Non-Coding RNAs in Cancer Radiosensitivity: MicroRNAs and lncRNAs as Regulators of Radiation-Induced Signaling Pathways
Abstract
1. Introduction
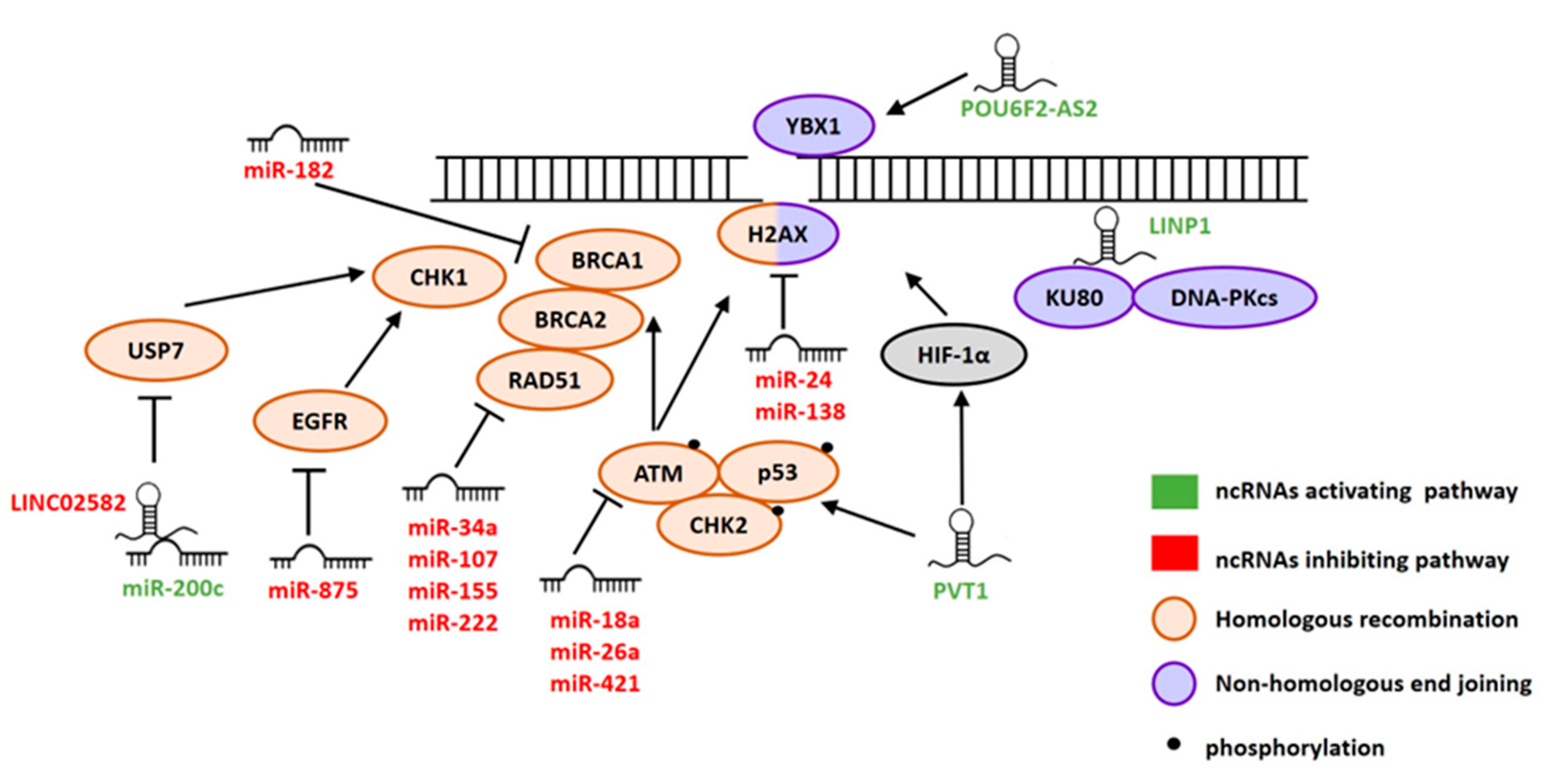
2. ncRNAs Involved in DSB Repair
2.1. miRNAs
2.2. lncRNAs
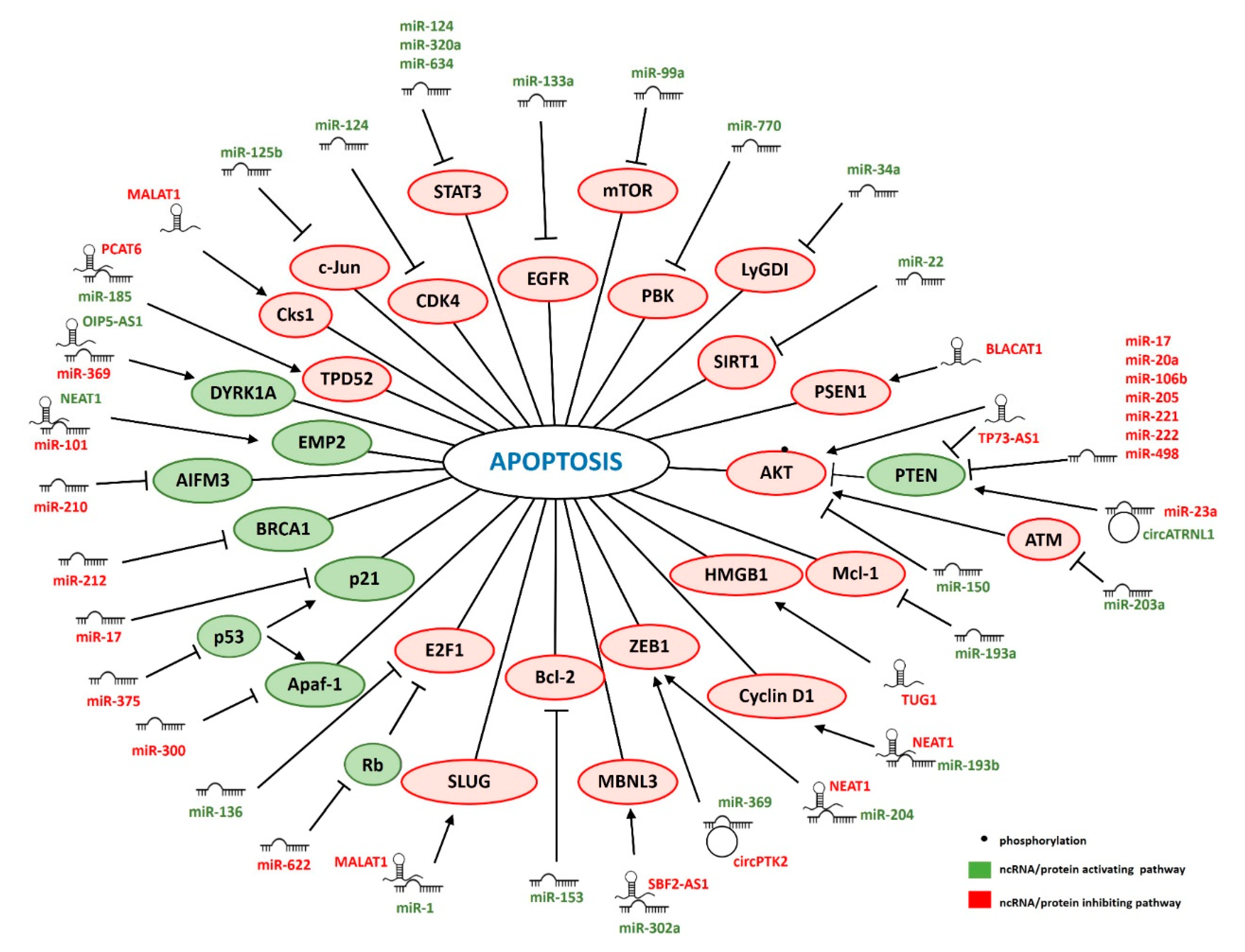
3. ncRNAs Regulating IR-Induced Apoptosis
3.1. miRNAs
3.2. lncRNAs
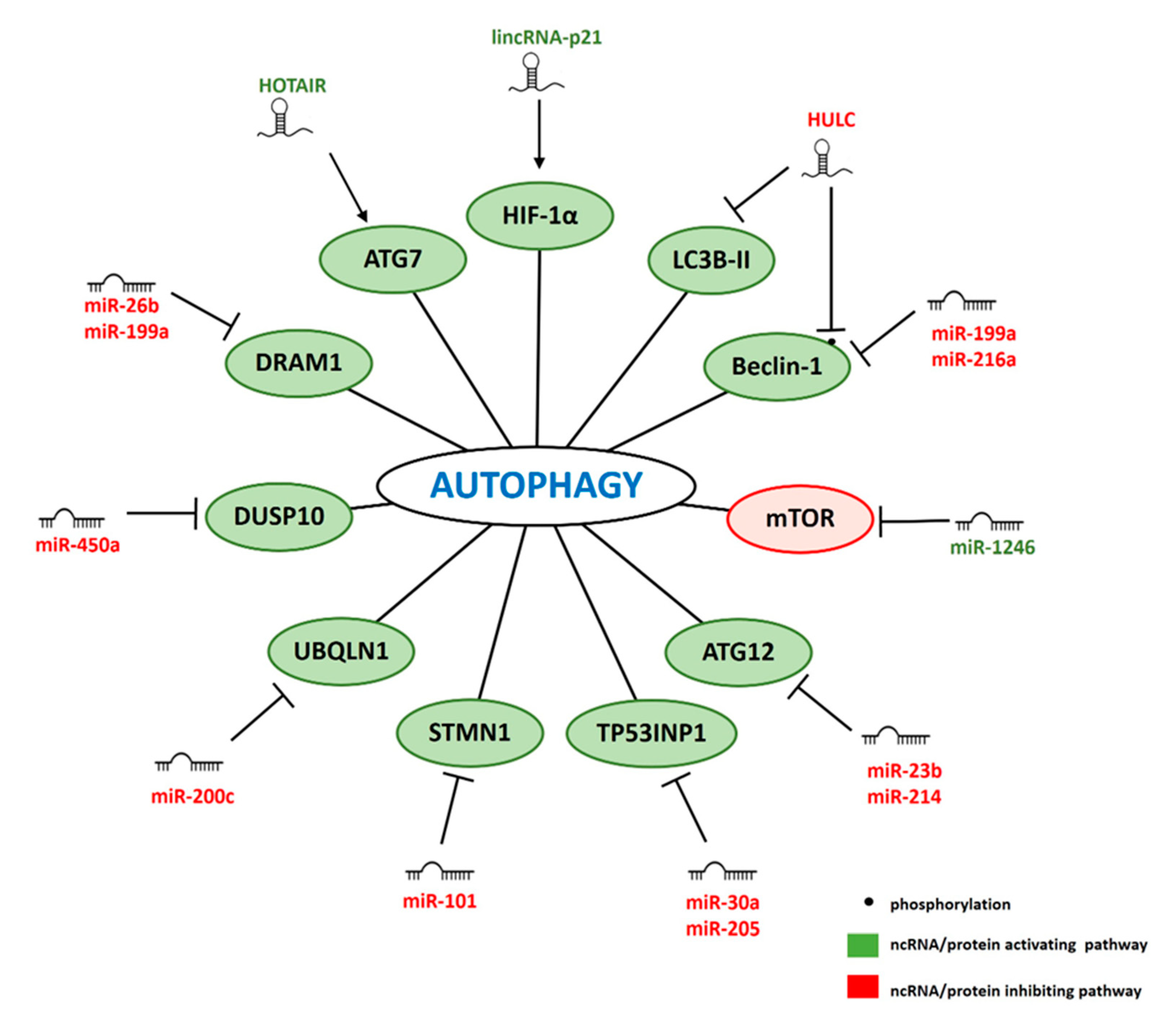
4. ncRNAs Involved in IR-Related Autophagy
4.1. miRNAs
4.2. lncRNAs
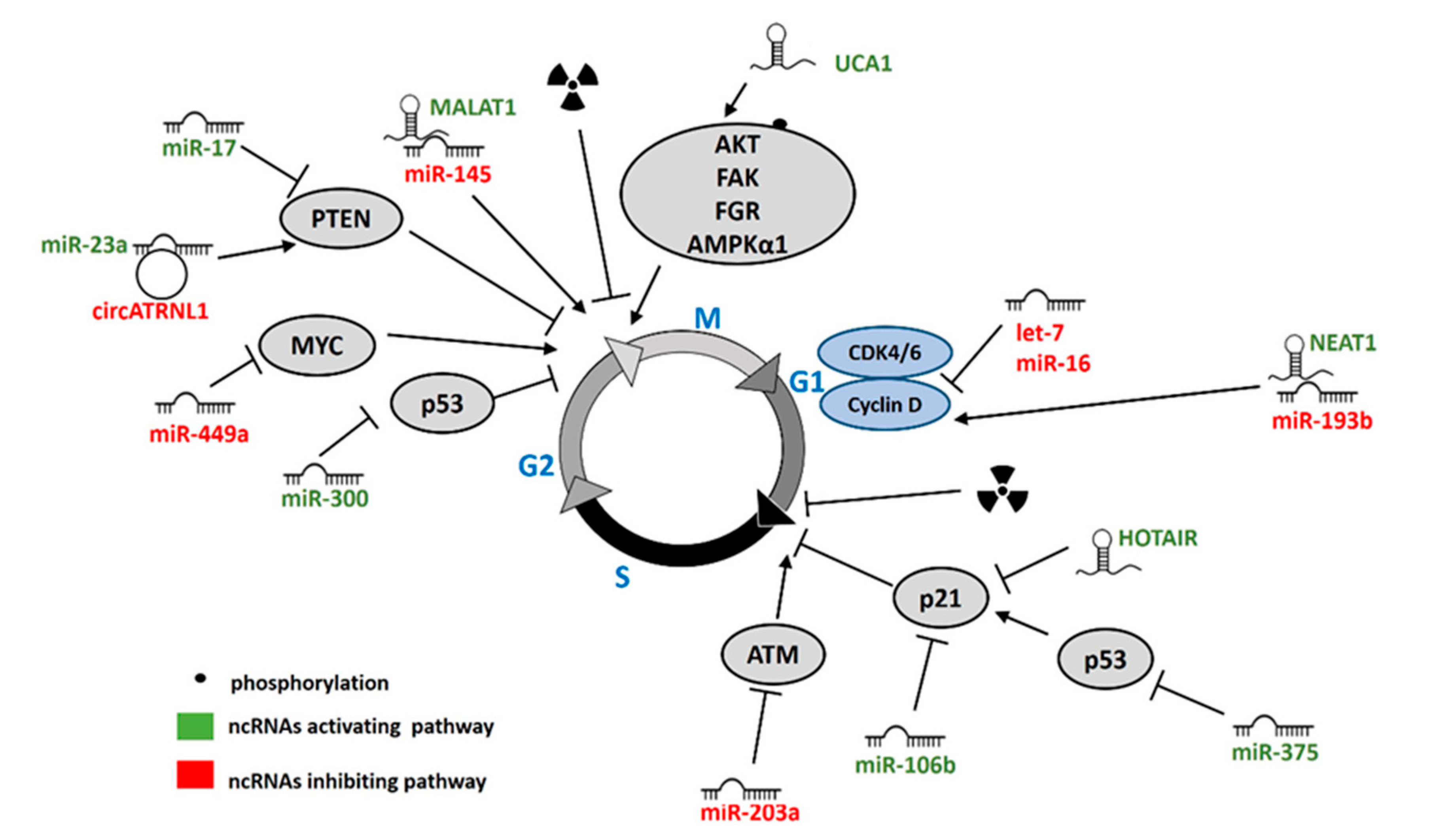
5. ncRNAs Regulating Cell Cycle in Response to IR
5.1. miRNAs
5.2. lncRNAs
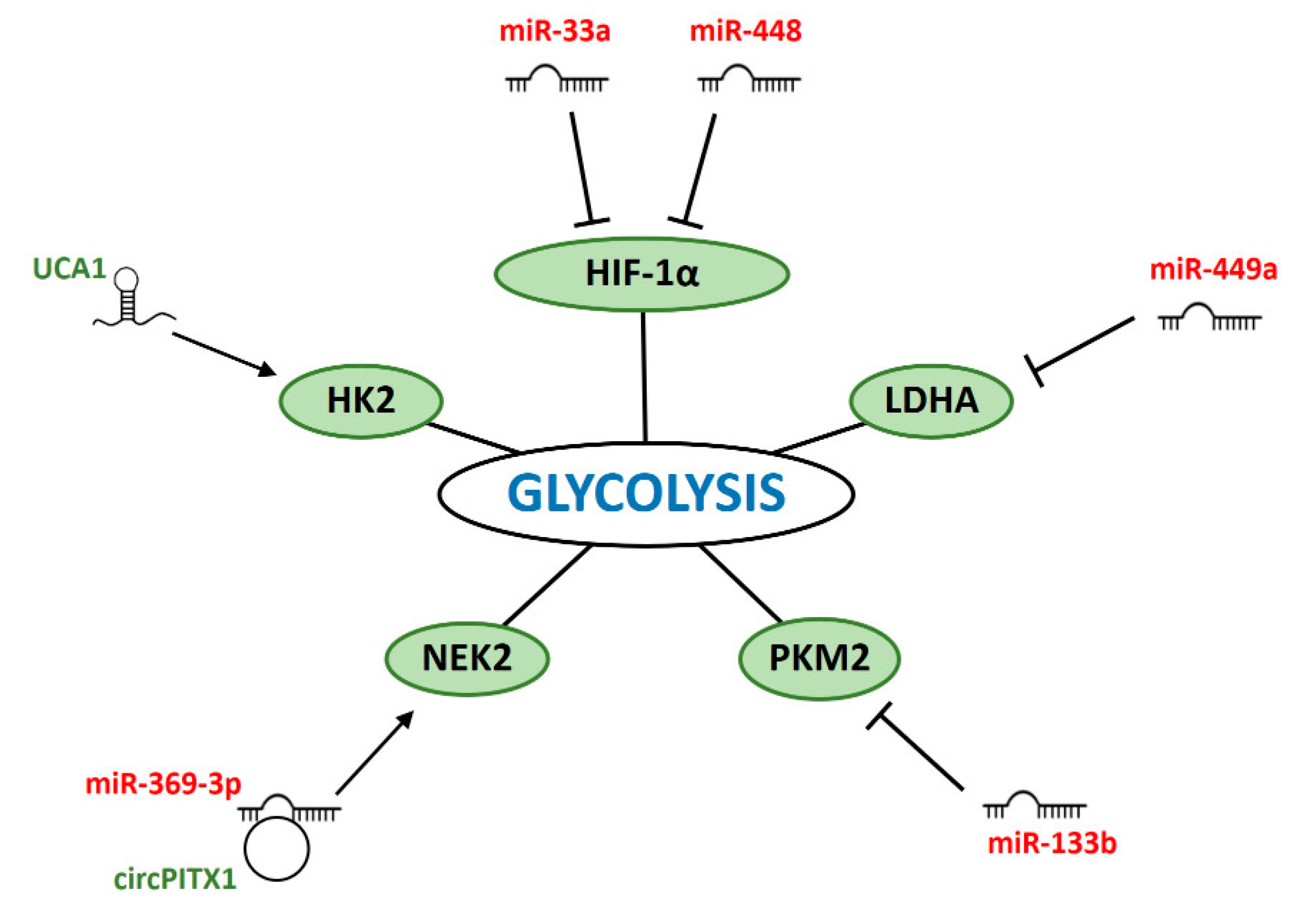
6. ncRNAs Modulating Glycolysis in IR-Response
6.1. miRNAs
6.2. lncRNAs
7. ncRNAs Affecting the Wnt/β-Catenin Pathway in Response to IR
7.1. miRNAs
7.2. lncRNAs
8. Conclusions
Funding
Conflicts of Interest
References
- Williams, J.R.; Zhang, Y.; Zhou, H.; Gridley, D.S.; Koch, C.J.; Russell, J.; Slater, J.S.; Little, J.B. A quantitative overview of radiosensitivity of human tumor cells across histological type and TP53 status. Int. J. Radiat. Biol. 2008, 84, 253–264. [Google Scholar] [CrossRef]
- Li, G.; Liu, Y.; Liu, C.; Su, Z.; Ren, S.; Wang, Y.; Deng, T.; Huang, D.; Tian, Y.; Qiu, Y. Genome-wide analyses of long noncoding RNA expression profiles correlated with radioresistance in nasopharyngeal carcinoma via next-generation deep sequencing. BMC Cancer 2016, 16, 719. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Cao, S.; Li, W.; Wei, D.; Wang, Z.; Li, G.; Pan, X.; Lei, D. Time-course differential lncRNA and mRNA expressions in radioresistant hypopharyngeal cancer cells. Oncotarget 2017, 8, 40994–41010. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.; Li, Y.; Ming, Z.; Wang, H.; Dong, Z.; Qiu, L.; Wang, T. Comprehensive circular RNA expression profile in radiation-treated HeLa cells and analysis of radioresistance-related circRNAs. PeerJ 2018, 6, e5011. [Google Scholar] [CrossRef] [PubMed]
- Su, H.; Lin, F.; Deng, X.; Shen, L.; Fang, Y.; Fei, Z.; Zhao, L.; Zhang, X.; Pan, H.; Xie, D.; et al. Profiling and bioinformatics analyses reveal differential circular RNA expression in radioresistant esophageal cancer cells. J. Transl. Med. 2016, 14, 225. [Google Scholar] [CrossRef] [PubMed]
- Chaudhry, M.A.; Sachdeva, H.; Omaruddin, R.A. Radiation-induced micro-RNA modulation in glioblastoma cells differing in DNA-repair pathways. DNA Cell Biol. 2010, 29, 553–561. [Google Scholar] [CrossRef]
- Chaudhry, M.A.; Omaruddin, R.A.; Brumbaugh, C.D.; Tariq, M.A.; Pourmand, N. Identification of radiation-induced microRNA transcriptome by next-generation massively parallel sequencing. J. Radiat. Res. 2013, 54, 808–822. [Google Scholar] [CrossRef]
- Fu, X.D. Non-coding RNA: A new frontier in regulatory biology. Natl. Sci. Rev. 2014, 1, 190–204. [Google Scholar] [CrossRef]
- Jonas, S.; Izaurralde, E. Towards a molecular understanding of microRNA-mediated gene silencing. Nat. Rev. Genet. 2015, 16, 421–433. [Google Scholar] [CrossRef]
- Baek, D.; Villen, J.; Shin, C.; Camargo, F.D.; Gygi, S.P.; Bartel, D.P.; Chi, N.C.; Shaw, R.M.; De Val, S.; Kang, G.; et al. Widespread changes in protein synthesis induced by microRNAs. Nature 2008, 455, 1710–1713. [Google Scholar] [CrossRef]
- Fang, S.; Zhang, L.; Guo, J.; Niu, Y.; Wu, Y.; Li, H.; Zhao, L.; Li, X.; Teng, X.; Sun, X.; et al. NONCODEV5: A comprehensive annotation database for long non-coding RNAs. Nucleic Acids Res. 2018, 46, D308–D314. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.L.; Yang, L. Regulation of circRNA biogenesis. RNA Biol. 2015, 12, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Mercer, T.R.; Dinger, M.E.; Mattick, J.S. Long non-coding RNAs: Insights into functions. Nat. Rev. Genet. 2009, 10, 155–159. [Google Scholar] [CrossRef] [PubMed]
- Ulitsky, I.; Bartel, D.P. XLincRNAs: Genomics, evolution, and mechanisms. Cell 2013, 154, 26–46. [Google Scholar] [CrossRef]
- Wang, K.C.; Chang, H.Y. Molecular mechanisms of long noncoding RNAs. Mol. Cell 2011, 43, 904–914. [Google Scholar] [CrossRef]
- Zhou, Y.; Zhang, L.; Fan, J.; Jia, R.; Song, X.; Xu, X.; Dai, L.; Zhuang, A.; Ge, S.; Fan, X. Let-7b overexpression leads to increased radiosensitivity of uveal melanoma cells. Melanoma Res. 2015, 25, 119–126. [Google Scholar] [CrossRef]
- Sun, H.; Ding, C.; Zhang, H.; Gao, J. Let-7 miRNAs sensitize breast cancer stem cells to radiation-induced repression through inhibition of the cyclin D1/Akt1/Wnt1 signaling pathway. Mol. Med. Rep. 2016, 14, 3285–3292. [Google Scholar] [CrossRef]
- Wang, F.; Mao, A.; Tang, J.; Zhang, Q.; Yan, J.; Wang, Y.; Di, C.; Gan, L.; Sun, C.; Zhang, H. microRNA-16-5p enhances radiosensitivity through modulating Cyclin D1/E1–pRb–E2F1 pathway in prostate cancer cells. J. Cell. Physiol. 2019, 234, 13182–13190. [Google Scholar] [CrossRef]
- Song, L.; Lin, C.; Wu, Z.; Gong, H.; Zeng, Y.; Wu, J.; Li, M.; Li, J. MiR-18a impairs DNA damage response through downregulation of Ataxia telangiectasia mutated (ATM) kinase. PLoS ONE 2011, 6, e25454. [Google Scholar] [CrossRef]
- Chen, X.; Wu, L.; Li, D.; Xu, Y.; Zhang, L.; Niu, K.; Kong, R.; Gu, J.; Xu, Z.; Chen, Z.; et al. Radiosensitizing effects of miR-18a-5p on lung cancer stem-like cells via downregulating both ATM and HIF-1α. Cancer Med. 2018, 7, 3834–3847. [Google Scholar] [CrossRef]
- Zhang, X.; Li, Y.; Wang, D.; Wei, X. miR-22 suppresses tumorigenesis and improves radiosensitivity of breast cancer cells by targeting Sirt1. Biol. Res. 2017, 50. [Google Scholar] [CrossRef]
- Wang, P.; Zhang, J.; Zhang, L.; Zhu, Z.; Fan, J.; Chen, L.; Zhuang, L.; Luo, J.; Chen, H.; Liu, L.; et al. MicroRNA 23b regulates autophagy associated with radioresistance of pancreatic cancer cells. Gastroenterology 2013, 145. [Google Scholar] [CrossRef]
- Lal, A.; Pan, Y.; Navarro, F.; Dykxhoorn, D.M.; Moreau, L.; Meire, E.; Bentwich, Z.; Lieberman, J.; Chowdhury, D. MiR-24-mediated downregulation of H2AX suppresses DNA repair in terminally differentiated blood cells. Nat. Struct. Mol. Biol. 2009, 16, 492–498. [Google Scholar] [CrossRef] [PubMed]
- Guo, P.; Lan, J.; Ge, J.; Nie, Q.; Guo, L.; Qiu, Y.; Mao, Q. MiR-26a enhances the radiosensitivity of glioblastoma multiforme cells through targeting of ataxia-telangiectasia mutated. Exp. Cell Res. 2014, 320, 200–208. [Google Scholar] [CrossRef]
- Meng, C.; Liu, Y.; Shen, Y.; Liu, S.; Wang, Z.; Ye, Q.; Liu, H.; Liu, X.; Jia, L. MicroRNA-26b suppresses autophagy in breast cancer cells by targeting DRAM1 mRNA, and is downregulated by irradiation. Oncol. Lett. 2018, 15, 1435–1440. [Google Scholar] [CrossRef] [PubMed]
