Monoclonal Antibody-Based Immunotherapy and Its Role in the Development of Cardiac Toxicity
Abstract
Simple Summary
Abstract
1. Introduction
2. Contemporary Cancer Immunotherapy
3. Monoclonal Antibodies (mAbs) and Their Application in Cancer Immunotherapy
3.1. Development of Therapeutic mAbs
3.2. Types of mAbs
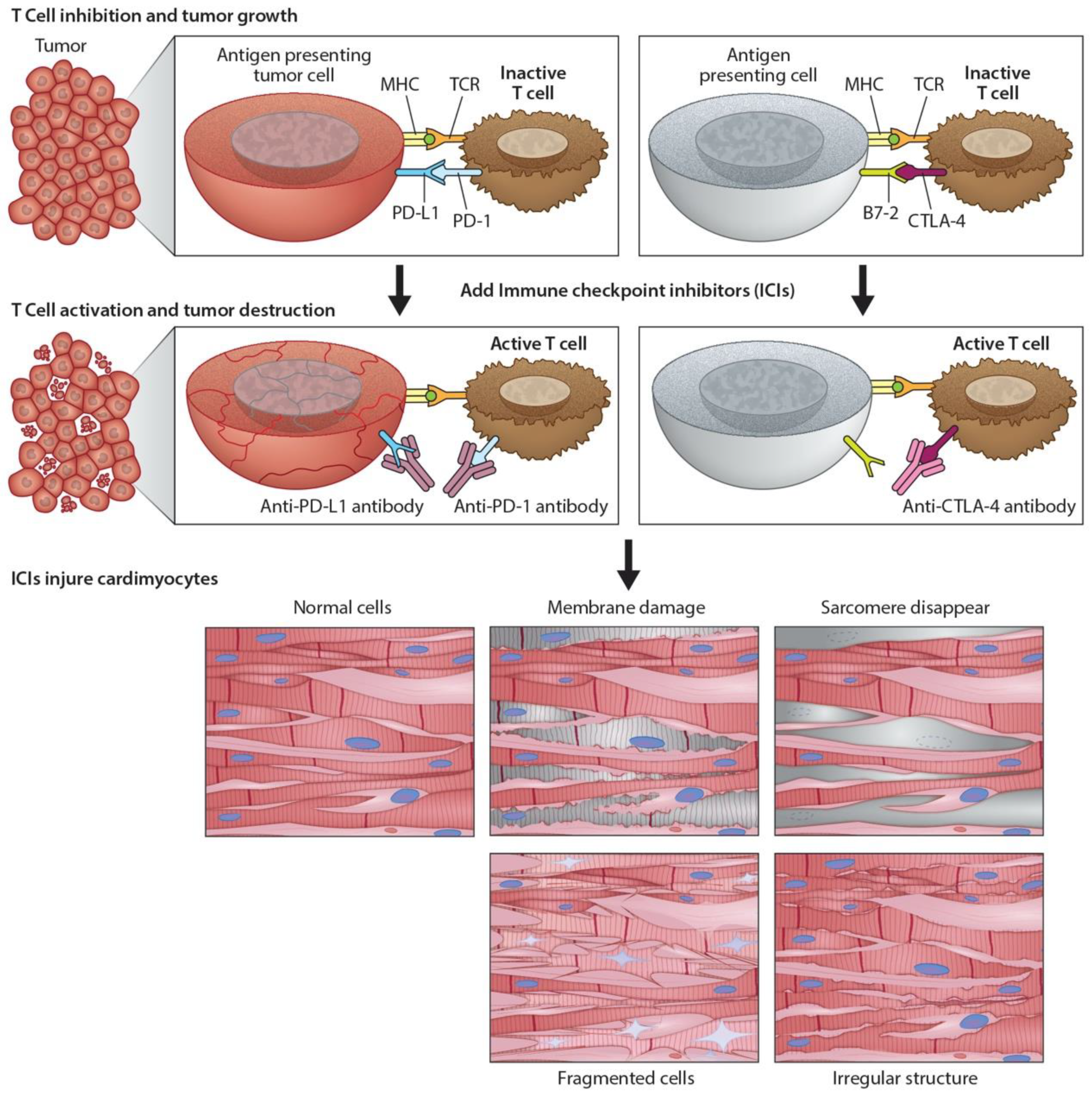
3.3. Mechanism of Action of mAbs in Cancer Treatment
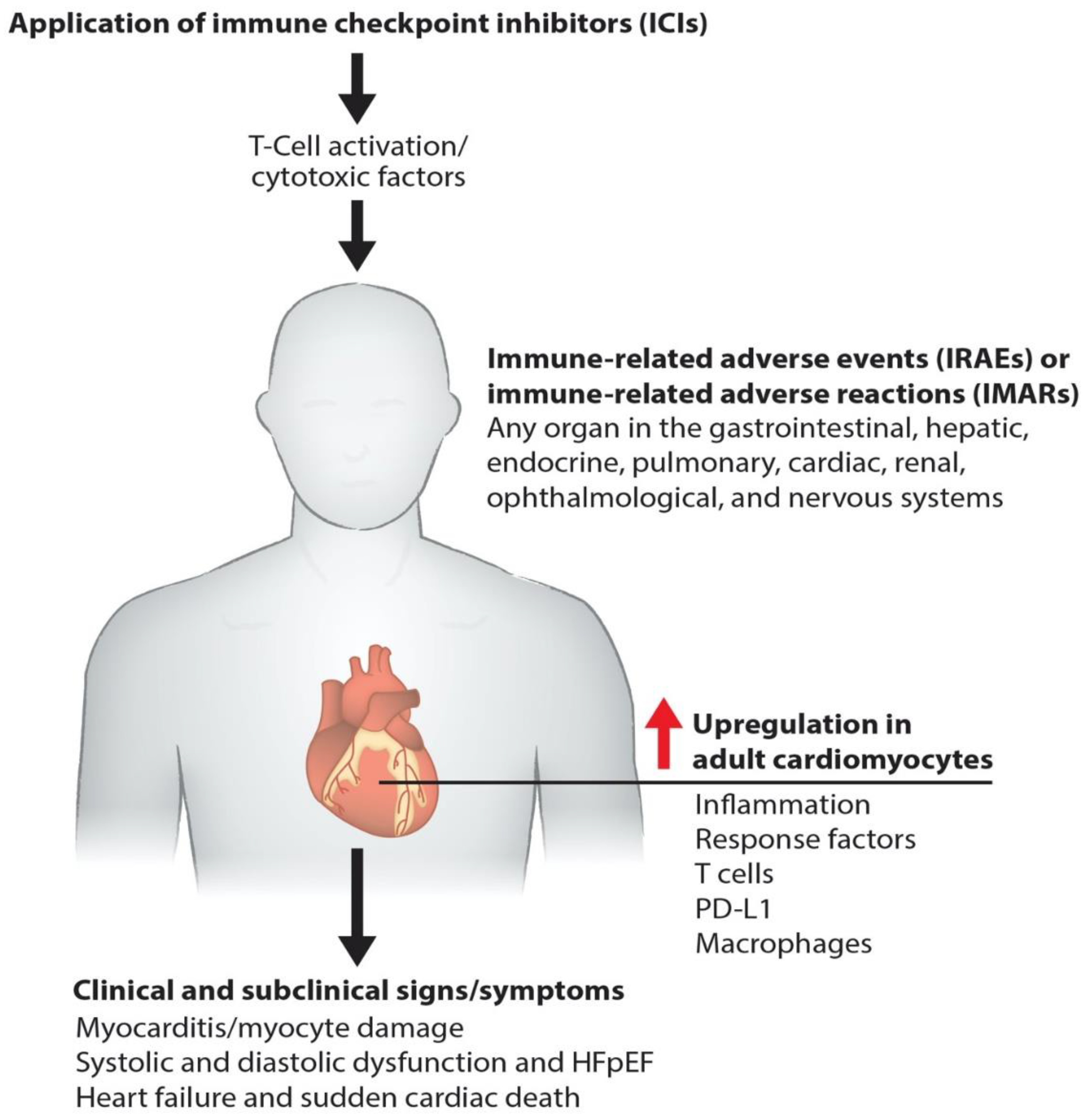
3.4. Monoclonal Antibodies (mAbs) in Cancer Treatment and the Development of Cardiac Toxicity
3.5. Cardio-Oncology: Clinical Presentation, Diagnosis, and Management of Cardiotoxicity
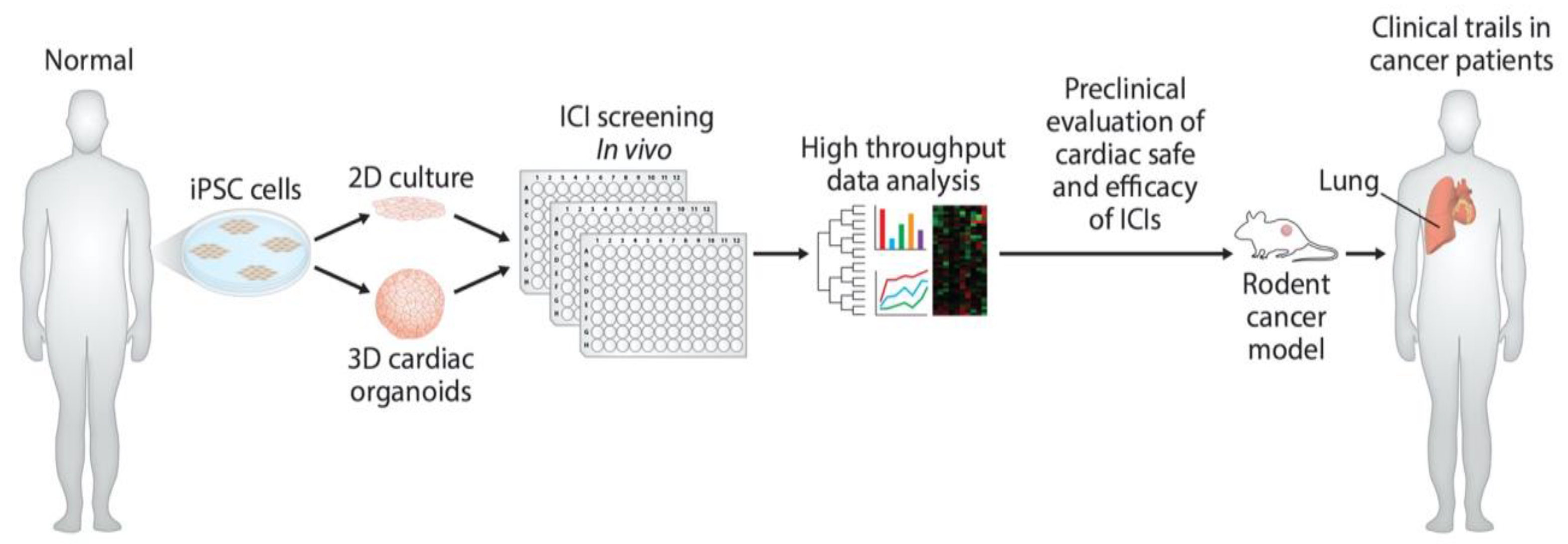
4. Preclinical Evaluation of mAbs in the Promotion of Cardiotoxicity
5. Future Directions
6. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ACT | Adoptive cell therapy |
| TIL | Tumor-Infiltrating Lymphocyte |
| TCR | T-Cell Receptor |
| CAR | Chimeric Antigen Receptor |
| hiPSC-CMs | human-induced pluripotent stem cell-derived cardiomyocytes |
| CTLA-4 | Cytotoxic T-lymphocyte-associated antigen 4 |
| ASE | American Society of Echocardiography |
| EACVI | European Association of Cardiovascular Imaging |
| IRAEs | Immune-related adverse events |
| IMARs | Immune-related adverse reactions |
| ICI | Immune checkpoint inhibitors |
| LVD | Left ventricular dysfunction |
| HFpEF | Heart failure with preserved ejection fraction |
| PD-1 | Programmed cell death protein-1 |
| PD-L1 | Programmed death ligand-1 |
References
- Lenneman, C.G.; Kimmick, G.G.; Sawyer, D.B. Epidemiology of Cardio-Oncology. In Cardio-Oncology: The Clinical Overlap of Cancer and Heart Disease; Kimmick, G.G., Lenihan, D.J., Sawyer, D.B., Mayer, E.L., Hershman, D.L., Eds.; Springer: Cham, Switzerland, 2017; pp. 1–14. [Google Scholar]
- Palaskas, N.; Lopez-Mattei, J.; Durand, J.B.; Iliescu, C.; Deswal, A. Immune Checkpoint Inhibitor Myocarditis: Pathophysiological Characteristics, Diagnosis, and Treatment. J. Am. Heart Assoc. 2020, 9, e013757. [Google Scholar] [CrossRef]
- Zarifa, A.; Lopez-Mattei, J.; Palaskas, N.; Iliescu, C.; Durand, J.B.; Kim, P.Y. Immune Checkpoint Inhibitors (ICIs)-Related Cardiotoxicity. Adv. Exp. Med. Biol. 2020, 1244, 277–285. [Google Scholar]
