RETRACTED: HSP70–eIF4G Interaction Promotes Protein Synthesis and Cell Proliferation in Hepatocellular Carcinoma
Abstract
1. Introduction
2. Results
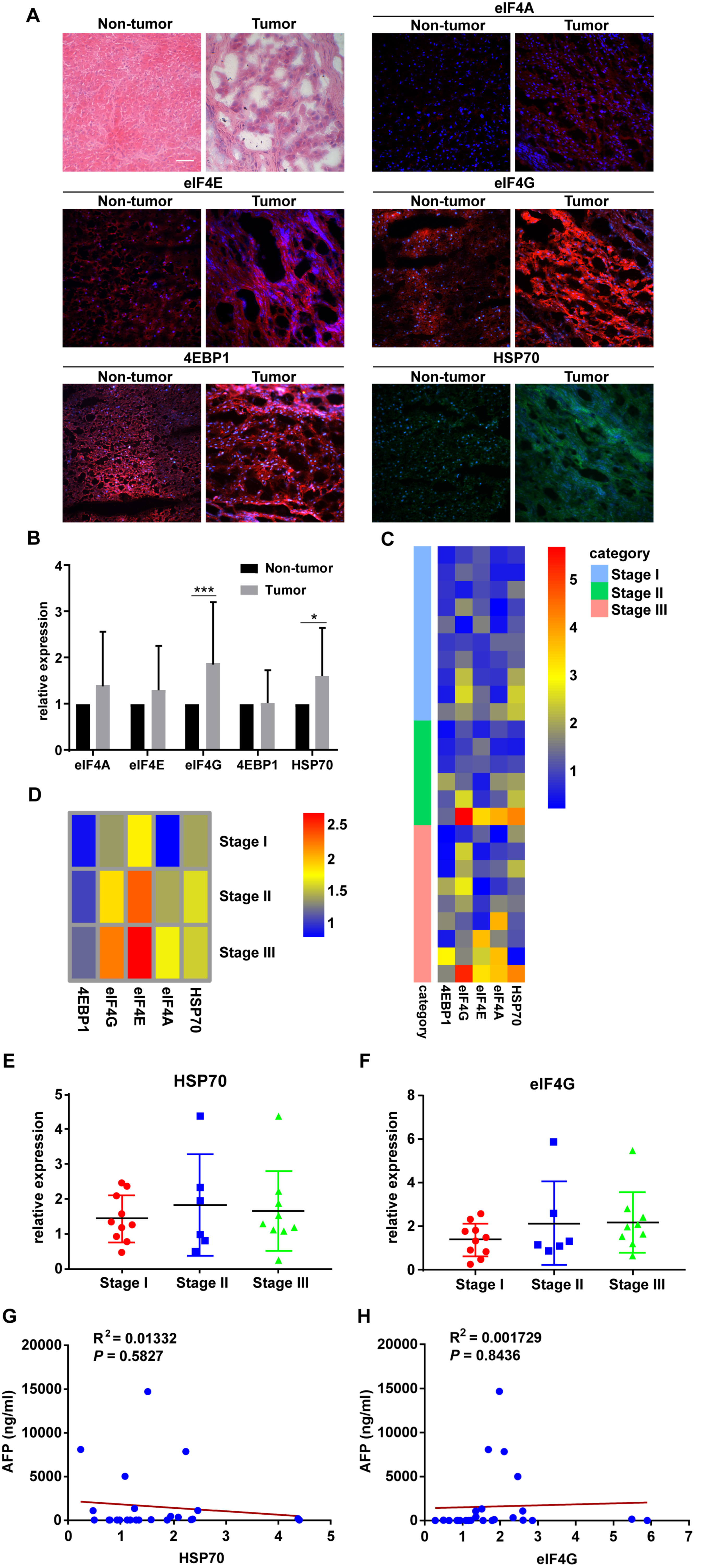
2.1. HSP70 and eIF4G Expression Are Significantly Higher in HCC Tumor Specimens
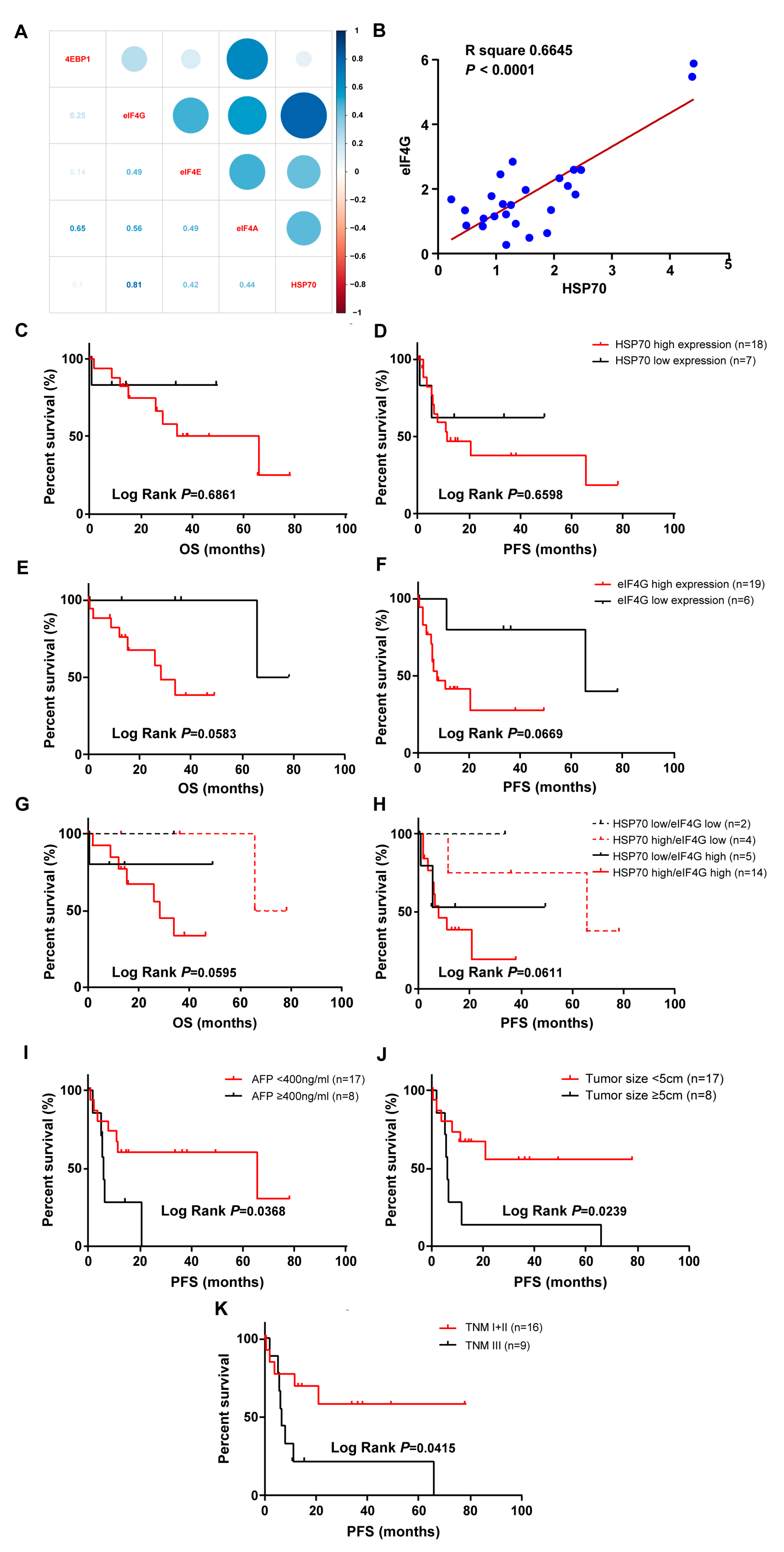
2.2. HSP70 and eIF4G Expression is Positively Correlated in Tumor Specimens of Patients with HCC
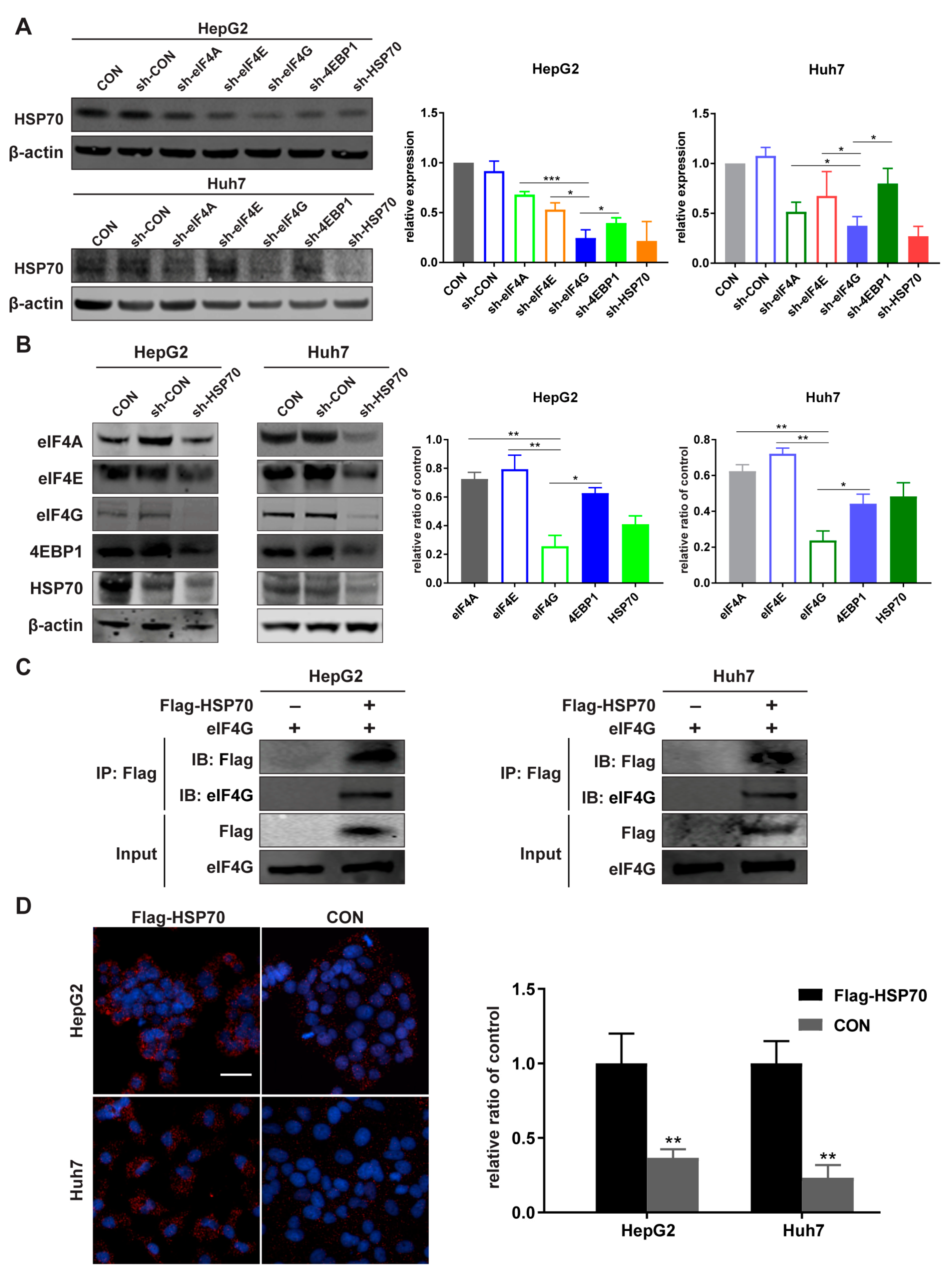
2.3. HSP70 Interacts with eIF4G in HCC Cell Lines
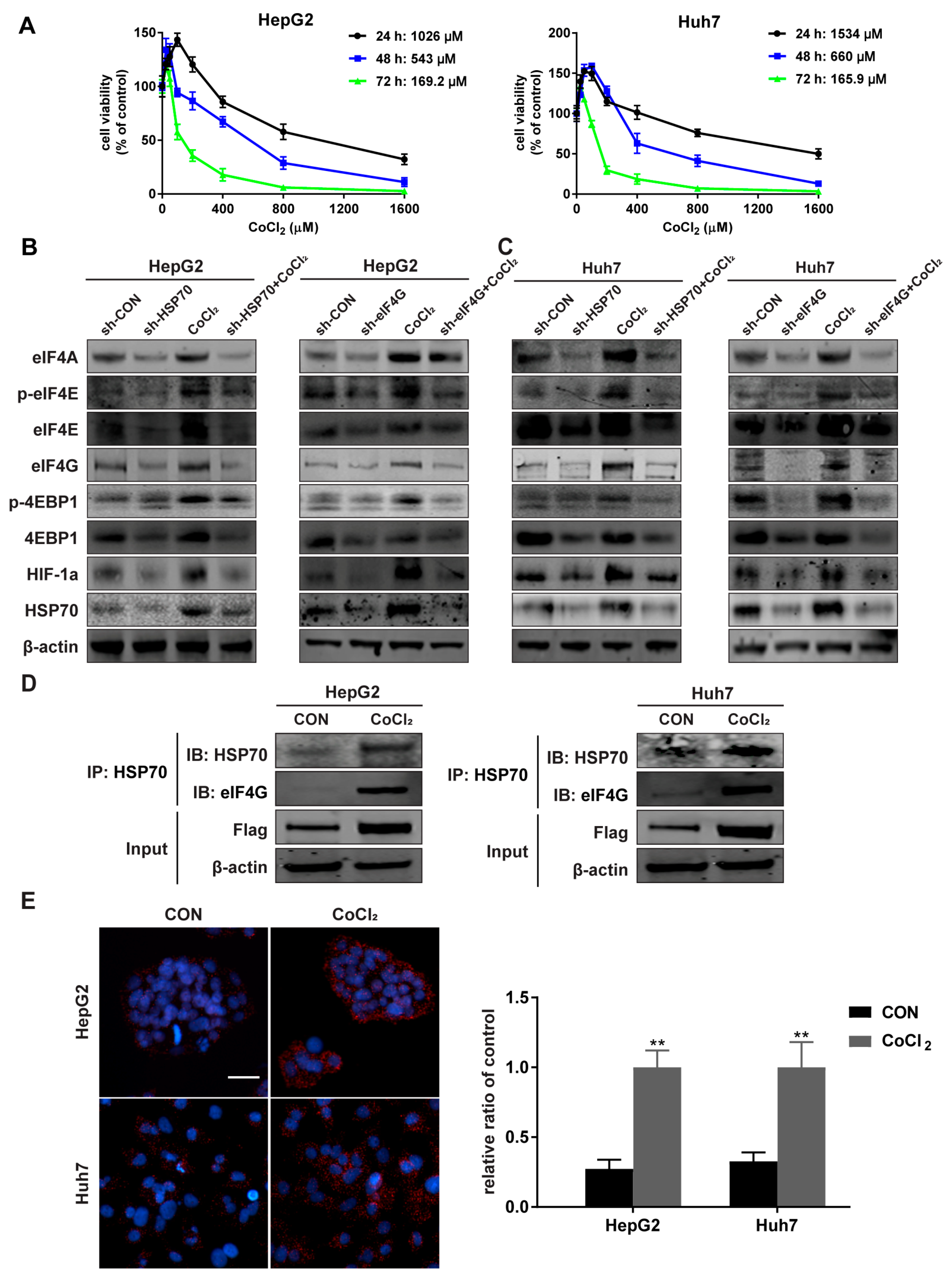
2.4. The Interaction between HSP70 and eIF4G Increases under Hypoxia
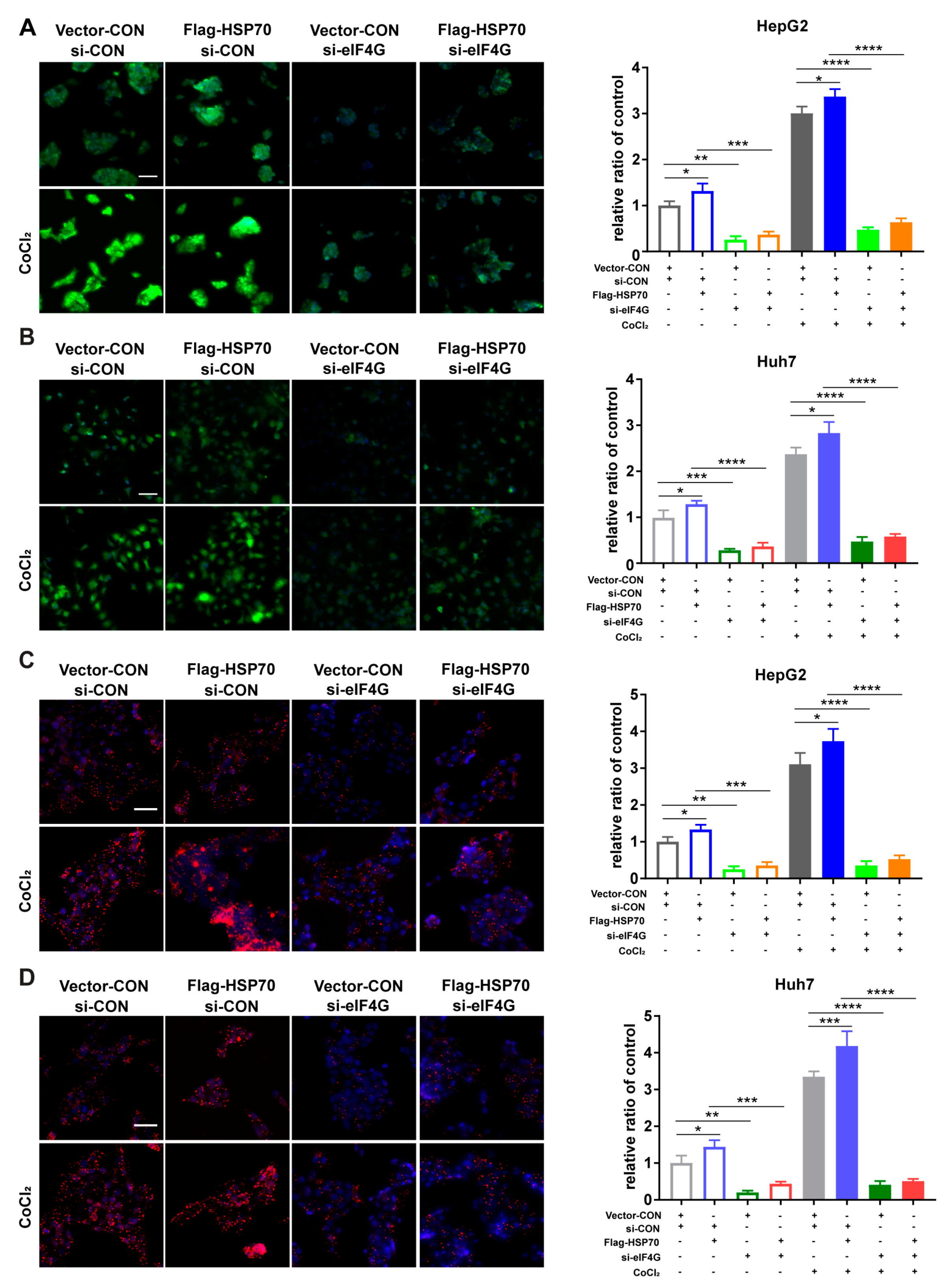
2.5. HSP70–eIF4G Interaction Promotes Cellular Protein Synthesis
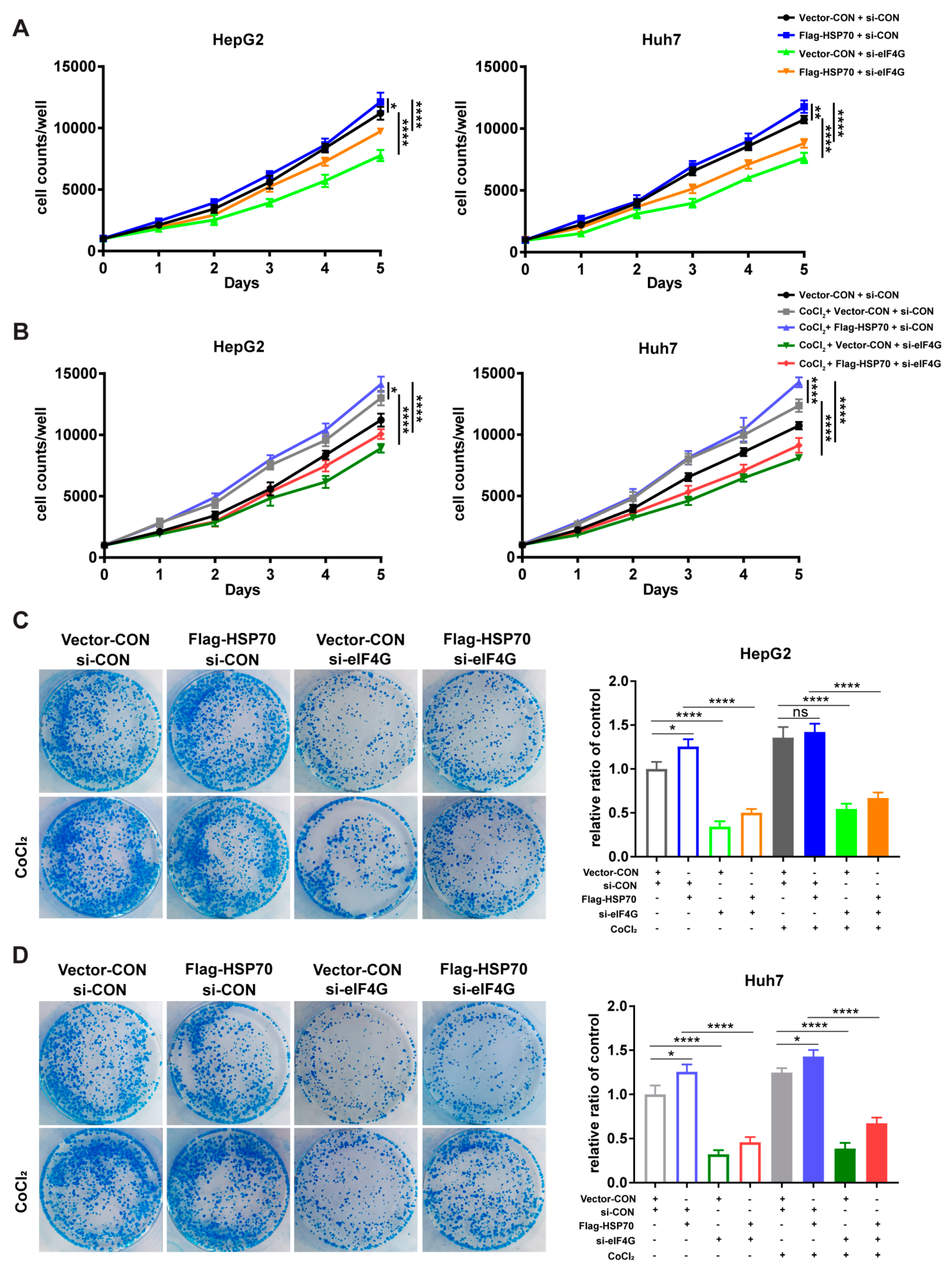
2.6. HSP70–eIF4G Interaction Promotes Cell Proliferation
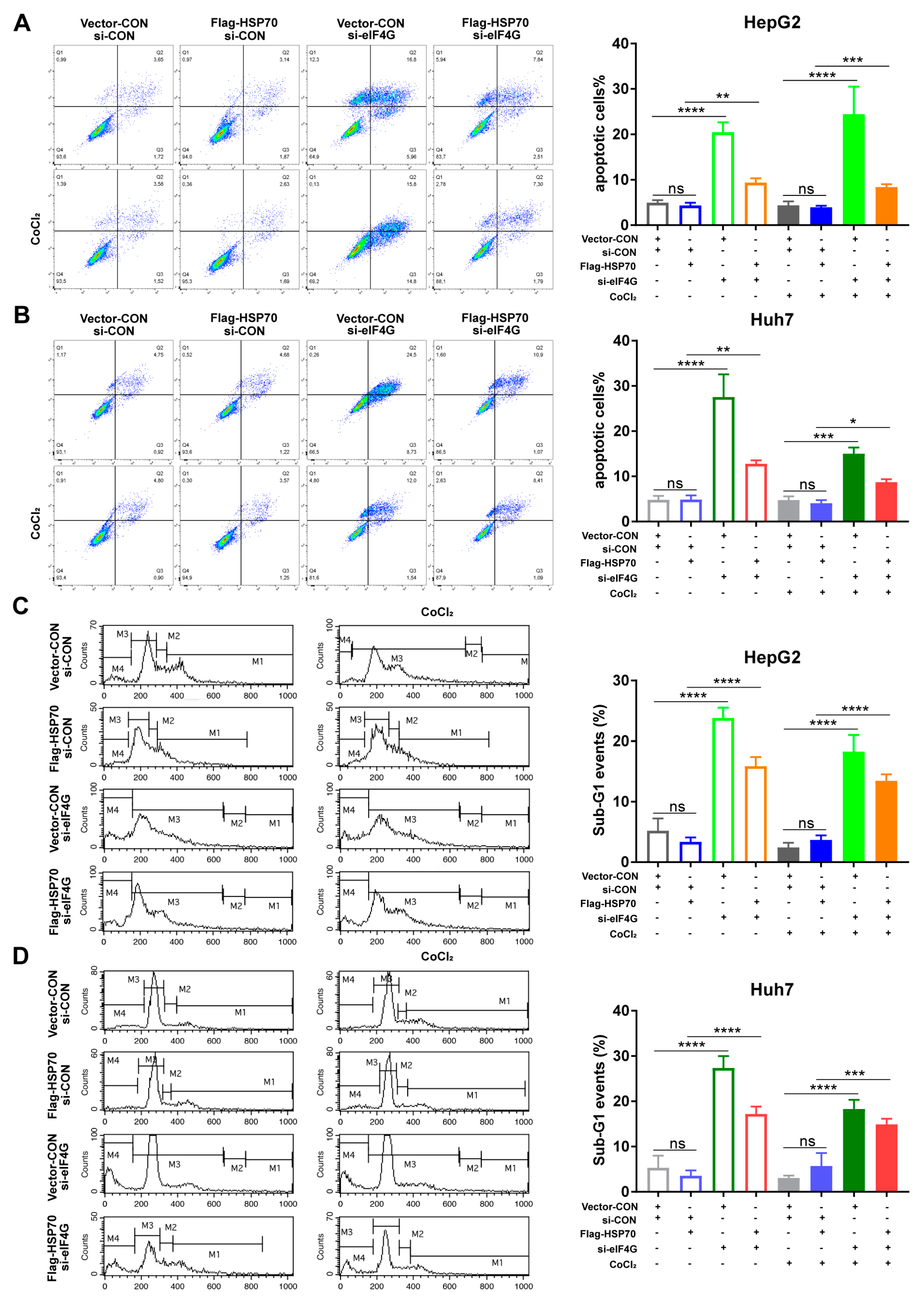
2.7. HSP70–eIF4G Interaction Inhibits Cell Apoptosis
3. Discussion
4. Materials and Methods
4.1. Clinical Specimens
4.2. Cell Culture and Reagents
4.3. Immunofluorescence (IF)
4.4. Gene Survival Analysis
4.5. Short Hairpin RNA (shRNA), Small Interfering RNA (siRNA), and Flag-HSP70 Plasmid Transfection
4.6. Co-Immunoprecipitation (Co-IP)
4.7. Cell Proliferation Assay
4.8. Western Blot
4.9. Colony Formation Assay
4.10. Annexin V-FITC/PI Staining Apoptosis Assay and Cell Cycle Assay
4.11. Click-iT® HPG Alexa Fluor® Protein Synthesis Assay
4.12. In Situ Proximity Ligation Assay
4.13. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| HCC | Hepatocellular carcinoma |
| OS | Overall survival |
| HSPs | Heat shock proteins |
| HSP70 | Heat shock protein 70 |
| DFS | Disease-free survival |
| eIF4G | Eukaryotic translation initiation factor 4G |
| eIF4F | Eukaryotic translation initiation factor 4F |
| eIF4A | Eukaryotic translation initiation factor 4A |
| eIF4E | Eukaryotic translation initiation factor 4E |
| 4EBP1 | Eukaryotic translation initiation factor 4E binding protein 1 |
| IRES | internal ribosomal entry site |
| H&E | hematoxylin and eosin stain |
| AFP | alpha-fetoprotein |
| NASH | nonal- coholic steatohepatitis |
| ECGO PS | Eastern Cooperative Oncology Group Performance Status |
| BCLC | Barcelona clinic liver cancer |
| TNM | Tumor, Node, Metastasis |
| PFS | Progression-free survival |
| Co-IP | Co-Immunoprecipitation |
| CoCl2 | Cobalt chloride |
| IC50 | Proximity Ligation Assay |
| Apaf-1 | apoptosis protease activating factor 1 |
| Ask-1 | apoptosis signal-regulated kinase 1 |
| MAPK | mitogen-activated protein kinase |
| mHSP70 | membrane HSP70 |
| NSCLC | non-small cell lung carcinoma |
| RCT | Radiochemotherapy |
| IL-2 | Interleukin-2 |
| IL-5 | Interleukin-5 |
| ERK | extracellular-signal-regulated kinase |
| IF | Immunofluorescence |
| shRNA | Short hairpin RNA |
| siRNA | Small interfering RNA |
References
- El-Serag, H.B.; Rudolph, K.L. Hepatocellular carcinoma: Epidemiology and molecular carcinogenesis. Gastroenterology 2007, 132, 2557–2576. [Google Scholar] [CrossRef] [PubMed]
- Jemal, A.; Ward, E.M.; Johnson, C.J.; Cronin, K.A.; Ma, J.; Ryerson, B.; Mariotto, A.; Lake, A.J.; Wilson, R.; Sherman, R.L.; et al. Annual Report to the Nation on the Status of Cancer, 1975–2014, Featuring Survival. J. Natl Cancer Inst. 2017, 109, djx030. [Google Scholar] [CrossRef] [PubMed]
