Innate and Adaptive Immunity Linked to Recognition of Antigens Shared by Neural Crest-Derived Tumors
Abstract
1. Introduction
2. Neural Crest-Derived Cells (NCDCs)
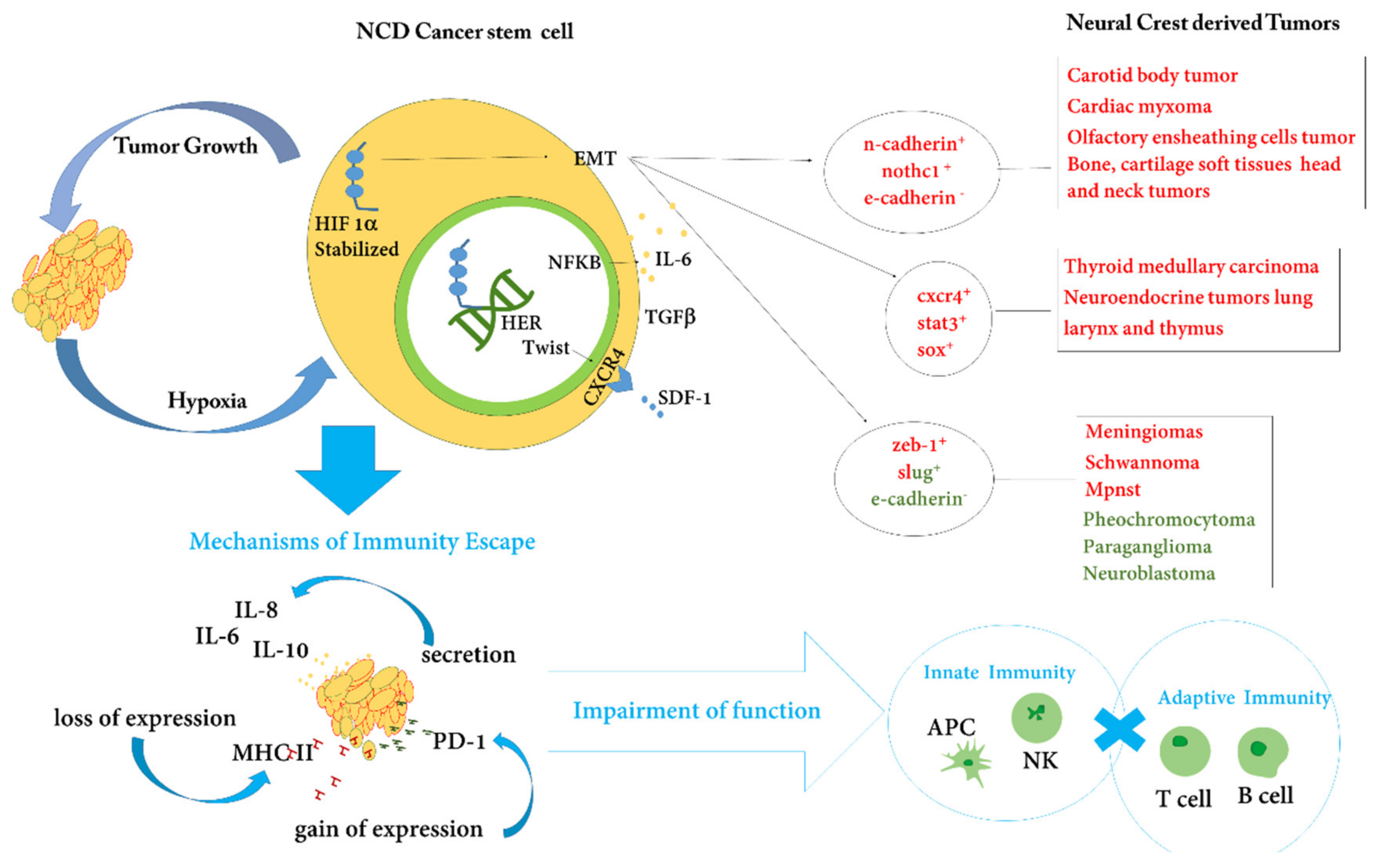
2.1. Epithelial-Mesenchymal Transitions in NCDCs
2.2. NCDCs and the Immune System
3. Neural Crest-Derived Tumors (NCDTs)
3.1. Melanocytic Tumors Cutaneous/Mucosal
3.2. Medullary Thyroid Carcinoma
3.3. Pheocromocytoma and Paragangliomas
3.4. Neuroblastoma
3.5. Schwannoma, Neurofibroma, and MPNST
3.6. Meningioma
3.7. Tumors Of Not Established NC Origin
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ALDH | Aldehyde dehydrogenases |
| alpha-SMA | Alpha-smooth muscle actin |
| c-Myc | Avian myelocytomatosis virus oncogene cellular homolog |
| bFGF | Basic fibroblast growth factor |
| BST2 | Bone Marrow Stromal Cell Antigen 2 |
| BMP | Bone morphogenetic proteins |
| CXCR-4 | C-X-C chemokine receptor type 4 |
| CXCL12 | C-X-C Motif Chemokine Ligand 12 |
| CMs | Cardiac myxomas |
| CC | Carney complex |
| CNS | Central nervous system |
| CDKN2A | Cyclin-dependent kinase Inhibitor 2A |
| DGS | Di George Syndrome |
| DNS | Dispersed neuroendocrine system |
| EED | Embryonic Ectoderm Development |
| ET3 | Endothelin 3 |
| ETRB | Endothelin receptor B |
| EMA | Epithelial membrane antigen |
| EMT | Epithelial–mesenchymal transition |
| ERK | Extracellular signal-regulated kinase |
| FMTC | Familial medullary thyroid carcinoma |
| GFRa1 | GDNF Family Receptor Alpha 1 |
| GDNF | Glial cell line–derived neurotrophic factor |
| GSK | Glycogen synthase kinase |
| GAP43 | Growth Associated Protein 43 |
| HER | Hypoxia-elements response |
| HIF | Hypoxia-inducible factor |
| EZH1/2 | Histone methyltransferases enhancer of zeste homolog 1 and 2 |
| iNOS | Inducible nitric oxide synthase |
| IFN-γ | Interferon- γ |
| IL | Interleukin |
| JNK | Jun N-terminal kinase |
| KRT 19 | Keratin, type I cytoskeletal 19 |
| KLF4 | Kruppel Like Factor 4 |
| LIF | Leukemia Inhibitory Factor |
| LOXL2 | Lysyl oxidase-like 2 |
| MPNST | Malignant peripheral nerve sheath tumor |
| MTC | Medullary thyroid carcinoma |
| MS | Melanotic schwannoma |
| MSCs | Mesenchymal stem cells |
| MMP | Metalloprotease enzymes |
| H3K27 | Methylation of histone H3 on lysine 27 |
| MITF-M | Microphthalmia-associated transcription factor |
| MAPK | Mitogen-activated protein kinase |
| Merlin | Moesin-ezrin-radixin-like protein |
| MCP-1 | Monocyte chemoattractant protein-1 |
| MEN2A | Multiple Endocrine Neoplasia type 2A |
| CDH2 | N-cadherin-2 |
| NK cells | Natural Killer cells |
| NC | Neural crest |
| NCDT | Neural Crest Cell-Derived Tumors |
| NCC | Neural crest cells |
| NCDCs | Neural crest-derived cells |
| NTs | Neuroblastic tumors |
| NBs | Neuroblastoma |
| NRAS | Neuroblastoma ras viral oncogene homolog |
| NF2 | Neurofibromatosis type 2 |
| NFKB | Nuclear factor kappa-light-chain-enhancer of activated B cells |
| NF-kB | Nuclear factor κ B |
| PG | Paraganglia |
| PI3K | Phosphatidylinositol-3-Kinase |
| PCP | Planar cell polarity |
| PRC2 | Polycomb repressive complex 2 |
| AKT | Protein kinase B |
| PRKAR1A | Protein Kinase CAMP-Dependent Type I Regulatory Subunit Alpha |
| ZEB1 | Proto-oncogene zinc finger E-box binding homeobox 1 |
| RHOB | Ras homolog gene family, member B |
| RAC1 | Ras-related C3 botulinum toxin substrate 1 |
| Ret | Rearranged During Transfection |
| c-Kit | Receptor tyrosine kinase |
| ROCK | Rho-associated coiled coil-containing protein kinases |
| SC | Schwannomas |
| STAT | Signal transducer and activator of transcription |
| SCLC | Small cell lung cancer |
| SMAD | Small mother against decapentaplegic |
| SMO | Smoothened homolog |
| SNAIL | Snail family transcriptional repressor |
| SOX | Sry-box transcription factors |
| SCF | Stem cell factor |
| SDF1 | Stromal cell-derived factor 1 |
| SDHB | Succinate dehydrogenase complex iron sulfur subunit B |
| SUZ12 | Suppressor of Zeste 12 Protein Homolog |
| TRAF7 | TNF Receptor Associated Factor 7 |
| TKR | Total knee replacement |
| TCF 3 | Transcription factor 3 |
| TAM | Tumor-associated Macrophages |
| TIL | Tumor infiltrating lymphocytes |
| TNF | Tumor necrosis factor |
| TP53 | Tumor Protein P53 |
| Twist 1 | Twist Family BHLH Transcription Factor 1 |
| BRAF | V-raf murine sarcoma viral oncogene homolog B1 |
| VEGF | Vascular endothelial growth factor |
| Wnt | Wingless-type MMTV integration site family member |
| ZIKV | ZIKA virus |
| ZIC | Zinc finger of the cerebellum protein family |
| ZEB-protein | Zinc-finger E-box binding homeobox |
References
