Pathophysiology of Bone Loss in Patients with Prostate Cancer Receiving Androgen-Deprivation Therapy and Lifestyle Modifications for the Management of Bone Health: A Comprehensive Review
Abstract
1. Introduction
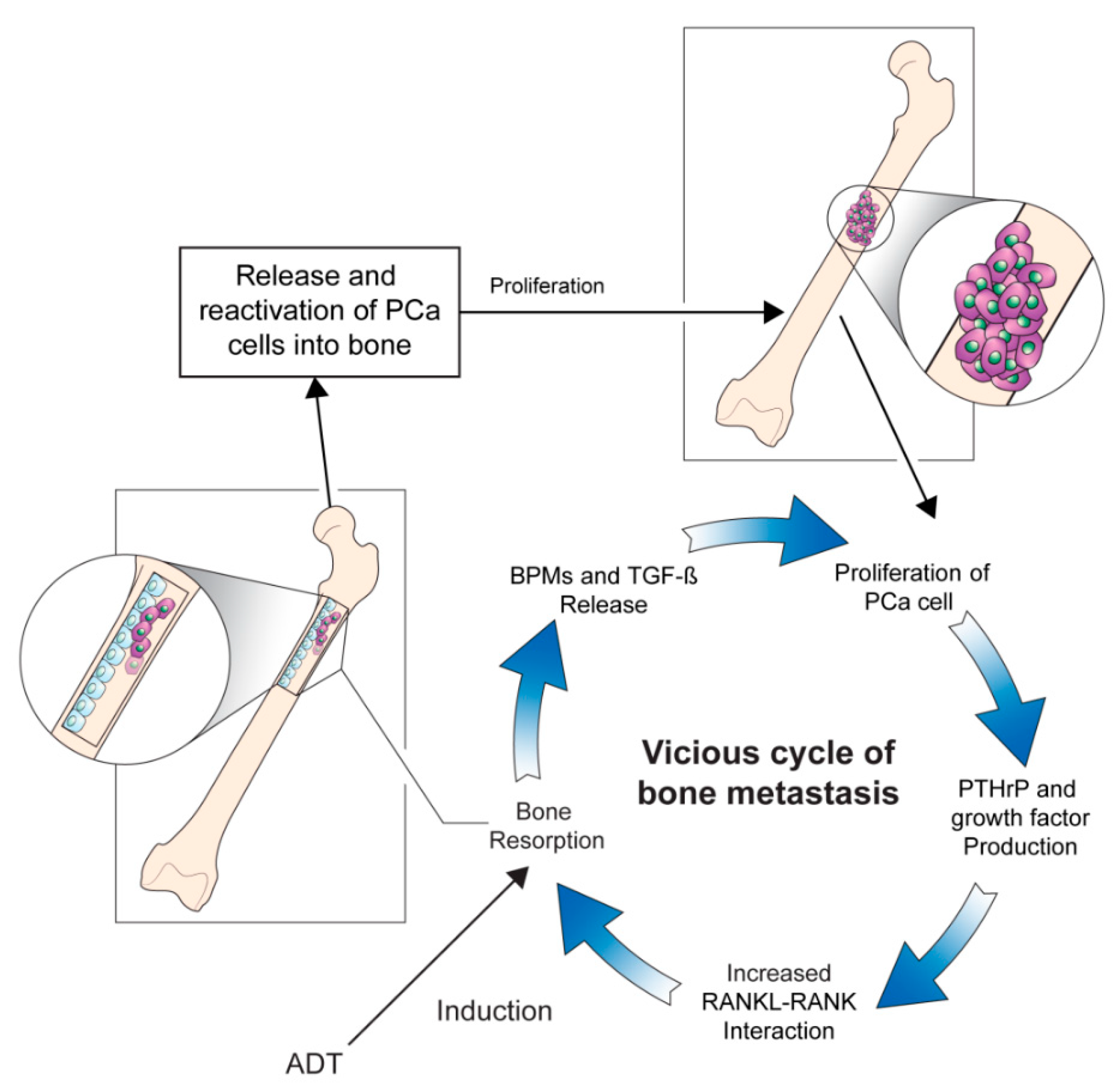
2. Pathophysiology Underlying Prostate Cancer Bone Metastasis
3. Pathophysiology of Cancer Treatment-Induced Bone Loss
4. Clinical Factors of Bone Loss
4.1. Primary Causes
4.2. Secondary Causes
5. Bone Health Assessment in Patients with Prostate Cancer
6. Monitoring Bone Health and Cancer Treatment-Induced Bone Loss in Patients with Prostate Cancer Treated by Androgen-Deprivation Therapy
7. Management Strategies for Cancer Treatment-Induced Bone Loss
7.1. Awareness and Education
7.2. Dietary Supplementation
7.3. Lifestyle Modification and Physical Exercise
8. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA A Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Koo, K.C.; Park, S.U.; Kim, K.H.; Rha, K.H.; Hong, S.J.; Yang, S.C.; Chung, B.H. Predictors of survival in prostate cancer patients with bone metastasis and extremely high prostate-specific antigen levels. Prostate Int. 2015, 3, 10–15. [Google Scholar] [CrossRef] [PubMed]
- Singh, J.; Trabulsi, E.J.; Gomella, L.G. The Quality-of-Life Impact of Prostate Cancer Treatments. Curr. Urol. Rep. 2010, 11, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Bourke, L.; Boorjian, S.A.; Briganti, A.; Klotz, L.; Mucci, L.; Resnick, M.J.; Rosario, D.J.; Skolarus, T.A.; Penson, D. Survivorship and Improving Quality of Life in Men with Prostate Cancer. Eur. Urol. 2015, 68, 374–383. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.K.; Lee, J.Y.; Kim, K.J.; Hong, N.; Kim, J.W.; Hah, Y.S.; Koo, K.C.; Kim, J.H.; Cho, K.S. Effect of Androgen-Deprivation Therapy on Bone Mineral Density in Patients with Prostate Cancer: A Systematic Review and Meta-Analysis. J. Clin. Med. 2019, 8, 113. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, P.L.; Alibhai, S.M.; Basaria, S.; D’Amico, A.V.; Kantoff, P.W.; Keating, N.L.; Penson, D.; Rosario, D.J.; Tombal, B.; Smith, M.R. Adverse Effects of Androgen Deprivation Therapy and Strategies to Mitigate Them. Eur. Urol. 2015, 67, 825–836. [Google Scholar] [CrossRef] [PubMed]
- So, A.; Chin, J.; Fleshner, N.; Saad, F. Management of skeletal-related events in patients with advanced prostate cancer and bone metastases: Incorporating new agents into clinical practice. Can. Urol. Assoc. J. 2012, 6, 465–470. [Google Scholar] [CrossRef] [PubMed]
- Tannock, I.F.; De Wit, R.; Berry, W.R.; Horti, J.; Pluzanska, A.; Chi, K.N.; Oudard, S.; Theodore, C.; James, N.D.; Turesson, I.; et al. Docetaxel plus Prednisone or Mitoxantrone plus Prednisone for Advanced Prostate Cancer. New Engl. J. Med. 2004, 351, 1502–1512. [Google Scholar] [CrossRef] [PubMed]
- Saad, F.; Ivanescu, C.; Phung, D.; Loriot, Y.; Abhyankar, S.; Beer, T.M.; Tombal, B.; Holmstrom, S. Skeletal-related events significantly impact health-related quality of life in metastatic castration-resistant prostate cancer: Data from PREVAIL and AFFIRM trials. Prostate Cancer Prostatic Dis. 2017, 20, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Vignani, F.; Bertaglia, V.; Buttigliero, C.; Tucci, M.; Scagliotti, G.V.; Di Maio, M. Skeletal metastases and impact of anticancer and bone-targeted agents in patients with castration-resistant prostate cancer. Cancer Treat. Rev. 2016, 44, 61–73. [Google Scholar] [CrossRef] [PubMed]
- Cook, R.J.; Coleman, R.; Brown, J.E.; Lipton, A.; Major, P.; Hei, Y.J.; Saad, F.; Smith, M.R. Markers of Bone Metabolism and Survival in Men with Hormone-Refractory Metastatic Prostate Cancer. Clin. Cancer Res. 2006, 12, 3361–3367. [Google Scholar] [CrossRef] [PubMed]
