Ferrocifen Loaded Lipid Nanocapsules: A Promising Anticancer Medication against Multidrug Resistant Tumors
Abstract
Simple Summary
Abstract
1. Introduction
2. Ferrocifens: Promising Molecules against Cancer
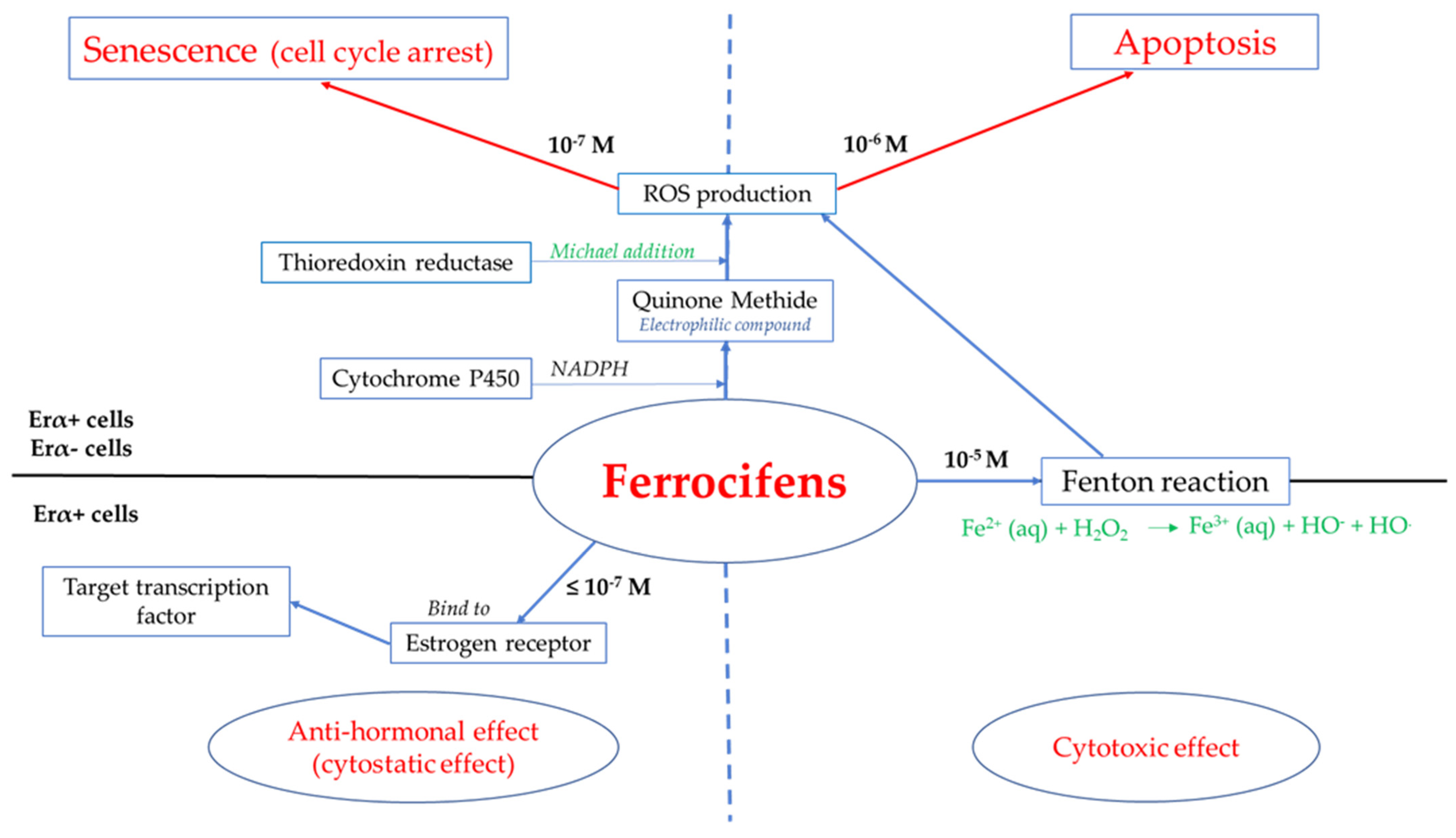
2.1. Ferrocifen Mechanisms of Action
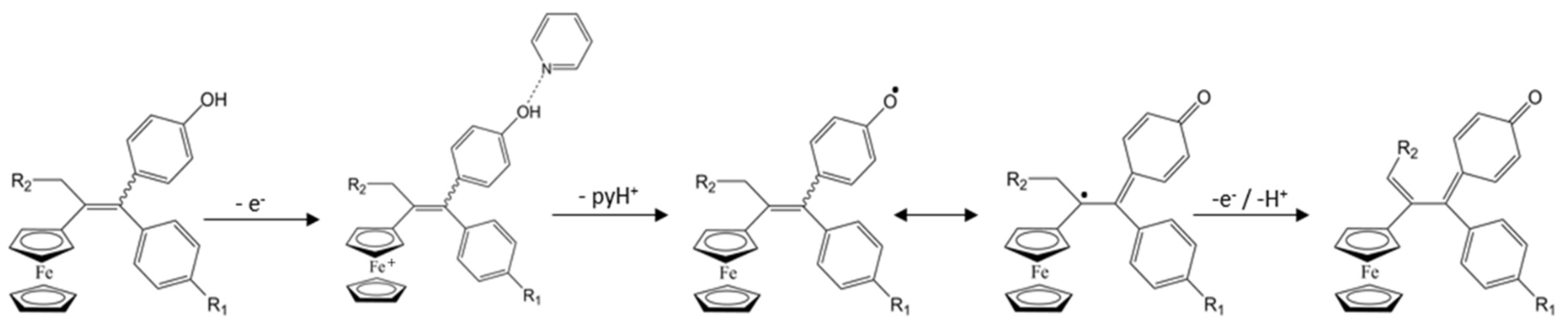
2.1.1. Formation of the Quinone Methide
2.1.2. Ferrocifen Mechanisms of Action at the Cellular Level
Antiestrogenic Proliferative Effect
Cytotoxic Effect
3. Lipid Nanocapsule Characteristics
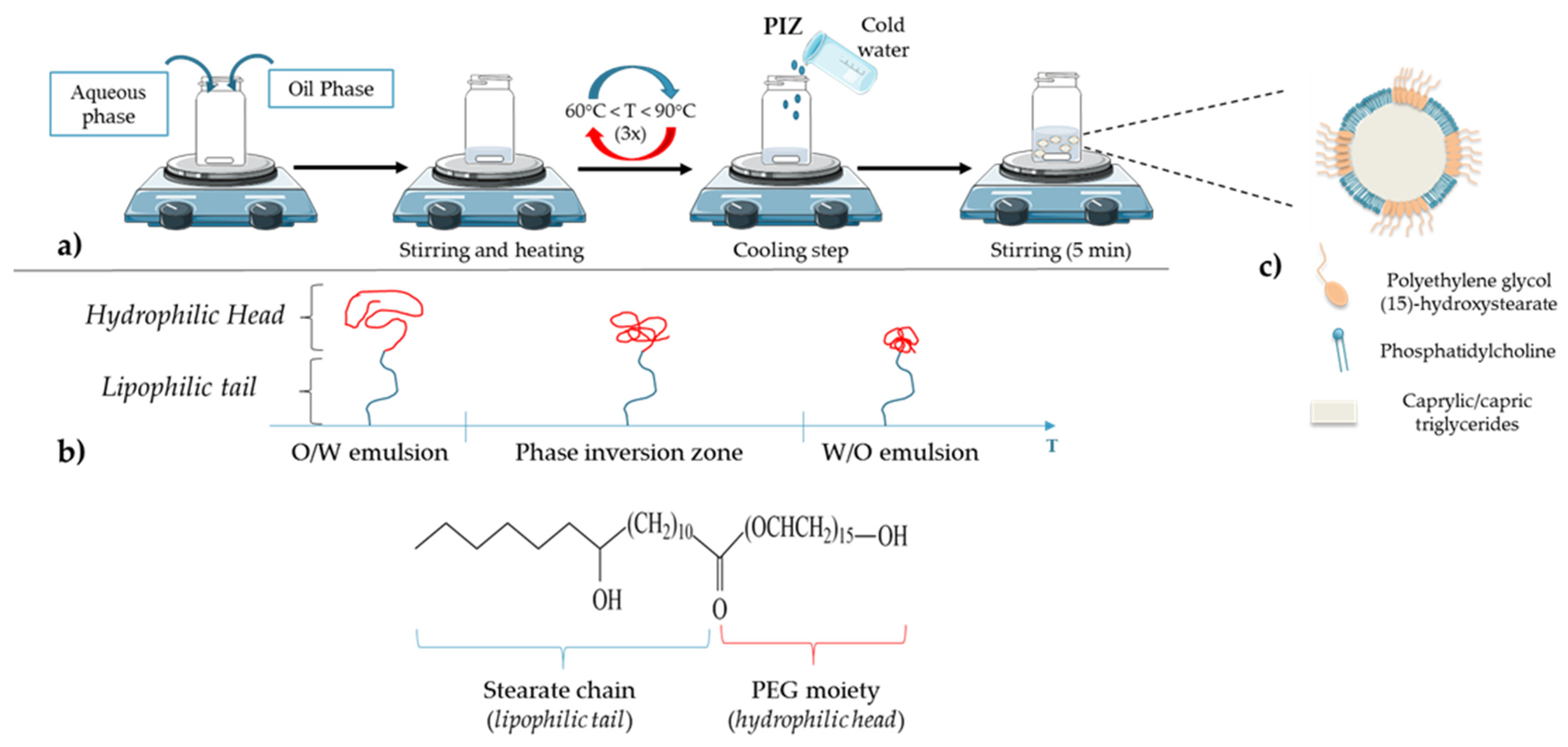
3.1. Composition and Formulation Process
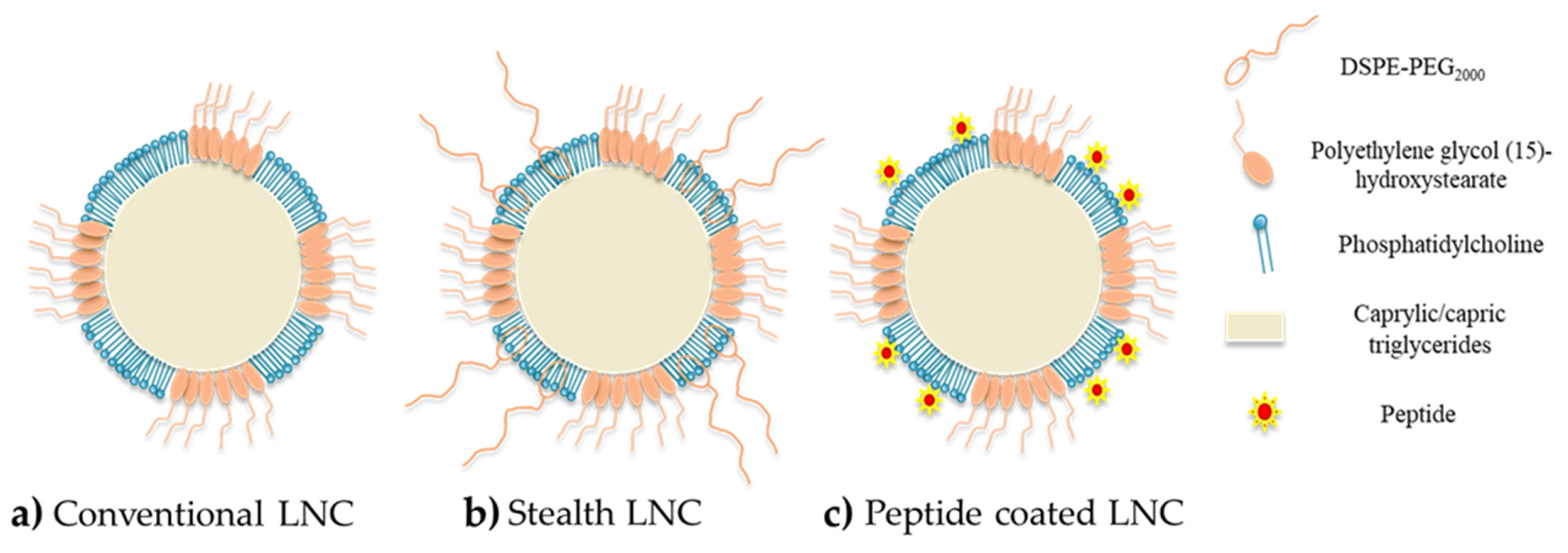
3.2. Surface Modification (Passive and Active Targeting)
4. Ferrocifen-Loaded LNCs: In Vitro Studies
5. Ferrocifen-Loaded Lipid Nanocapsules: In Vivo Experiments
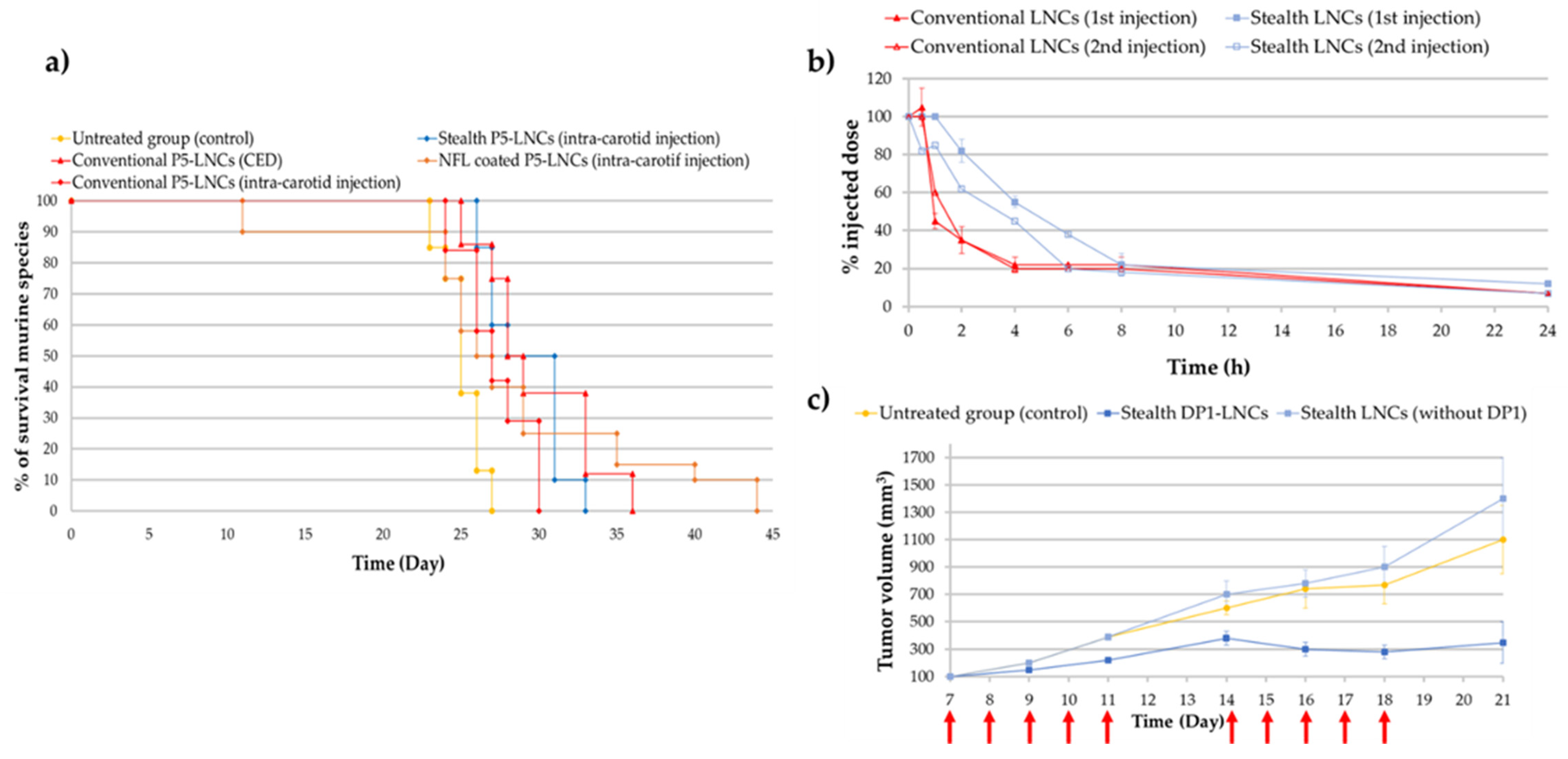
5.1. Glioblastoma
5.2. Other Cancers
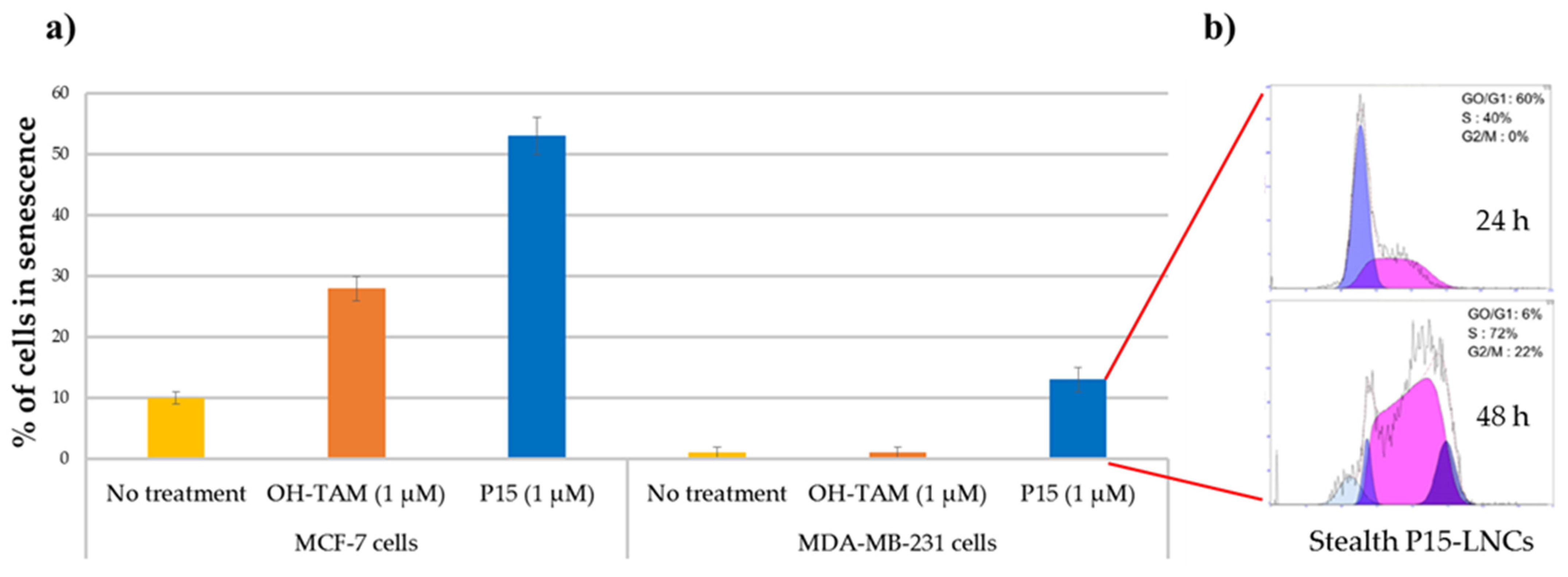
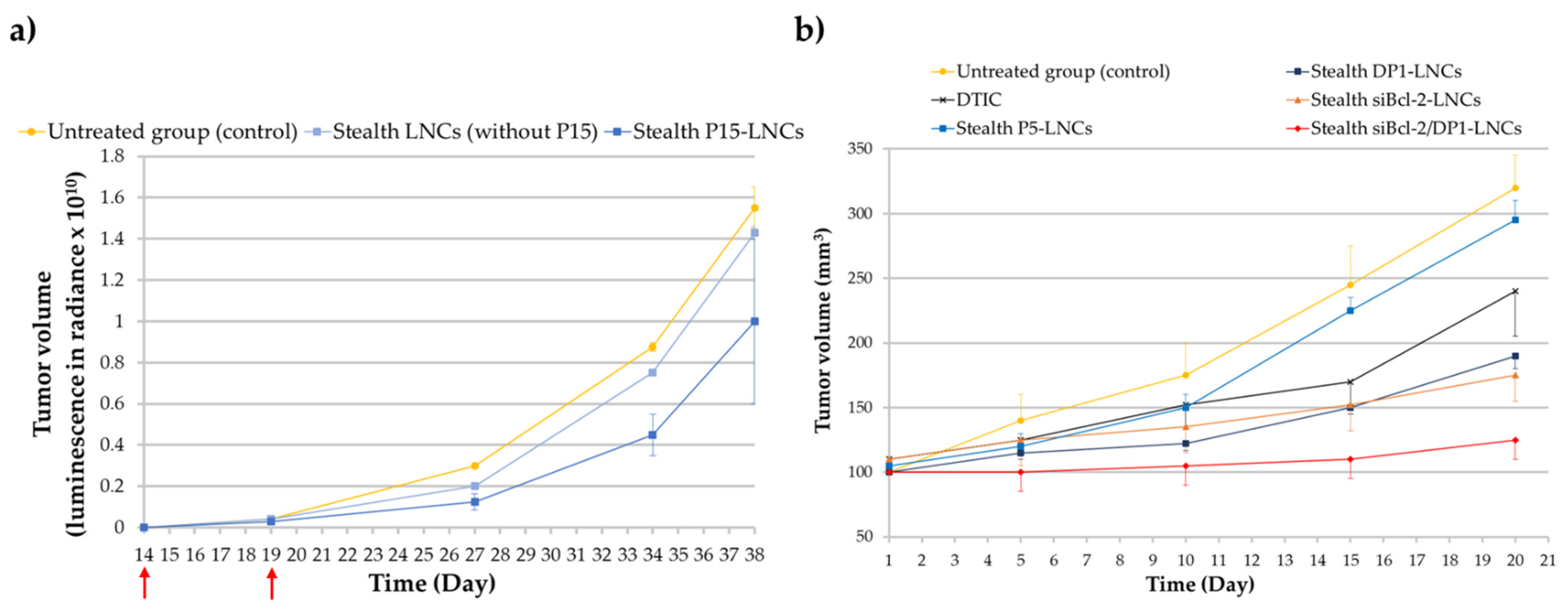
5.2.1. Breast Cancer
5.2.2. Metastatic Melanoma
6. Association with Other Therapies: An Interesting Perspective
6.1. Ferrocifens and Radiotherapy
6.2. Ferrocifens and Gene Therapy
6.3. Ferrocifen-LNC-Loaded Cellular Vectors
7. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Uziely, B.; Jeffers, S.; Isacson, R.; Kutsch, K.; Wei-Tsao, D.; Yehoshua, Z.; Libson, E.; Muggia, F.M.; Gabizon, A. Liposomal Doxorubicin: Antitumor Activity and Unique Toxicities during Two Complementary Phase I Studies. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 1995, 13, 1777–1785. [Google Scholar] [CrossRef]
- Bobo, D.; Robinson, K.J.; Islam, J.; Thurecht, K.J.; Corrie, S.R. Nanoparticle-Based Medicines: A Review of FDA-Approved Materials and Clinical Trials to Date. Pharm. Res. 2016, 33, 2373–2387. [Google Scholar] [CrossRef] [PubMed]
- Couvreur, P. Nanomedicine: From Where Are We Coming and Where Are We Going? J. Control. Release Off. J. Control. Release Soc. 2019, 311–312, 319–321. [Google Scholar] [CrossRef]
- Reedijk, J. Platinum Anticancer Coordination Compounds: Study of DNA Binding Inspires New Drug Design. Eur. J. Inorg. Chem. 2009, 2009, 1303–1312. [Google Scholar] [CrossRef]
- Al-Malky, H.S.; Al Harthi, S.E.; Osman, A.-M.M. Major Obstacles to Doxorubicin Therapy: Cardiotoxicity and Drug Resistance. J. Oncol. Pharm. Pract. Off. Publ. Int. Soc. Oncol. Pharm. Pract. 2020, 26, 434–444. [Google Scholar] [CrossRef]