- Cha, C.H.; Saif, M.W.; Yamane, B.H.; Weber, S.M. Hepatocellular carcinoma: Current management. Curr. Probl. Surg. 2010, 47, 10–67. [Google Scholar] [CrossRef] [PubMed]
- Rampone, B.; Schiavone, B.; Martino, A.; Viviano, C.; Confuorto, G. Current management strategy of hepatocellular carcinoma. World J. Gastroenterol. 2009, 15, 3210–3216. [Google Scholar] [CrossRef] [PubMed]
- Llovet, J.M.; Zucman-Rossi, J.; Pikarsky, E.; Sangro, B.; Schwartz, M.; Sherman, M.; Gores, G. Hepatocellular carcinoma. Nat. Rev. Dis. Primers 2016, 2, 16018. [Google Scholar] [CrossRef]
- Llovet, J.M.; Ricci, S.; Mazzaferro, V.; Hilgard, P.; Gane, E.; Blanc, J.F.; de Oliveira, A.C.; Santoro, A.; Raoul, J.L.; Forner, A.; et al. Sorafenib in advanced hepatocellular carcinoma. N. Engl. J. Med. 2008, 359, 378–390. [Google Scholar] [CrossRef]
- Morimoto, R.I. Regulation of the heat shock transcriptional response: Cross talk between a family of heat shock factors, molecular chaperones, and negative regulators. Genes. Dev. 1998, 12, 3788–3796. [Google Scholar] [CrossRef]
- Murphy, M.E. The HSP70 family and cancer. Carcinogenesis 2013, 34, 1181–1188. [Google Scholar] [CrossRef]
- Kim, J.Y.; Han, Y.; Lee, J.E.; Yenari, M.A. The 70-kDa heat shock protein (Hsp70) as a therapeutic target for stroke. Expert. Opin. Ther. Targets 2018, 22, 191–199. [Google Scholar] [CrossRef]
- Zorzi, E.; Bonvini, P. Inducible hsp70 in the regulation of cancer cell survival: Analysis of chaperone induction, expression and activity. Cancers 2011, 3, 3921–3956. [Google Scholar] [CrossRef]
- Milner, C.M.; Campbell, R.D. Structure and expression of the three MHC-linked HSP70 genes. Immunogenetics 1990, 32, 242–251. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.; Hunt, C.; Morimoto, R. Structure and expression of the human gene encoding major heat shock protein HSP70. Mol. Cell Biol. 1985, 5, 330–341. [Google Scholar] [CrossRef] [PubMed]
- Bukau, B.; Weissman, J.; Horwich, A. Molecular chaperones and protein quality control. Cell 2006, 125, 443–451. [Google Scholar] [CrossRef] [PubMed]
- Hartl, F.U.; Hayer-Hartl, M. Molecular chaperones in the cytosol: From nascent chain to folded protein. Science 2002, 295, 1852–1858. [Google Scholar] [CrossRef] [PubMed]
- Young, J.C.; Barral, J.M.; Ulrich Hartl, F. More than folding: Localized functions of cytosolic chaperones. Trends Biochem Sci. 2003, 28, 541–547. [Google Scholar] [CrossRef] [PubMed]
- De Los Rios, P.; Ben-Zvi, A.; Slutsky, O.; Azem, A.; Goloubinoff, P. Hsp70 chaperones accelerate protein translocation and the unfolding of stable protein aggregates by entropic pulling. Proc. Natl. Acad Sci. USA 2006, 103, 6166–6171. [Google Scholar] [CrossRef]
- Yang, X.; Wang, J.; Zhou, Y.; Wang, Y.; Wang, S.; Zhang, W. Hsp70 promotes chemoresistance by blocking Bax mitochondrial translocation in ovarian cancer cells. Cancer Lett. 2012, 321, 137–143. [Google Scholar] [CrossRef]
- Kim, G.; Meriin, A.B.; Gabai, V.L.; Christians, E.; Benjamin, I.; Wilson, A.; Wolozin, B.; Sherman, M.Y. The heat shock transcription factor Hsf1 is downregulated in DNA damage-associated senescence, contributing to the maintenance of senescence phenotype. Aging Cell 2012, 11, 617–627. [Google Scholar] [CrossRef]