- Xu, C.G.; Yang, M.F.; Fan, J.X.; Wang, W. MiR-30a and miR-205 are downregulated in hypoxia and modulate radiosensitivity of prostate cancer cells by inhibiting autophagy via TP53INP1. Eur. Rev. Med. Pharmacol. Sci. 2016, 20, 1501–1508. [Google Scholar] [PubMed]
- Cao, K.; Li, J.; Chen, J.; Qian, L.; Wang, A.; Chen, X.; Xiong, W.; Tang, J.; Tang, S.; Chen, Y.; et al. microRNA-33a-5p increases radiosensitivity by inhibiting glycolysis in melanoma. Oncotarget 2017, 8, 83660–83672. [Google Scholar] [CrossRef]
- Duan, W.; Xu, Y.; Dong, Y.; Cao, L.; Tong, J.; Zhou, X. Ectopic expression of miR-34a enhances radiosensitivity of non-small cell lung cancer cells, partly by suppressing the LyGDI signaling pathway. J. Radiat. Res. 2013, 54, 611–619. [Google Scholar] [CrossRef] [PubMed]
- Cortez, M.A.; Valdecanas, D.; Niknam, S.; Peltier, H.J.; Diao, L.; Giri, U.; Komaki, R.; Calin, G.A.; Gomez, D.R.; Chang, J.Y.; et al. In vivo delivery of miR-34a sensitizes lung tumors to radiation through RAD51 regulation. Mol. Ther. Nucleic Acids 2015, 4, e270. [Google Scholar] [CrossRef]
- Yin, H.; Ma, J.; Chen, L.; Piao, S.; Zhang, Y.; Zhang, S.; Ma, H.; Li, Y.; Qu, Y.; Wang, X.; et al. MiR-99a enhances the radiation sensitivity of non-small cell lung cancer by targeting mTOR. Cell. Physiol. Biochem. 2018, 46, 471–481. [Google Scholar] [CrossRef]
- Ng, W.L.; Yan, D.; Zhang, X.; Mo, Y.Y.; Wang, Y. Over-expression of miR-100 is responsible for the low-expression of ATM in the human glioma cell line: M059J. DNA Repair (Amst) 2010, 9, 1170–1175. [Google Scholar] [CrossRef] [PubMed]
- Yan, D.; Ng, W.L.; Zhang, X.; Wang, P.; Zhang, Z.; Mo, Y.Y.; Mao, H.; Hao, C.; Olson, J.J.; Curran, W.J.; et al. Targeting DNA-PKcs and ATM with miR-101 sensitizes tumors to radiation. PLoS ONE 2010, 5, e11397. [Google Scholar] [CrossRef] [PubMed]
- Sun, Q.; Liu, T.; Zhang, T.; Du, S.; Xie, G.; Lin, X.; Chen, L.; Yuan, Y. MiR-101 sensitizes human nasopharyngeal carcinoma cells to radiation by targeting stathmin 1. Mol. Med. Rep. 2015, 11, 3330–3336. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Neijenhuis, S.; Bajrami, I.; Miller, R.; Lord, C.J.; Ashworth, A. Identification of miRNA modulators to PARP inhibitor response. DNA Repair (Amst) 2013, 12, 394–402. [Google Scholar] [CrossRef] [PubMed]
- Deng, X.; Ma, L.; Wu, M.; Zhang, G.; Jin, C.; Guo, Y.; Liu, R. MiR-124 radiosensitizes human glioma cells by targeting CDK4. J. Neurooncol. 2013, 114, 263–274. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.H.; Wang, Q.Q.; Li, H.; Ye, T.; Gao, F.; Liu, Y.C. miR-124 radiosensitizes human esophageal cancer cell TE-1 by targeting CDK4. Genet. Mol. Res. 2016, 15. [Google Scholar] [CrossRef]
- Wang, M.; Meng, B.; Yu, J.; Chen, Q.; Liu, Y. MiR-124 inhibits growth and enhances radiation-induced apoptosis in non-small cell lung cancer by inhibiting STAT3. Cell. Physiol. Biochem. 2018, 44, 2017–2028. [Google Scholar] [CrossRef]
- Fu, Y.; Xiong, J. MicroRNA-124 enhances response to radiotherapy in human epidermal growth factor receptor 2-positive breast cancer cells by targeting signal transducer and activator of transcription 3. Croat. Med. J. 2016, 57, 457–464. [Google Scholar] [CrossRef]
- Gu, H.; Liu, M.; Ding, C.; Wang, X.; Wang, R.; Wu, X.; Fan, R. Hypoxia-responsive miR-124 and miR-144 reduce hypoxia-induced autophagy and enhance radiosensitivity of prostate cancer cells via suppressing PIM1. Cancer Med. 2016, 5, 1174–1182. [Google Scholar] [CrossRef]
- Pedroza-Torres, A.; Campos-Parra, A.D.; Millan-Catalan, O.; Loissell-Baltazar, Y.A.; Zamudio-Meza, H.; De Leon, D.C.; Montalvo-Esquivel, G.; Isla-Ortiz, D.; Herrera, L.A.; Ángeles-Zaragoza, Ó.; et al. MicroRNA-125 modulates radioresistance through targeting p21 in cervical cancer. Oncol. Rep. 2018, 39, 1532–1540. [Google Scholar] [CrossRef] [PubMed]
- Metheetrairut, C.; Adams, B.D.; Nallur, S.; Weidhaas, J.B.; Slack, F.J. Cel-mir-237 and its homologue, hsa-miR-125b, modulate the cellular response to ionizing radiation. Oncogene 2017, 36, 512–524. [Google Scholar] [CrossRef]
- Yang, Q.S.; Jiang, L.P.; He, C.Y.; Tong, Y.N.; Liu, Y.Y. Up-regulation of microRNA-133a inhibits the MEK/ERK signaling pathway to promote cell apoptosis and enhance radio-sensitivity by targeting EGFR in esophageal cancer in vivo and in vitro. J. Cell. Biochem. 2017, 118, 2625–2634. [Google Scholar] [CrossRef]
- Liu, G.; Li, Y.; Gao, X. Overexpression of microRNA-133b sensitizes non-small cell lung cancer cells to irradiation through the inhibition of glycolysis. Oncol. Lett. 2016, 11, 2903–2908. [Google Scholar] [CrossRef]
- Lu, H.J.; Jin, P.Y.; Tang, Y.; Fan, S.H.; Zhang, Z.F.; Wang, F.; Wu, D.M.; Lu, J.; Zheng, Y.L. microRNA-136 inhibits proliferation and promotes apoptosis and radiosensitivity of cervical carcinoma through the NF-κB pathway by targeting E2F1. Life Sci. 2018, 199, 167–178. [Google Scholar] [CrossRef]
- Wang, Y.; Huang, J.W.; Li, M.; Cavenee, W.K.; Mitchell, P.S.; Zhou, X.; Tewari, M.; Furnari, F.B.; Taniguchi, T. MicroRNA-138 modulates DNA damage response by repressing histone H2AX expression. Mol. Cancer Res. 2011, 9, 1100–1111. [Google Scholar] [CrossRef]
- Wu, S.J.; Chen, J.; Wu, B.; Wang, Y.J.; Guo, K.Y. MicroRNA-150 enhances radiosensitivity by inhibiting the AKT pathway in NK/T cell lymphoma. J. Exp. Clin. Cancer Res. 2018, 37, 18. [Google Scholar] [CrossRef]
- Sun, D.; Mu, Y.; Piao, H. MicroRNA-153-3p enhances cell radiosensitivity by targeting BCL2 in human glioma. Biol. Res. 2018, 51, 56. [Google Scholar] [CrossRef]
- Gasparini, P.; Lovat, F.; Fassan, M.; Casadei, L.; Cascione, L.; Jacob, N.K.; Carasi, S.; Palmieri, D.; Costinean, S.; Shapiro, C.L.; et al. Protective role of miR-155 in breast cancer through RAD51 targeting impairs homologous recombination after irradiation. Proc. Natl. Acad. Sci. USA 2014, 111, 4536–4541. [Google Scholar] [CrossRef]
- Moskwa, P.; Buffa, F.M.; Pan, Y.; Panchakshari, R.; Gottipati, P.; Muschel, R.J.; Beech, J.; Kulshrestha, R.; Abdelmohsen, K.; Weinstock, D.M.; et al. MiR-182-mediated downregulation of BRCA1 impacts DNA repair and sensitivity to PARP inhibitors. Mol. Cell 2011, 41, 210–220. [Google Scholar] [CrossRef]
- Li, G.; Wang, Y.; Liu, Y.; Su, Z.; Liu, C.; Ren, S.; Deng, T.; Huang, D.; Tian, Y.; Qiu, Y. miR-185-3p regulates nasopharyngeal carcinoma radioresistance by targeting WNT2B in vitro. Cancer Sci. 2014, 105, 1560–1568. [Google Scholar] [CrossRef]
- Kwon, J.E.; Kim, B.Y.; Kwak, S.Y.; Bae, I.H.; Han, Y.H. Ionizing radiation-inducible microRNA miR-193a-3p induces apoptosis by directly targeting Mcl-1. Apoptosis 2013, 18, 896–909. [Google Scholar] [CrossRef]