- Ali, S.; Rasool, M.; Chaoudhry, H.; N Pushparaj, P.; Jha, P.; Hafiz, A.; Mahfooz, M.; Abdus Sami, G.; Azhar Kamal, M.; Bashir, S.; et al. Molecular Mechanisms and Mode of Tamoxifen Resistance in Breast Cancer. Bioinformation 2016, 12, 135–139. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Chen, L. Progress in Research on Paclitaxel and Tumor Immunotherapy. Cell. Mol. Biol. Lett. 2019, 24. [Google Scholar] [CrossRef]
- Cao, Y.; Wei, Z.; Li, M.; Wang, H.; Yin, L.; Chen, D.; Wang, Y.; Chen, Y.; Yuan, Q.; Pu, X.; et al. Formulation, Pharmacokinetic Evaluation and Cytotoxicity of an Enhanced- Penetration Paclitaxel Nanosuspension. Curr. Cancer Drug Targets 2019, 19, 338–347. [Google Scholar] [CrossRef]
- Jaouen, G.; Vessieres, A. Transition Metal Carbonyl Oestrogen Receptor Assay. Pure Appl. Chem. 1985, 57, 1865–1874. [Google Scholar] [CrossRef]
- Jaouen, G.; Vessieres, A.; Top, S.; Ismail, A.A.; Butler, I.S. Metal Carbonyl Fragments as a New Class of Markers in Molecular Biology. J. Am. Chem. Soc. 1985, 107, 4778–4780. [Google Scholar] [CrossRef]
- Hillard, E.; Vessières, A.; Le Bideau, F.; Plazuk, D.; Spera, D.; Huché, M.; Jaouen, G. A Series of Unconjugated Ferrocenyl Phenols: Prospects as Anticancer Agents. ChemMedChem 2006, 1, 551–559. [Google Scholar] [CrossRef]
- Top, S.; Vessières, A.; Cabestaing, C.; Laios, I.; Leclercq, G.; Provot, C.; Jaouen, G. Studies on Organometallic Selective Estrogen Receptor Modulators. (SERMs) Dual Activity in the Hydroxy-Ferrocifen Series. J. Organomet. Chem. 2001, 637–639, 500–506. [Google Scholar] [CrossRef]
- Vessières, A.; Top, S.; Beck, W.; Hillard, E.; Jaouen, G. Metal Complex SERMs (Selective Oestrogen Receptor Modulators). The Influence of Different Metal Units on Breast Cancer Cell Antiproliferative Effects. Dalton Trans. Camb. Engl. 2003 2006, 529–541. [Google Scholar] [CrossRef] [PubMed]
- Maeda, H. Macromolecular Therapeutics in Cancer Treatment: The EPR Effect and Beyond. J. Control. Release Off. J. Control. Release Soc. 2012, 164, 138–144. [Google Scholar] [CrossRef] [PubMed]
- Maeda, H. The 35th Anniversary of the Discovery of EPR Effect: A New Wave of Nanomedicines for Tumor-Targeted Drug Delivery-Personal Remarks and Future Prospects. J. Pers. Med. 2021, 11, 229. [Google Scholar] [CrossRef]
