Neoadjuvant Intraperitoneal Chemotherapy in Patients with Pseudomyxoma Peritonei—A Novel Treatment Approach
Abstract
1. Introduction
1.1. Cytoreductive Surgery and Hyperthermic Intraperitoneal Chemotherapy
1.2. Neoadjuvant Intraperitoneal Chemotherapy
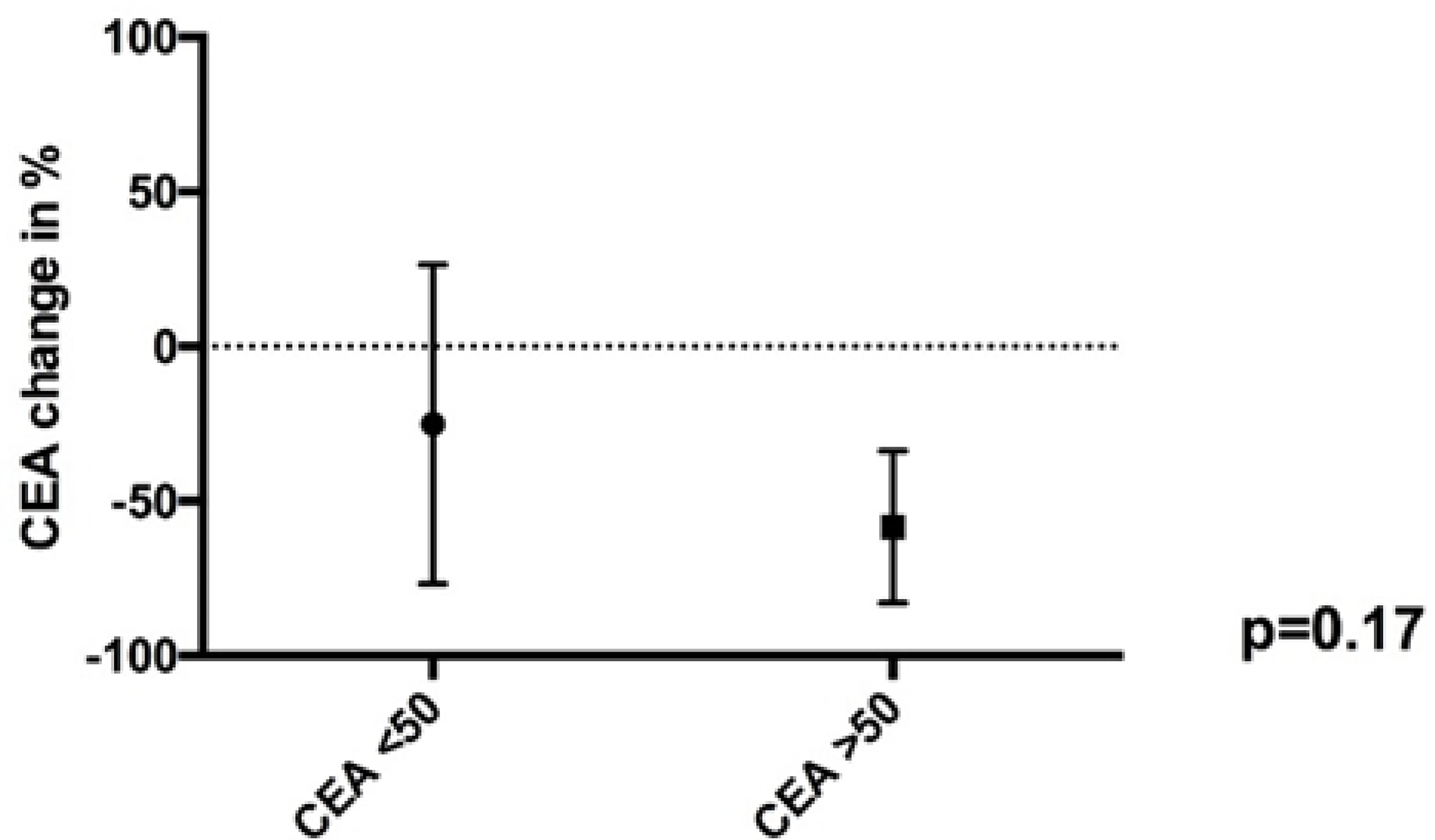
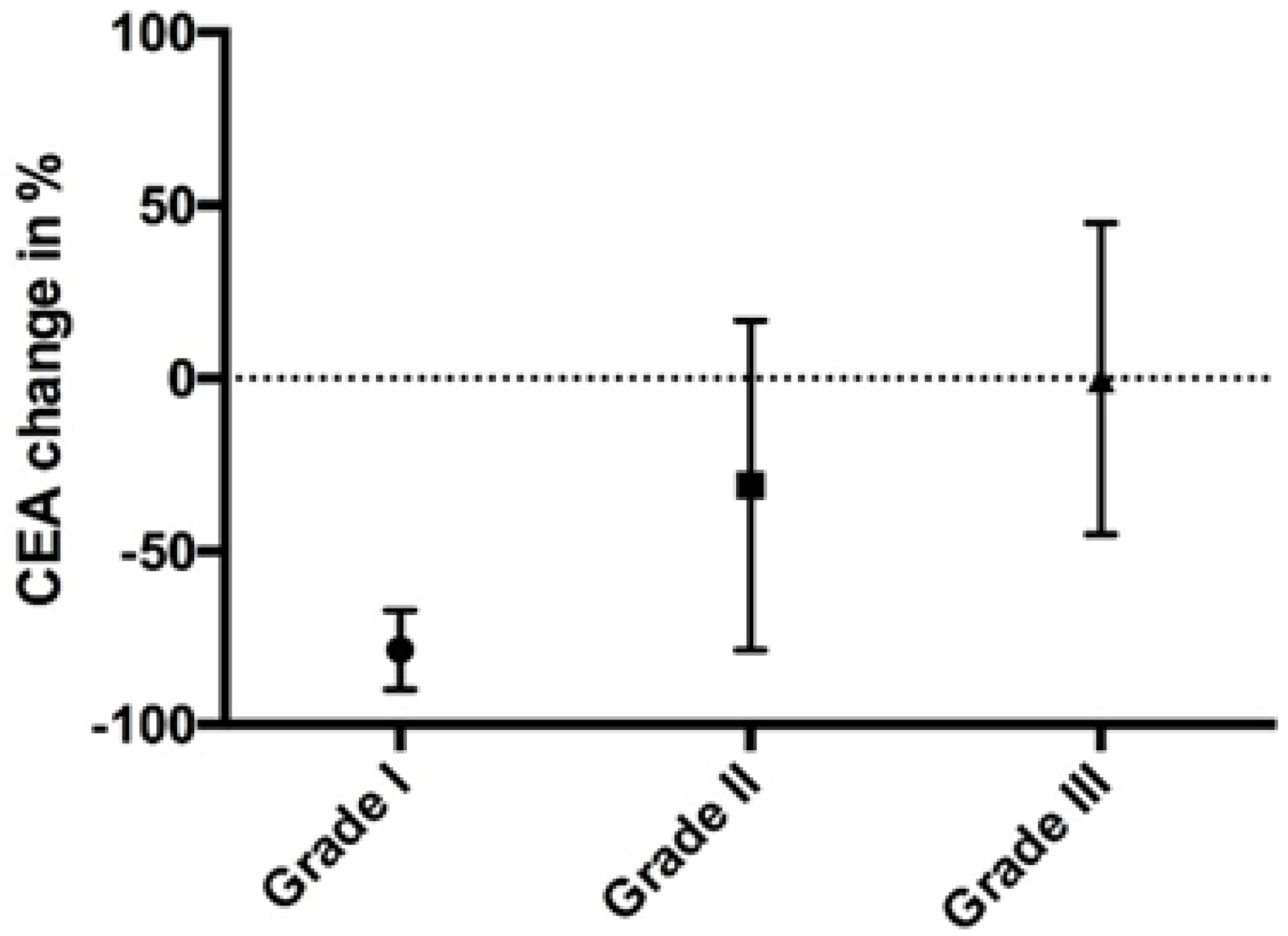
2. Results
3. Discussion
3.1. NIPS
3.2. Tumor Marker Correlation
3.3. Tumor Regression Grading
3.4. Study Limitations
4. Material and Methods
4.1. Neoadjuvant Laparoscopic HIPEC
4.2. Normothermic Intraperitoneal Chemotherapy
4.3. CRS and HIPEC
4.4. Response Evaluation
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Pai, R.K.; Longacre, T.A. Appendiceal mucinous tumors and pseudomyxoma peritonei: Histologic features, diagnostic problems and proposed classification. Adv. Anat. Pathol. 2005, 12, 291–311. [Google Scholar] [CrossRef]
- Young, R.H. Pseudomyxoma peritonei and selected other aspects of the spread of appendiceal neoplasms. Semin. Diagn. Pathol. 2004, 21, 134–150. [Google Scholar] [CrossRef] [PubMed]
- Miner, T.J.; Shia, J.; Jaques, D.P.; Klimstra, D.S.; Brennan, M.F.; Coit, D.G. Long-term survival following treatment of pseudomyxoma peritonei: An analysis of surgical therapy. Ann. Surg. 2005, 241, 300–308. [Google Scholar] [CrossRef]
- Gough, D.B.; Donohue, J.H.; Schutt, A.J.; Gonchoroff, N.; Goellner, J.R.; Wilson, T.O.; Naessens, J.M.; O’Brien, P.C.; van Heerden, J.A. Pseudomyxoma peritonei. Long- term patient survival with an aggressive regional approach. Ann. Surg. 1994, 219, 112–119. [Google Scholar] [CrossRef] [PubMed]
- Chua, T.C.; Moran, B.J.; Sugarbaker, P.H.; Levine, E.A.; Glehen, O.; Gilly, F.N.; Baratti, D.; Deraco, M.; Elias, D.; Sardi, A.; et al. Early- and long-term outcome data of patients with pseudomyxoma peritonei from appendiceal origin treated by a strategy of cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. J. Clin. Oncol. 2012, 30, 2449–2456. [Google Scholar] [CrossRef] [PubMed]