- Etchevers, H.C.; Dupin, E.; Le Douarin, N.M. The diverse neural crest: From embryology to human pathology. Development 2019, 146, dev169821. [Google Scholar] [CrossRef] [PubMed]
- Dupin, E.; Le Douarin, N.M. The neural crest, a multifaceted structure of the vertebrates. Birth Defects Res. C Embryo Today 2014, 102, 187–209. [Google Scholar] [CrossRef] [PubMed]
- Candeloro, P.; Grande, E.; Raimondo, R.; Di Mascolo, D.; Gentile, F.; Coluccio, M.L.; Perozziello, G.; Malara, N.; Francardi, M.; Di Fabrizio, E. Raman database of amino acids solutions: A critical study of Extended Multiplicative Signal Correction. Analyst 2013, 138, 7331–7340. [Google Scholar] [CrossRef]
- Perozziello, G.; La Rocca, R.; Cojoc, G.; Liberale, C.; Malara, N.; Simone, G.; Candeloro, P.; Anichini, A.; Tirinato, L.; Gentile, F.; et al. Microfluidic devices modulate tumor cell line susceptibility to NK cell recognition. Small 2012, 8, 2886–2894. [Google Scholar] [CrossRef] [PubMed]
- Maguire, L.H.; Thomas, A.R.; Goldstein, A.M. Tumors of the neural crest: Common themes in development and cancer. Dev. Dyn. 2015, 244, 311–322. [Google Scholar] [CrossRef] [PubMed]
- Gall, T.M.H.; Frampton, A.E. Gene of the month: E-cadherin (CDH1). J. Clin. Pathol. 2013, 66, 928–932. [Google Scholar] [CrossRef]
- Hatta, K.; Takeichi, M. Expression of N-cadherin adhesion molecules associated with early morphogenetic events in chick development. Nature 1986, 320, 447–449. [Google Scholar] [CrossRef]
- Brafman, D.; Willert, K. Wnt/β-catenin signaling during early vertebrate neural development. Dev. Neurobiol. 2017, 77, 1239–1259. [Google Scholar] [CrossRef]
- Semenza, G.L. Hypoxia and cancer. Cancer Metastasis Rev. 2007, 26, 223–224. [Google Scholar] [CrossRef]
- Barriga, E.H.; Maxwell, P.H.; Reyes, A.E.; Mayor, R. The hypoxia factor Hif-1α controls neural crest chemotaxis and epithelial to mesenchymal transition. J. Cell Biol. 2013, 201, 759–776. [Google Scholar] [CrossRef]
- Kucia, M.; Reca, R.; Miekus, K.; Wanzeck, J.; Wojakowski, W.; Janowska-Wieczorek, A.; Ratajczak, J.; Ratajczak, M.Z. Trafficking of Normal Stem Cells and Metastasis of Cancer Stem Cells Involve Similar Mechanisms: Pivotal Role of the SDF-1-CXCR4 Axis. Stem Cells 2005, 23, 879–894. [Google Scholar] [CrossRef] [PubMed]
- Di Vito, A.; Scali, E.; Ferraro, G.; Mignogna, C.; Presta, I.; Camastra, C.; Palmieri, C.; Donato, G.; Barni, T. Elastofibroma dorsi: A histochemical and immunohistochemical study. Eur. J. Histochem. 2015, 59, 2459. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Vega, F.M.; Ridley, A.J. The RhoB small GTPase in physiology and disease. Small GTPases 2018, 9, 384–393. [Google Scholar] [CrossRef] [PubMed]
- Cano, A.; Pérez-Moreno, M.A.; Rodrigo, I.; Locascio, A.; Blanco, M.J.; Del Barrio, M.G.; Portillo, F.; Nieto, M.A. The transcription factor Snail controls epithelial-mesenchymal transitions by repressing E-cadherin expression. Nat. Cell Biol. 2000, 2, 76–83. [Google Scholar] [CrossRef] [PubMed]
- Hegarty, S.V.; Sullivan, A.M.; O’Keeffe, G.W. Zeb2: A multifunctional regulator of nervous system development. Prog. Neurobiol. 2015, 132, 81–95. [Google Scholar] [CrossRef]
- Nieszporek, A.; Skrzypek, K.; Adamek, G.; Majka, M. Molecular mechanisms of epithelial to mesenchymal transition in tumor metastasis. Acta Biochim. Pol. 2019, 66, 509–520. [Google Scholar] [CrossRef]
- Nichane, M.; Ren, X.; Bellefroid, E.J. Self-regulation of Stat3 activity coordinates cell-cycle progression and neural crest specification. EMBO J. 2010, 29, 55–67. [Google Scholar] [CrossRef]
- Zage, P.E.; Whittle, S.B.; Shohet, J.M. CD114: A New Member of the Neural Crest-Derived Cancer Stem Cell Marker Family. J. Cell. Biochem. 2017, 118, 221–231. [Google Scholar] [CrossRef]
- Kudo-Saito, C.; Shirako, H.; Ohike, M.; Tsukamoto, N.; Kawakami, Y. CCL2 is critical for immunosuppression to promote cancer metastasis. Clin. Exp. Metastasis 2013, 30, 393–405. [Google Scholar] [CrossRef]
- Kudo-Saito, C.; Shirako, H.; Takeuchi, T.; Kawakami, Y. Cancer Metastasis Is Accelerated through Immunosuppression during Snail-Induced EMT of Cancer Cells. Cancer Cell 2009, 15, 195–206. [Google Scholar] [CrossRef]
- Vaglia, J.L.; Hall, B.K. Regulation of neural crest cell populations: Occurrence, distribution and underlying mechanisms. Int. J. Dev. Biol. 1999, 43, 95–110. [Google Scholar] [PubMed]
- Gosain, A. Established and emerging concepts in Hirschsprung’s-associated enterocolitis. Pediatr. Surg. Int. 2016, 32, 313–320. [Google Scholar] [CrossRef] [PubMed]
