Genetics and Opioids: Towards More Appropriate Prescription in Cancer Pain
Abstract
1. Introduction
2. Methods
3. Genetic Variation and Drug Response
3.1. Pharmacokinetic Phenotype (PK)
3.1.1. Cytochrome P-450 Genetic Variability and Opioid Pharmacokinetics
3.1.2. UGP Genetic Variability and Opioid Pharmacokinetics (PK)
3.2. Pharmacodynamic Profile
OPRM1 and COMT Genes Variability Influences Opioid Pharmacodynamics
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Bruera, E.; Paice, J.A. Cancer pain management: Safe and effective use of opioids. Am. Soc. Clin. Oncol. Educ. Book 2015, 35, 593–599. [Google Scholar] [CrossRef] [PubMed]
- Varrassi, G.; Fusco, M.; Skaper, S.D.; Battelli, D.; Zis, P.; Coaccioli, S.; Pace, M.C.; Paladini, A. A pharmacological rationale to reduce the incidence of opioid induced tolerance and hyperalgesia: A review. Pain Ther. 2018, 7, 59–75. [Google Scholar] [CrossRef]
- Sacerdote, P. Opioid-induced immunosuppression. Curr. Opin. Support Palliat. Care 2008, 2, 14–18. [Google Scholar] [CrossRef]
- Fountas, A.; Van Uum, S.; Karavitaki, N. Opioid-induced endocrinopathies. Lancet Diabetes Endocrinol. 2020, 8, 68–80. [Google Scholar] [CrossRef]
- Paice, J.A. Cancer pain management and the opioid crisis in America: How to preserve hard-earned gains in improving the quality of cancer pain management. Cancer 2018, 124, 2491–2497. [Google Scholar] [CrossRef]
- De Gregori, M.; Diatchenko, L.; Belfer, I.; Allegri, M. OPRM1 receptor as new biomarker to help the prediction of post mastectomy pain and recurrence in breast cancer. Minerva Anestesiol. 2015, 81, 894–900. [Google Scholar]
- Ruano, G.; Kost, J.A. Fundamental considerations for genetically-guided pain management with opioids based on CYP2D6 and OPRM1 polymorphisms. Pain Physician 2018, 21, E611–e621. [Google Scholar] [CrossRef]
- Black, J.L., III; Walker, D.L.; O’Kane, D.J.; Harmandayan, M. Frequency of undetected CYP2D6 hybrid genes in clinical samples: Impact on phenotype prediction. Drug Metab. Dispos. 2012, 40, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Samer, C.F.; Daali, Y.; Wagner, M.; Hopfgartner, G.; Eap, C.B.; Rebsamen, M.C.; Rossier, M.F.; Hochstrasser, D.; Dayer, P.; Desmeules, J.A. Genetic polymorphisms and drug interactions modulating CYP2D6 and CYP3A activities have a major effect on oxycodone analgesic efficacy and safety. Br. J. Pharmacol. 2010, 160, 919–930. [Google Scholar] [CrossRef] [PubMed]
- Neafsey, P.; Ginsberg, G.; Hattis, D.; Sonawane, B. Genetic polymorphism in cytochrome P450 2D6 (CYP2D6): Population distribution of CYP2D6 activity. J. Toxicol. Environ. Health B Crit. Rev. 2009, 12, 334–361. [Google Scholar] [CrossRef] [PubMed]
- Haufroid, V.; Hantson, P. CYP2D6 genetic polymorphisms and their relevance for poisoning due to amfetamines, opioid analgesics and antidepressants. Clin. Toxicol. 2015, 53, 501–510. [Google Scholar] [CrossRef] [PubMed]
- Koren, G.; Cairns, J.; Chitayat, D.; Gaedigk, A.; Leeder, S.J. Pharmacogenetics of morphine poisoning in a breastfed neonate of a codeine-prescribed mother. Lancet 2006, 368, 704. [Google Scholar] [CrossRef]
- Orliaguet, G.; Hamza, J.; Couloigner, V.; Denoyelle, F.; Loriot, M.A.; Broly, F.; Garabedian, E.N. A case of respiratory depression in a child with ultrarapid CYP2D6 metabolism after tramadol. Pediatrics 2015, 135, e753–e755. [Google Scholar] [CrossRef] [PubMed]
- Stamer, U.M.; Stuber, F.; Muders, T.; Musshoff, F. Respiratory depression with tramadol in a patient with renal impairment and CYP2D6 gene duplication. Anesth. Analg. 2008, 107, 926–929. [Google Scholar] [CrossRef] [PubMed]
- Rocco, R.; Thiels, C.A.; Ubl, D.S.; Moyer, A.M.; Habermann, E.B.; Cassivi, S.D. Use of pharmacogenetic data to guide individualized opioid prescribing after surgery. Surgery 2019, 166, 476–482. [Google Scholar] [CrossRef] [PubMed]
