Retrospective Analysis of Patients with Signet Ring Subtype of Colorectal Cancer with Peritoneal Metastasis Treated with CRS & HIPEC
Simple Summary
Abstract
1. Introduction
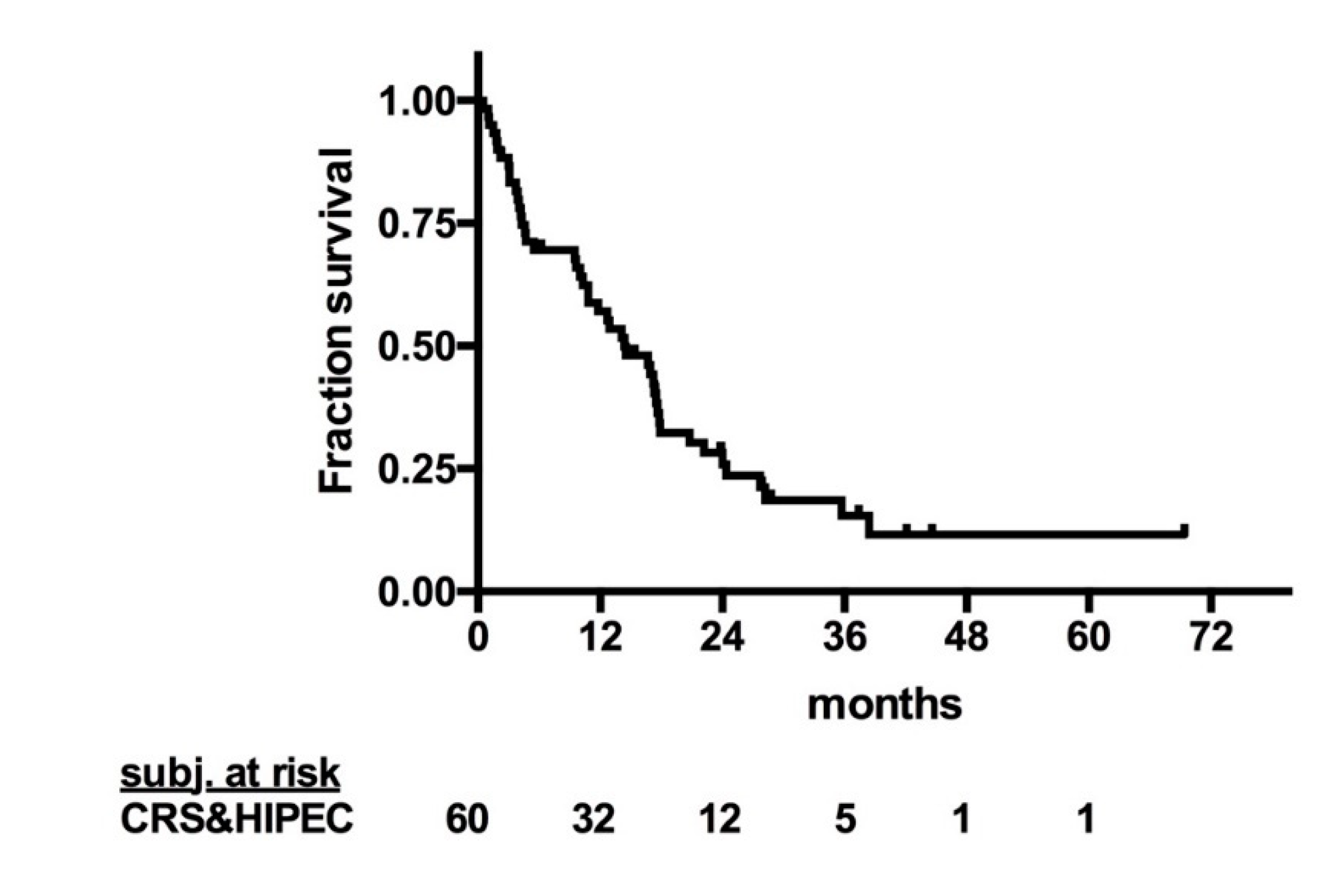
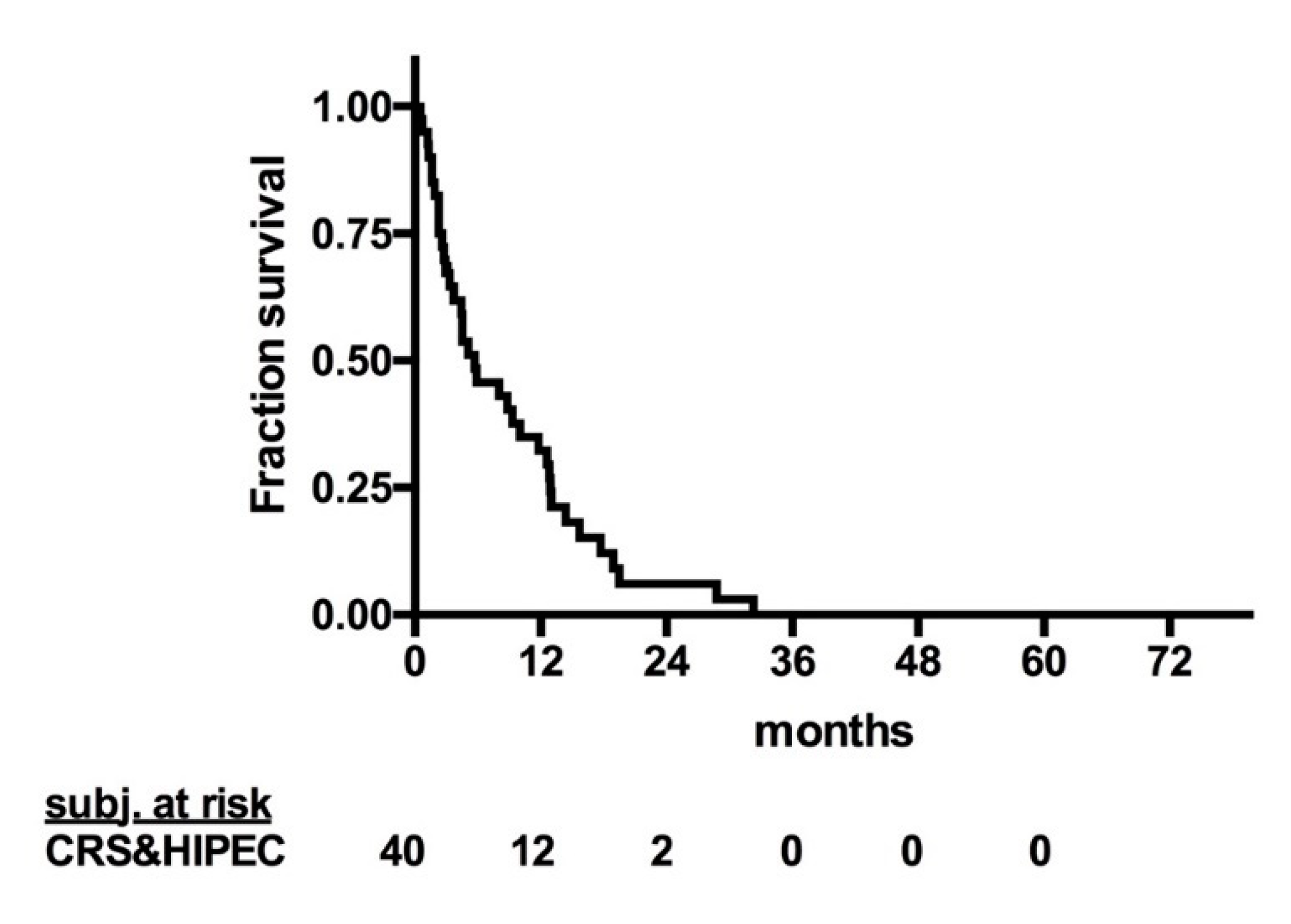
2. Results
2.1. Analysis of Predictive Factors
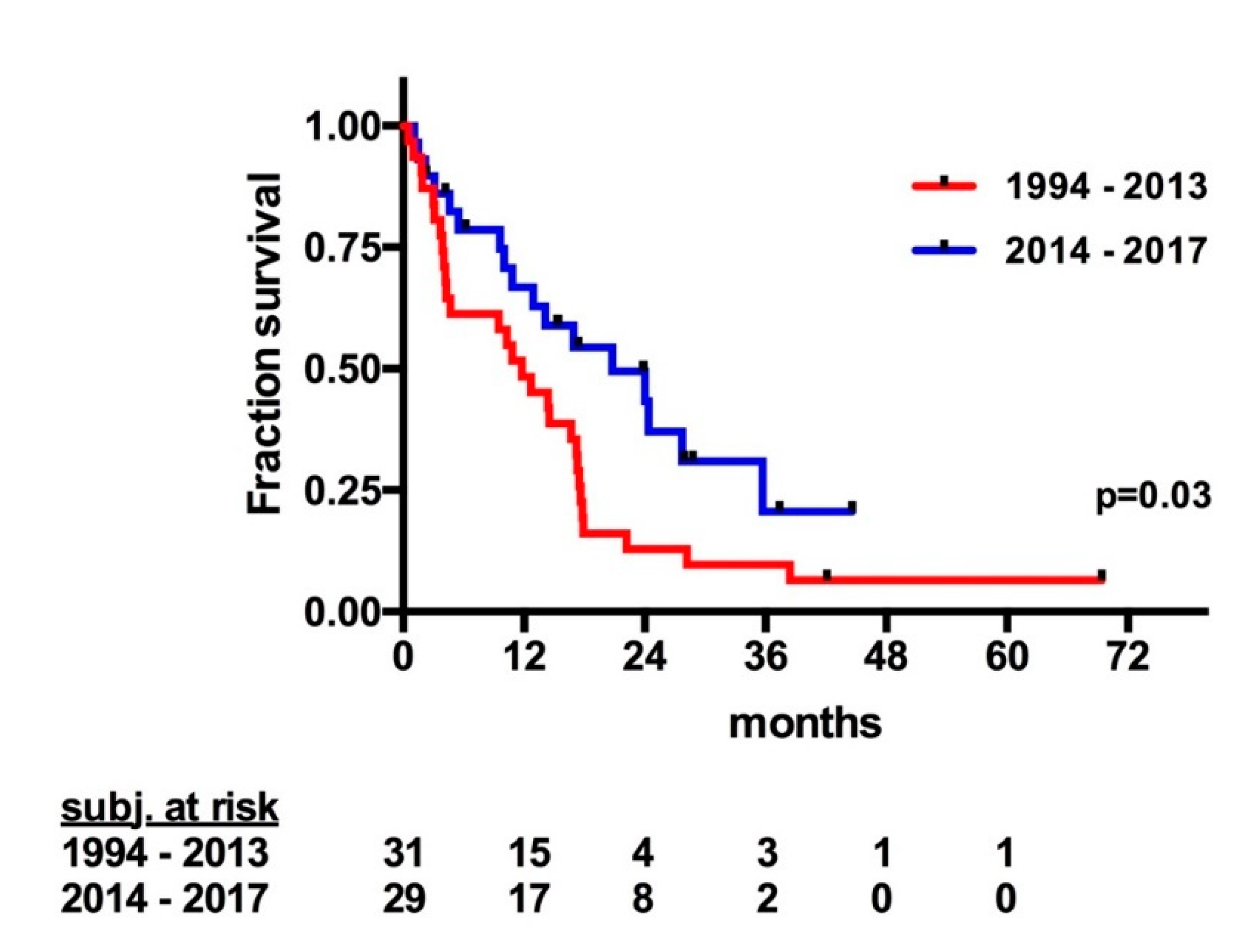
2.2. Tumor Recurrence
3. Discussion
4. Methods
4.1. Treatment and Follow-Up Protocol
4.2. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Data Availability Statement
References
- Lemmens, V.E.; Klaver, Y.L.; Verwaal, V.J.; Rutten, H.J.; Coebergh, J.W.; de Hingh, I.H. Predictors and survival of synchronous peritoneal carcinomatosis of colorectal origin: A population-based study. Int. J. Cancer 2011, 128, 2717–2725. [Google Scholar] [CrossRef] [PubMed]
- Segelman, J.; Granath, F.; Holm, T.; Machado, M.; Mahteme, H.; Martling, A. Incidence, prevalence and risk factors for peritoneal carcinomatosis from colorectal cancer. Br. J. Surg. 2012, 99, 699–705. [Google Scholar] [CrossRef] [PubMed]
- Jahne, J. Cytoreduction including total gastrectomy for pseudomyxoma peritonei. Br. J. Surg. 2002, 89, 938–939. [Google Scholar]
- Sadeghi, B.; Arvieux, C.; Glehen, O.; Beaujard, A.C.; Rivoire, M.; Baulieux, J.; Fontaumard, E.; Brachet, A.; Caillot, J.L.; Faure, J.L.; et al. Peritoneal carcinomatosis from non-gynecologic malignancies: Results of the EVOCAPE 1 multicentric prospective study. Cancer 2000, 88, 358–363. [Google Scholar] [CrossRef]
- Franko, J.; Shi, Q.; Goldman, C.D.; Pockaj, B.A.; Nelson, G.D.; Goldberg, R.M.; Pitot, H.C.; Grothey, A.; Alberts, S.R.; Sargent, D.J. Treatment of colorectal peritoneal carcinomatosis with systemic chemotherapy: A pooled analysis of north central cancer treatment group phase III trials N9741 and N9841. J. Clin. Oncol. 2012, 30, 263–267. [Google Scholar] [CrossRef] [PubMed]
