Reconstitution of T Cell Subsets Following Allogeneic Hematopoietic Cell Transplantation
Abstract
1. Introduction
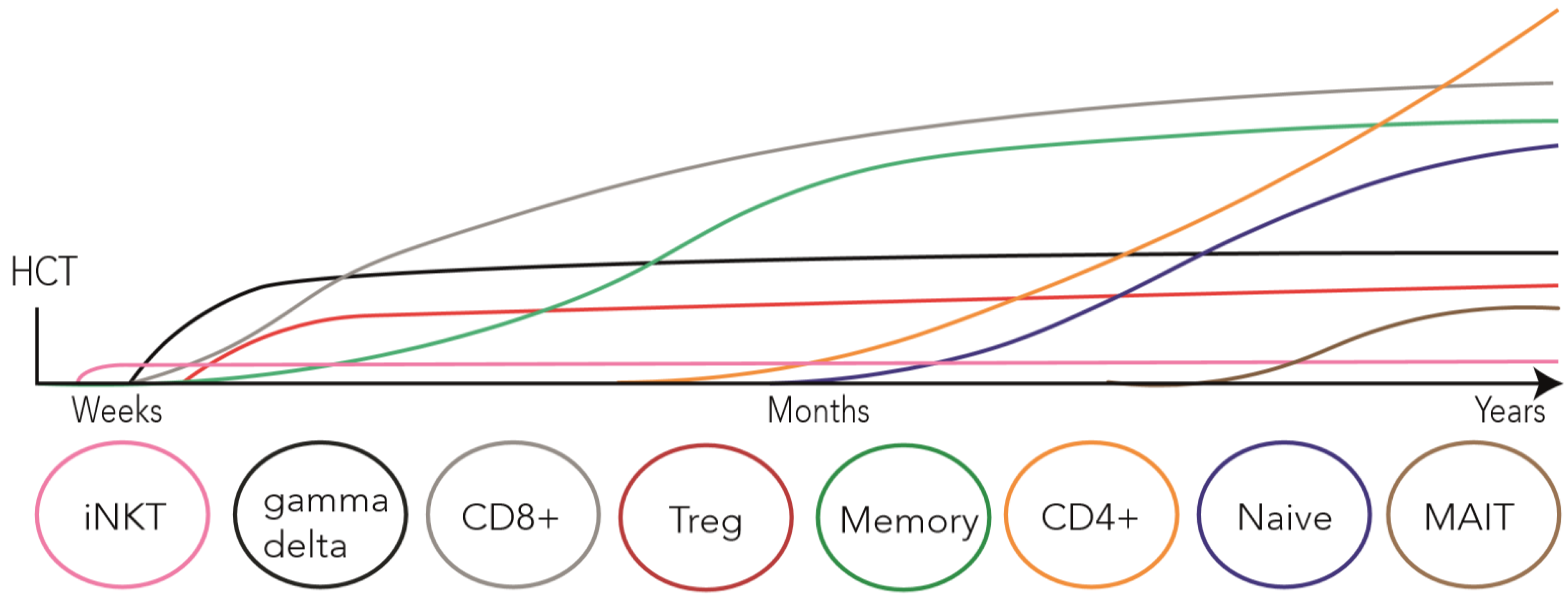
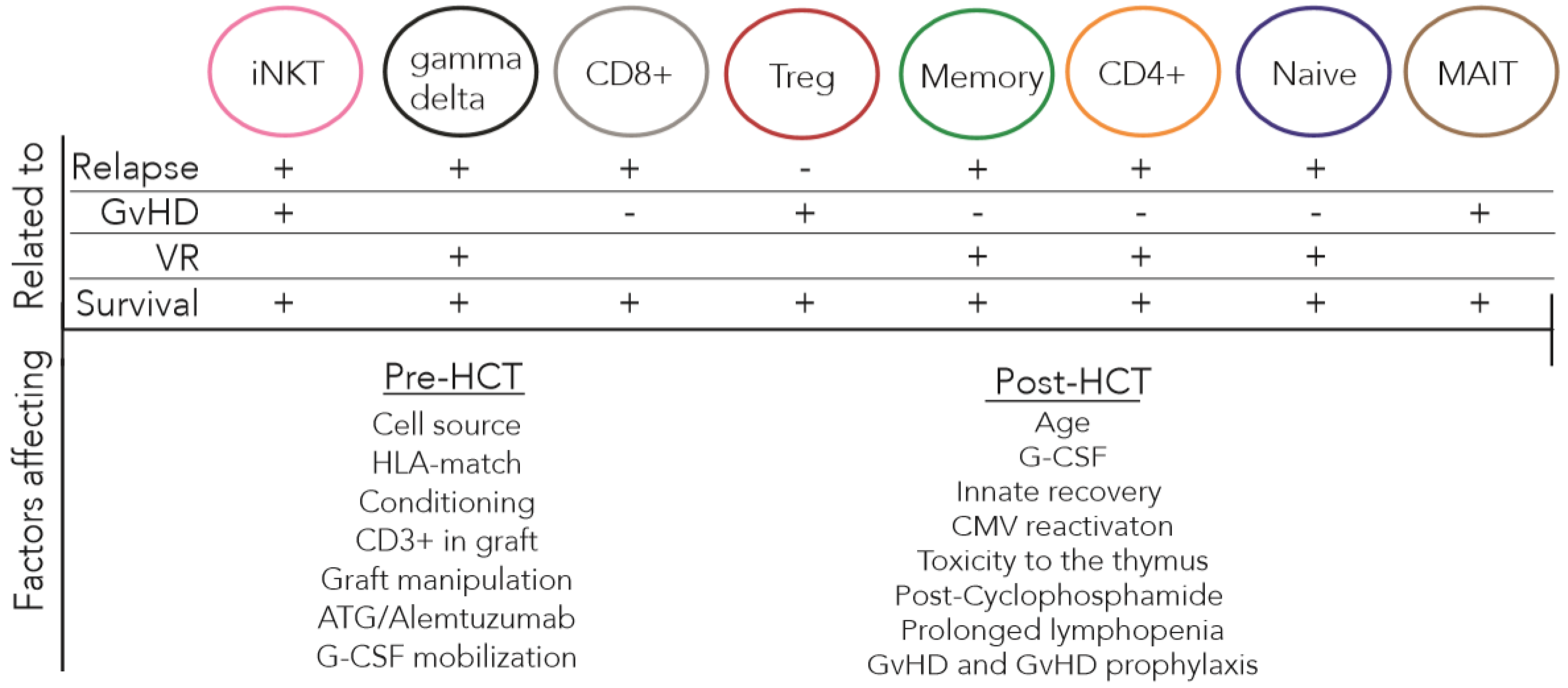
2. T Cell Reconstitution after allo-HCT
3. CD4+ T Cells
3.1. Th Cells
3.2. Tregs
4. CD8+ T Cells
5. γδ T Cells
6. MAIT Cells
7. iNKT Cells
8. Naive T Cells
9. Memory T Cells
10. Concluding Remarks and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- de Koning, C.; Plantinga, M.; Besseling, P.; Boelens, J.J.; Nierkens, S. Immune Reconstitution after Allogeneic Hematopoietic Cell Transplantation in Children. Biol. Blood Marrow Transpl. 2016, 22, 195–206. [Google Scholar] [CrossRef]
- Bartelink, I.H.; Belitser, S.V.; Knibbe, C.A.J.; Danhof, M.; de Pagter, A.J.; Egberts, T.C.G.; Boelens, J.J. Immune Reconstitution Kinetics as an Early Predictor for Mortality using Various Hematopoietic Stem Cell Sources in Children. Biol. Blood Marrow Transpl. 2013, 19, 305–313. [Google Scholar] [CrossRef]
- Fedele, R.; Martino, M.; Garreffa, C.; Messina, G.; Console, G.; Princi, D.; Dattola, A.; Moscato, T.; Massara, E.; Spiniello, E.; et al. The impact of early CD4+ lymphocyte recovery on the outcome of patients who undergo allogeneic bone marrow or peripheral blood stem cell transplantation. Blood Transfus. 2012, 10, 174–180. [Google Scholar] [PubMed]
- Kim, D.H.; Sohn, S.K.; Won, D.I.; Lee, N.Y.; Suh, J.S.; Lee, K.B. Rapid helper T-cell recovery above 200 × 106/l at 3 months correlates to successful transplant outcomes after allogeneic stem cell transplantation. Bone Marrow Transplant. 2006, 37, 1119–1128. [Google Scholar] [CrossRef] [PubMed]
- Komanduri, K.V.; St. John, L.S.; De Lima, M.; McMannis, J.; Rosinski, S.; McNiece, I.; Bryan, S.G.; Kaur, I.; Martin, S.; Wieder, E.D.; et al. Delayed immune reconstitution after cord blood transplantation is characterized by impaired thymopoiesis and late memory T-cell skewing. Blood 2007, 110, 4543–4551. [Google Scholar] [PubMed]
- Lindemans, C.A.; Chiesa, R.; Amrolia, P.J.; Rao, K.; Nikolajeva, O.; De Wildt, A.; Gerhardt, C.E.; Gilmour, K.C.; Bierings, M.B.; Veys, P.; et al. Impact of thymoglobulin prior to pediatric unrelated umbilical cord blood transplantation on immune reconstitution and clinical outcome. Blood 2014, 123, 126–132. [Google Scholar] [CrossRef]
- Bosch, M.; Dhadda, M.; Hoegh-Petersen, M.; Liu, Y.; Hagel, L.M.; Podgorny, P.; Ugarte-Torres, A.; Khan, F.M.; Luider, J.; Auer-Grzesiak, I.; et al. Immune Reconstitution After Antithymocyte Globulin-Conditioned Hematopoietic Cell Transplantation. Bone 2011, 23, 1–7. [Google Scholar] [CrossRef]
- Törlén, J.; Gaballa, A.; Remberger, M.; Mörk, L.M.; Sundberg, B.; Mattsson, J.; Uhlin, M. Effect of Graft-versus-Host Disease Prophylaxis Regimens on T and B Cell Reconstitution after Allogeneic Hematopoietic Stem Cell Transplantation. Biol. Blood Marrow Transpl. 2019, 25, 1260–1268. [Google Scholar]
- Bae, K.W.; Kim, B.E.; Koh, K.N.; Im, H.J.; Seo, J.J. Factors influencing lymphocyte reconstitution after allogeneic hematopoietic stem cell transplantation in children. Korean J. Hematol. 2012, 47, 44–52. [Google Scholar] [CrossRef]
- Park, B.G.; Park, C.-J.; Jang, S.; Chi, H.-S.; Kim, D.-Y.; Lee, J.-H.; Lee, J.-H.; Lee, K.-H. Reconstitution of lymphocyte subpopulations after hematopoietic stem cell transplantation: Comparison of hematologic malignancies and donor types in event-free patients. Leuk. Res. 2015, 39, 1334–1341. [Google Scholar] [CrossRef]
- Petersen, S.L.; Ryder, L.P.; Björk, P.; Madsen, H.O.; Heilmann, C.; Jacobsen, N.; Sengeløv, H.; Vindeløv, L.L. A comparison of T-, B- and NK-cell reconstitution following conventional or nonmyeloablative conditioning and transplantation with bone marrow or peripheral blood stem cells from human leucocyte antigen identical sibling donors. Bone Marrow Transplant. 2003, 32, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Williams, K.; Hakim, F.T.; Gress, R.E. T-cell reconstitution following lymphodepletion. Brain. Behav. Immun. 2008, 22, 629. [Google Scholar] [CrossRef]
- Admiraal, R.; Chiesa, R.; Bierings, M.; Versluijs, A.B.; Hiwarkar, P.; Silva, J.; Veys, P.; Boelens, J. Early CD4+ Immune Reconstitution Predicts Probability of Relapse in Pediatric AML after Unrelated Cord Blood Transplantation: Importance of Preventing in Vivo T-Cell Depletion Using Thymoglobulin®. Biol. Blood Marrow Transpl. 2015, 21, S206. [Google Scholar] [CrossRef]
- Parkman, R.; Cohen, G.; Carter, S.L.; Weinberg, K.I.; Masinsin, B.; Guinan, E.; Kurtzberg, J.; Wagner, J.E.; Kernan, N.A. Successful Immune Reconstitution Decreases Leukemic Relapse and Improves Survival in Recipients of Unrelated Cord Blood Transplantation. Boil. Blood Marrow Transpl. 2006, 12, 919–927. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Bühlmann, L.; Buser, A.S.; Cantoni, N.; Gerull, S.; Tichelli, A.; Gratwohl, A.; Stern, M. Lymphocyte subset recovery and outcome after T-cell replete allogeneic hematopoietic SCT. Bone Marrow Transplant. 2010, 46, 1357–1362. [Google Scholar] [CrossRef] [PubMed]
- Khandelwal, P.; Lane, A.; Chaturvedi, V.; Owsley, E.; Davies, S.M.; Marmer, D.; Filipovich, A.H.; Jordan, M.B.; Marsh, R.A. Peripheral Blood CD38 Bright CD8+ Effector Memory T Cells Predict Acute Graft-versus-Host Disease. Biol. Blood Marrow Transpl. 2015, 21, 1215–1222. [Google Scholar] [CrossRef] [PubMed]
- Oshrine, B.R.; Li, Y.; Teachey, D.T.; Heimall, J.; Barrett, D.M.; Bunin, N. Immunologic recovery in children after alternative donor allogeneic transplantation for hematologic malignancies: Comparison of recipients of partially T cell-depleted peripheral blood stem cells and umbilical cord blood. Biol. Blood Marrow Transpl. 2013, 19, 1581–1589. [Google Scholar] [CrossRef]