- Roodman, G.D. Mechanisms of Bone Metastasis. N. Engl. J. Med. 2004, 350, 1655–1664. [Google Scholar] [CrossRef] [PubMed]
- Sottnik, J.L.; Dai, J.; Zhang, H.; Campbell, B.; Keller, E.T. Tumor-induced pressure in the bone microenvironment causes osteocytes to promote the growth of prostate cancer bone metastases. Cancer Res. 2015, 75, 2151–2158. [Google Scholar] [CrossRef] [PubMed]
- Park, S.H.; Keller, E.T.; Shiozawa, Y. Bone Marrow Microenvironment as a Regulator and Therapeutic Target for Prostate Cancer Bone Metastasis. Calcif. Tissue Int. 2017, 102, 152–162. [Google Scholar] [CrossRef] [PubMed]
- Greenspan, S.L.; Coates, P.; Sereika, S.M.; Nelson, J.B.; Trump, N.L.; Resnick, N.M. Bone Loss after Initiation of Androgen Deprivation Therapy in Patients with Prostate Cancer. J. Clin. Endocrinol. Metab. 2005, 90, 6410–6417. [Google Scholar] [CrossRef] [PubMed]
- Almeida, M.; Laurent, M.R.; Dubois, V.; Claessens, F.; O’Brien, C.A.; Bouillon, R.; Vanderschueren, D.; Manolagas, S.C. Estrogens and Androgens in Skeletal Physiology and Pathophysiology. Physiol. Rev. 2017, 97, 135–187. [Google Scholar] [CrossRef] [PubMed]
- Mohamad, N.V.; Soelaiman, I.-N.; Chin, K.-Y. A concise review of testosterone and bone health. Clin. Interv. Aging 2016, 11, 1317–1324. [Google Scholar] [CrossRef] [PubMed]
- Vanderschueren, D.; Laurent, M.R.; Claessens, F.; Gielen, E.; Lagerquist, M.; Vandenput, L.; Börjesson, A.E.; Ohlsson, C. Sex steroid actions in male bone. Endocr. Rev. 2014, 35, 906–960. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Henning, P.; Sjögren, K.; Koskela, A.; Tuukkanen, J.; Movérare-Skrtic, S.; Ohlsson, C. The androgen receptor is required for maintenance of bone mass in adult male mice. Mol. Cell. Endocrinol. 2019, 479, 159–169. [Google Scholar] [CrossRef] [PubMed]
- Clarke, B.L.; Khosla, S. Androgens and bone. Steroids 2008, 74, 296–305. [Google Scholar] [CrossRef] [PubMed]
- Kasperk, C.; Wakley, G.K.; Huempfner-Hierl, H.; Ziegler, R. Gonadal and Adrenal Androgens Are Potent Regulators of Human Bone Cell Metabolism In Vitro. J. Bone Miner. Res. 1997, 12, 464–471. [Google Scholar] [CrossRef] [PubMed]
- Khosla, S. New Insights Into Androgen and Estrogen Receptor Regulation of the Male Skeleton. J. Bone Miner. Res. 2015, 30, 1134–1137. [Google Scholar] [CrossRef] [PubMed]
- Todenhöfer, T.; Stenzl, A.; Hofbauer, L.C.; Rachner, T.D. Targeting Bone Metabolism in Patients with Advanced Prostate Cancer: Current Options and Controversies. Int. J. Endocrinol. 2015, 2015, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Argoud, T.; Boutroy, S.; Claustrat, B.; Chapurlat, R.; Szulc, P. Association Between Sex Steroid Levels and Bone Microarchitecture in Men: The STRAMBO Study. J. Clin. Endocrinol. Metab. 2014, 99, 1400–1410. [Google Scholar] [CrossRef] [PubMed]
- Drake, M.T.; Khosla, S. Male osteoporosis. Endocrinol. Metab. Clin. North Am. 2012, 41, 629–641. [Google Scholar] [CrossRef] [PubMed]
- Piot, A.; Chapurlat, R.; Claustrat, B.; Szulc, P. Relationship Between Sex Steroids and Deterioration of Bone Microarchitecture in Older Men: The Prospective STRAMBO Study. J. Bone Miner. Res. 2019, 34, 1562–1573. [Google Scholar] [CrossRef] [PubMed]
- Kiratli, B.; Srinivas, S.; Perkash, I.; Terris, M.K. Progressive decrease in bone density over 10 years of androgen deprivation therapy in patients with prostate cancer. Urology 2001, 57, 127–132. [Google Scholar] [CrossRef]
- Uyl, D.D.; Bultink, I.; Lems, W. Advances in Glucocorticoid-Induced Osteoporosis. Curr. Rheumatol. Rep. 2011, 13, 233–240. [Google Scholar] [CrossRef]