- Brousseau, D.C.; McCarver, D.G.; Drendel, A.L.; Divakaran, K.; Panepinto, J.A. The effect of CYP2D6 polymorphisms on the response to pain treatment for pediatric sickle cell pain crisis. J. Pediatr. 2007, 150, 623–626. [Google Scholar] [CrossRef]
- Dagostino, C.; Allegri, M.; Napolioni, V.; D’Agnelli, S.; Bignami, E.; Mutti, A.; van Schaik, R.H. CYP2D6 genotype can help to predict effectiveness and safety during opioid treatment for chronic low back pain: Results from a retrospective study in an Italian cohort. Pharmgenomics Pers. Med. 2018, 11, 179–191. [Google Scholar] [CrossRef]
- Linares, O.A.; Daly, D.; Linares, A.D.; Stefanovski, D.; Boston, R.C. Personalized oxycodone dosing: Using pharmacogenetic testing and clinical pharmacokinetics to reduce toxicity risk and increase effectiveness. Pain Med. 2014, 15, 791–806. [Google Scholar] [CrossRef]
- Mosley, S.A.; Hicks, J.K.; Portman, D.G.; Donovan, K.A.; Gopalan, P.; Schmit, J.; Starr, J.; Silver, N.; Gong, Y.; Langaee, T.; et al. Design and rational for the precision medicine guided treatment for cancer pain pragmatic clinical trial. Contemp. Clin. Trials 2018, 68, 7–13. [Google Scholar] [CrossRef]
- Smith, D.M.; Weitzel, K.W.; Elsey, A.R.; Langaee, T.; Gong, Y.; Wake, D.T.; Duong, B.Q.; Hagen, M.; Harle, C.A.; Mercado, E.; et al. CYP2D6-guided opioid therapy improves pain control in CYP2D6 intermediate and poor metabolizers: A pragmatic clinical trial. Genet. Med. 2019, 21, 1842–1850. [Google Scholar] [CrossRef]
- Tanaka, H.; Naito, T.; Sato, H.; Hiraide, T.; Yamada, Y.; Kawakami, J. Impact of CYP genotype and inflammatory markers on the plasma concentrations of tramadol and its demethylated metabolites and drug tolerability in cancer patients. Eur. J. Clin. Pharmacol. 2018, 74, 1461–1469. [Google Scholar] [CrossRef] [PubMed]
- Andreassen, T.N.; Eftedal, I.; Klepstad, P.; Davies, A.; Bjordal, K.; Lundstrom, S.; Kaasa, S.; Dale, O. Do CYP2D6 genotypes reflect oxycodone requirements for cancer patients treated for cancer pain? A cross-sectional multicentre study. Eur. J. Clin. Pharmacol. 2012, 68, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Naito, T.; Tashiro, M.; Ishida, T.; Ohnishi, K.; Kawakami, J. Cancer cachexia raises the plasma concentration of oxymorphone through the reduction of CYP3A but not CYP2D6 in oxycodone-treated patients. J. Clin. Pharmacol. 2013, 53, 812–818. [Google Scholar] [CrossRef] [PubMed]
- Naito, T.; Tashiro, M.; Yamamoto, K.; Ohnishi, K.; Kagawa, Y.; Kawakami, J. Impact of cachexia on pharmacokinetic disposition of and clinical responses to oxycodone in cancer patients. Eur. J. Clin. Pharmacol. 2012, 68, 1411–1418. [Google Scholar] [CrossRef] [PubMed]
- Mercadante, S. Opioid metabolism and clinical aspects. Eur. J. Clin. Pharmacol. 2015, 769, 71–78. [Google Scholar] [CrossRef]
- Guitton, J.; Buronfosse, T.; Désage, M.; Lepape, A.; Brazier, J.L.; Beaune, P. Possible involvement of multiple cytochrome P450S in fentanyl and sufentanil metabolism as opposed to alfentanil. Biochem. Pharmacol. 1997, 53, 1613–1619. [Google Scholar] [CrossRef]
- Gasche, Y.; Daali, Y.; Fathi, M.; Chiappe, A.; Cottini, S.; Dayer, P.; Desmeules, J. Codeine intoxication associated with ultrarapid CYP2D6 metabolism. N. Engl. J. Med. 2004, 351, 2827–2831. [Google Scholar] [CrossRef]
- van Schaik, R.H.; van der Heiden, I.P.; van den Anker, J.N.; Lindemans, J. CYP3A5 variant allele frequencies in Dutch Caucasians. Clin. Chem. 2002, 48, 1668–1671. [Google Scholar] [CrossRef]