- Matsuoka, K.; Kim, H.T.; Mcdonough, S.; Bascug, G.; Warshauer, B.; Koreth, J.; Cutler, C.; Ho, V.T.; Alyea, E.P.; Antin, J.H.; et al. Altered regulatory T cell homeostasis in patients with CD4+ lymphopenia following allogeneic hematopoietic stem cell transplantation. J. Clin. Investig. 2010, 120, 1479–1493. [Google Scholar] [CrossRef]
- Mackall, C.L.; Fleisher, T.A.; Brown, M.R.; Andrich, M.P.; Chen, C.C.; Feuerstein, I.M.; Horowitz, M.E.; Magrath, I.T.; Shad, A.T.; Steinberg, S.M.; et al. Age, thymopoiesis, and CD4+ T-lymphocyte regeneration after intensive chemotherapy. N. Engl. J. Med. 1995, 332, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Moutuou, M.M.; Page, G.; Zaid, I.; Lesage, S.; Guimond, M. Restoring T Cell Homeostasis After Allogeneic Stem Cell Transplantation; Principal Limitations and Future Challenges. Front. Immunol. 2018, 9, 1237. [Google Scholar] [CrossRef]
- Castermans, E.; Hannon, M.; Dutrieux, J.; Humblet-Baron, S.; Seidel, L.; Cheynier, R.; Willems, E.; Gothot, A.; Vanbellinghen, J.-F.; Geenen, V.; et al. Thymic recovery after allogeneic hematopoietic cell transplantation with non-myeloablative conditioning is limited to patients younger than 60 years of age. Haematol. 2010, 96, 298–306. [Google Scholar] [CrossRef] [PubMed]
- Moretta, A.; Maccario, R.; Fagioli, F.; Giraldi, E.; Busca, A.; Montagna, D.; Miniero, R.; Comoli, P.; Giorgiani, G.; Zecca, M.; et al. Analysis of immune reconstitution in children undergoing cord blood transplantation. Exp. Hematol. 2001, 29, 371–379. [Google Scholar] [CrossRef]
- Talvensaari, K.; Clave, E.; Douay, C.; Rabian, C.; Garderet, L.; Busson, M.; Garnier, F.; Douek, D.; Gluckman, E.; Charron, D.; et al. A broad T-cell repertoire diversity and an efficient thymic function indicate a favorable long-term immune reconstitution after cord blood stem cell transplantation. Blood 2002, 99, 1458–1464. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Xu, L. Evaluation of TCR repertoire diversity in patients after hematopoietic stem cell transplantation. Stem Cell Investig. 2015, 2, 17. [Google Scholar] [PubMed]
- Clave, E.; Lisini, D.; Douay, C.; Giorgiani, G.; Busson, M.; Zecca, M.; Moretta, F.; Acquafredda, G.; Brescia, L.P.; Locatelli, F.; et al. Thymic function recovery after unrelated donor cord blood or T-cell depleted HLA-haploidentical stem cell transplantation correlates with leukemia relapse. Front. Immunol. 2013, 4, 25. [Google Scholar] [CrossRef] [PubMed]
- Gkazi, A.S.; Margetts, B.K.; Attenborough, T.; Mhaldien, L.; Standing, J.F.; Oakes, T.; Heather, J.M.; Booth, J.; Pasquet, M.; Chiesa, R.; et al. Clinical T cell receptor repertoire deep sequencing and analysis: An application to monitor immune reconstitution following cord blood transplantation. Front. Immunol. 2018, 9, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Ph, D.; Weisdorf, D.J.; Wingard, J.R.; Cutler, C.S.; Johnston, L.; Maziarz, R.T.; Pulsipher, M.A.; Bredeson, C.; Carter, S.L.; Sc, D.; et al. Peripheral-Blood Stem Cells versus Bone Marrow from Unrelated Donors. N. Engl. J. Med. 2013, 367, 1–16. [Google Scholar]
- Hiwarkar, P.; Qasim, W.; Ricciardelli, I.; Gilmour, K.; Quezada, S.A.; Saudemont, A.; Amrolia, P.; Veys, P. Cord blood T cells mediate enhanced antitumor effects compared with adult peripheral blood T cells. Blood 2015, 126, 2882–2891. [Google Scholar] [CrossRef]
- Mold, J.E.; Venkatasubrahmanyam, S.; Burt, T.D.; Michaëlsson, J.; Rivera, J.M.; Galkina, S.A.; Weinberg, K.; Stoddart, C.A.; McCune, J.M. Fetal and adult hematopoietic stem cells give rise to distinct T cell lineages in humans. Science 2010, 330, 1695–1699. [Google Scholar] [CrossRef]
- Azevedo, R.I.; Soares, M.V.D.; Albuquerque, A.S.; Tendeiro, R.; Soares, R.S.; Martins, M.; Ligeiro, D.; Victorino, R.M.M.; Lacerda, J.F.; Sousa, A.E. Long-term immune reconstitution of naive and memory t cell pools after haploidentical hematopoietic stem cell transplantation. Biol. Blood Marrow Transpl. 2013, 19, 703–712. [Google Scholar] [CrossRef][Green Version]
- Tian, D.M.; Wang, Y.; Zhang, X.H.; Liu, K.Y.; Huang, X.J.; Chang, Y.J. Rapid recovery of CD3+CD8+ T cells on day 90 predicts superior survival after unmanipulated haploidentical blood and marrow transplantation. PLoS ONE 2016, 11, e0156777. [Google Scholar] [CrossRef] [PubMed]
- Simonetta, F.; Alvarez, M.; Negrin, R.S. Natural killer cells in graft-versus-host-disease after allogeneic hematopoietic cell transplantation. Front. Immunol. 2017, 8, 465. [Google Scholar] [CrossRef]
- Minculescu, L.; Marquart, H.V.; Ryder, L.P.; Andersen, N.S.; Schjoedt, I.; Friis, L.S.; Kornblit, B.T.; Petersen, S.L.; Haastrup, E.; Fischer-Nielsen, A.; et al. Improved Overall Survival, Relapse-Free-Survival, and Less Graft-vs.-Host-Disease in Patients With High Immune Reconstitution of TCR Gamma Delta Cells 2 Months after Allogeneic Stem Cell Transplantation. Front. Immunol. 2019, 10, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Airoldi, I.; Bertaina, A.; Prigione, I.; Zorzoli, A.; Pagliara, D.; Cocco, C.; Meazza, R.; Loiacono, F.; Lucarelli, B.; Bernardo, M.E.; et al. γδ T-cell reconstitution after HLA-haploidentical hematopoietic transplantation depleted of TCR-αβ+/CD19+ lymphocytes. Blood 2015, 125, 2349–2358. [Google Scholar] [CrossRef]