- Danhier, F. To Exploit the Tumor Microenvironment: Since the EPR Effect Fails in the Clinic, What Is the Future of Nanomedicine? J. Control. Release Off. J. Control. Release Soc. 2016, 244, 108–121. [Google Scholar] [CrossRef]
- Sindhwani, S.; Syed, A.M.; Ngai, J.; Kingston, B.R.; Maiorino, L.; Rothschild, J.; MacMillan, P.; Zhang, Y.; Rajesh, N.U.; Hoang, T.; et al. The Entry of Nanoparticles into Solid Tumours. Nat. Mater. 2020, 19, 566–575. [Google Scholar] [CrossRef]
- Poste, G.; Kirsh, R. Site–Specific (Targeted) Drug Delivery in Cancer Therapy. Bio/Technology 1983, 1, 869–878. [Google Scholar] [CrossRef]
- Rolland, A.; Bourel, D.; Genetet, B.; Le Verge, R. Monoclonal Antibodies Covalently Coupled to Polymethacrylic Nanoparticles: In Vitro Specific Targeting to Human T Lymphocytes. Int. J. Pharm. 1987, 39, 173–180. [Google Scholar] [CrossRef]
- Barbet, J.; Machy, P.; Leserman, L.D. Monoclonal Antibody Covalently Coupled to Liposomes: Specific Targeting to Cells. J. Supramol. Struct. Cell. Biochem. 1981, 16, 243–258. [Google Scholar] [CrossRef] [PubMed]
- Hirsjärvi, S.; Passirani, C.; Benoit, J.-P. Passive and Active Tumour Targeting with Nanocarriers. Curr. Drug Discov. Technol. 2011, 8, 188–196. [Google Scholar] [CrossRef]
- Clemons, T.D.; Singh, R.; Sorolla, A.; Chaudhari, N.; Hubbard, A.; Iyer, K.S. Distinction Between Active and Passive Targeting of Nanoparticles Dictate Their Overall Therapeutic Efficacy. Langmuir ACS J. Surf. Colloids 2018, 34, 15343–15349. [Google Scholar] [CrossRef]
- Attia, M.F.; Anton, N.; Wallyn, J.; Omran, Z.; Vandamme, T.F. An Overview of Active and Passive Targeting Strategies to Improve the Nanocarriers Efficiency to Tumour Sites. J. Pharm. Pharmacol. 2019, 71, 1185–1198. [Google Scholar] [CrossRef]
- Heurtault, B.; Saulnier, P.; Pech, B.; Proust, J.-E.; Benoit, J.-P. A Novel Phase Inversion-Based Process for the Preparation of Lipid Nanocarriers. Pharm. Res. 2002, 19, 875–880. [Google Scholar] [CrossRef] [PubMed]
- Heurtault, B.; Saulnier, P.; Benoit, J.-P.; Proust, J.-E.; Pech, B.; Richard, J. Lipid Nanocapsules, Preparation Process and Use as Medicine. U.S. Patent 8057823B2, 15 November 2011. Available online: https://patents.google.com/patent/US8057823B2/en (accessed on 17 April 2020).
