Clinical Characteristics of Korean Breast Cancer Patients Who Carry Pathogenic Germline Mutations in Both BRCA1 and BRCA2: A Single-Center Experience
Abstract
1. Introduction
2. Results
2.1. Clinical Characteristics of Patients with Pathogenic Germline BRCA1 and 2 Mutations
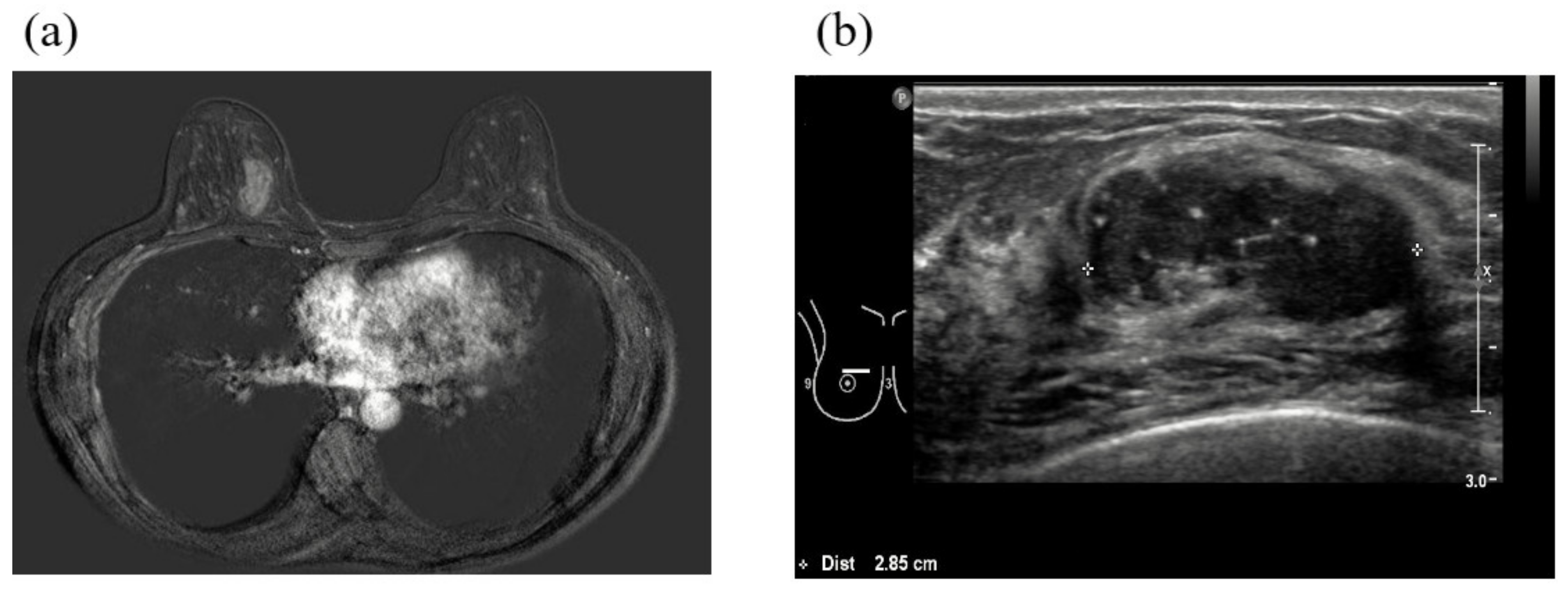
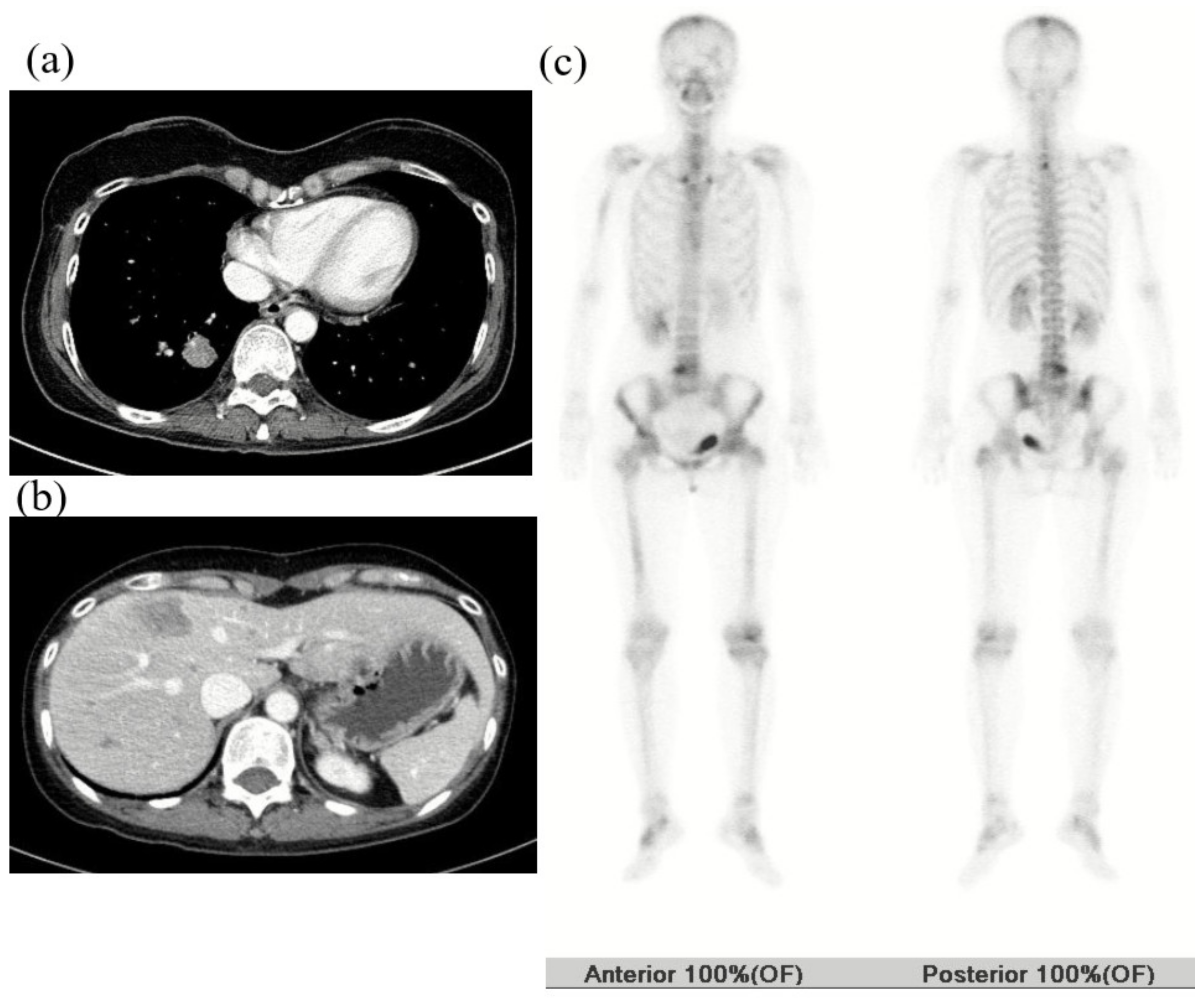
2.2. First Case
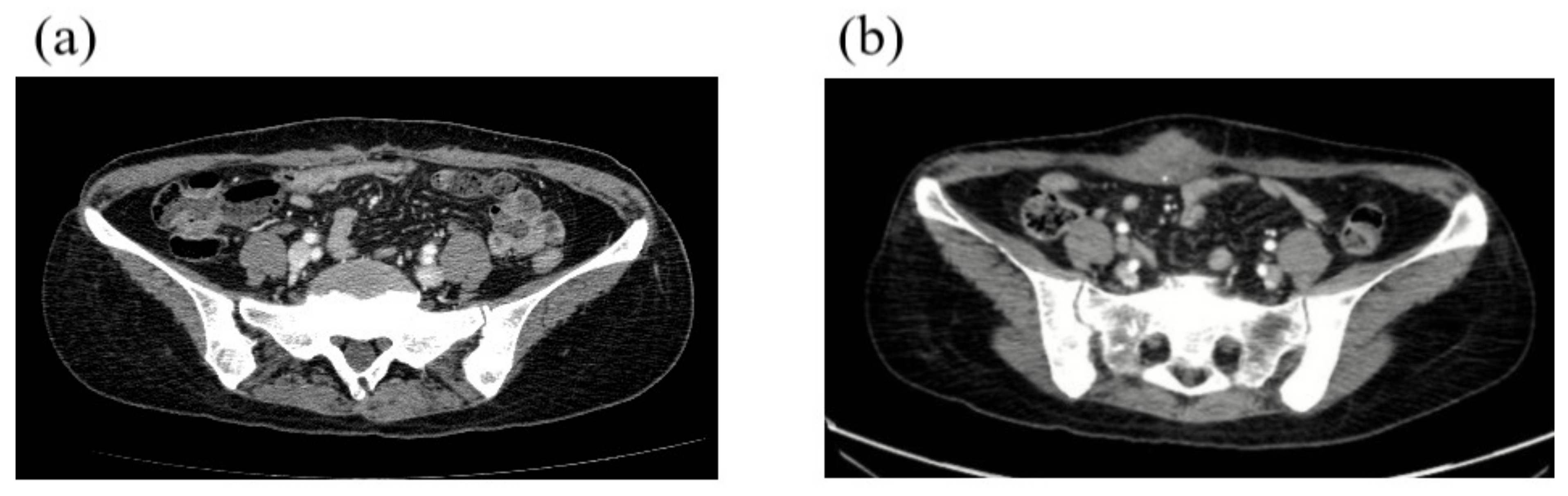
2.3. Second Case
2.4. Third Case
2.5. Fourth Case
2.6. Clinical Characteristics of Patients with a Pathogenic gBRCA1 Mutation and gBRCA2 Variants of Uncertain Significance
3. Discussion
4. Materials and Methods
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- King, M.C. “The race” to clone BRCA1. Science 2014, 343, 1462–1465. [Google Scholar] [CrossRef] [PubMed]
- Lord, C.J.; Ashworth, A. PARP inhibitors: Synthetic lethality in the clinic. Science 2017, 355, 1152–1158. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Iversen, E.S.; Friebel, T.; Finkelstein, D.; Weber, B.L.; Eisen, A.; Peterson, L.E.; Schildkraut, J.M.; Isaacs, C.; Peshkin, B.N.; et al. Characterization of BRCA1 and BRCA2 Mutations in a Large United States Sample. J. Clin. Oncol. 2006, 24, 863–871. [Google Scholar] [CrossRef] [PubMed]
- Kuchenbaecker, K.B.; Hopper, J.L.; Barnes, D.R.; Phillips, K.A.; Mooij, T.M.; Roos-Blom, M.J.; Jervis, S.; van Leeuwen, F.E.; Milne, R.L.; Andrieu, N.; et al. Risks of Breast, Ovarian, and Contralateral Breast Cancer for BRCA1 and BRCA2 Mutation Carriers. JAMA 2017, 317, 2402–2416. [Google Scholar] [CrossRef] [PubMed]
- Domchek, S.M. Risk-Reducing Mastectomy in BRCA1 and BRCA2 Mutation Carriers: A Complex DiscussionRisk-Reducing Mastectomy in BRCA1 and BRCA2 Mutation Carriers—A Complex DiscussionRisk-Reducing Mastectomy in BRCA1 and BRCA2 Mutation Carriers—A Complex Discussion. JAMA 2019, 321, 27. [Google Scholar] [CrossRef] [PubMed]
- Han, S.A.; Park, S.K.; Ahn, S.H.; Lee, M.H.; Noh, D.Y.; Kim, L.S.; Noh, W.C.; Jung, Y.; Kim, K.S.; Kim, S.W. The Korean Hereditary Breast Cancer (KOHBRA) study: Protocols and interim report. Clin. Oncol. R. Coll. Radiol. Great Br. 2011, 23, 434–441. [Google Scholar] [CrossRef]
