The Perfect Combination: Enhancing Patient Response to PD-1-Based Therapies in Epithelial Ovarian Cancer
Abstract
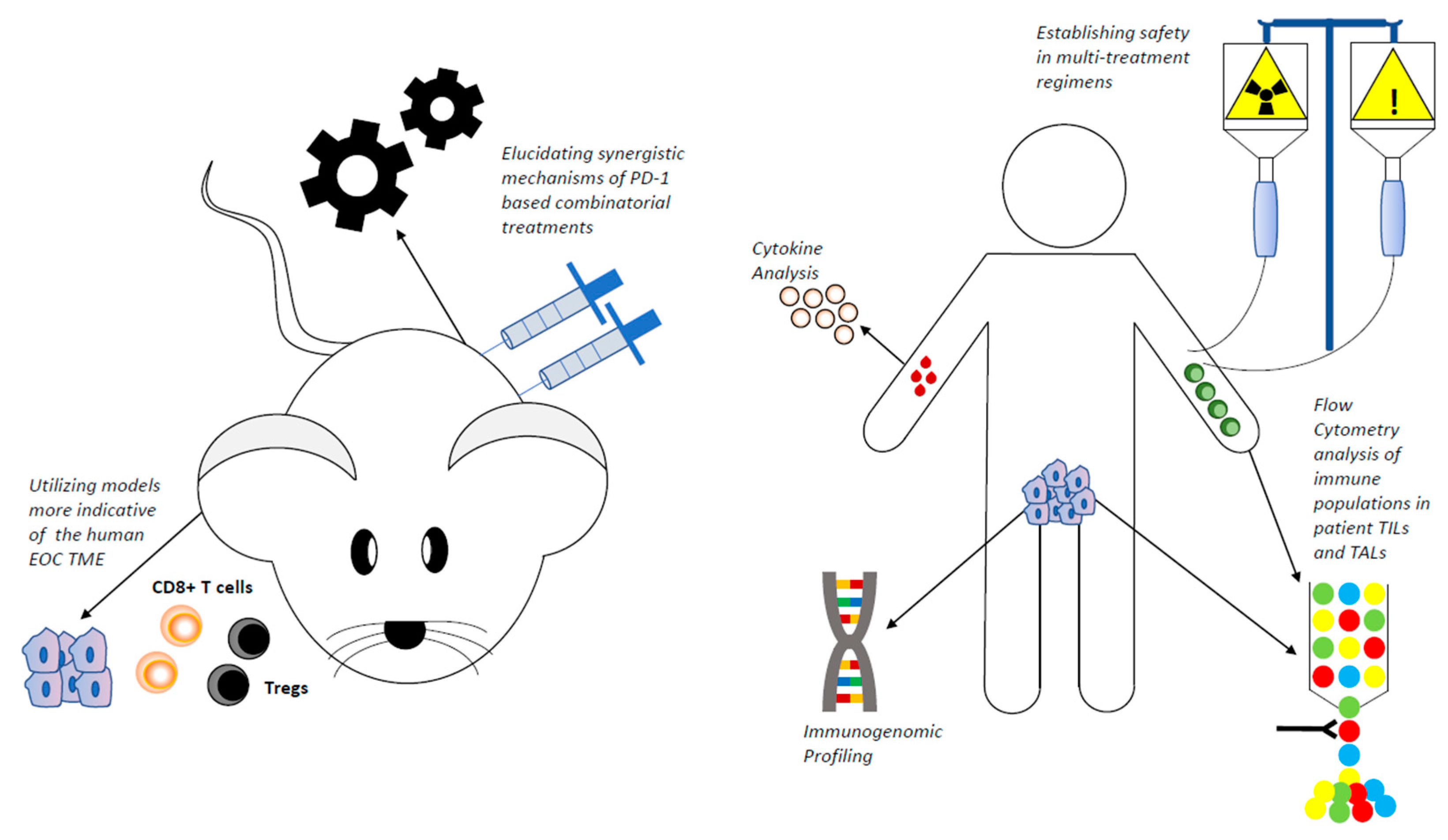
1. Introduction
2. Challenges to PD-1 Therapy
2.1. Unique Tumor Microenvironment
2.2. Elevated Levels of Highly Activated T Regulatory Cells
2.3. Patient Selection Barriers
3. PD-1 Based Combinatorial Approaches
3.1. PD-1 and Chemotherapy
3.1.1. Preclinical Studies
3.1.2. Clinical Studies
3.2. PD-1 and Anti-Angiogenic Therapy
3.2.1. Preclinical Studies
3.2.2. Clinical Studies
3.3. PD-1 and PARP Therapy
3.3.1. Preclinical Studies
3.3.2. Clinical Studies
3.4. PD-1 and Additional Immune Receptor Targeting
3.4.1. Preclinical Studies
3.4.2. Clinical Studies
3.5. PD-1 and Novel Immunotherapies
3.5.1. Preclinical Studies
3.5.2. Clinical Studies
3.6. PD-1 and Other Agents
3.6.1. Preclinical Studies
3.6.2. Clinical Studies
4. Discussion
5. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- American Cancer Society. Cancer Statistics Center—Ovary. Available online: https://cancerstatisticscenter.cancer.org/#!/cancer-site/Ovary (accessed on 9 April 2020).
- Pignata, S.; Cecere, S.C.; Du Bois, A.; Harter, P.; Heitz, F. Treatment of recurrent ovarian cancer. Ann. Oncol. 2017, 28 (Suppl. 8), viii51–viii56. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Lang, J. Programmed death-1 pathway blockade produces a synergistic antitumor effect: Combined application in ovarian cancer. J. Gynecol. Oncol. 2017, 28, 1093851. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, E.M.; Refae, A.A.; Bayer, A.M.; Sagr, E.R. Poly(ADP-ribose) polymerase inhibitors as maintenance treatment in patients with newly diagnosed advanced ovarian cancer: A meta-analysis. Future Oncol. 2020, 16, 585–596. [Google Scholar] [CrossRef]
- Kerliu, L.; Myruski, S.; Bhatti, A.; Soni, P.; Petrosius, P.; Pervanas, H.C.; Horton, E.R. Niraparib for the treatment of recurrent epithelial ovarian, fallopian tube, or primary peritoneal cancer [published online ahead of print, 2020 Mar 16]. Ann. Pharmacother. 2020, 1060028020912749. [Google Scholar]
- Rossi, L.; Verrico, M.; Zaccarelli, E.; Papa, A.; Colonna, M.; Strudel, M.; Vici, P.; Bianco, V.; Tomao, F. Bevacizumab in ovarian cancer: A critical review of phase III studies. Oncotarget 2017, 8, 12389–12405. [Google Scholar] [CrossRef] [PubMed]
- Cojocaru, E.; Parkinson, C.A.; Brenton, J.D. Personalising Treatment for High-Grade Serous Ovarian Carcinoma. Clin. Oncol. 2018, 30, 515–524. [Google Scholar] [CrossRef]
- Kamphorst, A.O.; Wieland, A.; Nasti, T.H.; Yang, S.; Zhang, R.; Barber, D.L.; Konieczny, B.T.; Daugherty, C.Z.; Koenig, L.; Yu, K.; et al. Rescue of exhausted CD8 T cells by PD-1-targeted therapies is CD28-dependent. Science 2017, 355, 1423–1427. [Google Scholar] [CrossRef]
- Hui, E.; Cheung, J.; Zhu, J.; Su, X.; Taylor, M.J.; Wallweber, H.A.; Sasmal, D.K.; Huang, J.; Kim, J.M.; Mellman, I.; et al. T cell costimulatory receptor CD28 is a primary target for PD-1-mediated inhibition. Science 2017, 355, 1428–1433. [Google Scholar] [CrossRef]
- Nowicki, T.S.; Hu-Lieskovan, S. Mechanisms of Resistance to PD-1 and PD-L1 Blockade. Cancer J. 2018, 24, 47. [Google Scholar] [CrossRef]
- Mayoux, M.; Roller, A.; Pulko, V.; Sammicheli, S.; Chen, S.; Sum, E.; Jost, C.; Fransen, M.F.; Buser, R.B.; Kowanetz, M.; et al. Dendritic cells dictate responses to PD-L1 blockade cancer immunotherapy. Sci. Transl. Med. 2020, 12, eaav7431. [Google Scholar] [CrossRef]
- Raedler, L.A. Keytruda (Pembrolizumab): First PD-1 Inhibitor Approved for Previously Treated Unresectable or Metastatic Melanoma. Am. Health Drug Benefits 2015, 8, 96–100. [Google Scholar]
- Raedler, L.A. Opdivo (Nivolumab): Second PD-1 Inhibitor Receives FDA Approval for Unresectable or Metastatic Melanoma. Am. Health Drug Benefits 2015, 8, 180–183. [Google Scholar] [PubMed]
- Sui, H.; Ma, N.; Wang, Y.; Li, H.; Liu, X.; Su, Y.; Yang, J. Anti-PD-1/PD-L1 Therapy for Non-Small-Cell Lung Cancer: Toward Personalized Medicine and Combination Strategies. J. Immunol. Res. 2018, 2018, 6984948. [Google Scholar] [CrossRef] [PubMed]
- Zolkind, P.; Uppaluri, R. Checkpoint immunotherapy in head and neck cancers. Cancer Metastasis Rev. 2017, 36, 475–489. [Google Scholar] [CrossRef] [PubMed]
- Botta, G.P.; Granowicz, E.; Costantini, C. Advances on immunotherapy in genitourinary and renal cell carcinoma. Transl. Cancer Res. 2017, 6, 17–29. [Google Scholar] [CrossRef] [PubMed]
- Hamanishi, J.; Mandai, M.; Ikeda, T.; Minami, M.; Kawaguchi, A.; Murayama, T.; Kanai, M.; Mori, Y.; Matsumoto, S.; Chikuma, S. Safety and Antitumor Activity of Anti-PD-1 Antibody, Nivolumab, in Patients with Platinum-Resistant Ovarian Cancer. J. Clin. Oncol. 2015, 33, 4015–4022. [Google Scholar] [CrossRef] [PubMed]
- Varga, A.; Piha-Paul, S.A.; Ott, P.A.; Mehnert, J.M.; Berton-Rigaud, D.; Johnson, E.A.; Cheng, J.D.; Yuan, S.; Rubin, E.H.; Matei, D.E. Antitumor activity and safety of pembrolizumab in patients (pts) with PD-L1 positive advanced ovarian cancer: Interim results from a phase Ib study. J. Clin. Oncol. 2015, 33 (Suppl. 15), 5510. [Google Scholar] [CrossRef]
- Disis, M.L.; Patel, M.R.; Pant, S.; Hamilton, E.P.; Lockhart, A.C. Avelumab (MSB0010718C; anti-PD-L1) in Patients with Recurrent/Refractory Ovarian Cancer from the JAVELIN Solid Tumor Phase Ib Trial: Safety and Clinical Activity. J. Clin. Oncol 2016, 33 (Suppl. 15), 5533. [Google Scholar] [CrossRef]
- Infante, J.R.; Braiteh, F.; Emens, L.A.; Balmanoukian, A.S.; Oaknin, A.; Wang, Y.; Liu, B.; Molinero, L.; Fasso, M.; O’Hear, C.; et al. Safety, clinical activity and biomarkers of atezolizumab (atezo) in advanced ovarian cancer (OC). Ann. Oncol. 2016, 27, vi300. [Google Scholar] [CrossRef]
- Zhang, L.; Chen, Y.; Li, F.; Bao, L.; Liu, W. Atezolizumab and Bevacizumab Attenuate Cisplatin Resistant Ovarian Cancer Cells Progression Synergistically via Suppressing Epithelial-Mesenchymal Transition. Front. Immunol. 2019, 10, 867. [Google Scholar] [CrossRef]
- Rodriguez, G.M.; Galpin, K.J.C.; McCloskey, C.W.; Vanderhyden, B.C. The tumor microenvironment of epithelial ovarian cancer and its influence on response to immunotherapy. Cancers 2018, 10, 242. [Google Scholar] [CrossRef] [PubMed]
- Greppi, M.; Tabellini, G.; Patrizi, O.; Candiani, S.; Decensi, A.; Parolini, S.; Sivori, S.; Pesce, S.; Paleari, L.; Marcenaro, E. Strengthening the antitumor NK cell function for the treatment of ovarian cancer. Int. J. Mol. Sci. 2019, 20, 890. [Google Scholar] [CrossRef] [PubMed]
- Pesce, S.; Greppi, M.; Tabellini, G.; Rampinelli, F.; Parolini, S.; Olive, D.; Moretta, L.; Moretta, A.; Marcenaro, E. Identification of a subset of human natural killer cells expressing high levels of programmed death 1: A phenotypic and functional characterization. J. Allergy Clin. Immunol. 2016, 139, 335–346.e3. [Google Scholar] [CrossRef] [PubMed]
- Meza-Perez, S.; Randall, T.D. Immunological functions of the omentum. Trends Immunol. 2017, 38, 526–536. [Google Scholar] [CrossRef]
- Nieman, K.M.; Kenny, H.A.; Penicka, C.V.; Ladanyi, A.; Buell-Gutbrod, R.; Zillhardt, M.R.; Romero, I.R.; Carey, M.S.; Mills, G.B.; Hotamisligil, G.S.; et al. Adipocytes promote ovarian cancer metastasis and provide energy for rapid tumor growth. Nat. Med. 2011, 17, 1498–1503. [Google Scholar] [CrossRef]