- Daugaard, M.; Kirkegaard-Sorensen, T.; Ostenfeld, M.S.; Aaboe, M.; Hoyer-Hansen, M.; Orntoft, T.F.; Rohde, M.; Jaattela, M. Lens epithelium-derived growth factor is an Hsp70-2 regulated guardian of lysosomal stability in human cancer. Cancer Res. 2007, 67, 2559–2567. [Google Scholar] [CrossRef]
- Gong, J.; Weng, D.; Eguchi, T.; Murshid, A.; Sherman, M.Y.; Song, B.; Calderwood, S.K. Targeting the hsp70 gene delays mammary tumor initiation and inhibits tumor cell metastasis. Oncogene 2015, 34, 5460–5471. [Google Scholar] [CrossRef]
- Gullo, C.A.; Teoh, G. Heat shock proteins: To present or not, that is the question. Immunol. Lett 2004, 94, 1–10. [Google Scholar] [CrossRef] [PubMed]
- 2Bruemmer-Smith, S.; Stuber, F.; Schroeder, S. Protective functions of intracellular heat-shock protein (HSP) 70-expression in patients with severe sepsis. Intensive Care. Med. 2001, 27, 1835–1841. [Google Scholar] [CrossRef] [PubMed]
- Weinstein, P.R.; Hong, S.; Sharp, F.R. Molecular identification of the ischemic penumbra. Stroke 2004, 35, 2666–2670. [Google Scholar] [CrossRef]
- International Consensus Group for Hepatocellular Neoplasia. Pathologic diagnosis of early hepatocellular carcinoma: A report of the international consensus group for hepatocellular neoplasia. Hepatology 2009, 49, 658–664. [Google Scholar] [CrossRef] [PubMed]
- Farinati, F.; Marino, D.; De Giorgio, M.; Baldan, A.; Cantarini, M.; Cursaro, C.; Rapaccini, G.; Del Poggio, P.; Di Nolfo, M.A.; Benvegnu, L.; et al. Diagnostic and prognostic role of alpha-fetoprotein in hepatocellular carcinoma: Both or neither? Am. J. Gastroenterol. 2006, 101, 524–532. [Google Scholar] [CrossRef] [PubMed]
- Shin, E.; Ryu, H.S.; Kim, S.H.; Jung, H.; Jang, J.J.; Lee, K. The clinicopathological significance of heat shock protein 70 and glutamine synthetase expression in hepatocellular carcinoma. J. Hepatobiliary Pancreat Sci. 2011, 18, 544–550. [Google Scholar] [CrossRef]
- Kang, G.H.; Lee, B.S.; Lee, E.S.; Kim, S.H.; Lee, H.Y.; Kang, D.Y. Prognostic significance of p53, mTOR, c-Met, IGF-1R, and HSP70 overexpression after the resection of hepatocellular carcinoma. Gut. Liver. 2014, 8, 79–87. [Google Scholar] [CrossRef]
- Silvera, D.; Formenti, S.C.; Schneider, R.J. Translational control in cancer. Nat. Rev. Cancer 2010, 10, 254–266. [Google Scholar] [CrossRef]
- Moerke, N.J.; Aktas, H.; Chen, H.; Cantel, S.; Reibarkh, M.Y.; Fahmy, A.; Gross, J.D.; Degterev, A.; Yuan, J.; Chorev, M.; et al. Small-molecule inhibition of the interaction between the translation initiation factors eIF4E and eIF4G. Cell 2007, 128, 257–267. [Google Scholar] [CrossRef]
- Pestova, T.V.; Kolupaeva, V.G.; Lomakin, I.B.; Pilipenko, E.V.; Shatsky, I.N.; Agol, V.I.; Hellen, C.U. Molecular mechanisms of translation initiation in eukaryotes. Proc. Nat.l Acad. Sci. USA 2001, 98, 7029–7036. [Google Scholar] [CrossRef]
- Grifo, J.A.; Tahara, S.M.; Morgan, M.A.; Shatkin, A.J.; Merrick, W.C. New initiation factor activity required for globin mRNA translation. J. Biol. Chem. 1983, 258, 5804–5810. [Google Scholar] [PubMed]
- Parsyan, A.; Svitkin, Y.; Shahbazian, D.; Gkogkas, C.; Lasko, P.; Merrick, W.C.; Sonenberg, N. mRNA helicases: The tacticians of translational control. Nat. Rev. Mol. Cell Biol. 2011, 12, 235–245. [Google Scholar] [CrossRef] [PubMed]