- Iwashita, T.; Kruger, G.M.; Pardal, R.; Kiel, M.J.; Morrison, S.J. Hirschsprung disease is linked to defects in neural crest stem cell function. Science 2003, 301, 972–976. [Google Scholar] [CrossRef] [PubMed]
- Fu, M.; Tam, P.K.H.; Sham, M.H.; Lui, V.C.H. Embryonic development of the ganglion plexuses and the concentric layer structure of human gut: A topographical study. Anat. Embryol. 2004, 208, 33–41. [Google Scholar] [CrossRef]
- Escot, S.; Blavet, C.; Faure, E.; Zaffran, S.; Duband, J.L.; Fournier-Thibault, C. Disruption of CXCR4 signaling in pharyngeal neural crest cells causes DiGeorge syndrome-like malformations. Development 2016, 143, 582–588. [Google Scholar] [CrossRef]
- Allen, C.D.C.; Ansel, K.M.; Low, C.; Lesley, R.; Tamamura, H.; Fujii, N.; Cyster, J.G. Germinal center dark and light zone organization is mediated by CXCR4 and CXCR5. Nat. Immunol. 2004, 5, 943–952. [Google Scholar] [CrossRef]
- Hargreaves, D.C.; Hyman, P.L.; Lu, T.T.; Ngo, V.N.; Bidgol, A.; Suzuki, G.; Zou, Y.R.; Littman, D.R.; Cyster, J.G. A coordinated change in chemokine responsiveness guides plasma cell movements. J. Exp. Med. 2001, 194, 45–56. [Google Scholar] [CrossRef]
- Malara, N.; Trunzo, V.; Foresta, U.; Amodio, N.; De Vitis, S.; Roveda, L.; Fava, M.; Coluccio, M.L.; Macrì, R.; Di Vito, A.; et al. Ex-vivo characterization of circulating colon cancer cells distinguished in stem and differentiated subset provides useful biomarker for personalized metastatic risk assessment. J. Transl. Med. 2016, 14, 133. [Google Scholar] [CrossRef]
- Elmaci, I.; Altinoz, M.A.; Sari, R. Immune Pathobiology of Schwannomas: A Concise Review. J. Neurol. Surg. Part A Cent. Eur. Neurosurg. 2018, 79, 159–162. [Google Scholar] [CrossRef]
- Bayless, N.L.; Greenberg, R.S.; Swigut, T.; Wysocka, J.; Blish, C.A. Zika Virus Infection Induces Cranial Neural Crest Cells to Produce Cytokines at Levels Detrimental for Neurogenesis. Cell Host Microb. 2016, 20, 423–428. [Google Scholar] [CrossRef]
- Yang, E.V.; Kim, S.J.; Donovan, E.L.; Chen, M.; Gross, A.C.; Webster Marketon, J.I.; Barsky, S.H.; Glaser, R. Norepinephrine upregulates VEGF, IL-8, and IL-6 expression in human melanoma tumor cell lines: Implications for stress-related enhancement of tumor progression. Brain. Behav. Immun. 2009, 23, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Le Douarin, N.M.; Dupin, E. Multipotentiality of the neural crest. Curr. Opin. Genet. Dev. 2003, 13, 529–536. [Google Scholar] [CrossRef] [PubMed]
- Young, H.M.; Hearn, C.J.; Farlie, P.G.; Canty, A.J.; Thomas, P.Q.; Newgreen, D.F. GDNF is a chemoattractant for enteric neural cells. Dev. Biol. 2001, 229, 503–516. [Google Scholar] [CrossRef] [PubMed]
- Natarajan, D.; Marcos-Gutierrez, C.; Pachnis, V.; de Graaff, E. Requirement of signaling by receptors tyrosine kinase RET for the directed migration of enteric nervous system progenitor cells during mammalian embryogenesis. Development 2002, 129, 5151–5160. [Google Scholar] [PubMed]
- Johnson, M.D.; O’Connell, M.J.; Vito, F.; Pilcher, W. Bone morphogenetic protein 4 and its receptors are expressed in the leptomeninges and meningiomas and signal via the smad pathway. J. Neuropathol. Exp. Neurol. 2009, 68, 1177–1183. [Google Scholar] [CrossRef]
- Montesano, R.; Sarközi, R.; Schramek, H. Bone morphogenetic protein-4 strongly potentiates growth factor-induced proliferation of mammary epithelial cells. Biochem. Biophys. Res. Commun. 2008, 374, 164–168. [Google Scholar] [CrossRef]
- Fujii, S.; Yoshida, S.; Inagaki, E.; Hatou, S.; Tsubota, K.; Takahashi, M.; Shimmura, S.; Sugita, S. Immunological Properties of Neural Crest Cells Derived from Human Induced Pluripotent Stem Cells. Stem Cells Dev. 2019, 28, 28–43. [Google Scholar] [CrossRef]
- Sugita, S.; Kamao, H.; Iwasaki, Y.; Okamoto, S.; Hashiguchi, T.; Iseki, K.; Hayashi, N.; Mandai, M.; Takahashi, M. Inhibition of T-cell activation by retinal pigment epithelial cells derived from induced pluripotent stem cells. Investig. Ophthalmol. Vis. Sci. 2015, 56, 1051–1062. [Google Scholar] [CrossRef]
- Liu, Y.; Jing, H.; Kou, X.; Chen, C.; Liu, D.; Jin, Y.; Lu, L.; Shi, S. PD-1 is required to maintain stem cell properties in human dental pulp stem cells. Cell Death Differ. 2018, 25, 1350–1360. [Google Scholar] [CrossRef]
- Dondero, A.; Pastorino, F.; Della Chiesa, M.; Corrias, M.V.; Morandi, F.; Pistoia, V.; Olive, D.; Bellora, F.; Locatelli, F.; Castellano, A.; et al. PD-L1 expression in metastatic neuroblastoma as an additional mechanism for limiting immune surveillance. Oncoimmunology 2016, 5, e1064578. [Google Scholar] [CrossRef]