- Liu, Y.; Metzinger, M.N.; Lewellen, K.A.; Cripps, S.N.; Carey, K.D.; Harper, E.I.; Shi, Z.; Tarwater, L.; Grisoli, A.; Lee, E.; et al. Obesity Contributes to Ovarian Cancer Metastatic Success through Increased Lipogenesis, Enhanced Vascularity, and Decreased Infiltration of M1 Macrophages. Cancer Res. 2015, 75, 5046–5057. [Google Scholar] [CrossRef]
- Deying, W.; Feng, G.; Shumei, L.; Hui, Z.; Ming, L.; Hongqing, W. CAF-derived HGF promotes cell proliferation and drug resistance by up-regulating the c-Met/PI3K/Akt and GRP78 signalling in ovarian cancer cells. Biosci. Rep. 2017, 37, BSR20160470. [Google Scholar] [CrossRef]
- Givel, A.-M.; Kieffer, Y.; Scholer-Dahirel, A.; Sirven, P.; Cardon, M.; Pelon, F.; Magagna, I.; Gentric, G.; Costa, A.; Bonneau, C.; et al. miR200-regulated CXCL12β promotes fibroblast heterogeneity and immunosuppression in ovarian cancers. Nat. Commun. 2018, 9, 1056. [Google Scholar] [CrossRef]
- Wei, R.; Lv, M.; Li, F.; Cheng, T.; Zhang, Z.; Jiang, G.; Zhou, Y.; Gao, R.; Wei, X.; Lou, J.; et al. Human CAFs promote lymphangiogenesis in ovarian cancer via the Hh-VEGF-C signaling axis. Oncotarget 2017, 8, 67315–67328. [Google Scholar] [CrossRef]
- McCloskey, C.W.; Cook, D.P.; Kelly, B.S.; Azzi, F.; Allen, C.H.; Forsyth-Greig, A.; Upham, J.; Rayner, K.J.; Gray, D.A.; Boyd, R.W.; et al. Metformin Abrogates Age-Associated Ovarian Fibrosis. Clin. Cancer Res. 2020, 26, 632–642. [Google Scholar] [CrossRef]
- Briley, S.M.; Jasti, S.; McCracken, J.M.; Hornick, J.E.; Fegley, B.; Pritchard, M.T.; Duncan, F.E. Reproductive age-associated fibrosis in the stroma of the mammalian ovary. Reproduction 2016, 152, 245–260. [Google Scholar] [CrossRef] [PubMed]
- Miyazawa, A.; Ito, S.; Asano, S.; Tanaka, I.; Sato, M.; Kondo, M.; Hasegawa, Y. Regulation of PD-L1 expression by matrix stiffness in lung cancer cells. Biochem. Biophys. Res. Commun. 2018, 495, 2344–2349. [Google Scholar] [CrossRef] [PubMed]
- Pearce, O.M.T.; Delaine-Smith, R.M.; Maniati, E.; Nichols, S.; Wang, J.; Böhm, S.; Rajeeve, V.; Ullah, D.; Chakravarty, P.; Jones, R.R.; et al. Deconstruction of a metastatic tumor microenvironment reveals a common matrix response in human cancers. Cancer Discov. 2018, 8, 304–319. [Google Scholar] [CrossRef] [PubMed]
- Salmon, H.; Franciszkiewicz, K.; Damotte, D.; Dieu-Nosjean, M.-C.; Validire, P.; Trautmann, A.; Mami-Chouaib, F.; Donnadieu, E. Matrix architecture defines the preferential localization and migration of T cells into the stroma of human lung tumors. J. Clin. Investig. 2012, 122, 899–910. [Google Scholar] [CrossRef] [PubMed]
- Mariathasan, S.; Turley, S.J.; Nickles, D.; Castiglioni, A.; Yuen, K.; Wang, Y.; Kadel III, E.E.; Koeppen, H.; Astarita, J.L.; Cubas, R.; et al. TGFβ attenuates tumour response to PD-L1 blockade by contributing to exclusion of T cells. Nature 2018, 554, 544–548. [Google Scholar] [CrossRef] [PubMed]
- Jensen, C.; Madsen, D.H.; Hansen, M.; Schmidt, H.; Svane, I.M.; Karsdal, M.A.; Willumsen, N. Non-invasive biomarkers derived from the extracellular matrix associate with response to immune checkpoint blockade (anti-CTLA-4) in metastatic melanoma patients. J. Immunother. Cancer 2018, 6, 152. [Google Scholar] [CrossRef]
- Zhang, L.; Conejo-Garcia, J.R.; Katsaros, D.; Gimotty, P.A.; Massobrio, M.; Regnani, G.; Makrigiannakis, A.; Gray, H.; Schlienger, K.; Liebman, M.N.; et al. Intratumoral T cells, recurrence, and survival in epithelial ovarian cancer. N. Engl. J. Med. 2003, 348, 203–213. [Google Scholar] [CrossRef]
- Webb, J.R.; Milne, K.; Nelson, B.H. Location, location, location: CD103 demarcates intraepithelial, prognostically favorable CD8(+) tumor-infiltrating lymphocytes in ovarian cancer. Oncoimmunology 2014, 3, e27668. [Google Scholar] [CrossRef]
- Webb, J.R.; Milne, K.; Watson, P.; Deleeuw, R.J.; Nelson, B.H. Tumor-infiltrating lymphocytes expressing the tissue resident memory marker CD103 are associated with increased survival in high-grade serous ovarian cancer. Clin. Cancer Res. 2014, 20, 434–444. [Google Scholar] [CrossRef]
- Bu, M.; Shen, Y.; Seeger, W.L.; An, S.; Qi, R.; Sanderson, J.A.; Cai, Y. Ovarian carcinoma-infiltrating regulatory T cells were more potent suppressors of CD8(+) T cell inflammation than their peripheral counterparts, a function dependent on TIM3 expression. Tumour Biol. 2016, 37, 3949–3956. [Google Scholar] [CrossRef]
- Landskron, J.; Helland, Ø.; Torgersen, K.M.; Aandahl, E.M.; Gjertsen, B.T.; Bjørge, L.; Tasḱen, K. Activated regulatory and memory T-cells accumulate in malignant ascites from ovarian carcinoma patients. Cancer Immunol. Immunother. 2015, 64, 337–347. [Google Scholar] [CrossRef] [PubMed]
- Landskron, J.; Taskén, K. CD147 in regulatory T cells. Cell. Immunol. 2013, 282, 17–20. [Google Scholar] [CrossRef] [PubMed]
- Xiong, L.; Edwards, C.K.; Zhou, L. The biological function and clinical utilization of CD147 in human diseases: A review of the current scientific literature. Int. J. Mol. Sci. 2014, 15, 17411–17441. [Google Scholar] [CrossRef] [PubMed]
- Toker, A.; Nguyen, L.T.; Stone, S.C.; Yang, S.Y.C.; Katz, S.R.; Shaw, P.A.; Clarke, B.A.; Ghazarian, D.; Al-Habeeb, A.; Easson, A.; et al. Regulatory T Cells in Ovarian Cancer Are Characterized by a Highly Activated Phenotype Distinct from that in Melanoma. Clin. Cancer Res. 2018, 24, 5685–5696. [Google Scholar] [CrossRef] [PubMed]
- Gaillard, S.L.; Coleman, R.L. Identifying markers of immune response in ovarian cancer: Does PD-L1 expression meet the mark? Ann. Oncol. 2019, 30, 1025–1028. [Google Scholar] [CrossRef] [PubMed]
- Chin, C.D.; Fares, C.M.; Konecny, G.E.; Rao, J. Biomarkers that may predict response to immunotherapy in ovarian malignancies. Curr. Opin. Obstet. Gynecol. 2020, 32, 84–90. [Google Scholar] [CrossRef]
- Ghisoni, E.; Imbimbo, M.; Zimmermann, S.; Valabrega, G. Ovarian cancer immunotherapy: Turning up the heat. Int. J. Mol. Sci. 2019, 20, 2927. [Google Scholar] [CrossRef]
- Disis, M.L.; Taylor, M.H.; Kelly, K.; Beck, J.T.; Gordon, M.; Moore, K.M.; Patel, M.R.; Chaves, J.; Park, H.; Mita, A.C.; et al. Efficacy and safety of avelumab for patients with recurrent or refractory ovarian cancer: Phase 1b results from the JAVELIN solid tumor trial. JAMA Oncol. 2019, 5, 393–401. [Google Scholar] [CrossRef]
- Zhu, X.; Xu, J.; Cai, H.; Lang, J. Carboplatin and programmed death-ligand 1 blockade synergistically produce a similar antitumor effect to carboplatin alone in murine ID8 ovarian cancer model. J. Obstet. Gynaecol. Res. 2018, 44, 303–311. [Google Scholar] [CrossRef]
- Darb-Esfahani, S.; Kunze, C.A.; Kulbe, H.; Sehouli, J.; Wienert, S.; Lindner, J.; Budczies, J.; Bockmayr, M.; Dietal, M.; Denkert, C.; et al. Prognostic impact of programmed cell death-1 (PD-1) and PD-ligand 1 (PD-L1) expression in cancer cells and tumor-infiltrating lymphocytes in ovarian high grade serous carcinoma. Oncotarget 2016, 7, 1486–1499. [Google Scholar] [CrossRef]
- Webb, J.R.; Milne, K.; Kroeger, D.R.; Nelson, B.H. PD-L1 expression is associated with tumor-infiltrating T cells and favorable prognosis in high-grade serous ovarian cancer. Gynecol. Oncol. 2016, 141, 293–302. [Google Scholar] [CrossRef] [PubMed]
- Martin de la Fuente, L.; Westbom-Fremer, S.; Arildsen, N.S.; Hartman, L.; Malander, S.; Kannisto, P.; Måsbäck, A.; Hedenfalk, I. PD-1/PD-L1 expression and tumor-infiltrating lymphocytes are prognostically favorable in advanced high-grade serous ovarian carcinoma. Virchows Arch. 2020, 477, 83–91. [Google Scholar] [CrossRef] [PubMed]
