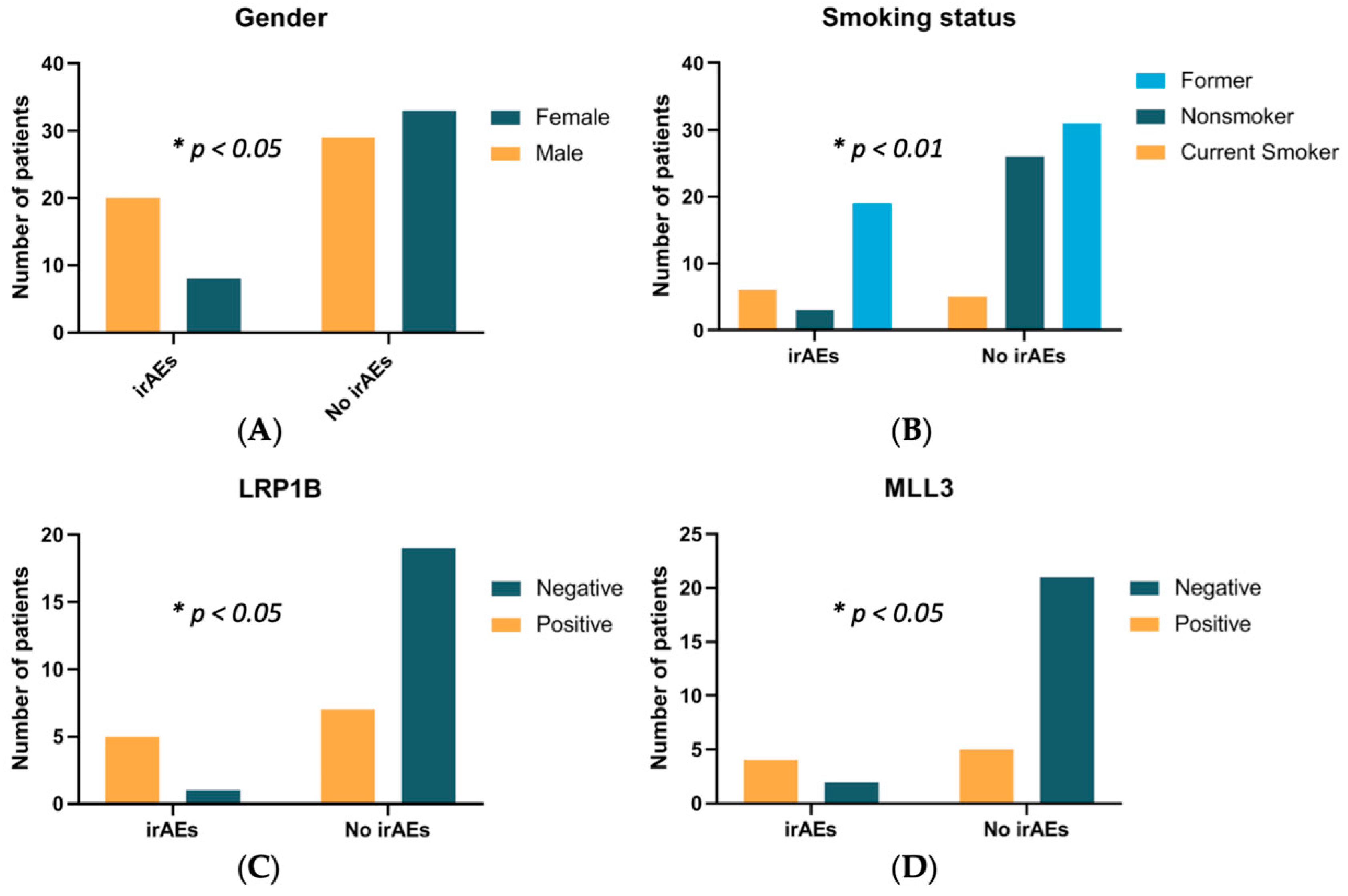
3.2. Clinical Features in irAEs and Non-irAE Population
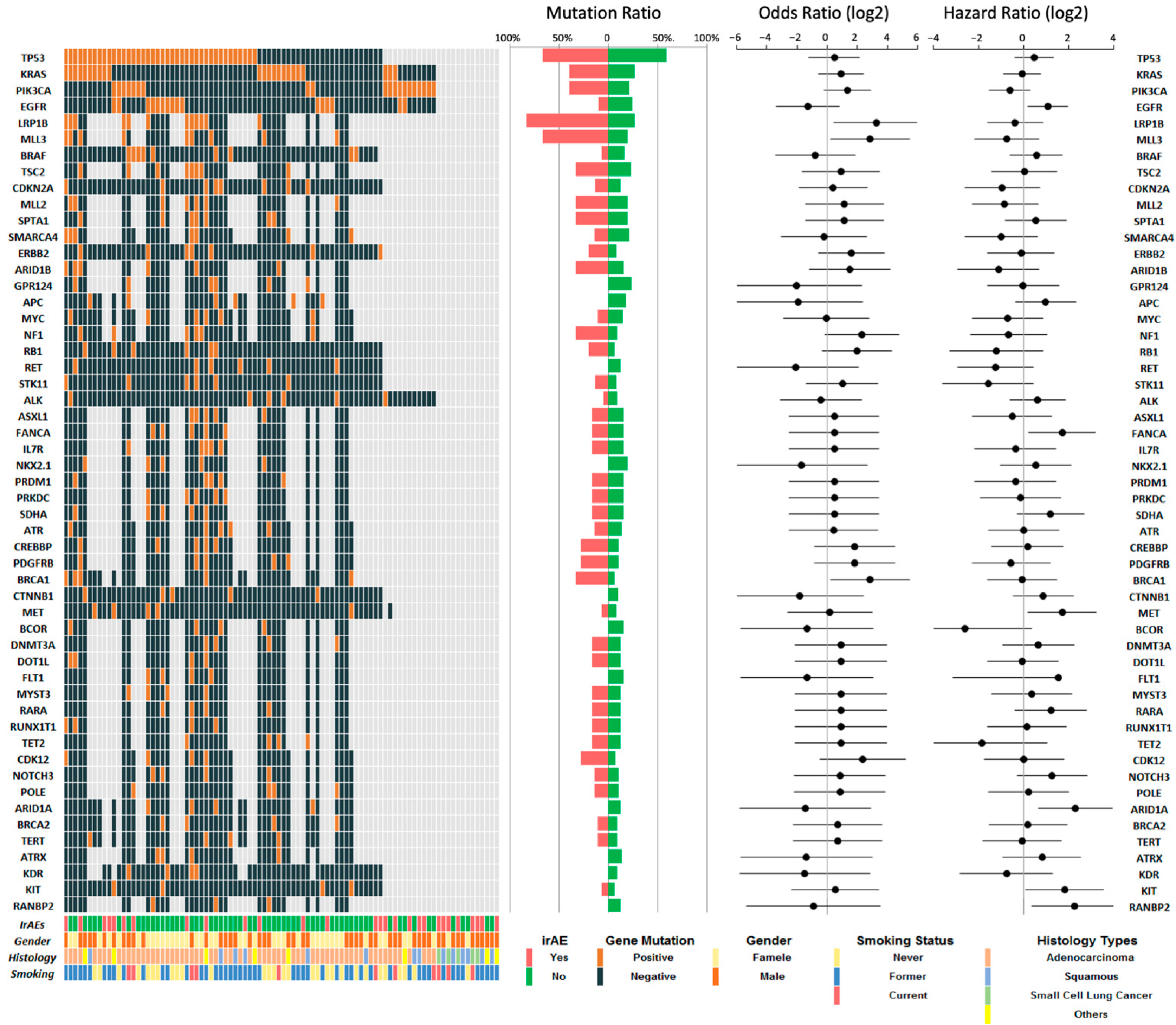
3.3. Molecular Features in irAE and Non-irAE Population
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| CI | Confidence interval |
| CTLA-4 | Cytotoxic T-lymphocyte–associated antigen 4 |
| DCR | Disease control rate |
| EGD | Esophagogastroduodenoscopy |
| FANCA | Fanconi anemia complementation group A |
| FDA | Food and Drug Administration |
| GAs | Genomic alterations |
| HGF | Hepatocyte growth factor |
| HR | Hazard ratio |
| ICIs | Immune checkpoint inhibitors |
| IHC | Immunohistochemistry |
| IrAEs | Immune-related adverse events |
| IRB | Institutional review board |
| KMT2C | Lysine (K) methyltransferase 2C |
| LRP1B | Low-density lipoprotein (LDL) receptor-related protein 1B |
| LOS | Length of stay |
| MLL3 | Mixed-lineage leukemia protein 3 |
| NGS | Next-generation sequencing |
| NSCC-NOS | Non-small cell carcinoma, not otherwise specified |
| NSCLC | Non-small cell lung cancer |
| OR | Odds ratio |
| ORR | Overall response rate |
| OS | Overall survival |
| PD-1 | Program death -1 |
| PD-L1 | Programmed death-ligand 1 |
| PFS | Progression-free survival |
| SCLC | Small cell lung cancer |
| TMB | Tumor mutation burden |
| VS | Versus |
References
- Mok, T.S.K.; Wu, Y.L.; Kudaba, I.; Kowalski, D.M.; Cho, B.C.; Turna, H.Z.; Castro, G., Jr.; Srimuninnimit, V.; Laktionov, K.K.; Bondarenko, I.; et al. Pembrolizumab versus chemotherapy for previously untreated, PD-L1-expressing, locally advanced or metastatic non-small-cell lung cancer (KEYNOTE-042): A randomised, open-label, controlled, phase 3 trial. Lancet 2019, 393, 1819–1830. [Google Scholar] [CrossRef]
- Gandhi, L.; Rodriguez-Abreu, D.; Gadgeel, S.; Esteban, E.; Felip, E.; De Angelis, F.; Domine, M.; Clingan, P.; Hochmair, M.J.; Powell, S.F.; et al. Pembrolizumab plus Chemotherapy in Metastatic Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2018, 378, 2078–2092. [Google Scholar] [CrossRef] [PubMed]
- Paz-Ares, L.; Luft, A.; Vicente, D.; Tafreshi, A.; Gumus, M.; Mazieres, J.; Hermes, B.; Cay Senler, F.; Csoszi, T.; Fulop, A.; et al. Pembrolizumab plus Chemotherapy for Squamous Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2018, 379, 2040–2051. [Google Scholar] [CrossRef]
- Reck, M.; Rodríguez-Abreu, D.; Robinson, A.G.; Hui, R.; Csőszi, T.; Fülöp, A.; Gottfried, M.; Peled, N.; Tafreshi, A.; Cuffe, S.; et al. Pembrolizumab versus chemotherapy for PD-L1–positive non–small-cell lung cancer. N. Engl. J. Med. 2016, 375, 1823–1833. [Google Scholar] [CrossRef] [PubMed]
- Horn, L.; Mansfield, A.S.; Szczesna, A.; Havel, L.; Krzakowski, M.; Hochmair, M.J.; Huemer, F.; Losonczy, G.; Johnson, M.L.; Nishio, M.; et al. First-Line Atezolizumab plus Chemotherapy in Extensive-Stage Small-Cell Lung Cancer. N. Engl. J. Med. 2018, 379, 2220–2229. [Google Scholar] [CrossRef]