- Robert, C. A decade of immune-checkpoint inhibitors in cancer therapy. Nat. Commun. 2020, 11, 3801. [Google Scholar] [CrossRef]
- Dong, J.; Chen, H. Cardiotoxicity of Anticancer Therapeutics. Front. Cardiovasc. Med. 2018, 5, 9. [Google Scholar] [CrossRef]
- Jain, V.; Bahia, J.; Mohebtash, M.; Barac, A. Cardiovascular Complications Associated with Novel Cancer Immunotherapies. Curr. Treat. Options Cardiovasc. Med. 2017, 19, 36. [Google Scholar] [CrossRef]
- Said, R.; Nickolich, M.; Lenihan, D.J.; Tsimberidou, A.M. Cardiotoxicity of Anticancer Therapies. In Cardio-Oncology: The Clinical Overlap of Cancer and Heart Disease; Kimmick, G.G., Lenihan, D.J., Sawyer, D.B., Mayer, E.L., Hershman, D.L., Eds.; Springer: Cham, Switzerland, 2017; pp. 15–42. [Google Scholar]
- Berger, M.; Shankar, V.; Vafai, A. Therapeutic applications of monoclonal antibodies. Am. J. Med. Sci. 2002, 324, 14–30. [Google Scholar] [CrossRef]
- Hafeez, U.; Gan, H.K.; Scott, A.M. Monoclonal antibodies as immunomodulatory therapy against cancer and autoimmune diseases. Curr. Opin. Pharmacol. 2018, 41, 114–121. [Google Scholar] [CrossRef]
- Scott, A.M.; Wolchok, J.D.; Old, L.J. Antibody therapy of cancer. Nat. Rev. Cancer 2012, 12, 278–287. [Google Scholar] [CrossRef]
- Rohaan, M.W.; Wilgenhof, S.; Haanen, J. Adoptive cellular therapies: The current landscape. Virchows Arch. Int. J. Pathol. 2019, 474, 449–461. [Google Scholar] [CrossRef]
- Feins, S.; Kong, W.; Williams, E.F.; Milone, M.C.; Fraietta, J.A. An introduction to chimeric antigen receptor (CAR) T-cell immunotherapy for human cancer. Am. J. Hematol. 2019, 94, S3–S9. [Google Scholar] [CrossRef]
- Stanculeanu, D.L.; Daniela, Z.; Lazescu, A.; Bunghez, R.; Anghel, R. Development of new immunotherapy treatments in different cancer types. J. Med. Life 2016, 9, 240–248. [Google Scholar]
- Hollingsworth, R.E.; Jansen, K. Turning the corner on therapeutic cancer vaccines. NPJ Vaccines 2019, 4, 7. [Google Scholar] [CrossRef]
- Zhang, X.; Soori, G.; Dobleman, T.J.; Xiao, G.G. The application of monoclonal antibodies in cancer diagnosis. Exp. Rev. Mol. Diagn. 2014, 14, 97–106. [Google Scholar] [CrossRef]