- Klaver, Y.L.B.; Simkens, L.H.J.; Lemmens, V.E.P.P.; Koopman, M.; Teerenstra, S.; Bleichrodt, R.P.; De Hingh, I.H.J.T.; Punt, C.J.A. Outcomes of colorectal cancer patients with peritoneal carcinomatosis treated with chemotherapy with and without targeted therapy. Eur. J. Surg. Oncol. 2012, 38, 617–623. [Google Scholar] [CrossRef]
- Koopman, M.; Antonini, N.F.; Douma, J.; Wals, J.; Honkoop, A.H.; Erdkamp, F.L.; de Jong, R.S.; Rodenburg, C.J.; Vreugdenhil, G.; Loosveld, O.J.; et al. Sequential versus combination chemotherapy with capecitabine, irinotecan, and oxaliplatin in advanced colorectal cancer (CAIRO): A phase III randomised controlled trial. Lancet 2007, 370, 135–142. [Google Scholar] [CrossRef]
- Tol, J.; Koopman, M.; Rodenburg, C.J.; Cats, A.; Creemers, G.J.; Schrama, J.G.; Erdkamp, F.L.G.; Vos, A.H.; Mol, L.; Antonini, N.F.; et al. A randomised phase III study on capecitabine, oxaliplatin and bevacizumab with or without cetuximab in first-line advanced colorectal cancer, the CAIRO2 study of the Dutch Colorectal Cancer Group (DCCG). An interim analysis of toxicity. Ann. Oncol. 2008, 19, 734–738. [Google Scholar] [CrossRef]
- Elias, D.; Lefevre, J.H.; Chevalier, J.; Brouquet, A.; Marchal, F.; Classe, J.M.; Ferron, G.; Guilloit, J.M.; Meeus, P.; Goéré, D.; et al. Complete cytoreductive surgery plus intraperitoneal chemohyperthermia with oxaliplatin for peritoneal carcinomatosis of colorectal origin. J. Clin. Oncol. 2009, 27, 681–685. [Google Scholar] [CrossRef]
- Verwaal, V.J.; van Ruth, S.; de Bree, E.; van Slooten, G.W.; van Tinteren, H.; Boot, H.; Zoetmulder, F.A. Randomized trial of cytoreduction and hyperthermic intraperitoneal chemotherapy versus systemic chemotherapy and palliative surgery in patients with peritoneal carcinomatosis of colorectal cancer. J. Clin. Oncol. 2003, 21, 3737–3743. [Google Scholar] [CrossRef]
- Franko, J.; Ibrahim, Z.; Gusani, N.J.; Holtzman, M.P.; Bartlett, D.L.; Zeh, H.J., 3rd. Cytoreductive surgery and hyperthermic intraperitoneal chemoperfusion versus systemic chemotherapy alone for colorectal peritoneal carcinomatosis. Cancer 2010, 116, 3756–3762. [Google Scholar] [CrossRef] [PubMed]