- Ansari, N.; Chandrakumaran, K.; Dayal, S.; Mohamed, F.; Cecil, T.D.; Moran, B.J. Cytoreductive surgery and hyperthermic intraperitoneal chemotherapy in 1000 patients with perforated appendiceal epithelial tumours. Eur. J. Surg. Oncol. 2016, 42, 1035–1041. [Google Scholar] [CrossRef]
- Youssef, H.; Newman, C.; Chandrakumaran, K.; Mohamed, F.; Cecil, T.D.; Moran, B.J. Operative findings, early complications, and long-term survival in 456 patients with pseudomyxoma peritonei syndrome of appendiceal origin. Dis. Colon. Rectum. 2011, 54, 293–299. [Google Scholar] [CrossRef]
- Austin, F.; Mavanur, A.; Sathaiah, M.; Steel, J.; Lenzner, D.; Ramalingam, L.; Holtzman, M.; Ahrendt, S.; Pingpank, J.; Zeh, H.J.; et al. Aggressive management of peritoneal carcinomatosis from mucinous appendiceal neoplasms. Ann. Surg. Oncol. 2012, 19, 1386–1393. [Google Scholar] [CrossRef]
- Yonemura, Y.; Bandou, E.; Sawa, T.; Yoshimitsu, Y.; Endou, Y.; Sasaki, T.; Sugarbaker, P.H. Neoadjuvant treatment of gastric cancer with peritoneal dissemination. Eur. J. Surg. Oncol. 2006, 32, 661–665. [Google Scholar] [CrossRef]
- Fujiwara, Y.; Takiguchi, S.; Nakajima, K.; Miyata, H.; Yamasaki, M.; Kurokawa, Y.; Okada, K.; Mori, M.; Doki, Y. Neoadjuvant Intraperitoneal and systemic chemotherapy for gastric patients with peritoneal dissemination. Ann. Surg. Oncol. 2011, 18, 3726–3731. [Google Scholar] [CrossRef]
- Muñoz-Casares, F.C.; Rufián, S.; Arjona-Sánchez, Á.; Rubio, M.J.; Díaz, R.; Casado, Á.; Naranjo, Á.; Díaz-Iglesias, C.J.; Ortega, R.; Muñoz-Villanueva, M.C.; et al. Neoadjuvant intraperitoneal chemotherapy with paclitaxel for the radical surgical treatment of peritoneal carcinomatosis in ovarian cancer: A prospective pilot study. Cancer Chemother. Pharmacol. 2011, 68, 267–274. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Los, G.; Mutsaers, P.H.; van der Vijgh, W.J.; Baldew, G.S.; de Graaf, P.W.; McVie, J.G. Direct diffusion of cis-damminedichloroplatinum (II) in intraperitoneal rat tumors after intraperitoneal chemotherapy: A comparison with systemic chemotherapy. Cancer Res. 1989, 49, 3380–3384. [Google Scholar] [PubMed]