- Buderath, P.; Mairinger, F.; Mairinger, E.; Böhm, K.; Mach, P.; Schmid, K.W.; Kimming, R.; Kasimir-Bauer, S.; Bankfalvi, A.; Westerwick, D.; et al. Prognostic significance of PD-1 and PD-L1 positive tumor-infiltrating immune cells in ovarian carcinoma. Int. J. Gynecol. Cancer 2019, 29, 1389–1395. [Google Scholar] [CrossRef]
- Chatterjee, J.; Dai, W.; Aziz, N.H.A.; Teo, P.Y.; Wahba, J.; Phelps, D.L.; Maine, C.J.; Whilding, L.M.; Dina, R.; Trevisan, G.; et al. Clinical Use of Programmed Cell Death-1 and Its Ligand Expression as Discriminatory and Predictive Markers in Ovarian Cancer. Clin. Cancer Res. 2017, 23, 3453–3460. [Google Scholar] [CrossRef] [PubMed]
- Hamanishi, J.; Mandai, M.; Iwasaki, M.; Okazaki, T.; Tanaka, Y.; Yamaguchi, K.; Hoguchi, T.; Yagi, H.; Takakura, K.; Minato, N.; et al. Programmed cell death 1 ligand 1 and tumor-infiltrating CD8+ T lymphocytes are prognostic factors of human ovarian cancer. Proc. Natl. Acad. Sci. USA 2007, 104, 3360–3365. [Google Scholar] [CrossRef] [PubMed]
- Drakes, M.L.; Mehrotra, S.; Aldulescu, M.; Potkul, R.K.; Liu, Y.; Grisoli, A.; Joyce, C.; O’Brien, T.E.; Stack, M.S.; Stiff, P.J. Stratification of ovarian tumor pathology by expression of programmed cell death-1 (PD-1) and PD-ligand- 1 (PD-L1) in ovarian cancer. J. Ovarian Res. 2018, 11, 43. [Google Scholar] [CrossRef]
- Huang, L.-J.; Deng, X.-F.; Chang, F.; Wu, X.-L.; Wu, Y.; Diao, Q.-Z. Prognostic significance of programmed cell death ligand 1 expression in patients with ovarian carcinoma: A systematic review and meta-analysis. Medicine 2018, 97, e12858. [Google Scholar] [CrossRef]
- Grabosch, S.; Zeng, F.; Zhang, L.; Strange, M.; Brozick, J.; Edwards, R.P.; Vlad, A. PD-L1 biology in response to chemotherapy in vitro and in vivo in ovarian cancer. J. Immunother. Cancer 2015, 3, P302. [Google Scholar] [CrossRef]
- Wahba, J.; Natoli, M.; Whilding, L.M.; Parente-Pereira, A.C.; Jung, Y.; Zona, S.; Lam, E.W.F.; Simth, J.R.; Maher, J.; Ghaem-Maghami, S. Chemotherapy-induced apoptosis, autophagy and cell cycle arrest are key drivers of synergy in chemo-immunotherapy of epithelial ovarian cancer. Cancer Immunol. Immunother. 2018, 67, 1753–1765. [Google Scholar] [CrossRef]
- Böhm, S.; Montfort, A.; Pearce, O.M.T.; Topping, J.; Chakravarty, P.; Everitt, G.L.A.; Clear, A.; McDermott, J.R.; Ennis, D.; Dowe, T.; et al. Neoadjuvant Chemotherapy Modulates the Immune Microenvironment in Metastases of Tubo-Ovarian High-Grade Serous Carcinoma. Clin. Cancer Res. 2016, 22, 3025–3036. [Google Scholar] [CrossRef]
- Peng, J.; Hamanishi, J.; Matsumura, N.; Abiko, K.; Murat, K.; Baba, T.; yamaguchi, K.; Horikawa, N.; Hosoe, Y.; Murphy, S.K.; et al. Chemotherapy Induces Programmed Cell Death-Ligand 1 Overexpression via the Nuclear Factor-κB to Foster an Immunosuppressive Tumor Microenvironment in Ovarian Cancer. Cancer Res. 2015, 75, 5034–5045. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.M.; Liu, Y.C.; Li, W.; Liu, S.; Liu, H.X.; Li, L.W.; Ma, R. Nivolumab effectively inhibit platinum-resistant ovarian cancer cells via induction of cell apoptosis and inhibition of ADAM17 expression. Eur. Rev. Med. Pharmacol. Sci. 2017, 21, 1198–1205. [Google Scholar] [PubMed]
- Moss, M.L.; Minond, D. Recent advances in ADAM17 research: A promising target for cancer and inflammation. Mediat. Inflamm. 2017, 2017, 9673537. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Tao, Z.; Hai, B.; Liang, H.; Shi, Y.; Wang, T.; Song, W.; Chen, Y.; OuYang, J.; Chen, J.; et al. miR-424(322) reverses chemoresistance via T-cell immune response activation by blocking the PD-L1 immune checkpoint. Nat. Commun. 2016, 7, 11406. [Google Scholar] [CrossRef] [PubMed]
- D’Incalci, M.; Galmarini, C.M. A review of trabectedin (ET-743): A unique mechanism of action. Mol. Cancer Ther. 2010, 9, 2157–2163. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Wang, H.; Meng, F.; Li, J.; Zhang, S. Combined Trabectedin and anti-PD1 antibody produces a synergistic antitumor effect in a murine model of ovarian cancer. J. Transl. Med. 2015, 13, 247. [Google Scholar] [CrossRef] [PubMed]
- Germano, G.; Frapolli, R.; Belgiovine, C.; Anselmo, A.; Pesce, S.; Liguori, M.; Erba, E.; Uboldi, S.; Zucchetti, M.; Pasqualini, F.; et al. Role of macrophage targeting in the antitumor activity of trabectedin. Cancer Cell 2013, 23, 249–262. [Google Scholar] [CrossRef]
- Colmegna, B.; Uboldi, S.; Frapolli, R.; Licandro, S.A.; Panini, N.; Galmarini, C.M.; Badri, N.; Spanswick, V.J.; Bingham, J.P.; Kiakos, K.; et al. Increased sensitivity to platinum drugs of cancer cells with acquired resistance to trabectedin. Br. J. Cancer 2015, 113, 1687–1693. [Google Scholar] [CrossRef]
- Ventriglia, J.; Paciolla, I.; Cecere, S.C.; Pisano, C.; Di Napoli, M.; Arenare, L.; Setola, S.V.; Losito, N.S.; Califano, D.; Orsitura, M.; et al. Trabectedin in ovarian cancer: Is it now a standard of care? Clin. Oncol. 2018, 30, 498–503. [Google Scholar] [CrossRef]
- Ghaffari, A.; Peterson, N.; Khalaj, K.; Vitkin, N.; Robinson, A.; Francis, J.A.; Madhuri, K. STING agonist therapy in combination with PD-1 immune checkpoint blockade enhances response to carboplatin chemotherapy in high-grade serous ovarian cancer. Br. J. Cancer 2018, 119, 440–449. [Google Scholar] [CrossRef]
- Hartl, C.A.; Bertschi, A.; Puerto, R.B.; Andresen, C.; Cheney, E.M.; Mittendorf, E.A.; Guerriero, J.L.; Goldberg, M.S. Combination therapy targeting both innate and adaptive immunity improves survival in a pre-clinical model of ovarian cancer. J. Immunother. Cancer 2019, 7, 199. [Google Scholar] [CrossRef] [PubMed]
- Pujade-Lauraine, E.; Fujiwara, K. Avelumab alone or in combination with pegylated liposomal doxorubicin versus pegylated liposomal doxorubicin alone in platinum-resistant or refractory epithelial ovarian cancer: Primary and biomarker analysis of the phase III JAVELIN Ovarian 200 trial. Gynecol. Oncol. 2019, 154, 21–22. [Google Scholar] [CrossRef]
- Matulonis, U.A.; Barry, W.; Penson, R.T. Phase II study of pembrolizumab (pembro) combined with pegylated liposomal doxorubicin (PLD) for recurrent platinum-resistant ovarian, fallopian tube or peritoneal cancer. Gynecol. Oncol. 2018, 149, 24. [Google Scholar] [CrossRef]
- Inayama, Y.; Hamanishi, J.; Matsumura, N.; Murakami, R.; Abiko, K.; Yamaguchi, K.; baba, T.; Horie, K.; Konishi, I.; Mandai, M.l. Antitumor Effect of Nivolumab on Subsequent Chemotherapy for Platinum-Resistant Ovarian Cancer. Oncologist 2018, 23, 1382–1384. [Google Scholar] [CrossRef]
- Shimada, M.; Itamochi, H.; Kigawa, J. Nedaplatin: A cisplatin derivative in cancer chemotherapy. Cancer Manag. Res. 2013, 5, 67–76. [Google Scholar] [CrossRef]
- Zeng, Y.; Li, B.; Liang, Y.; Reeves, P.M.; Qu, X.; Ran, C.; Liu, Q.; Callahan, M.V.; Sluder, A.E.; Gelfand, J.A.; et al. Dual blockade of CXCL12-CXCR4 and PD-1-PD-L1 pathways prolongs survival of ovarian tumor-bearing mice by prevention of immunosuppression in the tumor microenvironment. FASEB J. 2019, 33, 6596–6608. [Google Scholar] [CrossRef]
- Chatterjee, S.; Behnam Azad, B.; Nimmagadda, S. The intricate role of CXCR4 in cancer. Adv. Cancer Res. 2014, 124, 31–82. [Google Scholar]
- Meder, L.; Schuldt, P.; Thelen, M.; Schmitt, A.; Dietlein, F.; Klein, S.; Borchmann, S.; Wennhold, K.; Vlasic, I.; Oberbeck, S.; et al. Combined VEGF and PD-L1 Blockade Displays Synergistic Treatment Effects in an Autochthonous Mouse Model of Small Cell Lung Cancer. Cancer Res. 2018, 78, 4270–4281. [Google Scholar] [CrossRef]