- Noh, J.M.; Choi, D.H.; Nam, S.J.; Lee, J.E.; Kim, J.W.; Kim, S.W.; Kang, E.; Lee, M.H.; Ahn, S.H.; Kim, K.S.; et al. Characteristics of double heterozygosity for BRCA1 and BRCA2 germline mutations in Korean breast cancer patients. Breast Cancer Res. Treat. 2012, 131, 217–222. [Google Scholar] [CrossRef]
- Robson, M.; Im, S.-A.; Senkus, E.; Xu, B.; Domchek, S.M.; Masuda, N.; Delaloge, S.; Li, W.; Tung, N.; Armstrong, A.; et al. Olaparib for Metastatic Breast Cancer in Patients with a Germline BRCA Mutation. N. Engl. J. Med. 2017, 377, 523–533. [Google Scholar] [CrossRef]
- Frank, T.S.; Deffenbaugh, A.M.; Reid, J.E.; Hulick, M.; Ward, B.E.; Lingenfelter, B.; Gumpper, K.L.; Scholl, T.; Tavtigian, S.V.; Pruss, D.R.; et al. Clinical characteristics of individuals with germline mutations in BRCA1 and BRCA2: Analysis of 10,000 individuals. J. Clin. Oncol. Off. J. Am. Soc. Clin. Oncol. 2002, 20, 1480–1490. [Google Scholar] [CrossRef]
- Cancer Genome Atlas Network. Comprehensive molecular portraits of human breast tumours. Nature 2012, 490, 61–70. [Google Scholar] [CrossRef]
- Lord, C.J.; Ashworth, A. BRCAness revisited. Nat. Rev. Cancer 2016, 16, 110–120. [Google Scholar] [CrossRef] [PubMed]
- Litton, J.K.; Rugo, H.S.; Ettl, J.; Hurvitz, S.A.; Gonçalves, A.; Lee, K.-H.; Fehrenbacher, L.; Yerushalmi, R.; Mina, L.A.; Martin, M.; et al. Talazoparib in Patients with Advanced Breast Cancer and a Germline BRCA Mutation. N. Engl. J. Med. 2018, 379, 753–763. [Google Scholar] [CrossRef] [PubMed]
- Kwong, A.; Shin, V.Y.; Ho, J.C.; Kang, E.; Nakamura, S.; Teo, S.H.; Lee, A.S.; Sng, J.H.; Ginsburg, O.M.; Kurian, A.W.; et al. Comprehensive spectrum of BRCA1 and BRCA2 deleterious mutations in breast cancer in Asian countries. J. Med. Genet. 2016, 53, 15–23. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J. The role of BRCA1 in homologous recombination repair in response to replication stress: Significance in tumorigenesis and cancer therapy. Cell Biosci. 2013, 3, 11. [Google Scholar] [CrossRef] [PubMed]
