Abstract
Aberrant regulation of the cell cycle is a typical feature of all forms of cancer. In head and neck squamous cell carcinoma (HNSCC), it is often associated with the overexpression of cyclin D1 (CCND1). However, it remains unclear how CCND1 expression changes between tumor and normal tissues and whether human papillomavirus (HPV) affects differential CCND1 expression. Here, we evaluated the expression of D-type cyclins in a cohort of 94 HNSCC patients of which 82 were subjected to whole genome expression profiling of primary tumors and paired normal mucosa. Comparative analysis of paired samples showed that CCND1 was upregulated in 18% of HNSCC tumors. Counterintuitively, CCND1 was downregulated in 23% of carcinomas, more frequently in HPV-positive samples. There was no correlation between the change in D-type cyclin expression and patient survival. Intriguingly, among the tumors with downregulated CCND1, one-third showed an increase in cyclin D2 (CCND2) expression. On the other hand, one-third of tumors with upregulated CCND1 showed a decrease in CCND2. Collectively, we have shown that CCND1 was frequently downregulated in HNSCC tumors. Furthermore, regardless of the HPV status, our data suggested that a change in CCND1 expression was alleviated by a compensatory change in CCND2 expression.
1. Introduction
Head and neck squamous cell carcinomas (HNSCC) are malignant neoplasms that arise from the mucosal epithelial surface of the upper respiratory and digestive tracts. The worldwide annual incidence of head and neck cancers is more than 550,000 cases, thus making HNSCC the sixth-most common cancer in the world [1]. Unfortunately, the incidence of this tumor type continues to increase.
HNSCC is more prevalent in men than in women, and the majority of HNSCC cases occur in patients over the age of 60 [2]. The most affected subsites are the oral cavity, oropharynx, hypopharynx, nasopharynx and larynx. Early-stage, locally contained disease responds favorably to treatment, presenting very good cure rates of 70–90%. However, advanced HNSCC exhibit aggressive loco-regional invasions, frequent second primary tumors and lymph node metastases, while distant metastases are relatively rare. Despite advances in the treatment of HNSCC using molecularly targeted therapies, the five-year survival rate of loco-regional or recurrent/metastatic diseases has remained at 50–60% [3,4].
The main risk factors for the development of head and neck cancers include tobacco exposure and alcohol consumption, which are associated with more than 70% of all HNSCC cases [5,6], and the infection of high-risk oncogenic types of human papillomavirus (HPV) [7]. Among HPV high-risk types, HPV16 and HPV18 are the most common, accounting for more than 85% of all HPV-positive (HPV(+)) tumors [8,9]. HPV status, combined with the traditional tumor-node-metastasis (TNM) staging system, is considered to be a particularly significant biological prognostic marker and distinguishes two etiologically different subtypes of HNSCC [10]. Individuals with HPV(+) HNSCCs have a considerably better prognosis compared to those who possess HPV-negative (HPV(−)) cancers [11].
In HNSCC, the alterations which lead to functional loss of the tumor suppressor function are much more frequent than oncogene-activating mutations. These changes in both HPV subtypes almost invariably deregulate cell cycle entry and progression and, thus, are detrimental to cell proliferation. In HPV(−) tumors, p53 tumor suppressor is inactivated by genetic mutations, and retinoblastoma protein (pRb) is inactivated by cyclin D1 (CCND1) and the CDK4/6 complex. On the other hand, in HPV(+) tumors, viral proteins E6 and E7 inactivate p53 and pRb, thus enabling these tumors to evade cell cycle checkpoints [8,12]. Functional loss of p53, either by mutation or proteasomal degradation, occurs early in HNSCC development in approximately 75% of cases [13,14]. Likewise, another tumor suppressor gene, CDKN2A, that encodes cyclin-dependent kinase inhibitor p16INK4a, is frequently inactivated by a copy number loss in HPV(−) HNSCC [15,16]. In contrast, p16INK4a overexpression is typical for HPV(+) HNSCC [17] and is used as a surrogate marker of HPV infection [13,16].
A mutation detected in negative regulators of the cell cycle is often paralleled by the increased expression of cyclin D1, a potent oncogene and positive regulator of the cell cycle that is encoded by the CCND1 gene [15]. CCND1 is a member of the D-type cyclin family that includes cyclin D2 (CCND2) and cyclin D3 (CCND3). Cyclins D1, D2 and D3 show roughly 50% of sequence identity at the amino acid level, and all have been shown to activate CDK4/6 and promote G1/S transition [18]. In nontransformed cells, the expression of D-type cyclins is controlled by mitogenic, adhesion and differentiation signals and, thus, integrates extracellular signals with the cell cycle. Overexpression of the D-type cyclins bypasses the requirement for mitogenic stimuli promoting unchecked proliferation and is believed to be an early cause of tumor formation [18,19,20]. Cyclin D1 protein overexpression has been reported in up to 70% of HNSCC cases [21,22], and gene amplification represents the most prominent mechanism of increased CCND1 expression [15]. In addition, amplification of CCND2 in HNSCC has also been reported [23]. Despite the frequent CCND1 upregulation and its positive role in cell proliferation, the value of CCND1 as an independent prognostic marker in HNSCC is inconsistent. Several studies have suggested that elevated CCND1 expression correlates with poor prognosis of some types of HNSCC [21,24,25]; however, other studies have not found a significant correlation [26,27,28,29].
In the present study, we examined the expression of D-type cyclins in a new cohort of 94 HNSCC patients with known clinical data. For 82 of these patients, the expression profiles of both the tumor and matching normal mucosa were obtained using DNA microarrays. Analysis of expression changes between tumor and normal tissues revealed that a considerable fraction of tumors downregulates CCND1 expression. However, the reduction of cyclin D1 expression did not correlate with a favorable clinical outcome. We also found that tumors with downregulated cyclin D1 expression frequently upregulate the expression of cyclin D2. Taken together, these results suggest a compensatory mechanism where the upregulation of cyclin D2 may be a direct consequence of the loss of related cyclin D1 function.
2. Results
2.1. Patient Characteristics
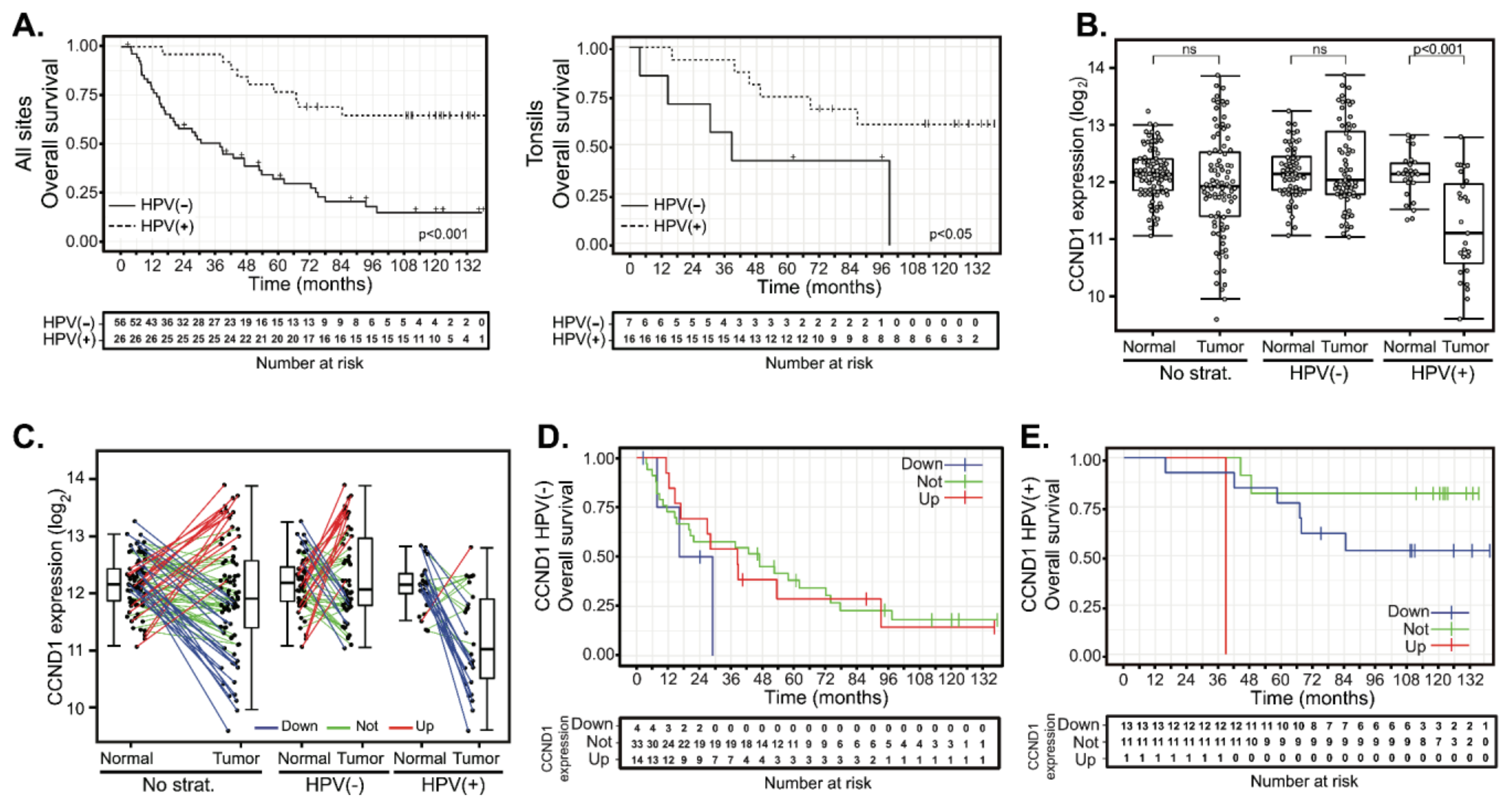
Normal (n = 86) and tumor (n = 90) samples were obtained from a total of 94 HNSCC patients from which clinical and pathological data were collected (Table 1 and Table S1). Overall, patients were users of tobacco (81%) and alcohol (59%). HPV was detected in 26 cases (28%). In contrast to previous reports [30,31,32], there was almost no difference in the average age of HNSCC diagnosis between HPV(−) and HPV(+) subgroups. Primary treatment consisted of surgery alone in 15 (16%) cases, surgery combined with adjuvant radiotherapy in 73 (78%) cases and surgery combined with adjuvant chemoradiotherapy in 6 (6%) cases. Median follow-up length was 110 months for HPV(+) patients and 29 months for HPV(−) patients. HPV infection was associated with significantly longer overall survival (OS) (Figure 1A, left panel). Five-year OS was 77% (CI 62–95%) and 32% (CI 22–48%) in HPV(+) and HPV(−) groups, respectively. Hazard ratio (HR) of HPV(−) vs HPV(+) 2.13–9.15, p < 0.001). Disease-free survival (DFS) was also shorter for HPV(−) patients (five-year survival 61%; CI 46–81%) than for HPV(+) patients (five-year survival 85%; CI 72–100%) (Figure S1A). However, the difference between HPV(−) and HPV(+) patients did not reach statistical significance (HR = 2.89, CI 0.95-8.77, p = 0.06).