- Quenet, F.; Goéré, D.; Mehta, S.S.; Roca, L.; Dumont, F.; Hessissen, M.; Saint-Aubert, B.; Elias, D. Results of two bi-institutional prospective studies using intraperitoneal oxaliplatin with or without irinotecan during HIPEC after cytoreductive surgery for colorectal carcinomatosis. Ann. Surg. 2011, 254, 294–301. [Google Scholar] [CrossRef] [PubMed]
- Froysnes, I.S.; Larsen, S.G.; Spasojevic, M.; Dueland, S.; Flatmark, K. Complete cytoreductive surgery and hyperthermic intraperitoneal chemotherapy for colorectal peritoneal metastasis in Norway: Prognostic factors and oncologic outcome in a national patient cohort. J. Surg. Oncol. 2016, 114, 222–227. [Google Scholar] [CrossRef] [PubMed]
- Baratti, D.; Kusamura, S.; Pietrantonio, F.; Guaglio, M.; Niger, M.; Deraco, M. Progress in treatments for colorectal cancer peritoneal metastases during the years 2010–2015. A systematic review. Crit. Rev. Oncol. Hematol. 2016, 100, 209–222. [Google Scholar] [CrossRef]
- Quenet, F.; Elias, D.; Roca, L.; Goere, D.; Ghouti, L.; Pocard, M.; Facy, O.; Arvieux, C.; Lorimier, G.; Pezet, D. A UNICANCER phase III trial of hyperthermic intra-peritoneal chemotherapy (HIPEC) for colorectal peritoneal carcinomatosis (PC): PRODIGE 7. J. Clin. Oncol. 2018, 36 (Suppl. S18), 3503. [Google Scholar] [CrossRef]
- Kwakman, R.; Schrama, A.M.; van Olmen, J.P.; Otten, R.H.; de Lange-de Klerk, E.S.; Erienne, M.; Kazemier, G.; te Velde, E.A. Clinicopathological Parameters in Patient Selection for Cytoreductive Surgery and Hyperthermic Intraperitoneal Chemotherapy for Colorectal Cancer Metastases: A Meta-analysis. Ann. Surg. 2016, 263, 1102–1111. [Google Scholar] [CrossRef]
- Kang, H.; O’Connell, J.B.; Maggard, M.A.; Sack, J.; Ko, C.Y. A 10-year outcomes evaluation of mucinous and signet-ring cell carcinoma of the colon and rectum. Dis. Colon Rectum 2005, 48, 1161–1168. [Google Scholar] [CrossRef]