- Wang, S.; Liechty, B.; Patel, S.; Weber, J.S.; Hollmann, T.J.; Snuderl, M.; Karajannis, M.A. Programmed death ligand 1 expression and tumor infiltrating lymphocytes in neurofibromatosis type 1 and 2 associated tumors. J. Neurooncol. 2018, 138, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Obeid, J.M.; Erdag, G.; Smolkin, M.E.; Deacon, D.H.; Patterson, J.W.; Chen, L.; Bullock, T.N.; Slingluff, C.L. PD-L1, PD-L2 and PD-1 expression in metastatic melanoma: Correlation with tumor-infiltrating immune cells and clinical outcome. Oncoimmunology 2016, 5, e1235107. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.D. PD-L1 expression in meningiomas. J. Clin. Neurosci. 2018, 57, 149–151. [Google Scholar] [CrossRef] [PubMed]
- Shurell, E.; Singh, A.S.; Crompton, J.G.; Jensen, S.; Li, Y.; Dry, S.; Nelson, S.; Chmielowski, B.; Bernthal, N.; Federman, N.; et al. Characterizing the immune microenvironment of malignant peripheral nerve sheath tumor by PD-L1 expression and presence of CD8+ tumor infiltrating lymphocytes. Oncotarget 2016, 7, 64300–64308. [Google Scholar] [CrossRef]
- Kemper, K.; De Goeje, P.L.; Peeper, D.S.; Van Amerongen, R. Phenotype switching: Tumor cell plasticity as a resistance mechanism and target for therapy. Cancer Res. 2014, 74, 5937–5941. [Google Scholar] [CrossRef]
- Hodis, E.; Watson, I.R.; Kryukov, G.V.; Arold, S.T.; Imielinski, M.; Theurillat, J.P.; Nickerson, E.; Auclair, D.; Li, L.; Place, C.; et al. A landscape of driver mutations in melanoma. Cell 2012, 150, 251–263. [Google Scholar] [CrossRef]
- Davies, H.; Bignell, G.R.; Cox, C.; Stephens, P.; Edkins, S.; Clegg, S.; Teague, J.; Woffendin, H.; Garnett, M.J.; Bottomley, W.; et al. Mutations of the BRAF gene in human cancer. Nature 2002, 417, 949–954. [Google Scholar] [CrossRef]
- Delmas, A.; Cherier, J.; Pohorecka, M.; Medale-Giamarchi, C.; Meyer, N.; Casanova, A.; Sordet, O.; Lamant, L.; Savina, A.; Pradines, A.; et al. The c-Jun/RHOB/AKT pathway confers resistance of BRAF-mutant melanoma cells to MAPK inhibitors. Oncotarget 2015, 6, 15250–15264. [Google Scholar] [CrossRef]
- Joyce, J.A.; Pollard, J.W. Microenvironmental regulation of metastasis. Nat. Rev. Cancer 2009, 9, 239–252. [Google Scholar] [CrossRef]
- Christofori, G. New signals from the invasive front. Nature 2006, 441, 444–450. [Google Scholar] [CrossRef]
- Murtas, D.; Maxia, C.; Diana, A.; Pilloni, L.; Corda, C.; Minerba, L.; Tomei, S.; Piras, F.; Ferreli, C.; Perra, M.T. Role of epithelial–mesenchymal transition involved molecules in the progression of cutaneous melanoma. Histochem. Cell Biol. 2017, 148, 639–649. [Google Scholar] [CrossRef]
- Stahl, J.M.; Sharma, A.; Cheung, M.; Zimmerman, M.; Cheng, J.Q.; Bosenberg, M.W.; Kester, M.; Sandirasegarane, L.; Robertson, G.P. Deregulated Akt3 activity promotes development of malignant melanoma. Cancer Res. 2004, 64, 7002–7010. [Google Scholar] [CrossRef]
- Paluncic, J.; Kovacevic, Z.; Jansson, P.J.; Kalinowski, D.; Merlot, A.M.; Huang, M.L.H.; Lok, H.C.; Sahni, S.; Lane, D.J.R.; Richardson, D.R. Roads to melanoma: Key pathways and emerging players in melanoma progression and oncogenic signaling. Biochim. Biophys. Acta Mol. Cell Res. 2016, 1863, 770–784. [Google Scholar] [CrossRef]
- Prasad, C.P.; Mohapatra, P.; Andersson, T. Therapy for BRAFI-resistant melanomas: Is WNT5A the answer? Cancers 2015, 7, 1900–1924. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Huo, J.W.; Chen, S.S.; Xue, J.X.; Gao, W.Y.; Li, X.Y.; Song, Y.H.; Xu, H.T.; Zhu, X.W.; Chen, K. MicroRNA-22 targets FMNL2 to inhibit Melanoma progression via the regulation of the Wnt/β-catenin signaling pathway and epithelial-mesenchymal transition. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 5332–5342. [Google Scholar] [PubMed]
- Zhu, L.; Liu, Z.; Dong, R.; Wang, X.; Zhang, M.; Guo, X.; Yu, N.; Zeng, A. MicroRNA-3662 targets ZEB1 and attenuates the invasion of the highly aggressive melanoma cell line A375. Cancer Manag. Res. 2019, 11, 5845–5856. [Google Scholar] [CrossRef] [PubMed]
- Barbero, G.; Castro, M.V.; Villanueva, M.B.; Quezada, M.J.; Fernández, N.B.; DeMorrow, S.; Lopez-Bergami, P. An Autocrine Wnt5a Loop Promotes NF-κB Pathway Activation and Cytokine/Chemokine Secretion in Melanoma. Cells 2019, 8, 1060. [Google Scholar] [CrossRef]
- Mignogna, C.; Staropoli, N.; Botta, C.; De Marco, C.; Rizzuto, A.; Morelli, M.; Di Cello, A.; Franco, R.; Camastra, C.; Presta, I.; et al. Aurora Kinase A expression predicts platinum-resistance and adverse outcome in high-grade serous ovarian carcinoma patients. J. Ovarian Res. 2016, 9, 31. [Google Scholar] [CrossRef]