- Socinski, M.A.; Jotte, R.M.; Cappuzzo, F.; Orlandi, F.; Stroyakovskiy, D.; Nogami, N.; Rodriguez-Abreu, D.; Moro-Sibilot, D.; Thomas, C.A.; Barlesi, F.; et al. Atezolizumab for First-Line Treatment of Metastatic Nonsquamous NSCLC. N. Engl. J. Med. 2018, 378, 2288–2301. [Google Scholar] [CrossRef]
- Paz-Ares, L.; Dvorkin, M.; Chen, Y.; Reinmuth, N.; Hotta, K.; Trukhin, D.; Statsenko, G.; Hochmair, M.J.; Özgüroğlu, M.; Ji, J.H.; et al. Durvalumab plus platinum–etoposide versus platinum–etoposide in first-line treatment of extensive-stage small-cell lung cancer (CASPIAN): A randomised, controlled, open-label, phase 3 trial. Lancet 2019, 394, 1929–1939. [Google Scholar] [CrossRef]
- Antonia, S.J.; Lopez-Martin, J.A.; Bendell, J.; Ott, P.A.; Taylor, M.; Eder, J.P.; Jager, D.; Pietanza, M.C.; Le, D.T.; de Braud, F.; et al. Nivolumab alone and nivolumab plus ipilimumab in recurrent small-cell lung cancer (CheckMate 032): A multicentre, open-label, phase 1/2 trial. Lancet Oncol. 2016, 17, 883–895. [Google Scholar] [CrossRef]
- Brahmer, J.; Reckamp, K.L.; Baas, P.; Crino, L.; Eberhardt, W.E.; Poddubskaya, E.; Antonia, S.; Pluzanski, A.; Vokes, E.E.; Holgado, E.; et al. Nivolumab versus Docetaxel in Advanced Squamous-Cell Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2015, 373, 123–135. [Google Scholar] [CrossRef]
- Brahmer, J.R.; Govindan, R.; Anders, R.A.; Antonia, S.J.; Sagorsky, S.; Davies, M.J.; Dubinett, S.M.; Ferris, A.; Gandhi, L.; Garon, E.B.; et al. The Society for Immunotherapy of Cancer consensus statement on immunotherapy for the treatment of non-small cell lung cancer (NSCLC). J. Immunother. Cancer 2018, 6, 75. [Google Scholar] [CrossRef]
- Hellmann, M.D.; Paz-Ares, L.; Bernabe Caro, R.; Zurawski, B.; Kim, S.-W.; Carcereny Costa, E.; Park, K.; Alexandru, A.; Lupinacci, L.; de la Mora Jimenez, E.; et al. Nivolumab plus Ipilimumab in Advanced Non‚ÄìSmall-Cell Lung Cancer. N. Engl. J. Med. 2019, 381, 2020–2031. [Google Scholar] [CrossRef] [PubMed]
- Reck, M.; Ciuleanu, T.-E.; Dols, M.C.; Schenker, M.; Zurawski, B.; Menezes, J.; Richardet, E.; Bennouna, J.; Felip, E.; Juan-Vidal, O.; et al. Nivolumab (NIVO) + ipilimumab (IPI) + 2 cycles of platinum-doublet chemotherapy (chemo) vs. 4 cycles chemo as first-line (1L) treatment (tx) for stage IV/recurrent non-small cell lung cancer (NSCLC): CheckMate 9LA. J. Clin. Oncol. 2020, 38, 9501. [Google Scholar] [CrossRef]
- Antonia, S.J.; Villegas, A.; Daniel, D.; Vicente, D.; Murakami, S.; Hui, R.; Yokoi, T.; Chiappori, A.; Lee, K.H.; de Wit, M.; et al. Durvalumab after Chemoradiotherapy in Stage III Non-Small-Cell Lung Cancer. N. Engl. J. Med. 2017, 377, 1919–1929. [Google Scholar] [CrossRef]
- Forde, P.M.; Chaft, J.E.; Smith, K.N.; Anagnostou, V.; Cottrell, T.R.; Hellmann, M.D.; Zahurak, M.; Yang, S.C.; Jones, D.R.; Broderick, S.; et al. Neoadjuvant PD-1 Blockade in Resectable Lung Cancer. N. Engl. J. Med. 2018, 378, 1976–1986. [Google Scholar] [CrossRef] [PubMed]