- Krainer, M.; Silva-Arrieta, S.; FitzGerald, M.G.; Shimada, A.; Ishioka, C.; Kanamaru, R.; MacDonald, D.J.; Unsal, H.; Finkelstein, D.M.; Bowcock, A.; et al. Differential contributions of BRCA1 and BRCA2 to early-onset breast cancer. N. Engl. J. Med. 1997, 336, 1416–1421. [Google Scholar] [CrossRef]
- Rashid, M.U.; Muhammad, N.; Bajwa, S.; Faisal, S.; Tahseen, M.; Bermejo, J.L.; Amin, A.; Loya, A.; Hamann, U. High prevalence and predominance of BRCA1 germline mutations in Pakistani triple-negative breast cancer patients. BMC Cancer 2016, 16, 673. [Google Scholar] [CrossRef]
- Heidemann, S.; Fischer, C.; Engel, C.; Fischer, B.; Harder, L.; Schlegelberger, B.; Niederacher, D.; Goecke, T.O.; Doelken, S.C.; Dikow, N.; et al. Double heterozygosity for mutations in BRCA1 and BRCA2 in German breast cancer patients: Implications on test strategies and clinical management. Breast Cancer Res. Treat. 2012, 134, 1229–1239. [Google Scholar] [CrossRef]
- Roy, R.; Chun, J.; Powell, S.N. BRCA1 and BRCA2: Different roles in a common pathway of genome protection. Nat. Rev. Cancer 2011, 12, 68–78. [Google Scholar] [CrossRef]
- Park, B.; Sohn, J.Y.; Yoon, K.A.; Lee, K.S.; Cho, E.H.; Lim, M.C.; Yang, M.J.; Park, S.J.; Lee, M.H.; Lee, S.Y.; et al. Characteristics of BRCA1/2 mutations carriers including large genomic rearrangements in high risk breast cancer patients. Breast Cancer Res. Treat. 2017, 163, 139–150. [Google Scholar] [CrossRef]
- Fong, P.C.; Boss, D.S.; Yap, T.A.; Tutt, A.; Wu, P.; Mergui-Roelvink, M.; Mortimer, P.; Swaisland, H.; Lau, A.; O’Connor, M.J.; et al. Inhibition of poly (ADP-ribose) polymerase in tumors from BRCA mutation carriers. N. Engl. J. Med. 2009, 361, 123–134. [Google Scholar] [CrossRef]
