Cross-Cultural Validation of the Italian Version of the Bt-DUX: A Subjective Measure of Health-Related Quality of Life in Patients Who Underwent Surgery for Lower Extremity Malignant Bone Tumour
Abstract
1. Introduction
2. Method
2.1. Translation, Adaptation, and Validation
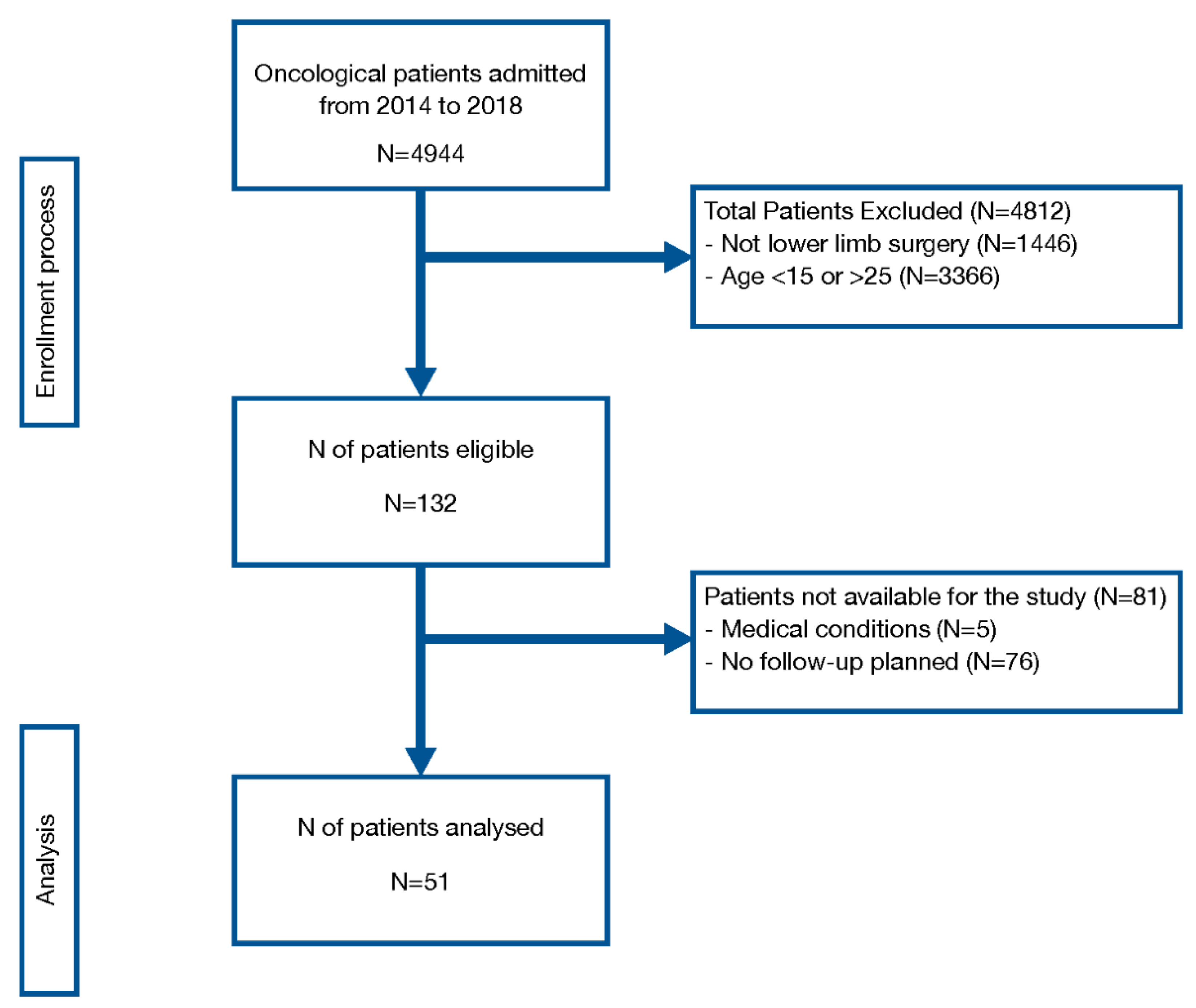
2.2. Patients
2.3. Assessment
2.4. Statistical Analysis
2.5. Sample Size
3. Result
3.1. The Translation Process
3.2. Patient Characteristics
3.3. Internal Consistency
3.4. Construct Validity
3.5. Discriminant Validity
4. Discussion
5. Conclusion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- AIRTUM 2012. Epidemiol. Prev. 2013, 37 (Suppl. 1), pp. 1–296. Available online: https://www.registri-tumori.it/cms/pubblicazioni/i-tumori-italia-rapporto-2012-i-tumori-dei-bambini-e-degli-adolescenti (accessed on 17 May 2020).