- Elens, L.; Nieuweboer, A.; Clarke, S.J.; Charles, K.A.; de Graan, A.J.; Haufroid, V.; Mathijssen, R.H.; van Schaik, R.H. CYP3A4 intron 6 C > T SNP (CYP3A4*22) encodes lower CYP3A4 activity in cancer patients, as measured with probes midazolam and erythromycin. Pharmacogenomics 2013, 14, 137–149. [Google Scholar] [CrossRef]
- Matic, M.; de Wildt, S.N.; Tibboel, D.; van Schaik, R.H.N. Analgesia and opioids: A pharmacogenetics shortlist for implementation in clinical practice. Clin. Chem. 2017, 63, 1204–1213. [Google Scholar] [CrossRef]
- Barratt, D.T.; Bandak, B.; Klepstad, P.; Dale, O.; Kaasa, S.; Christrup, L.L.; Tuke, J.; Somogyi, A.A. Genetic, pathological and physiological determinants of transdermal fentanyl pharmacokinetics in 620 cancer patients of the EPOS study. Pharmacogenet. Genom. 2014, 24, 185–194. [Google Scholar] [CrossRef] [PubMed]
- Ren, Z.Y.; Xu, X.Q.; Bao, Y.P.; He, J.; Shi, L.; Deng, J.H.; Gao, X.J.; Tang, H.L.; Wang, Y.M.; Lu, L. The impact of genetic variation on sensitivity to opioid analgesics in patients with postoperative pain: A systematic review and meta-analysis. Pain Physician 2015, 18, 131–152. [Google Scholar] [PubMed]
- Takashina, Y.; Naito, T.; Mino, Y.; Yagi, T.; Ohnishi, K.; Kawakami, J. Impact of CYP3A5 and ABCB1 gene polymorphisms on fentanyl pharmacokinetics and clinical responses in cancer patients undergoing conversion to a transdermal system. Drug Metab. Pharmacokinet. 2012, 27, 414–421. [Google Scholar] [CrossRef] [PubMed]
- Ross, J.R.; Rutter, D.; Welsh, K.; Joel, S.P.; Goller, K.; Wells, A.U.; Du Bois, R.; Riley, J. Clinical response to morphine in cancer patients and genetic variation in candidate genes. Pharmacogenomics J. 2005, 5, 324–336. [Google Scholar] [CrossRef]
- Klepstad, P.; Rakvåg, T.T.; Kaasa, S.; Holthe, M.; Dale, O.; Borchgrevink, P.C.; Baar, C.; Vikan, T.; Krokan, H.E.; Skorpen, F. The 118 A > G polymorphism in the human mu-opioid receptor gene may increase morphine requirements in patients with pain caused by malignant disease. Acta Anaesthesiol. Scand. 2004, 48, 1232–1239. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.C.; Wang, W.S. Human mu-opioid receptor gene A118G polymorphism predicts the efficacy of tramadol/acetaminophen combination tablets (ultracet) in oxaliplatin-induced painful neuropathy. Cancer 2012, 118, 1718–1725. [Google Scholar] [CrossRef]
- Matic, M.; Jongen, J.L.; Elens, L.; de Wildt, S.N.; Tibboel, D.; Sillevis Smitt, P.A.; van Schaik, R.H. Advanced cancer pain: The search for genetic factors correlated with interindividual variability in opioid requirement. Pharmacogenomics 2017, 18, 1133–1142. [Google Scholar] [CrossRef]
- Reyes-Gibby, C.C.; Shete, S.; Rakvag, T.; Bhat, S.V.; Skorpen, F.; Bruera, E.; Kaasa, S.; Klepstad, P. Exploring joint effects of genes and the clinical efficacy of morphine for cancer pain: OPRM1 and COMT gene. Pain 2007, 130, 25–30. [Google Scholar] [CrossRef]
- Gray, K.; Adhikary, S.D.; Janicki, P. Pharmacogenomics of analgesics in anesthesia practice: A current update of literature. J. Anaesthesiol. Clin. Pharmacol. 2018, 34, 155–160. [Google Scholar] [CrossRef]
- Meineke, I.; Freudenthaler, S.; Hofmann, U.; Schaeffeler, E.; Mikus, G.; Schwab, M.; Prange, H.W.; Gleiter, C.H.; Brockmoller, J. Pharmacokinetic modelling of morphine, morphine-3-glucuronide and morphine-6-glucuronide in plasma and cerebrospinal fluid of neurosurgical patients after short-term infusion of morphine. Br. J. Clin. Pharmacol. 2002, 54, 592–603. [Google Scholar] [CrossRef]
- Lotsch, J.; von Hentig, N.; Freynhagen, R.; Griessinger, N.; Zimmermann, M.; Doehring, A.; Rohrbacher, M.; Sittl, R.; Geisslinger, G. Cross-sectional analysis of the influence of currently known pharmacogenetic modulators on opioid therapy in outpatient pain centers. Pharmacogenet. Genom. 2009, 19, 429–436. [Google Scholar] [CrossRef] [PubMed]