- Postow, M.A.; Sidlow, R.; Hellmann, M.D. Immune-Related Adverse Events Associated with Immune Checkpoint Blockade. N. Engl. J. Med. 2018, 378, 158–168. [Google Scholar] [CrossRef] [PubMed]
- Weber, J.S.; Kahler, K.C.; Hauschild, A. Management of immune-related adverse events and kinetics of response with ipilimumab. J. Clin. Oncol. 2012, 30, 2691–2697. [Google Scholar] [CrossRef]
- Arnaud-Coffin, P.; Maillet, D.; Gan, H.K.; Stelmes, J.J.; You, B.; Dalle, S.; Peron, J. A systematic review of adverse events in randomized trials assessing immune checkpoint inhibitors. Int. J. Cancer 2019, 145, 639–648. [Google Scholar] [CrossRef]
- Wang, D.Y.; Salem, J.E.; Cohen, J.V.; Chandra, S.; Menzer, C.; Ye, F.; Zhao, S.; Das, S.; Beckermann, K.E.; Ha, L.; et al. Fatal Toxic Effects Associated With Immune Checkpoint Inhibitors: A Systematic Review and Meta-analysis. JAMA Oncol. 2018, 4, 1721–1728. [Google Scholar] [CrossRef]
- Thompson, J.A.; Schneider, B.J.; Brahmer, J.; Andrews, S.; Armand, P.; Bhatia, S.; Budde, L.E.; Costa, L.; Davies, M.; Dunnington, D.; et al. Management of Immunotherapy-Related Toxicities, Version 1.2019. J. Natl. Compr. Cancer Netw. 2019, 17, 255–289. [Google Scholar] [CrossRef]
- Puzanov, I.; Diab, A.; Abdallah, K.; Bingham, C.O., 3rd; Brogdon, C.; Dadu, R.; Hamad, L.; Kim, S.; Lacouture, M.E.; LeBoeuf, N.R.; et al. Managing toxicities associated with immune checkpoint inhibitors: Consensus recommendations from the Society for Immunotherapy of Cancer (SITC) Toxicity Management Working Group. J. Immunother. Cancer 2017, 5, 95. [Google Scholar] [CrossRef]
- Brahmer, J.R.; Lacchetti, C.; Schneider, B.J.; Atkins, M.B.; Brassil, K.J.; Caterino, J.M.; Chau, I.; Ernstoff, M.S.; Gardner, J.M.; Ginex, P.; et al. Management of Immune-Related Adverse Events in Patients Treated With Immune Checkpoint Inhibitor Therapy: American Society of Clinical Oncology Clinical Practice Guideline. J. Clin. Oncol. 2018, 36, 1714–1768. [Google Scholar] [CrossRef]
- Hellmann, M.D.; Nathanson, T.; Rizvi, H.; Creelan, B.C.; Sanchez-Vega, F.; Ahuja, A.; Ni, A.; Novik, J.B.; Mangarin, L.M.B.; Abu-Akeel, M.; et al. Genomic Features of Response to Combination Immunotherapy in Patients with Advanced Non-Small-Cell Lung Cancer. Cancer Cell 2018, 33, 843–852.e4. [Google Scholar] [CrossRef]
- Skoulidis, F.; Goldberg, M.E.; Greenawalt, D.M.; Hellmann, M.D.; Awad, M.M.; Gainor, J.F.; Schrock, A.B.; Hartmaier, R.J.; Trabucco, S.E.; Gay, L.; et al. STK11/LKB1 Mutations and PD-1 Inhibitor Resistance in KRAS-Mutant Lung Adenocarcinoma. Cancer Discov. 2018, 8, 822–835. [Google Scholar] [CrossRef]