- Han, G.; Wang, Y.; Bi, W.Z. Study on the health-related quality of life in patients after surgery for malignant bone tumors. Asian Pac. J. Cancer Prev. 2012, 13, 127–130. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Stokke, J.; Sung, L.; Gupta, A.; Lindber, A.; Rosenberg, A.R. Systematic review and meta-analysis of objective and subjective quality of life among pediatric, adolescent and young adult bone tumor survivors. Pediatr. Blood Cancer 2015, 62, 1616–1629. [Google Scholar] [CrossRef] [PubMed]
- Singh, V.A.; Heng, C.W.; Yasin, N.F. Gait Analysis in Patients with Wide Resection and Endoprosthesis Replacement around the Knee. Indian J. Orthop. 2018, 52, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Goryn, T.; Pienkowski, A.; Szostakowski, B.; Zdzienicki, M.; Ługowska, I.; Rutkowski, P. Functional outcome of surgical treatment of adults with extremity osteosarcoma after megaprosthetic reconstruction-single-center experience. J. Orthop. Surg. Res. 2019, 14, 346. [Google Scholar] [CrossRef] [PubMed]
- Frances, J.M.; Morris, C.D.; Arkader, A.; Nikolic, Z.G.; Healey, J.H. What is quality of life in children with bone sarcoma? Clin. Orthop. Relat. Res. 2007, 459, 34–39. [Google Scholar] [CrossRef]
- Tabone, M.D.; Rodary, C.; Oberlin, O.; Gentet, J.C.; Pacquement, H.; Kalifa, C. Quality of life of patients treated during childhood for a bone tumor: Assessment by the Child Health Questionnaire. Pediatr. Blood Cancer 2005, 45, 207–211. [Google Scholar] [CrossRef]
- Ruggieri, P.; Mavrogenis, A.F.; Mercuri, M. Quality of life following limb-salvage surgery for bone sarcomas. Expert Rev. Pharmacoecon Outcomes Res. 2011, 11, 59–73. [Google Scholar] [CrossRef]
- Aaronson, N.K.; Ahmedzai, S.; Bergman, B.; Bullinger, M.; Cull, A.; Duez, N.J.; Filiberti, A.; Flechtner, H.; Fleishman, S.B.; de Haes, J.C.; et al. The European Organization for Research and Treatment of Cancer QLQ-C30: A quality-of-life instrument for use in international clinical trials in oncology. J. Natl. Cancer Inst. 1993, 85, 365–376. [Google Scholar] [CrossRef]
- Akahane, T.; Shimizu, T.; Isobe, K.; Yoshimura, Y.; Fujioka, F.; Kato, H. Evaluation of postoperative general quality of life for patients with osteosarcoma around the knee joint. J. Pediatr. Orthop. B 2007, 16, 269–272. [Google Scholar] [CrossRef]
- Bekkering, W.P.; Vliet Vlieland, T.P.; Fiocco, M.; Koopman, H.M.; Schoones, J.W.; Nelissen, R.G.; Taminiau, A.H. Quality of life, functional ability and physical activity after different surgical interventions for bone cancer of the leg: A systematic review. Surg. Oncol. 2012, 21, e39–e47. [Google Scholar] [CrossRef]