- Lang, T.; Hitzl, M.; Burk, O.; Mornhinweg, E.; Keil, A.; Kerb, R.; Klein, K.; Zanger, U.M.; Eichelbaum, M.; Fromm, M.F. Genetic polymorphisms in the multidrug resistance-associated protein 3 (ABCC3, MRP3) gene and relationship to its mRNA and protein expression in human liver. Pharmacogenetics 2004, 14, 155–164. [Google Scholar] [CrossRef] [PubMed]
- Venkatasubramanian, R.; Fukuda, T.; Niu, J.; Mizuno, T.; Chidambaran, V.; Vinks, A.A.; Sadhasivam, S. ABCC3 and OCT1 genotypes influence pharmacokinetics of morphine in children. Pharmacogenomics 2014, 15, 1297–1309. [Google Scholar] [CrossRef] [PubMed]
- Tzvetkov, M.V.; Saadatmand, A.R.; Lotsch, J.; Tegeder, I.; Stingl, J.C.; Brockmoller, J. Genetically polymorphic OCT1: Another piece in the puzzle of the variable pharmacokinetics and pharmacodynamics of the opioidergic drug tramadol. Clin. Pharmacol. Ther. 2011, 90, 143–150. [Google Scholar] [CrossRef]
- Stamer, U.M.; Musshoff, F.; Stuber, F.; Brockmoller, J.; Steffens, M.; Tzvetkov, M.V. Loss-of-function polymorphisms in the organic cation transporter OCT1 are associated with reduced postoperative tramadol consumption. Pain 2016, 157, 2467–2475. [Google Scholar] [CrossRef]
- Tzvetkov, M.V.; dos Santos Pereira, J.N.; Meineke, I.; Saadatmand, A.R.; Stingl, J.C.; Brockmoller, J. Morphine is a substrate of the organic cation transporter OCT1 and polymorphisms in OCT1 gene affect morphine pharmacokinetics after codeine administration. Biochem. Pharmacol. 2013, 86, 666–678. [Google Scholar] [CrossRef]
- Coffman, B.L.; Rios, G.R.; King, C.D.; Tephly, T.R. Human UGT2B7 catalyzes morphine glucuronidation. Drug Metab. Dispos. 1997, 25, 1–4. [Google Scholar]
- Christrup, L.L. Morphine metabolites. Acta Anaesthesiol. Scand. 1997, 41, 116–122. [Google Scholar] [CrossRef]
- De Gregori, S.; Minella, C.E.; De Gregori, M.; Tinelli, C.; Ranzani, G.N.; Govoni, S.; Allegri, M.; Regazzi, M. Clinical pharmacokinetics of morphine and its metabolites during morphine dose titration for chronic cancer pain. Ther. Drug Monit. 2014, 36, 335–344. [Google Scholar] [CrossRef]
- Holthe, M.; Klepstad, P.; Zahlsen, K.; Borchgrevink, P.C.; Hagen, L.; Dale, O.; Kaasa, S.; Krokan, H.E.; Skorpen, F. Morphine glucuronide-to-morphine plasma ratios are unaffected by the UGT2B7 H268Y and UGT1A1*28 polymorphisms in cancer patients on chronic morphine therapy. Eur. J. Clin. Pharmacol. 2002, 58, 353–356. [Google Scholar] [CrossRef]
- Holthe, M.; Rakvag, T.N.; Klepstad, P.; Idle, J.R.; Kaasa, S.; Krokan, H.E.; Skorpen, F. Sequence variations in the UDP-glucuronosyltransferase 2B7 (UGT2B7) gene: Identification of 10 novel single nucleotide polymorphisms (SNPs) and analysis of their relevance to morphine glucuronidation in cancer patients. Pharmacogenomics J. 2003, 3, 17–26. [Google Scholar] [CrossRef] [PubMed]
- Duguay, Y.; Baar, C.; Skorpen, F.; Guillemette, C. A novel functional polymorphism in the uridine diphosphate-glucuronosyltransferase 2B7 promoter with significant impact on promoter activity. Clin. Pharmacol. Ther. 2004, 75, 223–233. [Google Scholar] [CrossRef] [PubMed]
- Klepstad, P.; Fladvad, T.; Skorpen, F.; Bjordal, K.; Caraceni, A.; Dale, O.; Davies, A.; Kloke, M.; Lundstrom, S.; Maltoni, M.; et al. Influence from genetic variability on opioid use for cancer pain: A European genetic association study of 2294 cancer pain patients. Pain 2011, 152, 1139–1145. [Google Scholar] [CrossRef]
- Fujita, K.; Ando, Y.; Yamamoto, W.; Miya, T.; Endo, H.; Sunakawa, Y.; Araki, K.; Kodama, K.; Nagashima, F.; Ichikawa, W.; et al. Association of UGT2B7 and ABCB1 genotypes with morphine-induced adverse drug reactions in Japanese patients with cancer. Cancer Chemother. Pharmacol. 2010, 65, 251–258. [Google Scholar] [CrossRef]