- Auchus, R.J.; Yu, M.K.; Nguyen, S.; Mundle, S.D. Use of Prednisone With Abiraterone Acetate in Metastatic Castration-Resistant Prostate Cancer. Oncology 2014, 19, 1231–1240. [Google Scholar] [CrossRef] [PubMed]
- Dorff, T.; Crawford, E.D. Management and challenges of corticosteroid therapy in men with metastatic castrate-resistant prostate cancer. Ann. Oncol. 2013, 24, 31–38. [Google Scholar] [CrossRef] [PubMed]
- Rizzo, S.; Galvano, A.; Pantano, F.; Iuliani, M.; Vincenzi, B.; Passiglia, F.; Spoto, S.; Tonini, G.; Bazan, V.; Russo, A.; et al. The effects of enzalutamide and abiraterone on skeletal related events and bone radiological progression free survival in castration resistant prostate cancer patients: An indirect comparison of randomized controlled trials. Crit. Rev. Oncol. 2017, 120, 227–233. [Google Scholar] [CrossRef] [PubMed]
- Tombal, B.; Borre, M.; Rathenborg, P.; Werbrouck, P.; Van Poppel, H.; Heidenreich, A.; Iversen, P.; Braeckman, J.; Heracek, J.; Baskin-Bey, E.; et al. Enzalutamide monotherapy in hormone-naive prostate cancer: Primary analysis of an open-label, single-arm, phase 2 study. Lancet Oncol. 2014, 15, 592–600. [Google Scholar] [CrossRef]
- Smith, M.R.; Goode, M.; Zietman, A.L.; McGovern, F.J.; Lee, H.; Finkelstein, J.S. Bicalutamide Monotherapy Versus Leuprolide Monotherapy for Prostate Cancer: Effects on Bone Mineral Density and Body Composition. J. Clin. Oncol. 2004, 22, 2546–2553. [Google Scholar] [CrossRef] [PubMed]
- Iuliani, M.; Pantano, F.; Buttigliero, C.; Fioramonti, M.; Bertaglia, V.; Vincenzi, B.; Zoccoli, A.; Ribelli, G.; Tucci, M.; Vignani, F.; et al. Biological and clinical effects of abiraterone on anti-resorptive and anabolic activity in bone microenvironment. Oncotarget 2015, 6, 12520–12528. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kokontis, J.M.; Hay, N.; Liao, S. Progression of LNCaP Prostate Tumor Cells during Androgen Deprivation: Hormone-Independent Growth, Repression of Proliferation by Androgen, and Role for p27Kip1 in Androgen-Induced Cell Cycle Arrest. Mol. Endocrinol. 1998, 12, 941–953. [Google Scholar] [CrossRef] [PubMed]
- Schweizer, M.T.; Antonarakis, E.S.; Wang, H.; Ajiboye, A.S.; Spitz, A.; Cao, H.; Luo, J.; Haffner, M.C.; Yegnasubramanian, S.; Carducci, M.A.; et al. Effect of bipolar androgen therapy for asymptomatic men with castration-resistant prostate cancer: Results from a pilot clinical study. Sci. Transl. Med. 2015, 7, 269ra2. [Google Scholar] [CrossRef] [PubMed]
- Denmeade, S.R.; Isaacs, J.T. Bipolar androgen therapy: The rationale for rapid cycling of supraphysiologic androgen/ablation in men with castration resistant prostate cancer. Prostate 2010, 70, 1600–1607. [Google Scholar] [CrossRef] [PubMed]
- Schweizer, M.T.; Wang, H.; Luber, B.; Nadal, R.; Spitz, A.; Rosen, D.M.; Cao, H.; Antonarakis, E.S.; Eisenberger, M.A.; Carducci, M.A.; et al. Bipolar Androgen Therapy for Men With Androgen Ablation Naïve Prostate Cancer: Results From the Phase II BATMAN Study. Prostate 2016, 76, 1218–1226. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.R.; Saad, F.; Chowdhury, S.; Oudard, S.; Hadaschik, B.; Graff, J.N.; Olmos, D.; Mainwaring, P.N.; Lee, J.Y.; Uemura, H.; et al. Apalutamide Treatment and Metastasis-free Survival in Prostate Cancer. N. Engl. J. Med. 2018, 378, 1408–1418. [Google Scholar] [CrossRef] [PubMed]
- Saylor, P.J.; Morton, R.A.; Hancock, M.L.; Barnette, K.G.; Steiner, M.S.; Smith, M.R. Factors Associated With Vertebral Fractures in Men Treated With Androgen Deprivation Therapy for Prostate Cancer. J. Urol. 2011, 186, 482–486. [Google Scholar] [CrossRef] [PubMed]