Table 1.
Summary of clinical data for all patients in the present cohort (n = 94). For four patients, only samples from normal mucosa were available, and thus, the human papillomavirus (HPV) status was not determined. Statistical significance gauges the association between a variable and the HPV subtypes: (n.s.) non-significant, (*) p < 0.05 and (***) p < 0.001. Percentages are column-wise by default, row-wise if “r” is behind the value. (NA) not available.
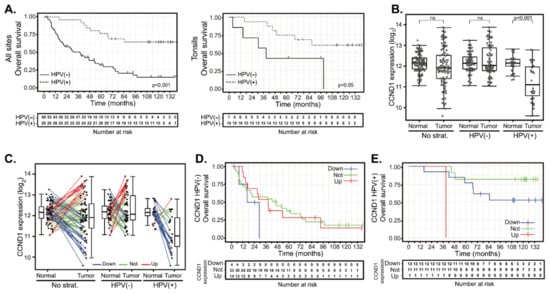
Figure 1.
CCND1 gene expression changes between tumor and normal tissues and patient survival in human papillomavirus (HPV) (+) and HPV(−) groups. (A) Kaplan-Meier plots of overall survival (OS) for HPV(−) and HPV(+) groups: entire cohort (left panel, n = 82) and tonsillar carcinoma (right panel, n = 23). (B) CCND1 gene expression in a cohort of 94 patients (left panel) and stratified according to the HPV status (middle and right panel). The boxplot displays interquartile range with the whiskers indicating the greatest and smallest observations, excluding outliers. (C) CCND1 deregulation between paired tumor and normal tissues. Paired samples are connected by the lines color-coded according to the change of CCND1 expression (red—CCND1 upregulation, fold-changes (FC) > 2; green—no deregulation, 0.5 < FC < 2 and blue—CCND1 downregulation, FC < 0.5). (D) Kaplan–Meier plots for OS stratified according to CCND1 gene deregulation in the HPV(−) group. (E) Kaplan–Meier plot for OS by CCND1 deregulation in the HPV(+) group. Up—group with CCND1 upregulated, not—group without CCND1 deregulation and down—group with CCND1 downregulated. Tick marks and crosses in (A,D,E) indicate right censoring.
We next analyzed whether HPV infection results in better prognosis of patients with the same tumor location. We analyzed tumors from the tonsils and the base of the tongue, as these subsites were significantly represented in respect of both the number and the proportion of HPV(+) tumors. Similar to the entire cohort, HPV infection of tonsil tumors was associated with significantly longer overall survival (Figure 1A, right panel). Five-year OS was 69% (CI 49–96%) and 43% (CI 18–100%) in the HPV(+) and HPV(−) groups, respectively. The hazard ratio of HPV(+) tonsil tumors was 0.3 (CI 0.09–0.99, p < 0.05). A similar trend was observed in patients with tumors originating in the base of the tongue, although in this case, statistical significance was not reached (Supplementary Figure S1B). Five-year OS was 71% (CI 24–100%) and 29% (CI 9–92%) in the HPV(+) and HPV(−) groups, respectively. The hazard ratio of the HPV(+) base of the tongue tumors was 0.27 (CI 0.07–1.11, p = 0.07).
Whole genome expression profiles were obtained for a cohort of 94 patients (90 tumor samples, 86 normal tissue samples). From this cohort, a group of 82 patients had expression profiles from both tumor and matched normal tissues. Of these 82 patients, 25 and 57 individuals had HPV(+) and HPV(−) tumors, respectively.
2.2. Deregulation of Cyclin D1 Expression in HPV(−) and in HPV(+) HNSCC Tumors Does Not Correlate with Patient Outcomes
Analysis of expression profiles from the cohort of 94 HNSCC cases revealed that the mean expression of CCND1 does not differ between the normal mucosae and carcinomas. However, CCND1 expression in tumors showed a visible spread toward both increased and decreased values (Figure 1B). Decreased CCND1 expression was significant mainly in the HPV(+) group. We thus determined the changes in expression of CCND1 between 82 paired tumor and normal tissues patient by patient. We considered CCND1 to be deregulated in the tumor if the fold-change of expression between tumor and normal tissues was above two (CCND1 upregulation) or less than 0.5 (CCND1 downregulation), respectively. This categorization resulted in the formation of three different clusters: 47 cases (57.3%) in which there was no change in CCND1 expression and 15 cases (18.3%) where CCND1 was upregulated as expected in proliferating tumor tissue. However, a substantial number of tumors downregulated CCND1 (20 cases, 24.4%) (Figure 1C). Further stratification according to HPV status revealed that CCND1 downregulation was typical for HPV(+) tumors (13 cases, 52.0%), while almost all of the remaining 11 (44.0%) HPV(+) tumors did not deregulate CCND1 expression. Only in one HPV(+) case (4.0%), CCND1 was found to be upregulated (Figure 1C). On the other hand, HPV(−) tumors were spread across all three categories: tumors with CCND1 expression upregulated (14 cases, 24.6%), unchanged (36 cases, 63.2%) or downregulated (7 cases, 12.3%). We examined whether these changes in CCND1 expression correlated with patient outcomes and clustered 76 patients with known follow-up according to their CCND1 regulation. Survival analysis revealed that neither OS (Figure 1D, Table 2 and Table S2) nor DFS (Supplementary Figure S1C and Table S2) were affected by CCND1 upregulation in the HPV(−) group. CCND1 downregulation was associated with a shorter five-year OS (Figure 1D and Table 2); however, the clinical data for this group was available for four patients only, showing no statistically significant difference (Table 2 and Table S2). In the HPV(+) group, CCND1 downregulation did not affect OS or DFS significantly (Figure 1E, Supplementary Figure S1D and Table 2). This data suggested that there is no significant association between worse clinical outcome and CCND1 upregulation and, conversely, between better outcomes in patients with CCND1 downregulation.

Table 2.
Survival analysis, overall survival (OS). Patients with paired samples and known survival (n = 76, see Table 1) were clustered according to the deregulation of D-type cyclin expression in matching samples. Thresholds for deregulation were the following: not (0.5 < fold-changes (FC) < 2), up (FC > 2) and down (FC < 0.5). HR—hazard ratio and NA—not available.
2.3. Cyclin D1 Upregulation Correlates with Bona Fide Amplification of Its Genomic Locus
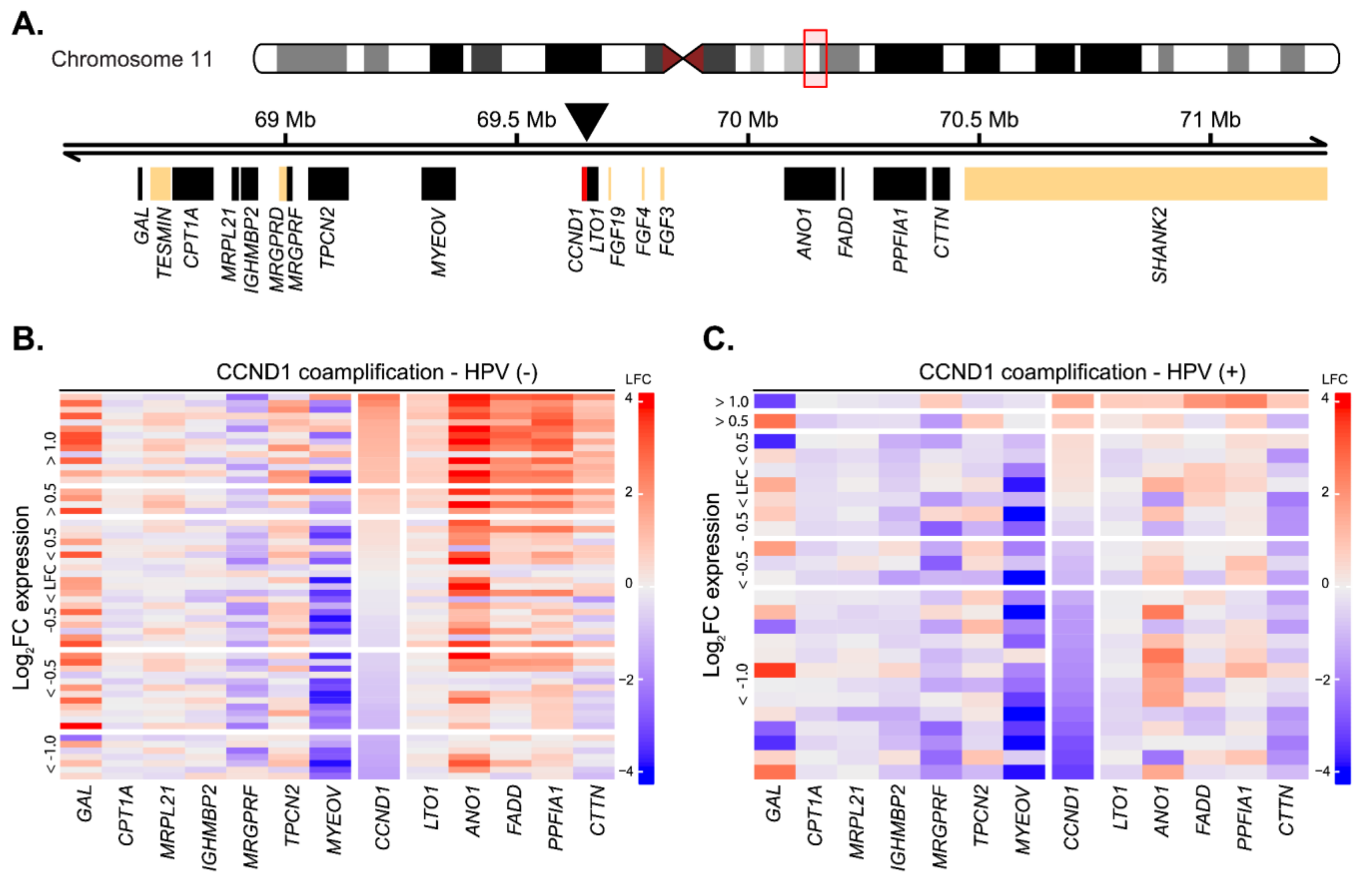
The observed low number of tumors with upregulated CCND1 expression was unexpected, as CCND1 overexpression was generally reported in up to 70% of patients at the protein level [21,22]. The common mechanism underlying CCND1 upregulation is its gene locus amplification, with frequency ranging from 17% to 50% of cases [33,34]. While we do not possess direct data on the amplification, we analyzed changes in the expression of genes surrounding the CCND1 gene as its surrogate marker [15,22]. Locus amplification would lead to upregulation of other genes in the locus, together with the CCND1 upregulation. Out of 20 genes present in the two-megabase window around the CCND1 genomic locus, we could analyze changes in the expression of 12 genes, while the expression of the other six genes was not detected by the microarray technology (Figure 2A). In the group of 15 predominantly HPV(−) cases where CCND1 expression was upregulated in the tumor tissues (FC > 2), we observed upregulation of the genes that were downstream of CCND1 in 14 (93%) cases (Figure 2B,C). Notably, ANO1 was upregulated both in the majority of HPV(−) tumors and in HPV(+) tumors, probably reflecting the fact that this protein is a potential driver oncogene [35]. With the exception of TPCN2, the genes upstream of CCND1 were not upregulated along with CCND1 in HPV(−) tumors. The expression of MRGPRF and MYEOV were rather downregulated (Figure 2B,C). The downregulation of MYEOV may be explained by frequent epigenomic silencing of the gene [36]. In summary, we observed a strong signal for amplification of the CCND1 genomic loci in HPV(−) tumors. In the HPV(+) group, the co-amplification was observed only in one case, in agreement with previously published data that amplification of the CCND1 locus is rare in HPV(+) tumors [22].