- Oroszi, G.; Anton, R.F.; O’Malley, S.; Swift, R.; Pettinati, H.; Couper, D.; Yuan, Q.; Goldman, D. OPRM1 Asn40Asp predicts response to naltrexone treatment: A haplotype-based approach. Alcohol Clin. Exp. Res. 2009, 33, 383–393. [Google Scholar] [CrossRef] [PubMed]
- Mura, E.; Govoni, S.; Racchi, M.; Carossa, V.; Ranzani, G.N.; Allegri, M.; van Schaik, R.H. Consequences of the 118A>G polymorphism in the OPRM1 gene: Translation from bench to bedside? J. Pain Res. 2013, 6, 331–353. [Google Scholar] [CrossRef] [PubMed]
- Ravindranathan, A.; Joslyn, G.; Robertson, M.; Schuckit, M.A.; Whistler, J.L.; White, R.L. Functional characterization of human variants of the mu-opioid receptor gene. Proc. Natl. Acad. Sci. USA 2009, 106, 10811–10816. [Google Scholar] [CrossRef] [PubMed]
- Bauer, I.E.; Soares, J.C.; Nielsen, D.A. The role of opioidergic genes in the treatment outcome of drug addiction pharmacotherapy: A systematic review. Am. J. Addict. 2015, 24, 15–23. [Google Scholar] [CrossRef]
- Hwang, I.C.; Park, J.Y.; Myung, S.K.; Ahn, H.Y.; Fukuda, K.; Liao, Q. OPRM1 A118G gene variant and postoperative opioid requirement: A systematic review and meta-analysis. Anesthesiology 2014, 121, 825–834. [Google Scholar] [CrossRef]
- Yuan, J.J.; Hou, J.K.; Zhang, W.; Chang, Y.Z.; Li, Z.S.; Wang, Z.Y.; Du, Y.Y.; Ma, X.J.; Zhang, L.R.; Kan, Q.C.; et al. CYP3A4 * 1G Genetic polymorphism influences metabolism of fentanyl in human liver microsomes in Chinese patients. Pharmacology 2015, 96, 55–60. [Google Scholar] [CrossRef]
- De Gregori, M.; Diatchenko, L.; Ingelmo, P.M.; Napolioni, V.; Klepstad, P.; Belfer, I.; Molinaro, V.; Garbin, G.; Ranzani, G.N.; Alberio, G.; et al. Human genetic variability contributes to postoperative morphine consumption. J. Pain 2016, 17, 628–636. [Google Scholar] [CrossRef] [PubMed]
- Andersen, S.; Skorpen, F. Variation in the COMT gene: Implications for pain perception and pain treatment. Pharmacogenomics 2009, 10, 669–684. [Google Scholar] [CrossRef] [PubMed]
- Belfer, I.; Segall, S.K.; Lariviere, W.R.; Smith, S.B.; Dai, F.; Slade, G.D.; Rashid, N.U.; Mogil, J.S.; Campbell, C.M.; Edwards, R.R.; et al. Pain modality- and sex-specific effects of COMT genetic functional variants. Pain 2013, 154, 1368–1376. [Google Scholar] [CrossRef] [PubMed]
- Diatchenko, L.; Slade, G.D.; Nackley, A.G.; Bhalang, K.; Sigurdsson, A.; Belfer, I.; Goldman, D.; Xu, K.; Shabalina, S.A.; Shagin, D.; et al. Genetic basis for individual variations in pain perception and the development of a chronic pain condition. Hum. Mol. Genet. 2005, 14, 135–143. [Google Scholar] [CrossRef] [PubMed]
- Meloto, C.B.; Segall, S.K.; Smith, S.; Parisien, M.; Shabalina, S.A.; Rizzatti-Barbosa, C.M.; Gauthier, J.; Tsao, D.; Convertino, M.; Piltonen, M.H.; et al. COMT gene locus: New functional variants. Pain 2015, 156, 2072–2083. [Google Scholar] [CrossRef]
- Nascimento, T.D.; Yang, N.; Salman, D.; Jassar, H.; Kaciroti, N.; Bellile, E.; Danciu, T.; Koeppe, R.; Stohler, C.; Zubieta, J.K.; et al. µ-Opioid activity in chronic TMD pain is associated with COMT Polymorphism. J. Dent. Res. 2019, 98, 1324–1331. [Google Scholar] [CrossRef]
- Kowarik, M.C.; Einhauser, J.; Jochim, B.; Buttner, A.; Tolle, T.R.; Riemenschneider, M.; Platzer, S.; Berthele, A. Impact of the COMT Val(108/158)Met polymorphism on the mu-opioid receptor system in the human brain: Mu-opioid receptor, met-enkephalin and beta-endorphin expression. Neurosci. Lett. 2012, 506, 214–219. [Google Scholar] [CrossRef]
- Matsuoka, H.; Arao, T.; Makimura, C.; Takeda, M.; Kiyota, H.; Tsurutani, J.; Fujita, Y.; Matsumoto, K.; Kimura, H.; Otsuka, M.; et al. Expression changes in arrestin beta 1 and genetic variation in catechol-O-methyltransferase are biomarkers for the response to morphine treatment in cancer patients. Oncol. Rep. 2012, 27, 1393–1399. [Google Scholar] [CrossRef]