- Secco, G.B.; Fardelli, R.; Campora, E.; Lapertosa, G.; Gentile, R.; Zoli, S.; Prior, C. Primary mucinous adenocarcinomas and signet-ring cell carcinomas of colon and rectum. Oncology 1994, 51, 30–34. [Google Scholar] [CrossRef]
- Makino, T.; Tsujinaka, T.; Mishima, H.; Ikenaga, M.; Sawamura, T.; Nakamori, S.; Fujitani, K.; Hirao, M.; Kashiwazaki, M.; Masuda, N.; et al. Primary signet-ring cell carcinoma of the colon and rectum: Report of eight cases and review of 154 Japanese cases. Hepatogastroenterology 2006, 53, 845–849. [Google Scholar]
- Belli, S.; Aytac, H.O.; Karagulle, E.; Yabanoglu, H.; Kayaselcuk, F.; Yildirim, S. Outcomes of surgical treatment of primary signet ring cell carcinoma of the colon and rectum: 22 cases reviewed with literature. Int. Surg. 2014, 99, 691–698. [Google Scholar] [CrossRef]
- Khoury, J.; Chacko, R.; Macari, D.; Gbadamosi, B.; Ezekwudo, D.; Blankenship, L.; Anderson, J.M.; Jaiyesimi, I.A. Metastatic patterns and prognostic significance of signet ring cell carcinoma of the colon: Retrospective analysis of SEER database. J. Clin. Oncol. 2018, 36, e15594. [Google Scholar] [CrossRef]
- Nitsche, U.; Zimmermann, A.; Späth, C.; Müller, T.; Maak, M.; Schuster, T.; Slotta-Huspenina, J.; Käser, S.A.; Michalski, C.W.; Janssen, K.P.; et al. Mucinous and signet-ring cell colorectal cancers differ from classical adenocarcinomas in tumor biology and prognosis. Ann. Surg. 2013, 258, 775–782. [Google Scholar] [CrossRef]
- Verwaal, V.J.; van Tinteren, H.; van Ruth, S.; Zoetmulder, F.A. Predicting the survival of patients with peritoneal carcinomatosis of colorectal origin treated by aggressive cytoreduction and hyperthermic intraperitoneal chemotherapy. Br. J. Surg. 2004, 91, 739–746. [Google Scholar] [CrossRef]
- Chua, T.C.; Pelz, J.O.; Kerscher, A.; Morris, D.L.; Esquivel, J. Critical analysis of 33 patients with peritoneal carcinomatosis secondary to colorectal and appendiceal signet ring cell carcinoma. Ann. Surg. Oncol. 2009, 16, 2765–2770. [Google Scholar] [CrossRef]