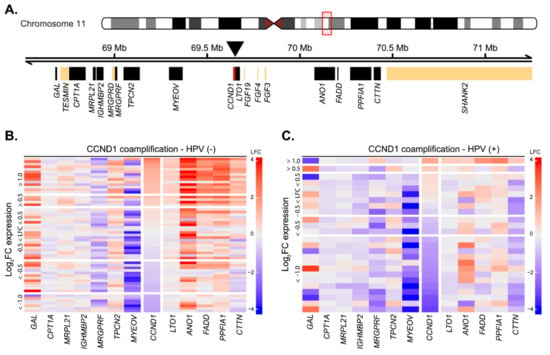
Figure 2.
CCND1 gene locus amplification as estimated from expression changes between tumor and normal tissues in HPV(+) and HPV(−) patient groups. (A) Schematics of the CCND1 gene locus spanning ~ 2 Mb indicating position of the surrounding genes. Position of the CCND1 gene (red) is indicated by the arrowhead. Black rectangles indicate expressed genes, and beige rectangles indicate genes expressed below the detection threshold. Position on chromosome 11 is given in Mb, million base pairs. (B) Heatmap of gene expression changes between tumor and normal tissues in the HPV(−) group for expressed genes from the CCND1 locus. LFC—log2 fold-change in expression. (C) Heatmap of gene expression changes between tumor and normal tissues in the HPV(+) group for expressed genes from the CCND1 locus.
2.4. Deregulation of Cyclin D2 Does Not Correlate with Patient Outcomes
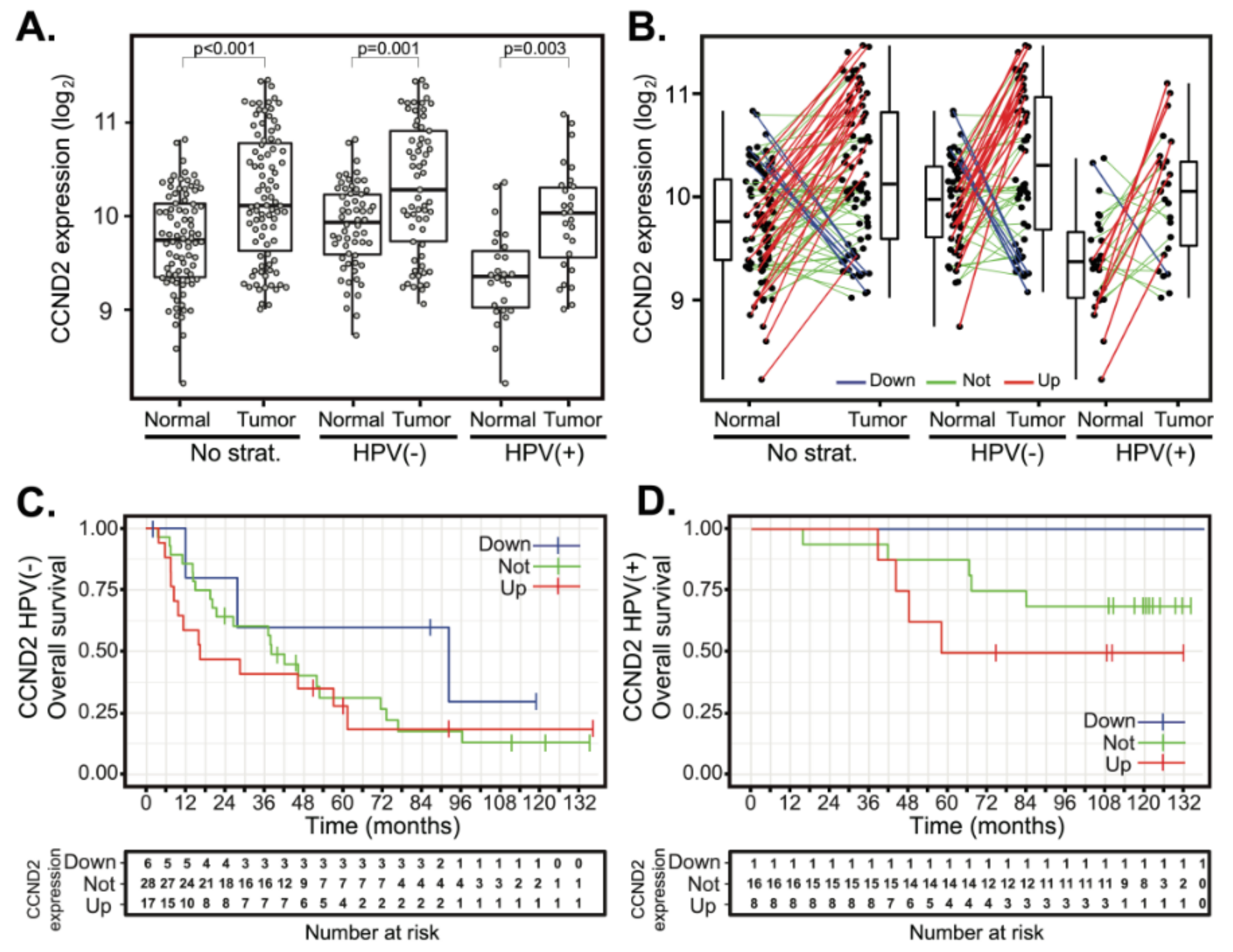
The expression levels of other D-type cyclins, CCND2 and CCND3, were also examined. In contrast to CCND1, the median expression of CCND2 was slightly elevated, yet it was statistically significant in both HPV(−) and HPV(+) tumors (Figure 3A). Comparative analysis of CCND2 gene expression between paired tumor and normal samples revealed upregulation of CCND2 expression in 26 cases (31.7%) (Figure 3B). In 49 cases (59.8%), there was no change in CCND2 expression, and downregulation of CCND2 was observed in seven cases (8.5%) only. Stratification according to HPV status revealed that both HPV(+) and HPV(−) tumors displayed significant upregulation of CCND2 (Figure 3B), while downregulation of CCND2 was observed only in a few HPV(−) tumors and a single HPV(+) tumor. Neither CCND2 upregulation nor downregulation affected the OS (Figure 3C,D and Table 2) and DFS (Supplementary Figure S1E,F and Table S2).
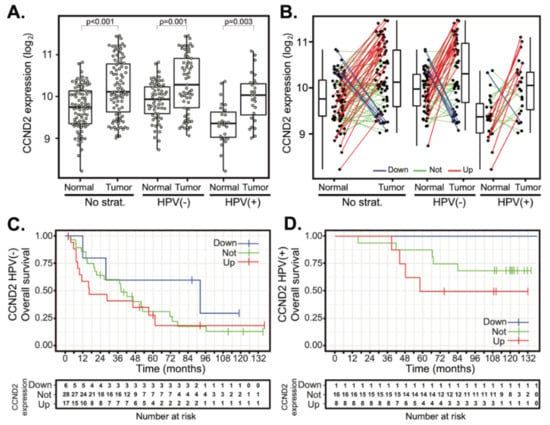
Figure 3.
CCND2 gene expression changes between tumor and normal tissues and patient survival in HPV(+) and HPV(−) groups. (A) CCND2 gene expression in a cohort of 94 patients (left panel) and stratified according to the HPV status (middle and right panels). The boxplot displays interquartile range with the whiskers indicating the greatest and smallest observations, excluding outliers. (B) CCND2 deregulation between paired tumor and normal tissues. Paired samples are connected by the lines color-coded according to the change of CCND2 expression (red—CCND2 upregulation, FC > 2; green—no deregulation, 0.5 < FC < 2 and blue—CCND2 downregulation, FC < 0.5). (C,D) Kaplan–Meier plots for OS stratified according to CCND2 gene deregulation in HPV(−) and HPV(+) groups. Up—group with CCND2 upregulated, not—group without CCND2 deregulation and down—CCND2 group with CCND2 downregulated. Tick marks in (C,D) indicate right censoring.