- Yi, H.; Liang, B.; Jia, J.; Liang, N.; Xu, H.; Ju, G.; Ma, S.; Liu, X. Differential roles of miR-199a-5p in radiation-induced autophagy in breast cancer cells. FEBS Lett. 2013, 587, 436–443. [Google Scholar] [CrossRef]
- Sun, Q.; Liu, T.; Yuan, Y.; Guo, Z.; Xie, G.; Du, S.; Lin, X.; Xu, Z.; Liu, M.; Wang, W.; et al. MiR-200c inhibits autophagy and enhances radiosensitivity in breast cancer cells by targeting UBQLN1. Int. J. Cancer 2015, 136, 1003–1012. [Google Scholar] [CrossRef]
- Liu, H.Y.; Zhang, Y.Y.; Zhu, B.L.; Feng, F.Z.; Zhang, H.T.; Yan, H.; Zhou, B. MiR-203a-3p regulates the biological behaviors of ovarian cancer cells through mediating the Akt/GSK-3β/Snail signaling pathway by targeting ATM. J. Ovarian Res. 2019, 12. [Google Scholar] [CrossRef]
- Hu, J.L.; He, G.Y.; Lan, X.L.; Zeng, Z.C.; Guan, J.; Ding, Y.; Qian, X.L.; Liao, W.T.; Ding, Y.Q.; Liang, L. Inhibition of ATG12-mediated autophagy by miR-214 enhances radiosensitivity in colorectal cancer. Oncogenesis 2018, 7, 16. [Google Scholar] [CrossRef]
- Zhang, X.; Shi, H.; Lin, S.; Ba, M.; Cui, S. MicroRNA-216a enhances the radiosensitivity of pancreatic cancer cells by inhibiting beclin-1-mediated autophagy. Oncol. Rep. 2015, 34, 1557–1564. [Google Scholar] [CrossRef]
- Liang, L.; Zhu, J.; Zaorsky, N.G.; Deng, Y.; Wu, X.; Liu, Y.; Liu, F.; Cai, G.; Gu, W.; Shen, L.; et al. MicroRNA-223 enhances radiation sensitivity of U87MG cells in vitro and in vivo by targeting ataxia telangiectasia mutated. Int. J. Radiat. Oncol. Biol. Phys. 2014, 88, 955–960. [Google Scholar] [CrossRef]
- Su, H.; Wu, Y.; Fang, Y.; Shen, L.; Fei, Z.; Xie, C.; Chen, M. MicroRNA-301a targets WNT1 to suppress cell proliferation and migration and enhance radiosensitivity in esophageal cancer cells. Oncol. Rep. 2019, 41, 599–607. [Google Scholar] [CrossRef]
- Liang, Z.; Ahn, J.; Guo, D.; Votaw, J.R.; Shim, H. MicroRNA-302 replacement therapy sensitizes breast cancer cells to ionizing radiation. Pharm. Res. 2013, 30, 1008–1016. [Google Scholar] [CrossRef]
- Lv, Q.; Hu, J.X.; Li, Y.J.; Xie, N.; Song, D.D.; Zhao, W.; Yan, Y.F.; Li, B.S.; Wang, P.Y.; Xie, S.Y. MiR-320a effectively suppresses lung adenocarcinoma cell proliferation and metastasis by regulating STAT3 signals. Cancer Biol. Ther. 2017, 18, 142–151. [Google Scholar] [CrossRef]
- Yang, C.X.; Zhang, S.M.; Li, J.; Yang, B.; Ouyang, W.; Mei, Z.J.; Chen, J.; Dai, J.; Ke, S.; Zhou, F.X.; et al. MicroRNA-320 regulates the radiosensitivity of cervical cancer cells C33AR by targeting β-catenin. Oncol. Lett. 2016, 12, 4983–4990. [Google Scholar] [CrossRef]
- Li, G.; Liu, Y.; Su, Z.; Ren, S.; Zhu, G.; Tian, Y.; Qiu, Y. MicroRNA-324-3p regulates nasopharyngeal carcinoma radioresistance by directly targeting WNT2B. Eur. J. Cancer 2013, 49, 2596–2607. [Google Scholar] [CrossRef] [PubMed]
- Mansour, W.Y.; Bogdanova, N.V.; Kasten-Pisula, U.; Rieckmann, T.; Köcher, S.; Borgmann, K.; Baumann, M.; Krause, M.; Petersen, C.; Hu, H.; et al. Aberrant overexpression of miR-421 downregulates ATM and leads to a pronounced DSB repair defect and clinical hypersensitivity in SKX squamous cell carcinoma. Radiother. Oncol. 2013, 106, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Lan, F.; Qin, Q.; Yu, H.; Yue, X. Effect of glycolysis inhibition by miR-448 on glioma radiosensitivity. J. Neurosurg. 2020, 132, 1456–1464. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Liu, H.; Du, L.; Xi, P.; Wang, Q.; Li, Y.; Liu, D. MiR-449a suppresses LDHA-mediated glycolysis to enhance the sensitivity of non-small cell lung cancer cells to ionizing radiation. Oncol. Res. 2018, 26, 547–556. [Google Scholar] [CrossRef]
- Mao, A.; Zhao, Q.; Zhou, X.; Sun, C.; Si, J.; Zhou, R.; Gan, L.; Zhang, H. MicroRNA-449a enhances radiosensitivity by downregulation of c-Myc in prostate cancer cells. Sci. Rep. 2016, 6, 27346. [Google Scholar] [CrossRef]
- Chen, H.; Yao, X.; Di, X.; Zhang, Y.; Zhu, H.; Liu, S.; Chen, T.; Yu, D.; Sun, X. MiR-450a-5p inhibits autophagy and enhances radiosensitivity by targeting dual-specificity phosphatase 10 in esophageal squamous cell carcinoma. Cancer Lett. 2020, 483, 114–126. [Google Scholar] [CrossRef]
- Yang, B.; Kuai, F.; Chen, Z.; Fu, D.; Liu, J.; Wu, Y.; Zhong, J. miR-634 decreases the radioresistance of human breast cancer cells by targeting STAT3. Cancer Biother. Radiopharm. 2020, 35, 241–248. [Google Scholar] [CrossRef]
- Lee, H.C.; Her, N.-G.; Kang, D.; Jung, S.H.; Shin, J.; Lee, M.; Bae, I.H.; Kim, Y.-N.; Park, H.J.; Ko, Y.-G.; et al. Radiation-inducible miR-770-5p sensitizes tumors to radiation through direct targeting of PDZ-binding kinase. Cell Death Dis. 2017, 8, e2693. [Google Scholar] [CrossRef]
- El Bezawy, R.; Cominetti, D.; Fenderico, N.; Zuco, V.; Beretta, G.L.; Dugo, M.; Arrighetti, N.; Stucchi, C.; Rancati, T.; Valdagni, R.; et al. miR-875-5p counteracts epithelial-to-mesenchymal transition and enhances radiation response in prostate cancer through repression of the EGFR-ZEB1 axis. Cancer Lett. 2017, 395, 53–62. [Google Scholar] [CrossRef]
- Xie, C.; Wu, Y.; Fei, Z.; Fang, Y.; Xiao, S.; Su, H. MicroRNA-1275 induces radiosensitization in oesophageal cancer by regulating epithelial-to-mesenchymal transition via Wnt/β-catenin pathway. J. Cell. Mol. Med. 2020, 24, 747–759. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.Y.; Lin, K.C.; Chiou, J.F.; Jeng, S.C.; Cheng, W.H.; Chang, C.L.; Lin, W.C.; Wu, L.L.; Lee, H.L.; Chen, R.J. MicroRNA-17-5p post-transcriptionally regulates p21 expression in irradiated betel quid chewing-related oral squamous cell carcinoma cells. Strahlenther. Onkol. 2013, 189, 675–683. [Google Scholar] [CrossRef]
- Hu, Z.; Zhou, S.; Luo, H.; Ji, M.; Zheng, J.; Huang, F.; Wang, F. miRNA-17 promotes nasopharyngeal carcinoma radioresistance by targeting PTEN/AKT. Int. J. Clin. Exp. Pathol. 2019, 12, 229–240. [Google Scholar] [PubMed]
- Zhang, Y.; Zheng, L.; Ding, Y.; Li, Q.; Wang, R.; Liu, T.; Sun, Q.; Yang, H.; Peng, S.; Wang, W.; et al. MIR-20a induces cell radioresistance by activating the PTEN/PI3K/Akt signaling pathway in hepatocellular carcinoma. Int. J. Radiat. Oncol. Biol. Phys. 2015, 92, 1132–1140. [Google Scholar] [CrossRef]
- Zhou, L.; Bai, H.; Deng, C.; Wang, C.; Xu, R.; Shao, S. MicroRNA-21 is involved in X-ray irradiation resistance in K562 leukaemia cells. Hematology 2015, 20, 343–348. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.L.; Wang, H.; Liu, J.; Wang, Z.X. MicroRNA-21 (miR-21) expression promotes growth, metastasis, and chemo- or radioresistance in non-small cell lung cancer cells by targeting PTEN. Mol. Cell. Biochem. 2013, 372, 35–45. [Google Scholar] [CrossRef]