- Mazieres, J.; Drilon, A.; Lusque, A.; Mhanna, L.; Cortot, A.B.; Mezquita, L.; Thai, A.A.; Mascaux, C.; Couraud, S.; Veillon, R.; et al. Immune checkpoint inhibitors for patients with advanced lung cancer and oncogenic driver alterations: Results from the IMMUNOTARGET registry. Ann. Oncol. 2019, 30, 1321–1328. [Google Scholar] [CrossRef]
- Schoenfeld, A.J.; Arbour, K.C.; Rizvi, H.; Iqbal, A.N.; Gadgeel, S.M.; Girshman, J.; Kris, M.G.; Riely, G.J.; Yu, H.A.; Hellmann, M.D. Severe immune-related adverse events are common with sequential PD-(L)1 blockade and osimertinib. Ann. Oncol. 2019, 30, 839–844. [Google Scholar] [CrossRef] [PubMed]
- June, C.H.; Warshauer, J.T.; Bluestone, J.A. Is autoimmunity the Achilles' heel of cancer immunotherapy? Nat. Med. 2017, 23, 540–547. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Core Team: Vienna, Austria, 2019. [Google Scholar]
- Garon, E.B.; Hellmann, M.D.; Rizvi, N.A.; Carcereny, E.; Leighl, N.B.; Ahn, M.J.; Eder, J.P.; Balmanoukian, A.S.; Aggarwal, C.; Horn, L.; et al. Five-Year Overall Survival for Patients With Advanced NonSmall-Cell Lung Cancer Treated With Pembrolizumab: Results From the Phase I KEYNOTE-001 Study. J. Clin. Oncol. 2019, 37, 2518. [Google Scholar] [CrossRef] [PubMed]
- Topalian, S.L.; Hodi, F.S.; Brahmer, J.R.; Gettinger, S.N.; Smith, D.C.; McDermott, D.F.; Powderly, J.D.; Sosman, J.A.; Atkins, M.B.; Leming, P.D.; et al. Five-Year Survival and Correlates Among Patients With Advanced Melanoma, Renal Cell Carcinoma, or Non-Small Cell Lung Cancer Treated With Nivolumab. JAMA Oncol. 2019, 5, 1411–1420. [Google Scholar] [CrossRef]
- Parikh, A.B.; Zhong, X.; Mellgard, G.; Qin, Q.; Patel, V.G.; Wang, B.; Alerasool, P.; Garcia, P.; Leiter, A.; Gallagher, E.J.; et al. Risk Factors for Emergency Room and Hospital Care Among Patients With Solid Tumors on Immune Checkpoint Inhibitor Therapy. Am. J. Clin. Oncol. 2021, 44, 114–120. [Google Scholar] [CrossRef]
- Sato, K.; Akamatsu, H.; Murakami, E.; Sasaki, S.; Kanai, K.; Hayata, A.; Tokudome, N.; Akamatsu, K.; Koh, Y.; Ueda, H.; et al. Correlation between immune-related adverse events and efficacy in non-small cell lung cancer treated with nivolumab. Lung Cancer 2018, 115, 71–74. [Google Scholar] [CrossRef]
- Sattar, J.; Kartolo, A.; Hopman, W.M.; Lakoff, J.M.; Baetz, T. The efficacy and toxicity of immune checkpoint inhibitors in a real-world older patient population. J. Geriatr. Oncol. 2018. [Google Scholar] [CrossRef]
- Ricciuti, B.; Genova, C.; De Giglio, A.; Bassanelli, M.; Dal Bello, M.G.; Metro, G.; Brambilla, M.; Baglivo, S.; Grossi, F.; Chiari, R. Impact of immune-related adverse events on survival in patients with advanced non-small cell lung cancer treated with nivolumab: Long-term outcomes from a multi-institutional analysis. J. Cancer Res. Clin. Oncol. 2019, 145, 479–485. [Google Scholar] [CrossRef]
- Ahn, B.C.; Pyo, K.H.; Xin, C.F.; Jung, D.; Shim, H.S.; Lee, C.Y.; Park, S.Y.; Yoon, H.I.; Hong, M.H.; Cho, B.C.; et al. Comprehensive analysis of the characteristics and treatment outcomes of patients with non-small cell lung cancer treated with anti-PD-1 therapy in real-world practice. J. Cancer Res. Clin. Oncol. 2019, 145, 1613–1623. [Google Scholar] [CrossRef]