- Richter, J.D.; Sonenberg, N. Regulation of cap-dependent translation by eIF4E inhibitory proteins. Nature 2005, 433, 477–480. [Google Scholar] [CrossRef] [PubMed]
- Kapp, L.D.; Lorsch, J.R. The molecular mechanics of eukaryotic translation. Annu. Rev. Biochem. 2004, 73, 657–704. [Google Scholar] [CrossRef]
- Mader, S.; Lee, H.; Pause, A.; Sonenberg, N. The translation initiation factor eIF-4E binds to a common motif shared by the translation factor eIF-4 gamma and the translational repressors 4E-binding proteins. Mol. Cell Biol. 1995, 15, 4990–4997. [Google Scholar] [CrossRef]
- De Benedetti, A.; Graff, J.R. eIF-4E expression and its role in malignancies and metastases. Oncogene 2004, 23, 3189–3199. [Google Scholar] [CrossRef]
- Pause, A.; Belsham, G.J.; Gingras, A.C.; Donze, O.; Lin, T.A.; Lawrence, J.C., Jr.; Sonenberg, N. Insulin-dependent stimulation of protein synthesis by phosphorylation of a regulator of 5′-cap function. Nature 1994, 371, 762–767. [Google Scholar] [CrossRef]
- Sonenberg, N.; Hinnebusch, A.G. Regulation of translation initiation in eukaryotes: Mechanisms and biological. targets. Cell 2009, 136, 731–745. [Google Scholar] [CrossRef]
- Laplante, M.; Sabatini, D.M. mTOR signaling in growth control and disease. Cell 2012, 149, 274–293. [Google Scholar] [CrossRef]
- Tu, L.; Liu, Z.; He, X.; He, Y.; Yang, H.; Jiang, Q.; Xie, S.; Xiao, G.; Li, X.; Yao, K.; et al. Over-expression of eukaryotic translation initiation factor 4 gamma 1 correlates with tumor progression and poor prognosis in nasopharyngeal carcinoma. Mol. Cancer 2010, 9, 78. [Google Scholar] [CrossRef]
- Silvera, D.; Arju, R.; Darvishian, F.; Levine, P.H.; Zolfaghari, L.; Goldberg, J.; Hochman, T.; Formenti, S.C.; Schneider, R.J. Essential role for eIF4GI overexpression in the pathogenesis of inflammatory breast cancer. Nat. Cell Biol. 2009, 11, 903–908. [Google Scholar] [CrossRef] [PubMed]
- Comtesse, N.; Keller, A.; Diesinger, I.; Bauer, C.; Kayser, K.; Huwer, H.; Lenhof, H.P.; Meese, E. Frequent overexpression of the genes FXR1, CLAPM1 and EIF4G located on amplicon 3q26-27 in squamous cell carcinoma of the lung. Int. J. Cancer 2007, 120, 2538–2544. [Google Scholar] [CrossRef] [PubMed]
- An, S.L.; Xiao, T.; Wang, L.M.; Rong, W.Q.; Wu, F.; Feng, L.; Liu, F.Q.; Tian, F.; Wu, J.X. Prognostic Significance of Preoperative Serum Alpha- fetoprotein in Hepatocellular Carcinoma and Correlation with Clinicopathological Factors: A Single-center Experience from China. Asian. Pac. J. Cancer Prev. 2015, 16, 4421–4427. [Google Scholar] [CrossRef] [PubMed]
- Blank, S.; Wang, Q.; Fiel, M.I.; Luan, W.; Kim, K.W.; Kadri, H.; Mandeli, J.; Hiotis, S.P. Assessing prognostic significance of preoperative alpha-fetoprotein in hepatitis B-associated hepatocellular carcinoma: Normal is not the new normal. Ann. Surg. Oncol. 2014, 21, 986–994. [Google Scholar] [CrossRef]
- Sauzay, C.; Petit, A.; Bourgeois, A.M.; Barbare, J.C.; Chauffert, B.; Galmiche, A.; Houessinon, A. Alpha-foetoprotein (AFP): A multi-purpose marker in hepatocellular carcinoma. Clin. Chim.Acta. 2016, 463, 39–44. [Google Scholar] [CrossRef]
- Edge SB, B.D.; Compton, C.C.; Fritz, A.G.; Greene, F.L.; Trotti, A. (Eds.) AJCC Cancer Staging Manual, 7th ed.; Springer: New York, NY, USA, 2010; pp. 593–597. [Google Scholar]