- Wang, J.; Xu, J.; Fu, J.; Yuan, D.; Guo, F.; Zhou, C.; Shao, C. MiR-29a regulates radiosensitivity in human intestinal cells by targeting PTEN gene. Radiat. Res. 2016, 186, 292–301. [Google Scholar] [CrossRef]
- Vahabi, M.; Pulito, C.; Sacconi, A.; Donzelli, S.; D’Andrea, M.; Manciocco, V.; Pellini, R.; Paci, P.; Sanguineti, G.; Strigari, L.; et al. MiR-96-5p targets PTEN expression affecting radio-chemosensitivity of HNSCC cells. J. Exp. Clin. Cancer Res. 2019, 38, 141. [Google Scholar] [CrossRef]
- Zheng, L.; Zhang, Y.; Liu, Y.; Zhou, M.; Lu, Y.; Yuan, L.; Zhang, C.; Hong, M.; Wang, S.; Li, X. MiR-106b induces cell radioresistance via the PTEN/PI3K/AKT pathways and p21 in colorectal cancer. J. Transl. Med. 2015, 13, 252. [Google Scholar] [CrossRef]
- Xiao, S.; Yang, Z.; Lv, R.; Zhao, J.; Wu, M.; Liao, Y.; Liu, Q. miR-135b contributes to the radioresistance by targeting GSK3β in human glioblastoma multiforme cells. PLoS ONE 2014, 9, e108810. [Google Scholar] [CrossRef]
- Huang, Y.; Tan, D.; Xiao, J.; Li, Q.; Zhang, X.; Luo, Z. MIR-150 contributes to the radioresistance in nasopharyngeal carcinoma cells by targeting glycogen synthase kinase-3β. J. Cancer Res. Ther. 2018, 14, 111–118. [Google Scholar] [CrossRef]
- Yang, F.; Liu, Q.; Hu, C.-M. Epstein-Barr virus-encoded LMP1 increases miR-155 expression, which promotes radioresistance of nasopharyngeal carcinoma via suppressing UBQLN1. Eur. Rev. Med. Pharmacol. Sci. 2015, 19, 4507–4515. [Google Scholar] [PubMed]
- Qu, C.; Liang, Z.; Huang, J.L.; Zhao, R.; Su, C.; Wang, S.; Wang, X.; Zhang, R.; Lee, M.H.; Yang, H. MiR-205 determines the radioresistance of human nasopharyngeal carcinoma by directly targeting PTEN. Cell Cycle 2012, 11, 785–796. [Google Scholar] [CrossRef] [PubMed]
- Tang, Y.; Cui, Y.; Li, Z.; Jiao, Z.; Zhang, Y.; He, Y.; Chen, G.; Zhou, Q.; Wang, W.; Zhou, X.; et al. Radiation-induced miR-208a increases the proliferation and radioresistance by targeting p21 in human lung cancer cells. J. Exp. Clin. Cancer Res. 2016, 35, 7. [Google Scholar] [CrossRef]
- Yang, W.; Sun, T.; Cao, J.; Liu, F.; Tian, Y.; Zhu, W. Downregulation of miR-210 expression inhibits proliferation, induces apoptosis and enhances radiosensitivity in hypoxic human hepatoma cells in vitro. Exp. Cell Res. 2012, 318, 944–954. [Google Scholar] [CrossRef]
- He, X.; Fan, S. hsa-miR-212 modulates the radiosensitivity of glioma cells by targeting BRCA1. Oncol. Rep. 2018, 39, 977–984. [Google Scholar] [CrossRef]
- Zhang, Q.; Zhang, S. MIR-214 promotes radioresistance in Human ovarian cancer cells by targeting PETN. Biosci. Rep. 2017, 37. [Google Scholar] [CrossRef]
- Xue, Q.; Sun, K.; Deng, H.J.; Lei, S.T.; Dong, J.Q.; Li, G.X. Anti-miRNA-221 sensitizes human colorectal carcinoma cells to radiation by upregulating PTEN. World J. Gastroenterol. 2013, 19, 9307–9317. [Google Scholar] [CrossRef]
- Li, G.; Deng, Y.; Wu, Y.; Liang, L.; Zhang, Z. The role of MiR-221 in regulating gastric cancer radiation sensitivity. Int. J. Radiat. Oncol. 2016, 96, E168. [Google Scholar] [CrossRef]
- Zhang, C.; Kang, C.; Wang, P.; Cao, Y.; Lv, Z.; Yu, S.; Wang, G.; Zhang, A.; Jia, Z.; Han, L.; et al. MICRORNA-221 and -222 regulate radiation sensitivity by targeting the PTEN pathway. Int. J. Radiat. Oncol. Biol. Phys. 2011, 80, 240–248. [Google Scholar] [CrossRef]
- Zhang, C.-z.; Han, L.; Zhang, A.-l.; Fu, Y.-c.; Yue, X.; Wang, G.-x.; Jia, Z.-f.; Pu, P.-y.; Zhang, Q.-y.; Kang, C.-s. MicroRNA-221 and microRNA-222 regulate gastric carcinoma cell proliferation and radioresistance by targeting PTEN. BMC Cancer 2010, 10. [Google Scholar] [CrossRef]
- He, J.; Feng, X.; Hua, J.; Wei, L.; Lu, Z.; Wei, W.; Cai, H.; Wang, B.; Shi, W.; Ding, N.; et al. miR-300 regulates cellular radiosensitivity through targeting p53 and apaf1 in human lung cancer cells. Cell Cycle 2017, 16, 1943–1953. [Google Scholar] [CrossRef]
- Yue, X.; Lan, F.; Xia, T. Hypoxic glioma cell-secreted exosomal miR-301a activates Wnt/β-catenin signaling and promotes radiation resistance by targeting TCEAL7. Mol. Ther. 2019, 27, 1939–1949. [Google Scholar] [CrossRef]
- Liu, Y.; Xing, R.; Zhang, X.; Dong, W.; Zhang, J.; Yan, Z.; Li, W.; Cui, J.; Lu, Y. MiR-375 targets the p53 gene to regulate cellular response to ionizing radiation and etoposide in gastric cancer cells. DNA Repair (Amst) 2013, 12, 741–750. [Google Scholar] [CrossRef]
- Duan, X.M.; Liu, X.N.; Li, Y.X.; Cao, Y.Q.; Silayiding, A.; Zhang, R.K.; Wang, J.P. MicroRNA-498 promotes proliferation, migration, and invasion of prostate cancer cells and decreases radiation sensitivity by targeting PTEN. Kaohsiung J. Med. Sci. 2019, 35, 659–671. [Google Scholar] [CrossRef]
- Ma, W.; Yu, J.; Qi, X.; Liang, L.; Zhang, Y.; Ding, Y.; Lin, X.; Li, G.; Ding, Y. Radiation-induced microRNA-622 causes radioresistance in colorectal cancer cells by down-regulating Rb. Oncotarget 2015, 6, 15984–15994. [Google Scholar] [CrossRef]
- Fan, L.; Wang, J.; Cao, Q.; Ding, X.; Li, B. Aberrant miR-1246 expression promotes radioresistance in non-small cell lung cancer: A potential prognostic biomarker and radiotherapy sensitization target. Am. J. Cancer Res. 2020, 10, 314–335. [Google Scholar]
- Chen, G.; Li, Y.; He, Y.; Zeng, B.; Yi, C.; Wang, C.; Zhang, X.; Zhao, W.; Yu, D. Upregulation of circular RNA circATRNL1 to sensitize oral squamous cell carcinoma to irradiation. Mol. Ther. Nucleic Acids 2020, 19, 961–973. [Google Scholar] [CrossRef]
- Chen, L.; Yuan, D.; Yang, Y.; Ren, M. LincRNA-p21 enhances the sensitivity of radiotherapy for gastric cancer by targeting the beta-catenin signaling pathway. J. Cell Biochem 2019, 120, 6178–6187. [Google Scholar] [CrossRef]
- Wang, G.; Li, Z.; Zhao, Q.; Zhu, Y.; Zhao, C.; Li, X.; Ma, Z.; Li, X.; Zhang, Y. LincRNA-p21 enhances the sensitivity of radiotherapy for human colorectal cancer by targeting the Wnt/beta-catenin signaling pathway. Oncol Rep. 2014, 31, 1839–1845. [Google Scholar] [CrossRef]
- Liu, Y.; Yue, P.; Zhou, T.; Zhang, F.; Wang, H.; Chen, X. LncRNA MEG3 enhances (131)I sensitivity in thyroid carcinoma via sponging miR-182. Biomed. Pharmacother. 2018, 105, 1232–1239. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, C.; Chen, C.; Wu, F.; Shen, P.; Zhang, P.; He, G.; Li, X. Long non-coding RNA NEAT1 regulates epithelial membrane protein 2 expression to repress nasopharyngeal carcinoma migration and irradiation-resistance through miR-101-3p as a competing endogenous RNA mechanism. Oncotarget 2017, 8, 70156–70171. [Google Scholar] [CrossRef]
