Divergent Patterns and Trends in Breast Cancer Incidence, Mortality and Survival Among Older Women in Germany and the United States
Abstract
1. Introduction
2. Materials and Methods
2.1. Data Sources
2.2. Definition of Variables
2.3. Statistical Analysis
2.4. Ethics Approval and Consent of Participate
3. Results
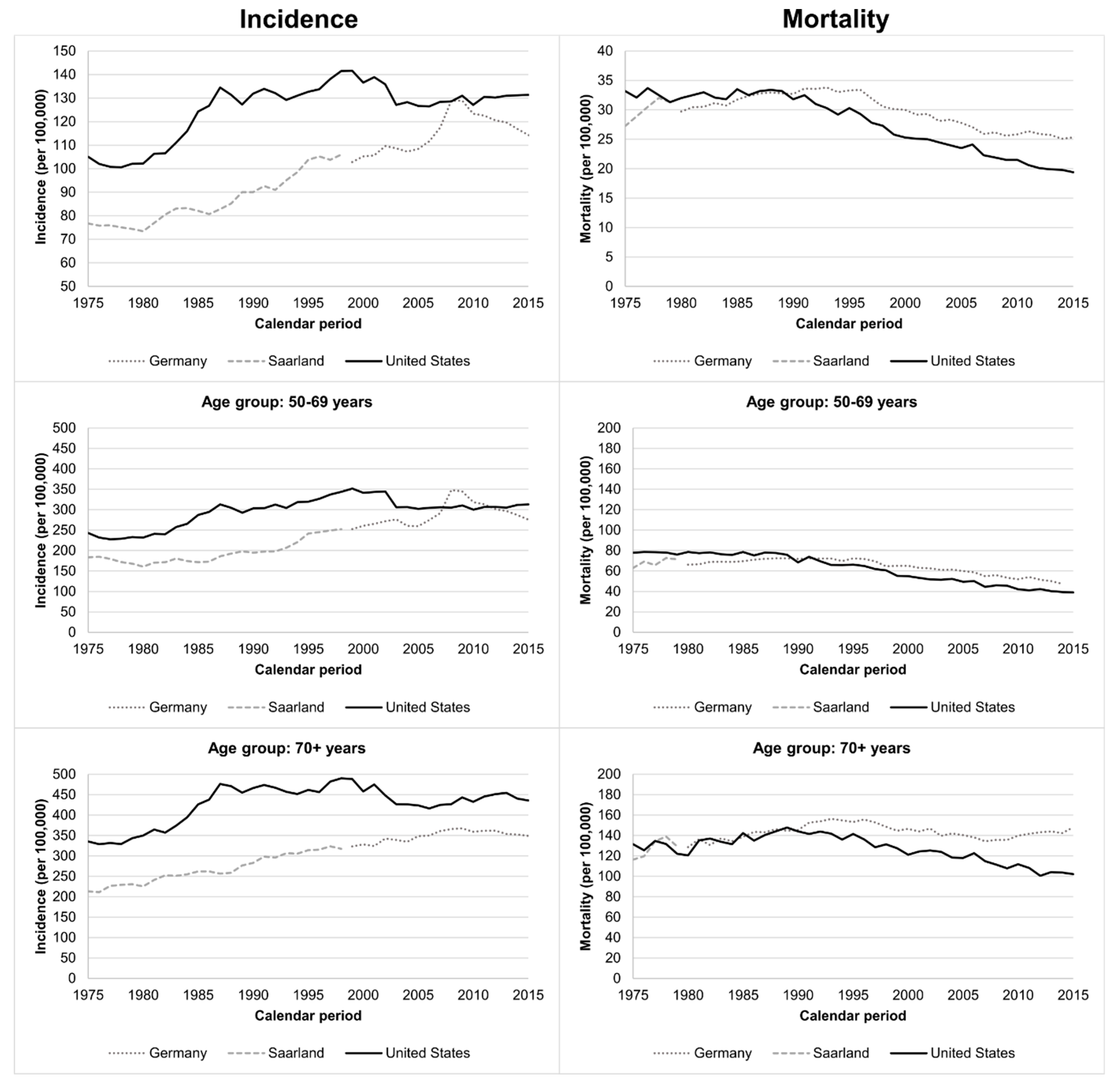
3.1. Trends in Incidence and Mortality
3.2. Patient Characteristics in 2013–2015
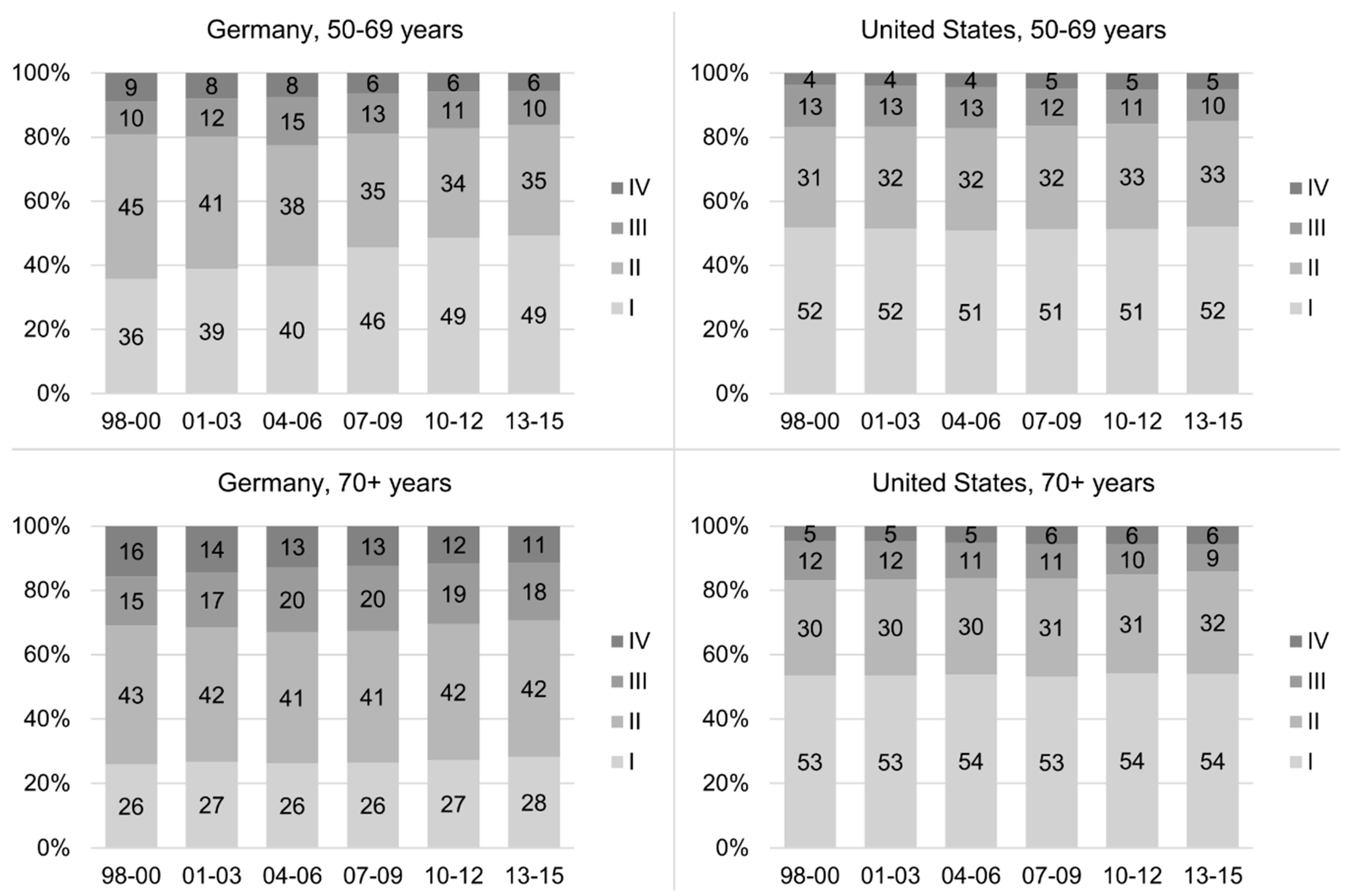