- Schmittnaegel, M.; Rigamonti, N.; Kadioglu, E.; Cassará, A.; Wyser Rmili, C.; Kiialainen, A.; Kienast, Y.; Mueller, H.J.; Ooi, C.H.; Laoui, D.; et al. Dual angiopoietin-2 and VEGFA inhibition elicits antitumor immunity that is enhanced by PD-1 checkpoint blockade. Sci. Transl. Med. 2017, 9, eaak9670. [Google Scholar] [CrossRef]
- Liu, J.F.; Herold, C.; Gray, K.P.; Penson, R.T.; Horowitz, N.; Konstantinopoulos, P.A.; Castro, C.M.; Hill, S.J.; Curtis, J.; Luo, W.; et al. Assessment of combined nivolumab and bevacizumab in relapsed ovarian cancer: A phase 2 clinical trial. JAMA Oncol. 2019, 5, 1731–1738. [Google Scholar] [CrossRef]
- Huang, Y.; Lichtenberger, L.M.; Taylor, M. Antitumor and antiangiogenic effects of aspirin-PC in ovarian cancer. Mol. Cancer Ther. 2016, 15, 2894–2904. [Google Scholar] [CrossRef] [PubMed]
- Jhiang, S.M. The RET proto-oncogene in human cancers. Oncogene 2000, 19, 5590–5597. [Google Scholar] [CrossRef] [PubMed]
- Wieser, V.; Gaugg, I.; Fleischer, M.; Shivalingaiah, G.; Wenzel, S.; Sprung, S.; Lax, S.F.; Zeiment, A.G.; Fiegl, H.; Marth, C. BRCA1/2 and TP53 mutation status associates with PD-1 and PD-L1 expression in ovarian cancer. Oncotarget 2018, 9, 17501–17511. [Google Scholar] [CrossRef]
- Strickland, K.C.; Howitt, B.E.; Shukla, S.A.; Rodig, S.; Ritterhouse, L.L.; Liu, J.F.; Garbar, J.E.; Chowdhury, D.; Wu, C.J.; D’Andrea, A.D.; et al. Association and prognostic significance of BRCA1/2-mutation status with neoantigen load, number of tumor-infiltrating lymphocytes and expression of PD-1/PD-L1 in high grade serous ovarian cancer. Oncotarget 2016, 7, 13587–13598. [Google Scholar] [CrossRef]
- Ledermann, J.A. PARP inhibitors in ovarian cancer. Ann. Oncol. 2016, 27 (Suppl. 1), i40–i44. [Google Scholar] [CrossRef]
- Wang, Z.; Sun, K.; Xiao, Y.; Feng, B.; Mikule, K.; Ma, X.; Feng, N.; Vellano, C.P.; Federico, L.; Marszalek, J.R.; et al. Niraparib activates interferon signaling and potentiates anti-PD-1 antibody efficacy in tumor models. Sci. Rep. 2019, 9, 1853. [Google Scholar] [CrossRef] [PubMed]
- Ding, L.; Kim, H.-J.; Wang, Q.; Kearns, M.; Jiang, T.; Ohlson, C.E.; Li, B.B.; Xie, S.; Liu, J.F.; Stover, E.H.; et al. PARP Inhibition Elicits STING-Dependent Antitumor Immunity in Brca1-Deficient Ovarian Cancer. Cell Rep. 2018, 25, 2972–2980.e5. [Google Scholar] [CrossRef] [PubMed]
- Konstantinopoulos, P.A.; Waggoner, S.E.; Vidal, G.A.; Mita, M.M.; Fleming, G.F.; Holloway, R.W.; Van Lee, L.; Sachdev, J.C.; Chapman-Davis, E.; Colon-otero, G.; et al. TOPACIO/Keynote-162 (NCT02657889): A phase 1/2 study of niraparib + pembrolizumab in patients (pts) with advanced triple-negative breast cancer or recurrent ovarian cancer (ROC)—Results from ROC cohort. J. Clin. Oncol. 2018, 36 (Suppl. 15), 106. [Google Scholar] [CrossRef]
- Färkkilä, A.; Gulhan, D.C.; Casado, J.; Jacobson, C.A.; Nguyen, H.; Kochupurakkal, B.; Maliga, Z.; Yapp, C.; Chen, Y.-A.; Schapiro, D.; et al. Immunogenomic profiling determines responses to combined PARP and PD-1 inhibition in ovarian cancer. Nat. Commun. 2020, 11, 1459. [Google Scholar] [CrossRef]
- Friedlander, M.; Meniawy, T.; Markman, B.; Mileshkin, L.R.; Harnett, P.; Millward, M.; Lundy, J.; Freimund, A.E.; Norris, C.; Wu, J.; et al. A phase 1b study of the anti-PD-1 monoclonal antibody BGB-A317 (A317) in combination with the PARP inhibitor BGB-290 (290) in advanced solid tumors. J. Clin. Oncol. 2018, 36 (Suppl. 5), 48. [Google Scholar] [CrossRef]
- Drew, Y.; de Jonge, M.; Hong, S.H.; Park, Y.H.; Wolfer, A.; Brown, J.; Ferguson, M.; Gore, M.E.; Alvarez, R.; Gresty, C.; et al. An open-label, phase II basket study of olaparib and durvalumab (MEDIOLA): Results in germline BRCA-mutated (gBRCA m) platinum-sensitive relapsed (PSR) ovarian cancer (OC). Gynecol. Oncol. 2018, 149, 246–247. [Google Scholar] [CrossRef]
- Lee, J.M.; Annunziata, C.M.; Houston, N.; Kohn, E.C. 936PD A phase 2 study of durvalumab, a PD-L1 inhibitor and olaparib in recurrent ovarian cancer (OvCa). Ann. Oncol. 2018, 29 (Suppl. 8), mdy285-145. [Google Scholar] [CrossRef]
- Doo, D.W.; Norian, L.A.; Arend, R.C. Checkpoint inhibitors in ovarian cancer: A review of preclinical data. Gynecol. Oncol. Rep. 2019, 29, 48–54. [Google Scholar] [CrossRef] [PubMed]
- Wei, H.; Zhao, L.; Li, W.; Fan, K.; Qian, W.; Hou, S.; Wang, H.; Dai, M.; Hellstrom, I.; Hellstrom, K.E.; et al. Combinatorial PD-1 blockade and CD137 activation has therapeutic efficacy in murine cancer models and synergizes with cisplatin. PLoS ONE 2013, 8, e84927. [Google Scholar] [CrossRef] [PubMed]
- Guo, Z.; Wang, X.; Cheng, D.; Xia, Z.; Luan, M.; Zhang, S. Correction: PD-1 Blockade and OX40 Triggering Synergistically Protects against Tumor Growth in a Murine Model of Ovarian Cancer. PLoS ONE 2017, 12, e0186965. [Google Scholar] [CrossRef]
- Lu, L.; Xu, X.; Zhang, B.; Zhang, R.; Ji, H.; Wang, X. Combined PD-1 blockade and GITR triggering induce a potent antitumor immunity in murine cancer models and synergizes with chemotherapeutic drugs. J. Transl. Med. 2014, 12, 36. [Google Scholar] [CrossRef]
- Sawada, M.; Goto, K.; Morimoto-Okazawa, A.; Haruna, M.; Yamamoto, K.; Yamamoto, Y.; Nakagawa, S.; Hiramatsu, K.; Matsuzaki, S.; Kobayashi, E.; et al. PD-1+ Tim3+ tumor-infiltrating CD8 T cells sustain the potential for IFN-γ production, but lose cytotoxic activity in ovarian cancer. Int. Immunol. 2020, 32, 397–405. [Google Scholar] [CrossRef]
- Rådestad, E.; Klynning, C.; Stikvoort, A.; Mogensen, O.; Nava, S.; Magalhaes, I.; Uhlin, M. Immune profiling and identification of prognostic immune-related risk factors in human ovarian cancer. Oncoimmunology 2019, 8, e1535730. [Google Scholar] [CrossRef]
- Liu, Y.; Yu, Y.; Yang, S.; Zeng, B.; Zhang, Z.; Jiao, G.; Zhang, Y.; Cai, L.; Yang, R. Regulation of arginase I activity and expression by both PD-1 and CTLA-4 on the myeloid-derived suppressor cells. Cancer Immunol. Immunother. 2009, 58, 687–697. [Google Scholar] [CrossRef]
- Duraiswamy, J.; Kaluza, K.M.; Freeman, G.J.; Coukos, G. Dual blockade of PD-1 and CTLA-4 combined with tumor vaccine effectively restores T-cell rejection function in tumors. Cancer Res. 2013, 73, 3591–3603. [Google Scholar] [CrossRef]
- Duraiswamy, J.; Freeman, G.J.; Coukos, G. Therapeutic PD-1 pathway blockade augments with other modalities of immunotherapy T-cell function to prevent immune decline in ovarian cancer. Cancer Res. 2013, 73, 6900–6912. [Google Scholar] [CrossRef] [PubMed]
- Dai, M.; Wei, H.; Yip, Y.Y.; Feng, Q.; He, K.; Popov, V.; Hellstrom, I.; Hellstrom, K.E. Long-lasting complete regression of established mouse tumors by counteracting Th2 inflammation. J. Immunother. 2013, 36, 248–257. [Google Scholar] [CrossRef] [PubMed]
- Huang, R.-Y.; Francois, A.; McGray, A.R.; Miliotto, A.; Odunsi, K. Compensatory upregulation of PD-1, LAG-3, and CTLA-4 limits the efficacy of single-agent checkpoint blockade in metastatic ovarian cancer. Oncoimmunology 2017, 6, e1249561. [Google Scholar] [CrossRef] [PubMed]
- Gao, Q.; Dong, X.; Xu, Q.; Zhu, L.; Wang, F.; Hou, Y.; Chao, C.C. Therapeutic potential of CRISPR/Cas9 gene editing in engineered T-cell therapy. Cancer Med. 2019, 8, 4254–4264. [Google Scholar] [CrossRef] [PubMed]