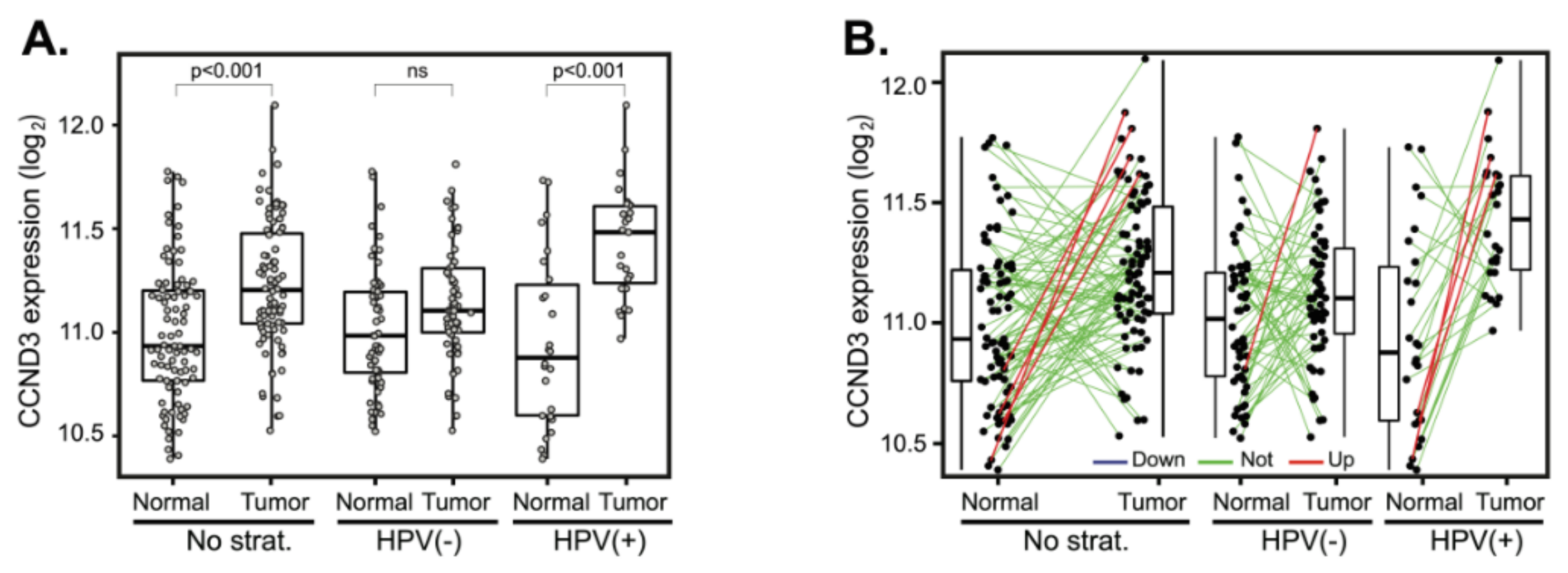
The analysis of CCND3 mean expressions revealed elevated CCND3 expression only in HPV(+) tumors (Figure 4A). Expression changes between paired tumor and normal samples also revealed the upregulation of CCND3 expression in four cases (6.6%), predominantly in HPV(−) tumors (Figure 4B). Downregulation of CCND3 was not observed. Due to the small number of patients with CCND3 expression changes, statistical analysis did not yield any significant results (Table 2 and Table S2).
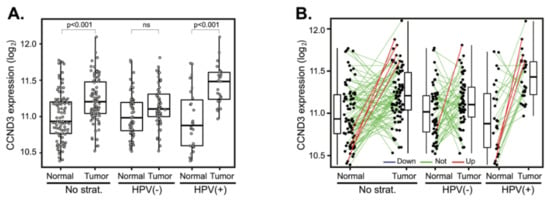
Figure 4.
CCND3 gene expression changes between tumor and normal tissues. (A) CCND3 gene expression in a cohort of 94 patients (left panel) and stratified according to the HPV status (middle and right panels). The boxplot displays interquartile range with the whiskers indicating the greatest and smallest observations, excluding outliers. (B) CCND3 deregulation between paired tumor and normal tissues. Paired samples are connected by the lines color-coded according to the change of CCND3 expression (red—CCND3 upregulation, FC > 2; green—no deregulation, 0.5 < FC < 2 and blue—CCND3 downregulation, FC < 0.5).
2.5. Cyclin D2 is Often Upregulated in Tumors with Downregulated Cyclin D1 and Vice Versa
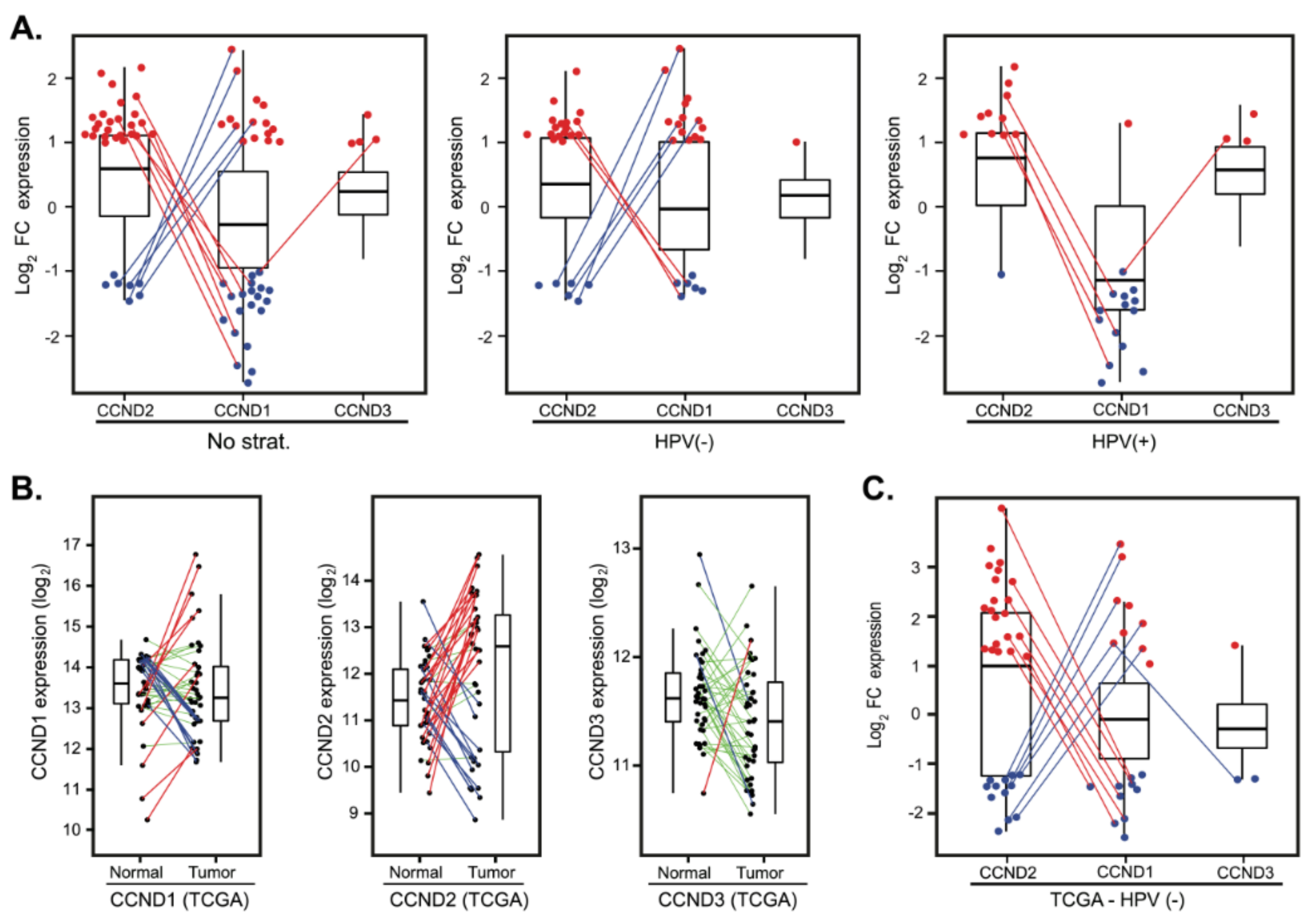
The finding that CCND1 was downregulated in more than a fourth of the cases (Figure 1C) and that these patients did not display a better prognosis (Table 2) suggests the hypothesis that additional cyclins could compensate for the CCND1 deficiency. We thus examined whether CCND1 mRNA downregulation is paralleled by upregulation of CCND2 and/or CCND3. Indeed, the comparison revealed that a substantial number of tumors with downregulated CCND1 upregulated CCND2 (7 out of 20; 35%) and CCND3 in one individual case (Figure 5A). Intriguingly, we also found that tumors that upregulated CCND1 often downregulated CCND2 expression (5 out of 15; 33.3%) (Figure 5A). This observation suggests that expression of CCND1 and CCND2 is coupled.
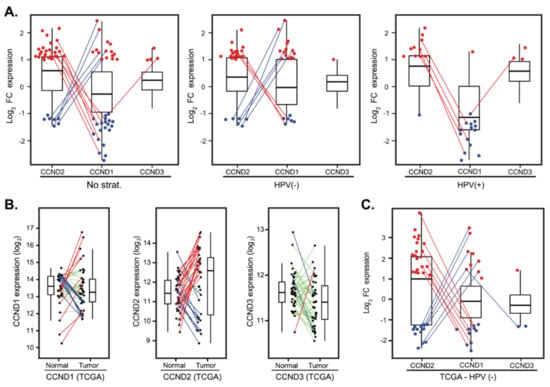
Figure 5.