3.3. Trends in Stage Distributions
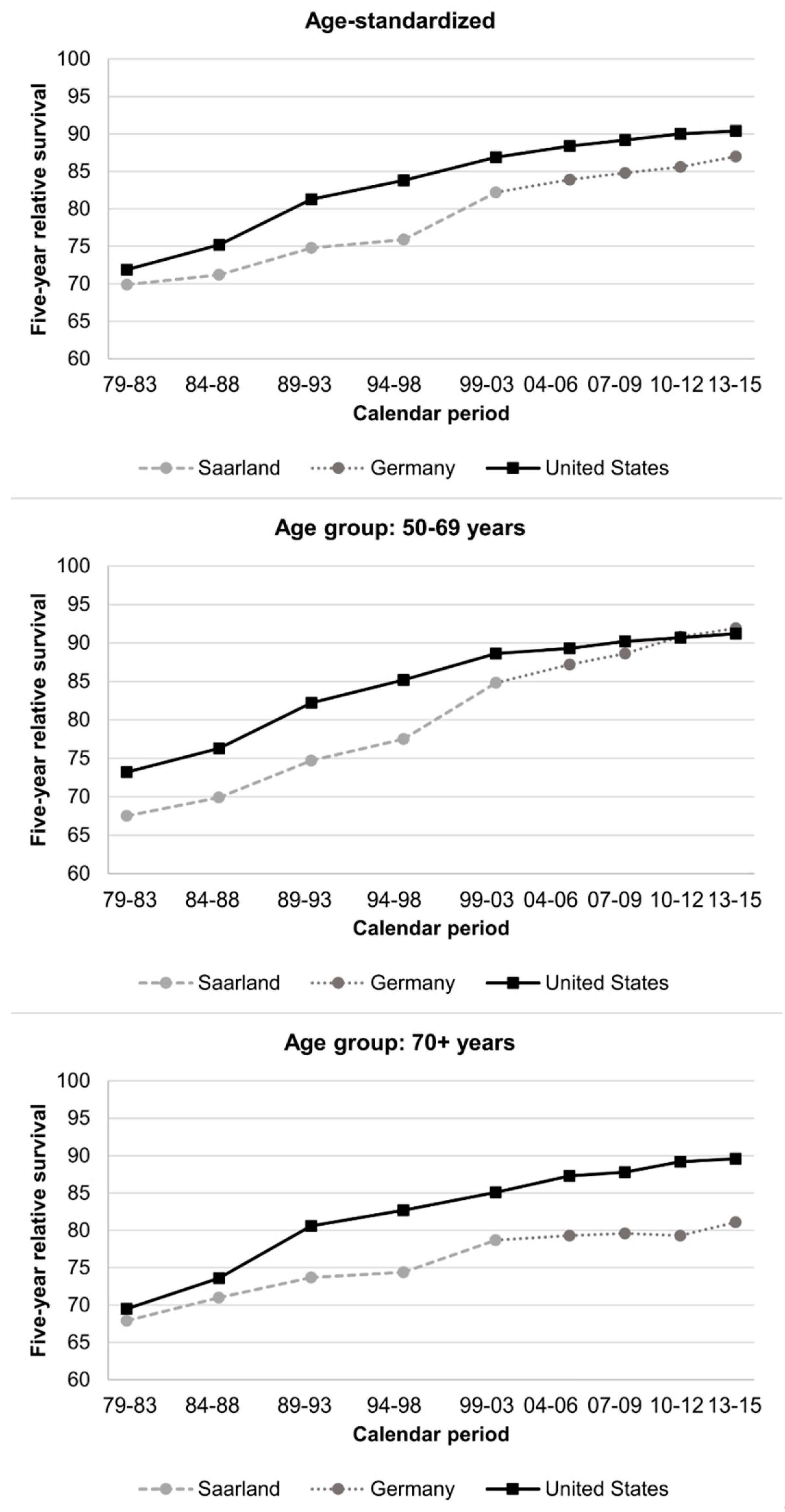
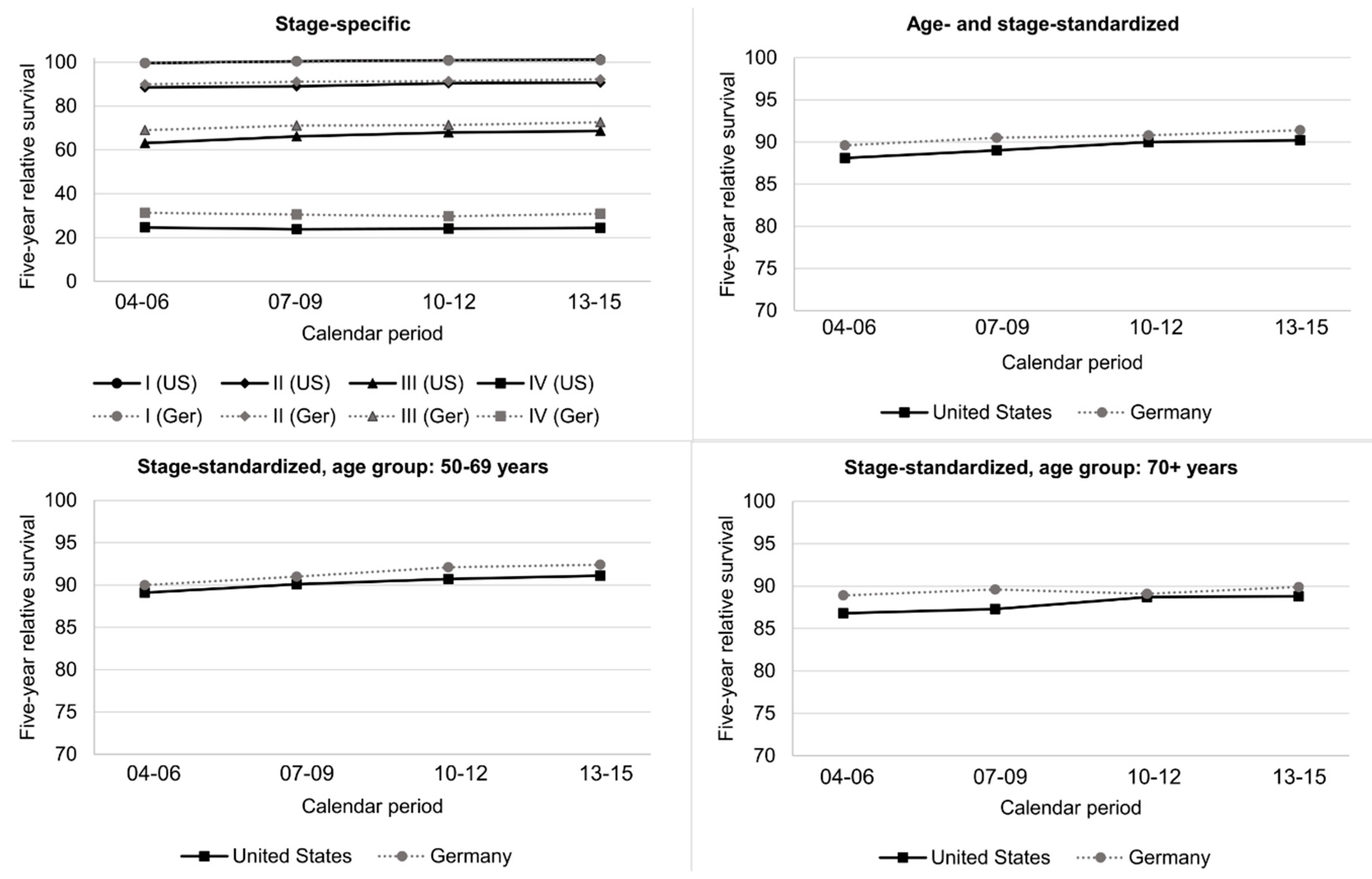
3.4. Trends in Five-Year RS
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References and Notes
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2020. CA Cancer J. Clin. 2020, 70, 7–30. [Google Scholar] [CrossRef] [PubMed]
- Robert Koch Institute. German Centre for Cancer Registry Data, Database Query. Available online: http://www.krebsdaten.de/database (accessed on 7 August 2020).