- Cheung, A.S.; Pattison, D.A.; Bretherton, I.; Hoermann, R.; Joon, D.L.; Ho, E.; Jenkins, T.; Hamilton, E.J.; Bate, K.; Chan, I.; et al. Cardiovascular risk and bone loss in men undergoing androgen deprivation therapy for non-metastatic prostate cancer: Implementation of standardized management guidelines. Andrology 2013, 1, 583–589. [Google Scholar] [CrossRef] [PubMed]
- Saad, F.; Lattouf, J.-B. Bisphosphonates: Prevention of Bone Metastases in Prostate Cancer. Adv. Struct. Saf. Stud. 2012, 192, 109–126. [Google Scholar] [CrossRef]
- Walsh, J.; Eastell, R. Osteoporosis in men. Nat. Rev. Endocrinol. 2013, 9, 637–645. [Google Scholar] [CrossRef] [PubMed]
- Hendrickx, G.; Boudin, E.; Van Hul, W. A look behind the scenes: The risk and pathogenesis of primary osteoporosis. Nat. Rev. Rheumatol. 2015, 11, 462–474. [Google Scholar] [CrossRef] [PubMed]
- Qian, S.-Z.; Xu, Y.C.; Zhang, J. Hormonal deficiency in elderly males. Int. J. Androl. 2000, 23, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Edwards, M.H.; Jameson, K.; Denison, H.J.; Harvey, N.C.; Sayer, A.A.; Dennison, E.M.; Cooper, C. Clinical risk factors, bone density and fall history in the prediction of incident fracture among men and women. Bone 2012, 52, 541–547. [Google Scholar] [CrossRef] [PubMed]
- Israeli, R.S. Managing Bone Loss and Bone Metastases in Prostate Cancer Patients: A Focus on Bisphosphonate Therapy. Rev. Urol. 2008, 10, 99–110. [Google Scholar] [PubMed]
- Keller, E.T.; Brown, J. Prostate cancer bone metastases promote both osteolytic and osteoblastic activity. J. Cell. Biochem. 2004, 91, 718–729. [Google Scholar] [CrossRef] [PubMed]
- Mottet, N.; Bellmunt, J.; Bolla, M.; Briers, E.; Cumberbatch, M.G.; De Santis, M.; Fossati, N.; Gross, T.; Henry, A.M.; Joniau, S.; et al. EAU-ESTRO-SIOG Guidelines on Prostate Cancer. Part 1: Screening, Diagnosis, and Local Treatment with Curative Intent. Eur. Urol. 2017, 71, 618–629. [Google Scholar] [CrossRef] [PubMed]
- Krohn, K.; Schwartz, E.N.; Chung, Y.-S.; Lewiecki, E.M. Dual-energy X-ray Absorptiometry Monitoring with Trabecular Bone Score: 2019 ISCD Official Position. J. Clin. Densitom. 2019, 22, 501–505. [Google Scholar] [CrossRef] [PubMed]
- Kanis, J.A.; Melton, L.J.; Christiansen, C.; Johnston, C.C.; Khaltaev, N. The diagnosis of osteoporosis. J. Bone Miner. Res. 2009, 9, 1137–1141. [Google Scholar] [CrossRef] [PubMed]
- Siris, E.S.; Miller, P.; Barrett-Connor, E.; Faulkner, K.G.; Wehren, L.; Abbott, T.A.; Berger, M.L.; Santora, A.C.; Sherwood, L.M. Identification and Fracture Outcomes of Undiagnosed Low Bone Mineral Density in Postmenopausal Women. JAMA 2001, 286, 2815. [Google Scholar] [CrossRef] [PubMed]
- Azagra, R.; Zwart, M.; Sanchez, J.C.M.; Aguye, A.; Groimap, G. La herramienta FRAX® en la prevención de fracturas asociadas al tratamiento de privación androgénica en el cáncer de próstata. Med. Clínica 2013, 142, 231–232. [Google Scholar] [CrossRef] [PubMed]
- Santini, D.; Berruti, A.; Di Maio, M.; Procopio, G.; Bracarda, S.; Ibrahim, T.; Bertoldo, F. Bone health management in the continuum of prostate cancer disease: A review of the evidence with an expert panel opinion. ESMO Open 2020, 5, e000652. [Google Scholar] [CrossRef] [PubMed]
- Collins, G.S.; Mallett, S.; Altman, U.G. Predicting risk of osteoporotic and hip fracture in the United Kingdom: Prospective independent and external validation of QFractureScores. BMJ 2011, 342, d3651. [Google Scholar] [CrossRef] [PubMed]
- Hippisley-Cox, J.; Coupland, C.; Brindle, P. The performance of seven QPrediction risk scores in an independent external sample of patients from general practice: A validation study. BMJ Open 2014, 4, e005809. [Google Scholar] [CrossRef] [PubMed]
- Pothuaud, L.; Carceller, P.; Hans, D. Correlations between grey-level variations in 2D projection images (TBS) and 3D microarchitecture: Applications in the study of human trabecular bone microarchitecture. Bone 2008, 42, 775–787. [Google Scholar] [CrossRef] [PubMed]