- Haratani, K.; Hayashi, H.; Chiba, Y.; Kudo, K.; Yonesaka, K.; Kato, R.; Kaneda, H.; Hasegawa, Y.; Tanaka, K.; Takeda, M.; et al. Association of Immune-Related Adverse Events With Nivolumab Efficacy in Non-Small-Cell Lung Cancer. JAMA Oncol. 2018, 4, 374–378. [Google Scholar] [CrossRef]
- Grangeon, M.; Tomasini, P.; Chaleat, S.; Jeanson, A.; Souquet-Bressand, M.; Khobta, N.; Bermudez, J.; Trigui, Y.; Greillier, L.; Blanchon, M.; et al. Association Between Immune-related Adverse Events and Efficacy of Immune Checkpoint Inhibitors in Non-small-cell Lung Cancer. Clin. Lung Cancer 2019, 20, 201–207. [Google Scholar] [CrossRef] [PubMed]
- Shafqat, H.; Gourdin, T.; Sion, A. Immune-related adverse events are linked with improved progression-free survival in patients receiving anti-PD-1/PD-L1 therapy. Semin. Oncol. 2018, 45, 156–163. [Google Scholar] [CrossRef] [PubMed]
- Durbin, S.M.; Zubiri, L.; Niemierko, A.; Bardia, A.; Sullivan, R.J.; McEwen, C.; Mulvey, T.M.; Allen, I.M.; Lawrence, D.P.; Cohen, J.V.; et al. Clinical Outcomes of Patients with Metastatic Cancer Receiving Immune Checkpoint Inhibitors in the Inpatient Setting. Oncologist 2021, 26, 49–55. [Google Scholar] [CrossRef]
- Chen, M.; Li, Q.; Xu, Y.; Zhao, J.; Zhang, L.; Wei, L.; Zhong, W.; Wang, M. Immunotherapy as second-line treatment and beyond for non-small cell lung cancer in a single center of China: Outcomes, toxicities, and clinical predictive factors from a real-world retrospective analysis. Thorac. Cancer 2020, 11, 1955–1962. [Google Scholar] [CrossRef]
- Ksienski, D.; Wai, E.S.; Croteau, N.; Fiorino, L.; Brooks, E.; Poonja, Z.; Fenton, D.; Geller, G.; Glick, D.; Lesperance, M. Efficacy of Nivolumab and Pembrolizumab in Patients With Advanced Non-Small-Cell Lung Cancer Needing Treatment Interruption Because of Adverse Events: A Retrospective Multicenter Analysis. Clin. Lung Cancer 2019, 20, e97–e106. [Google Scholar] [CrossRef] [PubMed]
- Mouri, A.; Kaira, K.; Yamaguchi, O.; Shiono, A.; Miura, Y.; Hashimoto, K.; Nishihara, F.; Murayama, Y.; Kobayashi, K.; Kagamu, H. Clinical difference between discontinuation and retreatment with nivolumab after immune-related adverse events in patients with lung cancer. Cancer Chemother. Pharmacol. 2019, 84, 873–880. [Google Scholar] [CrossRef] [PubMed]
- Jia, X.-H.; Geng, L.-Y.; Jiang, P.-P.; Xu, H.; Nan, K.-J.; Yao, Y.; Jiang, L.-L.; Sun, H.; Qin, T.-J.; Guo, H. The biomarkers related to immune related adverse events caused by immune checkpoint inhibitors. J. Exp. Clin. Cancer Res. 2020, 39, 284. [Google Scholar] [CrossRef] [PubMed]