- Béduneau, A.; Hindré, F.; Clavreul, A.; Leroux, J.-C.; Saulnier, P.; Benoit, J.-P. Brain Targeting Using Novel Lipid Nanovectors. J. Control. Release Off. J. Control. Release Soc. 2008, 126, 44–49. [Google Scholar] [CrossRef]
- Roger, E.; Lagarce, F.; Garcion, E.; Benoit, J.-P. Lipid Nanocarriers Improve Paclitaxel Transport throughout Human Intestinal Epithelial Cells by Using Vesicle-Mediated Transcytosis. J. Control. Release Off. J. Control. Release Soc. 2009, 140, 174–181. [Google Scholar] [CrossRef] [PubMed]
- Laine, A.-L.; Huynh, N.T.; Clavreul, A.; Balzeau, J.; Béjaud, J.; Vessieres, A.; Benoit, J.-P.; Eyer, J.; Passirani, C. Brain Tumour Targeting Strategies via Coated Ferrociphenol Lipid Nanocapsules. Eur. J. Pharm. Biopharm. Off. J. Arbeitsgemeinschaft Pharm. Verfahrenstechnik EV 2012, 81, 690–693. [Google Scholar] [CrossRef]
- Balzeau, J.; Pinier, M.; Berges, R.; Saulnier, P.; Benoit, J.-P.; Eyer, J. The Effect of Functionalizing Lipid Nanocapsules with NFL-TBS.40-63 Peptide on Their Uptake by Glioblastoma Cells. Biomaterials 2013, 34, 3381–3389. [Google Scholar] [CrossRef]
- Carradori, D.; Saulnier, P.; Préat, V.; des Rieux, A.; Eyer, J. NFL-Lipid Nanocapsules for Brain Neural Stem Cell Targeting in Vitro and in Vivo. J. Control. Release Off. J. Control. Release Soc. 2016, 238, 253–262. [Google Scholar] [CrossRef]
- David, S.; Montier, T.; Carmoy, N.; Resnier, P.; Clavreul, A.; Mével, M.; Pitard, B.; Benoit, J.-P.; Passirani, C. Treatment Efficacy of DNA Lipid Nanocapsules and DNA Multimodular Systems after Systemic Administration in a Human Glioma Model. J. Gene Med. 2012, 14, 769–775. [Google Scholar] [CrossRef] [PubMed]
- Allard, E.; Hindré, F.; Passirani, C.; Lemaire, L.; Lepareur, N.; Noiret, N.; Menei, P.; Benoit, J.-P. 188Re-Loaded Lipid Nanocapsules as a Promising Radiopharmaceutical Carrier for Internal Radiotherapy of Malignant Gliomas. Eur. J. Nucl. Med. Mol. Imaging 2008, 35, 1838–1846. [Google Scholar] [CrossRef] [PubMed]
- Top, S.; Tang, J.; Vessières, A.; Carrez, D.; Provot, C.; Jaouen, G. Ferrocenyl Hydroxytamoxifen: A Prototype for a New Range of Oestradiol Receptor Site-Directed Cytotoxics. Chem. Commun. 1996, 955–956. [Google Scholar] [CrossRef]
- Top, S.; Dauer, B.; Vaissermann, J.; Jaouen, G. Facile Route to Ferrocifen, 1-[4-(2-Dimethylaminoethoxy)]-1-(Phenyl-2-Ferrocenyl-but-1-Ene), First Organometallic Analogue of Tamoxifen, by the McMurry Reaction. J. Organomet. Chem. 1997, 541, 355–361. [Google Scholar] [CrossRef]
- Vessières, A.; Wang, Y.; McGlinchey, M.J.; Jaouen, G. Multifaceted Chemical Behaviour of Metallocene (M = Fe, Os) Quinone Methides. Their Contribution to Biology. Coord. Chem. Rev. 2021, 430, 213658. [Google Scholar] [CrossRef]
- Citta, A.; Folda, A.; Bindoli, A.; Pigeon, P.; Top, S.; Vessières, A.; Salmain, M.; Jaouen, G.; Rigobello, M.P. Evidence for Targeting Thioredoxin Reductases with Ferrocenyl Quinone Methides. A Possible Molecular Basis for the Antiproliferative Effect of Hydroxyferrocifens on Cancer Cells. J. Med. Chem. 2014, 57, 8849–8859. [Google Scholar] [CrossRef]
- Michard, Q.; Jaouen, G.; Vessieres, A.; Bernard, B.A. Evaluation of Cytotoxic Properties of Organometallic Ferrocifens on Melanocytes, Primary and Metastatic Melanoma Cell Lines. J. Inorg. Biochem. 2008, 102, 1980–1985. [Google Scholar] [CrossRef]
- Nguyen, A.; Vessières, A.; Hillard, E.A.; Top, S.; Pigeon, P.; Jaouen, G. Ferrocifens and Ferrocifenols as New Potential Weapons against Breast Cancer. CHIMIA 2007, 61, 716–724. [Google Scholar] [CrossRef]
- Allard, E.; Passirani, C.; Garcion, E.; Pigeon, P.; Vessières, A.; Jaouen, G.; Benoit, J.-P. Lipid Nanocapsules Loaded with an Organometallic Tamoxifen Derivative as a Novel Drug-Carrier System for Experimental Malignant Gliomas. J. Control. Release Off. J. Control. Release Soc. 2008, 130, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Soares, M.B.P.; Costa, J.F.O.; de Sá, M.S.; dos Santos, R.R.; Pigeon, P.; Jaouen, G.; Santana, A.E.G.; Goulart, M.O.F.; Hillard, E. Antiparasitic and Immunomodulatory Activities of 1,1-Bis(4-Hydroxyphenyl)-2-Phenyl-but-1-Ene and Its Protected and Free 2-Ferrocenyl Derivatives. Drug Dev. Res. 2010. [Google Scholar] [CrossRef]
- Jaouen, G.; Vessières, A.; Top, S. Ferrocifen Type Anti Cancer Drugs. Chem. Soc. Rev. 2015, 44, 8802–8817. [Google Scholar] [CrossRef]
- Hillard, E.; Vessières, A.; Thouin, L.; Jaouen, G.; Amatore, C. Ferrocene-Mediated Proton-Coupled Electron Transfer in a Series of Ferrocifen-Type Breast-Cancer Drug Candidates. Angew. Chem. Int. Ed. 2006, 45, 285–290. [Google Scholar] [CrossRef] [PubMed]
- Kovacic, P. Unifying Mechanism for Anticancer Agents Involving Electron Transfer and Oxidative Stress: Clinical Implications. Med. Hypotheses 2007, 69, 510–516. [Google Scholar] [CrossRef] [PubMed]
- Messina, P.; Labbé, E.; Buriez, O.; Hillard, E.A.; Vessières, A.; Hamels, D.; Top, S.; Jaouen, G.; Frapart, Y.M.; Mansuy, D.; et al. Deciphering the Activation Sequence of Ferrociphenol Anticancer Drug Candidates. Chem. Eur. J. 2012, 18, 6581–6587. [Google Scholar] [CrossRef]
- Hillard, E.A.; de Abreu, F.C.; Ferreira, D.C.M.; Jaouen, G.; Goulart, M.O.F.; Amatore, C. Electrochemical Parameters and Techniques in Drug Development, with an Emphasis on Quinones and Related Compounds. Chem. Commun. 2008, 2612–2628. [Google Scholar] [CrossRef]
- Zekri, O.; Hillard, E.A.; Top, S.; Vessières, A.; Pigeon, P.; Plamont, M.-A.; Huché, M.; Boutamine, S.; McGlinchey, M.J.; Müller-Bunz, H.; et al. Role of Aromatic Substituents on the Antiproliferative Effects of Diphenyl Ferrocenyl Butene Compounds. Dalton Trans. Camb. Engl. 2003 2009, 4318–4326. [Google Scholar] [CrossRef]
- Wang, Y.; Pigeon, P.; Top, S.; McGlinchey, M.J.; Jaouen, G. Organometallic Antitumor Compounds: Ferrocifens as Precursors to Quinone Methides. Angew. Chem. Int. Ed. Engl. 2015, 54, 10230–10233. [Google Scholar] [CrossRef]
- Wang, Y.; Pigeon, P.; Top, S.; Sanz García, J.; Troufflard, C.; Ciofini, I.; McGlinchey, M.J.; Jaouen, G. Atypical Lone Pair-π Interaction with Quinone Methides in a Series of Imido-Ferrociphenol Anticancer Drug Candidates. Angew. Chem. Int. Ed. Engl. 2019, 58, 8421–8425. [Google Scholar] [CrossRef]
- Richard, M.-A.; Hamels, D.; Pigeon, P.; Top, S.; Dansette, P.M.; Lee, H.Z.S.; Vessières, A.; Mansuy, D.; Jaouen, G. Oxidative Metabolism of Ferrocene Analogues of Tamoxifen: Characterization and Antiproliferative Activities of the Metabolites. ChemMedChem 2015, 10, 981–990. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Dansette, P.M.; Pigeon, P.; Top, S.; McGlinchey, M.J.; Mansuy, D.; Jaouen, G. A New Generation of Ferrociphenols Leads to a Great Diversity of Reactive Metabolites, and Exhibits Remarkable Antiproliferative Properties. Chem. Sci. 2018, 9, 70–78. [Google Scholar] [CrossRef]
- Görmen, M.; Pigeon, P.; Top, S.; Hillard, E.A.; Huché, M.; Hartinger, C.G.; de Montigny, F.; Plamont, M.-A.; Vessières, A.; Jaouen, G. Synthesis, Cytotoxicity, and COMPARE Analysis of Ferrocene and [3]Ferrocenophane Tetrasubstituted Olefin Derivatives against Human Cancer Cells. ChemMedChem 2010, 5, 2039–2050. [Google Scholar] [CrossRef]
- Scalcon, V.; Citta, A.; Folda, A.; Bindoli, A.; Salmain, M.; Ciofini, I.; Blanchard, S.; de Jésús Cázares-Marinero, J.; Wang, Y.; Pigeon, P.; et al. Enzymatic Oxidation of Ansa-Ferrocifen Leads to Strong and Selective Thioredoxin Reductase Inhibition in Vitro. J. Inorg. Biochem. 2016, 165, 146–151. [Google Scholar] [CrossRef][Green Version]
- Jaouen, G.; Top, S.; Vessières, A.; Leclercq, G.; Quivy, J.; Jin, L.; Croisy, A. The First Organometallic Antioestrogens and Their Antiproliferative Effects. Comptes Rendus Académie Sci. Ser. IIC Chem. 2000, 3, 89–93. [Google Scholar] [CrossRef]
- Vessières, A.; Corbet, C.; Heldt, J.M.; Lories, N.; Jouy, N.; Laïos, I.; Leclercq, G.; Jaouen, G.; Toillon, R.-A. A Ferrocenyl Derivative of Hydroxytamoxifen Elicits an Estrogen Receptor-Independent Mechanism of Action in Breast Cancer Cell Lines. J. Inorg. Biochem. 2010, 104, 503–511. [Google Scholar] [CrossRef] [PubMed]
- Lainé, A.-L.; Adriaenssens, E.; Vessières, A.; Jaouen, G.; Corbet, C.; Desruelles, E.; Pigeon, P.; Toillon, R.-A.; Passirani, C. The in Vivo Performance of Ferrocenyl Tamoxifen Lipid Nanocapsules in Xenografted Triple Negative Breast Cancer. Biomaterials 2013, 34, 6949–6956. [Google Scholar] [CrossRef]
- Wang, Y.; Richard, M.-A.; Top, S.; Dansette, P.M.; Pigeon, P.; Vessières, A.; Mansuy, D.; Jaouen, G. Ferrocenyl Quinone Methide-Thiol Adducts as New Antiproliferative Agents: Synthesis, Metabolic Formation from Ferrociphenols, and Oxidative Transformation. Angew. Chem. Int. Ed. Engl. 2016, 55, 10431–10434. [Google Scholar] [CrossRef] [PubMed]
- Clavreul, A.; Montagu, A.; Lainé, A.-L.; Tétaud, C.; Lautram, N.; Franconi, F.; Passirani, C.; Vessières, A.; Montero-Menei, C.N.; Menei, P. Targeting and Treatment of Glioblastomas with Human Mesenchymal Stem Cells Carrying Ferrociphenol Lipid Nanocapsules. Int. J. Nanomed. 2015, 10, 1259–1271. [Google Scholar] [CrossRef][Green Version]