- Soderberg, O.; Gullberg, M.; Jarvius, M.; Ridderstrale, K.; Leuchowius, K.J.; Jarvius, J.; Wester, K.; Hydbring, P.; Bahram, F.; Larsson, L.G.; et al. Direct observation of individual endogenous protein complexes in situ by proximity ligation. Nat. Methods 2006, 3, 995–1000. [Google Scholar] [CrossRef]
- Stankiewicz, A.R.; Lachapelle, G.; Foo, C.P.; Radicioni, S.M.; Mosser, D.D. Hsp70 inhibits heat-induced apoptosis upstream of mitochondria by preventing Bax translocation. J. Biol. Chem. 2005, 280, 38729–38739. [Google Scholar] [CrossRef]
- Beere, H.M.; Wolf, B.B.; Cain, K.; Mosser, D.D.; Mahboubi, A.; Kuwana, T.; Tailor, P.; Morimoto, R.I.; Cohen, G.M.; Green, D.R. Heat-shock protein 70 inhibits apoptosis by preventing recruitment of procaspase-9 to the Apaf-1 apoptosome. Nat. Cell Biol. 2000, 2, 469–475. [Google Scholar] [CrossRef]
- Gao, Y.; Han, C.; Huang, H.; Xin, Y.; Xu, Y.; Luo, L.; Yin, Z. Heat shock protein 70 together with its co-chaperone CHIP inhibits TNF-alpha induced apoptosis by promoting proteasomal degradation of apoptosis signal-regulating kinase1. Apoptosis 2010, 15, 822–833. [Google Scholar] [CrossRef]
- Ammirante, M.; Rosati, A.; Arra, C.; Basile, A.; Falco, A.; Festa, M.; Pascale, M.; d’Avenia, M.; Marzullo, L.; Belisario, M.A.; et al. IKK{gamma} protein is a target of BAG3 regulatory activity in human tumor growth. Proc Natl Acad Sci. USA 2010, 107, 7497–7502. [Google Scholar] [CrossRef]
- Colvin, T.A.; Gabai, V.L.; Gong, J.; Calderwood, S.K.; Li, H.; Gummuluru, S.; Matchuk, O.N.; Smirnova, S.G.; Orlova, N.V.; Zamulaeva, I.A.; et al. Hsp70-Bag3 interactions regulate cancer-related signaling networks. Cancer Res. 2014, 74, 4731–4740. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.V.; Semenza, G.L. RACK1 vs. HSP90: Competition for HIF-1 alpha degradation vs. stabilization. Cell Cycle 2007, 6, 656–659. [Google Scholar] [CrossRef] [PubMed]
- Sharp, F.R.; Lu, A.; Tang, Y.; Millhorn, D.E. Multiple molecular penumbras after focal cerebral ischemia. J. Cereb. Blood Flow Metab. 2000, 20, 1011–1032. [Google Scholar] [CrossRef] [PubMed]
- Ren, A.; Yan, G.; You, B.; Sun, J. Down-regulation of mammalian sterile 20-like kinase 1 by heat shock protein 70 mediates cisplatin resistance in prostate cancer cells. Cancer Res. 2008, 68, 2266–2274. [Google Scholar] [CrossRef] [PubMed]
- Pocaly, M.; Lagarde, V.; Etienne, G.; Ribeil, J.A.; Claverol, S.; Bonneu, M.; Moreau-Gaudry, F.; Guyonnet-Duperat, V.; Hermine, O.; Melo, J.V.; et al. Overexpression of the heat-shock protein 70 is associated to imatinib resistance in chronic myeloid leukemia. Leukemia 2007, 21, 93–101. [Google Scholar] [CrossRef] [PubMed]
- Sheng, L.; Tang, T.; Liu, Y.; Ma, Y.; Wang, Z.; Tao, H.; Zhang, Y.; Qi, Z. Inducible HSP70 antagonizes cisplatininduced cell apoptosis through inhibition of the MAPK signaling pathway in HGC27 cells. Int. J. Mol Med 2018, 42, 2089–2097. [Google Scholar]
- Horton, L.E.; James, P.; Craig, E.A.; Hensold, J.O. The yeast hsp70 homologue Ssa is required for translation and interacts with Sis1 and Pab1 on translating ribosomes. J. Biol. Chem. 2001, 276, 14426–14433. [Google Scholar] [CrossRef]
- Cuesta, R.; Laroia, G.; Schneider, R.J. Chaperone hsp27 inhibits translation during heat shock by binding eIF4G and facilitating dissociation of cap-initiation complexes. Genes Dev. 2000, 14, 1460–1470. [Google Scholar]