- Kholmanskikh, O.; Van Baren, N.; Brasseur, F.; Ottaviani, S.; Vanacker, J.; Arts, N.; Van Der Bruggen, P.; Coulie, P.; De Plaen, E. Interleukins 1α and 1β secreted by some melanoma cell lines strongly reduce expression of MITF-M and melanocyte differentiation antigens. Int. J. Cancer 2010, 127, 1625–1636. [Google Scholar] [CrossRef]
- Perozziello, G.; Simone, G.; Candeloro, P.; Gentile, F.; Malara, N.; Larocca, R.; Coluccio, M.; Pullano, S.; Tirinato, L.; Geschke, O.; et al. A Fluidic Motherboard for Multiplexed Simultaneous and Modular Detection in Microfluidic Systems for Biological Application. Micro Nanosyst. 2010, 2, 227–238. [Google Scholar] [CrossRef]
- Kuphal, S.; Winklmeier, A.; Warnecke, C.; Bosserhoff, A.K. Constitutive HIF-1 activity in malignant melanoma. Eur. J. Cancer 2010, 46, 1159–1169. [Google Scholar] [CrossRef]
- Ball, D.W. American Thyroid Association guidelines for management of medullary thyroid cancer: An adult endocrinology perspective. Thyroid 2009, 19, 547–550. [Google Scholar] [CrossRef]
- Donis-keller, H.; Dou, S.; Chi, D.; Carlson, K.M.; Toshima, K.; Lairmore, T.C.; Howe, J.R.; Moley, J.F.; Goodfellow, P.; Wells, S.A. Mutations in the RET proto-oncogene are associated with MEN 2a and FMTC. Hum. Mol. Genet. 1993, 2, 851–856. [Google Scholar] [CrossRef] [PubMed]
- Quadro, L.; Panariello, L.; Salvatore, D.; Carlomagno, F.; Del Prete, M.; Nunziata, V.; Colantuoni, V.; Di Giovanni, G.; Brandi, M.L.; Mannelli, M. Frequent RET protooncogene mutations in multiple endocrine neoplasia type 2A. J. Clin. Endocrinol. Metab. 1994, 79, 590–594. [Google Scholar] [PubMed]
- Eng, C. Multiple Endocrine Neoplasia Type 2; Wiley Online Library: Hoboken, NJ, USA, 1993. [Google Scholar]
- Sun, X.; Cheng, G.; Hao, M.; Zheng, J.; Zhou, X.; Zhang, J.; Taichman, R.S.; Pienta, K.J.; Wang, J. CXCL12/CXCR4/CXCR7 chemokine axis and cancer progression. Cancer Metastasis Rev. 2010, 29, 709–722. [Google Scholar] [CrossRef] [PubMed]
- Bradley, C.A. CXCR4–HIF-1α interaction drives metastasis. Nat. Rev. Urol. 2018, 15, 726. [Google Scholar] [CrossRef]
- Qiao, N.; Wang, L.; Wang, T.; Li, H. Inflammatory CXCL12-CXCR4/CXCR7 axis mediates G-protein signaling pathway to influence the invasion and migration of nasopharyngeal carcinoma cells. Tumor Biol. 2016, 37, 8169–8179. [Google Scholar] [CrossRef] [PubMed]
- Werner, T.A.; Forster, C.M.; Dizdar, L.; Verde, P.E.; Raba, K.; Schott, M.; Knoefel, W.T.; Krieg, A. CXCR4/CXCR7/CXCL12 axis promotes an invasive phenotype in medullary thyroid carcinoma. Br. J. Cancer 2017, 117, 1837–1845. [Google Scholar] [CrossRef] [PubMed]
- Varricchi, G.; Loffredo, S.; Marone, G.; Modestino, L.; Fallahi, P.; Ferrari, S.M.; de Paulis, A.; Antonelli, A.; Galdiero, M.R. The immune landscape of thyroid cancer in the context of immune checkpoint inhibition. Int. J. Mol. Sci. 2019, 20, 3934. [Google Scholar] [CrossRef] [PubMed]
- Heriot, K. The Neuroendocrine System and Paragangliomas. In Practical Surgical Pathology Integrating Molecular Pathology into Your Morphologic Practice; American Society of Clinical Pathology: Hong Kong, China, 2013; pp. 60–61. ISBN 978-089189-5886. [Google Scholar]
- Wong, A.; Miller, A.R.; Metter, J.; Thomas, C.R. Locally advanced duodenal gangliocytic paraganglioma treated with adjuvant radiation therapy: Case report and review of the literature. World J. Surg. Oncol. 2005, 3, 15. [Google Scholar] [CrossRef] [PubMed]
- Astuti, D.; Latif, F.; Dallol, A.; Dahia, P.L.M.; Douglas, F.; George, E.; Sköldberg, F.; Husebye, E.S.; Eng, C.; Maher, E.R. Gene mutations in the succinate dehydrogenase subunit SDHB cause susceptibility to familial pheochromocytoma and to familial paraganglioma. Am. J. Hum. Genet. 2001, 69, 49–54. [Google Scholar] [CrossRef] [PubMed]
- Loriot, C.; Burnichon, N.; Gadessaud, N.; Vescovo, L.; Amar, L.; Libé, R.; Bertherat, J.; Plouin, P.F.; Jeunemaitre, X.; Gimenez-Roqueplo, A.P.; et al. Epithelial to mesenchymal transition is activated in metastatic pheochromocytomas and paragangliomas caused by SDHB gene mutations. J. Clin. Endocrinol. Metab. 2012, 97, 954–962. [Google Scholar] [CrossRef] [PubMed]
- Waldmann, J.; Slater, E.P.; Langer, P.; Buchholz, M.; Ramaswamy, A.; Walz, M.K.; Schmid, K.W.; Feldmann, G.; Bartsch, D.K.; Fendrich, V. Expression of the transcription factor Snail and its target gene Twist are associated with malignancy in pheochromocytomas. Ann. Surg. Oncol. 2009, 16, 1997–2005. [Google Scholar] [CrossRef]
- Lee, S.Y.; Ju, M.K.; Jeon, H.M.; Lee, Y.J.; Kim, C.H.; Park, H.G.; Han, S.I.; Kang, H.S. Oncogenic metabolism acts as a prerequisite step for induction of cancer metastasis and cancer stem cell phenotype. Oxid. Med. Cell. Longev. 2018, 2018, 1027453. [Google Scholar] [CrossRef] [PubMed]