- de Bree, E.; Tsiftsis, D.D. Experimental and pharmacological studies in intraperitoneal chemotherapy from laboratory bench to bedside. In Advances in Peritoneal Surface Oncology; Gonzalez-Moleno, S., Ed.; Springer: Berlin/Heidelberg, Germany; New York, NY, USA; Dordrecht, The Netherlands; London, UK, 2007; pp. 53–73. [Google Scholar]
- Yonemura, Y.; Elnemr, A.; Endou, Y.; Ishibashi, H.; Mizumoto, A.; Miura, M.; Li, Y. Effects of Neoadjuvant Intraperitoneal/Systemic Chemotherapy (Bidirectional Chemotherapy) for the treatment of patients with peritoneal metastasis from gastric cancer. Int. J. Surg. Oncol. 2012, 2012, 148420. [Google Scholar] [CrossRef] [PubMed]
- Ishigami, H.; Kitayama, J.; Kaisaki, S.; Hidemura, A.; Kato, M.; Otani, K.; Kamei, T.; Soma, D.; Miyato, H.; Yamashita, H.; et al. Phase II study of weekly intravenous and intraperitoneal paclitaxel combined with S1 for advanced gastric cancer with peritoneal metastasis. Ann. Oncol. 2010, 21, 67–70. [Google Scholar] [CrossRef]
- Yonemura, Y.; Endou, Y.; Shinbo, M.; Sasaki, T.; Hirano, M.; Mizumoto, A.; Matsuda, T.; Takao, N.; Ichinose, M.; Mizuno, M.; et al. Safety and efficacy of bidirectional chemotherapy for treatment of patients with peritoneal dissemination for gastric cancer. Selection for cytoreductive surgery. J. Surg. Oncol. 2009, 15, 311–316. [Google Scholar] [CrossRef]
- Farquharson, A.L.; Pranesh, N.; Witham, G.; Swindell, R.; Taylor, M.B.; Renehan, A.G.; Rout, S.; Wilson, M.S.; O’Dwyer, S.T.; Saunders, M.P. A phase II study evaluating the use of concurrent mitomycin C and capecitabine in patients with advanced unresectable pseudomyxoma peritonei. Br. J. Cancer 2008, 99, 591–596. [Google Scholar] [CrossRef]
- Shapiro, J.F.; Chase, J.L.; Wolff, R.A.; Lambert, L.A.; Mansfield, P.F.; Overman, M.J.; Ohinata, A.; Liu, J.; Wang, X.; Eng, C. Modern systemic chemotherapy in surgically unresectable neoplasms of appendiceal origin: A single-institution experience. Cancer 2010, 116, 316–322. [Google Scholar] [CrossRef]