- Lu, R.M.; Hwang, Y.C.; Liu, I.J.; Lee, C.C.; Tsai, H.Z.; Li, H.J.; Wu, H.C. Development of therapeutic antibodies for the treatment of diseases. J. Biomed. Sci. 2020, 27, 1. [Google Scholar] [CrossRef]
- Chen, Y.; Hu, Z.; Wang, Q.; Ge, Y.; Bai, L.; Wang, X.; Zhang, X. Generation and characterization of four novel monoclonal antibodies against human programmed death-1 molecule. Hybridoma 2010, 29, 153–160. [Google Scholar] [CrossRef]
- Wei, G.; Wang, J.; Huang, H.; Zhao, Y. Novel immunotherapies for adult patients with B-lineage acute lymphoblastic leukemia. J. Hematol. Oncol. 2017, 10, 150. [Google Scholar] [CrossRef]
- Jin, J.; Park, G.; Park, J.B.; Kim, S.; Kim, H.; Chung, J. An anti-EGFR x cotinine bispecific antibody complexed with cotinine-conjugated duocarmycin inhibits growth of EGFR-positive cancer cells with KRAS mutations. Exp. Mol. Med. 2018, 50, 1–14. [Google Scholar] [CrossRef]
- Marhelava, K.; Pilch, Z.; Bajor, M.; Graczyk-Jarzynka, A.; Zagozdzon, R. Targeting Negative and Positive Immune Checkpoints with Monoclonal Antibodies in Therapy of Cancer. Cancers 2019, 11, 1756. [Google Scholar] [CrossRef]
- Pento, J.T. Monoclonal Antibodies for the Treatment of Cancer. Anticancer Res. 2017, 37, 5935–5939. [Google Scholar]
- Spallarossa, P.; Tini, G.; Sarocchi, M.; Arboscello, E.; Grossi, F.; Queirolo, P.; Zoppoli, G.; Ameri, P. Identification and Management of Immune Checkpoint Inhibitor-Related Myocarditis: Use Troponin Wisely. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2019, 37, 2201–2205. [Google Scholar] [CrossRef]
- Wang, D.Y.; Salem, J.E.; Cohen, J.V.; Chandra, S.; Menzer, C.; Ye, F.; Zhao, S.; Das, S.; Beckermann, K.E.; Ha, L.; et al. Fatal Toxic Effects Associated With Immune Checkpoint Inhibitors: A Systematic Review and Meta-analysis. JAMA Oncol. 2018, 4, 1721–1728. [Google Scholar] [CrossRef]
- Imazio, M.; Gaita, F.; LeWinter, M. Evaluation and Treatment of Pericarditis: A Systematic Review. JAMA 2015, 314, 1498–1506. [Google Scholar] [CrossRef]
- Varricchi, G.; Marone, G.; Mercurio, V.; Galdiero, M.R.; Bonaduce, D.; Tocchetti, C.G. Immune Checkpoint Inhibitors and Cardiac Toxicity: An Emerging Issue. Curr. Med. Chem. 2018, 25, 1327–1339. [Google Scholar] [CrossRef]