Compensatory expression of CCND2 in tumors with deregulated CCND1. (A) D-type cyclins expression changes presented as log2 fold-change between tumor and normal tissues in a cohort of 82 patients (left panel) and stratified according to the HPV status (middle and right panels). For clarity, only tumors with FC > 2 (red dots) and FC < 0.5 (blue dots) are shown. Lines connect the same patients. (B) D-type cyclins deregulation between paired tumor and normal tissues in The Cancer Genome Atlas (TCGA) cohort of 39 HPV(−) patients. Paired samples are connected by the lines color-coded according to the change in D-type cyclin expression (red—upregulation, FC > 2; green—no deregulation, 0.5 < FC < 2 and blue—downregulation, FC < 0.5). (C) D-type cyclins expression changes presented as log2 fold-change between paired tumor and normal tissues in the TCGA cohort of 39 HPV(−) patients. For clarity, only tumors with FC > 2 (red dots) and FC < 0.5 (blue dots) are shown. Lines connect the same patients.
2.6. Validation in an Independent Cohort
To validate our findings in an independent cohort, we analyzed the expression profiles from a cohort of 490 patients from The Cancer Genome Atlas (TCGA) obtained from cBioPortal (see Materials and Methods for details). In this cohort, there were 42 paired tumor and normal mucosa samples with follow-up clinical data; however, only three of these pairs contained HPV(+) tumors. Hence, we focused on HPV(−) pairs. Analysis of CCND1, CCND2 and CCND3 expression recapitulated the findings in our dataset. CCND1 deregulation between tumor and normal tissues clustered patients in three groups (Figure 5B) with no correlation with OS and DFS (Table S3). Substantial fraction of patients had CCND2 in tumors upregulated (20 cases, 50.0%) or downregulated (12 cases, 30.0%) (Figure 5B). Importantly, we observed again that a significant fraction of the tumors with upregulated CCND1 had downregulated CCND2 (6 out of 9; 60.0%) or vice versa (7 out of 10, 70.0%) (Figure 5C).
2.7. Immunohistochemical Analysis of Tumor Samples
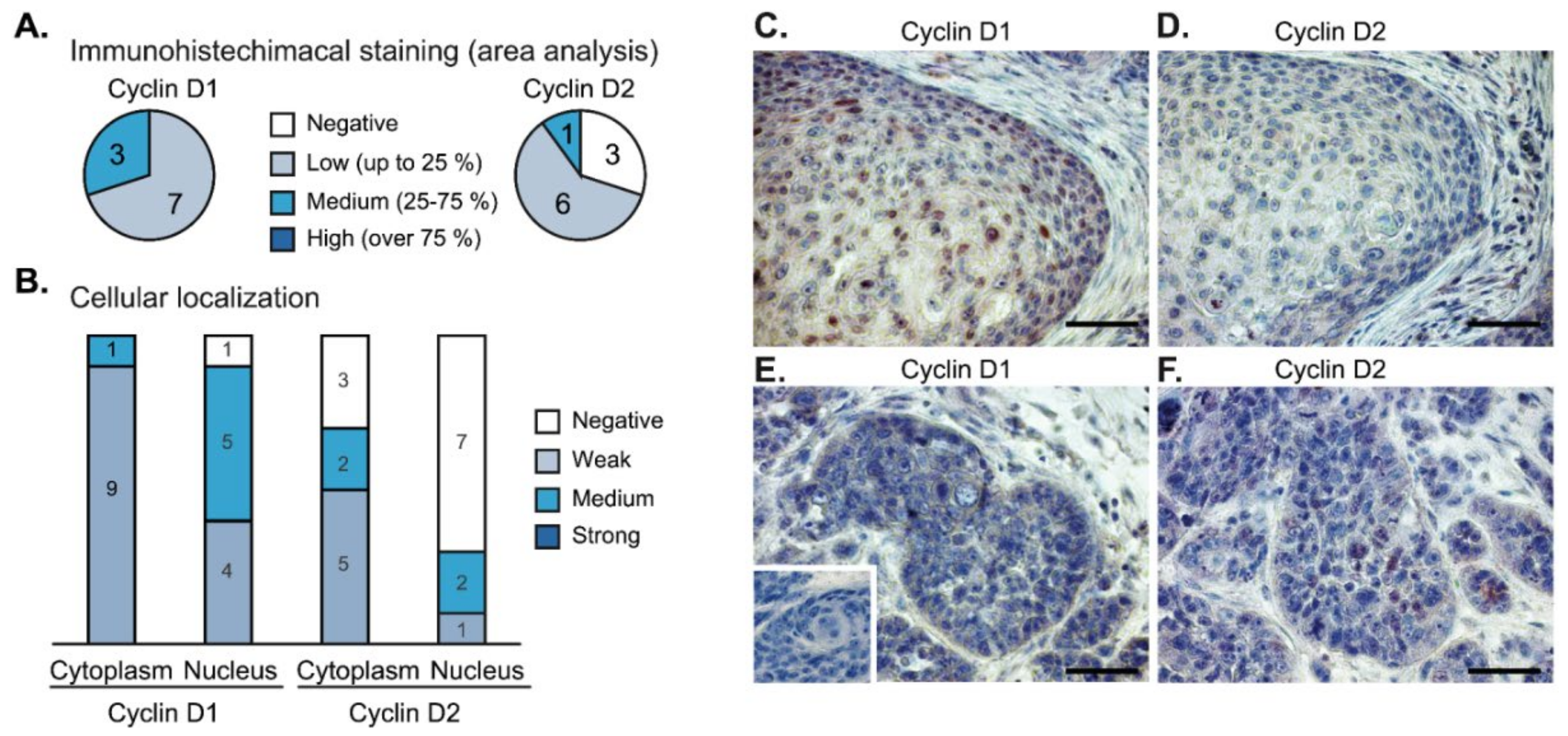
To evaluate the expression of cyclins at the protein level, we performed immunohistochemical analysis of 10 independent tumor samples (Figure 6). We observed cyclin D1 focal staining with variable intensity in the cytoplasm (10/10) and in the nuclei as well (9/10) (Figure 6A,B). The expression of cyclin D2 was weaker, and it was completely negative in three samples (Figure 6A); the nuclear signal of cyclin D2 was observed only in three cases (3/10) (Figure 6B). Frequently, we observed that the nuclear signal of cyclin D1 was accompanied by low or negative nuclear signals of cyclin D2 (Figure 6C,D). Vice versa, in the regions with high nuclear staining of cyclin D2, we observed that the level of cyclin D1 was low (Figure 6E,F).
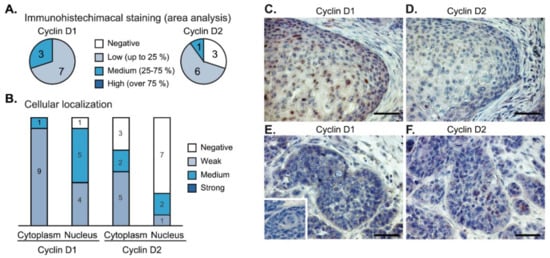
Figure 6.
Immunohistochemistry of cyclin D1 and cyclin D2 expression in head and neck squamous cell carcinomas (n = 10). (A) Low-to-medium intensity staining of cyclin D1 was detected in all tumor samples. The staining of cyclin D2 was negative in three cases. (B) Nuclear and cytoplasmic distribution of cyclin D1 and cyclin D2 staining in the tumor samples. (C,D) In cases with high nuclear intensity of cyclin D1 (panel C), the nuclear staining of cyclin D2 was weak or negative (panel D). (E,F) Vice versa, higher intensity of cyclin D2 (panel F) was observed in tumor buds with a lack of nuclear cyclin D1 (panel E). Inset in panel E shows a negative control. The bar is 0.1 mm.
3. Discussion
Amplification of the CCND1 gene encoding cyclin D1 is one of the most frequent genomic alterations in human cancers [37]. Altered CCND1 expression has been reported in many different cancers [18,19], including head and neck squamous cell carcinoma [13,38]. In this work, we have focused on the expression patterns of cyclin D1, as well as the other two members of the cyclin D family, in a new cohort of 94 HNSCC patients. This dataset contains expression data from 82 tumor samples and paired normal mucosa and, to our best knowledge, represents the largest collection of paired HNSCC samples. Analysis of expression changes between paired tumor and normal samples revealed that expression of the CCND1 gene is upregulated mainly in HPV(−) patients, and, unexpectedly, it may also be downregulated, predominantly in HPV(+) patients. Nonetheless, there was no statistically significant correlation between CCND1 deregulation and the disease outcome. Further analysis showed that CCND1 upregulation was frequently accompanied by CCND2 downregulation, and, conversely, its downregulation was found to be often accompanied by upregulation of CCND2 or, in one case, of CCND3. Based on these data, we speculate that upregulation of one D-type cyclin could be compensated by downregulation of another one. Such compensatory effects may explain previously observed poor correlations between CCND1 expression and patient prognosis [26,27,28].
The importance of D-type cyclins for tumorigenesis has been demonstrated in mouse models, where the presence of cyclin D proteins is required for both tumor initiation and maintenance, while they seem largely dispensable for normal development [39,40]. It is presumed that the increase in the cyclin D expression is an early oncogenic event that causes tumor formation and continuous uncontrolled proliferation of tumor cells [18,19,41]. Concordantly, with these studies, the analysis of almost 500 HNSCC samples showed amplification of chromosomal region 11q13 containing the CCND1 gene in 31% of cases [15], although the CCND1 amplification frequency has been reported to range from 17% to 50% [33,34]. The amplified 11q13 locus harbors several other genes that could promote tumorigenesis. Among them, ANO1 coding for anoctamin-1 and CTTN coding for cortactin are frequently coamplified with CCND1. These genes were shown to promote HNSCC progression and to be associated with poor prognosis [22,29,35,42]. In agreement with that, we found that LTO1, ANO1 and CTTN are coregulated in the HPV(−) tumors, and they could cooperate with CCND1 to affect the clinical outcome.
In addition to gene amplification, other mechanisms likely contribute to cyclin D1 protein upregulation, as its overexpression estimated by immunohistochemistry has been documented in up to 70% of HNSCC tumors [21,22]. In our dataset, we observed CCND1 mRNA upregulation in approximately 20% of cases, and, surprisingly, roughly 25% of tumors showed a decreased expression of the CCND1 gene when compared to normal mucosa. Since the repression of cyclin D1 promotes cell cycle exit, cellular quiescence and, in some cases, cell differentiation [43] and, in HNSCC tumor models, reduces cell growth and survival [44], these findings indicated that a CCND1 decrease may have a positive impact on patient outcome. However, our data did not show any statistically significant correlation between CCND1 expression and disease outcomes.