- Van Sweringen, H.L.; Hanseman, D.J.; Ahmad, S.A.; Edwards, M.J.; Sussman, J.J. Predictors of survival in patients with high-grade peritoneal metastases undergoing cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. Surgery 2012, 152, 617–624. [Google Scholar] [CrossRef]
- Winer, J.; Zenati, M.; Ramalingam, L.; Jones, H.; Zureikat, A.; Holtzman, M.; Lee, K.; Ahrendt, S.; Pingpank, J.; Zeh, H.J.; et al. Impact of aggressive histology and location of primary tumor on the efficacy of surgical therapy for peritoneal carcinomatosis of colorectal origin. Ann. Surg. Oncol. 2014, 21, 1456–1462. [Google Scholar] [CrossRef]
- Van Oudheusden, T.R.; Braam, H.J.; Nienhuijs, S.W.; Wiezer, M.J.; Van Ramshorst, B.; Luyer, P.; de Hingh, I.H. Poor outcome after cytoreductive surgery and HIPEC for colorectal peritoneal carcinomatosis with signet ring cell histology. J. Surg. Oncol. 2015, 111, 237–242. [Google Scholar] [CrossRef]
- Elias, D.; Mariani, A.; Cloutier, A.S.; Blot, F.; Goéré, D.; Dumont, F.; Honoré, C.; Billard, V.; Dartigues, P.; Ducreux, M. Modified selection criteria for complete cytoreductive surgery plus HIPEC based on peritoneal cancer index and small bowel involvement for peritoneal carcinomatosis of colorectal origin. Eur. J. Surg. Oncol. 2014, 40, 1467–1473. [Google Scholar] [CrossRef]
- Yonemura, Y.; Bandou, E.; Sawa, T.; Yoshimitsu, Y.; Endou, Y.; Sasaki, T.; Sugarbaker, P.H. Neoadjuvant treatment of gastric cancer with peritoneal dissemination. Eur. J. Surg. Oncol. 2006, 32, 661–665. [Google Scholar] [CrossRef]
- Jacquet, P.; Sugarbaker, P.H. Clinical research methodologies in diagnosis and staging of patients with peritoneal carcinomatosis. Cancer Treat Res. 1996, 82, 359–374. [Google Scholar]
- Sugarbaker, P.H. Successful management of microscopic residual disease in large bowel cancer. Cancer Chemother. Pharmacol. 1999, 43, S15–S25. [Google Scholar] [CrossRef] [PubMed]