- Mariotti, V.; Page, D.B.; Davydov, O.; Hans, D.; Hudis, C.A.; Patil, S.; Kunte, S.; Girotra, M.; Farooki, A.; Fornier, M.N. Assessing fracture risk in early stage breast cancer patients treated with aromatase-inhibitors: An enhanced screening approach incorporating trabecular bone score. J. Bone Oncol. 2016, 7, 32–37. [Google Scholar] [CrossRef] [PubMed]
- Heidenreich, A.; Bellmunt, J.; Bolla, M.; Joniau, S.; Mason, M.; Matveev, V.; Mottet, N.; Schmid, H.; Van Der Kwast, T.H.; Wiegel, T.; et al. Guía de la EAU sobre el cáncer de próstata. Parte I: Cribado, diagnóstico y tratamiento del cáncer clínicamente localizado. Actas Urológicas Españolas 2011, 35, 501–514. [Google Scholar] [CrossRef]
- Mohler, J.L.; Antonarakis, E.S. NCCN Guidelines Updates: Management of Prostate Cancer. J. Natl. Compr. Cancer Netw. 2019, 17, 583–586. [Google Scholar]
- Nanes, M.S.; Kallen, C. Osteoporosis. Semin. Nucl. Med. 2014, 44, 439–450. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, M.M.; Khandelwal, N.; Mandal, A.K.; Rana, S.V.; Gupta, V.; Mohan, V.C.; Kishore, G.V.M.K. Factors affecting bone mineral density in patients with prostate carcinoma before and after orchidectomy. Cancer 2005, 103, 2042–2052. [Google Scholar] [CrossRef] [PubMed]
- Grossmann, M.; Hamilton, E.J.; Gilfillan, C.; Bolton, D.; Joon, D.L.; Zajac, J.D. Bone and metabolic health in patients with non-metastatic prostate cancer who are receiving androgen deprivation therapy. Med. J. Aust. 2011, 194, 301–306. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.; Leslie, W.D.; Czaykowski, P.; Gingerich, J.; Geirnaert, M.; Lau, Y. A comprehensive bone-health management approach for men with prostate cancer receiving androgen deprivation therapy. Curr. Oncol. 2011, 18, e163–e172. [Google Scholar] [CrossRef] [PubMed]
- Owen, P.J.; Daly, R.M.; Livingston, P.M.; Fraser, S.F. Lifestyle guidelines for managing adverse effects on bone health and body composition in men treated with androgen deprivation therapy for prostate cancer: An update. Prostate Cancer Prostatic Dis. 2017, 20, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Saylor, P.J.; Rumble, R.B.; Michalski, J.M. Bone Health and Bone-Targeted Therapies for Prostate Cancer: American Society of Clinical Oncology Endorsement Summary of a Cancer Care Ontario Guideline. JCO Oncol. Pr. 2020. [Google Scholar] [CrossRef] [PubMed]
- Lassemillante, A.-C.M.; Skinner, T.L.; Hooper, J.; Prins, J.B.; Wright, O.R.L. Osteoporosis-Related Health Behaviors in Men With Prostate Cancer and Survivors. Am. J. Men’s Health 2016, 11, 13–23. [Google Scholar] [CrossRef] [PubMed]
- Mckean, H.; Looker, S.; Hartmann, L.C.; Hayman, S.R.; Kaur, J.S.; McWilliams, R.R.; Peethambaram, P.P.; Stahl, J.F.; Jatoi, A. Are Cancer Survivors/Patients Knowledgeable About Osteoporosis? Results from a Survey of 285 Chemotherapy-treated Cancer Patients and Their Companions. J. Nutr. Educ. Behav. 2008, 40, 144–148. [Google Scholar] [CrossRef] [PubMed]
- Nadler, M.; Alibhai, S.M.; Catton, P.; Catton, C.; To, M.J.; Jones, J. Osteoporosis knowledge, health beliefs, and healthy bone behaviours in patients on androgen-deprivation therapy (ADT) for prostate cancer. BJU Int. 2013, 111, 1301–1309. [Google Scholar] [CrossRef] [PubMed]
- Tripathy, D.; Durie, B.G.M.; Mautner, B.; Ferenz, K.S.; Moul, J.W. Awareness, concern, and communication between physicians and patients on bone health in cancer. Support. Care Cancer 2014, 22, 1601–1610. [Google Scholar] [CrossRef] [PubMed]