- Shang, J.; Song, Q.; Yang, Z.; Sun, X.; Xue, M.; Chen, W.; Yang, J.; Wang, S. Analysis of PD-1 related immune transcriptional profile in different cancer types. Cancer Cell Int. 2018, 18, 1–11. [Google Scholar] [CrossRef]
- Shao, J.B.; Gao, Z.M.; Huang, W.Y.; Lu, Z.B. The mechanism of epithelial-mesenchymal transition induced by TGF-β1 in neuroblastoma cells. Int. J. Oncol. 2017, 50, 1623–1633. [Google Scholar] [CrossRef]
- Wu, J.; Cheng, P.; Huang, Z.; Tan, Q.; Qu, Y. Nodal increases the malignancy of childhood neuroblastoma cells via regulation of Zeb1. BioFactors 2019, 45, 355–363. [Google Scholar] [CrossRef]
- Selmi, A.; de Saint-Jean, M.; Jallas, A.C.; Garin, E.; Hogarty, M.D.; Bénard, J.; Puisieux, A.; Marabelle, A.; Valsesia-Wittmann, S. TWIST1 is a direct transcriptional target of MYCN and MYC in neuroblastoma. Cancer Lett. 2015, 357, 412–418. [Google Scholar] [CrossRef]
- Zhao, Z.; Rahman, M.A.; Chen, Z.G.; Shin, D.M. Multiple biological functions of Twist1 in various cancers. Oncotarget 2017, 8, 20380–20393. [Google Scholar] [CrossRef]
- Dyberg, C.; Fransson, S.; Andonova, T.; Sveinbjörnsson, B.; Lännerholm-Palm, J.; Olsen, T.K.; Forsberg, D.; Herlenius, E.; Martinsson, T.; Brodin, B.; et al. Rho-associated kinase is a therapeutic target in neuroblastoma. Proc. Natl. Acad. Sci. USA 2017, 114, E6603–E6612. [Google Scholar] [CrossRef]
- Hadjidaniel, M.D.; Muthugounder, S.; Hung, L.T.; Sheard, M.A.; Shirinbak, S.; Chan, R.Y.; Nakata, R.; Borriello, L.; Malvar, J.; Kennedy, R.J.; et al. Tumor-associated macrophages promote neuroblastoma via STAT3 phosphorylation and up-regulation of c-MYC. Oncotarget 2017, 8, 91516–91529. [Google Scholar] [CrossRef] [PubMed]
- Antonescu, C.; Louis, D.N.; Hunter, S.; Perry, A.; Reuss, D.; Stemmer-Racmaninov, A. Schwannoma. In WHO Classification of Tumours of the Central Nervous System; Louis, D.N., Ohgaki, H., Wiestler, O.D., Cavenee, W.K., Eds.; Agency for Research on Cancer: Lyon, France, 2016; pp. 214–218. ISBN 978-92-832-4492-9. [Google Scholar]
- Taurone, S.; Bianchi, E.; Attanasio, G.; Di Gioia, C.; Ierinó, R.; Carubbi, C.; Galli, D.; Pastore, F.S.; Giangaspero, F.; Filipo, R.; et al. Immunohistochemical profile of cytokines and growth factors expressed in vestibular schwannoma and in normal vestibular nerve tissue. Mol. Med. Rep. 2015, 12, 737–745. [Google Scholar] [CrossRef] [PubMed]
- De Vries, W.M.; Briaire-de Bruijn, I.H.; van Benthem, P.P.G.; van der Mey, A.G.L.; Hogendoorn, P.C.W. M-CSF and IL-34 expression as indicators for growth in sporadic vestibular schwannoma. Virchows Arch. 2019, 474, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Guadagno, E.; Presta, I.; Maisano, D.; Donato, A.; Pirrone, C.K.; Cardillo, G.; Corrado, S.D.; Mignogna, C.; Mancuso, T.; Donato, G.; et al. Role of macrophages in brain tumor growth and progression. Int. J. Mol. Sci. 2018, 19, 1005. [Google Scholar] [CrossRef] [PubMed]
- De Vries, M.; Briaire-De Bruijn, I.; Malessy, M.J.A.; De Bruïne, S.F.T.; Van Der Mey, A.G.L.; Hogendoorn, P.C.W. Tumor-associated macrophages are related to volumetric growth of vestibular schwannomas. Otol. Neurotol. 2013, 34, 347–352. [Google Scholar] [CrossRef]
- Perry, A.; Graffeo, C.S.; Carlstrom, L.P.; Raghunathan, A.; Driscoll, C.L.W.; Neff, B.A.; Carlson, M.L.; Parney, I.F.; Link, M.J.; Van Gompel, J.J. Predominance of M1 subtype among tumor-associated macrophages in phenotypically aggressive sporadic vestibular schwannoma. J. Neurosurg. 2019, 1, 1–9. [Google Scholar] [CrossRef]
- Arima, Y.; Hayashi, H.; Kamata, K.; Goto, T.M.; Sasaki, M.; Kuramochi, A.; Saya, H. Decreased expression of neurofibromin contributes to epithelial-mesenchymal transition in neurofibromatosis type 1. Exp. Dermatol. 2010, 19, e136–e141. [Google Scholar] [CrossRef]
- Liao, C.P.; Booker, R.C.; Brosseau, J.P.; Chen, Z.; Mo, J.; Tchegnon, E.; Wang, Y.; Clapp, D.W.; Le, L.Q. Contributions of inflammation and tumor microenvironment to neurofibroma tumorigenesis. J. Clin. Investig. 2018, 128, 2848–2861. [Google Scholar] [CrossRef]
- Meyer, A.; Billings, S.D. What’s new in nerve sheath tumors. Virchows Arch. 2020, 476, 65–80. [Google Scholar] [CrossRef]
- Haworth, K.B.; Arnold, M.A.; Pierson, C.R.; Choi, K.; Yeager, N.D.; Ratner, N.; Roberts, R.D.; Finlay, J.L.; Cripe, T.P. Immune profiling of NF1-associated tumors reveals histologic subtype distinctions and heterogeneity: Implications for immunotherapy. Oncotarget 2017, 8, 82037–82048. [Google Scholar] [CrossRef]
- Ng, J.; Celebre, A.; Munoz, D.G.; Keith, J.L.; Karamchandani, J.R. Sox10 is superior to s100 in the diagnosis of meningioma. Appl. Immunohistochem. Mol. Morphol. 2015, 23, 215–219. [Google Scholar] [CrossRef] [PubMed]