Why patient outcome does not correlate with the changes in CCND1 expression remains unknown. According to our data, we speculate that a functional significance of change in CCND1 expression could be alleviated by the compensatory change in the expression of another D-type cyclin. Compensation among D-type cyclins has been observed in mice, where cyclins D1, D2 and D3 are expressed redundantly in most tissues. Genetic ablation of individual D-type cyclin does not generally affect normal development; however, the simultaneous ablation of cyclins resulted in more severe developmental defects and embryonal lethality. These studies suggested that D-cyclins can compensate for the loss of another family member [45]. A compensatory effect was notably evident in mice engineered to express only one D-type cyclin. In embryos, the sole expressed cyclin became upregulated in most tissues which were otherwise negative for this type of cyclin [46]. Cyclin compensation was also observed in adult mice in the proliferating uterine epithelium, where the cyclin D1 absence can be compensated by cyclin D2 [47], and in B-lymphocytes, where D2 absence is compensated by upregulation of cyclin D3 [48]. All these observations point to a mechanism where perturbation of one cyclin D member may induce the compensatory expression of other family members and correspond to our findings, where one-third of tumors with downregulated cyclin D1 mRNA upregulated the expression of cyclin D2. We also observed the opposite trend, where in one-third of tumors with cyclin D1 upregulation, cyclin D2 was downregulated, further supporting the hypothesis that genetic compensation in response to the perturbation of cyclin D function is a common phenomenon in HNSCC. We have validated our results in an independent TCGA dataset and also by immunohistochemical staining.
The best prognostic factor for HNSCC remains to be HPV infection, as HPV(+) patients display better clinical outcomes. The worse prognosis of HPV(−) patients may be the consequence of different mutational burdens in HPV(+) and HPV(−) HNSCC tumors [38]. It may also reflect the fact that the CCND1 locus is rarely amplified in HPV(+) tumors [22,49], which is consistent with our findings that a positive correlation of expression of CCND1 and the genes in the 11q13 locus is observed almost exclusively in HPV(−) tumors. The genes coamplified with CCND1, such as LTO1/ORAOV1, ANO1 and CTTN, are associated with HNSCC metastasis and recurrence and may therefore account for the worse prognosis of HPV(−) patients [22,29,42,50,51]. Our observations that cyclin D1 expression is downregulated in half of HPV(+) tumors was surprising. Given that cyclin D1 is required for tumor formation induced by HPV proteins E6 and E7 [52], it indicated that cyclin D1 downregulation may also contribute to a better prognosis of HPV(+) patients. Indeed, previous studies have associated low cyclin D1 expression in HPV(+) tumors with improved survival rates when in combination with high p16INK4a, low pRb and low p53 [53,54]. However, we have not observed an improved outcome in HPV(+) patients who exhibit low expressions of cyclin D1 mRNA. In addition to the proposed compensatory effect of cyclin D2, it is also possible that the decrease in CCND1 expression in HPV(+) tumors is a consequence of low pressure on cyclin D expression and lower CDK4/6 activity in general, as the G1 transcriptional repressor complex pRb/E2F is already disrupted by the viral protein E7 [14,38]. It is of note that, in HPV-associated cervical tumors, D-type cyclins and CDK4/6 activity inhibition by p16INK4a promotes tumor cell survival [55]. It is possible to speculate that cyclin D1 downregulation contributes to the survival of HPV(+) HNSCC tumor cells, similarly to cervical cancer.
In conclusion, in our study with independent validation, we have analyzed the mRNA expression level changes between paired tumor and normal samples at the patient level. Availability of paired samples provided us with the opportunity to show a significant variation in D-type cyclin gene expression between individual patients and to further stratify the patients according to the changes in the expression of the D-type cyclins. Although our results do not support a role for a specific D-cyclin in patient prognosis, it is possible to speculate about a compensatory mechanism between D-type cyclin expressions. These results are in agreement with the current opinion that tumors can be addicted to cyclin D-dependent CDK4/6 kinase activity [56] and that CDK4/6–cyclin D complexes represent promising targets in cancer therapy [18,20,41]. In the clinical trials conducted in HNSCC patients with radioresistant recurrent tumors, a combination of cetuximab and palbociclib targeting the EGF receptor and CDK4/6, respectively, indeed show promising results [57,58]. Analysis of cyclin expression changes between patient tumors and normal tissues may lead to the employment of more effective personalized therapies that could enhance the efficiency of currently adopted cancer treatment regimens.
4. Materials and Methods
4.1. Sample Collection
Sample collection, detection of HPV and microarray analysis were performed as described in Valach et al. [59] and Szabo et al. [60]. In short, normal mucosa and tumor tissue specimens were collected from 94 patients suffering from HNSCC, after their informed consent in full agreement with the local ethical committee and the Declaration of Helsinki. Specimens were chemically protected from RNA degradation and stored at –85 °C. Detection of high-risk oncogenic HPV types (16, 19, 31, 33 and 45) in tumor samples was done by RT-qPCR of the E6 and E7 genes.
For immunohistochemical analysis, the tissues were fixed in 10% neutral buffered formalin and routinely processed for paraffin block preparation. Tissue sections (5-μm thick) were deparaffinized in xylene and ethanol baths with decreasing concentrations of ethanol (5 min each). Heat-induced epitope retrieval was performed at pH = 6.0 (citrate buffer). Sections were blocked in hydrogen peroxide blocking reagent and protein block (ab64218 and ab64226, respectively; both Abcam, Cambridge, UK). Primary antibodies (cyclin D1, clone DCS-6, mouse monoclonal; Dako, Glostrup, Denmark and cyclin D2 (D52F9) rabbit monoclonal #3741; Cell Signaling Technology, Danvers (MA), USA) were diluted 1:100, and sections were incubated at room temperature for 2 hours followed by incubation with HRP-tagged secondary antibody (Histofine simple stain MAX PO MULTI; Nichirei Biosciences, Tokyo, Japan) for 15 minutes. The reaction was developed for 10 minutes using Histofine simple stain AEC solution (Nichirei Biosciences, Tokyo, Japan) and counterstained with Gill´s hematoxylin (Sigma Aldrich, Prague, Czech Republic). For the negative control, the primary antibody was replaced by control nonimmune serum (X0910 and X0903; Dako, Glostrup, Denmark). Imaging was performed with a Leica DM2000 microscope using the LAS software package (Leica Microsystems GmbH, Wetzlar, Germany). The annotation of immunohistochemical results was performed using the method described in detail in Protein Atlas version 19.2 (https://www.proteinatlas.org/about/assays+annotation).
4.2. Transcription Profiling
Whole genome transcription profiling was performed on HumanWG-6 v3 Expression BeadChip microarrays (Illumina, San Diego, California, USA). Subsequent data analysis was done in R statistical environment [61]: raw data was processed using the limma package [62] of the Bioconductor Project [63]. Background corrected and quantile normalized data were corrected for batch effects using the ComBat function from the R package sva [64], and expression intensities of technical replicates were averaged at the probe level. Detected transcripts were annotated using the provided manifest file (HumanWG-6_V3_0_R2_11282955_A.bgx; Illumina, San Diego, CA, USA). Expression changes between the sample groups (tumor HPV(−), tumor HPV(+) and normal) were detected by the moderated t-test within the limma package.
4.3. Clinical Data
The following patient data was recorded: TNM stages, tumor grade and localization, presence of keratinization, extracapsular extensions, angiogenesis and perineural spread. Information regarding treatment was also recorded (surgery and adjuvant chemo-/radiotherapy), together with patient characteristics (smoking, alcohol usage, personal and family cancer anamnesis). Vital status was recorded by the clinician/investigator at time of last follow-up. For overall survival, vital status was alive or dead. For disease-free survival, four statuses were recorded: disease free, exitus of other reason, recidive and exitus of the disease. The latter two vital statuses were taken as events in the disease-free survival analysis. Association between the HPV(−) and HPV(+) groups and clinical variables was tested using either a Kruskal-Wallis or Fisher’s exact test.
4.4. Analysis of CCND1 Coamplification
To verify the co-amplification of the 11q13 region in HPV(−) tumors, we calculated log2 fold-changes of genes near CCND1 (one megabase upstream and downstream of the CCND1 start and end, respectively). The intensity of the probes targeting a particular gene was averaged, and genes with a mean probe log2-intensity lower than 5 were omitted. We used Gviz [65] and ComplexHeatmap [66] packages of the Bioconductor Project for visualization.
4.5. The Cancer Genome Atlas (TCGA) Validation Dataset
To provide an external validation, we used the head and neck squamous cell carcinoma TCGA PanCancer RNA-seq dataset of 490 HNSCC patients [15,67], provided through cBioPortal [68,69]. Read count matrices, clinical and follow-up data were downloaded and imported to the R statistical environment. DESeq2 [70] package was used for sequencing depth and variance-stabilizing normalization. DESeq2 was also used for statistical testing of the mean difference between sample groups (normal, tumor HPV(−) and tumor HPV(+)) using the Wald test.
4.6. Survival Analysis
We calculated fold-changes (FC) in expression of the D-type cyclins between tumor and normal mucosa samples for each patient and split the patients in three groups depending on FC: patients with cyclin upregulation (FC > 2), no deregulation (0.5 < FC < 2) and downregulation (FC < 0.5). These groups were used in survival analyses using the R packages survival [71] and survminer [72]. For each D-type cyclin, univariate Cox proportional hazard model, Kaplan-Meier estimator and its associated log-rank test were computed, using patients with no deregulation of cyclin as a reference.
4.7. Data Availability
The present dataset is available in MIAME-compliant form from the ArrayExpress database under accession E-MTAB-8588. The dataset used to validate our findings is available from The Cancer Genome Atlas via cBioPortal (https://www.cbioportal.org).
4.8. Ethics Approval and Consent to Participate
Ethical approval for patient recruitment, sample collection, clinical follow-up and data analysis based on the Declaration of Helsinki was granted by the Ethics Committee for Multi-Centric Clinical Trials of the University Hospital Motol and 2nd Faculty of Medicine, Charles University in Prague (Approval No EK-890/15).