- Jimbo, K.; Konuma, T.; Watanabe, E.; Kohara, C.; Mizukami, M.; Nagai, E.; Oiwa-Monna, M.; Mizusawa, M.; Isobe, M.; Kato, S.; et al. T memory stem cells after allogeneic haematopoietic cell transplantation: Unique long-term kinetics and influence of chronic graft-versus-host disease. Br. J. Haematol. 2019, 186, 866–878. [Google Scholar] [CrossRef] [PubMed]
- Storek, J.; Joseph, A.; Espino, G.; Dawson, M.A.; Douek, D.C.; Sullivan, K.M.; Flowers, M.E.D.; Martin, P.; Mathioudakis, G.; Nash, R.A.; et al. Immunity of patients surviving 20 to 30 years after allogeneic or syngeneic bone marrow transplantation. Blood 2001, 98, 3505–3512. [Google Scholar] [CrossRef]
- Ishaqi, M.K.; Afzal, S.; Dupuis, A.; Doyle, J.; Gassas, A. Early lymphocyte recovery post-allogeneic hematopoietic stem cell transplantation is associated with significant graft-versus-leukemia effect without increase in graft-versus-host disease in pediatric acute lymphoblastic leukemia. Bone Marrow Transplant. 2008, 41, 245–252. [Google Scholar] [CrossRef]
- Fu, J.; Heinrichs, J.; Yu, X.-Z. Helper T-Cell Differentiation in Graft-Versus-Host Disease After Allogeneic Hematopoietic Stem Cell Transplantation. Archivum Immunologiae et Therapiae Experimentalis 2014, 62, 277–301. [Google Scholar] [CrossRef]
- Admiraal, R.; de Koning, C.C.H.; Lindemans, C.A.; Bierings, M.B.; Wensing, A.M.J.; Versluys, A.B.; Wolfs, T.F.W.; Nierkens, S.; Boelens, J.J. Viral reactivations and associated outcomes in the context of immune reconstitution after pediatric hematopoietic cell transplantation. J. Allergy Clin. Immunol. 2017, 140, 1643–1650. [Google Scholar] [CrossRef]
- Admiraal, R.; Lindemans, C.A.; Van Kesteren, C.; Bierings, M.B.; Versluijs, A.B.; Nierkens, S.; Boelens, J.J.; Versluys, A.B. Excellent T-cell reconstitution and survival depend on low ATG exposure after pediatric cord blood transplantation. Blood 2016, 128, 2734–2741. [Google Scholar] [CrossRef] [PubMed]
- Chiesa, R.; Gilmour, K.; Qasim, W.; Adams, S.; Worth, A.J.J.; Zhan, H.; Montiel-Equihua, C.A.; Derniame, S.; Cale, C.; Rao, K.; et al. Omission of in vivo T-cell depletion promotes rapid expansion of naïve CD4+ cord blood lymphocytes and restores adaptive immunity within 2 months after unrelated cord blood transplant. Br. J. Haematol. 2012, 156, 656–666. [Google Scholar] [CrossRef] [PubMed]
- De Koning, C.; Gabelich, J.-A.; Langenhorst, J.; Admiraal, R.; Kuball, J.; Boelens, J.J.; Nierkens, S. Filgrastim enhances T-cell clearance by antithymocyte globulin exposure after unrelated cord blood transplantation. Blood Adv. 2018, 2, 565–574. [Google Scholar] [CrossRef] [PubMed]
- Bejanyan, N.; Brunstein, C.G.; Cao, Q.; Lazaryan, A.; Luo, X.; Curtsinger, J.; Mehta, R.S.; Warlick, E.; Cooley, S.A.; Blazar, B.R.; et al. Delayed immune reconstitution after allogeneic transplantation increases the risks of mortality and chronic GVHD. Blood Adv. 2018, 2, 909–922. [Google Scholar] [CrossRef] [PubMed]
- Admiraal, R.; Van Kesteren, C.; Der Zijde, E.C.J.-V.; Lankester, A.C.; Bierings, M.B.; Egberts, T.; Van Tol, M.J.D.; Knibbe, C.A.J.; Bredius, R.G.M.; Boelens, J.J. Association between anti-thymocyte globulin exposure and CD4+ immune reconstitution in paediatric haemopoietic cell transplantation: A multicentre, retrospective pharmacodynamic cohort analysis. Lancet Haematol. 2015, 2, e194–e203. [Google Scholar] [CrossRef]
- Waller, E.K.; Logan, B.R.; Fei, M.; Lee, S.J.; Confer, D.; Howard, A.; Chandrakasan, S.; Anasetti, C.; Fernando, S.M.; Giver, C.R. Kinetics of immune cell reconstitution predict survival in allogeneic bone marrow and G-CSF–mobilized stem cell transplantation. Blood Adv. 2019, 3, 2250–2263. [Google Scholar] [CrossRef]
- Hoare, R.L.; Veys, P.; Klein, N.; Callard, R.; Standing, J.F. Predicting CD4 T-Cell Reconstitution Following Pediatric Hematopoietic Stem Cell Transplantation. Clin. Pharmacol. Ther. 2017, 102, 349–357. [Google Scholar] [CrossRef]
- Eyrich, M.; Leiler, C.; Lang, P.; Schilbach, K.; Schumm, M.; Bader, P.; Greil, J.; Klingebiel, T.; Handgretinger, R.; Niethammer, D.; et al. A prospective comparison of immune reconstitution in pediatric recipients of positively selected CD34+ peripheral blood stem cells from unrelated donors vs recipients of unmanipulated bone marrow from related donors. Bone Marrow Transplant. 2003, 32, 379–390. [Google Scholar] [CrossRef]
- Koehl, U.; Bochennek, K.; Zimmermann, S.Y.; Lehrnbecher, T.; Sörensen, J.; Esser, R.; Andreas, C.; Kramm, C.; Grüttner, H.P.; Falkenberg, E.; et al. Immune recovery in children undergoing allogeneic stem cell transplantation: Absolute CD8+CD3+ count reconstitution is associated with survival. Bone Marrow Transplant. 2007, 39, 269–278. [Google Scholar] [CrossRef]
- Politikos, I.; Lavery, J.A.; Hilden, P.; Cho, C.; Borrill, T.; Maloy, M.A.; Giralt, S.A.; Brink, M.R.M.V.D.; Perales, M.-A.; Barker, J.N. Robust CD4+ T-cell recovery in adults transplanted with cord blood and no antithymocyte globulin. Blood Adv. 2020, 4, 191–202. [Google Scholar] [CrossRef]
- Soiffer, R.J.; Kim, H.T.; McGuirk, J.; Horwitz, M.; Johnston, L.; Patnaik, M.M.; Rybka, W.; Artz, A.; Porter, D.L.; Shea, T.C.; et al. Prospective, Randomized, Double-Blind, Phase III Clinical Trial of Anti–T-Lymphocyte Globulin to Assess Impact on Chronic Graft-Versus-Host Disease–Free Survival in Patients Undergoing HLA-Matched Unrelated Myeloablative Hematopoietic Cell Transplantation. J. Clin. Oncol. 2017, 35, 4003–4011. [Google Scholar] [CrossRef]
- Xhaard, A.; Moins-Teisserenc, H.; Busson, M.; Robin, M.; Ribaud, P.; Dhedin, N.; Abbes, S.; Carmagnat, M.; Kheav, V.D.; Maki, G.; et al. Reconstitution of regulatory T-cell subsets after allogeneic hematopoietic SCT. Bone Marrow Transplant. 2014, 49, 1089–1092. [Google Scholar] [CrossRef]