- Bekkering, W.P.; Vlieland, T.P.; Koopman, H.M.; Schaap, G.R.; Schreuder, H.W.; Beishuizen, A.; Tissing, W.J.; Hoogerbrugge, P.M.; Anninga, J.K.; Taminiau, A.H. The Bt-DUX: Development of a subjective measure of health-related quality of life in patients who underwent surgery for lower extremity malignant bone tumor. Pediatr. Blood Cancer 2009, 53, 348–355. [Google Scholar] [CrossRef] [PubMed]
- Bekkering, W.P.; Billing, L.; Grimer, R.J.; Vlieland, T.P.; Koopman, H.M.; Nelissen, R.G.; Taminiau, A.H. Translation and preliminary validation of the English version of the DUX questionnaire for lower extremity bone tumor patients (Bt-DUX): A disease-specific measure for quality of life. J. Surg. Oncol. 2013, 107, 353–359. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Hu, A.; Zhang, M.; Shi, C.; Zhang, X.; Zhang, J. Correlation between functional status and quality of life after surgery in patients with primary malignant bone tumor of the lower extremities. Orthop. Nurs. 2014, 33, 163–170. [Google Scholar] [CrossRef] [PubMed]
- Bekkering, W.P.; Vliet Vlieland, T.P.; Koopman, H.M.; Schaap, G.R.; Beishuizen, A.; Anninga, J.K.; Wolterbeek, R.; Nelissen, R.G.; Taminiau, A.H. A prospective study on quality of life and functional outcome in children and adolescents after malignant bone tumor surgery. Pediatr. Blood Cancer 2012, 58, 978–985. [Google Scholar] [CrossRef] [PubMed]
- Bekkering, W.P.; van Egmond-van Dam, J.C.; Bramer, J.A.M.; Beishuizen, A.; Fiocco, M.; Dijkstra, P.D.S. Quality of life after bone sarcoma surgery around the knee: A long-term follow-up study. Eur. J. Cancer Care (Engl.) 2017, 26, e12603. [Google Scholar] [CrossRef] [PubMed]
- Beaton, D.E.; Bombardier, C.; Guillemin, F.; Ferraz, M.B. Guidelines for the process of cross cultural adaptation of self-report measures. Spine 2000, 24, 3186–3191. [Google Scholar] [CrossRef]
- Guillemin, F.; Bombardier, C.; Beaton, D. Cross-cultural adaptation of health-related quality of life measures: Literature review and proposed guidelines. J. Clin. Epidemiol. 1993, 46, 1417–1432. [Google Scholar] [CrossRef]
- Wild, D.; Grove, A.; Martin, M.; Eremenco, S.; McElroy, S.; Verjee-Lorenz, A.; Erikson, P. ISPOR Task Force for Translation and Cultural Adaptation. Principles of Good Practice for the Translation and Cultural Adaptation Process for Patient-Reported Outcomes (PRO) Measures: Report of the ISPOR Task Force for Translation and Cultural Adaptation. Value Health 2005, 8, 94–104. [Google Scholar] [CrossRef]
- Lucisano, P.; Piemontese, M.E. Gulpease: A formula to predict readability of texts written in Italian Language. Scuola Città 1988, 39. (in Italian). [Google Scholar]