- Allard, E.; Jarnet, D.; Vessières, A.; Vinchon-Petit, S.; Jaouen, G.; Benoit, J.-P.; Passirani, C. Local Delivery of Ferrociphenol Lipid Nanocapsules Followed by External Radiotherapy as a Synergistic Treatment against Intracranial 9L Glioma Xenograft. Pharm. Res. 2010, 27, 56–64. [Google Scholar] [CrossRef]
- Resnier, P.; Galopin, N.; Sibiril, Y.; Clavreul, A.; Cayon, J.; Briganti, A.; Legras, P.; Vessières, A.; Montier, T.; Jaouen, G.; et al. Efficient Ferrocifen Anticancer Drug and Bcl-2 Gene Therapy Using Lipid Nanocapsules on Human Melanoma Xenograft in Mouse. Pharmacol. Res. 2017, 126, 54–65. [Google Scholar] [CrossRef]
- Patra, M.; Gasser, G. The Medicinal Chemistry of Ferrocene and Its Derivatives. Nat. Rev. Chem. 2017, 1, 1–12. [Google Scholar] [CrossRef]
- Rao, A.K.; Ziegler, Y.S.; McLeod, I.X.; Yates, J.R.; Nardulli, A.M. Thioredoxin and Thioredoxin Reductase Influence Estrogen Receptor Alpha-Mediated Gene Expression in Human Breast Cancer Cells. J. Mol. Endocrinol. 2009, 43, 251–261. [Google Scholar] [CrossRef]
- Paterni, I.; Granchi, C.; Katzenellenbogen, J.A.; Minutolo, F. Estrogen Receptors Alpha (ERα) and Beta (ERβ): Subtype-Selective Ligands and Clinical Potential. Steroids 2014, 90, 13–29. [Google Scholar] [CrossRef]
- Hillard, E.A.; Vessières, A.; Jaouen, G. Ferrocene Functionalized Endocrine Modulators as Anticancer Agents. In Medicinal Organometallic Chemistry; Topics in Organometallic Chemistry; Jaouen, G., Metzler-Nolte, N., Eds.; Springer: Berlin/Heidelberg, Germany, 2010; pp. 81–117. [Google Scholar] [CrossRef]
- Osella, D.; Mahboobi, H.; Colangelo, D.; Cavigiolio, G.; Vessières, A.; Jaouen, G. FACS Analysis of Oxidative Stress Induced on Tumour Cells by SERMs. Inorganica Chim. Acta 2005, 358, 1993–1998. [Google Scholar] [CrossRef]
- Fouda, M.F.R.; Abd-Elzaher, M.M.; Abdelsamaia, R.A.; Labib, A.A. On the Medicinal Chemistry of Ferrocene. Appl. Organomet. Chem. 2007, 21, 613–625. [Google Scholar] [CrossRef]
- Hillard, E.A.; Vessières, A.; Top, S.; Pigeon, P.; Kowalski, K.; Huché, M.; Jaouen, G. Organometallic Diphenols: The Importance of the Organometallic Moiety on the Expression of a Cytotoxic Effect on Breast Cancer Cells. J. Organomet. Chem. 2007, 692, 1315–1326. [Google Scholar] [CrossRef]
- Tonolo, F.; Salmain, M.; Scalcon, V.; Top, S.; Pigeon, P.; Folda, A.; Caron, B.; McGlinchey, M.J.; Toillon, R.-A.; Bindoli, A.; et al. Small Structural Differences between Two Ferrocenyl Diphenols Determine Large Discrepancies of Reactivity and Biological Effects. ChemMedChem 2019, 14, 1717–1726. [Google Scholar] [CrossRef]
- Hamels, D.; Dansette, P.M.; Hillard, E.A.; Top, S.; Vessières, A.; Herson, P.; Jaouen, G.; Mansuy, D. Ferrocenyl Quinone Methides as Strong Antiproliferative Agents: Formation by Metabolic and Chemical Oxidation of Ferrocenyl Phenols. Angew. Chem. Int. Ed. 2009, 48, 9124–9126. [Google Scholar] [CrossRef]
- Zhang, J.; Li, X.; Han, X.; Liu, R.; Fang, J. Targeting the Thioredoxin System for Cancer Therapy. Trends Pharmacol. Sci. 2017, 38, 794–808. [Google Scholar] [CrossRef]
- Scalcon, V.; Salmain, M.; Folda, A.; Top, S.; Pigeon, P.; Shirley Lee, H.Z.; Jaouen, G.; Bindoli, A.; Vessières, A.; Rigobello, M.P. Tamoxifen-like Metallocifens Target the Thioredoxin System Determining Mitochondrial Impairment Leading to Apoptosis in Jurkat Cells. Met. Integr. Biometal Sci. 2017, 9, 949–959. [Google Scholar] [CrossRef]
- Kardeh, S.; Ashkani-Esfahani, S.; Alizadeh, A.M. Paradoxical Action of Reactive Oxygen Species in Creation and Therapy of Cancer. Eur. J. Pharmacol. 2014, 735, 150–168. [Google Scholar] [CrossRef]
- Carneiro, B.A.; El-Deiry, W.S. Targeting Apoptosis in Cancer Therapy. Nat. Rev. Clin. Oncol. 2020, 1–23. [Google Scholar] [CrossRef]
- Heurtault, B.; Saulnier, P.; Pech, B.; Venier-Julienne, M.-C.; Proust, J.-E.; Phan-Tan-Luu, R.; Benoît, J.-P. The Influence of Lipid Nanocapsule Composition on Their Size Distribution. Eur. J. Pharm. Sci. Off. J. Eur. Fed. Pharm. Sci. 2003, 18, 55–61. [Google Scholar] [CrossRef]
- Shinoda, K.; Saito, H. The Stability of O/W Type Emulsions as Functions of Temperature and the HLB of Emulsifiers: The Emulsification by PIT-Method. J. Colloid Interface Sci. 1969, 30, 258–263. [Google Scholar] [CrossRef]
- Anton, N.; Gayet, P.; Benoit, J.-P.; Saulnier, P. Nano-Emulsions and Nanocapsules by the PIT Method: An Investigation on the Role of the Temperature Cycling on the Emulsion Phase Inversion. Int. J. Pharm. 2007, 344, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Garcion, E.; Lamprecht, A.; Heurtault, B.; Paillard, A.; Aubert-Pouessel, A.; Denizot, B.; Menei, P.; Benoît, J.-P. A New Generation of Anticancer, Drug-Loaded, Colloidal Vectors Reverses Multidrug Resistance in Glioma and Reduces Tumor Progression in Rats. Mol. Cancer Ther. 2006, 5, 1710–1722. [Google Scholar] [CrossRef]
- Heurtault, B.; Saulnier, P.; Pech, B.; Benoît, J.P.; Proust, J. Interfacial Stability of Lipid Nanocapsules. Colloids Surf. B Biointerfaces 2003, 30, 225–235. [Google Scholar] [CrossRef]
- Vu, V.P.; Gifford, G.B.; Chen, F.; Benasutti, H.; Wang, G.; Groman, E.V.; Scheinman, R.; Saba, L.; Moghimi, S.M.; Simberg, D. Immunoglobulin Deposition on Biomolecule Corona Determines Complement Opsonization Efficiency of Preclinical and Clinical Nanoparticles. Nat. Nanotechnol. 2019, 14, 260–268. [Google Scholar] [CrossRef] [PubMed]
- Afshar-Kharghan, V. The Role of the Complement System in Cancer. J. Clin. Investig. 2017, 127, 780–789. [Google Scholar] [CrossRef] [PubMed]
- Passirani, C.; Barratt, G.; Devissaguet, J.P.; Labarre, D. Interactions of Nanoparticles Bearing Heparin or Dextran Covalently Bound to Poly(Methyl Methacrylate) with the Complement System. Life Sci. 1998, 62, 775–785. [Google Scholar] [CrossRef]
- Knop, K.; Hoogenboom, R.; Fischer, D.; Schubert, U.S. Poly(Ethylene Glycol) in Drug Delivery: Pros and Cons as Well as Potential Alternatives. Angew. Chem. Int. Ed. Engl. 2010, 49, 6288–6308. [Google Scholar] [CrossRef]
- Béduneau, A.; Saulnier, P.; Anton, N.; Hindré, F.; Passirani, C.; Rajerison, H.; Noiret, N.; Benoit, J.-P. Pegylated Nanocapsules Produced by an Organic Solvent-Free Method: Evaluation of Their Stealth Properties. Pharm. Res. 2006, 23, 2190–2199. [Google Scholar] [CrossRef] [PubMed]
- Lainé, A.-L.; Gravier, J.; Henry, M.; Sancey, L.; Béjaud, J.; Pancani, E.; Wiber, M.; Texier, I.; Coll, J.-L.; Benoit, J.-P.; et al. Conventional versus Stealth Lipid Nanoparticles: Formulation and in Vivo Fate Prediction through FRET Monitoring. J. Control. Release Off. J. Control. Release Soc. 2014, 188, 1–8. [Google Scholar] [CrossRef]
- Perrier, T.; Saulnier, P.; Fouchet, F.; Lautram, N.; Benoît, J.-P. Post-Insertion into Lipid NanoCapsules (LNCs): From Experimental Aspects to Mechanisms. Int. J. Pharm. 2010, 396, 204–209. [Google Scholar] [CrossRef]
- Resnier, P.; LeQuinio, P.; Lautram, N.; André, E.; Gaillard, C.; Bastiat, G.; Benoit, J.-P.; Passirani, C. Efficient in Vitro Gene Therapy with PEG SiRNA Lipid Nanocapsules for Passive Targeting Strategy in Melanoma. Biotechnol. J. 2014, 9, 1389–1401. [Google Scholar] [CrossRef] [PubMed]
- Vonarbourg, A.; Passirani, C.; Saulnier, P.; Simard, P.; Leroux, J.C.; Benoit, J.P. Evaluation of Pegylated Lipid Nanocapsules versus Complement System Activation and Macrophage Uptake. J. Biomed. Mater. Res. A 2006, 78, 620–628. [Google Scholar] [CrossRef]
- Thakur, S.; Kesharwani, P.; Tekade, R.K.; Jain, N.K. Impact of Pegylation on Biopharmaceutical Properties of Dendrimers. Polymer 2015, 59, 67–92. [Google Scholar] [CrossRef]
- Huynh, N.T.; Morille, M.; Bejaud, J.; Legras, P.; Vessieres, A.; Jaouen, G.; Benoit, J.-P.; Passirani, C. Treatment of 9L Gliosarcoma in Rats by Ferrociphenol-Loaded Lipid Nanocapsules Based on a Passive Targeting Strategy via the EPR Effect. Pharm. Res. 2011, 28, 3189–3198. [Google Scholar] [CrossRef]
- Morille, M.; Montier, T.; Legras, P.; Carmoy, N.; Brodin, P.; Pitard, B.; Benoît, J.-P.; Passirani, C. Long-Circulating DNA Lipid Nanocapsules as New Vector for Passive Tumor Targeting. Biomaterials 2010, 31, 321–329. [Google Scholar] [CrossRef]
- Pasut, G.; Veronese, F.M. State of the Art in PEGylation: The Great Versatility Achieved after Forty Years of Research. J. Control. Release Off. J. Control. Release Soc. 2012, 161, 461–472. [Google Scholar] [CrossRef] [PubMed]
- Béduneau, A.; Saulnier, P.; Hindré, F.; Clavreul, A.; Leroux, J.-C.; Benoit, J.-P. Design of Targeted Lipid Nanocapsules by Conjugation of Whole Antibodies and Antibody Fab’ Fragments. Biomaterials 2007, 28, 4978–4990. [Google Scholar] [CrossRef]
- Resnier, P.; Lepeltier, E.; Emina, A.L.; Galopin, N.; Bejaud, J.; David, S.; Ballet, C.; Benvegnu, T.; Pecorari, F.; Chourpa, I.; et al. Model Affitin and PEG Modifications onto SiRNA Lipid Nanocapsules: Cell Uptake and in Vivo Biodistribution Improvements. RSC Adv. 2019, 9, 27264–27278. [Google Scholar] [CrossRef]
- Benoit, J.-P.; Perrier, T. Method for Preparing Functionalized Lipid Capsules. U.S. Patent 9333181B2, 5 October 2016. Available online: https://patents.google.com/patent/US9333181B2/en (accessed on 9 June 2020).