- Finn, R.S.; Qin, S.; Ikeda, M.; Galle, P.R.; Ducreux, M.; Kim, T.Y.; Kudo, M.; Breder, V.; Merle, P.; Kaseb, A.O.; et al. Atezolizumab plus Bevacizumab in Unresectable Hepatocellular Carcinoma. N. Engl. J. Med. 2020, 382, 1894–1905. [Google Scholar] [CrossRef]
- Garcia, J.; Hurwitz, H.I.; Sandler, A.B.; Miles, D.; Coleman, R.L.; Deurloo, R.; Chinot, O.L. Bevacizumab (Avastin(R)) in cancer treatment: A review of 15 years of clinical experience and future outlook. Cancer Treat Rev. 2020, 86, 102017. [Google Scholar] [CrossRef]
- Kokowski, K.; Stangl, S.; Seier, S.; Hildebrandt, M.; Vaupel, P.; Multhoff, G. Radiochemotherapy combined with NK cell transfer followed by second-line PD-1 inhibition in a patient with NSCLC stage IIIb inducing long-term tumor control: A case study. Strahlenther Onkol. 2019, 195, 352–361. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.K.; Na, H.J.; Lee, W.R.; Jeoung, M.H.; Lee, S. Heat shock protein 70-1A is a novel angiogenic regulator. Biochem. Biophys Res. Commun. 2016, 469, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Park, S.L.; Chung, T.W.; Kim, S.; Hwang, B.; Kim, J.M.; Lee, H.M.; Cha, H.J.; Seo, Y.; Choe, S.Y.; Ha, K.T.; et al. HSP70-1 is required for interleukin-5-induced angiogenic responses through eNOS pathway. Sci. Rep. 2017, 7, 44687. [Google Scholar] [CrossRef] [PubMed]
- Menyhart, O.; Nagy, A.; Gyorffy, B. Determining consistent prognostic biomarkers of overall survival and vascular invasion in hepatocellular carcinoma. R. Soc. Open Sci. 2018, 5, 181006. [Google Scholar] [CrossRef]
| Characteristic | Group | N = 25 | Ratio (% of Total N) | p Value |
|---|---|---|---|---|
| Gender | Male | 16 | 64% | |
| Female | 9 | 36% | ||
| Age (years) | <50 | 3 | 12% | |
| ≥50 | 22 | 88% | ||
| AFP (ng/mL) | <400 | 17 | 68% | |
| ≥400 | 8 | 32% | ||
| Etiology | Hepatitis B | 2 | 8% | |
| Hepatitis C | 7 | 28% | ||
| Alcohol | 6 | 24% | ||
| NASH | 2 | 8% | ||
| Autoimmune | 1 | 4% | ||
| Vinyl chloride | 1 | 4% | ||
| Colorectal metastasis | 1 | 4% | ||
| Unknown | 5 | 20% | ||
| Liver cirrhosis | Present | 12 | 48% | |
| Absent | 13 | 52% | ||
| Tumor size (cm) | <5 | 17 | 68% | |
| ≥5 | 8 | 32% | ||
| Tumor number | Single | 19 | 76% | |
| Multiple | 6 | 24% | ||
| Edmondson-Steiner grade | G1 | 1 | 4% | |
| G2 | 14 | 56% | ||
| G3 | 10 | 40% | ||
| ECGO PS | 0 | 23 | 92% | |
| 1 | 2 | 8% | ||
| Child-Pugh class | A | 24 | 96% | |
| B | 1 | 4% | ||
| BCLC stage | 0 + A | 20 | 80% | |
| B | 5 | 20% | ||
| TNM stage | I + II | 16 | 64% | |
| III | 9 | 36% | ||
| Recurrence | Positive | 12 | 48% | |
| Negative | 13 | 52% | ||
| Recurrence Pattern | Intrahepatic only | 9 | 75% | |
| n = 12 | Extrahepatic only | 1 | 8.33% | |
| Both | 2 | 16.67% | ||
| Ki-67 expression | High | 11 | 44% | |
| Low | 14 | 56% | ||
| Midkine expression | High | 15 | 60% | |
| Low | 10 | 40% | ||
| HSP70 high expression | Tumor tissue | 18 | 72% | 0.0042 ** |
| Non-Tumor tissue | 7 | 30% | ||
| eIF4A high expression | Tumor tissue | 12 | 48% | >0.9999 |
| Non-Tumor tissue | 13 | 52% | ||
| eIF4E high expression | Tumor tissue | 14 | 56% | 0.5721 |
| Non-Tumor tissue | 11 | 44% | ||
| eIF4G high expression | Tumor tissue | 19 | 76% | 0.0005 *** |