- Castrillon, J.A.; Eng, C.; Cheng, F. Pharmacogenomics for Immunotherapy and Immune-related Cardiotoxicity. Hum. Mol. Genet. 2020, 29, R186–R196. [Google Scholar] [CrossRef]
- Minasian, L.; Dimond, E.; Davis, M.; Adhikari, B.; Fagerstrom, R.; Fabian, C.; Floyd, J.; Unger, J.M.; Douglas, P.S.; Mustian, K.M.; et al. The Evolving Design of NIH-Funded Cardio-Oncology Studies to Address Cancer Treatment-Related Cardiovascular Toxicity. JACC Cardio Oncol. 2019, 1, 105–113. [Google Scholar] [CrossRef]
- Zaidi, N.; Jaffee, E.M. Immunotherapy transforms cancer treatment. J. Clin. Investig. 2019, 129, 46–47. [Google Scholar] [CrossRef]
- Saiki, H.; Petersen, I.A.; Scott, C.G.; Bailey, K.R.; Dunlay, S.M.; Finley, R.R.; Ruddy, K.J.; Yan, E.; Redfield, M.M. Risk of Heart Failure with Preserved Ejection Fraction in Older Women After Contemporary Radiotherapy for Breast Cancer. Circulation 2017, 135, 1388–1396. [Google Scholar] [CrossRef]
- Upadhrasta, S.; Elias, H.; Patel, K.; Zheng, L. Managing cardiotoxicity associated with immune checkpoint inhibitors. Chronic Dis. Transl. Med. 2019, 5, 6–14. [Google Scholar] [CrossRef]
- Ryan, T.D.; Nagarajan, R.; Godown, J. Pediatric Cardio-Oncology: Development of Cancer Treatment-Related Cardiotoxicity and the Therapeutic Approach to Affected Patients. Curr. Treat. Options Oncol. 2019, 20, 56. [Google Scholar] [CrossRef]
- Jain, V.; Mohebtash, M.; Rodrigo, M.E.; Ruiz, G.; Atkins, M.B.; Barac, A. Autoimmune Myocarditis Caused by Immune Checkpoint Inhibitors Treated With Antithymocyte Globulin. J. Immunother. 2018, 41, 332–335. [Google Scholar] [CrossRef]
- Tajmir-Riahi, A.; Bergmann, T.; Schmid, M.; Agaimy, A.; Schuler, G.; Heinzerling, L. Life-threatening Autoimmune Cardiomyopathy Reproducibly Induced in a Patient by Checkpoint Inhibitor Therapy. J. Immunother. 2018, 41, 35–38. [Google Scholar] [CrossRef]
- Xia, W.; Chen, H.; Chen, D.; Ye, Y.; Xie, C.; Hou, M. PD-1 inhibitor inducing exosomal miR-34a-5p expression mediates the cross talk between cardiomyocyte and macrophage in immune checkpoint inhibitor-related cardiac dysfunction. J. Immunother. Cancer 2020, 8, e001293. [Google Scholar] [CrossRef]