- Alibhai, S.M.; Rahman, S.; Warde, P.; Jewett, M.A.; Jaffer, T.; Cheung, A.M. Prevention and management of osteoporosis in men receiving androgen deprivation therapy: A survey of urologists and radiation oncologists. Urology 2006, 68, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Damji, A.N.; Bies, K.; Alibhai, S.M.; Jones, J. Bone health management in men undergoing ADT: Examining enablers and barriers to care. Osteoporos. Int. 2014, 26, 951–959. [Google Scholar] [CrossRef] [PubMed]
- Alibhai, S.M.; Breunis, H.; Timilshina, N.; Hamidi, M.S.; Cheung, A.M.; Tomlinson, G.; Manokumar, T.; Samadi, O.; Sandoval, J.; Durbano, S.; et al. Improving bone health in men with prostate cancer receiving androgen deprivation therapy: Results of a randomized phase 2 trial. Cancer 2017, 124, 1132–1140. [Google Scholar] [CrossRef] [PubMed]
- Bordes, J.K.A.D.; Suarez-Almazor, M.E.; Volk, R.; Lu, H.; Edwards, B.; Lopez-Olivo, M.A. Online Educational Tool to Promote Bone Health in Cancer Survivors. J. Health Commun. 2017, 22, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Batai, K.; Murphy, A.B.; Ruden, M.; Newsome, J.; Shah, E.; Dixon, M.A.; Jacobs, E.T.; Hollowell, C.M.P.; Ahaghotu, C.; Kittles, R.A. Race and BMI modify associations of calcium and vitamin D intake with prostate cancer. BMC Cancer 2017, 17, 64. [Google Scholar] [CrossRef] [PubMed]
- Capiod, T.; Delongchamps, N.B.; Pigat, N.; Souberbielle, J.-C.; Goffin, V. Do dietary calcium and vitamin D matter in men with prostate cancer? Nat. Rev. Urol. 2018, 15, 453–461. [Google Scholar] [CrossRef] [PubMed]
- Datta, M.; Schwartz, G.G. Calcium and Vitamin D Supplementation During Androgen Deprivation Therapy for Prostate Cancer: A Critical Review. Oncology 2012, 17, 1171–1179. [Google Scholar] [CrossRef] [PubMed]
- Nelson, S.M.; Batai, K.; Ahaghotu, C.; Agurs-Collins, T.; Kittles, R.A. Association between Serum 25-Hydroxy-Vitamin D and Aggressive Prostate Cancer in African American Men. Nutrients 2016, 9, 12. [Google Scholar] [CrossRef] [PubMed]
- Saylor, P.J.; Keating, N.L.; Smith, M.R. Prostate Cancer Survivorship: Prevention and Treatment of the Adverse Effects of Androgen Deprivation Therapy. J. Gen. Intern. Med. 2009, 24, 389–394. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Nowson, C.; McGrath, J.J.; Ebeling, P.R.; Haikerwal, A.; Daly, R.; Sanders, K.M.; Seibel, M.; Mason, R.S. Vitamin D and health in adults in Australia and New Zealand: A position statement. Med. J. Aust. 2012, 196, 686–687. [Google Scholar] [CrossRef] [PubMed]
- Aune, D.; Rosenblatt, D.A.N.; Chan, D.S.; Vieira, A.R.; Vieira, R.; Greenwood, D.C.; Vatten, L.J.; Norat, T. Dairy products, calcium, and prostate cancer risk: A systematic review and meta-analysis of cohort studies. Am. J. Clin. Nutr. 2014, 101, 87–117. [Google Scholar] [CrossRef] [PubMed]
- Marshall, D.T.; Savage, S.J.; Garrett-Mayer, E.; Keane, T.E.; Hollis, B.W.; Horst, R.L.; Ambrose, L.H.; Kindy, M.S.; Gattoni-Celli, S. Vitamin D3 supplementation at 4000 international units per day for one year results in a decrease of positive cores at repeat biopsy in subjects with low-risk prostate cancer under active surveillance. J. Clin. Endocrinol. Metab. 2012, 97, 2315–2324. [Google Scholar] [CrossRef] [PubMed]
- Owen, P.J.; Daly, R.M.; Via, J.D.; Mundell, N.L.; Livingston, P.M.; Rantalainen, T.; Fraser, S.F. Does Use of Androgen Deprivation Therapy (ADT) in Men with Prostate Cancer Increase the Risk of Sarcopenia? Calcif. Tissue Int. 2019, 105, 403–411. [Google Scholar] [CrossRef] [PubMed]
- Grossmann, M.; Cheung, A.S.; Zajac, J.D. Androgens and prostate cancer; pathogenesis and deprivation therapy. Best Pr. Res. Clin. Endocrinol. Metab. 2013, 27, 603–616. [Google Scholar] [CrossRef] [PubMed]