- Pearl, J.P.; Parris, J.; Hale, D.A.; Hoffmann, S.C.; Bernstein, W.B.; McCoy, K.L.; Swanson, S.J.; Mannon, R.B.; Roederer, M.; Kirk, A. Immunocompetent T-Cells with a Memory-Like Phenotype are the Dominant Cell Type Following Antibody-Mediated T-Cell Depletion. Arab. Archaeol. Epigr. 2005, 5, 465–474. [Google Scholar] [CrossRef] [PubMed]
- De Koning, C.; Langenhorst, J.; Van Kesteren, C.; Lindemans, C.A.; Huitema, A.D.; Nierkens, S.; Boelens, J.J. Innate Immune Recovery Predicts CD4+ T Cell Reconstitution after Hematopoietic Cell Transplantation. Boil. Blood Marrow Transpl. 2019, 25, 819–826. [Google Scholar] [CrossRef] [PubMed]
- Ding, L.; Zhu, H.; Yang, Y.; Yan, H.-M.; Zhang, H.-H.; Han, D.-M.; Wang, Z.-D.; Zheng, X.-L.; Liu, J.; Zhu, L.; et al. The absolute number of regulatory T cells in unmanipulated peripheral blood grafts predicts the occurrence of acute graft-versus-host disease post haplo-identical hematopoietic stem cell transplantation. Leuk. Res. 2017, 56, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Kanakry, C.G.; Ganguly, S.; Zahurak, M.; Bolaños-Meade, J.; Thoburn, C.; Perkins, B.; Fuchs, E.J.; Jones, R.J.; Hess, A.D.; Luznik, L. Aldehyde Dehydrogenase Expression Drives Human Regulatory T Cell Resistance to Posttransplantation Cyclophosphamide. Sci. Transl. Med. 2013, 5, 201ra119. [Google Scholar] [CrossRef]
- Nunes, N.S.; Kanakry, C.G. Mechanisms of Graft-versus-Host Disease Prevention by Post-transplantation Cyclophosphamide: An Evolving Understanding. Front. Immunol. 2019, 10, 2668. [Google Scholar] [CrossRef]
- Reubsaet, L.L.; De Pagter, A.P.J.; Van Baarle, D.; Keukens, L.; Nanlohy, N.; Sanders, E.A.M.; Prakken, B.J.; Boelens, J.J.; De Kleer, I.M. Stem cell source-dependent reconstitution of FOXP3+ T cells after pediatric SCT and the association with allo-reactive disease. Bone Marrow Transplant. 2012, 48, 502–507. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Watanabe, N.; Narita, M.; Furukawa, T.; Nakamura, T.; Yamahira, A.; Masuko, M.; Toba, K.; Fuse, I.; Aizawa, Y.; Takahashi, M. Kinetics of pDCs, mDCs, γδT cells and regulatory T cells in association with graft versus host disease after hematopoietic stem cell transplantation. Int. J. Lab. Hematol. 2011, 33, 378–390. [Google Scholar] [CrossRef] [PubMed]
- Wu, K.N.; Emmons, R.V.; Lisanti, M.P.; Farber, J.L.; Witkiewicz, A.K. Foxp3-expressing T regulatory cells and mast cells in acute graft-versus-host disease of the skin. Cell Cycle 2009, 8, 3601–3605. [Google Scholar] [CrossRef]
- Charrier, E.; Cordeiro, P.; Brito, R.-M.; Mezziani, S.; Herblot, S.; Le Deist, F.; Duval, M. Reconstitution of maturating and regulatory lymphocyte subsets after cord blood and BMT in children. Bone Marrow Transplant. 2012, 48, 376–382. [Google Scholar] [CrossRef]
- Magenau, J.M.; Qin, X.; Tawara, I.; Rogers, C.E.; Kitko, C.; Schlough, M.; Bickley, D.; Braun, T.M.; Jang, P.-S.; Lowler, K.P.; et al. Frequency of CD4+CD25hiFOXP3+ Regulatory T Cells Has Diagnostic and Prognostic Value as a Biomarker for Acute Graft-versus-Host-Disease. Boil. Blood Marrow Transpl. 2010, 16, 907–914. [Google Scholar] [CrossRef]
- Rezvani, K.; Mielke, S.; Ahmadzadeh, M.; Kilical, Y.; Savani, B.N.; Zeilah, J.; Keyvanfar, K.; Montero, A.; Hensel, N.; Kurlander, R.; et al. High donor FOXP3-positive regulatory T-cell (Treg) content is associated with a low risk of GVHD following HLA-matched allogeneic SCT. Blood 2006, 108, 1291–1297. [Google Scholar] [CrossRef]
- Lu, S.; Liu, K.; Liu, D.-H.; Xu, L.; Huang, X.-J. High frequencies of CD62L+ naive regulatory T cells in allografts are associated with a low risk of acute graft-versus-host disease following unmanipulated allogeneic haematopoietic stem cell transplantation. Clin. Exp. Immunol. 2011, 165, 264–277. [Google Scholar] [CrossRef] [PubMed]
- Soares, M.V.; Azevedo, R.I.; Ferreira, I.A.; Bucar, S.; Ribeiro, A.C.; Vieira, A.; Pereira, P.N.G.; Ribeiro, R.M.; Ligeiro, D.; Alho, A.C.; et al. Naive and Stem Cell Memory T Cell Subset Recovery Reveals Opposing Reconstitution Patterns in CD4 and CD8 T Cells in Chronic Graft vs. Host Disease. Front. Immunol. 2019, 10, 334. [Google Scholar] [CrossRef] [PubMed]
- Alho, A.C.; Kim, H.T.; Chammas, M.J.; Reynolds, C.G.; Matos, T.R.; Forcade, E.; Whangbo, J.; Nikiforow, S.; Cutler, C.S.; Koreth, J.; et al. Unbalanced recovery of regulatory and effector T cells after allogeneic stem cell transplantation contributes to chronic GVHD. Blood 2016, 127, 646–657. [Google Scholar] [CrossRef] [PubMed]
- Jacopo, P.; Forcina, A.; Clerici, D.; Crocchiolo, R.; Vago, L.; Stanghellini, M.T.L.; Noviello, M.; Messina, C.; Crotta, A.; Assanelli, A.; et al. Sirolimus-based graft-versus-host disease prophylaxis promotes the in vivo expansion of regulatory T cells and permits peripheral blood stem cell transplantation from haploidentical donors. Leukemia 2014, 29, 396–405. [Google Scholar]
- Furukawa, A.; Wisel, S.A.; Tang, Q. Impact of Immune-Modulatory Drugs on Regulatory T Cell. Transplantation 2016, 100, 2288–2300. [Google Scholar] [CrossRef]
- Wachsmuth, L.P.; Patterson, M.; Eckhaus, M.A.; Venzon, D.J.; Gress, R.E.; Kanakry, C.G. Posttransplantation cyclophosphamide prevents graft-versus-host disease by inducing alloreactive T cell dysfunction and suppression. J. Clin. Investig. 2019, 129, 2357–2373. [Google Scholar] [CrossRef]
- Rocha, V.; Wagner, J.E.; Sobocinski, K.A.; Klein, J.P.; Zhang, M.-J.; Horowitz, M.M.; Gluckman, E. Graft-Versus-Host Disease in Children Who Have Received a Cord-Blood or Bone Marrow Transplant from an HLA-Identical Sibling. N. Engl. J. Med. 2000, 342, 1846–1854. [Google Scholar] [CrossRef]