- Masuda, T.; Kuramoto, M.; Shimada, S.; Ikeshima, S.; Yamamoto, K.; Nakamura, K.; Yoshimatsu, S.; Urata, M.; Baba, H. The effect of extensive intraoperative peritoneal lavage therapy (EIPL) on stage III B + C and cytology-positive gastric cancer patients. Int. J. Clin. Oncol. 2016, 21, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Dindo, D.; Demartines, N.; Clavien, P.A. Classification of surgical complications: A new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann. Surg. 2004, 240, 205–213. [Google Scholar] [CrossRef] [PubMed]
| Characteristics | Value | |
|---|---|---|
| Sex (%) | Male | 56.7 (34/60) |
| Female | 43.3 (26/60) | |
| Age (years) | 51.4 (SD: 13.9) | |
| Primary Tumor Location (%) | Cecum | 20 (12/60) |
| Ascending colon | 23.3 (14/60) | |
| Transverse colon | 8.3 (5/60) | |
| Descending colon | 3.3 (2/60) | |
| Sigmoid colon | 30 (18/60) | |
| Recto-sigmoid | 15 (9/60) | |
| Appearance of PM (%) | Synchronous | 60.3 (35/58) |
| Metachronous | 39.7 (23/58) | |
| Not known | 2 | |
| PCI (Mean) | Total PCI | 13.1 (SD: 10.4) |
| Small bowel PCI | 3.6 (SD: 3.8) | |
| Patients Treated with HIPEC (%) | Yes | 75 (45/60) |
| No | 25 (15/60) | |
| HIPEC Drug Used (%) | MMC + CDDP | 60 (27/45) |
| Oxaliplatin + 5FU | 33.3 (15/45) | |
| MMC | 2.2 (1/45) | |
| Taxotere + CDDP | 2.2 (1/45) | |
| Oxaliplatin | 2.2 (1/45) | |
| Mean Surgical Time (min) | 302.6 (SD: 258.8) | |
| Mean Blood Loss (mL) | 1457.6 (SD: 1160.5) | |
| Mean No. of Blood Units Transfused | 4.4 (SD: 5.7) | |
| Completeness of Cytoreduction (%) | CC0 | 61.7 (37/60) |
| CC1 | 5 (3/60) | |
| CC2 | 13.3 (8/60) | |
| CC3 | 20 (12/60) | |
| Clavien–Dindo (%) | Grade 0 | 50 (30/60) |
| Grade I–II | 23.3 (14/60) | |
| Grade III | 13.3 (8/60) | |
| Grade IV | 11.7 (7/60) | |
| Grade V | 1.7 (1/60) | |
| Preoperative Chemotherapy (%) | No Chemotherapy | 1.7 (1/60) |
| NACT | 91.7 (55/60) | |
| NACT without antibody | 58.9 (32/60) | |
| NACT + anti-VEGF antibody | 23.6 (13/55) | |
| NACT + anti EGFR antibody | 16.4 (9/55) | |
| NACT + multiple targeted drugs | 1.8 (1/55) | |
| Biphasic chemotherapy (IV + IP) | 5 (3/60) | |