- Imai, Y.; Hasegawa, K.; Matsushita, H.; Fujieda, N.; Sato, S.; Miyagi, E.; Kakimi, K.; Fujiwara, K. Expression of multiple immune checkpoint molecules on T cells in malignant ascites from epithelial ovarian carcinoma. Oncol. Lett. 2018, 15, 6457–6468. [Google Scholar] [CrossRef] [PubMed]
- Zamarin, D.; Burger, R.A.; Sill, M.W.; Powell, D.J.; Lankes, H.A.; Feldman, M.D.; Zivanovic, O.; Gunderson, C.; Ko, E.; Matthews, C. Randomized phase II trial of nivolumab versus nivolumab and ipilimumab for recurrent or persistent ovarian cancer: An NRG oncology study. J. Clin. Oncol. 2020, 38, 1814–1823. [Google Scholar] [CrossRef]
- Bekes, I.; Löb, S.; Holzheu, I.; Janni, W.; Baumann, L.; Wöckel, A.; Wulff, C. Nectin-2 in ovarian cancer: How is it expressed and what might be its functional role? Cancer Sci. 2019, 110, 1872–1882. [Google Scholar] [CrossRef]
- Whelan, S.; Ophir, E.; Kotturi, M.F.; Levy, O.; Ganguly, S.; Leung, L.; Vaknon, I.; Kumar, S.; Dassa, L.; Hansen, K.; et al. PVRIG and PVRL2 Are Induced in Cancer and Inhibit CD8+ T-cell Function. Cancer Immunol. Res. 2019, 7, 257–268. [Google Scholar] [CrossRef]
- Gitto, S.B.; Kim, H.; Rafail, S.; Omran, D.K.; Medvedev, S.; Kinose, Y.; Rodriquez-Garcia, A.; Flowers, A.J.; Xu, H.; Schwartz, L.E.; et al. An autologous humanized patient-derived-xenograft platform to evaluate immunotherapy in ovarian cancer. Gynecol. Oncol. 2020, 156, 222–232. [Google Scholar] [CrossRef]
- Oyer, J.L.; Gitto, S.B.; Altomare, D.A.; Copik, A.J. PD-L1 blockade enhances anti-tumor efficacy of NK cells. Oncoimmunology 2018, 7, e1509819. [Google Scholar] [CrossRef]
- McGray, A.J.R.; Huang, R.-Y.; Battaglia, S.; Eppolito, C.; Miliotto, A.; Stephenson, K.B.; lugade, A.A.; Webster, G.; Lichty, B.D.; Seshadri, M.; et al. Oncolytic Maraba virus armed with tumor antigen boosts vaccine priming and reveals diverse therapeutic response patterns when combined with checkpoint blockade in ovarian cancer. J. Immunother. Cancer 2019, 7, 189. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Ravindranathan, R.; Kalinski, P.; Guo, Z.S.; Bartlett, D.L. Rational combination of oncolytic vaccinia virus and PD-L1 blockade works synergistically to enhance therapeutic efficacy. Nat. Commun. 2017, 8, 14754. [Google Scholar] [CrossRef] [PubMed]
- Kowalsky, S.J.; Liu, Z.; Feist, M.; Berkey, S.E.; Ma, C.; Ravindranathan, R.; Dai, E.; Roy, E.J.; Guo, Z.S.; Bartlett, D.L. Superagonist IL-15-Armed Oncolytic Virus Elicits Potent Antitumor Immunity and Therapy That Are Enhanced with PD-1 Blockade. Mol. Ther. 2018, 26, 2476–2486. [Google Scholar] [CrossRef] [PubMed]
- Qiao, G.; Wang, X.; Zhou, X.; Morse, M.A.; Wu, J.; Wang, S.; Song, Y.; Jiang, N.; Zhao, Y.; Zhou, L.; et al. Immune correlates of clinical benefit in a phase I study of hyperthermia with adoptive T cell immunotherapy in patients with solid tumors. Int. J. Hyperth. 2019, 36 (Suppl. 1), 74–82. [Google Scholar] [CrossRef] [PubMed]
- Yeku, O.; Zamarin, D.; Gallagher, J.; Aghajanian, C.A.; Konner, J. A phase II trial of TPIV200 (a polypeptide vaccine against folate receptor alpha) plus durvalumab (anti-PD-L1 antibody) in patients with platinum-resistant ovarian cancer. Gynecol. Oncol. 2018, 149, 56–57. [Google Scholar] [CrossRef]
- O’Cearbhaill, R.E.; Gnjatic, S.; Aghajanian, C.; Iasonos, A.; Konner, J.A.; Losada, N.; Sarasohn, D.; rasalan, T.; Tew, W.P.; Zamarin, D. A phase I study of concomitant galinpepimut-s (GPS) in combination with nivolumab (nivo) in patients (pts) with WT1+ ovarian cancer (OC) in second or third remission. JCO 2018, 36 (Suppl. 15), 5553. [Google Scholar] [CrossRef]
- Sanborn, R.E.; Pishvaian, M.J.; Kluger, H.M.; Callahan, M.K. Clinical results with combination of anti-CD27 agonist antibody, varlilumab, with anti-PD1 antibody nivolumab in advanced cancer patients. J. Clin. Oncol. 2017, 35 (Suppl. 15), 3007. [Google Scholar] [CrossRef]
- Padmanabhan, S.; Zou, Y.; Vancurova, I. Immunoblotting Analysis of Intracellular PD-L1 Levels in Interferon-γ-Treated Ovarian Cancer Cells Stably Transfected with Bcl3 shRNA. Methods Mol. Biol. 2020, 2108, 211–220. [Google Scholar]
- Zou, Y.; Uddin, M.M.; Padmanabhan, S.; Zhu, Y.; Bu, P.; Vancura, A.; Vancurova, I. The proto-oncogene Bcl3 induces immune checkpoint PD-L1 expression, mediating proliferation of ovarian cancer cells. J. Biol. Chem. 2018, 293, 15483–15496. [Google Scholar] [CrossRef]
- Guo, T.; Li, B.; Gu, C.; Chen, X.; Han, M.; Liu, X.; Xu, C. PGC-1α inhibits polyamine metabolism in Cyclin E1-driven ovarian cancer. Cancer Med. 2019, 8, 7754–7761. [Google Scholar] [CrossRef]
- Natoli, M.; Bonito, N.; Robinson, J.D.; Ghaem-Maghami, S.; Mao, Y. Human ovarian cancer intrinsic mechanisms regulate lymphocyte activation in response to immune checkpoint blockade. Cancer Immunol. Immunother. 2020, 69, 1391–1401. [Google Scholar] [CrossRef] [PubMed]
- Padmanabhan, S.; Zou, Y.; Vancurova, I. Flow Cytometry Analysis of Surface PD-L1 Expression Induced by IFNγ and Romidepsin in Ovarian Cancer Cells. Methods Mol. Biol. 2020, 2108, 221–228. [Google Scholar] [PubMed]
- Li, X.; Fu, Y.; Yang, B.; Guo, E.; Wu, Y.; Huang, J.; Zhang, X.; Xiao, R.; Li, K.; Wang, B.; et al. BRD4 inhibition by AZD5153 promotes antitumor immunity via depolarizing M2 macrophages. Front. Immunol. 2020, 11, 89. [Google Scholar] [CrossRef] [PubMed]
- Crawford, A.; Haber, L.; Kelly, M.P.; Vazzana, K.; Canova, L.; Ram, P.; Pawashe, A.; Finney, J.; Jalal, S.; Chiu, D.; et al. A Mucin 16 bispecific T cell-engaging antibody for the treatment of ovarian cancer. Sci. Transl. Med. 2019, 11, eaau7534. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.-J.; Siu, M.K.; Jiang, Y.-X.; Leung, T.H.; Chan, D.W.; Cheng, R.R.; Cheung, A.N.; Ngan, H.Y.; Chan, K.K. Aberrant upregulation of PDK1 in ovarian cancer cells impairs CD8+ T cell function and survival through elevation of PD-L1. Oncoimmunology 2019, 8, e1659092. [Google Scholar] [CrossRef]
- Zhang, Y.; Yang, S.; Yang, Y.; Liu, T. Resveratrol induces immunogenic cell death of human and murine ovarian carcinoma cells. Infect. Agents Cancer 2019, 14, 27. [Google Scholar] [CrossRef]
- Cao, Q.; Wang, W.; Zhou, M.; Huang, Q.; Wen, X.; Zhao, J.; Shi, S.; Geng, K.; Li, F.; Hatakeyama, H.; et al. Induction of antitumor immunity in mice by the combination of nanoparticle-based Photothermolysis and anti-PD-1 checkpoint inhibition. Nanomedicine 2020, 25, 102169. [Google Scholar] [CrossRef]
- Teo, P.Y.; Yang, C.; Whilding, L.M.; Parente-Pereira, A.C.; Maher, J.; George, A.J.T.; Hedrick, J.L.; Yang, Y.Y.; Ghaem-Maghami, S. Ovarian cancer immunotherapy using PD-L1 siRNA targeted delivery from folic acid-functionalized polyethylenimine: Strategies to enhance T cell killing. Adv. Healthc. Mater. 2015, 4, 1180–1189. [Google Scholar] [CrossRef]
- Cadoo, K.A.; Meyers, M.L.; Burger, R.A.; Armstrong, D.K.; Penson, R.T.; Gordon, M.S.; Fleming, G.F.; Moroney, J.W.; Hamilton, E.P.; Duska, L.R.; et al. A Phase II Randomized Study of Avelumab Plus Entinostat Versus Avelumab Plus Placebo in Patients (pts) with Advanced Epithelial Ovarian Cancer (EOC). J. Clin. Oncol. 2019, 37 (Suppl. 15), 5511. [Google Scholar] [CrossRef]
- Matulonis, U.A.; Moore, K.N. Initial safety and activity findings from a phase IB escalation study of mirvetuximab soravtansine, a folate receptor alpha (FRα-targeting antibody-drug conjugate (ADC), with pembrolizumab in platinum-resistant epithelial ovarian cancer (EOC) patients. Gynecol. Oncol. 2018, 149, 38. [Google Scholar] [CrossRef]