- Blackham, A.U.; Swett, K.; Eng, C.; Sirintrapun, J.; Bergman, S.; Geisinger, K.R.; Votanopoulos, K.; Stewart, J.H.; Shen, P.; Levine, E.A. Perioperative systemic chemotherapy for appendiceal mucinous carcinoma peritonei treated with cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. J. Surg. Oncol. 2014, 109, 740–745. [Google Scholar] [CrossRef]
- Van Ruth, S.; Hart, A.A.M.; Bonfrer, J.M.G.; Verwaal, V.J.; Zoetmulder, F.A.N. Prognostic value of baseline and serial carcinoembryonic antigen and carbohydrate antigen 19.9 measurements in patients with pseudomyxoma peritonei treated with cytoreduction and hyperthermic intraperitoneal chemotherapy. Ann. Surg. Oncol. 2002, 9, 961–967. [Google Scholar] [CrossRef]
- Baratti, D.; Kusamura, S.; Martinetti, A.; Seregni, E.; Laterza, B.; Oliva, D.G.; Deraco, M. Prognostic value of circulating tumor markers in patients with pseudomyxoma peritonei treated with cytoreductive surgery and hyperthermic intraperitoneal chemotherapy. Ann. Surg. Oncol. 2007, 14, 2300–2308. [Google Scholar] [CrossRef]
- Yamao, T.; Kai, S.; Kazami, A.; Koizumi, K.; Handa, T.; Takemoto, N.; Maruyama, M. Tumor markers CEA, CA19-9 and CA125 in monitoring of response to systemic chemotherapy in patients with advanced gastric cancer. Jpn. J. Clin. Oncol. 1999, 29, 550–555. [Google Scholar] [CrossRef] [PubMed]
- Lee, D.W.; Im, S.A.; Kim, Y.J.; Yang, Y.; Rhee, J.; Na, I.I.; Lee, K.H.; Kim, T.Y.; Han, S.W.; Choi, I.S.; et al. CA19-9 or CEA Decline after the First Cycle of Treatment Predicts Survival in Advanced Biliary Tract Cancer Patients Treated with S-1 and Cisplatin Chemotherapy. Cancer Res. Treat. 2017, 49, 807–815. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.N.; Xiao, W.W.; Xi, S.Y.; OuYang, P.Y.; You, K.Y.; Zeng, Z.F.; Ding, P.R.; Zhang, H.Z.; Pan, Z.Z.; Xu, R.H.; et al. Pathological Assessment of the AJCC Tumor Regression Grading System After Preoperative Chemoradiotherapy for Chinese Locally Advanced Rectal Cancer. Medicine 2016, 95, e2272. [Google Scholar] [CrossRef]