- Di Ianni, M.; Falzetti, F.; Carotti, A.; Terenzi, A.; Castellino, F.; Bonifacio, E.; Del Papa, B.; Zei, T.; Ostini, R.I.; Cecchini, D.; et al. Tregs prevent GVHD and promote immune reconstitution in HLA-haploidentical transplantation. Blood 2011, 117, 3921–3928. [Google Scholar] [CrossRef]
- Trovillion, E.M.; Gloude, N.J.; Anderson, E.J.; Morris, G.P. Relationship of post-transplant thymopoiesis with CD4+FoxP3+ regulatory T cell recovery associated with freedom from chronic graft versus host disease. Bone Marrow Transplant. 2019, 54, 917–920. [Google Scholar] [CrossRef] [PubMed]
- Mittrücker, H.-W.; Visekruna, A.; Huber, M. Heterogeneity in the Differentiation and Function of CD8+ T Cells. Archivum Immunologiae et Therapiae Experimentalis 2014, 62, 449–458. [Google Scholar] [CrossRef] [PubMed]
- Stikvoort, A.; Gaballa, A.; Solders, M.; Nederlof, I.; Önfelt, B.; Sundberg, B.; Remberger, M.; Sundin, M.; Mattsson, J.; Uhlin, M. Risk Factors for Severe Acute Graft-versus-Host Disease in Donor Graft Composition. Boil. Blood Marrow Transpl. 2018, 24, 467–477. [Google Scholar] [CrossRef] [PubMed]
- Stikvoort, A.; Chen, Y.; Rådestad, E.; Törlén, J.K.; Lakshmikanth, T.; Björklund, A.; Mikes, J.; Achour, A.; Gertow, J.; Sundberg, B.; et al. Combining Flow and Mass Cytometry in the Search for Biomarkers in Chronic Graft-versus-Host Disease. Front. Immunol. 2017, 8. [Google Scholar] [CrossRef] [PubMed]
- Klein, A.K.; Patel, D.D.; Gooding, M.E.; Sempowski, G.D.; Chen, B.J.; Liu, C.; Kurtzberg, F.; Haynes, B.F.; Chao, N.J. T-cell recovery in adults and children following umbilical cord blood transplantation. Boil. Blood Marrow Transpl. 2001, 7, 454–466. [Google Scholar] [CrossRef]
- Kanakry, C.G.; Coffey, D.; Towlerton, A.M.; Vulic, A.; Storer, B.E.; Chou, J.; Yeung, C.; Gocke, C.D.; Robins, H.S.; O’Donnell, P.V.; et al. Origin and evolution of the T cell repertoire after posttransplantation cyclophosphamide. JCI Insight 2016, 1, 86252. [Google Scholar] [CrossRef] [PubMed]
- Ogonek, J.; Varanasi, P.; Luther, S.; Schweier, P.; Kühnau, W.; Göhring, G.; Dammann, E.; Stadler, M.; Ganser, A.; Borchers, S.; et al. Possible Impact of Cytomegalovirus-Specific CD8 + T Cells on Immune Reconstitution and Conversion to Complete Donor Chimerism after Allogeneic Stem Cell Transplantation. Boil. Blood Marrow Transpl. 2017, 23, 1046–1053. [Google Scholar] [CrossRef] [PubMed]
- Suessmuth, Y.; Mukherjee, R.; Watkins, B.; Koura, D.T.; Finstermeier, K.; Desmarais, C.; Stempora, L.; Horan, J.T.; Langston, A.; Qayed, M.; et al. CMV reactivation drives posttransplant T-cell reconstitution and results in defects in the underlying TCRβ repertoire. Blood 2015, 125, 3835–3850. [Google Scholar] [CrossRef]
- Jacobson, C.A.; Turki, A.T.; McDonough, S.M.; Stevenson, K.E.; Kim, H.T.; Kao, G.; Herrera, M.I.; Reynolds, C.G.; Alyea, E.P.; Ho, V.T.; et al. Immune Reconstitution after Double Umbilical Cord Blood Stem Cell Transplantation: Comparison with Unrelated Peripheral Blood Stem Cell Transplantation. Boil. Blood Marrow Transpl. 2012, 18, 565–574. [Google Scholar] [CrossRef]
- Scheper, W.; Van Dorp, S.; Kersting, S.; Pietersma, F.; Lindemans, C.; Hol, S.; Heijhuurs, S.; Sebestyen, Z.; Gründer, C.; Marcu-Malina, V.; et al. γδT cells elicited by CMV reactivation after allo-SCT cross-recognize CMV and leukemia. Leukemia 2013, 27, 1328–1338. [Google Scholar] [CrossRef]
- Handgretinger, R.; Schilbach, K. The potential role of gd T cells after allogeneic HCT for leukemia. Blood 2018, 131, 1063–1072. [Google Scholar] [CrossRef] [PubMed]
- Cela, M.E.; Holladay, M.S.; Rooney, C.M.; Richardson, S.; Alexander, B.; Krance, R.A.; Brenner, M.K.; Heslop, H.E. γδ T lymphocyte regeneration after T lymphocyte-depleted bone marrow transplantation from mismatched family members or matched unrelated donors. Bone Marrow Transplant. 1996, 17, 243–247. [Google Scholar] [PubMed]
- Godder, K.T.; Henslee-Downey, P.J.; Mehta, J.; Park, B.S.; Chiang, K.-Y.; Abhyankar, S.; Lamb, L.S. Long term disease-free survival in acute leukemia patients recovering with increased γδ T cells after partially mismatched related donor bone marrow transplantation. Bone Marrow Transplant. 2007, 39, 751–757. [Google Scholar] [CrossRef] [PubMed]
- Perko, R.; Kang, G.; Sunkara, A.; Leung, W.; Thomas, P.G.; Dallas, M.H. Gamma Delta T cell Reconstitution is Associated with Fewer Infections and Improved Event Free Survival following Hematopoietic Stem Cell Transplantation for Pediatric Leukemia Gamma Delta T Cells after HSCT Ross. Biol. Blood Marrow Transpl. 2015, 21, 130–136. [Google Scholar] [CrossRef] [PubMed]
- Lamb, L.; Gee, A.P.; Hazlett, L.J.; Musk, P.; Parrish, R.S.; O’Hanlon, T.P.; Geier, S.S.; Folk, R.S.; Harris, W.G.; McPherson, K.; et al. Influence of T cell depletion method on circulating gammadelta T cell reconstitution and potential role in the graft-versus-leukemia effect. Cytotherapy 1999, 1, 7–19. [Google Scholar] [CrossRef]
- Hirokawa, M.; Horiuchi, T.; Kawabata, Y.; Kitabayashi, A.; Miura, A.B. Reconstitution of γδ T cell repertoire diversity after human allogeneic hematopoietic cell transplantation and the role of peripheral expansion of mature T cell population in the graft. Bone Marrow Transplant. 2000, 26, 177–185. [Google Scholar] [CrossRef]
- Ravens, S.; Schultze-Florey, C.; Raha, S.; Sandrock, I.; Drenker, M.; Oberdörfer, L.; Reinhardt, A.; Ravens, I.; Beck, M.; Geffers, R.; et al. Human γδ T cells are quickly reconstituted after stem-cell transplantation and show adaptive clonal expansion in response to viral infection. Nat. Immunol. 2017, 18, 393–401. [Google Scholar] [CrossRef]