- Agrawal, N.; Khunger, A.; Vachhani, P.; Colvin, T.A.; Hattoum, A.; Spangenthal, E.; Curtis, A.B.; Dy, G.K.; Ernstoff, M.S.; Puzanov, I. Cardiac Toxicity Associated with Immune Checkpoint Inhibitors: Case Series and Review of the Literature. Case Rep. Oncol. 2019, 12, 260–276. [Google Scholar] [CrossRef]
- Wang, H.; Kwak, D.; Fassett, J.; Hou, L.; Xu, X.; Burbach, B.J.; Thenappan, T.; Xu, Y.; Ge, J.B.; Shimizu, Y.; et al. CD28/B7 Deficiency Attenuates Systolic Overload-Induced Congestive Heart Failure, Myocardial and Pulmonary Inflammation, and Activated T Cell Accumulation in the Heart and Lungs. Hypertension 2016, 68, 688–696. [Google Scholar] [CrossRef]
- Johnson, D.B.; Balko, J.M.; Compton, M.L.; Chalkias, S.; Gorham, J.; Xu, Y.; Hicks, M.; Puzanov, I.; Alexander, M.R.; Bloomer, T.L.; et al. Fulminant Myocarditis with Combination Immune Checkpoint Blockade. N. Engl. J. Med. 2016, 375, 1749–1755. [Google Scholar] [CrossRef]
- Saade, A.; Mansuet-Lupo, A.; Arrondeau, J.; Thibault, C.; Mirabel, M.; Goldwasser, F.; Oudard, S.; Weiss, L. Pericardial effusion under nivolumab: Case-reports and review of the literature. J. Immunother. Cancer 2019, 7, 266. [Google Scholar] [CrossRef]
- Chen, D.Y.; Huang, W.K.; Chien-Chia Wu, V.; Chang, W.C.; Chen, J.S.; Chuang, C.K.; Chu, P.H. Cardiovascular toxicity of immune checkpoint inhibitors in cancer patients: A review when cardiology meets immuno-oncology. J. Formos Med. Assoc. 2020, 119, 1461–1475. [Google Scholar] [CrossRef]
- Zhou, Y.W.; Zhu, Y.J.; Wang, M.N.; Xie, Y.; Chen, C.Y.; Zhang, T.; Xia, F.; Ding, Z.Y.; Liu, J.Y. Immune Checkpoint Inhibitor-Associated Cardiotoxicity: Current Understanding on Its Mechanism, Diagnosis and Management. Front. Pharmacol. 2019, 10, 1350. [Google Scholar] [CrossRef]
- Tocchetti, C.G.; Cadeddu, C.; Di Lisi, D.; Femmino, S.; Madonna, R.; Mele, D.; Monte, I.; Novo, G.; Penna, C.; Pepe, A.; et al. From Molecular Mechanisms to Clinical Management of Antineoplastic Drug-Induced Cardiovascular Toxicity: A Translational Overview. Antioxid. Redox Signal. 2019, 30, 2110–2153. [Google Scholar] [CrossRef]
- Lyon, A.R.; Yousaf, N.; Battisti, N.M.L.; Moslehi, J.; Larkin, J. Immune checkpoint inhibitors and cardiovascular toxicity. Lancet Oncol. 2018, 19, e447–e458. [Google Scholar] [CrossRef]