- Langer, R.; Ott, K.; Feith, M.; Lordick, F.; Siewert, J.R.; Becker, K. Prognostic significance of histopathological tumor regression after neoadjuvant chemotherapy in esophageal adenocarcinomas. Mod. Pathol. 2009, 22, 1555–1563. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Li, L.; Wang, X.; Wang, C.; Wang, D. Tumor regression grading after neoadjuvant chemotherapy predicts long-term outcome of stage IIIC epithelial ovarian cancer. J. Clin. Oncol. 2018, 36, 5547. [Google Scholar] [CrossRef]
- Rödel, C.; Martus, P.; Papadoupolos, T.; Füzesi, L.; Klimpfinger, M.; Fietkau, R.; Liersch, T.; Hohenberger, W.; Raab, R.; Sauer, R.; et al. Prognostic Significance of Tumor Regression After Preoperative Chemoradiotherapy for Rectal Cancer. J. Clin. Oncol. 2005, 23, 8688–8696. [Google Scholar] [CrossRef]
- D’Ugo, D.; Persiani, R.; Rausei, S.; Biondi, A.; Vigorita, V.; Boccia, S.; Ricci, R. Response to neoadjuvant chemotherapy and effects of tumor regression in gastric cancer. Eur. J. Surg. Oncol. 2006, 32, 1105–1109. [Google Scholar] [CrossRef]
- Dineen, S.P.; Royal, R.E.; Hughes, M.S.; Sagebiel, T.; Bhosale, P.; Overman, M.; Matamoros, A.; Mansfield, P.F.; Fournier, K.F. A Simplified Preoperative Assessment Predicts Complete Cytoreduction and Outcomes in Patients with Low-Grade Mucinous Adenocarcinoma of the Appendix. Ann. Surg. Oncol. 2015, 22, 3640–3646. [Google Scholar] [CrossRef]
- Yonemura, Y.; Canbay, E.; Sako, S.; Ishibashi, H.; Hirano, M.; Mizumoto, A.; Takeshita, K.; Takao, N.; Ichinose, M.; Lee, C.; et al. Effects of Laparoscopic Hyperthermic Intraperitoneal Chemotherapy for Peritoneal Metastasis from Gastric Cancer. Cancer Clin. Oncol. 2014, 3, 43–50. [Google Scholar]
- Jacquet, P.; Sugarbaker, P.H. Clinical research methodologies in diagnosis and staging of patients with peritoneal carcinomatosis. Cancer Treat. Res. 1996, 82, 359–374. [Google Scholar]
- Masuda, T.; Kuramoto, M.; Shimada, S.; Ikeshima, S.; Yamamoto, K.; Nakamura, K.; Yoshimatsu, S.; Urata, M.; Baba, H. The effects of extensive intraoperative peritoneal lavage (EIPL) in stage IIIB+C and cytology positive gastric cancer patients. Int. J. Clin. Oncol. 2016, 21, 289–294. [Google Scholar] [CrossRef] [PubMed]