| Non-Tumor tissue | 6 | 24% | ||
| 4EBP1 high expression | Tumor tissue | 10 | 40% | 0.2578 |
| Non-Tumor tissue | 15 | 60% | ||
| Stage | T | N | M | |
| I | T1 | N0 | M0 | |
| II | T2 | N0 | M0 | |
| IIIa | T3a | N0 | M0 | |
| IIIb | T3b | N0 | M0 | |
| IIIc | T4 | N0 | M0 | |
| IVa | Any T | N1 | M0 | |
| IVb | Any T | Any N | M1 |
| Characteristic | Group | HSP70 High Expression | HSP70 Low Expression | p Value |
|---|---|---|---|---|
| Gender | Male | 9 | 7 | 0.4013 |
| Female | 7 | 2 | ||
| Age (years) | <50 | 3 | 0 | 0.5343 |
| ≥50 | 15 | 7 | ||
| AFP (ng/mL) | <400 | 12 | 5 | >0.9999 |
| ≥400 | 6 | 2 | ||
| Etiology | Hepatitis B | 2 | 0 | 0.7124 |
| Hepatitis C | 4 | 3 | ||
| Alcohol | 5 | 1 | ||
| NASH | 1 | 1 | ||
| Autoimmune | 1 | 0 | ||
| Vinyl chloride | 0 | 1 | ||
| Colorectal metastasis | 0 | 1 | ||
| Unknown | 5 | 0 | ||
| Liver cirrhosis | Present | 9 | 3 | >0.9999 |
| Absent | 9 | 4 | ||
| Tumor size (cm) | <5 | 11 | 6 | 0.3623 |
| ≥5 | 7 | 1 | ||
| Tumor number | Single | 15 | 4 | 0.2985 |
| Multiple | 3 | 3 | ||
| Edmondson-Steiner grade | G1 | 0 | 1 | 0.0439 * |
| G2 | 9 | 5 | ||
| G3 | 9 | 1 | ||
| ECGO PS | 0 | 16 | 7 | >0.9999 |
| 1 | 2 | 0 | ||
| Child-Pugh class | A | 17 | 7 | >0.9999 |
| B | 1 | 0 | ||
| BCLC stage | 0 + A | 16 | 4 | 0.1130 |
| B | 2 | 3 | ||
| TNM stage | I + II | 10 | 6 | >0.9999 |
| III | 6 | 3 | ||
| Recurrence | Positive | 10 | 2 | 0.3783 |
| Negative | 8 | 5 | ||
| Recurrence Pattern | Intrahepatic only | 8 | 1 | 0.5665 |
| N = 12 | Extrahepatic only | 1 | 0 | |
| Both | 2 | 0 | ||
| Ki-67 expression | High | 7 | 4 | 0.6564 |
| Low | 11 | 3 | ||
| Midkine expression | High | 10 | 5 | 0.6592 |
| Low | 8 | 2 | ||
| Patient survival (%) | 1-year OS | 82.35 | 83.33 | 0.7055 |
| 1-year PFS | 47.06 | 62.5 | 0.1031 | |
| 3-year OS | 49.91 | 83.33 | 0.5712 | |
| 3-year PFS | 37.65 | 62.5 | 0.5290 | |
| 5-year OS | 49.91 | 0 | 0.5528 | |
| 5-year PFS | 37.65 | 0 | 0.3706 |
| Characteristic | Group | eIF4G High Expression | eIF4G Low Expression | p Value |
|---|---|---|---|---|
| Gender | Male | 13 | 3 | 0.6300 |
| Female | 6 | 3 | ||
| Age (years) | <50 | 1 | 2 | 0.1326 |
| ≥50 | 18 | 4 | ||
| AFP (ng/mL) | <400 | 11 | 6 | 0.1292 |
| ≥400 | 8 | 0 | ||
| Etiology | Hepatitis B | 1 | 1 | 0.0812 |
| Hepatitis C | 7 | 0 | ||
| Alcohol | 6 | 0 | ||
| NASH | 1 | 1 | ||
| Autoimmune | 0 | 1 | ||
| Vinyl chloride | 1 | 0 | ||
| Colorectal metastasis | 0 | 1 | ||
| Unknown | 3 | 2 | ||
| Liver cirrhosis | Present | 10 | 2 | 0.6447 |
| Absent | 9 | 4 | ||
| Tumor size (cm) | <5 | 13 | 4 | >0.9999 |
| ≥5 | 6 | 2 | ||
| Tumor number | Single | 13 | 6 | 0.2778 |
| Multiple | 6 | 0 | ||
| Edmondson-Steiner grade | G1 | 0 | 1 | 0.0694 |
| G2 | 10 | 4 | ||
| G3 | 9 | 1 | ||
| ECGO PS | 0 | 18 | 5 | 0.4300 |
| 1 | 1 | 1 | ||
| Child-Pugh class | A | 19 | 5 | 0.2400 |
| B | 0 | 1 | ||
| BCLC stage | 0 + A | 15 | 5 | >0.9999 |
| B | 4 | 1 | ||
| TNM stage | I + II | 11 | 5 | 0.3644 |
| III | 8 | 1 | ||
| Recurrence | Positive | 10 | 2 | 0.6447 |
| Negative | 9 | 4 | ||
| Recurrence Pattern | Intrahepatic only | 9 | 0 | 0.0271 * |
| n = 12 | Extrahepatic only | 0 | 1 | |
| Both | 1 | 1 | ||
| Ki-67 expression | High | 9 | 2 | 0.6609 |
| Low | 10 | 4 | ||
| Midkine expression | High | 12 | 3 | 0.6532 |