- Rakvag, T.T.; Klepstad, P.; Baar, C.; Kvam, T.M.; Dale, O.; Kaasa, S.; Krokan, H.E.; Skorpen, F. The Val158Met polymorphism of the human catechol-O-methyltransferase (COMT) gene may influence morphine requirements in cancer pain patients. Pain 2005, 116, 73–78. [Google Scholar] [CrossRef]
- Rakvag, T.T.; Ross, J.R.; Sato, H.; Skorpen, F.; Kaasa, S.; Klepstad, P. Genetic variation in the catechol-O-methyltransferase (COMT) gene and morphine requirements in cancer patients with pain. Mol. Pain 2008, 4, 64. [Google Scholar] [CrossRef]
- Cargnin, S.; Magnani, F.; Viana, M.; Tassorelli, C.; Mittino, D.; Cantello, R.; Sances, G.; Nappi, G.; Canonico, P.L.; Genazzani, A.A.; et al. An opposite-direction modulation of the COMT Val158Met polymorphism on the clinical response to intrathecal morphine and triptans. J. Pain 2013, 14, 1097–1106. [Google Scholar] [CrossRef]
- Henker, R.A.; Lewis, A.; Dai, F.; Lariviere, W.R.; Meng, L.; Gruen, G.S.; Sereika, S.M.; Pape, H.; Tarkin, I.S.; Gowda, I.; et al. The associations between OPRM 1 and COMT genotypes and postoperative pain, opioid use, and opioid-induced sedation. Biol. Res. Nurs. 2013, 15, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Kolesnikov, Y.; Gabovits, B.; Levin, A.; Voiko, E.; Veske, A. Combined catechol-O-methyltransferase and mu-opioid receptor gene polymorphisms affect morphine postoperative analgesia and central side effects. Anesth. Analg. 2011, 112, 448–453. [Google Scholar] [CrossRef] [PubMed]
- De Gregori, M.; Garbin, G.; De Gregori, S.; Minella, C.E.; Bugada, D.; Lisa, A.; Govoni, S.; Regazzi, M.; Allegri, M.; Ranzani, G.N. Genetic variability at COMT but not at OPRM1 and UGT2B7 loci modulates morphine analgesic response in acute postoperative pain. Eur. J. Clin. Pharmacol. 2013, 69, 1651–1658. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.F.; Aloyo, V.J.; Weiss, B. Continuous treatment with the D2 dopamine receptor agonist quinpirole decreases D2 dopamine receptors, D2 dopamine receptor messenger RNA and proenkephalin messenger RNA, and increases mu opioid receptors in mouse striatum. Neuroscience 1993, 54, 669–680. [Google Scholar] [CrossRef]
- Steiner, H.; Gerfen, C.R. Role of dynorphin and enkephalin in the regulation of striatal output pathways and behavior. Exp. Brain Res. 1998, 123, 60–76. [Google Scholar] [CrossRef] [PubMed]
- Choi, S.W.; Lam, D.M.H.; Wong, S.S.C.; Shiu, H.H.C.; Wang, A.X.M.; Cheung, C.W. Effects of Single Nucleotide Polymorphisms on Surgical and Postsurgical Opioid Requirements: A Systematic Review and Meta-Analysis. Clin. J. Pain 2017, 33, 1117–1130. [Google Scholar] [CrossRef]
- Lucenteforte, E.; Vannacci, A.; Crescioli, G.; Lombardi, N.; Vagnoli, L.; Giunti, L.; Cetica, V.; Coniglio, M.L.; Pugi, A.; Bonaiuti, R.; et al. Opioid response in paediatric cancer patients and the Val158Met polymorphism of the human catechol-O-methyltransferase (COMT) gene: An Italian study on 87 cancer children and a systematic review. BMC Cancer 2019, 19, 113. [Google Scholar] [CrossRef]
- Neufeld, N.J.; Elnahal, S.M.; Alvarez, R.H. Cancer pain: A review of epidemiology, clinical quality and value impact. Future Oncol. 2017, 13, 833–841. [Google Scholar] [CrossRef]
- Gammal, R.S.; Crews, K.R.; Haidar, C.E.; Hoffman, J.M.; Baker, D.K.; Barker, P.J.; Estepp, J.H.; Pei, D.; Broeckel, U.; Wang, W.; et al. Pharmacogenetics for Safe Codeine Use in Sickle Cell Disease. Pediatrics 2016, 138. [Google Scholar] [CrossRef]
- Crews, K.R.; Gaedigk, A.; Dunnenberger, H.M.; Leeder, J.S.; Klein, T.E.; Caudle, K.E.; Haidar, C.E.; Shen, D.D.; Callaghan, J.T.; Sadhasivam, S.; et al. Clinical pharmacogenetics implementation consortium guidelines for cytochrome P450 2D6 genotype and codeine therapy: 2014 update. Clin. Pharmacol. Ther. 2014, 95, 376–382. [Google Scholar] [CrossRef]