- Khouri, M.G.; Klem, I.; Shenoy, C.; Sulpher, J.; Dent, S.F. Screening and Monitoring for Cardiotoxicity During Cancer Treatment. In Cardio-Oncology: Clinical Overlap of Cancer and Heart Disease; Kimmick, G.G., Lenihan, D.J., Sawyer, D.B., Mayer, E.L., Hershman, D.L., Eds.; Springer: Cham, Switzerland, 2017; pp. 43–80. [Google Scholar]
- Cheng, R.K.; Kirkpatrick, J.N.; Sorror, M.L.; Barac, A. Cardio-Oncology and the Intersection of Cancer and Cardiotoxicity. JACC Cardio Oncol. 2019, 1, 314–317. [Google Scholar] [CrossRef]
- Brahmer, J.R.; Lacchetti, C.; Schneider, B.J.; Atkins, M.B.; Brassil, K.J.; Caterino, J.M.; Chau, I.; Ernstoff, M.S.; Gardner, J.M.; Ginex, P.; et al. Management of Immune-Related Adverse Events in Patients Treated With Immune Checkpoint Inhibitor Therapy: American Society of Clinical Oncology Clinical Practice Guideline. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2018, 36, 1714–1768. [Google Scholar] [CrossRef]
- Zamorano, J.L.; Lancellotti, P.; Rodriguez Muñoz, D.; Aboyans, V.; Asteggiano, R.; Galderisi, M.; Habib, G.; Lenihan, D.J.; Lip, G.Y.H.; Lyon, A.R.; et al. 2016 ESC Position Paper on cancer treatments and cardiovascular toxicity developed under the auspices of the ESC Committee for Practice Guidelines: The Task Force for cancer treatments and cardiovascular toxicity of the European Society of Cardiology (ESC). Eur. Heart J. 2016, 37, 2768–2801. [Google Scholar] [CrossRef]
- Curigliano, G.; Cardinale, D.; Suter, T.; Plataniotis, G.; de Azambuja, E.; Sandri, M.T.; Criscitiello, C.; Goldhirsch, A.; Cipolla, C.; Roila, F. Cardiovascular toxicity induced by chemotherapy, targeted agents and radiotherapy: ESMO Clinical Practice Guidelines. Ann. Oncol. 2012, 23, vii155–vii166. [Google Scholar] [CrossRef]
- Gilstrap, L.; Harrison, M.; Kimmick, G.G.; Nohria, A. Management of Chemotherapy-Associated Cardiomyopathy. In Cardio-Oncology: The Clinical Overlap of Cancer and Heart Disease; Kimmick, G.G., Lenihan, D.J., Sawyer, D.B., Mayer, E.L., Hershman, D.L., Eds.; Springer: Cham, Switzerland, 2017; pp. 81–104. [Google Scholar]