- Ferlay, J.; Colombet, M.; Bray, F. Cancer Incidence in Five Continents, CI5plus: IARC CancerBase No. 9; International Agency for Research on Cancer: Lyon, France, 2018; Available online: http://ci5.iarc.fr (accessed on 24 October 2019).
- Toriola, A.T.; Colditz, G.A. Trends in breast cancer incidence and mortality in the United States: Implications for prevention. Breast Cancer Res. Treat. 2013, 138, 665–673. [Google Scholar] [CrossRef] [PubMed]
- Li, N.; Deng, Y.; Zhou, L.; Tian, T.; Yang, S.; Wu, Y.; Zheng, Y.; Zhai, Z.; Hao, Q.; Song, D.; et al. Global burden of breast cancer and attributable risk factors in 195 countries and territories, from 1990 to 2017: Results from the Global Burden of Disease Study 2017. J. Hematol. Oncol. 2019, 12, 140. [Google Scholar] [CrossRef]
- Ahmad, A. Breast Cancer Statistics: Recent Trends. Adv. Exp. Med. Biol. 2019, 1152, 1–7. [Google Scholar] [CrossRef]
- International Agency for Research on Cancer. Breast Cancer Screening. In IARC Handbook of Cancer Prevention; IARC: Lyon, France, 2016; Volume 15. [Google Scholar]
- Allemani, C.; Matsuda, T.; Di Carlo, V.; Harewood, R.; Matz, M.; Niksic, M.; Bonaventure, A.; Valkov, M.; Johnson, C.J.; Esteve, J.; et al. Global surveillance of trends in cancer survival 2000–2014 (CONCORD-3): Analysis of individual records for 37 513 025 patients diagnosed with one of 18 cancers from 322 population-based registries in 71 countries. Lancet 2018, 391, 1023–1075. [Google Scholar] [CrossRef]
- Waks, A.G.; Winer, E.P. Breast Cancer Treatment: A Review. JAMA 2019, 321, 288–300. [Google Scholar] [CrossRef]
- Jubair, S.; Alkhateeb, A.; Tabl, A.A.; Rueda, L.; Ngom, A. A novel approach to identify subtype-specific network biomarkers of breast cancer survivability. Netw. Modeling Anal. Health Inform. Bioinform. 2020, 9, 43. [Google Scholar] [CrossRef]
- Tabl, A.A.; Alkhateeb, A.; ElMaraghy, W.; Rueda, L.; Ngom, A. A Machine Learning Approach for Identifying Gene Biomarkers Guiding the Treatment of Breast Cancer. Front. Genet. 2019, 10. [Google Scholar] [CrossRef]
- Sant, M.; Francisci, S.; Capocaccia, R.; Verdecchia, A.; Allemani, C.; Berrino, F. Time trends of breast cancer survival in Europe in relation to incidence and mortality. Int. J. Cancer 2006, 119, 2417–2422. [Google Scholar] [CrossRef]
- Wolters, R.; Regierer, A.C.; Schwentner, L.; Geyer, V.; Possinger, K.; Kreienberg, R.; Wischnewsky, M.B.; Wockel, A. A comparison of international breast cancer guidelines—Do the national guidelines differ in treatment recommendations? Eur. J. Cancer 2012, 48, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Malek, D.; Kaab-Sanyal, V. Implementation of the German Mammography Screening Program (German MSP) and First Results for Initial Examinations, 2005–2009. Breast Care 2016, 11, 183–187. [Google Scholar] [CrossRef] [PubMed]
- Siu, A.L. Screening for Breast Cancer: U.S. Preventive Services Task Force Recommendation Statement. Ann. Intern. Med. 2016, 164, 279–296. [Google Scholar] [CrossRef]
- Sant, M.; Allemani, C.; Santaquilani, M.; Knijn, A.; Marchesi, F.; Capocaccia, R. EUROCARE-4. Survival of cancer patients diagnosed in 1995–1999. Results and commentary. Eur. J. Cancer 2009, 45, 931–991. [Google Scholar] [CrossRef]
- Jansen, L.; Castro, F.A.; Gondos, A.; Krilaviciute, A.; Barnes, B.; Eberle, A.; Emrich, K.; Hentschel, S.; Holleczek, B.; Katalinic, A.; et al. Recent cancer survival in Germany: An analysis of common and less common cancers. Int. J. Cancer 2015, 136, 2649–2658. [Google Scholar] [CrossRef]
- Surveillance, Epidemiology, and End Results (SEER) Program (www.seer.cancer.gov) SEER*Stat Database: Mortality—All COD, Aggregated With State, Total U.S. (1969–2016) <Katrina/Rita Population Adjustment>, National Cancer Institute, DCCPS, Surveillance Research Program, released December 2018. Underlying mortality data provided by NCHS (www.cdc.gov/nchs).