- Bechara, C.; Sagan, S. Cell-Penetrating Peptides: 20 Years Later, Where Do We Stand? FEBS Lett. 2013, 587, 1693–1702. [Google Scholar] [CrossRef] [PubMed]
- Karim, R.; Lepeltier, E.; Esnault, L.; Pigeon, P.; Lemaire, L.; Lépinoux-Chambaud, C.; Clere, N.; Jaouen, G.; Eyer, J.; Piel, G.; et al. Enhanced and Preferential Internalization of Lipid Nanocapsules into Human Glioblastoma Cells: Effect of a Surface-Functionalizing NFL Peptide. Nanoscale 2018, 10, 13485–13501. [Google Scholar] [CrossRef]
- Topin-Ruiz, S.; Mellinger, A.; Lepeltier, E.; Bourreau, C.; Fouillet, J.; Riou, J.; Jaouen, G.; Martin, L.; Passirani, C.; Clere, N. P722 Ferrocifen Loaded Lipid Nanocapsules Improve Survival of Murine Xenografted-Melanoma via a Potentiation of Apoptosis and an Activation of CD8+ T Lymphocytes. Int. J. Pharm. 2021, 593, 120111. [Google Scholar] [CrossRef]
- Huynh, N.T.; Passirani, C.; Allard-Vannier, E.; Lemaire, L.; Roux, J.; Garcion, E.; Vessieres, A.; Benoit, J.-P. Administration-Dependent Efficacy of Ferrociphenol Lipid Nanocapsules for the Treatment of Intracranial 9L Rat Gliosarcoma. Int. J. Pharm. 2012, 423, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Roger, M.; Clavreul, A.; Huynh, N.T.; Passirani, C.; Schiller, P.; Vessières, A.; Montero-Menei, C.; Menei, P. Ferrociphenol Lipid Nanocapsule Delivery by Mesenchymal Stromal Cells in Brain Tumor Therapy. Int. J. Pharm. 2012, 423, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Lainé, A.-L.; Clavreul, A.; Rousseau, A.; Tétaud, C.; Vessieres, A.; Garcion, E.; Jaouen, G.; Aubert, L.; Guilbert, M.; Benoit, J.-P.; et al. Inhibition of Ectopic Glioma Tumor Growth by a Potent Ferrocenyl Drug Loaded into Stealth Lipid Nanocapsules. Nanomed. Nanotechnol. Biol. Med. 2014, 10, 1667–1677. [Google Scholar] [CrossRef]
- Omuro, A.; DeAngelis, L.M. Glioblastoma and Other Malignant Gliomas: A Clinical Review. JAMA 2013, 310, 1842–1850. [Google Scholar] [CrossRef]
- Vinchon-Petit, S.; Jarnet, D.; Paillard, A.; Benoit, J.-P.; Garcion, E.; Menei, P. In Vivo Evaluation of Intracellular Drug-Nanocarriers Infused into Intracranial Tumours by Convection-Enhanced Delivery: Distribution and Radiosensitisation Efficacy. J. Neurooncol. 2010, 97, 195–205. [Google Scholar] [CrossRef]
- Allard, E.; Passirani, C.; Benoit, J.-P. Convection-Enhanced Delivery of Nanocarriers for the Treatment of Brain Tumors. Biomaterials 2009, 30, 2302–2318. [Google Scholar] [CrossRef]
- MacKay, J.A.; Deen, D.F.; Szoka, F.C. Distribution in Brain of Liposomes after Convection Enhanced Delivery; Modulation by Particle Charge, Particle Diameter, and Presence of Steric Coating. Brain Res. 2005, 1035, 139–153. [Google Scholar] [CrossRef] [PubMed]
- Ishida, T.; Kiwada, H. Accelerated Blood Clearance (ABC) Phenomenon upon Repeated Injection of PEGylated Liposomes. Int. J. Pharm. 2008, 354, 56–62. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.; Qin, Z.; Wu, J.; Li, L.; Jin, Q.; Ji, J. Zwitterionic Stealth Peptide-Protected Gold Nanoparticles Enable Long Circulation without the Accelerated Blood Clearance Phenomenon. Biomater. Sci. 2017, 6, 200–206. [Google Scholar] [CrossRef]
- Bruyère, C.; Mathieu, V.; Vessières, A.; Pigeon, P.; Top, S.; Jaouen, G.; Kiss, R. Ferrocifen Derivatives That Induce Senescence in Cancer Cells: Selected Examples. J. Inorg. Biochem. 2014, 141, 144–151. [Google Scholar] [CrossRef] [PubMed]
- Sun, Y.-S.; Zhao, Z.; Yang, Z.-N.; Xu, F.; Lu, H.-J.; Zhu, Z.-Y.; Shi, W.; Jiang, J.; Yao, P.-P.; Zhu, H.-P. Risk Factors and Preventions of Breast Cancer. Int. J. Biol. Sci. 2017, 13, 1387–1397. [Google Scholar] [CrossRef]
- Scully, O.J.; Bay, B.-H.; Yip, G.; Yu, Y. Breast Cancer Metastasis. Cancer Genomics Proteomics 2012, 9, 311–320. [Google Scholar]
- Yang, Y.; Pan, W.; Tang, X.; Wu, S.; Sun, X. A Meta-Analysis of Randomized Controlled Trials Comparing the Efficacy and Safety of Anastrozole versus Tamoxifen for Breast Cancer. Oncotarget 2017, 8, 48362–48374. [Google Scholar] [CrossRef]
- Pautu, V.; Leonetti, D.; Lepeltier, E.; Clere, N.; Passirani, C. Nanomedicine as a Potent Strategy in Melanoma Tumor Microenvironment. Pharmacol. Res. 2017, 126, 31–53. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Yang, G.; Zhang, Y.-Y.; Yao, Y.; Dong, L.-H. A Comparison between Oral Chemotherapy Combined with Radiotherapy and Radiotherapy for Newly Diagnosed Glioblastoma: A Systematic Review and Meta-Analysis. Medicine 2017, 96, e8444. [Google Scholar] [CrossRef] [PubMed]