- Inagaki, J.; Noguchi, M.; Kurauchi, K.; Tanioka, S.; Fukano, R.; Okamura, J. Effect of Cytomegalovirus Reactivation on Relapse after Allogeneic Hematopoietic Stem Cell Transplantation in Pediatric Acute Leukemia. Boil. Blood Marrow Transpl. 2016, 22, 300–306. [Google Scholar] [CrossRef]
- Lukasik, Z.; Elewaut, D.; Venken, K. MAIT Cells Come to the Rescue in Cancer Immunotherapy? Cancers 2020, 12, 413. [Google Scholar] [CrossRef]
- Gold, M.; McLaren, J.E.; Reistetter, J.A.; Smyk-Pearson, S.; Ladell, K.; Swarbrick, G.M.; Yu, Y.Y.L.; Hansen, T.H.; Lund, O.; Nielsen, M.; et al. MR1-restricted MAIT cells display ligand discrimination and pathogen selectivity through distinct T cell receptor usage. J. Exp. Med. 2014, 211, 1601–1610. [Google Scholar] [CrossRef]
- Ussher, J.E.; Bilton, M.; Attwod, E.; Shadwell, J.; Richardson, R.; De Lara, C.; Mettke, E.; Kurioka, A.; Hansen, T.H.; Klenerman, P.; et al. CD161++CD8+T cells, including the MAIT cell subset, are specifically activated by IL-12+IL-18 in a TCR-independent manner. Eur. J. Immunol. 2013, 44, 195–203. [Google Scholar] [CrossRef] [PubMed]
- Konuma, T.; Kohara, C.; Watanabe, E.; Takahashi, S.; Ozawa, G.; Suzuki, K.; Mizukami, M.; Nagai, E.; Jimbo, K.; Kaito, Y.; et al. Reconstitution of Circulating Mucosal-Associated Invariant T Cells after Allogeneic Hematopoietic Cell Transplantation: Its Association with the Riboflavin Synthetic Pathway of Gut Microbiota in Cord Blood Transplant Recipients. J. Immunol. 2020, 204, 1462–1473. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharyya, A.; Hanafi, L.-A.; Sheih, A.; Golob, J.L.; Srinivasan, S.; Boeckh, M.J.; Pergam, S.A.; Mahmood, S.; Baker, K.K.; Gooley, T.A.; et al. Graft-Derived Reconstitution of Mucosal-Associated Invariant T Cells after Allogeneic Hematopoietic Cell Transplantation. Boil. Blood Marrow Transpl. 2017, 24, 242–251. [Google Scholar] [CrossRef] [PubMed]
- Kawaguchi, K.; Umeda, K.; Hiejima, E.; Iwai, A.; Mikami, M.; Nodomi, S.; Saida, S.; Kato, I.; Hiramatsu, H.; Yasumi, T.; et al. Influence of post-transplant mucosal-associated invariant T cell recovery on the development of acute graft-versus-host disease in allogeneic bone marrow transplantation. Int. J. Hematol. 2018, 108, 66–75. [Google Scholar] [CrossRef] [PubMed]
- Mengge, G.; Hong, Y.; Sun, Y.; Kong, J.; Yan, C.; Wang, Z.; Wang, Y.; Huang, X.; Zhao, X. The Low Number of Mucosal-Associated Invariant T Cells in the Graft Was Associated with Occurrence of Gut Graft-Versus-Host Disease. Blood 2019, 134, 2001. [Google Scholar] [CrossRef]
- Ben Youssef, G.; Tourret, M.; Salou, M.; Ghazarian, L.; Houdouin, V.; Mondot, S.; Mburu, Y.; Lambert, M.; Azarnoush, S.; Diana, J.-S.; et al. Ontogeny of human mucosal-associated invariant T cells and related T cell subsets. J. Exp. Med. 2018, 215, 459–479. [Google Scholar] [CrossRef]
- Turtle, C.J.; Delrow, J.; Joslyn, R.C.; Swanson, H.M.; Basom, R.; Tabellini, L.; Delaney, C.; Heimfeld, S.; Hansen, J.A.; Riddell, S.R. Innate signals overcome acquired TCR signaling pathway regulation and govern the fate of human CD161hi CD8α+ semi-invariant T cells. Blood 2011, 118, 2752–2762. [Google Scholar] [CrossRef]
- Solders, M.; Erkers, T.; Gorchs, L.; Poiret, T.; Remberger, M.; Magalhaes, I.; Kaipe, H. Mucosal-Associated Invariant T Cells Display a Poor Reconstitution and Altered Phenotype after Allogeneic Hematopoietic Stem Cell Transplantation. Front. Immunol. 2017, 8, 1861. [Google Scholar] [CrossRef]
- Wang, W.; Xu, S.; Ren, Z.; Jiang, J.; Zheng, S. Gut microbiota and allogeneic transplantation. J. Transl. Med. 2015, 13, 275. [Google Scholar] [CrossRef]
- Dias, J.; Boulouis, C.; Gorin, J.B.; van den Biggelaar, R.H.; Lal, K.G.; Gibbs, A.; Loh, L.; Gulam, M.Y.; Sia, W.R.; Bari, S.; et al. The CD4−CD8− MAIT cell subpopulation is a functionally distinct subset developmentally related to the main CD8+ MAIT cell pool. Proc. Natl. Acad. Sci. USA 2018, 115, E11513–E11522. [Google Scholar] [CrossRef]
- Haraguchi, K.; Takahashi, T.; Hiruma, K.; Kanda, Y.; Tanaka, Y.; Ogawa, S.; Chiba, S.; Miura, O.; Sakamaki, H.; Hirai, H. Recovery of Vα24+ NKT cells after hematopoietic stem cell transplantation. Bone Marrow Transplant. 2004, 34, 595–602. [Google Scholar] [CrossRef] [PubMed]
- Chaidos, A.; Patterson, S.; Szydlo, R.; Chaudhry, M.S.; Dazzi, F.; Kanfer, E.; McDonald, N.; Marin, D.; Milojkovic, D.; Pavlu, J.; et al. Graft invariant natural killer T-cell dose predicts risk of acute graft-versus-host disease in allogeneic hematopoietic stem cell transplantation. Blood 2012, 119, 5030–5036. [Google Scholar] [CrossRef] [PubMed]
- Rubio, M.-T.; Moreira-Teixeira, L.; Bachy, E.; Bouillié, M.; Milpied, P.; Coman, T.; Suarez, F.; Marçais, A.; Sibon, D.; Buzyn, A.; et al. Early posttransplantation donor-derived invariant natural killer T-cell recovery predicts the occurrence of acute graft-versus-host disease and overall survival. Blood 2012, 120, 2144–2154. [Google Scholar] [CrossRef] [PubMed]
- Malard, F.; Labopin, M.; Chevallier, P.; Guillaume, T.; Duquesne, A.; Rialland, F.; Derenne, S.; Peterlin, P.; Leauté, A.-G.; Brissot, E.; et al. Larger number of invariant natural killer T cells in PBSC allografts correlates with improved GVHD-free and progression-free survival. Blood 2016, 127, 1828–1835. [Google Scholar] [CrossRef] [PubMed]