- Zou, Y.; Yao, S.; Chen, X.; Liu, D.; Wang, J.; Yuan, X.; Rao, J.; Xiong, H.; Yu, S.; Yuan, X.; et al. LncRNA OIP5-AS1 regulates radioresistance by targeting DYRK1A through miR-369-3p in colorectal cancer cells. Eur. J. Cell Biol. 2018, 97, 369–378. [Google Scholar] [CrossRef]
- Zhang, H.; Luo, H.; Hu, Z.; Peng, J.; Jiang, Z.; Song, T.; Wu, B.; Yue, J.; Zhou, R.; Xie, R.; et al. Targeting WISP1 to sensitize esophageal squamous cell carcinoma to irradiation. Oncotarget 2015, 6, 6218–6234. [Google Scholar] [CrossRef][Green Version]
- Gou, C.; Han, P.; Li, J.; Gao, L.; Ji, X.; Dong, F.; Su, Q.; Zhang, Y.; Liu, X. Knockdown of lncRNA BLACAT1 enhances radiosensitivity of head and neck squamous cell carcinoma cells by regulating PSEN1. Br. J. Radiol. 2020, 93, 20190154. [Google Scholar] [CrossRef]
- Lai, Y.; Chen, Y.; Lin, Y.; Ye, L. Down-regulation of LncRNA CCAT1 enhances radiosensitivity via regulating miR-148b in breast cancer. Cell Biol. Int. 2018, 42, 227–236. [Google Scholar] [CrossRef]
- Guan, Y.; Cao, Z.; Du, J.; Liu, T.; Wang, T. Circular RNA circPITX1 knockdown inhibits glycolysis to enhance radiosensitivity of glioma cells by miR-329-3p/NEK2 axis. Cancer Cell Int. 2020, 20. [Google Scholar] [CrossRef]
- Liu, Y.; Yao, K.; Zhang, K.; Wang, J.; Dai, Q.; Wang, R. Circular RNA PTK2 modifies the progression and radiosensitivity in gastric cancer via miR-369-3p/ZEB1 axis. RSC Adv. 2020, 10, 1711–1723. [Google Scholar] [CrossRef]
- Zhang, M.; Gao, C.; Yang, Y.; Li, G.; Dong, J.; Ai, Y.; Chen, N.; Li, W. Long noncoding RNA CRNDE/PRC2 participated in the radiotherapy resistance of human lung adenocarcinoma through targeting p21 expression. Oncol. Res. 2018, 26, 1245–1255. [Google Scholar] [CrossRef]
- Zhang, J.; Li, W. Long noncoding RNA CYTOR sponges miR-195 to modulate proliferation, migration, invasion and radiosensitivity in nonsmall cell lung cancer cells. Biosci. Rep. 2018, 38. [Google Scholar] [CrossRef]
- Zhang, H.; Hua, Y.; Jiang, Z.; Yue, J.; Shi, M.; Zhen, X.; Zhang, X.; Yang, L.; Zhou, R.; Wu, S. Cancer-associated fibroblast-promoted LncRNA DNM3OS confers radioresistance by regulating DNA damage response in esophageal squamous cell carcinoma. Clin. Cancer Res. 2019, 25, 1989–2000. [Google Scholar] [CrossRef]
- Dou, Q.; Xu, Y.; Zhu, Y.; Hu, Y.; Yan, Y.; Yan, H. LncRNA FAM83H-AS1 contributes to the radioresistance, proliferation, and metastasis in ovarian cancer through stabilizing HuR protein. Eur. J. Pharmacol. 2019, 852, 134–141. [Google Scholar] [CrossRef]
- Jing, L.; Yuan, W.; Ruofan, D.; Jinjin, Y.; Haifeng, Q. HOTAIR enhanced aggressive biological behaviors and induced radio-resistance via inhibiting p21 in cervical cancer. Tumour Biol. 2015, 36, 3611–3619. [Google Scholar] [CrossRef]
- Jiang, Y.; Li, Z.; Zheng, S.; Chen, H.; Zhao, X.; Gao, W.; Bi, Z.; You, K.; Wang, Y.; Li, W.; et al. The long non-coding RNA HOTAIR affects the radiosensitivity of pancreatic ductal adenocarcinoma by regulating the expression of Wnt inhibitory factor 1. Tumour Biol. 2016, 37, 3957–3967. [Google Scholar] [CrossRef]
- Hu, X.; Ding, D.; Zhang, J.; Cui, J. Knockdown of lncRNA HOTAIR sensitizes breast cancer cells to ionizing radiation through activating miR-218. Biosci. Rep. 2019, 39. [Google Scholar] [CrossRef]
- Wu, C.; Yang, L.; Qi, X.; Wang, T.; Li, M.; Xu, K. Inhibition of long non-coding RNA HOTAIR enhances radiosensitivity via regulating autophagy in pancreatic cancer. Cancer Manag. Res. 2018, 10, 5261–5271. [Google Scholar] [CrossRef]
- Chen, C.; Wang, K.; Wang, Q.; Wang, X. LncRNA HULC mediates radioresistance via autophagy in prostate cancer cells. Braz. J. Med. Biol. Res. 2018, 51, e7080. [Google Scholar] [CrossRef]
- Zhang, N.; Zeng, X.; Sun, C.; Guo, H.; Wang, T.; Wei, L.; Zhang, Y.; Zhao, J.; Ma, X. LncRNA LINC00963 promotes tumorigenesis and radioresistance in breast cancer by sponging miR-324-3p and inducing ACK1 expression. Mol. Ther. Nucleic Acids 2019, 18, 871–881. [Google Scholar] [CrossRef]
- Wang, B.; Zheng, J.; Li, R.; Tian, Y.; Lin, J.; Liang, Y.; Sun, Q.; Xu, A.; Zheng, R.; Liu, M.; et al. Long noncoding RNA LINC02582 acts downstream of miR-200c to promote radioresistance through CHK1 in breast cancer cells. Cell Death Dis. 2019, 10, 764. [Google Scholar] [CrossRef]
- Shen, Y.; Liu, Y.; Sun, T.; Yang, W. LincRNA-p21 knockdown enhances radiosensitivity of hypoxic tumor cells by reducing autophagy through HIF-1/Akt/mTOR/P70S6K pathway. Exp. Cell Res. 2017, 358, 188–198. [Google Scholar] [CrossRef]
- Yang, P.; Yang, Y.; An, W.; Xu, J.; Zhang, G.; Jie, J.; Zhang, Q. The long noncoding RNA-ROR promotes the resistance of radiotherapy for human colorectal cancer cells by targeting the p53/miR-145 pathway. J. Gastroenterol. Hepatol. 2017, 32, 837–845. [Google Scholar] [CrossRef]
- Wang, X.; Liu, H.; Shi, L.; Yu, X.; Gu, Y.; Sun, X. LINP1 facilitates DNA damage repair through non-homologous end joining (NHEJ) pathway and subsequently decreases the sensitivity of cervical cancer cells to ionizing radiation. Cell Cycle 2018, 17, 439–447. [Google Scholar] [CrossRef]
- Li, Z.; Zhou, Y.; Tu, B.; Bu, Y.; Liu, A.; Kong, J. Long noncoding RNA MALAT1 affects the efficacy of radiotherapy for esophageal squamous cell carcinoma by regulating Cks1 expression. J. Oral Pathol. Med. 2017, 46, 583–590. [Google Scholar] [CrossRef]
- Jin, C.; Yan, B.; Lu, Q.; Lin, Y.; Ma, L. The role of MALAT1/miR-1/slug axis on radioresistance in nasopharyngeal carcinoma. Tumour Biol. 2016, 37, 4025–4033. [Google Scholar] [CrossRef]
- Lu, H.; He, Y.; Lin, L.; Qi, Z.; Ma, L.; Li, L.; Su, Y. Long non-coding RNA MALAT1 modulates radiosensitivity of HR-HPV+ cervical cancer via sponging miR-145. Tumour Biol. 2016, 37, 1683–1691. [Google Scholar] [CrossRef] [PubMed]
- Lu, Y.; Li, T.; Wei, G.; Liu, L.; Chen, Q.; Xu, L.; Zhang, K.; Zeng, D.; Liao, R. The long non-coding RNA NEAT1 regulates epithelial to mesenchymal transition and radioresistance in through miR-204/ZEB1 axis in nasopharyngeal carcinoma. Tumour Biol. 2016, 37, 11733–11741. [Google Scholar] [CrossRef] [PubMed]
- Han, D.; Wang, J.; Cheng, G. LncRNA NEAT1 enhances the radio-resistance of cervical cancer via miR-193b-3p/CCND1 axis. Oncotarget 2018, 9, 2395–2409. [Google Scholar] [CrossRef]
- Shi, R.; Wu, P.; Liu, M.; Chen, B.; Cong, L. Knockdown of lncRNA PCAT6 enhances radiosensitivity in triple-negative breast cancer cells by regulating miR-185-5p/TPD52 axis. Onco Targets Ther. 2020, 13, 3025–3037. [Google Scholar] [CrossRef]
- Liu, J.; Sun, X.; Zhu, H.; Qin, Q.; Yang, X.; Sun, X. Long noncoding RNA POU6F2-AS2 is associated with oesophageal squamous cell carcinoma. J. Biochem. 2016, 160, 195–204. [Google Scholar] [CrossRef]