- Surveillance, Epidemiology, and End Results (SEER) Program (www.seer.cancer.gov) SEER*Stat Database: Incidence–SEER 9 Regs Research Data, Nov 2017 Sub (1973–2015) <Katrina/Rita Population Adjustment>–Linked To County Attributes–Total U.S., 1969–2016 Counties; & Research Data (1973–2015), National Cancer Institute, DCCPS, Surveillance Research Program, released April 2018, based on the November 2017 submission.
- Sinn, H.P.; Kreipe, H. A Brief Overview of the WHO Classification of Breast Tumors, 4th Edition, Focusing on Issues and Updates from the 3rd Edition. Breast Care 2013, 8, 149–154. [Google Scholar] [CrossRef]
- Brenner, H.; Gefeller, O.; Hakulinen, T. Period analysis for ‘up-to-date’ cancer survival data: Theory, empirical evaluation, computational realisation and applications. Eur. J. Cancer 2004, 40, 326–335. [Google Scholar] [CrossRef]
- Corazziari, I.; Quinn, M.; Capocaccia, R. Standard cancer patient population for age standardising survival ratios. Eur. J. Cancer 2004, 40, 2307–2316. [Google Scholar] [CrossRef]
- Ederer, F.; Heise, H. Instructions to IBM 650 Programmers in Processing Survival Computations; National Cancer Institute: Bethesda, MD, USA, 1959. [Google Scholar]
- Federal Statistical Office. Genesis Online Database. Life Tables (Period Life Tables): Germany, Years, Sex, Age. Table 12621-0001; years 2004-2015; Federal Statistical Office: Wiesbaden, Germany.
- Expected Survival Life Tables. Surveillance, Epidemiology, and End Results Program Web Site. Available online: http://seer.cancer.gov/expsurvival/ (accessed on 15 September 2016).