- Dindo, D.; Demartines, N.; Clavien, P.-A. Classification of surgical complications: A new proposal with evaluation in a cohort of 6336 patients and results of a survey. Ann. Surg. 2004, 240, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Dworak, O.; Keilholz, L.; Hoffmann, A. Pathological features of rectal cancer after preoperative radio-chemotherapy. Int. J. Colorectal Dis. 1997, 12, 19–23. [Google Scholar] [CrossRef] [PubMed]


| Patient Related | |
|---|---|
| Sex (%) | |
| Male | 37 (10/27) |
| Female | 63 (17/27) |
| Mean Age [years] | 54.6 (SD 12.7) |
| Mean PCI | 22.8 (SD 13.6) |
| Mean SB PCI | 5 (SD 4.7) |
| Tumor related | |
| Primary Tumor location (%) | |
| Appendix | 85.2 (23/27) |
| Urachus | 7.4 (2/27) |
| Ovary | 7.4 (2/27) |
| Appearance of PMP (%) | |
| Synchronous | 74.1 (20/27) |
| Metachronous | 25.9 (7/27) |
| Histological type of PMP at CRS (%) | |
| Acellular | 18.5 (5/27) |
| Low grade PMP | 44.4 (12/27) |
| High grade PMP | 14.8 (4/27) |
| High grade PMP with signet ring cells | 22.2 (6/27) |
| Surgery related (n = 22) | |
| Mean surgery time (min) | 314.4 (SD 122.7) |
| Mean blood loss (ml) | 2055.8 (SD 1312.7) |
| Mean no. of BT units | 7.8 (SD 5.8) |
| CCR status | |
| CC0 | 40.9 (9/22) |
| CC1 | 13.6 (3/22) |
| CC2 | 9.1 (2/22) |
| CC3 | 36.4 (8/22) |
| Clavien–Dindo class (%) | |
| Grade 0 | 68.2 (15/22) |
| Grade 1–2 | 0 (0/22) |
| Grade 3a | 13.6 (3/22) |
| Grade 3b | 9.1 (2/22) |
| Grade 4 | 4.5 (1/22) |
| Grade 5 | 4.5 (1/22) |
| Surgical complications (%) | 31.8 (7/22) |
| Anastomotic leak | 13.6 (3/22) |
| Intra-abdominal abscess | 4.5 (1/22) |
| Pancreatic fistula | 4.5 (1/22) |
| Small bowel perforation | 9.1 (2/22) |
| Medical complications (%) | 13.6 (3/22) |
| Pulmonary | 4.5 (1/22) |
| Deep venous thrombosis | 4.5 (1/22) |
| Sepsis | 4.5 (1/22) |
| Treatment related | |
| Tumor regression grade (%) | |