- Cole, A.J.; Dwight, T.; Gill, A.J.; Dickson, K.-A.; Zhu, Y.; Clarkson, A.; Gard, G.B.; Maidens, J.; Valmadre, S.; Clifton-Bligh, R.; et al. Assessing mutant p53 in primary high-grade serous ovarian cancer using immunohistochemistry and massively parallel sequencing. Sci. Rep. 2016, 6, 26191. [Google Scholar] [CrossRef] [PubMed]
- McCloskey, C.W.; Rodriguez, G.M.; Galpin, K.J.C.; Vanderhyden, B.C. Ovarian cancer immunotherapy: Preclinical models and emerging therapeutics. Cancers 2018, 10, 244. [Google Scholar] [CrossRef] [PubMed]
- McCloskey, C.W.; Goldberg, R.L.; Carter, L.E.; Gamwell, L.F.; Al-Hujaily, E.M.; Collins, O.; Macdonald, E.A.; Garson, K.; Daneshman, M.; Carmona, E.; et al. A new spontaneously transformed syngeneic model of high-grade serous ovarian cancer with a tumor-initiating cell population. Front. Oncol. 2014, 4, 53. [Google Scholar] [CrossRef] [PubMed]
- Okada, S.; Vaeteewoottacharn, K.; Kariya, R. Application of Highly Immunocompromised Mice for the Establishment of Patient-Derived Xenograft (PDX) Models. Cells 2019, 8, 889. [Google Scholar] [CrossRef] [PubMed]
- Gibellini, L.; De Biasi, S.; Porta, C.; Lo Tartaro, D.; Depenni, R.; Pellacani, G.; Sabbatini, R.; Cossarizza, A. Single-Cell Approaches to Profile the Response to Immune Checkpoint Inhibitors. Front. Immunol. 2020, 11, 490. [Google Scholar] [CrossRef]
| Study Title | Trial Identifier | Study Arm(s) | Primary Endpoint(s) | Results |
|---|---|---|---|---|
| A Phase 1b to Assess the Safety and Tolerability of Carboplatin-Cyclophosphamide Combined with Atezolizumab, an Antibody That Targets Programmed Death Ligand 1 (PD-L1), in Patients with Advanced Breast Cancer and Gynecologic Cancer | NCT02914470 | Single Arm | Toxicity; AEs | N/A |
| Matched Paired Pharmacodynamics and Feasibility Study of Durvalumab in Combination with Chemotherapy in Frontline Ovarian Cancer (N-Dur) | NCT02726997 | Single Arm | Pharmacodynamic changes | N/A |
| Anti-PD-1 Therapy in Combination with Platinum Chemotherapy for Platinum Resistant Ovarian, Fallopian Tube, and Primary Peritoneal Cancer | NCT03029598 | Single Arm | PFS | N/A |
| A Phase II Study of Pembrolizumab With Cisplatin and Gemcitabine Treatment in Patients with Recurrent Platinum-Resistant Ovarian Cancer | NCT02608684 | Single Arm | ORR by RECIST | CR: 5.6% PR: 55.6% SD: 27.8% PD: 11.1% |
| Phase II Open Label Nonrandomized Trial of the Anti PD-1 Therapy Pembrolizumab with First Line Platinum-Based Chemotherapy Followed by 12 Months Pembrolizumab Monotherapy for Patients with Stage III/IV Epithelial Ovarian Cancer | NCT02766582 | Single Arm | PFS | N/A |
| Matched Paired Pharmacodynamics and Feasibility Study of Pembrolizumab in Combination with Chemotherapy in Frontline Ovarian Cancer | NCT02520154 | Single Arm | PFS | N/A |
| Phase II Single Arm Study of Combination Pembrolizumab, Paclitaxel, and Carboplatin in Patients with Advanced Stage Ovarian, Fallopian Tube, or Peritoneal Carcinoma Receiving Neoadjuvant Chemotherapy | NCT02834975 | Single Arm | pORR | N/A |
| A Phase II Study of Pembrolizumab Combined with Pegylated Liposomal Doxorubicin (PLD) for Recurrent Platinum Resistant Ovarian, Fallopian Tube or Peritoneal Cancer | NCT02865811 | Single Arm | CBR | ORR: 11.5% [72] |
| A Randomized, Open-label, Multicenter, Phase 3 Study to Evaluate the Efficacy and Safety of Avelumab (MSB0010718C) in Combination with and/or Following Chemotherapy in Patients with Previously Untreated Epithelial Ovarian Cancer JAVELIN OVARIAN 100 | NCT02718417 | Arm 1: Chemotherapy Arm 2: Chemotherapy + avelumab maintenance Arm 3: Chemotherapy + avelumab + avelumab maintenance | PFS | Arm 1: N/A Arm 2: PFS: 16.8 months Arm 3: PFS: 18.1 months Study Terminated |
| A Phase 3, Multicenter, Randomized, Open-label Study of Avelumab (MSB0010718C) Alone or in Combination with Pegylated Liposomal Doxorubicin Alone in Patients with Platinum-Resistant/Refractory Ovarian Cancer | NCT02580058 | Arm 1: Avelumab Arm 2: Avelumab + PLD Arm 3: PLD | OS; PFS | Arm 1: OS: 11.8 months PFS: 1.9 months Arm 2: OS: 15.7 months PFS: 3.7 months Arm 3: OS: 13.1 months PFS: 3.5 months |
| Study Title | Trial Identifier | Study Arm(s) | Primary Endpoint(s) | Results |
|---|---|---|---|---|
| Phase Ib Trial of Pembrolizumab and Nintedanib | NCT02856425 | Single Arm | MTD of Nintedanib | N/A |
| A Phase II Study of the Anti-PDL1 Antibody Atezolizumab, Bevacizumab and Acetylsalicylic Acid to Investigate Safety and Efficacy of This Combination in Recurrent Platinum-Resistant Ovarian, Fallopian Tube or Primary Peritoneal Adenocarcinoma | NCT02659384 | Arm 1: Bevacizumab Arm 2: Atezolizumab + Bevacizumab Arm 3: Atezolizumab + Bevacizumab + Acetylsalicylic Acid | PFS | N/A |
| A Phase II Study with a Safety Lead-in of Nivolumab in Combination with Bevacizumab or in Combination with Bevacizumab and Rucaparib for the Treatment of Relapsed Epithelial Ovarian, Fallopian Tube or Peritoneal Cancer | NCT02873962 | Arm 1: Nivolumab + Bevacizumab Arm 2: Nivolumab + Bevacizumab + Rucaparib | ORR by RECIST | ORR: 28.9% (40% platinum-sensitive 16.7% platinum- resistant) [79] |
| A Randomized, Phase II/III Study of Pegylated Liposomal Doxorubicin and CTEP-Supplied Atezolizumab Versus Pegylated Liposomal Doxorubicin/Bevacizumab and CTEP-Supplied Atezolizumab Versus Pegylated Liposomal Doxorubicin/Bevacizumab in Platinum-Resistant Ovarian Cancer | NCT02839707 | Arm 1: PLD + Atezolizumab Arm 2: PLD + Bevacizumab + Atezolizumab Arm 3: PLD + Bevacizumab | DLT; PFS; OS | N/A |
| Atezolizumab in Combination with Bevacizumab and Chemotherapy versus Bevacizumab and Chemotherapy in Recurrent Ovarian Cancer—ARandomized Phase III Trial | NCT03353831 | Arm 1: Chemotherapy + Bevacizumab Arm 2: Chemotherapy + Bevacizumab + Atezolizumab | OS; PFS | N/A |
| A Phase III, Multicenter, Randomized, Study of Atezolizumab versus Placebo Administered in Combination with Paclitaxel, Carboplatin, and Bevacizumab to Patients with Newly Diagnosed Stage III or Stage IV Ovarian, Fallopian Tube, or Primary Peritoneal Cancer | NCT03038100 | Arm 1: Atezolizumab + Paclitaxel, Carboplatin and Bevacizumab Arm 2: Paclitaxel, Carboplatin and Bevacizumab | PFS, OS | N/A |
| A Randomized, Double-Blinded, Phase III Study of Atezolizumab versus Placebo in Patients with Late Relapse of Epithelial Ovarian, Fallopian Tube, or Peritoneal Cancer Treated by Platinum-Based Chemotherapy and Bevacizumab | NCT02891824 | Arm 1: Bevacizumab + Chemotherapy Arm 2: Atezolizumab + Bevacizumab + Chemotherapy | PFS | N/A |
| Study Title | Trial Identifier | Study Arm(s) | Primary Endpoint(s) | Results |
|---|---|---|---|---|
| A Phase 1/1b, Open Label, Multiple Dose, Dose Escalation and Expansion Study to Investigate the Safety, Pharmacokinetics and Antitumor Activity of the Anti-PD-1 Monoclonal Antibody BGB-A317 in Combination with the PARP Inhibitor BGB-290 in Subjects with Advanced Solid Tumors | NCT02660034 | Single Arm | Phase 1: AEs; DLT; MTD Phase 1b: ORR, PFS DOR, DCR, CBR, OS | PR: 7(5 EOC)/38 patients CR:1(EOC)/38 patients [89] |
| A Phase I/II Evaluation of Olaparib in Combination with Durvalumab (Medi4736) and Tremelimumab in the Treatment of Recurrent Platinum-Sensitive or Resistant or Refractory Epithelial Ovarian, Fallopian Tube, or Primary Peritoneal Cancer in Patients Who Carry a BRCA1 or BRCA2 Mutation | NCT02953457 | Single Arm | DLT, PFS | N/A |