- Kincaid, J.P.; Fishburne, R.P., Jr.; Rogers, R.L.; Chissom, B.S. Derivation of New Readability Formulas (Automated Readability Index, Fog Count and Flesch Reading Ease Formula) for Navy Enlisted Personnel. Institute for Simulation and Training. 1975. Available online: https://stars.library.ucf.edu/istlibrary/56 (accessed on 20 July 2020).
- Davis, A.M.; Wright, J.G.; Williams, J.I.; Bombardier, C.; Griffin, A.; Bell, R.S. Development of a measure of physical function for patients with bone and soft tissue sarcoma. Qual. Life Res. 1996, 5, 508–516. [Google Scholar] [CrossRef]
- Guilford, J.P. Fundamental Statistics in Psychology and Education; McGraw Hill Book Company: New York, NY, USA, 1956. [Google Scholar]
- Akiyama, T.; Uehara, K.; Ogura, K.; Shinoda, Y.; Iwata, S.; Saita, K.; Tanzawa, Y.; Nakatani, F.; Yonemoto, T.; Kawano, H.; et al. Cross-cultural adaptation and validation of the Japanese version of the Toronto Extremity Salvage Score (TESS) for patients with malignant musculoskeletal tumors in the upper extremities. J. Orthop Sci. 2017, 22, 127–132. [Google Scholar] [CrossRef]
- Saraiva, D.; de Camargo, B.; Davis, A.M. Cultural adaptation, translation and validation of a functional outcome questionnaire (TESS) to Portuguese with application to patients with lower extremity osteosarcoma. Pediatr. Blood Cancer 2008, 50, 1039–1042. [Google Scholar] [CrossRef] [PubMed]
- Morattel, B.; Mustaki, L.; Montemurro, M.; Letovanec, I.; Durham, A.D.; Becce, F.; Omoumi, P.; di Summa, P.G.; Matter, M.; Rüdiger, H.A.; et al. Oncological outcome, functional results and costs after unplanned excision of musculoskeletal soft tissue sarcoma. Eur. J. Surg. Oncol. 2020, 46, 898–904. [Google Scholar] [CrossRef] [PubMed]
- Kask, G.; Barner-Rasmussen, I.; Repo, J.P.; Kjäldman, M.; Kilk, K.; Blomqvist, C.; Tukiainen, E. Functional Outcome Measurement in Patients with Lower-Extremity Soft Tissue Sarcoma: A Systematic Literature Review. Ann. Surg Oncol. 2019, 26, 4707–4722. [Google Scholar] [CrossRef] [PubMed]
- Oh, E.; Seo, S.W.; Han, K.J. A longitudinal study of functional outcomes in patients with limb salvage surgery for soft tissue sarcoma. Sarcoma 2018, 12, 6846275. [Google Scholar] [CrossRef]
| Characteristics | N = 51 |
|---|---|
| Age, years, median (min-max) | 20 (15–25) |
| Follow-up, months, median (min-max) | 38 (14–60) |
| Sex, male (%) | 37 (72.5) |
| Diagnosis | |
| Osteosarcoma, n (%) Ewing sarcoma, n (%) Other, n (%) | 28 (54.9) |
| 18 (35.3) | |
| 5 (9.8) | |
| Surgery | |
| Prosthesis, n (%) | 21 (41.2) |
| Bone graft, n (%) | 23 (45.1) |
| Amputation, n (%) | 4 (7.8) |
| Resection, n (%) | 3 (5.9) |
| Site of the tumour | |
| Pelvis, n (%) Femur, n (%) Tibia, n (%) Foot, n (%) | 3 (5.9) |
| 29 (56.9) | |
| 17 (33.3) | |
| 2 (3.9) | |