| Biphasic chemotherapy without antibody | 66.6 (2/3) | |
| Biphasic chemotherapy + anti-VEGF antibody | 0 (0/3) | |
| Biphasic chemotherapy + anti EGFR antibody | 33.3 (1/3) | |
| No details | 1.7 (1/60) | |
| Postoperative Chemotherapy (%) | No chemotherapy | 1.7 (1/60) |
| Systemic chemotherapy | 63.3 (38/60) | |
| Systemic chemotherapy without antibody | 50 (19/38) | |
| Systemic chemotherapy + anti-VEGF antibody | 26.3 (10/38) | |
| Systemic chemotherapy + anti EGFR antibody | 18.4 (7/38) | |
| Systemic chemotherapy + multiple targeted drugs | 5.2 (2/38) | |
| Biphasic chemotherapy (IV + IP) | 1.7 (1/60) | |
| No details | 33.3 (20/60) | |
| Variable | Category | No. | Median Overall Survival (Months) | Univariate Analysis (p Value) | Multivariate Analysis | ||
|---|---|---|---|---|---|---|---|
| HR | 95% CI | p Value | |||||
| Gender | Female Male | 26 34 | 24.4 (10.1–38.7) 11.8 (8.6– 5.1) | 0.004 | 2.14 | 0.94–4.89 | 0.071 |
| Appearance of PM | Synchronous Metachronous | 35 23 | 14.4 (11.9–16.9) 16.7 (7.6–25.7) | 0.960 | |||
| HIPEC | HIPEC No HIPEC | 45 15 | 17.3 (16.2–18.4) 5.5 (0–16.1) | 0.007 | 2.20 | 1.01–4.81 | 0.049 |
| Complications | No Yes | 30 30 | 17.6 (3.5–31.70) 10.8 (0–23) | 0.006 | |||
| CCR | CC0-1 CC2-3 | 40 20 | 17.6 (16.7– 8.5) 4.7 (0–12) | <0.001 | 2.26 | 0.94–5.42 | 0.068 |
| Intraoperative Cytology | Negative Positive | 29 22 | 17.6 (13.1–22.1) 9.6 (1.8–17.3) | 0.017 | 1.78 | 0.86–3.71 | 0.122 |
| Age | ≤60 years >60 years | 40 20 | 12.9 (9.0–16.8) 17.5 (2.6–32.4) | 0.219 | |||
| Nodal status | N0 N+ | 15 45 | 16.9 (12.6–21.2) 12.9 (7.7–18.1) | 0.690 | |||
| Side of Primary | Right Left | 31 29 | 14.1 (9.8–18.4) 16.7 (9.6–23.8) | 0.829 | |||
| PCI | ≤12 >12 | 32 27 | 17.8 (10.1–25.5) 10.0 (8.4–11.5) | 0.004 | 0.27 | 0.07–1.10 | 0.068 |
| Small Bowel PCI | ≤2 >2 | 31 25 | 20.8 (13.1–28.5) 3.5 (5.8–15.5) | <0.001 | 6.51 | 1.65–25.74 | 0.008 |
| Blood loss | ≤1000 mL >1000 mL | 22 35 | 17.2 (7.7–26.7) 14.5 (8.9–20.1) | 0.398 | |||