- Fann, J.C.; Chang, K.J.; Hsu, C.Y.; Yen, A.M.; Yu, C.P.; Chen, S.L.; Kuo, W.H.; Tabár, L.; Chen, H.H. Impact of Overdiagnosis on Long-Term Breast Cancer Survival. Cancers 2019, 11, 325. [Google Scholar] [CrossRef] [PubMed]
- Autier, P.; Boniol, M.; Koechlin, A.; Pizot, C.; Boniol, M. Effectiveness of and overdiagnosis from mammography screening in the Netherlands: Population based study. BMJ 2017, 359, j5224. [Google Scholar] [CrossRef] [PubMed]
- Katalinic, A.; Eisemann, N.; Kraywinkel, K.; Noftz, M.R.; Hubner, J. Breast cancer incidence and mortality before and after implementation of the German mammography screening program. Int. J. Cancer 2019. [Google Scholar] [CrossRef] [PubMed]
- Glass, A.G.; Lacey, J.V., Jr.; Carreon, J.D.; Hoover, R.N. Breast cancer incidence, 1980–2006: Combined roles of menopausal hormone therapy, screening mammography, and estrogen receptor status. J. Natl. Cancer Inst. 2007, 99, 1152–1161. [Google Scholar] [CrossRef] [PubMed]
- Lerda, D.; Deandrea, S.; Freeman, C.; López-Alcalde, J.; Neamtiu, L.; Nicholl, C.; Nicholson, N.; Ulütuck, A.; Villanueva, S. Report of a European Survey on the Organisation of Breast Cancer Care Services; European Commission: Luxembourg, 2014. [Google Scholar]
- Perleth, M. Anwendung von Methoden des Qualitätsmanagements auf Präventionsmaßnahmen. Master’s Thesis, Hannover Medical School, Hanover, Germany, 1998. [Google Scholar]
- Holleczek, B.; Brenner, H. Trends of population-based breast cancer survival in Germany and the US: Decreasing discrepancies, but persistent survival gap of elderly patients in Germany. BMC Cancer 2012, 12, 317. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention (CDC). National Center for Health Statistics. Available online: https://www.cdc.gov/nchs/data/hus/2017/070.pdf (accessed on 21 August 2019).
- Kääb-Sanyal, V.; Wegener, B.; Malek, D. Ergebnisse des Mammographie-Screening-Programs in Deutschland. In Jahresbericht Qualitätssicherung 2014; Kooperationsgemeinschaft Mammographie: Berlin, Germany, 2016. [Google Scholar]
- Starker, A.; Buttmann-Schweiger, N.; Krause, L.; Barnes, B.; Kraywinkel, K.; Holmberg, C. Cancer screening in Germany: Availability and participation. Bundesgesundheitsblatt Gesundheitsforschung Gesundheitsschutz 2018, 61, 1491–1499. [Google Scholar] [CrossRef]
- Günster, C.; Klauber, J.; Robra, B.-P.; Schmacke, N.; Schmuker, C. (Eds.) Versorgungs-Report Früherkennung; Medizinisch Wissenschaftliche Verlagsgesellschaft: Berlin, Germany, 2019. [Google Scholar] [CrossRef]
- Berkemeyer, S.; Lemke, D.; Hense, H.W. Incidence and Mortality Trends in German Women with Breast Cancer Using Age, Period and Cohort 1999 to 2008. PLoS ONE 2016, 11, e0150723. [Google Scholar] [CrossRef][Green Version]
- Duggan, M.A.; Anderson, W.F.; Altekruse, S.; Penberthy, L.; Sherman, M.E. The Surveillance, Epidemiology, and End Results (SEER) Program and Pathology: Toward Strengthening the Critical Relationship. Am. J. Surg. Pathol. 2016, 40, e94–e102. [Google Scholar] [CrossRef]
| Factor | Germany N (%) b | United States N (%) b |
|---|---|---|
| N | 93,721 | 68,776 |
| Excluded DCO cases | 4209 (4.3) | 360 (0.5) |
| Age at diagnosis | ||
| 15–29 | 455 (0.5) | 380 (0.6) |
| 30–39 | 3111 (3.3) | 2819 (4.1) |
| 40–44 | 4482 (4.8) | 3910 (5.7) |
| 45–49 | 8609 (9.2) | 6080 (8.8) |
| 50–54 | 11,762 (12.6) | 7870 (11.4) |
| 55–59 | 9851 (10.5) | 8438 (12.3) |