- Cosman, F.; De Beur, S.J.; LeBoff, M.S.; Lewiecki, E.M.; Tanner, B.; Randall, S.; Lindsay, R.; Foundation, N.O. Clinician’s Guide to Prevention and Treatment of Osteoporosis. Osteoporos. Int. 2014, 25, 2359–2381. [Google Scholar] [CrossRef] [PubMed]
- Martone, A.M.; Marzetti, E.; Calvani, R.; Picca, A.; Tosato, M.; Santoro, L.; Di Giorgio, A.; Nesci, A.; Sisto, A.; Santoliquido, A.; et al. Exercise and Protein Intake: A Synergistic Approach against Sarcopenia. BioMed Res. Int. 2017, 2017, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Galvão, D.A.; Nosaka, K.; Taaffe, D.R.; Spry, N.; Kristjanson, L.J.; McGuigan, M.R.; Suzuki, K.; Yamaya, K.; Newton, R.U. Resistance Training and Reduction of Treatment Side Effects in Prostate Cancer Patients. Med. Sci. Sports Exerc. 2006, 38, 2045–2052. [Google Scholar] [CrossRef] [PubMed]
- Nilsen, T.S.; Raastad, T.; Skovlund, E.; Courneya, K.S.; Langberg, C.W.; Lilleby, W.; Fosså, S.D.; Thorsen, L. Effects of strength training on body composition, physical functioning, and quality of life in prostate cancer patients during androgen deprivation therapy. Acta Oncol. 2015, 54, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Cormie, P.; Galvão, D.A.; Spry, N.; Joseph, D.; Chee, R.; Taaffe, D.R.; Chambers, S.K.; Newton, R.U. Can supervised exercise prevent treatment toxicity in patients with prostate cancer initiating androgen-deprivation therapy: A randomised controlled trial. BJU Int. 2014, 115, 256–266. [Google Scholar] [CrossRef] [PubMed]
- Winters-Stone, K.; Dobek, J.C.; Bennett, J.A.; Maddalozzo, G.F.; Ryan, C.W.; Beer, T.M. Skeletal Response to Resistance and Impact Training in Prostate Cancer Survivors. Med. Sci. Sports Exerc. 2014, 46, 1482–1488. [Google Scholar] [CrossRef] [PubMed]
- Uth, J.; Hornstrup, T.; Christensen, J.F.; Christensen, K.B.; Jørgensen, N.R.; Schmidt, J.F.; Brasso, K.; Jakobsen, M.D.; Sundstrup, E.; Andersen, L.L.; et al. Efficacy of recreational football on bone health, body composition, and physical functioning in men with prostate cancer undergoing androgen deprivation therapy: 32-week follow-up of the FC prostate randomised controlled trial. Osteoporos. Int. 2015, 27, 1507–1518. [Google Scholar] [CrossRef] [PubMed]
- Kukuljan, S.; Nowson, C.; Bass, S.L.; Sanders, K.M.; Nicholson, G.C.; Seibel, M.; Salmon, J.; Daly, R. Effects of a multi-component exercise program and calcium–vitamin-D3-fortified milk on bone mineral density in older men: A randomised controlled trial. Osteoporos. Int. 2008, 20, 1241–1251. [Google Scholar] [CrossRef] [PubMed]
- Marques, E.A.; Mota, J.; Carvalho, J. Exercise effects on bone mineral density in older adults: A meta-analysis of randomized controlled trials. AGE 2011, 34, 1493–1515. [Google Scholar] [CrossRef] [PubMed]
- Park, S.; Kim, K.; Ahn, H.K.; Kim, J.W.; Min, G.; Chung, B.H.; Koo, K.C. Impact of Lifestyle Intervention for Patients with Prostate Cancer. Am. J. Health Behav. 2020, 44, 90–99. [Google Scholar] [CrossRef] [PubMed]
| Endocrine |
| Chronic kidney disease Diabetes mellitus |
| Hyperparathyroidism |
| Hypercortisolism |
| Hypogonadism, including aging |
| Hyperthyroidism |
| Nutritional/Gastrointestinal |
| Alcoholism |
| Chronic liver disease |
| Inflammatory bowel disease |
| Malabsorption syndromes |
| Malnutrition |
| Rheumatological/Connective Tissue |
| Ankylosing spondylitis |
| Marfan syndrome |
| Rheumatoid arthritis |
| Systemic lupus erythematosus |
| Hematological |
| Disseminated bone metastasis |
| Lymphoma/Leukemia |
| Multiple myeloma |
| At the Initiation of ADT Evaluation for Any History of Trauma-Induced Fractures and Risk Factors for Osteoporosis [49] |
| BMD assessment with DXA scan and subsequent scoring with FRAX [49] |