- Hau, E.; Shen, H.; Clark, C.; Graham, P.H.; Koh, E.-S.; L McDonald, K. The Evolving Roles and Controversies of Radiotherapy in the Treatment of Glioblastoma. J. Med. Radiat. Sci. 2016, 63, 114–123. [Google Scholar] [CrossRef]
- Grauer, O.; Jaber, M.; Hess, K.; Weckesser, M.; Schwindt, W.; Maring, S.; Wölfer, J.; Stummer, W. Combined Intracavitary Thermotherapy with Iron Oxide Nanoparticles and Radiotherapy as Local Treatment Modality in Recurrent Glioblastoma Patients. J. Neurooncol. 2019, 141, 83–94. [Google Scholar] [CrossRef]
- Lee, E.F.; Harris, T.J.; Tran, S.; Evangelista, M.; Arulananda, S.; John, T.; Ramnac, C.; Hobbs, C.; Zhu, H.; Gunasingh, G.; et al. BCL-XL and MCL-1 Are the Key BCL-2 Family Proteins in Melanoma Cell Survival. Cell Death Dis. 2019, 10, 342. [Google Scholar] [CrossRef]
- Ilmonen, S.; Hernberg, M.; Pyrhönen, S.; Tarkkanen, J.; Asko-Seljavaara, S. Ki-67, Bcl-2 and P53 Expression in Primary and Metastatic Melanoma. Melanoma Res. 2005, 15, 375–381. [Google Scholar] [CrossRef]
- David, S.; Resnier, P.; Guillot, A.; Pitard, B.; Benoit, J.-P.; Passirani, C. SiRNA LNCs--a Novel Platform of Lipid Nanocapsules for Systemic SiRNA Administration. Eur. J. Pharm. Biopharm. Off. J. Arbeitsgemeinschaft Pharm. Verfahrenstechnik EV 2012, 81, 448–452. [Google Scholar] [CrossRef][Green Version]
- David, S.; Carmoy, N.; Resnier, P.; Denis, C.; Misery, L.; Pitard, B.; Benoit, J.-P.; Passirani, C.; Montier, T. In Vivo Imaging of DNA Lipid Nanocapsules after Systemic Administration in a Melanoma Mouse Model. Int. J. Pharm. 2012, 423, 108–115. [Google Scholar] [CrossRef][Green Version]
- David, S.; Passirani, C.; Carmoy, N.; Morille, M.; Mevel, M.; Chatin, B.; Benoit, J.-P.; Montier, T.; Pitard, B. DNA Nanocarriers for Systemic Administration: Characterization and in Vivo Bioimaging in Healthy Mice. Mol. Ther. Nucleic Acids 2013, 2, e64. [Google Scholar] [CrossRef] [PubMed]
- Lagarce, F.; Passirani, C. Nucleic-Acid Delivery Using Lipid Nanocapsules. Curr. Pharm. Biotechnol. 2016, 17, 723–727. [Google Scholar] [CrossRef]
- Roger, M.; Clavreul, A.; Venier-Julienne, M.-C.; Passirani, C.; Sindji, L.; Schiller, P.; Montero-Menei, C.; Menei, P. Mesenchymal Stem Cells as Cellular Vehicles for Delivery of Nanoparticles to Brain Tumors. Biomaterials 2010, 31, 8393–8401. [Google Scholar] [CrossRef] [PubMed]
- Roger, M.; Clavreul, A.; Sindji, L.; Chassevent, A.; Schiller, P.C.; Montero-Menei, C.N.; Menei, P. In Vitro and in Vivo Interactions between Glioma and Marrow-Isolated Adult Multilineage Inducible (MIAMI) Cells. Brain Res. 2012, 1473, 193–203. [Google Scholar] [CrossRef] [PubMed]
- Roger, M.; Clavreul, A.; Venier-Julienne, M.-C.; Passirani, C.; Montero-Menei, C.; Menei, P. The Potential of Combinations of Drug-Loaded Nanoparticle Systems and Adult Stem Cells for Glioma Therapy. Biomaterials 2011, 32, 2106–2116. [Google Scholar] [CrossRef] [PubMed]
- Gengenbacher, N.; Singhal, M.; Augustin, H.G. Preclinical Mouse Solid Tumour Models: Status Quo, Challenges and Perspectives. Nat. Rev. Cancer 2017, 17, 751–765. [Google Scholar] [CrossRef]
- Decaudin, D. Primary Human Tumor Xenografted Models (‘tumorgrafts’) for Good Management of Patients with Cancer. Anticancer Drugs 2011, 22, 827–841. [Google Scholar] [CrossRef] [PubMed]
- Byrne, A.T.; Alférez, D.G.; Amant, F.; Annibali, D.; Arribas, J.; Biankin, A.V.; Bruna, A.; Budinská, E.; Caldas, C.; Chang, D.K.; et al. Interrogating Open Issues in Cancer Precision Medicine with Patient-Derived Xenografts. Nat. Rev. Cancer 2017, 17, 254–268. [Google Scholar] [CrossRef] [PubMed]
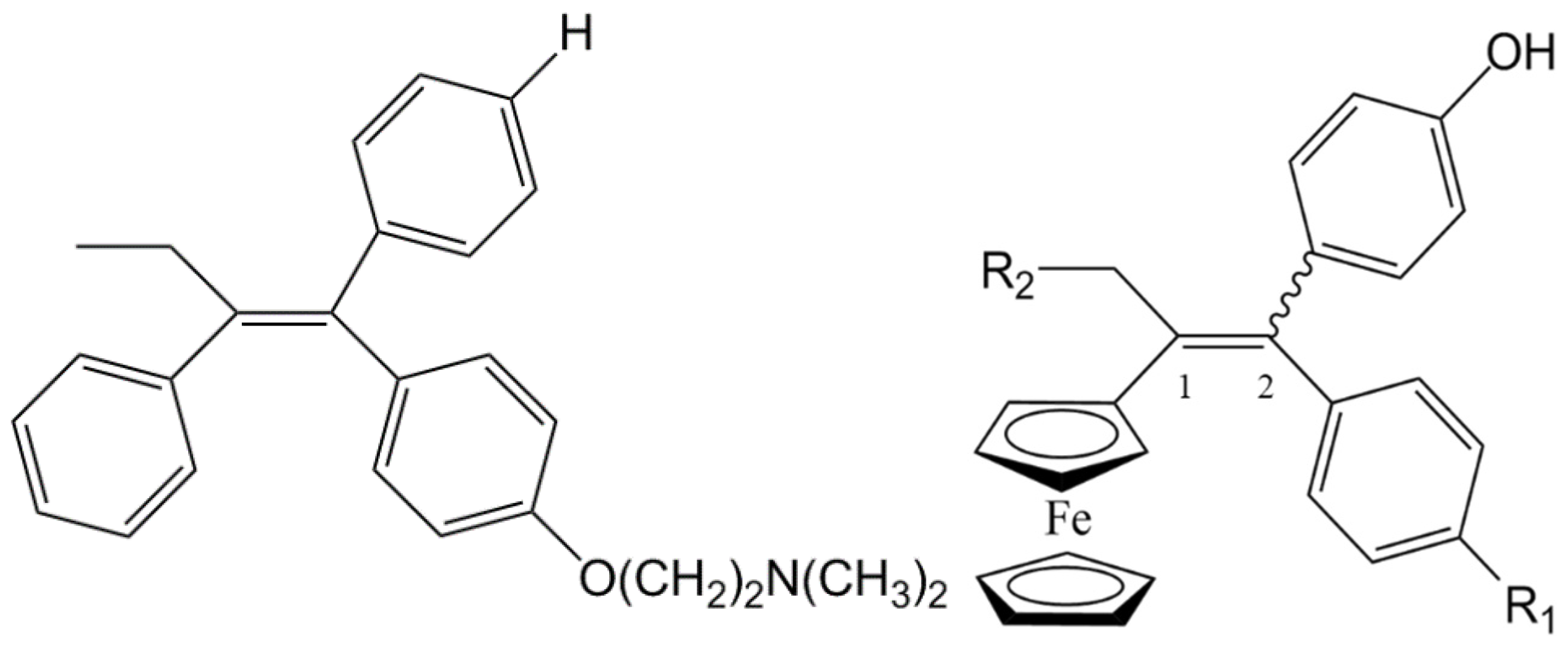
| Ferrocifen | Quinone Methide |
|---|---|
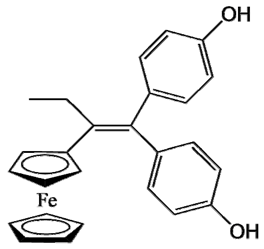 P5 (FcdiOH) |  Vinyl-QM-P5 |
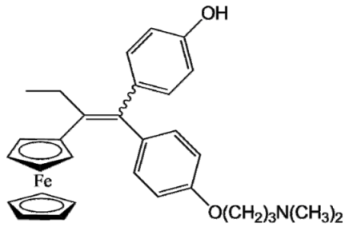 P15 (FcOHTam) | 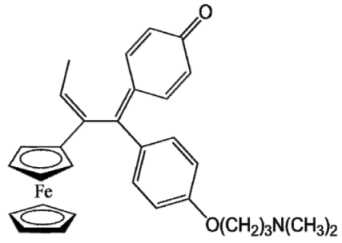 Vinyl-QM-P15 |
 P53 (FcTriOH) |  Furane-QM-P53 |
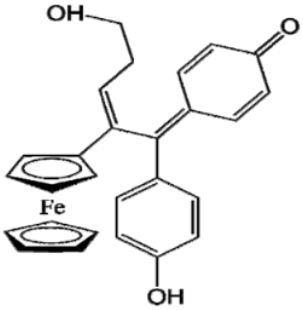 Vinyl-QM-P53 | |
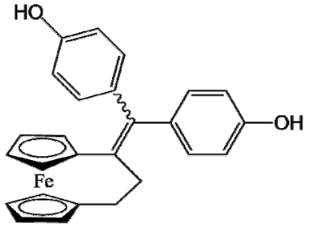 DP1 (Ansa-FcdiOH) | 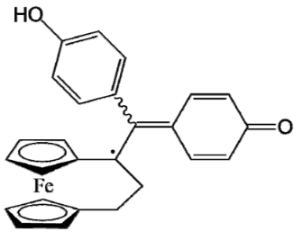 Radical DP1 |
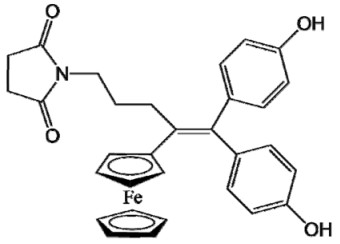 P722 | 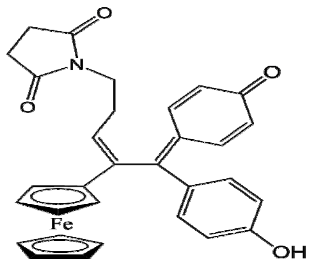 QM-P722 |
| Cell Line | P5 | P15 | |
|---|---|---|---|
| Breast | MCF7 | [55] | [55] |
| MDA-MB-231 | [56] | [56] | |