5. Conclusions
In this study, we examined the expression of cyclin D1 and other D-type cyclins in a new cohort of HNSCC patients. The availability of both tumor sample and normal tissue sample from the same individual allowed us to analyze the relative changes in D-type cyclin expression rather than their absolute expression levels. The comparison of the D-type cyclin expression in tumor and healthy tissue from the same patients revealed an unexpected pattern of their expression. A subset of tumors increased the cyclin D1 expression that was specific, with one exception, to HPV-negative tumors. Unexpectedly, we also observed that the cyclin D1 expression was often downregulated in some tumors, with a higher frequency observed in HPV-positive patients. We have not found any direct effect of the changes in D-type cyclin expression on patient prognosis, even after stratification of the patients according to their HPV status. This observation could be a consequence of cyclin D2 upregulation, which frequently compensates cyclin D1 downregulation and vice versa. The compensatory expression among D-type cyclins was also observed in an independent dataset obtained from The Cancer Genome Atlas, further validating our data. We thus propose that absent correlation between the cyclin D1 expression and patient survival in both subtypes of HNSCCs may be a consequence of a compensatory mechanism where the effect of change in the cyclin D1 expression could be alleviated by the reciprocal change in the expression of cyclin D2. These findings highlight the importance of analyses of matched tissues from the same individual, as they can reveal molecular changes associated with the cancer development.
Supplementary Materials
The following are available online at https://www.mdpi.com/2072-6694/12/4/792/s1: Figure S1: Disease-free survival analysis of groups stratified according to HPV status, CCND1 and CCND2 expression and overall survival of tumors from the base of the tongue stratified according to the HPV status. Table S1: Summary of clinical data for present cohort. Table S2: Survival analyses in the presented dataset. Table S3: Survival analyses in the TCGA dataset. MS Word file supplementary_captions.docx: Captions for supplementary files.
Author Contributions
Conceptualization, J.N., H.S., V.P., L.L., K.S.J., J.P., M.K. and T.V.; Data Curation, J.N., H.S. and M.K.; Formal Analysis, J.N., H.S. and M.K.; Funding Acquisition, H.S., V.P., L.L., K.S.J., J.P., M.K. and T.V.; Investigation, V.B., M.C., M.H., J.Š., M.Š., R.K. and L.L.; Methodology, H.S., K.S.J., J.P., M.K. and T.V.; Resources, H.S., K.S.J., J.P., M.K. and T.V.; Software, J.N., H.S. and M.K.; Supervision, M.K. and T.V.; Visualization, J.N., M.K. and T.V.; Writing Original Draft Preparation, J.N., J.Š., J.G., M.K. and T.V. and Writing Review & Editing, J.N., J.Š., J.G., M.K. and T.V. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Ministry of Health of the Czech Republic under grant no. AZV 16-29032A, by the Czech Science Foundation under grant no. 18-11908S, by the Operational Programme Research, Development and Education under the project “Center for Tumor Ecology—Research of the Cancer Microenvironment Supporting Cancer Growth and Spread” (reg. No. CZ.02.1.01/0.0/0.0/16_019/0000785) and by the Research and Development for Innovations Operational Program under project no. CZ.1.05/2.1.00/19.0400 (co-financed by the European Regional Development Fund and the state budget of the Czech Republic).
Acknowledgments
Authors are thankful to the service laboratory at Institute of Molecular Genetics of the Czech Academy of Sciences and especially to Martina Krausová and Šárka Kocourková for excellent technical assistance. Some results shown here are in whole or part based upon data generated by the TCGA Research Network: https://www.cancer.gov/tcga. We are deeply grateful to all specimen donors and research groups that have made the data publicly available. The authors thank J.E. Manning and Šárka Takáčová for critical reading of the manuscript and English grammar check.
Conflicts of Interest
The authors declare no conflicts of interest. The funders had no role in the design of the study; in the collection, analyses or interpretation of data; in the writing of the manuscript or in the decision to publish the results.
References
- Ferlay, J.; Shin, H.R.; Bray, F.; Forman, D.; Mathers, C.; Parkin, D.M. Estimates of worldwide burden of cancer in 2008: GLOBOCAN 2008. Int. J. Cancer 2010, 127, 2893–2917. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Steliarova-Foucher, E.; Lortet-Tieulent, J.; Rosso, S.; Coebergh, J.W.W.; Comber, H.; Forman, D.; Bray, F. Cancer incidence and mortality patterns in Europe: Estimates for 40 countries in 2012. Eur. J. Cancer 2013, 49, 1374–1403. [Google Scholar] [CrossRef] [PubMed]
- Kalavrezos, N.; Bhandari, R. Current trends and future perspectives in the surgical management of oral cancer. Oral Oncol. 2010, 46, 429–432. [Google Scholar] [CrossRef] [PubMed]
- Fung, C.; Grandis, J.R. Emerging drugs to treat squamous cell carcinomas of the head and neck. Expert Opin. Emerg. Drugs 2010, 15, 355–373. [Google Scholar] [CrossRef]
- Hashibe, M.; Brennan, P.; Chuang, S.C.; Boccia, S.; Castellsague, X.; Chen, C.; Curado, M.P.; Dal Maso, L.; Daudt, A.W.; Fabianova, E.; et al. Interaction between Tobacco and Alcohol Use and the Risk of Head and Neck Cancer: Pooled Analysis in the International Head and Neck Cancer Epidemiology Consortium. Cancer Epidemiol. Biomark. Prev. 2009, 18, 541–550. [Google Scholar]
- Warnakulasuriya, S. Global epidemiology of oral and oropharyngeal cancer. Oral Oncol. 2009, 45, 309–316. [Google Scholar] [CrossRef]
- Gillison, M.L.; Koch, W.M.; Capone, R.B.; Spafford, M.; Westra, W.H.; Wu, L.; Zahurak, M.L.; Daniel, R.W.; Viglione, M.; Symer, D.E.; et al. Evidence for a Causal Association Between Human Papillomavirus and a Subset of Head and Neck Cancers. J. Natl. Cancer Inst. 2000, 92, 709–720. [Google Scholar]
- Tommasino, M. The human papillomavirus family and its role in carcinogenesis. Semin. Cancer Biol. 2014, 26, 13–21. [Google Scholar] [CrossRef]
- Kreimer, A.R.; Clifford, G.M.; Boyle, P.; Franceschi, S. Human papillomavirus types in head and neck squamous cell carcinomas worldwide: A systemic review. Cancer Epidemiol. Biomark. Prev. 2005, 14, 467–475. [Google Scholar] [CrossRef]
- Psyrri, A.; Rampias, T.; Vermorken, J.B. The current and future impact of human papillomavirus on treatment of squamous cell carcinoma of the head and neck. Ann. Oncol. 2014, 25, 2101–2115. [Google Scholar] [CrossRef]
- Dayyani, F.; Etzel, C.J.; Liu, M.; Ho, C.-H.; Lippman, S.M.; Tsao, A.S. Meta-analysis of the impact of human papillomavirus (HPV) on cancer risk and overall survival in head and neck squamous cell carcinomas (HNSCC). Head Neck Oncol. 2010, 2, 15. [Google Scholar] [CrossRef] [PubMed]
- Ragin, C.C.R.; Modugno, F.; Gollin, S.M. The epidemiology and risk factors of head and neck cancer: A focus on human papillomavirus. J. Dent. Res. 2007, 86, 104–114. [Google Scholar] [CrossRef] [PubMed]
- Leemans, C.R.; Braakhuis, B.J.M.; Brakenhoff, R.H. The molecular biology of head and neck cancer. Nat. Rev. Cancer 2011, 11, 9–22. [Google Scholar] [CrossRef]
- Leemans, C.R.; Snijders, P.J.F.; Brakenhoff, R.H. The molecular landscape of head and neck cancer. Nat. Rev. Cancer 2018, 18, 269–282. [Google Scholar] [CrossRef]
- Lawrence, M.S.; Sougnez, C.; Lichtenstein, L.; Cibulskis, K.; Lander, E.; Gabriel, S.B.; Getz, G.; Ally, A.; Balasundaram, M.; Birol, I.; et al. Comprehensive genomic characterization of head and neck squamous cell carcinomas. Nature 2015, 517, 576–582. [Google Scholar]
- Chen, W.S.; Bindra, R.S.; Mo, A.; Hayman, T.; Husain, Z.; Contessa, J.N.; Gaffney, S.G.; Townsend, J.P.; Yu, J.B. CDKN2A Copy Number Loss Is an Independent Prognostic Factor in HPV-Negative Head and Neck Squamous Cell Carcinoma. Front. Oncol. 2018, 8, 95. [Google Scholar] [CrossRef]
- Dok, R.; Nuyts, S. HPV Positive Head and Neck Cancers: Molecular Pathogenesis and Evolving Treatment Strategies. Cancers 2016, 8, 41. [Google Scholar] [CrossRef]
- Musgrove, E.A.; Caldon, C.E.; Barraclough, J.; Stone, A.; Sutherland, R.L. Cyclin D as a therapeutic target in cancer. Nat. Rev. Cancer 2011, 11, 558–572. [Google Scholar] [CrossRef]
- Deshpande, A.; Sicinski, P.; Hinds, P.W. Cyclins and cdks in development and cancer: A perspective. Oncogene 2005, 24, 2909–2915. [Google Scholar] [CrossRef]