- Wu, D.; Li, Y.; Zhang, H.; Hu, X. Knockdown of lncrna PVT1 enhances radiosensitivity in non-small cell lung cancer by sponging Mir-195. Cell. Physiol. Biochem. 2017, 42, 2453–2466. [Google Scholar] [CrossRef]
- He, Y.; Jing, Y.; Wei, F.; Tang, Y.; Yang, L.; Luo, J.; Yang, P.; Ni, Q.; Pang, J.; Liao, Q.; et al. Long non-coding RNA PVT1 predicts poor prognosis and induces radioresistance by regulating DNA repair and cell apoptosis in nasopharyngeal carcinoma. Cell Death Dis. 2018, 9, 235. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, W.; Lian, J.; Zhang, H.; Yu, B.; Zhang, M.; Wei, F.; Wu, J.; Jiang, J.; Jia, Y.; et al. The lncRNA PVT1 regulates nasopharyngeal carcinoma cell proliferation via activating the KAT2A acetyltransferase and stabilizing HIF-1alpha. Cell Death Differ. 2020, 27, 695–710. [Google Scholar] [CrossRef] [PubMed]
- Yu, Z.; Wang, G.; Zhang, C.; Liu, Y.; Chen, W.; Wang, H.; Liu, H. LncRNA SBF2-AS1 affects the radiosensitivity of non-small cell lung cancer via modulating microRNA-302a/MBNL3 axis. Cell Cycle 2020, 19, 300–316. [Google Scholar] [CrossRef]
- Song, W.; Zhang, J.; Xia, Q.; Sun, M. Down-regulated lncRNA TP73-AS1 reduces radioresistance in hepatocellular carcinoma via the PTEN/Akt signaling pathway. Cell Cycle 2019, 18, 3177–3188. [Google Scholar] [CrossRef]
- Jiang, H.; Hu, X.; Zhang, H.; Li, W. Down-regulation of LncRNA TUG1 enhances radiosensitivity in bladder cancer via suppressing HMGB1 expression. Radiat. Oncol. 2017, 12, 65. [Google Scholar] [CrossRef]
- Fotouhi Ghiam, A.; Taeb, S.; Huang, X.; Huang, V.; Ray, J.; Scarcello, S.; Hoey, C.; Jahangiri, S.; Fokas, E.; Loblaw, A.; et al. Long non-coding RNA urothelial carcinoma associated 1 (UCA1) mediates radiation response in prostate cancer. Oncotarget 2017, 8, 4668–4689. [Google Scholar] [CrossRef]
- Fan, L.; Huang, C.; Li, J.; Gao, T.; Lin, Z.; Yao, T. Long non-coding RNA urothelial cancer associated 1 regulates radioresistance via the hexokinase 2/glycolytic pathway in cervical cancer. Int. J. Mol. Med. 2018, 42, 1875–1884. [Google Scholar] [CrossRef] [PubMed]
- Kakarougkas, A.; Jeggo, P.A. DNA DSB repair pathway choice: An orchestrated handover mechanism. Br. J. Radiol 2014, 87, 20130685. [Google Scholar] [CrossRef]
- Davis, A.J.; Chen, D.J. DNA double strand break repair via non-homologous end-joining. Transl. Cancer Res. 2013, 2, 130–143. [Google Scholar] [CrossRef]
- Wright, W.D.; Shah, S.S.; Heyer, W.D. Homologous recombination and the repair of DNA double-strand breaks. J. Biol. Chem. 2018, 293, 10524–10535. [Google Scholar] [CrossRef]
- Thapar, R. Regulation of DNA double-strand break repair by non-coding RNAs. Molecules 2018, 23, 2789. [Google Scholar] [CrossRef] [PubMed]
- Bekker-Jensen, S.; Mailand, N. Assembly and function of DNA double-strand break repair foci in mammalian cells. DNA Repair (Amst) 2010, 9, 1219–1228. [Google Scholar] [CrossRef] [PubMed]
- Zhou, L.; Yuan, R.; Serggio, L. Molecular mechanisms of irradiation-induced apoptosis. Front. Biosci. 2003, 8, d9–d19. [Google Scholar] [CrossRef] [PubMed]
- Sladky, V.; Schuler, F.; Fava, L.L.; Villunger, A. The resurrection of the PIDDosome—Emerging roles in the DNA-damage response and centrosome surveillance. J. Cell Sci. 2017, 130, 3779–3787. [Google Scholar] [CrossRef] [PubMed]
- Ray, P.; Guha, D.; Chakraborty, J.; Banerjee, S.; Adhikary, A.; Chakraborty, S.; Das, T.; Sa, G. Crocetin exploits p53-induced death domain (PIDD) and FAS-associated death domain (FADD) proteins to induce apoptosis in colorectal cancer. Sci. Rep. 2016, 6, 32979. [Google Scholar] [CrossRef]
- Pistritto, G.; Trisciuoglio, D.; Ceci, C.; Garufi, A.; D’Orazi, G. Apoptosis as anticancer mechanism: Function and dysfunction of its modulators and targeted therapeutic strategies. Aging (Albany NY) 2016, 8, 603–619. [Google Scholar] [CrossRef]
- Villanova, L.; Careccia, S.; De Maria, R.; Fiori, M.E. Micro-economics of apoptosis in cancer: ncRNAs modulation of BCL-2 family members. Int. J. Mol. Sci. 2018, 19, 958. [Google Scholar] [CrossRef]
- Rossi, M.N.; Antonangeli, F. LncRNAs: New players in apoptosis control. Int. J. Cell Biol. 2014, 2014, 473857. [Google Scholar] [CrossRef]
- Borras-Fresneda, M.; Barquinero, J.F.; Gomolka, M.; Hornhardt, S.; Rossler, U.; Armengol, G.; Barrios, L. Differences in DNA repair capacity, cell death and transcriptional response after irradiation between a radiosensitive and a radioresistant cell line. Sci Rep. 2016, 6, 27043. [Google Scholar] [CrossRef]
- Slack, F.J.; Chinnaiyan, A.M. The role of non-coding RNAs in oncology. Cell 2019, 179, 1033–1055. [Google Scholar] [CrossRef]
- Vousden, K.H.; Prives, C. Blinded by the light: The growing complexity of p53. Cell 2009, 137, 413–431. [Google Scholar] [CrossRef] [PubMed]
- Indovina, P.; Pentimalli, F.; Casini, N.; Vocca, I.; Giordano, A. RB1 dual role in proliferation and apoptosis: Cell fate control and implications for cancer therapy. Oncotarget 2015, 6, 17873–17890. [Google Scholar] [CrossRef] [PubMed]
- Knezevic, D.; Brash, D.E. Role of E2F1 in apoptosis: A case study in feedback loops. Cell Cycle 2004, 3, 729–732. [Google Scholar] [CrossRef] [PubMed]
- Xu, N.; Lao, Y.; Zhang, Y.; Gillespie, D.A. Akt: A double-edged sword in cell proliferation and genome stability. J. Oncol. 2012, 2012, 951724. [Google Scholar] [CrossRef] [PubMed]
- Fresno Vara, J.Á.; Casado, E.; De Castro, J.; Cejas, P.; Belda-Iniesta, C.; González-Barón, M. P13K/Akt signalling pathway and cancer. Cancer Treat. Rev. 2004, 30, 193–204. [Google Scholar] [CrossRef]
- Jiang, P.; Rao, E.Y.; Meng, N.; Zhao, Y.; Wang, J.J. MicroRNA-17-92 significantly enhances radioresistance in human mantle cell lymphoma cells. Radiat. Oncol. 2010, 5, 100. [Google Scholar] [CrossRef]
- Ummanni, R.; Teller, S.; Junker, H.; Zimmermann, U.; Venz, S.; Scharf, C.; Giebel, J.; Walther, R. Altered expression of tumor protein D52 regulates apoptosis and migration of prostate cancer cells. FEBS J. 2008, 275, 5703–5713. [Google Scholar] [CrossRef]
- White, E.; Mehnert, J.M.; Chan, C.S. Autophagy, metabolism, and cancer. Clin. Cancer Res. 2015, 21, 5037–5046. [Google Scholar] [CrossRef]
- Mizushima, N.; Ohsumi, Y.; Yoshimori, T. Autophagosome formation in mammalian cells. Cell Struct. Funct. 2002, 27, 421–429. [Google Scholar] [CrossRef]