| Glioblastoma | U87MG | [57] | - |
| 9L | [58] | - | |
| Melanoma | WM35 | [37] | [37] |
| WM793 | [37] | [37] | |
| WM9 | [37] | [37] | |
| SK-Mel28 | [59] | - | |
| Ovarian | A2780 | [56] | - |
| A2780cisR | [56] | - | |
| IC50 (µM) | |||
| 0.5–2 | |||
| 2–10 | |||
| Cell Line | DP1 [51] | P53 [50] | P722 [48] | |
|---|---|---|---|---|
| Breast | MCF7 | |||
| MDA-MB-231 | ||||
| CNS | SF-295 | |||
| SF-539 | ||||
| SF-268 | ||||
| Melanoma | SK-MEL-2 | |||
| SK-MEL-28 | ||||
| SK-MEL-5 | ||||
| Ovarian | IGROV1 | |||
| SK-OV-3 | ||||
| Leukemia | HL-60(TB) | |||
| MOLT-4 | ||||
| GI50 (µM) | ||||
| <0.1 | ||||
| 0.1–0.5 | ||||
| 0.5–2 | ||||
| 2–10 | ||||
| >10 | ||||
| Ferrocifens Used (Names) | Pathology | LNC Formulation | Encapsulation Efficiency (Drug Loading) | In Vivo Studies |
|---|---|---|---|---|
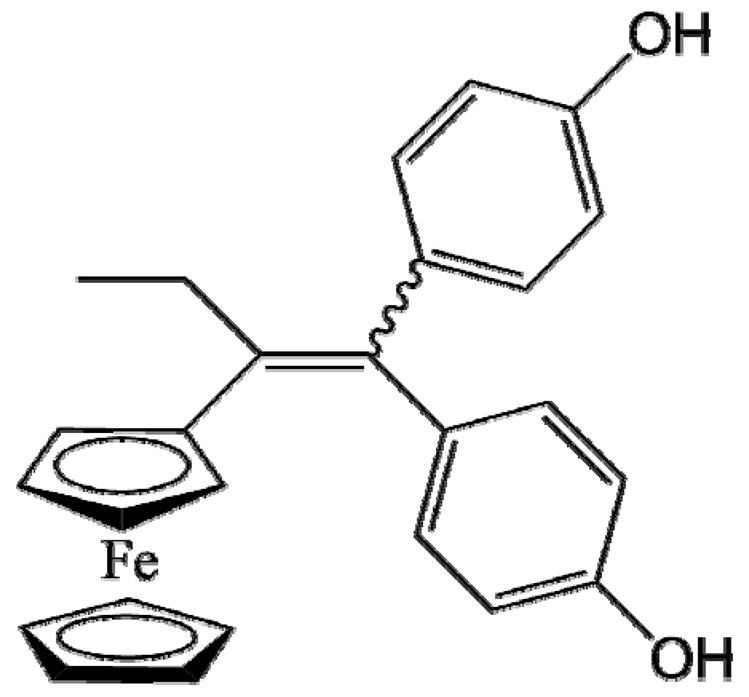 P5 (FcdiOH) | Glioblastoma (9L glioma cells) | Conventional LNCs | >98% (1.0 mg/g or 6.5 mg/g) | Single intratumoral injection (400 µL); ectopic model [39] |
| Glioblastoma (9L glioma cells) | Conventional LNCs | 96% (1.0 mg/g or 6.5 mg/g) | Single intracranial injection by CED (60 µL, 0.36 mg/rat) + 3 irradiations of 6 Gy doses; orthotopic model [58] | |
| Glioblastoma (9L glioma cells) | NFL-TBS-63 peptide-coated LNCs | (6.1 mg/mL) | Intracarotid injection (400 µL, 2.5 mg/rat); orthotopic model [28] | |
| (5.5 mg/mL) | Injection by CED (60 µL, 0.36 mg/kg rat); orthotopic model [28] | |||
| Glioblastoma (9L glioma cells) | Stealth LNCs | >98% (6.5 mg/g) | Single intravenous injection (400 µL, 2.4 mg/rat); ectopic model [88] | |
| Single intravenous injection (400 µL, 2.4 mg/rat); orthotopic model [88] | ||||
| Glioblastoma (9L glioma cells) | Stealth LNCs and conventional LNCs | (6.5 mg/g) | Intracarotid injection (400 µL, 2.4 mg/rat), orthotopic model [97] | |
| CED injection (60 µL, 0.36 mg/rat), orthotopic model [97] | ||||
| Glioblastoma (U87MG cells) | LNC-loaded MIAMI cells | (2.6 mg/mL) | Intratumoral injection (100 µL), heterotopic model [98] | |
| Glioblastoma (U87MG cells) | LNC-loaded MIAMI cells | (6.0 mg/g of LNCs) 20 pg of P5/MIAMI cells | Intratumoral injection (3.6 µg/mouse), orthotopic model [57] | |
| Melanoma (SK-Mel28 human cells) | Stealth LNCs | 92% (6.0 mg/mL) | Repeated intravenous injection (45 mg/kg); ectopic model [59] | |
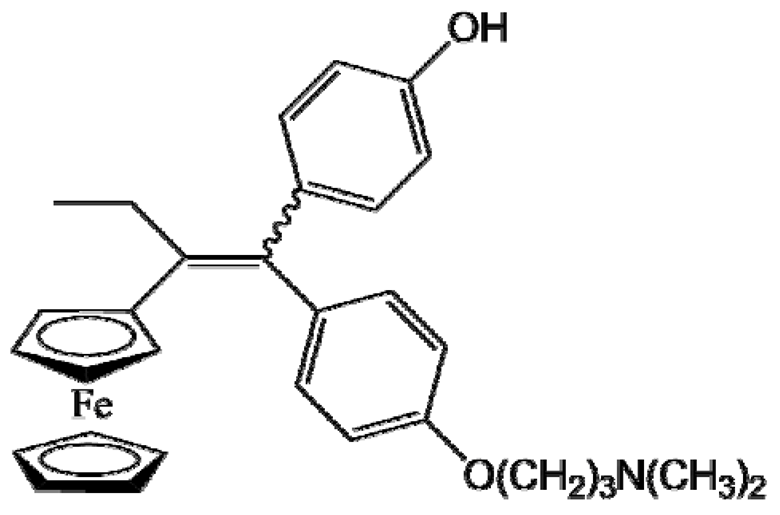 P15 (FcOHTam) | Breast cancer (MDA-MB-231 cells) | Stealth LNCs | 100% (8.0 mg/mL) | Repeated intraperitoneal injection (2×) (20 mg/kg); ectopic model [55] |
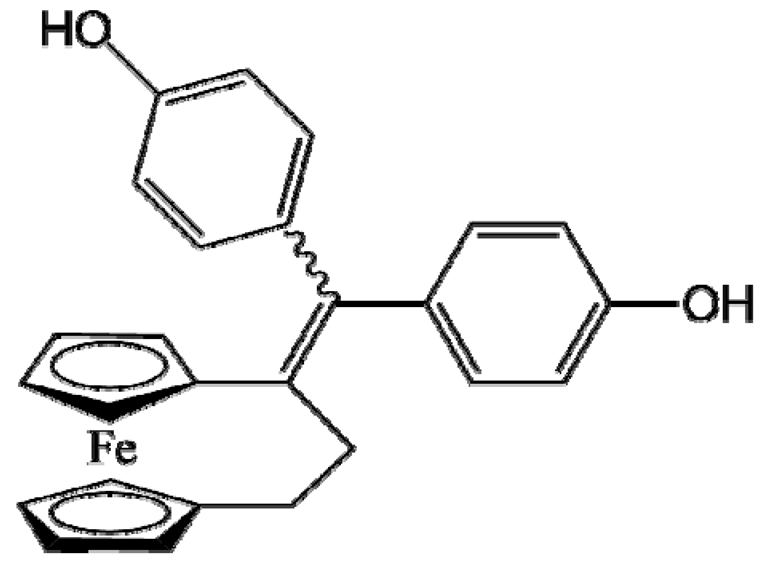 DP1 (Ansa-FcdiOH) | Melanoma (SK-Mel28 human cells) | Stealth LNCs | 96% (6.0 mg/mL) | Repeated intravenous injection (45 mg/kg); ectopic model [59] |
| Stealth LNCs with Bcl-2 siRNA | 85% (6.0 mg/mL) | Repeated intravenous injection (45 mg/kg); ectopic model [59] | ||
| Glioblastoma (9L glioma cells) | Stealth LNCs | (6.4 mg/g) | Repeated intravenous injection (10×) (20 mg/kg); ectopic model [99] | |
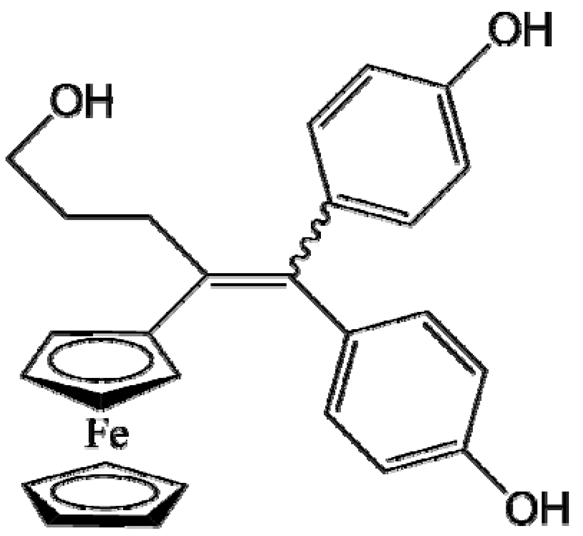 P53 (FctriOH) | Glioblastoma (U87MG cells) | NFL-TBS40-63 peptide-coated LNCs | >99% (2.67 mg/g) | Intravenous injection (2×) (70 µL, 20 mg/kg); ectopic model [95] |
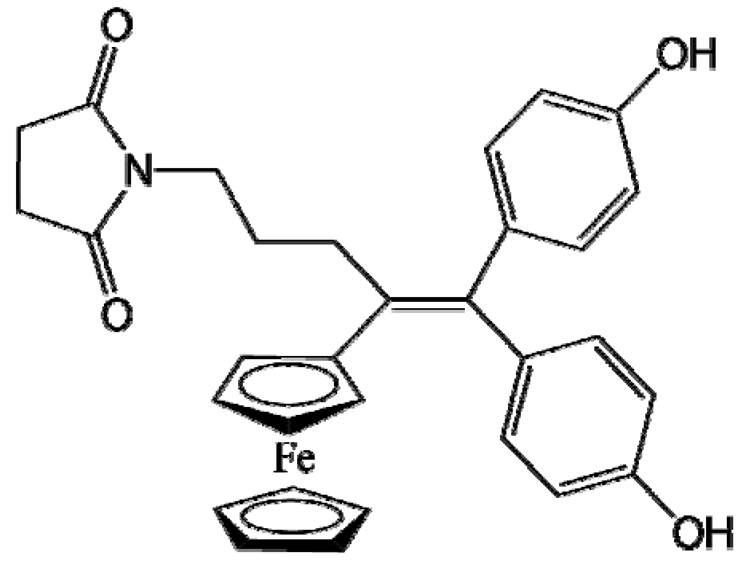 P722 | Melanoma (B16F10 cells) | Stealth LNCs | 65% | Intraperitoneal injection (7 mg/kg); orthotopic model [96] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Idlas, P.; Lepeltier, E.; Jaouen, G.; Passirani, C. Ferrocifen Loaded Lipid Nanocapsules: A Promising Anticancer Medication against Multidrug Resistant Tumors. Cancers 2021, 13, 2291. https://doi.org/10.3390/cancers13102291
Idlas P, Lepeltier E, Jaouen G, Passirani C. Ferrocifen Loaded Lipid Nanocapsules: A Promising Anticancer Medication against Multidrug Resistant Tumors. Cancers. 2021; 13(10):2291. https://doi.org/10.3390/cancers13102291
Chicago/Turabian StyleIdlas, Pierre, Elise Lepeltier, Gérard Jaouen, and Catherine Passirani. 2021. "Ferrocifen Loaded Lipid Nanocapsules: A Promising Anticancer Medication against Multidrug Resistant Tumors" Cancers 13, no. 10: 2291. https://doi.org/10.3390/cancers13102291
APA StyleIdlas, P., Lepeltier, E., Jaouen, G., & Passirani, C. (2021). Ferrocifen Loaded Lipid Nanocapsules: A Promising Anticancer Medication against Multidrug Resistant Tumors. Cancers, 13(10), 2291. https://doi.org/10.3390/cancers13102291