| Low | 7 | 3 | ||
| Patient survival (%) | 1-year OS | 75.89 | 100 | >0.9999 |
| 1-year PFS | 41.32 | 80 | 0.0953 | |
| 3-year OS | 38.55 | 100 | 0.1861 | |
| 3-year PFS | 27.55 | 80 | 0.2774 | |
| 5-year OS | 0 | 100 | 0.1753 | |
| 5-year PFS | 0 | 80 | 0.2335 |
| Characteristic | OS | PFS | ||
|---|---|---|---|---|
| No. of Patients (Died/Total) | p Value (Log-Rank-Test) | No. of Patients (Progress/Total) | p Value (Log-Rank-Test) | |
| Gender | ||||
| Male | 5/16 | 0.6030 | 7/16 | 0.9294 |
| Female | 4/9 | 6/9 | ||
| Age (years) | ||||
| <50 | 1/3 | 0.4527 | 1/3 | 0.2139 |
| ≥50 | 8/22 | 12/22 | ||
| AFP (ng/mL) | ||||
| <400 | 5/17 | 0.1863 | 7/17 | 0.0368 * |
| ≥400 | 4/8 | 6/8 | ||
| Etiology | ||||
| Hepatitis B | 0/2 | 0.2367 | 0/2 | 0.3502 |
| Hepatitis C | 2/7 | 3/7 | ||
| Alcohol | 2/6 | 3/6 | ||
| NASH | 1/2 | 1/2 | ||
| Autoimmune | 0/1 | 0/1 | ||
| Vinyl chloride | 0/1 | 1/1 | ||
| Colorectal metastasis | 0/1 | 0/1 | ||
| Unknown | 4/5 | 5/5 | ||
| Liver cirrhosis | ||||
| Present | 4/12 | 0.6010 | 5/12 | 0.2579 |
| Absent | 5/13 | 8/13 | ||
| Tumor size (cm) | ||||
| <5 | 5/17 | 0.4959 | 6/17 | 0.0239 * |
| ≥5 | 4/8 | 7/8 | ||
| Tumor number | ||||
| Single | 6/19 | 0.0744 | 9/19 | 0.1204 |
| Multiple | 3/6 | 4/6 | ||
| Edmondson-Steiner grade | ||||
| G1 | 0/1 | 0.5090 | 0/1 | 0.9778 |
| G2 | 4/14 | 7/14 | ||
| G3 | 5/10 | 6/10 | ||
| ECGO PS | ||||
| 0 | 8/23 | 0.7663 | 12/23 | 0.4295 |
| 1 | 1/2 | 1/2 | ||
| Child-Pugh class | ||||
| A | 9/24 | 0.4214 | 13/24 | 0.3483 |
| B | 0/1 | 0/1 | ||
| BCLC stage | ||||
| 0 + A | 8/20 | 0.4364 | 10/20 | 0.6930 |
| B | 1/5 | 3/5 | ||
| TNM stage | ||||
| I + II | 4/16 | 0.4385 | 5/16 | 0.0415 * |
| III | 5/9 | 8/9 | ||
| Recurrence | ||||
| Positive | 5/12 | 0.7374 | 9/12 | 0.3079 |
| Negative | 4/13 | 4/13 | ||
| Recurrence Pattern | ||||
| Intrahepatic only | 4/9 | 0.6937 | 6/9 | 0.3380 |
| Extrahepatic only | 1/1 | 1/1 | ||
| Both | 0/2 | 2/2 | ||
| Ki-67 expression | ||||
| High | 3/11 | 0.2863 | 5/11 | 0.6943 |
| Low | 6/14 | 8/14 | ||
| Midkine expression | ||||
| High | 4/15 | 0.5037 | 6/15 | 0.4995 |
| Low | 5/10 | 7/10 | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, M.; Wei, K.; Qian, B.; Feiler, S.; Lemekhova, A.; Büchler, M.W.; Hoffmann, K. RETRACTED: HSP70–eIF4G Interaction Promotes Protein Synthesis and Cell Proliferation in Hepatocellular Carcinoma. Cancers 2020, 12, 2262. https://doi.org/10.3390/cancers12082262
Wang M, Wei K, Qian B, Feiler S, Lemekhova A, Büchler MW, Hoffmann K. RETRACTED: HSP70–eIF4G Interaction Promotes Protein Synthesis and Cell Proliferation in Hepatocellular Carcinoma. Cancers. 2020; 12(8):2262. https://doi.org/10.3390/cancers12082262
Chicago/Turabian StyleWang, Meng, Kai Wei, Baifeng Qian, Svenja Feiler, Anastasia Lemekhova, Markus W. Büchler, and Katrin Hoffmann. 2020. "RETRACTED: HSP70–eIF4G Interaction Promotes Protein Synthesis and Cell Proliferation in Hepatocellular Carcinoma" Cancers 12, no. 8: 2262. https://doi.org/10.3390/cancers12082262
APA StyleWang, M., Wei, K., Qian, B., Feiler, S., Lemekhova, A., Büchler, M. W., & Hoffmann, K. (2020). RETRACTED: HSP70–eIF4G Interaction Promotes Protein Synthesis and Cell Proliferation in Hepatocellular Carcinoma. Cancers, 12(8), 2262. https://doi.org/10.3390/cancers12082262