- Rubio, M.-T.; Bouillié, M.; Bouazza, N.; Coman, T.; Trebeden-Nègre, H.; Gomez, A.; Suarez, F.; Sibon, D.; Brignier, A.; Paubelle, E.; et al. Pre-transplant donor CD4− invariant NKT cell expansion capacity predicts the occurrence of acute graft-versus-host disease. Leukemia 2016, 31, 903–912. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.W.; Park, S.-S.; Lim, J.-Y.; Min, G.J.; Park, S.; Jeon, Y.-W.; Yahng, S.-A.; Shin, S.-H.; Lee, S.-E.; Yoon, J.-H.; et al. Predictive Role of Circulating Immune Cell Subtypes Early after Allogeneic Hematopoietic Stem Cell Transplantation in Patients with Acute Leukemia. Int. J. Stem Cells 2018, 12, 73–83. [Google Scholar] [CrossRef]
- De Lalla, C.; Rinaldi, A.; Montagna, D.; Azzimonti, L.; Bernardo, M.E.; Sangalli, L.M.; Paganoni, A.M.; Maccario, R.; Di Cesare-Merlone, A.; Zecca, M.; et al. Invariant NKT Cell Reconstitution in Pediatric Leukemia Patients Given HLA-Haploidentical Stem Cell Transplantation Defines Distinct CD4+ and CD4− Subset Dynamics and Correlates with Remission State. J. Immunol. 2011, 186, 4490–4499. [Google Scholar] [CrossRef]
- Béziat, V.; Nguyen, S.; Exley, M.; Achour, A.; Simon, T.; Chevallier, P.; Sirvent, A.; Vigouroux, S.; Debre, P.; Rio, B.; et al. Shaping of iNKT cell repertoire after unrelated cord blood transplantation. Clin. Immunol. 2010, 135, 364–373. [Google Scholar] [CrossRef]
- Servais, S.; Menten-Dedoyart, C.; Beguin, Y.; Seidel, L.; Gothot, A.; Daulne, C.; Willems, É.; Delens, L.; Humblet-Baron, S.; Hannon, M.; et al. Impact of Pre-Transplant Anti-T Cell Globulin (ATG) on Immune Recovery after Myeloablative Allogeneic Peripheral Blood Stem Cell Transplantation. PLoS ONE 2015, 10, e0130026. [Google Scholar] [CrossRef]
- Broek, T.V.D.; Borghans, J.A.M.; Van Wijk, F. The full spectrum of human naive T cells. Nat. Rev. Immunol. 2018, 18, 363–373. [Google Scholar] [CrossRef]
- Ringhoffer, S.; Rojewski, M.; Döhner, H.; Bunjes, D.; Ringhoffer, M. T-cell reconstitution after allogeneic stem cell transplantation: Assessment by measurement of the sjTREC/βTREC ratio and thymic naïve T cells. Haematologica 2013, 98, 1600–1608. [Google Scholar] [CrossRef]
- Bleakley, M.; Heimfeld, S.; Loeb, K.R.; Jones, L.A.; Chaney, C.; Seropian, S.; Gooley, T.A.; Sommermeyer, F.; Riddell, S.R.; Shlomchik, W.D. Outcomes of acute leukemia patients transplanted with naive T cell-depleted stem cell grafts. J. Clin. Investig. 2015, 125, 2677–2689. [Google Scholar] [CrossRef] [PubMed]
- Ito, A.; Kitano, S.; Tajima, K.; Kim, Y.; Tanaka, T.; Inamoto, Y.; Kim, S.-W.; Yamamoto, N.; Fukuda, T.; Okamoto, S. Impact of low-dose anti-thymocyte globulin on immune reconstitution after allogeneic hematopoietic cell transplantation. Int. J. Hematol. 2019, 111, 120–130. [Google Scholar] [CrossRef] [PubMed]
- Shiratori, S.; Kosugi-Kanaya, M.; Hayase, E.; Okada, K.; Goto, H.; Sugita, J.; Onozawa, M.; Nakagawa, M.; Kahata, K.; Hashimoto, D.; et al. T-cell depletion effects of low-dose antithymocyte globulin for GVHD prophylaxis in HLA-matched allogeneic peripheral blood stem cell transplantation. Transpl. Immunol. 2018, 46, 21–22. [Google Scholar] [CrossRef] [PubMed]
- Hazenberg, M.D.; Otto, S.A.; De Pauw, E.S.; Roelofs, H.; Fibbe, W.E.; Hamann, D.; Miedema, F. T-cell receptor excision circle and T-cell dynamics after allogeneic stem cell transplantation are related to clinical events. Blood 2002, 99, 3449–3453. [Google Scholar] [CrossRef]
- Wils, E.-J.; Van Der Holt, B.; Broers, A.E.; Sluijs, S.J.P.-V.; Gratama, J.-W.; Braakman, E.; Cornelissen, J.J. Insufficient recovery of thymopoiesis predicts for opportunistic infections in allogeneic hematopoietic stem cell transplant recipients. Haematologica 2011, 96, 1846–1854. [Google Scholar] [CrossRef]
- Roberto, A.; Castagna, L.; Zanon, V.; Bramanti, S.; Crocchiolo, R.; McLaren, J.E.; Gandolfi, S.; Tentorio, P.; Sarina, B.; Timofeeva, I.; et al. Role of naive-derived T memory stem cells in T-cell reconstitution following allogeneic transplantation. Blood 2015, 125, 2855–2864. [Google Scholar] [CrossRef]
- Mensen, A.; Ochs, C.; Stroux, A.; Wittenbecher, F.; Szyska, M.; Imberti, L.; Fillatreau, S.; Uharek, L.; Renate, A.; Doerken, B.; et al. Utilization of TREC and KREC quantification for the monitoring of early T- and B-cell neogenesis in adult patients after allogeneic hematopoietic stem cell transplantation. J. Transl. Med. 2013, 11, 188. [Google Scholar] [CrossRef]
- Ruggeri, L.; Capanni, M.; Mancusi, A.; Perruccio, K.; Burchielli, E.; Martelli, M.F.; Velardi, A. Natural Killer Cell Alloreactivity in Haploidentical Hematopoietic Stem Cell Transplantation. Int. J. Hematol. 2005, 81, 13–17. [Google Scholar] [CrossRef]
- Pende, D.; Marcenaro, S.; Falco, M.; Martini, S.; Bernardo, M.E.; Montagna, D.; Romeo, E.; Cognet, C.; Martinetti, M.; Maccario, R.; et al. Anti-leukemia activity of alloreactive NK cells in KIR ligand-mismatched haploidentical HSCT for pediatric patients: Evaluation of the functional role of activating KIR and redefinition of inhibitory KIR specificity. Blood 2009, 113, 3119–3129. [Google Scholar] [CrossRef]
- Cieri, N.; Oliveira, G.; Greco, R.; Forcato, M.; Taccioli, C.; Cianciotti, B.C.; Valtolina, V.; Noviello, M.; Vago, L.; Bondanza, A.; et al. Generation of human memory stem T cells after haploidentical T-replete hematopoietic stem cell transplantation. Blood 2015, 125, 2865–2874. [Google Scholar] [CrossRef] [PubMed]
- Douek, D.C.; Vescio, R.A.; Betts, M.R.; Brenchley, J.M.; Hill, B.J.; Zhang, L.; Berenson, J.R.; Collins, R.H.; Koup, R.A. Assessment of thymic output in adults after haematopoietic stem-cell transplantation and prediction of T-cell reconstitution. Lancet 2020, 355, 1875–1881. [Google Scholar] [CrossRef]