- Beer, A.G.; Zenzmaier, C.; Schreinlechner, M.; Haas, J.; Dietrich, M.F.; Herz, J.; Marschang, P. Expression of a recombinant full-length LRP1B receptor in human non-small cell lung cancer cells confirms the postulated growth-suppressing function of this large LDL receptor family member. Oncotarget 2016, 7, 68721–68733. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.X.; Musco, S.; Lisitsina, N.M.; Yaklichkin, S.Y.; Lisitsyn, N.A. Genomic organization of a new candidate tumor suppressor gene, LRP1B. Genomics 2000, 69, 271–274. [Google Scholar] [CrossRef]
- Chen, H.; Chong, W.; Wu, Q.; Yao, Y.; Mao, M.; Wang, X. Association of LRP1B Mutation With Tumor Mutation Burden and Outcomes in Melanoma and Non-small Cell Lung Cancer Patients Treated With Immune Check-Point Blockades. Front. Immunol. 2019, 10, 1113. [Google Scholar] [CrossRef] [PubMed]
- Fountzilas, E.; Kotoula, V.; Koliou, G.A.; Giannoulatou, E.; Gogas, H.; Papadimitriou, C.; Tikas, I.; Zhang, J.; Papadopoulou, K.; Zagouri, F.; et al. Pathogenic mutations and overall survival in 3,084 patients with cancer: The Hellenic Cooperative Oncology Group Precision Medicine Initiative. Oncotarget 2020, 11, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Liu, Y.; Rappaport, A.R.; Kitzing, T.; Schultz, N.; Zhao, Z.; Shroff, A.S.; Dickins, R.A.; Vakoc, C.R.; Bradner, J.E.; et al. MLL3 is a haploinsufficient 7q tumor suppressor in acute myeloid leukemia. Cancer Cell 2014, 25, 652–665. [Google Scholar] [CrossRef]
- Rampias, T.; Karagiannis, D.; Avgeris, M.; Polyzos, A.; Kokkalis, A.; Kanaki, Z.; Kousidou, E.; Tzetis, M.; Kanavakis, E.; Stravodimos, K.; et al. The lysine-specific methyltransferase KMT2C/MLL3 regulates DNA repair components in cancer. EMBO Rep. 2019, 20. [Google Scholar] [CrossRef] [PubMed]
- Ricciuti, B.; Recondo, G.; Spurr, L.F.; Li, Y.Y.; Lamberti, G.; Venkatraman, D.; Umeton, R.; Cherniack, A.D.; Nishino, M.; Sholl, L.M.; et al. Impact of DNA Damage Response and Repair (DDR) Gene Mutations on Efficacy of PD-(L)1 Immune Checkpoint Inhibition in Non-Small Cell Lung Cancer. Clin. Cancer Res. 2020, 26, 4135–4142. [Google Scholar] [CrossRef] [PubMed]
- Zhao, D.; Mambetsariev, I.; Li, H.; Chen, C.; Fricke, J.; Fann, P.; Kulkarni, P.; Xing, Y.; Lee, P.P.; Bild, A.; et al. Association of molecular characteristics with survival in advanced non-small cell lung cancer patients treated with checkpoint inhibitors. Lung Cancer 2020, 146, 174–181. [Google Scholar] [CrossRef]
- Sabari, J.K.; Leonardi, G.C.; Shu, C.A.; Umeton, R.; Montecalvo, J.; Ni, A.; Chen, R.; Dienstag, J.; Mrad, C.; Bergagnini, I.; et al. PD-L1 expression, tumor mutational burden, and response to immunotherapy in patients with MET exon 14 altered lung cancers. Ann. Oncol. 2018, 29, 2085–2091. [Google Scholar] [CrossRef]
| IrAEs | No (%) |
|---|---|
| Pneumonitis | 10 (11.1%) |
| Adrenal insufficiency | 4 (4.4%) |
| Hypothyroidism | 4 (4.4%) |
| Colitis | 4 (4.4%) |
| Liver injury | 3 (3.3%) |
| Nephritis | 2 (2.2%) |
| Heart failure | 1 (1.1%) |