| Phase I/II Study of the Anti-Programmed Death Ligand-1 Antibody MEDI4736 in Combination with Olaparib and/or Cediranib for Advanced Solid Tumors and Advanced or Recurrent Ovarian, Triple Negative Breast, Lung, Prostate and Colorectal Cancers | NCT02484404 | Sequential Assignment | Phase I: RP2D Phase II: ORR | DCR: 53% [91] |
| Phase 1/2 Clinical Study of Niraparib in Combination with Pembrolizumab (MK-3475) in Patients with Advanced or Metastatic Triple-Negative Breast Cancer and in Patients with Recurrent Ovarian Cancer | NCT02657889 | Single Arm | Phase I: DLT, AEs Phase II: ORR | ORR:25%, [87] |
| A Phase I/II Study of MEDI4736 (Anti-PD-L1 Antibody) in Combination with Olaparib (PARP Inhibitor) in Patients with Advanced Solid Tumors | NCT02734004 | Single Arm | DCR, Safety and tolerability, ORR | DCR:81% ORR:63% [90] |
| A Phase 2, Open-label Study to Evaluate Rucaparib in Combination with Nivolumab in Patients with Selected Solid Tumors (ARIES) | NCT03824704 | Single Arm | ORR | N/A |
| Phase 2 Multicohort Study to Evaluate the Safety and Efficacy of Novel Treatment Combinations in Patients with Recurrent Ovarian Cancer | NCT03574779 | Single Arm | ORR | N/A |
| ATHENA (A Multicenter, Randomized, Double-Blind, Placebo-Controlled Phase 3 Study in Ovarian Cancer Patients Evaluating Rucaparib and Nivolumab as Maintenance Treatment Following Response to Front-Line Platinum-Based Chemotherapy) | NCT03522246 | Arm 1: Rucaparib + nivolumab Arm 2: Rucaparib Arm 3: Nivolumab Arm 4: Placebo | PFS | N/A |
| ENGOT-0V44 The FIRST (First-Line Ovarian Cancer Treatment with Niraparib Plus TSR-042) Study: A Randomized, Double-Blind, Phase 3 Comparison of Platinum-Based Therapy with TSR-042 and Niraparib Versus Standard of Care Platinum-Based Therapy as First-Line Treatment of Stage III or IV Nonmucinous Epithelial Ovarian Cancer | NCT03602859 | Arm 1: Chemotherapy + Bevacizumab Arm 2: Chemotherapy + Niraparib Arm 3: Chemotherapy + TSR-042 | PFS | N/A |
| A Randomized Phase 3, Double-Blind Study of Chemotherapy with or without Pembrolizumab Followed by Maintenance with Olaparib or Placebo for the First-Line Treatment of BRCA Non-Mutated Advanced Epithelial Ovarian Cancer (EOC) (KEYLYNK-001/ENGOT-ov43) | NCT03740165 | Arm 1: Pembrolizumab + Olaparib Arm 2: Pembrolizumab Arm 3: Placebo | PFS; OS | N/A |
| A Randomized, Open-Label Multicenter, Phase 3 Study to Evaluate the Efficacy and Safety of Avelumab in Combination with Chemotherapy Followed by Maintenance Therapy of Avelumab in Combination with the Poly (Adenosine Diphosphate [ADP]-Ribose) POLYMERASE (PARP) Inhibitor Talazoparib in Patients with Previously Untreated Advanced Ovarian Cancer (JAVELIN OVARIAN PARP100) | NCT03642132 | Arm 1: Chemotherapy + Avelumab + Talazoparib Arm 2: Chemotherapy + Talazoparib Arm 3: Chemotherapy + Bevacizumab | PFS | Study Discontinued |
| A Phase III Randomized, Double-Blind, Placebo-Controlled, Multicenter Study of Durvalumab in Combination with Chemotherapy and Bevacizumab, Followed by Maintenance Durvalumab, Bevacizumab and Olaparib in Newly Diagnosed Advanced Ovarian Cancer Patients (DUO-O). | NCT03737643 | Arm 1: Chemotherapy + Bevacizumab + maintenance Bevacizumab, Arm 2: Chemotherapy + Bevacizumab + Durvalumab + maintenance Bevacizumab + Durvalumab Arm 3: Chemotherapy + Bevacizumab +Durvalumab + maintenance Bevacizumab + Durvalumab + Olaparib. tBRCAm cohort: Chemotherapy + Bevacizumab + Durvalumab + maintenance Bevacizumab + Durvalumab + Olaparib. | PFS | N/A |
| Study Title | Trial Identifier | Study Arm(s) | Primary Endpoint(s) | Results |
|---|---|---|---|---|
| A Phase 1 Study to Evaluate the Safety and Tolerability of Anti-PD-L1, MEDI4736, in Combination with Tremelimumab in Subjects with Advanced Solid Tumors | NCT01975831 | Single Arm | AEs | N/A |
| A Phase 1, Open-Label, Dose Escalation and Dose Expansion Trial Evaluating the Safety, Pharmacokinetics, Pharmacodynamics, and Clinical Effects of Orally Administered CA-170 in Patients with Advanced Tumors and Lymphomas | NCT02812875 | Single Arm | DLT; MTD; RP2D | N/A |
| A Phase I Study of MEDI4736 (Anti-PD-L1 Antibody) in Combination with Tremelimumab (Anti-CTLA-4 Antibody) in Subjects with Advanced Solid Tumors | NCT02261220 | Single Arm | AEs, SAEs, DLT, ORR | N/A |
| A Phase 1, Open-Label, Dose Escalation Study of MGA271 in Combination with Pembrolizumab and in Combination with MGA012 in Patients with Melanoma, Squamous Cell Cancer of the Head and Neck, Non-Small Cell Lung Cancer, Urothelial Cancer, and Other Cancers | NCT02475213 | Sequential Assignment | AEs; SAEs | N/A |
| A Phase 1a/1b Study of COM701 as Monotherapy and in Combination with an Anti-PD-1 Antibody in Subjects with Advanced Solid Tumors | NCT03667716 | Arm 1: Monotherapy dose escalation Arm 2: Combination dose escalation Arm 3: Monotherapy expansion Arm 4: Combination dose expansion | AEs; MTD | N/A |
| A Phase Ib Study of the Safety and Pharmacology of Atezolizumab (Anti−Pd-L1 Antibody) Administered with Ipilimumab, Interferon-Alpha, or Other Immune-Modulating Therapies in Patients with Locally Advanced or Metastatic Solid Tumors | NCT02174172 | Arm 1: Atezolizumab + Ipilimumab Arm 2: Atezolizumab + Interferon alfa-2 Arm 3: Atezolizumab + Peg- Interferon alfa-2a Arm 4: Atezolizumab + Peg-Interferon alfa-2a+ Bevacizumab Arm 5: Atezolizumab + Obinutuzumab | RP2D; AEs | N/A |
| A Phase 1/2, Open-Label Study of Nivolumab Monotherapy or Nivolumab Combined with Ipilimumab in Subjects with Advanced or Metastatic Solid Tumors | NCT01928394 | Arm 1: Nivolumab Arm 2–5: Nivolumab + Ipilimumab at various dosages Arm 6: Nivolumab + Ipilimumab + Cobimetinib | ORR | OC Arm N-I Dose Level 2: ORR: 12.2% AEs: 58.54% OC Arm N-I Dose Level 2b: ORR: 7.0% AEs: 72.09% OC Arm N-I Dose Level 2c: ORR: 9.5% AEs: 69.05% |
| Phase II Randomized Trial of Nivolumab with or without Ipilimumab in Patients with Persistent or Recurrent Epithelial Ovarian, Primary Peritoneal, or Fallopian Tube Cancer | NCT02498600 | Arm 1: Nivolumab Arm 2: Nivolumab + Ipilimumab | ORR | ORR: 12.2% (Arm 1), 31.4% (Arm 2) PFS: 2 mo. (Arm 1), 3.9 mo. (Arm 2) [105] |
| A Phase II Clinical Trial Evaluating Ipilimumab and Nivolumab in Combination for the Treatment of Rare Gastrointestinal, Neuro-Endocrine and Gynecological Cancers | NCT02923934 | Single Arm | CBR | N/A |
| Modular Phase 2 Study to Link Combination Immune-Therapy to Patients with Advanced Solid and Hematologic Malignancies. Module 9: PDR001 Plus LAG525 for Patients with Advanced Solid and Hematologic Malignancies. | NCT03365791 | Single Arm | CBR; PFS | N/A |
| Randomized Phase II Trial of Durvalumab (MEDI4736) and Tremelimumab Administered in Combination versus Sequentially in Recurrent Platinum-Resistant Epithelial Ovarian Cancer | NCT03026062 | Arm 1: Sequential Tremelimumab + Durvalumab Arm 2: Combination Tremelimumab + Durvalumab | irPFS | N/A |
| Study Title | Trial Identifier | Study Arm(s) | Primary Endpoint(s) | Results |
|---|---|---|---|---|
| A Phase I Study of Concomitant WT1 Analog Peptide Vaccine with Montanide and GM-CSF in Combination with Nivolumab in Patients with Recurrent Ovarian Cancer Who Are in Second or Greater Remission | NCT02737787 | Single Arm | DLT | 1-year PFS: 64.0% [115] |
| A Phase 1a/1b Study of Cabiralizumab in Combination with Nivolumab in Patients with Selected Advanced Cancers | NCT02526017 | Arm 1: Cabiralizumab Arm 2: Cabiralizumab + Nivolumab | AEs; SAEs; RD; ORR | N/A |