| DXA scan or FRAX score only is not recommended. The following factors should be incorporated [49,53,54] |
| Age |
| BMD |
| History of corticosteroid therapy |
| Medical history of bone metastasis or fragility disease or treatment |
| Physical disability or risk factors of fall |
| Evaluations Recommended for Monitoring Skeletal Health (Perform at Baseline and Every 12–18 months Afterward) [54,59] |
| BMD measurement using DXA scan during the first 24 months of ADT |
| Bone turnover markers (e.g., serum ALP level) |
| Serum calcium levels |
| Serum vitamin D levels |
| Serum PTH levels |
| Height, weight, BMI |
| In the case of lumbar pain or loss of height, perform spine radiography and imaging studies |
| Tobacco and Alcohol Consumption |
| Smoking cessation should be recommended [62,63] |
| Alcohol consumption should be limited to two or fewer standard drinks per day [63] |
| Calcium |
| Integrate 3–4 daily servings of dairy products for dietary calcium intake [6,63,64,75,76,77,78] |
| Consider calcium supplements if the daily calcium intake is below 1000–1300 mg per day [6,63,64,75,79] |
| Vitamin D |
| Prior to ADT initiation, patients should have an assessment of serum 25(OH)D [6,63,74,75,77,79] |
| Men with 25(OH)D levels ≥50 nmol L−1 should consider a daily supplement intake of 800 IU. [6,63,64,75,77,79] |
| Men with vitamin D deficiency should supplement with 3000–5000 IU per day for at least 6–12 weeks under clinical supervision [75,76,77,78,80] |
| Physical Exercise Training |
| Progressive overloading should be applied to all exercises when possible, and sessions must be performed under professional supervision [64,65] |
| Progressive resistance training (PRT) [87,88,89,90,91] |
| Consider at least two times per week |
| At least 10 exercises focusing on major muscle groups, especially the muscles attached to the lumbar spine and hip |
| Two to three sets of 8–10 repetitions at moderate to high intensity |
| Weight-bearing impact exercise [89,90,91,92,93] |
| At least 4 days per week |
| Two to four impact exercises with a variation in magnitude and duration |
| PRT is recommended to patients with low muscle strength before initiation of impact exercises |
| Aerobic training [65] |
| At least five times per week |
| At least 30 min of continuous exercise |
| Target heart rate should be 55–75% of maximum predicted heart rate |
| Training sessions can be divided into shorter sessions if needed (three individual 10-min sessions) |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, T.J.; Koo, K.C. Pathophysiology of Bone Loss in Patients with Prostate Cancer Receiving Androgen-Deprivation Therapy and Lifestyle Modifications for the Management of Bone Health: A Comprehensive Review. Cancers 2020, 12, 1529. https://doi.org/10.3390/cancers12061529
Kim TJ, Koo KC. Pathophysiology of Bone Loss in Patients with Prostate Cancer Receiving Androgen-Deprivation Therapy and Lifestyle Modifications for the Management of Bone Health: A Comprehensive Review. Cancers. 2020; 12(6):1529. https://doi.org/10.3390/cancers12061529
Chicago/Turabian StyleKim, Tae Jin, and Kyo Chul Koo. 2020. "Pathophysiology of Bone Loss in Patients with Prostate Cancer Receiving Androgen-Deprivation Therapy and Lifestyle Modifications for the Management of Bone Health: A Comprehensive Review" Cancers 12, no. 6: 1529. https://doi.org/10.3390/cancers12061529
APA StyleKim, T. J., & Koo, K. C. (2020). Pathophysiology of Bone Loss in Patients with Prostate Cancer Receiving Androgen-Deprivation Therapy and Lifestyle Modifications for the Management of Bone Health: A Comprehensive Review. Cancers, 12(6), 1529. https://doi.org/10.3390/cancers12061529