- Swen, J.J.; Nijenhuis, M.; de Boer, A.; Grandia, L.; Maitland-van der Zee, A.H.; Mulder, H.; Rongen, G.A.; van Schaik, R.H.; Schalekamp, T.; Touw, D.J.; et al. Pharmacogenetics: From bench to byte—An update of guidelines. Clin. Pharmacol. Ther. 2011, 89, 662–673. [Google Scholar] [CrossRef] [PubMed]
- Bank, P.C.D.; Caudle, K.E.; Swen, J.J.; Gammal, R.S.; Whirl-Carrillo, M.; Klein, T.E.; Relling, M.V.; Guchelaar, H.J. Comparison of the Guidelines of the Clinical Pharmacogenetics Implementation Consortium and the Dutch Pharmacogenetics Working Group. Clin. Pharmacol. Ther. 2018, 103, 599–618. [Google Scholar] [CrossRef]
- van der Wouden, C.H.; van Rhenen, M.H.; Jama, W.O.M.; Ingelman-Sundberg, M.; Lauschke, V.M.; Konta, L.; Schwab, M.; Swen, J.J.; Guchelaar, H.J. Development of the PGx-Passport: A Panel of Actionable Germline Genetic Variants for Pre-Emptive Pharmacogenetic Testing. Clin. Pharmacol. Ther. 2019, 106, 866–873. [Google Scholar] [CrossRef] [PubMed]
- Singh, D.R.; Nag, K.; Shetti, A.N.; Krishnaveni, N. Tapentadol hydrochloride: A novel analgesic. Saudi J. Anaesth. 2013, 7, 322–326. [Google Scholar] [CrossRef]
- Lotsch, J.; Geisslinger, G. Pharmacogenetics of new analgesics. Br. J. Pharmacol. 2011, 163, 447–460. [Google Scholar] [CrossRef] [PubMed]
- Galvan, A.; Skorpen, F.; Klepstad, P.; Knudsen, A.K.; Fladvad, T.; Falvella, F.S.; Pigni, A.; Brunelli, C.; Caraceni, A.; Kaasa, S.; et al. Multiple Loci modulate opioid therapy response for cancer pain. Clin. Cancer Res. 2011, 17, 4581–4587. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Z.; Zhou, H.; Sherva, R.; Farrer, L.A.; Kranzler, H.R.; Gelernter, J. Genome-wide association study identifies a regulatory variant of RGMA associated with opioid dependence in European Americans. Biol. Psychiatry 2018, 84, 762–770. [Google Scholar] [CrossRef]
- Gelernter, J.; Kranzler, H.R.; Sherva, R.; Koesterer, R.; Almasy, L.; Zhao, H.; Farrer, L.A. Genome-wide association study of opioid dependence: Multiple associations mapped to calcium and potassium pathways. Biol. Psychiatry 2014, 76, 66–74. [Google Scholar] [CrossRef]
- Smith, A.H.; Jensen, K.P.; Li, J.; Nunez, Y.; Farrer, L.A.; Hakonarson, H.; Cook-Sather, S.D.; Kranzler, H.R.; Gelernter, J. Genome-wide association study of therapeutic opioid dosing identifies a novel locus upstream of OPRM1. Mol. Psychiatry 2017, 22, 346–352. [Google Scholar] [CrossRef]
- Lehmann-Che, J.; Poirot, B.; Boyer, J.C.; Evrard, A. Cancer genomics guide clinical practice in personalized medicine. Therapie 2017, 72, 439–451. [Google Scholar] [CrossRef] [PubMed]
- Anderson, J.L.; Horne, B.D.; Stevens, S.M.; Woller, S.C.; Samuelson, K.M.; Mansfield, J.W.; Robinson, M.; Barton, S.; Brunisholz, K.; Mower, C.P.; et al. A randomized and clinical effectiveness trial comparing two pharmacogenetic algorithms and standard care for individualizing warfarin dosing (CoumaGen-II). Circulation 2012, 125, 1997–2005. [Google Scholar] [CrossRef] [PubMed]
- Vogenberg, F.R.; Barash, C.I.; Pursel, M. Personalized medicine: Part 3: Challenges facing health care plans in implementing coverage policies for pharmacogenomic and genetic testing. Pharm. Ther. 2010, 35, 670–675. [Google Scholar]
- Xu, Y.; Johnson, A. Opioid therapy pharmacogenomics for noncancer pain: Efficacy, adverse events, and costs. Pain Res. Treat. 2013, 2013, 943014. [Google Scholar] [CrossRef] [PubMed]
- Pergolizzi, J.V., Jr.; Labhsetwar, S.A.; Amy Puenpatom, R.; Ben-Joseph, R.; Ohsfeldt, R.; Summers, K.H. Economic impact of potential CYP450 pharmacokinetic drug-drug interactions among chronic low back pain patients taking opioids. Pain Pract. 2012, 12, 45–56. [Google Scholar] [CrossRef]