- Curigliano, G.; Lenihan, D.; Fradley, M.; Ganatra, S.; Barac, A.; Blaes, A.; Herrmann, J.; Porter, C.; Lyon, A.R.; Lancellotti, P.; et al. Management of cardiac disease in cancer patients throughout oncological treatment: ESMO consensus recommendations. Ann. Oncol. 2020, 31, 171–190. [Google Scholar] [CrossRef]
- Teske, A.J.; Linschoten, M.; Kamphuis, J.A.M.; Naaktgeboren, W.R.; Leiner, T.; van der Wall, E.; Kuball, J.; van Rhenen, A.; Doevendans, P.A.; Cramer, M.J.; et al. Cardio-oncology: An overview on outpatient management and future developments. Neth. Heart J. 2018, 26, 521–532. [Google Scholar] [CrossRef]
- Sachinidis, A. Cardiotoxicity and Heart Failure: Lessons from Human-Induced Pluripotent Stem Cell-Derived Cardiomyocytes and Anticancer Drugs. Cells 2020, 9, 1001. [Google Scholar] [CrossRef]
- Sayed, N.; Ameen, M.; Wu, J.C. Personalized medicine in cardio-oncology: The role of induced pluripotent stem cell. Cardiovasc. Res. 2019, 115, 949–959. [Google Scholar] [CrossRef]
- Gintant, G.; Burridge, P.; Gepstein, L.; Harding, S.; Herron, T.; Hong, C.; Jalife, J.; Wu, J.C. Use of Human Induced Pluripotent Stem Cell-Derived Cardiomyocytes in Preclinical Cancer Drug Cardiotoxicity Testing: A Scientific Statement From the American Heart Association. Circ. Res. 2019, 125, e75–e92. [Google Scholar] [CrossRef]
- Sharma, A.; McKeithan, W.L.; Serrano, R.; Kitani, T.; Burridge, P.W.; Del Alamo, J.C.; Mercola, M.; Wu, J.C. Use of human induced pluripotent stem cell-derived cardiomyocytes to assess drug cardiotoxicity. Nat. Protoc. 2018, 13, 3018–3041. [Google Scholar] [CrossRef]
- Pinheiro, E.A.; Fetterman, K.A.; Burridge, P.W. hiPSCs in cardio-oncology: Deciphering the genomics. Cardiovasc. Res. 2019, 115, 935–948. [Google Scholar] [CrossRef]
- Niepel, M.; Hafner, M.; Duan, Q.; Wang, Z.; Paull, E.O.; Chung, M.; Lu, X.; Stuart, J.M.; Golub, T.R.; Subramanian, A.; et al. Common and cell-type specific responses to anti-cancer drugs revealed by high throughput transcript profiling. Nat. Commun. 2017, 8, 1186. [Google Scholar] [CrossRef]
- Kitani, T.; Ong, S.-G.; Lam, C.K.; Rhee, J.-W.; Zhang, J.Z.; Oikonomopoulos, A.; Ma, N.; Tian, L.; Lee, J.; Telli, M.L.; et al. Human-Induced Pluripotent Stem Cell Model of Trastuzumab-Induced Cardiac Dysfunction in Patients With Breast Cancer. Circulation 2019, 139, 2451–2465. [Google Scholar] [CrossRef] [PubMed]