- Tam, S.Y.; Wu, V.W.; Law, H.K. Influence of autophagy on the efficacy of radiotherapy. Radiat. Oncol. 2017, 12, 57. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Yang, Y.; Yang, X.; Zhu, H.; Guo, Q.; Chen, X.; Zhang, H.; Cheng, H.; Sun, X. Autophagy and its function in radiosensitivity. Tumour Biol. 2015, 36, 4079–4087. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Wang, H.; Huang, L.; Zhao, Y.; Wang, J. Crosstalk between autophagy and intracellular radiation response (Review). Int. J. Oncol. 2016, 49, 2217–2226. [Google Scholar] [CrossRef] [PubMed]
- Sharma, K.; Le, N.; Alotaibi, M.; Gewirtz, D.A. Cytotoxic autophagy in cancer therapy. Int. J. Mol. Sci. 2014, 15, 10034–10051. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.Y.; Liu, Y.W.; Wang, Y.K.; Lin, T.H.; Li, Y.Z.; Chen, S.H.; Lee, Y.R. Ionizing radiation induces autophagy in human oral squamous cell carcinoma. J. BUON 2014, 19, 137–144. [Google Scholar] [PubMed]
- Kang, R.; Zeh, H.J.; Lotze, M.T.; Tang, D. The Beclin 1 network regulates autophagy and apoptosis. Cell Death Differ. 2011, 18, 571–580. [Google Scholar] [CrossRef]
- Rothenberg, C.; Srinivasan, D.; Mah, L.; Kaushik, S.; Peterhoff, C.M.; Ugolino, J.; Fang, S.; Cuervo, A.M.; Nixon, R.A.; Monteiro, M.J. Ubiquilin functions in autophagy and is degraded by chaperone-mediated autophagy. Hum. Mol. Genet. 2010, 19, 3219–3232. [Google Scholar] [CrossRef]
- Guo, X.; Xiao, H.; Guo, S.; Li, J.; Wang, Y.; Chen, J.; Lou, G. Long noncoding RNA HOTAIR knockdown inhibits autophagy and epithelial-mesenchymal transition through the Wnt signaling pathway in radioresistant human cervical cancer HeLa cells. J. Cell. Physiol. 2019, 234, 3478–3489. [Google Scholar] [CrossRef]
- Reinhardt, H.C.; Schumacher, B. The p53 network: Cellular and systemic DNA damage responses in aging and cancer. Trends Genet. 2012, 28, 128–136. [Google Scholar] [CrossRef]
- Maier, P.; Hartmann, L.; Wenz, F.; Herskind, C. Cellular pathways in response to ionizing radiation and their targetability for tumor radiosensitization. Int. J. Mol. Sci. 2016, 17, 102. [Google Scholar] [CrossRef]
- Pawlik, T.M.; Keyomarsi, K. Role of cell cycle in mediating sensitivity to radiotherapy. Int. J. Radiat. Oncol. Biol. Phys. 2004, 59, 928–942. [Google Scholar] [CrossRef]
- Yang, X.; Liu, W.; Xu, X.; Zhu, J.; Wu, Y.; Zhao, K.; He, S.; Li, M.; Wu, Y.; Zhang, S.; et al. Downregulation of long noncoding RNA UCA1 enhances the radiosensitivity and inhibits migration via suppression of epithelialmesenchymal transition in colorectal cancer cells. Oncol. Rep. 2018, 40, 1554–1564. [Google Scholar] [CrossRef] [PubMed]
- Fadaka, A.; Ajiboye, B.; Ojo, O.; Adewale, O.; Olayide, I.; Emuowhochere, R. Biology of glucose metabolization in cancer cells. J. Oncol. Sci. 2017, 3, 45–51. [Google Scholar] [CrossRef]
- Yu, L.; Chen, X.; Sun, X.; Wang, L.; Chen, S. The glycolytic switch in tumors: How many players are involved? J. Cancer 2017, 8, 3430–3440. [Google Scholar] [CrossRef] [PubMed]
- Marbaniang, C.; Kma, L. Dysregulation of glucose metabolism by oncogenes and tumor suppressors in cancer cells. Asian Pac. J. Cancer Prev. 2018, 19, 2377–2390. [Google Scholar]
- Lin, J.; Xia, L.; Liang, J.; Han, Y.; Wang, H.; Oyang, L.; Tan, S.; Tian, Y.; Rao, S.; Chen, X.; et al. The roles of glucose metabolic reprogramming in chemo- and radio-resistance. J. Exp. Clin. Cancer Res. 2019, 38, 218. [Google Scholar] [CrossRef]
- Zhou, J.; Xu, D.; Xie, H.; Tang, J.; Liu, R.; Li, J.; Wang, S.; Chen, X.; Su, J.; Zhou, X.; et al. miR-33a functions as a tumor suppressor in melanoma by targeting HIF-1α. Cancer Biol. Ther. 2015, 16, 846–855. [Google Scholar] [CrossRef]
- Gu, Z.; Xia, J.; Xu, H.; Frech, I.; Tricot, G.; Zhan, F. NEK2 promotes aerobic glycolysis in multiple myeloma through regulating splicing of pyruvate kinase. J. Hematol. Oncol. 2017, 10, 1–11. [Google Scholar] [CrossRef]
- Zhan, T.; Rindtorff, N.; Boutros, M. Wnt signaling in cancer. Oncogene 2017, 36, 1461–1473. [Google Scholar] [CrossRef]
- Jackstadt, R.; Hodder, M.C.; Sansom, O.J. WNT and β-catenin in cancer: Genes and therapy. Annu. Rev. Cancer Biol. 2020, 4, 177–196. [Google Scholar] [CrossRef]
- El-Sahli, S.; Xie, Y.; Wang, L.; Liu, S. Wnt signaling in cancer metabolism and immunity. Cancers 2019, 11, 904. [Google Scholar] [CrossRef]
- Yang, Y.; Zhou, H.; Zhang, G.; Xue, X. Targeting the canonical Wnt/beta-catenin pathway in cancer radioresistance: Updates on the molecular mechanisms. J. Cancer Res. Ther. 2019, 15, 272–277. [Google Scholar] [CrossRef]
- Navarro, F.; Lieberman, J. miR-34 and p53: New insights into a complex functional relationship. PLoS ONE 2015, 10. [Google Scholar] [CrossRef] [PubMed]
- Schmitt, A.M.; Garcia, J.T.; Hung, T.; Flynn, R.A.; Shen, Y.; Qu, K.; Payumo, A.Y.; Peres-Da-Silva, A.; Broz, D.K.; Baum, R.; et al. An inducible long noncoding RNA amplifies DNA damage signaling. Nat. Genet. 2016, 48, 1370–1376. [Google Scholar] [CrossRef] [PubMed]
- Chaudhary, R.; Lal, A. Long noncoding RNAs in the p53 network. Wiley Interdiscip. Rev. RNA 2017, 8, e1410. [Google Scholar] [CrossRef]

| miRNA | Target | Pathway | Cancer Type | Reference |
|---|---|---|---|---|
| miRNA Promoting Radiosensitivity | ||||
| let-7 | Cyclin D1 | Cell cycle | Uveal melanoma *, breast cancer | [16,17] |
| miR-16 | Cyclin D1 | Cell cycle | Prostate cancer | [18] |
| miR-18a | ATM | DSB repair | Breast cancer, lung cancer * | [19,20] |
| HIF-1α | - | Lung cancer * | [20] | |
| miR-22 | SIRT1 | Apoptosis | Breast cancer | [21] |
| miR-23b | ATG12 | Autophagy | Pancreatic cancer * | [22] |
| miR-24 | H2AX | DSB repair | Leukemia | [23] |
| miR-26a | ATM | DSB repair | Glioma | [24] |
| miR-26b | DRAM1 | Autophagy | Breast cancer | [25] |
| miR-30a | TP53INP1 | Autophagy | Prostate cancer | [26] |
| miR-33a | HIF-1α | Glycolysis | Melanoma * | [27] |
| miR-34a | LyGDI | Apoptosis | NSCLC | [28] |
| RAD51 | DSB repair | Lung cancer * | [29] | |
| miR-99a | mTOR | Apoptosis | NSCLC * | [30] |
| miR-100 | ATM | - | Glioma | [31] |
| miR-101 | DNA-PKcs | - | Glioma * | [32] |
| ATM | - | Glioma * | [32] | |
| STMN1 | Autophagy | NPC | [33] | |
| miR-107 | RAD51 | DSB repair | Ovarian cancer | [34] |
| miR-124 | CDK4 | Apoptosis | Glioma, ESCC | [35,36] |
| STAT3 | Apoptosis | NSCLC *, breast cancer | [37,38] | |
| PIM 1 | - | Prostate cancer | [39] | |
| miR-125a | p21 | - | Cervical cancer | [40] |
| miR-125b | c-Jun | Apoptosis | Breast cancer | [41] |
| miR-133a | EGFR | Apoptosis | ESCC | [42] |
| miR-133b | PKM2 | Glycolysis | NSCLC | [43] |
| miR-136 | E2F1 | Apoptosis | Cervical cancer | [44] |
| miR-138 | H2AX | DSB repair | Osteosarcoma | [45] |
| miR-144 | PIM 1 | - | Prostate cancer | [39] |
| miR-150 | AKT | Apoptosis | NK/T cell lymphoma * | [46] |