| Pancreatitis | 1 (1.1%) |
| Diabetic ketone acidosis | 1 (1.1%) |
| Arthralgia | 1 (1.1%) |
| Rash | 2 (2.2%) |
| Other/Infection | 2 (2.2%) |
| Multiple irAEs | 7 (7.8%) |
| Total patients with irAEs | 28 (31.1%) 1 |
| Characteristics | n = 90 (%) | IrAEs n = 28 (%) | No irAEs n = 62 (%) | p Values 1 |
|---|---|---|---|---|
| Median age at ICI (range 36–88) | 68.5 | 70.5 | 67.5 | ns |
| Gender | <0.05 | |||
| Women | 41 (45.6%) | 8 (28.6%) | 33 (53.2%) | |
| Men | 49 (54.4%) | 20 (71.4%) | 29 (46.8%) | |
| Smoking status | <0.01 | |||
| Current | 11 (12.2%) | 6 (21.4%) | 5 (8.1%) | |
| Former | 50 (55.6%) | 19 (67.9%) | 31 (50.0%) | |
| Never | 29 (32.2%) | 3 (10.7%) | 26 (41.9%) | |
| Histology | <0.01 | |||
| Lung adenocarcinoma | 63 (70%) | 16 (57.1%) | 47 (75.8%) | |
| Lung squamous | 14 (15.6%) | 4 (14.3%) | 10 (16.1%) | |
| SCLC | 5 (5.6%) | 5 (17.9%) | 0 | |
| Others 2 | 8 (8.9%) | 3 (10.7%) | 5 (8.1%) | |
| ICIs with other therapy | ns | |||
| Yes | 12 (13.3%) | 2 (7.1%) | 10 (16.1%) | |
| No | 78 (86.7%) | 26 (92.9%) | 52 (83.9%) | |
| PD-L1 | ns | |||
| Negative | 16 (17.8%) | 5 (17.9%) | 11 (17.7%) | |
| 1% to <50% | 8 (8.9%) | 1 (3.6%) | 7 (11.3%) | |
| 50% and above | 21 (23.3%) | 7 (25.0%) | 14 (22.6%) | |
| Not tested | 45 (50.0%) | 15 (53.6%) | 30 (48.4%) | |
| Median lines of therapy (range 1–7) | 2 | 2 | 2 | ns |
| Brain metastasis | ns | |||
| Yes | 35 (38.9%) | 10 (35.7%) | 25 (40.3%) | |
| No | 55 (61.1%) | 18 (64.3%) | 37 (59.7%) | |
| Median length of stay (range 1–37) | 7 | 7 | 6 | ns |
| Invasive Procedures after ICIs | irAEs (n = 28) | No irAEs (n = 62) |
|---|---|---|
| Bronchoscopy/lung biopsy | 6 (21.4%) | 6 (9.7%) |
| EGD/Colonoscopy | 5 (17.9%) | 6 (9.7%) |
| Thoracentesis | 2 (7.1%) | 8 (12.9%) |
| Liver biopsy | 1 (3.6%) | 3 (4.8%) |
| Skin biopsy | 1 (3.6%) | 0 |
| Kidney biopsy | 1 (3.6%) | 0 |
| Brain surgery | 1 (3.6%) | 2 (3.2%) |
| Spine surgery | 0 | 1 (1.6%) |
| Pericardium biopsy | 0 | 1 (1.6%) |
| Total | 17 (60.7%) | 25 (40.3%) 1 |
| Genomics | All (%) | IrAEs (%) | No irAEs (%) | OR (95% CI) | p Values 1 |
|---|---|---|---|---|---|
| TP53 | ns | ||||
| Positive | 40 | 10 (66.7%) | 30 (58.8%) | ||
| Negative | 26 | 5 (33.3%) | 21 (41.2%) | ||
| Not tested | 24 | 13 | 11 | ||
| KRAS | ns | ||||
| Positive | 23 | 8 (40%) | 15 (26.3%) | ||
| Negative | 54 | 12 (60%) | 42 (73.7%) | ||
| Not tested | 13 | 8 | 5 | ||
| EGFR | ns | ||||
| Positive | 16 | 2 (10%) | 14 (24.6%) | ||
| Negative | 61 | 18 (90%) | 43 (75.4%) | ||
| Not tested | 13 | 8 | 5 | ||
| LRP1B | 13.5 (1.7–166.1) | <0.05 | |||
| Positive | 12 | 5 (83.3%) | 7 (26.9%) | ||
| Negative | 20 | 1 (16.7%) | 19 (73.1%) | ||
| Not tested | 58 | 22 | 36 | ||
| PIK3CA | ns | ||||
| Positive | 9 | 3 (20%) | 6 (11.8%) | ||
| Negative | 57 | 12 (80%) | 45 (88.2%) | ||
| Not tested | 24 | 13 | 11 | ||
| MLL3 | 8.4 (1.3–49.3) | <0.05 | |||