- Richards, S.; Aziz, N.; Bale, S.; Bick, D.; Das, S.; Gastier-Foster, J.; Grody, W.W.; Hegde, M.; Lyon, E.; Spector, E.; et al. Standards and guidelines for the interpretation of sequence variants: A joint consensus recommendation of the American College of Medical Genetics and Genomics and the Association for Molecular Pathology. Genet. Med. Off. J. Am. Coll. Med. Genet. 2015, 17, 405–424. [Google Scholar] [CrossRef] [PubMed]
- Park, K.S.; Cho, E.Y.; Nam, S.J.; Ki, C.S.; Kim, J.W. Comparative analysis of BRCA1 and BRCA2 variants of uncertain significance in patients with breast cancer: A multifactorial probability-based model versus ACMG standards and guidelines for interpreting sequence variants. Genet. Med. Off. J. Am. Coll. Med. Genet. 2016, 18, 1250–1257. [Google Scholar] [CrossRef] [PubMed]
- Lindor, N.M.; Guidugli, L.; Wang, X.; Vallee, M.P.; Monteiro, A.N.; Tavtigian, S.; Goldgar, D.E.; Couch, F.J. A review of a multifactorial probability-based model for classification of BRCA1 and BRCA2 variants of uncertain significance (VUS). Hum. Mutat. 2012, 33, 8–21. [Google Scholar] [CrossRef] [PubMed]
- Landrum, M.J.; Lee, J.M.; Benson, M.; Brown, G.R.; Chao, C.; Chitipiralla, S.; Gu, B.; Hart, J.; Hoffman, D.; Jang, W.; et al. ClinVar: Improving access to variant interpretations and supporting evidence. Nucleic Acids Res. 2018, 46, D1062–D1067. [Google Scholar] [CrossRef]
- Den Dunnen, J.T.; Dalgleish, R.; Maglott, D.R.; Hart, R.K.; Greenblatt, M.S.; McGowan-Jordan, J.; Roux, A.F.; Smith, T.; Antonarakis, S.E.; Taschner, P.E. HGVS Recommendations for the Description of Sequence Variants: 2016 Update. Hum. Mutat. 2016, 37, 564–569. [Google Scholar] [CrossRef]


| Characteristic | BRCA1 and BRCA2 (N = 4) | BRCA1 and BRCA2 VUS (N = 7) | |
|---|---|---|---|
| Age at diagnosis (year) | Mean (range) | 36 (31–43) | 32 (28–36) |
| Sex | Female Male | 4 0 | 7 0 |
| Reason for BRCA test | Family history of breast cancer Family history of ovarian cancer Bilateral breast cancer Age < 40 years | 4 1 2 3 | 1 2 0 5 |
| Married | Yes No | 3 1 | 3 4 |
| Menopausal status | Premenopausal | 4 | 7 |
| Family history of cancer | Breast Ovary Other | 3 1 0 | 2 2 4 |
| Prophylactic mastectomy | Yes No | 3 1 | 3 4 |