| 60–64 | 11,511 (12.3) | 9449 (13.7) |
| 65–69 | 10,577 (11.3) | 9635 (14.0) |
| 70–74 | 10,371 (11.1) | 7502 (10.9) |
| 75–79 | 10,994 (11.7) | 5415 (7.9) |
| 80+ | 11,998 (12.8) | 7278 (10.6) |
| Mean (standard deviation) | 63.7 (13.9) | 61.9 (13.6) |
| Morphology | ||
| Invasive carcinoma of no special type | 69,376 (74.0) | 49,938 (72.6) |
| Pleomorphic carcinoma | 2004 (2.1) | 3621 (5.3) |
| Invasive lobular carcinoma | 12,033 (12.8) | 7106 (10.3) |
| Tubular carcinoma | 875 (0.9) | 326 (0.5) |
| Mucinous carcinoma | 1590 (1.7) | 1319 (1.9) |
| Medullary carcinoma | 326 (0.3) | 103 (0.1) |
| Invasive micropapillary carcinoma | 180 (0.2) | 425 (0.6) |
| Metaplastic carcinoma of no special type | 430 (0.5) | 333 (0.5) |
| Invasive papillary carcinoma | 437 (0.5) | 280 (0.4) |
| Other/not specified | 6470 (6.9) | 5325 (7.7) |
| Grade | ||
| Well-differentiated (I) | 13,024 (13.9) | 16,273 (23.7) |
| Moderately differentiated (II) | 49,917 (53.3) | 28,737 (41.8) |
| Poorly differentiated (III) | 26,386 (28.2) | 19,810 (28.8) |
| Unknown | 4394 (4.7) | 3956 (5.8) |
| Tumor site (ICD-10 code C50.X) | ||
| Nipple and areola (0) | 995 (1.1) | 337 (0.5) |
| Central portion (1) | 4234 (4.5) | 3079 (4.5) |
| Upper-inner quadrant (2) | 10,186 (10.9) | 8590 (12.5) |
| Lower-inner quadrant (3) | 5281 (5.6) | 3873 (5.6) |
| Upper-outer quadrant (4) | 32,520 (34.7) | 22,774 (33.1) |
| Lower-outer quadrant (5) | 7355 (7.8) | 5245 (7.6) |
| Axillary tail (6) | 173 (0.2) | 352 (0.5) |
| Overlapping sites (7) | 13,008 (13.9) | 15,794 (23.0) |
| Unspecified site (9) | 19,969 (21.3) | 8732 (12.7) |
| Stage according to UICC/AJCCa | ||
| I | 36,708 (39.3) | 34,415 (50.1) |
| II | 36,758 (39.4) | 23,598 (34.4) |
| III | 12,777 (13.7) | 7119 (10.4) |
| IV | 7133 (7.6) | 3521 (5.1) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jansen, L.; Holleczek, B.; Kraywinkel, K.; Weberpals, J.; Schröder, C.C.; Eberle, A.; Emrich, K.; Kajüter, H.; Katalinic, A.; Kieschke, J.; et al. Divergent Patterns and Trends in Breast Cancer Incidence, Mortality and Survival Among Older Women in Germany and the United States. Cancers 2020, 12, 2419. https://doi.org/10.3390/cancers12092419
Jansen L, Holleczek B, Kraywinkel K, Weberpals J, Schröder CC, Eberle A, Emrich K, Kajüter H, Katalinic A, Kieschke J, et al. Divergent Patterns and Trends in Breast Cancer Incidence, Mortality and Survival Among Older Women in Germany and the United States. Cancers. 2020; 12(9):2419. https://doi.org/10.3390/cancers12092419
Chicago/Turabian StyleJansen, Lina, Bernd Holleczek, Klaus Kraywinkel, Janick Weberpals, Chloé Charlotte Schröder, Andrea Eberle, Katharina Emrich, Hiltraud Kajüter, Alexander Katalinic, Joachim Kieschke, and et al. 2020. "Divergent Patterns and Trends in Breast Cancer Incidence, Mortality and Survival Among Older Women in Germany and the United States" Cancers 12, no. 9: 2419. https://doi.org/10.3390/cancers12092419
APA StyleJansen, L., Holleczek, B., Kraywinkel, K., Weberpals, J., Schröder, C. C., Eberle, A., Emrich, K., Kajüter, H., Katalinic, A., Kieschke, J., Nennecke, A., Sirri, E., Heil, J., Schneeweiss, A., & Brenner, H., for the GEKID Cancer Survival Working Group. (2020). Divergent Patterns and Trends in Breast Cancer Incidence, Mortality and Survival Among Older Women in Germany and the United States. Cancers, 12(9), 2419. https://doi.org/10.3390/cancers12092419