| Phase Ib Trial of Pembrolizumab Administered in Combination with or Following Adoptive Cell Therapy—A Multiple Cohort Study; the ACTIVATE (Adoptive Cell Therapy InVigorated to Augment Tumor Eradication) Trial | NCT03158935 | Arm 1: Cyclophosphamide and fludarabine followed by TILs, IL-2, and Pembrolizumab (Advanced metastatic melanoma) Arm 2: Cyclophosphamide followed by TILs, IL-2, and Pembrolizumab (Advanced ovarian cancer) | SAEs | N/A |
| A Prospective Study of Hyperthermia Combined with Autologous Adoptive Cellular Immunotherapy in the Treatment of Abdominal and Pelvic Malignancies or Metastases | NCT03757858 | Arm 1: HT + ACT Arm 2: HT + ACT + Pembrolizumab Arm 3: HT + ACT + CT Arm 4: HT + CT | AEs; ORR | ORR: 30.0% DCR: 66.7% [113] |
| T-Cell Therapy in Combination with Checkpoint Inhibitors for Patients with Advanced Ovarian, Fallopian Tube and Primary Peritoneal Cancer | NCT03287674 | Single Arm | AEs | N/A |
| A Phase I/II Dose Escalation and Cohort Expansion Study of the Safety, Tolerability and Efficacy of Anti-CD27 Antibody (Varlilumab) Administered in Combination with Anti-PD-1 (Nivolumab) in Advanced Refractory Solid Tumors | NCT02335918 | Single Arm | Phase I: AEs Phase II: ORR; OS | PR:3(1 EOC)/36 patients SD: 11/36 patients [116] |
| Phase 1/2 Study of Chemoimmunotherapy with Toll-like Receptor 8 Agonist Motolimod (VTX-2337) + Anti-PD-L1 Antibody MEDI4736 in Subjects with Recurrent, Platinum-Resistant Ovarian Cancer for Whom Pegylated Liposomal Doxorubicin is Indicated | NCT02431559 | Arm 1: Durvalumab + PLD + motolimod dose 0a Arm 2: Durvalumab + PLD + motolimod dose 0b Arm 3: Durvalumab + PLD + motolimod dose level 1 Arm 4: Durvalumab + PLD | AEs; PFS | Arm 1: SAEs: 0.0% AEs: 100.0% PFS at 6 months: 33.3% PFS: 5.6 mo. CR: 0.0% PR: 33.3% SD: 33.3% PD: 33.3% Arm 2: SAEs: 50.0% AEs: 100.0% PFS at 6 months: 50.0% PFS: 5.7 mo. CR: 0.0% PR: 25.0% SD: 25.0% PD: 50.0% Arm 3: SAEs: 16.7% AEs: 100.0% PFS at 6 mo.: 33.3% PFS: 4.3 mo. CR: 0.0% PR: 33.3% SD: 16.7% PD: 50.0% Arm 4: SAEs: 57.5% AEs: 100.0% PFS at 6 mo.: 42.9% PFS: 5.5 mo. CR: 5.0% PR: 10.0% SD: 45.0% PD: 40.0% |
| Phase 1/2a Study of Double-Immune Suppression Blockade by Combining a CSF1R Inhibitor (PLX3397) with an Anti-PD-1 Antibody (Pembrolizumab) to Treat Advanced Melanoma and Other Solid Tumors | NCT02452424 | Single Arm | AEs | Study Terminated |
| A Phase II Trial of TPIV200/huFR-1 (A Multi-Epitope Anti-Folate Receptor Vaccine) Plus Anti-PD-L1 MEDI4736 (Durvalumab) in Patients with Platinum Resistant Ovarian Cancer | NCT02764333 | Single Arm | ORR | Acceptable Combinatorial Safety Profile (114) |
| A Randomized, Intra-Patient Crossover, Safety, Biomarker and Anti-Tumor Activity Assessment of the Combination of Atezolizumab and Vigil in Patients with Advanced Gynecological Cancers | NCT03073525 | Arm 1: Vigil + Atezolizumab Arm 2: Vigil and Vigil + Atezolizumab Arm 3: Atezolizumab and Vigil + Atezolizumab Arm 4: Atezolizumab | AEs | N/A |
| Pilot Study of Durvalumab (MEDI4736) in Combination with Vigil in Advanced Women’s Cancers | NCT02725489 | Arm 1: 1 × 106 cells Vigil Arm 2: 1 × 107 cells Vigil Arm 3: 1 × 105 cells Vigil | AEs | N/A |
| A Phase II Study Using Short-Term Cultured, Autologous Tumor-Infiltrating Lymphocytes Following a Lymphodepleting Regimen in Metastatic Cancers Plus the Administration of Pembrolizumab | NCT01174121 | Arm 1: CD8 + TILs + Aldesleukin + Cyclophosphamide + Fludarabine Arm 2: Unselected TILs + Aldesleukin+ Cyclophosphamide + Fludarabine Arm 3: Unselected TILs + Pembrolizumab + Cyclophosphamide Arm 4: Unselected TILs + Cyclophosphamide + Aldesleukin_ Fludarabine + Pembrolizumab | Response rate | N/A |
| Study Title | Trial Identifier | Study Arm(s) | Primary Endpoint(s) | Results |
|---|---|---|---|---|
| A Phase I Dose Finding Study of Oral LXH254 in Adult Patients with Advanced Solid Tumors Harboring MAPK Pathway Alterations | NCT02607813 | Arm 1: Dose escalation LXH254 Arm 2–4: Dose expansion LXH254 Arm 5: Dose expansion LXH254 + PDR001 Arm 6: Dose escalation LXH254 + PDR001 | AEs; DLT | N/A |
| A Phase 1A/B Study to Evaluate the Safety and Tolerability of ETC-1922159 in Advanced Solid Tumors | NCT02521844 | Arm 1: ETC-1922159 + Pembrolizumab Arm 2: ETC-1922159 until progression, then Pembrolizumab added | MTD; RD; AEs | N/A |
| A Randomized, Placebo-Controlled, Double-Blind, Multicenter Phase 1b/2 Study of Avelumab with or without Entinostat in Patients with Advanced Epithelial Ovarian Cancer which has Progressed or Recurred after First-Line Platinum-Based Chemotherapy and at Least Two Subsequent Lines of Treatment with a Safety Lead-In | NCT02915523 | Arm 1: Entinostat + Avelumab Arm 2: Avelumab | AEs; DLT; MTD /RP2D | PFS: 1.64 mo. (Arm 1) 1.51 mo. (Arm 2) AEs (any grade): 93% (Arm 1) 78% Arm (2) [128] |
| A Phase 1b/2 Study to Evaluate the Safety, Tolerability and Pharmacokinetics of Mirvetuximab Soravtansine (IMGN853) in Combination with Bevacizumab, Carboplatin, Pegylated Liposomal Doxorubicin, Pembrolizumab, or Bevacizumab + Carboplatin in Adults with Folate Receptor Alpha Positive Advanced Epithelial Ovarian Cancer, Primary Peritoneal Cancer or Fallopian Tube Cancer | NCT02606305 | Arm 1: Dose escalation and dose expansion IMGN853 +Bevacizumab Arm 2: Dose escalation IMGN853 + Carboplatin Arm 3: Dose escalation IMGN853 + PLD Arm 4: Dose escalation and dose expansion IMGN853 + Pembrolizumab Arm 5: Dose expansion IMGN853 + Bevacizumab + Carboplatin | TEAEs; SAEs; DLT; ORR | IMGN853 Combinations safe and tolerable [129] |
| A Phase 1/2 Study Exploring the Safety, Tolerability, and Efficacy of MK-3475 in Combination with INCB024360 in Subjects with Selected Cancers (ECHO-202/KEYNOTE-037) | NCT02178722 | Single Arm | AEs; ORR | ORR: EOC: 8.1% |
| Phase II Trial of Concurrent Anti-PD-L1 and SAbR for Patients with Persistent or Recurrent Epithelial Ovarian, Primary Peritoneal or Fallopian Tube Cancer (with Safety Lead-In) | NCT03312114 | Single Arm | ORR | Study Terminated |
| A Phase 2 Proof-of-Concept Study of ACP-196 Alone and in Combination with Pembrolizumab in Subjects with Recurrent Ovarian Cancer | NCT02537444 | Arm 1: Acalabrutinib Arm 2: Acalabrutinib + Pembrolizumab | OR | OR: 2.9% (Arm1) 9.1% (Arm 2) SAEs: 21.05% (Arm 1) 41.03% (Arm 2) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
James, N.E.; Woodman, M.; DiSilvestro, P.A.; Ribeiro, J.R. The Perfect Combination: Enhancing Patient Response to PD-1-Based Therapies in Epithelial Ovarian Cancer. Cancers 2020, 12, 2150. https://doi.org/10.3390/cancers12082150
James NE, Woodman M, DiSilvestro PA, Ribeiro JR. The Perfect Combination: Enhancing Patient Response to PD-1-Based Therapies in Epithelial Ovarian Cancer. Cancers. 2020; 12(8):2150. https://doi.org/10.3390/cancers12082150
Chicago/Turabian StyleJames, Nicole E., Morgan Woodman, Paul A. DiSilvestro, and Jennifer R. Ribeiro. 2020. "The Perfect Combination: Enhancing Patient Response to PD-1-Based Therapies in Epithelial Ovarian Cancer" Cancers 12, no. 8: 2150. https://doi.org/10.3390/cancers12082150
APA StyleJames, N. E., Woodman, M., DiSilvestro, P. A., & Ribeiro, J. R. (2020). The Perfect Combination: Enhancing Patient Response to PD-1-Based Therapies in Epithelial Ovarian Cancer. Cancers, 12(8), 2150. https://doi.org/10.3390/cancers12082150