| Prophylactic oophorectomy | Yes No | 3 1 | 0 7 |
| Surgical record | TM BCS | 2 2 | 1 6 |
| TNM stage | I II III | 1 3 0 | 2 4 1 |
| Pathology | Invasive ductal carcinoma | 4 | 7 |
| IHC subtype | ER (−)/PR (+)/HER2 (−) ER (+)/PR (+)/HER2 (−) ER (+)/PR (+)/HER2 (+) ER (−)/PR (−)/HER2 (−) | 0 1 0 3 | 1 0 1 5 |
| No. | Age at Diagnosis | Gene | Mutation | Pathogenic |
|---|---|---|---|---|
| 1 | 43 | BRCA1 BRCA2 | c.390C > A (p.Tyr130Ter) c.5576_5579del (p.Ile1859fs) | P P |
| 2 | 36 | BRCA1 BRCA2 | c.922_924delinsT (p.Lys307_Ser308insTer) c.3599_3600del (p.Asp1199_Cys1200insTer) | P P |
| 3 | 31 | BRCA1 BRCA2 | c.5496_5506delinsA (p.Val1833fs) c.7480C > T (p.Arg2494Ter) | P p |
| 4 | 35 | BRCA1 BRCA2 | c.5030_5033del (p.Thr1677fs) c.1399A > T (p.Lys467Ter) | P P |
| 5 | 34 | BRCA1 BRCA2 | c.5445G > A (p.Trp1815Ter) c.10150C > T (p.Arg3384Ter) | P VUS |
| 6 | 28 | BRCA1 BRCA2 | c.3627dupA (p.Glu1210Argfs) c.5969A > C (p.Asp1990Ala) | P VUS |
| 7 | 28 | BRCA1 BRCA2 BRCA2 BRCA2 | c.3627dupA (p.Glu1210Argfs) c.1362A > G (P.Lys454=) c.2127G > C (p.Leu709=) c.5785A > G (p.Ile1929Val) | P VUS → B VUS → B VUS → B |
| 8 | 36 | BRCA1 BRCA2 | c.4933delA (p.Arg1645Glyfs) c.8187G > T (p.Lys2729Asn) | P VUS → B |
| 9 | 35 | BRCA1 BRCA2 | c.302-2A > C c.1568A > G (p.His523Arg) | P VUS |
| 10 | 30 | BRCA1 BRCA2 | c.2405_2406delTG (p.Val802Glufs) c.8187G > T (p.Lys2729Asn) | P VUS → B |
| 11 | 35 | BRCA1 BRCA2 | c.5080G > T (p.Glu1694Ter) c.5969A > C (p.Asp1990Ala) | P VUS |
| No. | NeoadjuvantCTx. | Adjuvant Therapy | Recurrence | Palliative CTx. | OS (Months) |
|---|---|---|---|---|---|
| 1 | AC + D | RTx. | Yes | Yes (TC) | 42 |
| 2 | - | CTx. (TAC) + RTx. | No | No | 48 |
| 3 | - | CTx. (FAC) + Tamoxifen | No | No | 96 |
| 4 | - | - | No | No | 126 |
| 5 | AC + D | - | No | No | 42 |
| 6 | - | - | F/U loss | - | 3 |
| 7 | - | CTx. (FAC) + RTx. | No | - | 62 |
| 8 | AC | CTx. (docetaxel and Herceptin) + Tamoxifen + RTx. | No | - | 48 |
| 9 | AC + D | RTx. | Yes | Yes (TC) | 39 |
| 10 | AC + D | RTx | No | - | 40 |