| Grade III | 18.2 (4/22) |
| Grade II | 72.8 (16/22) |
| Grade I | 9.1 (2/22) |
| Grade 0 | 0 (0/22) |
| Histopathologic Subtype | Tumor Regression (Grade II and III) | No Tumor Regression (Grade 0 and I) |
|---|---|---|
| Acellular mucin | 4 | 0 |
| Low-grade | 1 | 7 |
| High-grade | 0 | 2 |
| High-grade with signet cells | 2 | 4 |
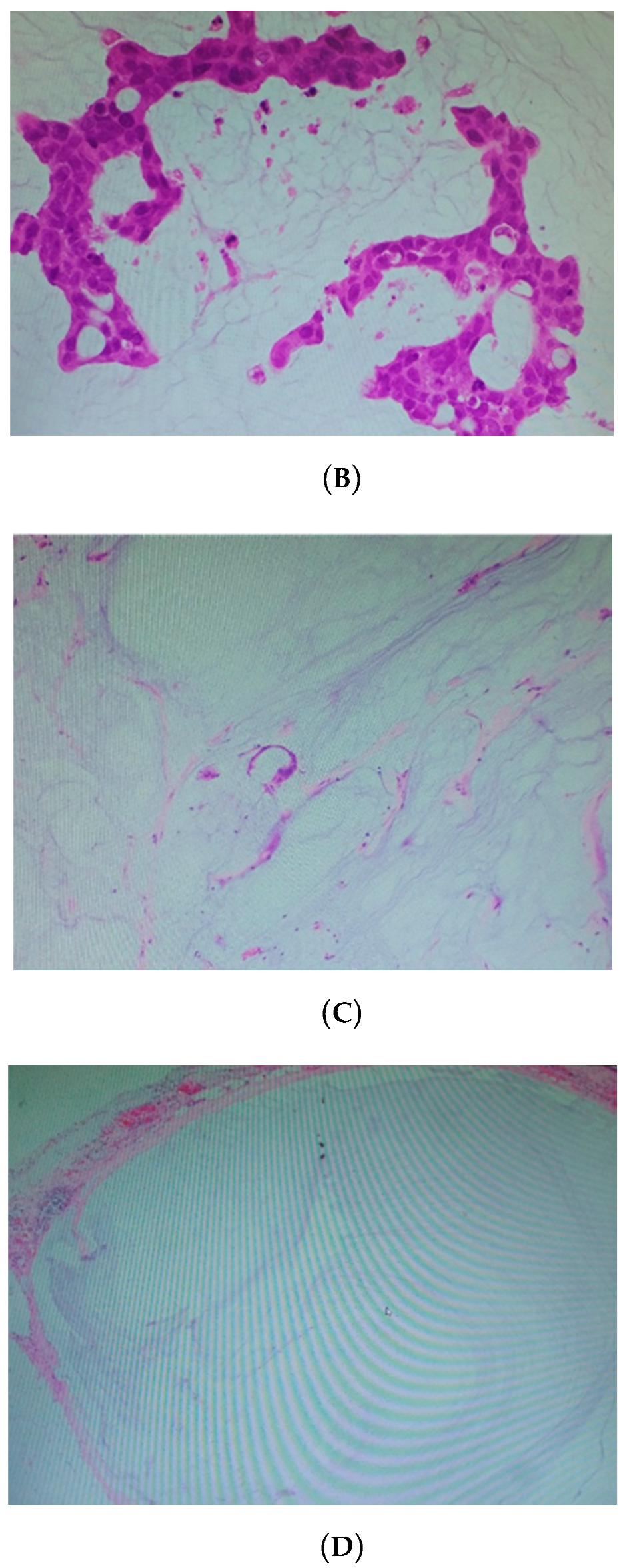
| Tumor Regression Grade | Pathological Finding (Figure 1a–d) |
|---|---|
| Grade 0 | No regression |
| Grade I | Predominantly tumors, along with mucinous material |
| Grade II | Predominantly mucinous material, with few scattered tumor cells |
| Grade III | No viable tumor cells detectable (only acellular mucin) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Prabhu, A.; Brandl, A.; Wakama, S.; Sako, S.; Ishibashi, H.; Mizumoto, A.; Takao, N.; Noguchi, K.; Motoi, S.; Ichinose, M.; et al. Neoadjuvant Intraperitoneal Chemotherapy in Patients with Pseudomyxoma Peritonei—A Novel Treatment Approach. Cancers 2020, 12, 2212. https://doi.org/10.3390/cancers12082212
Prabhu A, Brandl A, Wakama S, Sako S, Ishibashi H, Mizumoto A, Takao N, Noguchi K, Motoi S, Ichinose M, et al. Neoadjuvant Intraperitoneal Chemotherapy in Patients with Pseudomyxoma Peritonei—A Novel Treatment Approach. Cancers. 2020; 12(8):2212. https://doi.org/10.3390/cancers12082212
Chicago/Turabian StylePrabhu, Aruna, Andreas Brandl, Satoshi Wakama, Shouzou Sako, Haruaki Ishibashi, Akiyoshi Mizumoto, Nobuyuki Takao, Kousuke Noguchi, Shunsuke Motoi, Masumi Ichinose, and et al. 2020. "Neoadjuvant Intraperitoneal Chemotherapy in Patients with Pseudomyxoma Peritonei—A Novel Treatment Approach" Cancers 12, no. 8: 2212. https://doi.org/10.3390/cancers12082212
APA StylePrabhu, A., Brandl, A., Wakama, S., Sako, S., Ishibashi, H., Mizumoto, A., Takao, N., Noguchi, K., Motoi, S., Ichinose, M., Liu, Y., & Yonemura, Y. (2020). Neoadjuvant Intraperitoneal Chemotherapy in Patients with Pseudomyxoma Peritonei—A Novel Treatment Approach. Cancers, 12(8), 2212. https://doi.org/10.3390/cancers12082212