- Pergolizzi, J.V., Jr.; Labhsetwar, S.A.; Puenpatom, R.A.; Ben-Joseph, R.; Ohsfeldt, R.; Summers, K.H. Economic impact of potential drug-drug interactions among osteoarthritis patients taking opioids. Pain Pract. 2012, 12, 33–44. [Google Scholar] [CrossRef]
- Wu, A.C.; Fuhlbrigge, A.L. Economic evaluation of pharmacogenetic tests. Clin. Pharmacol. Ther. 2008, 84, 272–274. [Google Scholar] [CrossRef]
- Elliott, L.S.; Henderson, J.C.; Neradilek, M.B.; Moyer, N.A.; Ashcraft, K.C.; Thirumaran, R.K. Clinical impact of pharmacogenetic profiling with a clinical decision support tool in polypharmacy home health patients: A prospective pilot randomized controlled trial. PLoS ONE 2017, 12, e0170905. [Google Scholar] [CrossRef]
- Tollman, P.; Guy, P.; Altshuler, J.; Flanagan, A.; Steiner, M. A Revolution in R&D: How Genomics and Genetics are Transforming the Biopharmaceutical Industry; Boston Consulting Group: Boston, MA, USA, 2001. [Google Scholar]
- Compagnone, C.; Tagliaferri, F.; Allegri, M.; Fanelli, G. Ethical issues in pain and omics research. Some points to start the debate. Croat. Med. J. 2014, 55, 1–2. [Google Scholar] [CrossRef]
- Agarwal, D.; Udoji, M.A.; Trescot, A. Genetic testing for opioid pain management: A primer. Pain Ther. 2017, 6, 93–105. [Google Scholar] [CrossRef]
- Lipton, P. Pharmacogenetics: The ethical issues. Pharmacogenomics J. 2003, 3, 14–16. [Google Scholar] [CrossRef] [PubMed]
- Zittel, S.; Lohmann, K.; Bauer, P.; Klein, C.; Munchau, A. Munchausen syndrome by genetics: Next-generation challenges for clinicians. Neurology 2017, 88, 1000–1001. [Google Scholar] [CrossRef] [PubMed]
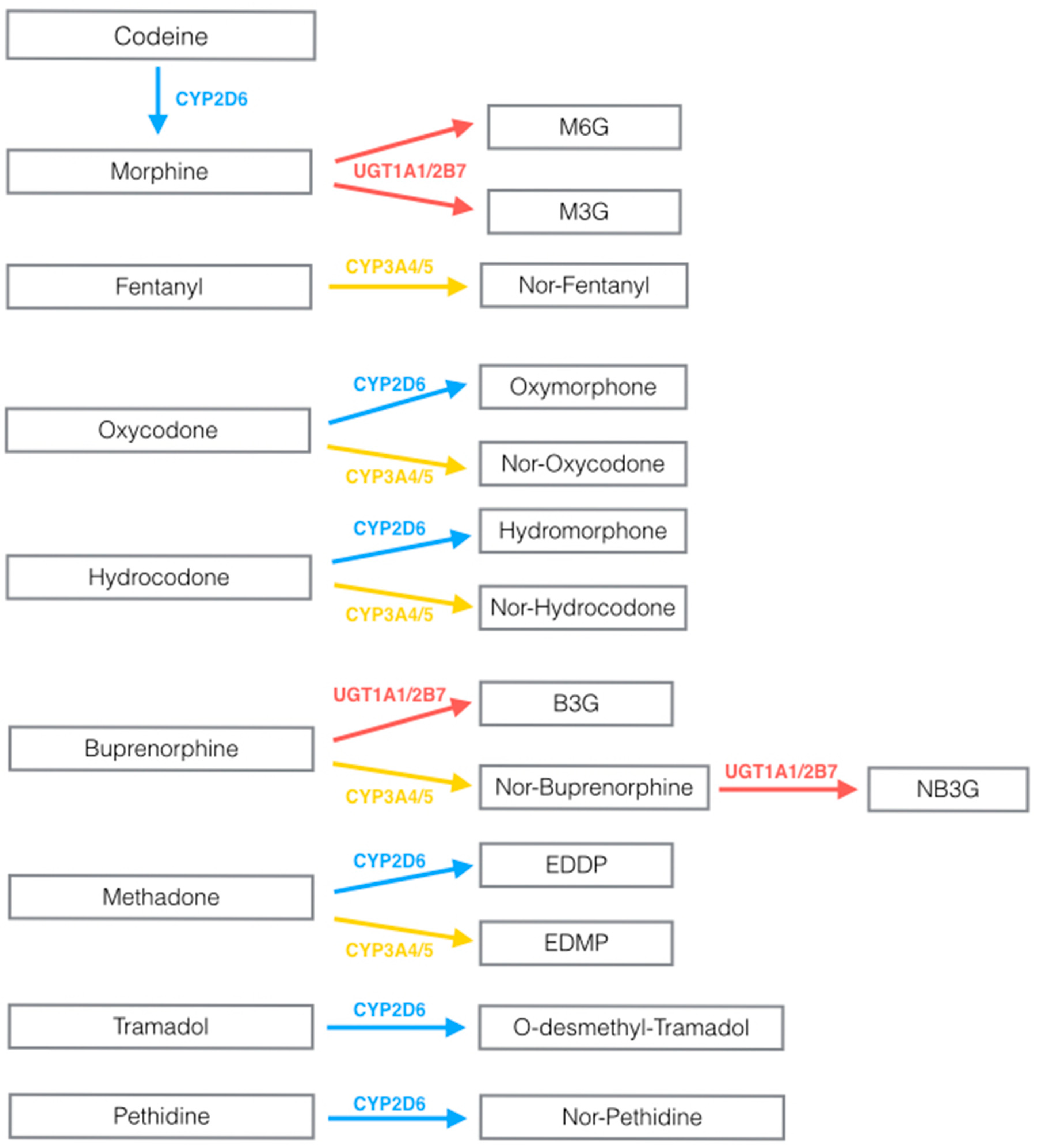
| PRO-Drug | Active Compound | Poor Metabolizers | Ultra-Rapid Metabolizers |
| Codeine | Morphine | ↓ efficacy | ↓ efficacy ↑ AE |
| Oxycodone | Oxymorphone | ↓ efficacy | ↓ efficacy ↑ AE |
| Hydrocodone | Hydromorphone | ↓ efficacy | ↓ efficacy ↑ AE |
| Tramadol | O-desmethyl-tramadol | ↓ efficacy | ↓ efficacy ↑ AE |
| Drug | Poor Metabolizers | Ultra-Rapid Metabolizers | |
| Pethidine (Meperidine) | Nor-pethidine | ↑ AE | ↓ efficacy |
| Morphine | M6G/M3G | ↑ AE | ↓ efficacy |
| Methadone | EDDP - EDMP | ↑ AE | ↓ efficacy |
| Gene (protein) | Drugs Involved | Function | Available Data |
|---|---|---|---|
| CYP2D6 (Cytochrome P450 2D6 isoenzyme) |
|
| |
| CYP3A4 (Cytochrome P450 family 3 subfamily A members 4) |
|
| |
| CYP3A5 (Cytochrome P450 family 3 subfamily A members 5) |
|
|
|
| OPRM1 (μ-opioid receptor) |
|
| |
| COMT (Catechol-O-methyltransferase) |
| ||
| ABCB1 (ATP binding cassette subfamily B member 1) |
|
|
|
| ABCC3 (ATP-binding cassette subfamily C member 3 transporter) |
|
| |
| SLC22A1 (Organic Cation Transporter 1) |
|
|
|
| KCNJ6 (G protein–activated inwardly rectifying potassium 2 channel- GIRK2) |
|
|
|
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bugada, D.; Lorini, L.F.; Fumagalli, R.; Allegri, M. Genetics and Opioids: Towards More Appropriate Prescription in Cancer Pain. Cancers 2020, 12, 1951. https://doi.org/10.3390/cancers12071951
Bugada D, Lorini LF, Fumagalli R, Allegri M. Genetics and Opioids: Towards More Appropriate Prescription in Cancer Pain. Cancers. 2020; 12(7):1951. https://doi.org/10.3390/cancers12071951
Chicago/Turabian StyleBugada, Dario, Luca F. Lorini, Roberto Fumagalli, and Massimo Allegri. 2020. "Genetics and Opioids: Towards More Appropriate Prescription in Cancer Pain" Cancers 12, no. 7: 1951. https://doi.org/10.3390/cancers12071951
APA StyleBugada, D., Lorini, L. F., Fumagalli, R., & Allegri, M. (2020). Genetics and Opioids: Towards More Appropriate Prescription in Cancer Pain. Cancers, 12(7), 1951. https://doi.org/10.3390/cancers12071951