| Outcome Measure | Median (min–max) (0–100) |
|---|---|
| EORTC | |
| QoL, General Health QoL, Function QoL, Symptom | 83.3 (33.3–100) |
| 88.9 (48.9–100) | |
| 7.7 (0–38.5) | |
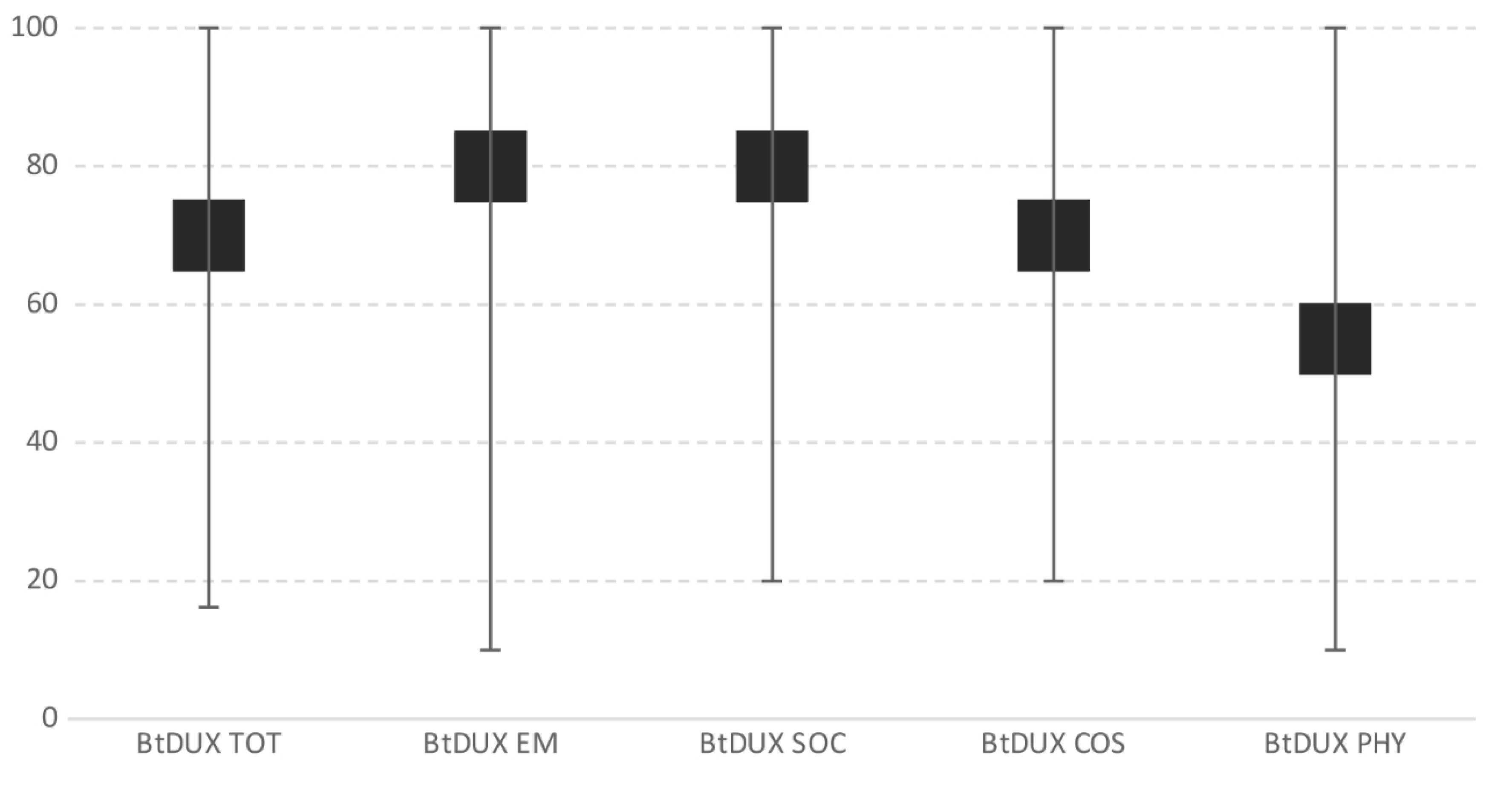
| Bt-DUX-It | |
| Total score Emotional Social Cosmetic Physical | 70 (16.25–100) |
| 80 (10–100) | |
| 80 (20–100) | |
| 70 (20–100) | |
| 55 (10–100) | |
| TESS | 89.2 (38–100) |
| Bt-DUX-It | EORTC QLQ General Health | EORTC QLQ Function | EORTC QLQ Symptom | TESS |
|---|---|---|---|---|
| Total score | 0.68 ** | 0.70 ** | −0.56 ** | 0.76 ** |
| Emotional | 0.66 ** | 0.63 ** | −0.52 ** | 0.60 ** |
| Social | 0.70 ** | 0.63 ** | −0.55 ** | 0.55 ** |
| Cosmetic | 0.59 ** | 0.60 ** | −0.47 ** | 0.64 ** |
| Physical | 0.54 ** | 0.69 ** | −0.49 ** | 0.79 ** |
| Bt-DUX-It | TESS < 89 N = 25 | TESS > 89 N = 26 | p-Value |
|---|---|---|---|
| Total score | 63.8 (16.2–90) | 83.8 (62.5–100) | <0.001 |
| Emotional | 65 (10–100) | 90 (60–100) | 0.001 |
| Social | 75 (20–100) | 85 (55–100) | 0.005 |
| Cosmetic | 60 (20–100) | 85 (55–100) | <0.001 |
| Physical | 40 (10–75) | 77.5 (35–100) | <0.001 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Morri, M.; Bekkering, P.W.; Cotti, M.; Meneghini, M.; Venturini, E.; Longhi, A.; Mariani, E.; Forni, C. Cross-Cultural Validation of the Italian Version of the Bt-DUX: A Subjective Measure of Health-Related Quality of Life in Patients Who Underwent Surgery for Lower Extremity Malignant Bone Tumour. Cancers 2020, 12, 2015. https://doi.org/10.3390/cancers12082015
Morri M, Bekkering PW, Cotti M, Meneghini M, Venturini E, Longhi A, Mariani E, Forni C. Cross-Cultural Validation of the Italian Version of the Bt-DUX: A Subjective Measure of Health-Related Quality of Life in Patients Who Underwent Surgery for Lower Extremity Malignant Bone Tumour. Cancers. 2020; 12(8):2015. https://doi.org/10.3390/cancers12082015
Chicago/Turabian StyleMorri, Mattia, Peter Willem Bekkering, Marco Cotti, Matilde Meneghini, Enrico Venturini, Alessandra Longhi, Elisabetta Mariani, and Cristiana Forni. 2020. "Cross-Cultural Validation of the Italian Version of the Bt-DUX: A Subjective Measure of Health-Related Quality of Life in Patients Who Underwent Surgery for Lower Extremity Malignant Bone Tumour" Cancers 12, no. 8: 2015. https://doi.org/10.3390/cancers12082015
APA StyleMorri, M., Bekkering, P. W., Cotti, M., Meneghini, M., Venturini, E., Longhi, A., Mariani, E., & Forni, C. (2020). Cross-Cultural Validation of the Italian Version of the Bt-DUX: A Subjective Measure of Health-Related Quality of Life in Patients Who Underwent Surgery for Lower Extremity Malignant Bone Tumour. Cancers, 12(8), 2015. https://doi.org/10.3390/cancers12082015