- Karamchandani, J.R.; Nielsen, T.O.; Van De Rijn, M.; West, R.B. Sox10 and s100 in the diagnosis of soft-tissue neoplasms. Appl. Immunohistochem. Mol. Morphol. 2012, 20, 445–450. [Google Scholar] [CrossRef] [PubMed]
- Dasgupta, K.; Jeong, J. Developmental biology of the meninges. Genesis 2019, 57, e23288. [Google Scholar] [CrossRef] [PubMed]
- Buerki, R.A.; Horbinski, C.M.; Kruser, T.; Horowitz, P.M.; James, C.D.; Lukas, R.V. An overview of meningiomas. Futur. Oncol. 2018, 14, 2161–2177. [Google Scholar] [CrossRef]
- Rouleau, G.A.; Merel, P.; Lutchman, M.; Sanson, M.; Zucman, J.; Marineau, C.; Hoang-Xuan, K.; Demczuk, S.; Desmaze, C.; Plougastel, B.; et al. Alteration in a new gene encoding a putative membrane-organizing protein causes neuro-fibromatosis type 2. Nature 1993, 363, 515–521. [Google Scholar] [CrossRef]
- Donato, G.; Ferraro, G.; Signorelli, F.; Iofrida, G.; Lavano, A.; Amorosi, A.; Maltese, L.; Perrotta, I.; Tripepi, S.; Pardatscher, K.; et al. Chordoid meningioma: Case report and literature review. Ultrastruct. Pathol. 2006, 30, 309–314. [Google Scholar] [CrossRef]
- Sugur, H.; Shastry, A.; Sadashiva, N.; Srinivas, D.; Santosh, V.; Somanna, S. Chromosomal aberrations in chordoid meningioma—An analysis. Neurol. India 2018, 66, 156–160. [Google Scholar]
- Wallesch, M.; Pachow, D.; Blücher, C.; Firsching, R.; Warnke, J.P.; Braunsdorf, W.E.K.; Kirches, E.; Mawrin, C. Altered expression of E-Cadherin-related transcription factors indicates partial epithelial-mesenchymal transition in aggressive meningiomas. J. Neurol. Sci. 2017, 380, 112–121. [Google Scholar] [CrossRef]
- Presta, I.; Guadagno, E.; Di Vito, A.; Malara, N.; Mignogna, C.; Maisano, D.; Donato, A.; Cardillo, G.; Del Basso De Caro, M.L.; Donato, G. Innate immunity may play a role in growth and relapse of chordoid meningioma. Int. J. Immunopathol. Pharmacol. 2017, 30, 429–433. [Google Scholar] [CrossRef]
- Nassehi, D.; Dyrbye, H.; Andresen, M.; Thomsen, C.; Juhler, M.; Laursen, H.; Broholm, H. Vascular endothelial growth factor A protein level and gene expression in intracranial meningiomas with brain edema. APMIS 2011, 119, 831–843. [Google Scholar] [CrossRef]
- Johnson, M.D. Transforming Growth Factor Beta Family in the Pathogenesis of Meningiomas. World Neurosurg. 2017, 104, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Reszec, J.; Rutkowski, R.; Chyczewski, L. The expression of hypoxia-inducible factor-1 in primary brain tumors. Int. J. Neurosci. 2013, 123, 657–662. [Google Scholar] [CrossRef] [PubMed]
- Presta, I.; Donato, A.; Chirchiglia, D.; Malara, N.; Donato, G. Cardiac myxoma and neural crests: A tense relationship. Cardiovasc. Pathol. 2020, 44, 107163. [Google Scholar] [CrossRef]
- Singhal, P.; Luk, A.; Rao, V.; Butany, J. Molecular basis of cardiac myxomas. Int. J. Mol. Sci. 2014, 15, 1315–1337. [Google Scholar] [CrossRef] [PubMed]
- Di Vito, A.; Mignogna, C.; Donato, G. The mysterious pathways of cardiac myxomas: A review of histogenesis, pathogenesis and pathology. Histopathology 2015, 66, 321–332. [Google Scholar] [CrossRef] [PubMed]
- Di Vito, A.; Santise, G.; Mignogna, C.; Chiefari, E.; Cardillo, G.; Presta, I.; Arturi, F.; Malara, N.; Brunetti, F.; Donato, A.; et al. Innate immunity in cardiac myxomas and its pathological and clinical correlations. Innate Immun. 2018, 24, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Pacheco-Velázquez, S.C.; Gallardo-Pérez, J.C.; Díaz, D.; Adán-Ladrón de Guevara, A.; Robledo-Cadena, D.X.; Saavedra, E.; Ruiz-Godoy, L.; Jimenez-Hernández, L.R.; Vargas-Barrón, J.; Aguilar-Ponce, J.L.; et al. Heart myxoma develops oncogenic and metastatic phenotype. J. Cancer Res. Clin. Oncol. 2019, 145, 1283–1295. [Google Scholar] [CrossRef]
- Perrotta, I.; Bruno, L.; Maltese, L.; Russo, E.; Donato, A.; Donato, G. Immunohistochemical Analysis of the Ubiquitin-conjugating Enzyme UbcH10 in Lung Cancer: A Useful Tool for Diagnosis and Therapy. J. Histochem. Cytochem. 2012, 60, 359–365. [Google Scholar] [CrossRef]
- Rosai, J. The origin of neuroendocrine tumors and the neural crest saga. Mod. Pathol. 2011, 24, S53–S57. [Google Scholar] [CrossRef]
- Becker, K.L.; Silva, O.L. Hypothesis: The bronchial Kulchitsky (K) cell as a source of humoral biologic activity. Med. Hypotheses 1981, 7, 943–949. [Google Scholar] [CrossRef]
- Gazdar, A.F.; Bunn, P.A.; Minna, J.D. Small-cell lung cancer: What we know, what we need to know and the path forward. Nat. Rev. Cancer 2017, 17, 725–737. [Google Scholar] [CrossRef] [PubMed]