| Positive | 9 | 4 (66.7%) | 5 (19.2%) | ||
| Negative | 23 | 2 (33.3%) | 21 (80.8%) | ||
| Not tested | 58 | 22 | 36 | ||
| TMB | ns | ||||
| TMB-Low | 5 | 1 (33.3%) | 4 (26.7%) | ||
| TMB-Intermediate | 9 | 1 (33.3%) | 8 (53.3%) | ||
| TMB-High | 4 | 1 (33.3%) | 3 (20%) | ||
| Not tested | 72 | 25 | 47 |
| Risk Factors | Odds Ratio (95% CI) | p Values 1 |
|---|---|---|
| Gender | ||
| Female | References | |
| Male | 1.47 (0.43–4.99) | 0.5358 |
| Smoking | ||
| Never | References | |
| Current | 3.61 (0.43–30.11) | 0.2363 |
| Former | 3.44 (0.76–15.45) | 0.1073 |
| MLL3 | ||
| Negative | References | |
| Positive | 6.52 (0.70–60.62) | 0.0991 |
| LRP1B | ||
| Negative | References | |
| Positive | 8.00 (0.65–98.01) | 0.1037 |
| Risk Factors | HR (95%CI) | p Values 1 |
|---|---|---|
| Gender | ||
| Female | Reference | |
| Male | 1.11 (0.57–2.15) | ns |
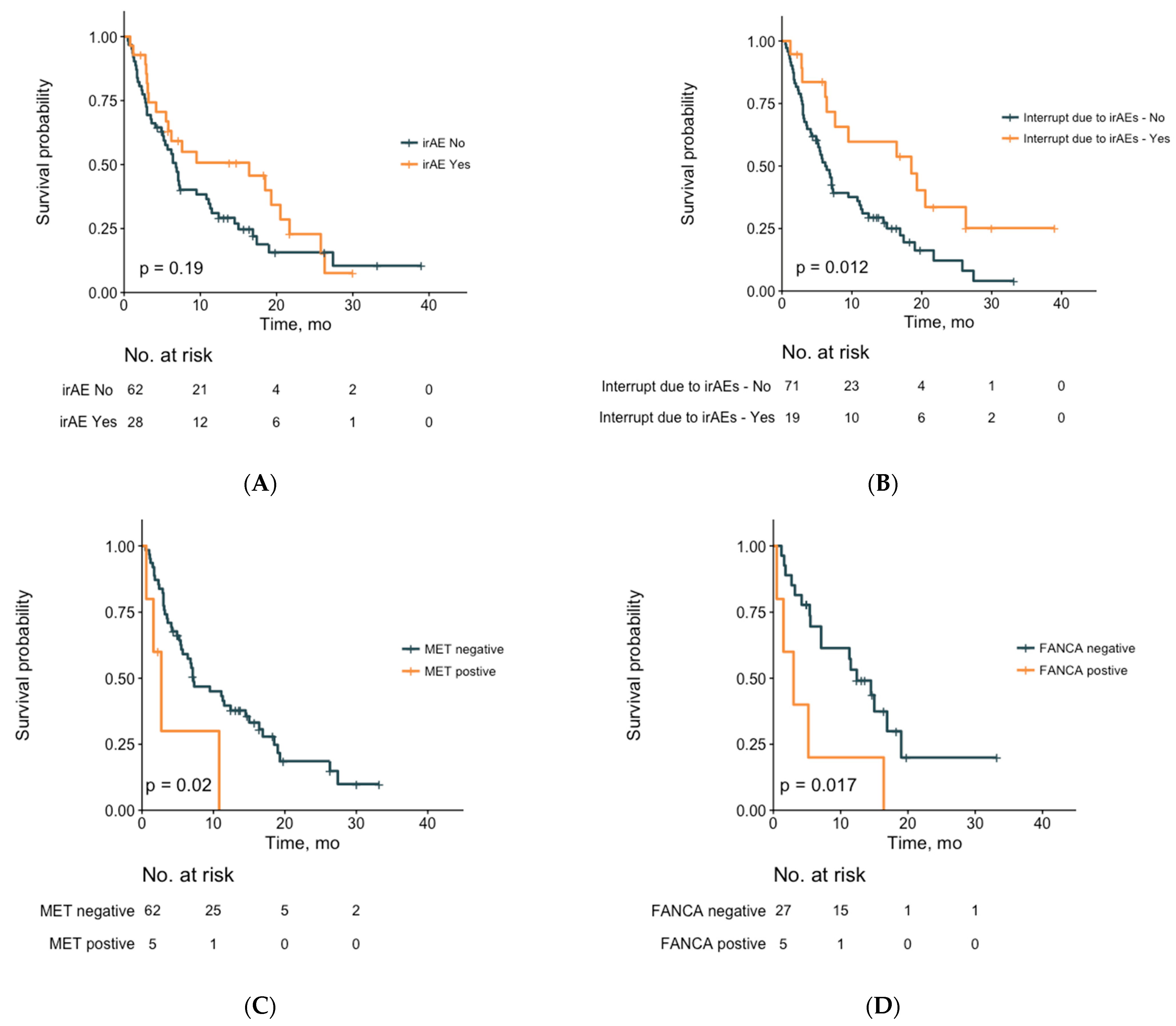
| IrAEs | ||
| No | Reference | |
| Yes | 1.21 (0.50–2.92) | ns |
| Interrupt ICIs due to irAEs | ||
| No | Reference | |
| Yes | 0.05 (0.01–0.19) | <0.001 |
| Lines of therapy | ||
| ≥3 lines | Reference | |
| 2nd line | 0.62 (0.25–1.49) | |
| 1st line | 0.21 (0.07–0.58) | <0.01 |
| EGFR | ||
| Negative | Reference | |
| Positive | 1.45 (0.55–3.77) | ns |
| FANCA | ||
| Negative | Reference | |
| Positive | 11.30 (3.36–38.01) | <0.001 |
| MET | ||
| Negative | Reference | |
| Positive | 11.17 (2.92–42.81) | <0.001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhao, D.; Li, H.; Mambetsariev, I.; Chen, C.; Pharaon, R.; Fricke, J.; Baroz, A.R.; Kulkarni, P.; Xing, Y.; Massarelli, E.; et al. Molecular and Clinical Features of Hospital Admissions in Patients with Thoracic Malignancies on Immune Checkpoint Inhibitors. Cancers 2021, 13, 2653. https://doi.org/10.3390/cancers13112653
Zhao D, Li H, Mambetsariev I, Chen C, Pharaon R, Fricke J, Baroz AR, Kulkarni P, Xing Y, Massarelli E, et al. Molecular and Clinical Features of Hospital Admissions in Patients with Thoracic Malignancies on Immune Checkpoint Inhibitors. Cancers. 2021; 13(11):2653. https://doi.org/10.3390/cancers13112653
Chicago/Turabian StyleZhao, Dan, Haiqing Li, Isa Mambetsariev, Chen Chen, Rebecca Pharaon, Jeremy Fricke, Angel R. Baroz, Prakash Kulkarni, Yan Xing, Erminia Massarelli, and et al. 2021. "Molecular and Clinical Features of Hospital Admissions in Patients with Thoracic Malignancies on Immune Checkpoint Inhibitors" Cancers 13, no. 11: 2653. https://doi.org/10.3390/cancers13112653
APA StyleZhao, D., Li, H., Mambetsariev, I., Chen, C., Pharaon, R., Fricke, J., Baroz, A. R., Kulkarni, P., Xing, Y., Massarelli, E., Koczywas, M., Reckamp, K. L., Margolin, K., & Salgia, R. (2021). Molecular and Clinical Features of Hospital Admissions in Patients with Thoracic Malignancies on Immune Checkpoint Inhibitors. Cancers, 13(11), 2653. https://doi.org/10.3390/cancers13112653