| BT | No Yes | 20 28 | 17.3 (13.1–1.5) 14.5 (7.1–22.0) | 0.295 | |||
| References | Total Number of Patients | Patients with Signet Ring Cell | Median OS | 5-Year OS | Mean PCI | CC0/CC1 |
|---|---|---|---|---|---|---|
| Verwaal et al. 2004 [23] | 102 | 15 | 13 | NR | NR | 49% * |
| Chua et al. 2009 [24] | 33 | 15 | 13 | NR | NR | 46.7% |
| van Sweringen et al. 2012 [25] | 36 | 5 | 7.2 # | NR | 15 # | 83% * |
| Winer et al. 2014 [26] | 67 | 30 | 12.2 | 7% | 11.5 | 77% |
| van Oudheusden et al. 2014 [27] | 351 | 20 | 14.1 | NR | NR | 87.5% |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Prabhu, A.; Brandl, A.; Wakama, S.; Sako, S.; Ishibashi, H.; Mizumoto, A.; Takao, N.; Noguchi, K.; Motoi, S.; Ichinose, M.; et al. Retrospective Analysis of Patients with Signet Ring Subtype of Colorectal Cancer with Peritoneal Metastasis Treated with CRS & HIPEC. Cancers 2020, 12, 2536. https://doi.org/10.3390/cancers12092536
Prabhu A, Brandl A, Wakama S, Sako S, Ishibashi H, Mizumoto A, Takao N, Noguchi K, Motoi S, Ichinose M, et al. Retrospective Analysis of Patients with Signet Ring Subtype of Colorectal Cancer with Peritoneal Metastasis Treated with CRS & HIPEC. Cancers. 2020; 12(9):2536. https://doi.org/10.3390/cancers12092536
Chicago/Turabian StylePrabhu, Aruna, Andreas Brandl, Satoshi Wakama, Shouzou Sako, Haruaki Ishibashi, Akiyoshi Mizumoto, Nobuyuki Takao, Kousuke Noguchi, Shunsuke Motoi, Masumi Ichinose, and et al. 2020. "Retrospective Analysis of Patients with Signet Ring Subtype of Colorectal Cancer with Peritoneal Metastasis Treated with CRS & HIPEC" Cancers 12, no. 9: 2536. https://doi.org/10.3390/cancers12092536
APA StylePrabhu, A., Brandl, A., Wakama, S., Sako, S., Ishibashi, H., Mizumoto, A., Takao, N., Noguchi, K., Motoi, S., Ichinose, M., Liu, Y., & Yonemura, Y. (2020). Retrospective Analysis of Patients with Signet Ring Subtype of Colorectal Cancer with Peritoneal Metastasis Treated with CRS & HIPEC. Cancers, 12(9), 2536. https://doi.org/10.3390/cancers12092536