| miR-153 | Bcl-2 | Apoptosis | Glioma * | [47] |
| miR-155 | RAD51 | DSB repair | Triple negative breast cancer * | [48] |
| miR-182 | BRCA1 | DSB repair | Breast cancer | [49] |
| miR-185 | Wnt2B | Wnt/β-catenin | NPC | [50] |
| miR-193a | Mcl-1 | Apoptosis | GBM, cervical cancer | [51] |
| miR-199a | Beclin-1, DRAM1 | Autophagy | Breast cancer | [52] |
| miR-200c | UBQLN1 | Autophagy | Breast cancer | [53] |
| miR-203a | ATM | Apoptosis, cell cycle, migration | Ovarian cancer | [54] |
| miR-205 | TP53INP1 | Autophagy | Prostate cancer | [26] |
| miR-214 | ATG12 | Autophagy | CRC * | [55] |
| miR-216a | Beclin-1 | Autophagy | Pancreatic cancer * | [56] |
| miR-222 | RAD51 | DSB repair | Ovarian cancer | [34] |
| miR-223 | ATM | - | Glioma * | [57] |
| miR-301a | Wnt | Wnt/β-catenin, migration | ESCC | [58] |
| miR-302a | AKT, RAD52 | - | Breast cancer * | [59] |
| miR-320a | STAT3 | Apoptosis, metastasis | NSCLC * | [60] |
| β-catenin | Wnt/β-catenin | Cervical cancer | [61] | |
| miR-324-3p | Wnt2B | Wnt/ β-catenin | NPC | [62] |
| miR-421 | ATM | DSB repair | SCC | [63] |
| miR-448 | HIF-1α | Glycolysis | Glioma | [64] |
| miR-449a | LDHA | Glycolysis, apoptosis | Lung cancer | [65] |
| miR-449a | c-MYC | Cell cycle | Prostate cancer * | [66] |
| miR-450a | DUSP10 | Autophagy | ESCC * | [67] |
| miR-634 | STAT3 | Apoptosis | Breast cancer | [68] |
| miR-770 | PBK | Apoptosis | CRC * | [69] |
| miR-875 | EGFR | DSB repair | Prostate cancer * | [70] |
| miR-1275 | Wnt1 | Wnt/β-catenin, EMT, | ESCC * | [71] |
| miRNA Promoting Radioresistance | ||||
| miR-17 | P21 | Apoptosis | OSCC * | [72] |
| PTEN | Apoptosis, cell cycle | NPC | [73] | |
| miR-20a | PTEN | Apoptosis | HCC | [74] |
| miR-21 | PTEN | Migration | Leukemia, NSCLC | [75,76] |
| miR-29a | PTEN | - | CRC | [77] |
| miR-96 | PTEN | - | HNSCC | [78] |
| miR-106b | PTEN, p21 | Apoptosis, cell cycle | CRC | [79] |
| miR-135b | GSK3β | - | GBM | [80] |
| miR-150 | GSK3β | - | NPC | [81] |
| miR-155 | UBQLN1 | - | NPC | [82] |
| miR-205 | PTEN | Apoptosis | NPC | [83] |
| miR-208a | p21 | Lung cancer | [84] | |
| miR-210 | AIFM3 | Apoptosis | Hepatoma | [85] |
| miR-212 | BRCA1 | Apoptosis | Glioma | [86] |
| miR-214 | PTEN | - | Ovarian cancer * | [87] |
| miR-221/miR-222 | PTEN | Apoptosis, migration | CRC, gastric cancer, GBM | [88,89,90,91] |
| miR-300 | p53, Apaf-1 | Apoptosis, cell cycle, senescence | Lung cancer | [92] |
| miR-301a | TCEAL7 | Wnt/β-catenin, apoptosis | GBM * | [93] |
| miR-375 | p53 | Apoptosis, cell cycle | Gastric cancer | [94] |
| miR-498 | PTEN | Apoptosis, migration | Prostate cancer | [95] |
| miR-622 | Rb | Apoptosis | CRC | [96] |
| miR-1246 | mTOR | Autophagy | NSCLC | [97] |
| lncRNA | Target | Pathway | Cancer Type | Reference |
|---|---|---|---|---|
| lncRNA Promoting Radiosensitivity | ||||
| circATRNL1 | miR-23a/PTEN | Apoptosis, cell cycle | OSCC | [98] |
| lincRNA-p21 | β-catenin | Wnt/β-catenin | Gastric cancer | [99] |
| - | Wnt/β-catenin | CRC | [100] | |
| MEG3 | miR-182 | DSB repair | Thyroid cancer | [101] |
| NEAT1 | miR-101/EMP2 | Apoptosis, EMT | NPC * | [102] |
| OIP5-AS1 | miR-369/DYRK1A | Apoptosis | CRC | [103] |
| lncRNA Promoting Radioresistance | ||||
| BOKAS | WISP1 | Wnt/β-catenin | ESCC | [104] |
| BLACAT1 | PSEN1 | Apoptosis, cell cycle DSB repair | HNSCC | [105] |
| CCAT1 | miR-148 | Apoptosis | Breast cancer | [106] |
| circPITX1 | miR-329-3p/NEK2 | Glycolysis | Glioma * | [107] |
| circPTK2 | miR-369/ZEB1 | Apoptosis | Gastric cancer * | [108] |
| CRNDE | - | Apoptosis | Lung adenocarcinoma | [109] |
| CYTOR | - | Apoptosis | NSCLC | [110] |
| DNM3OS | - | DSB repair | ESCC * | [111] |
| FAM83H-AS1 | HuR | DSB repair | Ovarian cancer | [112] |
| HOTAIR | p21 | Apoptosis, cell cycle | Cervical cancer* | [113] |
| WIF-1 | Wnt/β-catenin | Pancreatic cancer | [114] | |
| miR-218 | Apoptosis, cell cycle, DSB repair | Breast cancer | [115] | |
| LC3-II, ATG7 | Autophagy, apoptosis | Pancreatic cancer | [116] | |
| HULC | LC3B-II, Beclin-1 | Apoptosis, autophagy | Prostate cancer | [117] |
| LINC00963 | miR-324-3p/ACK1 | DSB repair | Breast cancer | [118] |
| LINC02582 | miR-200c/USP7/CHK1 | DSB repair | Breast cancer * | [119] |
| lincRNA-p21 | HIF-1α | Autophagy | Hepatoma, Glioma | [120] |
| lincRNA-ROR | P53, miR-145 | Apoptosis | CRC * | [121] |
| LINP1 | Ku80, DNA-PKcs | Apoptosis, DSB repair | Cervical cancer | [122] |
| MALAT1 | Cks1 | Apoptosis | ESCC * | [123] |
| miR-1/SLUG | Apoptosis | NPC * | [124] | |
| miR-145 | Apoptosis, cell cycle | Cervical cancer | [125] | |
| NEAT1 | miR-204/ZEB1 | Apoptosis | NPC | [126] |
| miR-193b/Cyclin D1 | Apoptosis, cell cycle | Cervical cancer * | [127] | |
| PCAT6 | miR-185/TPD52 | Apoptosis, cell cycle | Breast cancer | [128] |
| POU6F2-AS2 | YBX1 | DSB repair | ESCC | [129] |
| PVT1 | miR-195 | Apoptosis, DSB repair | NSCLC * | [130] |
| ATM, p53, CHK2 | Apoptosis, DSB repair | NPC * | [131] | |
| HIF-1α | DSB repair | NPC | [132] | |
| SBF2-AS1 | miR-302a/MBNL3 | Apoptosis | NSCLC * | [133] |
| TP73-AS1 | PTEN, AKT | Apoptosis | HCC * | [134] |
| TUG1 | HMGB1 | Apoptosis | Bladder cancer * | [135] |
| UCA1 | AKT, FAK, FGR, AMPKα1 | Cell cycle | Gastric cancer | [136] |
| UCA1 | HK2 | Glycolysis | Cervical cancer | [137] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Podralska, M.; Ciesielska, S.; Kluiver, J.; van den Berg, A.; Dzikiewicz-Krawczyk, A.; Slezak-Prochazka, I. Non-Coding RNAs in Cancer Radiosensitivity: MicroRNAs and lncRNAs as Regulators of Radiation-Induced Signaling Pathways. Cancers 2020, 12, 1662. https://doi.org/10.3390/cancers12061662
Podralska M, Ciesielska S, Kluiver J, van den Berg A, Dzikiewicz-Krawczyk A, Slezak-Prochazka I. Non-Coding RNAs in Cancer Radiosensitivity: MicroRNAs and lncRNAs as Regulators of Radiation-Induced Signaling Pathways. Cancers. 2020; 12(6):1662. https://doi.org/10.3390/cancers12061662
Chicago/Turabian StylePodralska, Marta, Sylwia Ciesielska, Joost Kluiver, Anke van den Berg, Agnieszka Dzikiewicz-Krawczyk, and Izabella Slezak-Prochazka. 2020. "Non-Coding RNAs in Cancer Radiosensitivity: MicroRNAs and lncRNAs as Regulators of Radiation-Induced Signaling Pathways" Cancers 12, no. 6: 1662. https://doi.org/10.3390/cancers12061662
APA StylePodralska, M., Ciesielska, S., Kluiver, J., van den Berg, A., Dzikiewicz-Krawczyk, A., & Slezak-Prochazka, I. (2020). Non-Coding RNAs in Cancer Radiosensitivity: MicroRNAs and lncRNAs as Regulators of Radiation-Induced Signaling Pathways. Cancers, 12(6), 1662. https://doi.org/10.3390/cancers12061662