| 11 | AC + D | RTx. | No | - | 40 |
| No. | Parous Status | Family History | No. of Relatives with Cancer | Type of Relative with Breast/Ovarian Cancer | Remarks |
|---|---|---|---|---|---|
| 1 | P | Breast cancer | 1 | Mother only | - |
| 2 | P | Breast cancer | 1 | Grandmother only | - |
| 3 | P | Breast cancer | 1 | Sister only | - |
| 4 | N | Ovarian cancer | 1 | Grandmother only | - |
| 5 | N | - | - | - | - |
| 6 | N | Breast cancer Ovarian cancer | 1 1 | Sister only Aunt only | gBRCA1m - |
| 7 | N | Ovarian cancer Cervical cancer Colon cancer | 111 | Grandmother only - - | - |
| 8 | P | Hepatocellular carcinoma | 1 | - | - |
| 9 | P | Gastric cancer | 1 | - | - |
| 10 | N | Colon cancer | 1 | - | - |
| 11 | P | Breast cancer | 1 | Mother only | - |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hur, J.Y.; Kim, J.-Y.; Ahn, J.S.; Im, Y.-H.; Lee, J.; Kwon, M.; Park, Y.H. Clinical Characteristics of Korean Breast Cancer Patients Who Carry Pathogenic Germline Mutations in Both BRCA1 and BRCA2: A Single-Center Experience. Cancers 2020, 12, 1306. https://doi.org/10.3390/cancers12051306
Hur JY, Kim J-Y, Ahn JS, Im Y-H, Lee J, Kwon M, Park YH. Clinical Characteristics of Korean Breast Cancer Patients Who Carry Pathogenic Germline Mutations in Both BRCA1 and BRCA2: A Single-Center Experience. Cancers. 2020; 12(5):1306. https://doi.org/10.3390/cancers12051306
Chicago/Turabian StyleHur, Joon Young, Ji-Yeon Kim, Jin Seok Ahn, Young-Hyuck Im, Jiyun Lee, Minsuk Kwon, and Yeon Hee Park. 2020. "Clinical Characteristics of Korean Breast Cancer Patients Who Carry Pathogenic Germline Mutations in Both BRCA1 and BRCA2: A Single-Center Experience" Cancers 12, no. 5: 1306. https://doi.org/10.3390/cancers12051306
APA StyleHur, J. Y., Kim, J.-Y., Ahn, J. S., Im, Y.-H., Lee, J., Kwon, M., & Park, Y. H. (2020). Clinical Characteristics of Korean Breast Cancer Patients Who Carry Pathogenic Germline Mutations in Both BRCA1 and BRCA2: A Single-Center Experience. Cancers, 12(5), 1306. https://doi.org/10.3390/cancers12051306