- Cornell, R.A.; Eisen, J.S. Notch in the pathway: The roles of Notch signaling in neural crest development. Semin. Cell Dev. Biol. 2005, 16, 663–672. [Google Scholar] [CrossRef] [PubMed]
- Miller, S.R.; Benito, C.; Mirsky, R.; Jessen, K.R.; Baker, C.V.H. Neural crest Notch/Rbpj signaling regulates olfactory gliogenesis and neuronal migration. Genesis 2018, 56, e23215. [Google Scholar] [CrossRef] [PubMed]
- Iriki, T.; Ohnishi, K.; Fujiwara, Y.; Horlad, H.; Saito, Y.; Pan, C.; Ikeda, K.; Mori, T.; Suzuki, M.; Ichiyasu, H.; et al. The cell-cell interaction between tumor-associated macrophages and small cell lung cancer cells is involved in tumor progression via STAT3 activation. Lung Cancer 2017, 106, 22–32. [Google Scholar] [CrossRef]
- Yasuda, Y.; Ozasa, H.; Kim, Y.H. PD-L1 Expression in Small Cell Lung Cancer. J. Thorac. Oncol. 2018, 13, e40–e41. [Google Scholar] [CrossRef]
- Doedens, A.L.; Stockmann, C.; Rubinstein, M.P.; Liao, D.; Zhang, N.; DeNardo, D.G.; Coussens, L.M.; Karin, M.; Goldrath, A.W.; Johnson, R.S. Macrophage expression of hypoxia-inducible factor-1α suppresses T-cell function and promotes tumor progression. Cancer Res. 2010, 70, 7465–7475. [Google Scholar] [CrossRef]
- Burke, B.; Giannoudis, A.; Corke, K.P.; Gill, D.; Wells, M.; Ziegler-Heitbrock, L.; Lewis, C.E. Hypoxia-induced gene expression in human macrophages: Implications for ischemic tissues and hypoxia-regulated gene therapy. Am. J. Pathol. 2003, 163, 1233–1243. [Google Scholar] [CrossRef]
- Chen, J.; Jiang, C.C.; Jin, L.; Zhang, X.D. Regulation of PD-L1: A novel role of pro-survival signaling in cancer. Ann. Oncol. 2016, 27, 409–416. [Google Scholar] [CrossRef]
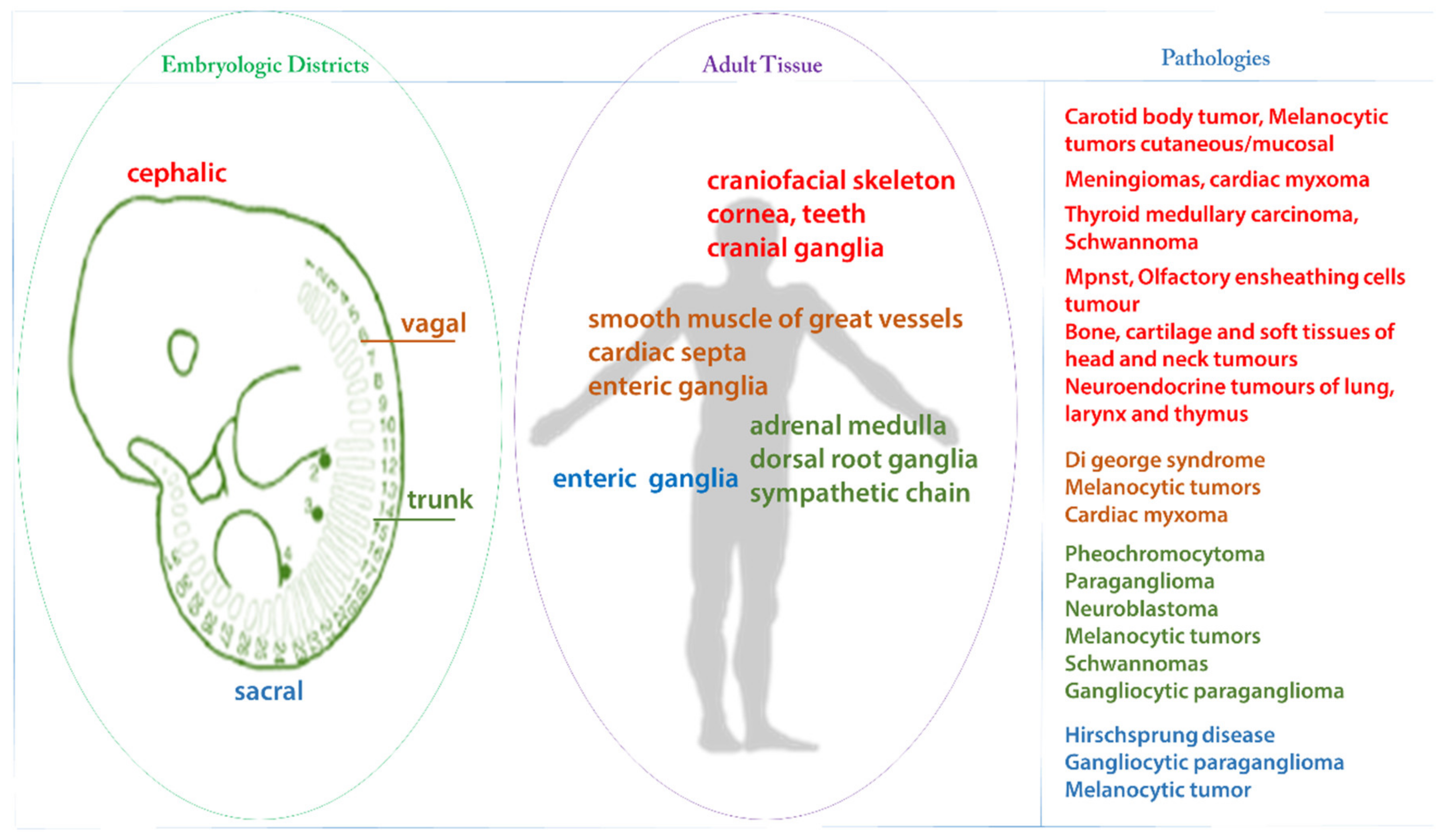
| Embryonic Derivation | Neural Crest-Derived Cell Types | Related Tumors | |||
|---|---|---|---|---|---|
| Neurons and Glial Cells | Pigment Cells | Endocrine Cells | Mesenchymal Cells | ||
| Cephalic | Sensory cranial ganglionic neurons Parasympathetic (ciliary)ganglionic neuron | Paraganglioma | |||
| Skin melanocytes, Choroid melanocytes, Other extracutaneous melanocytes (in gums, meninges, etc.) | Melanocytic tumors | ||||
| Schwann cells along PNS nerves | Schwannoma MPNST | ||||
| Olfactory Ensheating Cells | Olfactory ensheating cells tumors | ||||
| Calcitonin-producing cells | Thyroid medullary carcinoma | ||||
| Carotid body cells | Carotid body tumor | ||||
| Meninges (forebrain) | Meningiomas | ||||
| Craniofacial Osteocytes and Chondrocytes, Adipocytes, Dermal cells | Bone, cartilage and soft tissues of head and neck | ||||
| Olfactory neuroepithelium | Esthesioneuroblastoma | ||||
| Neuroendocrine cells in larynx, lungs, and thymus | Neuroendocrine tumors larynx, lungs, and thymus | ||||
| Vagal | Enteric ganglionic neurons | Melanocytes | Melanocytic tumors | ||
| Vascular smooth muscle cells Heart conotruncus | Cardiac Myxoma | ||||
| Trunk Enteric ganglionic neurons | Sympathetic ganglionic neurons Parasympathetic ganglionic neurons | Adrenal medullary cells | Pheochromocitoma Paraganglioma Neuroblastoma | ||
| Melanocytes | Melanocytic tumors | ||||
| Schwann cells along PNS nerves | Schwannoma | ||||
| Sensory (dorsal root) ganglionic neurons Satellite glial cells in PNS ganglia | Peripheral Gangliocytoma | ||||
| Enteric ganglionic neurons | Gangliocytic Paraganglioma | ||||
| Sacral | Melanocytes | Melanocytic tumors | |||
| Enteric ganglionic neurons | Gangliocytic paraganglioma | ||||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Donato, G.; Presta, I.; Arcidiacono, B.; Vismara, M.F.M.; Donato, A.; Garo, N.C.; Malara, N. Innate and Adaptive Immunity Linked to Recognition of Antigens Shared by Neural Crest-Derived Tumors. Cancers 2020, 12, 840. https://doi.org/10.3390/cancers12040840
Donato G, Presta I, Arcidiacono B, Vismara MFM, Donato A, Garo NC, Malara N. Innate and Adaptive Immunity Linked to Recognition of Antigens Shared by Neural Crest-Derived Tumors. Cancers. 2020; 12(4):840. https://doi.org/10.3390/cancers12040840
Chicago/Turabian StyleDonato, Giuseppe, Ivan Presta, Biagio Arcidiacono, Marco F.M. Vismara, Annalidia Donato, Nastassia C. Garo, and Natalia Malara. 2020. "Innate and Adaptive Immunity Linked to Recognition of Antigens Shared by Neural Crest-Derived Tumors" Cancers 12, no. 4: 840. https://doi.org/10.3390/cancers12040840
APA StyleDonato, G., Presta, I., Arcidiacono, B., Vismara, M. F. M., Donato, A., Garo, N. C., & Malara, N. (2020). Innate and Adaptive Immunity Linked to Recognition of Antigens Shared by Neural Crest-Derived Tumors. Cancers, 12(4), 840. https://doi.org/10.3390/cancers12040840