- Necela, B.M.; Axenfeld, B.C.; Serie, D.J.; Kachergus, J.M.; Perez, E.A.; Thompson, E.A.; Norton, N. The antineoplastic drug, trastuzumab, dysregulates metabolism in iPSC-derived cardiomyocytes. Clin. Transl. Med. 2017, 6, 5. [Google Scholar] [CrossRef] [PubMed]
| Cancer Type | mAb (ICI) | Cardiac Toxicity | Reference |
|---|---|---|---|
| Melanoma | Ipilimumab and Nivolumab | Myositis with rhabdomyolysis, early progressive cardiac electrical instability and myocarditis | [37] |
| Non-small cell lung cancer | Nivolumab | Massive pericardial effusion | [38] |
| Hodgkin’s lymphoma | Pembrolizumab (KEYTRUDA) | Myocardial infarction, pericardial effusion, pericarditis, arrhythmia and cardiac tamponade | [39] |
| Kidney cancer | Nivolumab | Myocarditis | [40] |
| Advanced urothelial carcinoma | Pembrolizumab and Atezolizumab | Immune myocarditis | [41] |
| Merkel cell carcinoma | Nivolumab | Myocarditis | [42] |
| Squamous cell neck cancer | Nivolumab | Myocarditis and ventricular arrhythmia | [42] |
| Squamous cell neck cancer | Pembrolizumab | Cardiac failure | [42] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumar, M.; Thangavel, C.; Becker, R.C.; Sadayappan, S. Monoclonal Antibody-Based Immunotherapy and Its Role in the Development of Cardiac Toxicity. Cancers 2021, 13, 86. https://doi.org/10.3390/cancers13010086
Kumar M, Thangavel C, Becker RC, Sadayappan S. Monoclonal Antibody-Based Immunotherapy and Its Role in the Development of Cardiac Toxicity. Cancers. 2021; 13(1):86. https://doi.org/10.3390/cancers13010086
Chicago/Turabian StyleKumar, Mohit, Chellappagounder Thangavel, Richard C. Becker, and Sakthivel Sadayappan. 2021. "Monoclonal Antibody-Based Immunotherapy and Its Role in the Development of Cardiac Toxicity" Cancers 13, no. 1: 86. https://doi.org/10.3390/cancers13010086
APA StyleKumar, M., Thangavel, C., Becker, R. C., & Sadayappan, S. (2021). Monoclonal Antibody-Based Immunotherapy and Its Role in the Development of Cardiac Toxicity. Cancers, 13(1), 86. https://doi.org/10.3390/cancers13010086