- Junge, S.; Kloeckener-Gruissem, B.; Zufferey, R.; Keisker, A.; Salgo, B.; Fauchère, J.-C.; Scherer, F.; Shalaby, T.; Grotzer, M.; Siler, U.; et al. Correlation between recent thymic emigrants and CD31+ (PECAM-1) CD4+ T cells in normal individuals during aging and in lymphopenic children. Eur. J. Immunol. 2007, 37, 3270–3280. [Google Scholar] [CrossRef] [PubMed]
- De Koning, C.; Admiraal, R.; Nierkens, S.; Boelens, J.J. Human herpesvirus 6 viremia affects T-cell reconstitution after allogeneic hematopoietic stem cell transplantation. Blood Adv. 2018, 2, 428–432. [Google Scholar] [CrossRef] [PubMed]
- Triplett, B.M.; Shook, D.R.; Eldridge, P.; Li, Y.; Kang, G.; Dallas, M.; Hartford, C.; Srinivasan, A.; Chan, W.K.; Suwannasaen, D.; et al. Rapid memory T-cell reconstitution recapitulating CD45RA-depleted haploidentical transplant graft content in patients with hematologic malignancies. Bone Marrow Transplant. 2015, 50, 968–977. [Google Scholar] [CrossRef] [PubMed]
- Gattinoni, L.; Speiser, D.E.; Lichterfeld, M.; Bonini, C. T memory stem cells in health and disease. Nat. Med. 2017, 23, 18–27. [Google Scholar] [CrossRef]
- Oliveira, G.; Ruggiero, E.; Stanghellini, M.T.L.; Cieri, N.; D’Agostino, M.; Fronza, R.; Lulay, C.; Dionisio, F.; Mastaglio, S.; Greco, R.; et al. Tracking genetically engineered lymphocytes long-term reveals the dynamics of T cell immunological memory. Sci. Transl. Med. 2015, 7, 317ra198. [Google Scholar] [CrossRef]
- Marraco, S.A.F.; Soneson, C.; Cagnon, L.; Gannon, P.O.; Allard, M.; Maillard, S.A.; Montandon, N.; Rufer, N.; Waldvogel, S.; Delorenzi, M.; et al. Long-lasting stem cell–like memory CD8 + T cells with a naïve-like profile upon yellow fever vaccination. Sci. Transl. Med. 2015, 7, 282ra48. [Google Scholar] [CrossRef]
- Biasco, L.; Scala, S.; Ricci, L.B.; Dionisio, F.; Baricordi, C.; Calabria, A.; Giannelli, S.; Cieri, N.; Barzaghi, F.; Pajno, R.; et al. In vivo tracking of T cells in humans unveils decade-long survival and activity of genetically modified T memory stem cells. Sci. Transl. Med. 2015, 7, 273ra13. [Google Scholar] [CrossRef]
- Gossel, G.; Hogan, T.; Cownden, D.; Seddon, B.; Yates, A.J. Memory CD4 T cell subsets are kinetically heterogeneous and replenished from naive T cells at high levels. eLife 2017, 6. [Google Scholar] [CrossRef]
- Ahmed, R.; Roger, L.; Del Amo, P.C.; Miners, K.L.; Jones, R.E.; Boelen, L.; Fali, T.; Elemans, M.; Zhang, Y.; Appay, V.; et al. Human Stem Cell-like Memory T Cells Are Maintained in a State of Dynamic Flux. Cell Rep. 2016, 17, 2811–2818. [Google Scholar] [CrossRef]
- Salit, R.B.; Hakim, F.T.; Bishop, M.R.; Friedman, T.M.; Korngold, R.; Goldgirsh, K.; Memon, S.; Steinberg, S.M.; Liewehr, D.; Peaceman, D.; et al. Influence of Graft Versus-Host Disease Prophylaxis Regimen On T-Cell Repertoire Diversity Following Reduced-Intensity HLA-Matched Unrelated Donor Allogeneic Hematopoietic Stem Cell Transplantation. Blood 2012, 120, 3054. [Google Scholar] [CrossRef]
- Wilson, A.; Laurenti, E.; Oser, G.; Van Der Wath, R.C.; Blanco-Bose, W.; Jaworski, M.; Offner, S.; Dunant, C.F.; Eshkind, L.; Bockamp, E.; et al. Hematopoietic Stem Cells Reversibly Switch from Dormancy to Self-Renewal during Homeostasis and Repair. Cell 2008, 135, 1118–1129. [Google Scholar] [CrossRef] [PubMed]
- Osorio, F.G.; Huber, A.R.; Oka, R.; Verheul, M.; Patel, S.H.; Hasaart, K.; De La Fonteijne, L.; Varela, I.; Camargo, F.D.; Van Boxtel, R. Somatic Mutations Reveal Lineage Relationships and Age-Related Mutagenesis in Human Hematopoiesis. Cell Rep. 2018, 25, 2308–2316. [Google Scholar] [CrossRef] [PubMed]
- Brunstein, C.G.; Miller, J.S.; Cao, Q.; McKenna, D.H.; Hippen, K.L.; Curtsinger, J.; DeFor, T.; Levine, B.L.; June, C.H.; Rubinstein, P.; et al. Infusion of ex vivo expanded T regulatory cells in adults transplanted with umbilical cord blood: Safety profile and detection kinetics. Blood 2011, 117, 1061–1070. [Google Scholar] [CrossRef]
- Chen, Y.-B.; Efebera, Y.A.; Johnston, L.; Ball, E.D.; Avigan, D.; Lekakis, L.J.; Bachier, C.R.; Martin, P.; Duramad, O.; Ishii, Y.; et al. Increased Foxp3+Helios+ Regulatory T Cells and Decreased Acute Graft-versus-Host Disease after Allogeneic Bone Marrow Transplantation in Patients Receiving Sirolimus and RGI-2001, an Activator of Invariant Natural Killer T Cells. Boil. Blood Marrow Transpl. 2017, 23, 625–634. [Google Scholar] [CrossRef]
- Triplett, B.M.; Muller, B.; Kang, G.; Li, Y.; Cross, S.J.; Moen, J.; Cunningham, L.; Janssen, W.; Mamcarz, E.; Shook, D.R.; et al. Selective T-cell depletion targeting CD45RA reduces viremia and enhances early T-cell recovery compared with CD3-targeted T-cell depletion. Transpl. Infect. Dis. 2018, 20, e12823. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dekker, L.; de Koning, C.; Lindemans, C.; Nierkens, S. Reconstitution of T Cell Subsets Following Allogeneic Hematopoietic Cell Transplantation. Cancers 2020, 12, 1974. https://doi.org/10.3390/cancers12071974
Dekker L, de Koning C, Lindemans C, Nierkens S. Reconstitution of T Cell Subsets Following Allogeneic Hematopoietic Cell Transplantation. Cancers. 2020; 12(7):1974. https://doi.org/10.3390/cancers12071974
Chicago/Turabian StyleDekker, Linde, Coco de Koning, Caroline Lindemans, and Stefan Nierkens. 2020. "Reconstitution of T Cell Subsets Following Allogeneic Hematopoietic Cell Transplantation" Cancers 12, no. 7: 1974. https://doi.org/10.3390/cancers12071974
APA StyleDekker, L., de Koning, C., Lindemans, C., & Nierkens, S. (2020). Reconstitution of T Cell Subsets Following Allogeneic Hematopoietic Cell Transplantation. Cancers, 12(7), 1974. https://doi.org/10.3390/cancers12071974