- VanArsdale, T.; Boshoff, C.; Arndt, K.T.; Abraham, R.T. Molecular Pathways: Targeting the Cyclin D-CDK4/6 Axis for Cancer Treatment. Clin. Cancer Res. 2015, 21, 2905–2910. [Google Scholar] [CrossRef]
- Bova, R.J.; Quinn, D.I.; Nankervis, J.S.; Cole, I.E.; Sheridan, B.F.; Jensen, M.J.; Morgan, G.J.; Hughes, C.J.; Sutherland, R.L. Cyclin D1 and p16INK4A expression predict reduced survival in carcinoma of the anterior tongue. Clin. Cancer Res. 1999, 5, 2810–2819. [Google Scholar] [PubMed]
- Hermida-Prado, F.; Menéndez, S.; Albornoz-Afanasiev, P.; Granda-Diaz, R.; Álvarez-Teijeiro, S.; Villaronga, M.; Allonca, E.; Alonso-Durán, L.; León, X.; Alemany, L.; et al. Distinctive Expression and Amplification of Genes at 11q13 in Relation to HPV Status with Impact on Survival in Head and Neck Cancer Patients. J. Clin. Med. 2018, 7, 501. [Google Scholar] [CrossRef]
- Li, H.; Wawrose, J.S.; Gooding, W.E.; Garraway, L.A.; Lui, V.W.Y.; Peyser, N.D.; Grandis, J.R. Genomic analysis of head and neck squamous cell carcinoma cell lines and human tumors: A rational approach to preclinical model selection. Mol. Cancer Res. 2014, 12, 571–582. [Google Scholar] [CrossRef] [PubMed]
- Michalides, R.; Van Veelen, N.; Hart, A.; Loftus, B.; Wientjens, E.; Balm, A. Overexpression of Cyclin D1 Correlates with Recurrence in a Group of Forty-seven Operable Squamous Cell Carcinomas of the Head and Neck. Cancer Res. 1995, 55, 975–978. [Google Scholar] [PubMed]
- Mineta, H.; Miura, K.; Takebayashi, S.; Ueda, Y.; Misawa, K.; Harada, H.; Wennerberg, J.; Dictor, M. Cyclin D1 overexpression correlates with poor prognosis in patients with tongue squamous cell carcinoma. Oral Oncol. 2000, 36, 194–198. [Google Scholar] [CrossRef]
- Wong, R.J.; Keel, S.B.; Glynn, R.J.; Varvares, M.A. Histological pattern of mandibular invasion by oral squamous cell carcinoma. Laryngoscope 2000, 110, 65–72. [Google Scholar] [CrossRef]
- Vielba, R.; Bilbao, J.; Ispizua, A.; Zabalza, I.; Alfaro, J.; Rezola, R.; Moreno, E.; Elorriaga, J.; Alonso, I.; Baroja, A.; et al. p53 and cyclin D1 as prognostic factors in squamous cell carcinoma of the larynx. Laryngoscope 2003, 113, 167–172. [Google Scholar] [CrossRef]
- Thomas, G.R.; Nadiminti, H.; Regalado, J. Molecular predictors of clinical outcome in patients with head and neck squamous cell carcinoma. Int. J. Exp. Pathol. 2005, 86, 347–363. [Google Scholar] [CrossRef]
- Rodrigo, J.P.; García-Carracedo, D.; García, L.A.; Menéndez, S.T.; Allonca, E.; González, M.V.; Fresno, M.F.; Suárez, C.; García-Pedrero, J.M. Distinctive clinicopathological associations of amplification of the cortactin gene at 11q13 in head and neck squamous cell carcinomas. J. Pathol. 2009, 217, 516–523. [Google Scholar] [CrossRef]
- Mellin, H.; Friesland, S.; Lewensohn, R.; Dalianis, T.; Munck-Wikland, E. Human papilloma virus (HPV) DNA in tonsillar cancer: Clinical correlates, risk of relapse, and survival. Int. J. Cancer 2000, 89, 300–304. [Google Scholar] [CrossRef]
- Strome, S.E.; Savva, A.; Brissett, A.E.; Gostout, B.S.; Lewis, J.; Clayton, A.C.; McGovern, R.; Weaver, A.L.; Persing, D.; Kasperbauer, J.L. Squamous cell carcinoma of the tonsils: A molecular analysis of HPV associations. Clin. Cancer Res. 2002, 8, 1093–1100. [Google Scholar]
- Chaturvedi, A.K.; Engels, E.A.; Anderson, W.F.; Gillison, M.L. Incidence trends for human papillomavirus-related and -unrelated oral squamous cell carcinomas in the United States. J. Clin. Oncol. 2008, 26, 612–619. [Google Scholar] [CrossRef] [PubMed]
- Schwaederlé, M.; Daniels, G.A.; Piccioni, D.E.; Fanta, P.T.; Schwab, R.B.; Shimabukuro, K.A.; Parker, B.A.; Kurzrock, R. Cyclin alterations in diverse cancers: Outcome and co-amplification network. Oncotarget 2015, 6, 3033–3042. [Google Scholar] [CrossRef] [PubMed]
- Vossen, D.; Verhagen, C.; Van der Heijden, M.; Essers, P.; Bartelink, H.; Verheij, M.; Wessels, L.; Van den Brekel, M.; Vens, C. Genetic Factors Associated with a Poor Outcome in Head and Neck Cancer Patients Receiving Definitive Chemoradiotherapy. Cancers 2019, 11, 445. [Google Scholar] [CrossRef]
- Britschgi, A.; Bill, A.; Brinkhaus, H.; Rothwell, C.; Clay, I.; Duss, S.; Rebhan, M.; Raman, P.; Guy, C.T.; Wetzel, K.; et al. Calcium-activated chloride channel ANO1 promotes breast cancer progression by activating EGFR and CAMK signaling. Proc. Natl. Acad. Sci. USA 2013, 110, E1026–E1034. [Google Scholar] [CrossRef]
- Janssen, J.W.G.; Imoto, I.; Inoue, J.; Shimada, Y.; Ueda, M.; Imamura, M.; Bartram, C.R.; Inazawa, J. MYEOV, a gene at 11q13, is coamplified with CCND1, but epigenetically inactivated in a subset of esophageal squamous cell carcinomas. J. Hum. Genet. 2002, 47, 460–464. [Google Scholar] [CrossRef]
- Beroukhim, R.; Mermel, C.H.; Porter, D.; Wei, G.; Raychaudhuri, S.; Donovan, J.; Barretina, J.; Boehm, J.S.; Dobson, J.; Urashima, M.; et al. The landscape of somatic copy-number alteration across human cancers. Nature 2010, 463, 899–905. [Google Scholar] [CrossRef]
- Faraji, F.; Schubert, A.D.; Kagohara, L.T.; Tan, M.; Xu, Y.; Zaidi, M.; Fortin, J.-P.; Fakhry, C.; Izumchenko, E.; Gaykalova, D.A.; et al. The Genome-Wide Molecular Landscape of HPV-Driven and HPV-Negative Head and Neck Squamous Cell Carcinoma. In Molecular Determinants of Head and Neck Cancer; Humana Press: Totowa, NJ, USA, 2018; pp. 293–325. [Google Scholar] [CrossRef]
- Yu, Q.; Geng, Y.; Sicinski, P. Specific protection against breast cancers by cyclin D1 ablation. Nature 2001, 411, 1017–1021. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.J.; Li, X.; Hydbring, P.; Sanda, T.; Stefano, J.; Christie, A.L.; Signoretti, S.; Look, A.T.; Kung, A.L.; Von Boehmer, H.; et al. The requirement for cyclin D function in tumor maintenance. Cancer Cell 2012, 22, 438–451. [Google Scholar] [CrossRef] [PubMed]
- Otto, T.; Sicinski, P. Cell cycle proteins as promising targets in cancer therapy. Nat. Rev. Cancer 2017, 17, 93–115. [Google Scholar] [CrossRef] [PubMed]
- Sugahara, K.; Michikawa, Y.; Ishikawa, K.; Shoji, Y.; Iwakawa, M.; Shibahara, T.; Imai, T. Combination effects of distinct cores in 11q13 amplification region on cervical lymph node metastasis of oral squamous cell carcinoma. Int. J. Oncol. 2011, 761–769. [Google Scholar]
- Klein, E.A.; Assoian, R.K. Transcriptional regulation of the cyclin D1 gene at a glance. J. Cell Sci. 2008, 121, 3853–3857. [Google Scholar] [CrossRef] [PubMed]
- Sauter, E.R.; Nesbit, M.; Litwin, S.; Klein-Szanto, A.J.; Cheffetz, S.; Herlyn, M. Antisense cyclin D1 induces apoptosis and tumor shrinkage in human squamous carcinomas. Cancer Res. 1999, 59, 4876–4881. [Google Scholar]
- Sherr, C.J.; Roberts, J.M. Living with or without cyclins and cyclin-dependent kinases. Genes Dev. 2004, 18, 2699–2711. [Google Scholar] [CrossRef]
- Ciemerych, M.A.; Kenney, A.M.; Sicinska, E.; Kalaszczynska, I.; Bronson, R.T.; Rowitch, D.H.; Gardner, H.; Sicinski, P. Development of mice expressing a single D-type cyclin. Genes Dev. 2002, 16, 3277–3289. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Pollard, J.W. Cyclin D2 compensates for the loss of cyclin D1 in estrogen-induced mouse uterine epithelial cell proliferation. Mol. Endocrinol. 2003, 17, 1368–1381. [Google Scholar] [CrossRef] [PubMed]
- Lam, E.W.-F.; Glassford, J.; Banerji, L.; Thomas, N.S.B.; Sicinski, P.; Klaus, G.G.B. Cyclin D3 Compensates for Loss of Cyclin D2 in Mouse B-lymphocytes Activated via the Antigen Receptor and CD40. J. Biol. Chem. 2000, 275, 3479–3484. [Google Scholar] [CrossRef] [PubMed]
- Van Kempen, P.M.W.; Noorlag, R.; Braunius, W.W.; Moelans, C.B.; Rifi, W.; Savola, S.; Koole, R.; Grolman, W.; Van Es, R.J.J.; Willems, S.M. Clinical relevance of copy number profiling in oral and oropharyngeal squamous cell carcinoma. Cancer Med. 2015, 4, 1525–1535. [Google Scholar] [CrossRef]
- Clark, E.S.; Brown, B.; Whigham, A.S.; Kochaishvili, A.; Yarbrough, W.G.; Weaver, A.M. Aggressiveness of HNSCC tumors depends on expression levels of cortactin, a gene in the 11q13 amplicon. Oncogene 2009, 28, 431–444. [Google Scholar] [CrossRef]
- Togashi, Y.; Arao, T.; Kato, H.; Matsumoto, K.; Terashima, M.; Hayashi, H.; De Velasco, M.A.; Fujita, Y.; Kimura, H.; Yasuda, T.; et al. Frequent amplification of oraov1 gene in esophageal squamous cell cancer promotes an aggressive phenotype via proline metabolism and ros production. Oncotarget 2014, 5, 2962–2973. [Google Scholar] [CrossRef]
- Al Moustafa, A.-E.; Foulkes, W.D.; Wong, A.; Jallal, H.; Batist, G.; Yu, Q.; Herlyn, M.; Sicinski, P.; Alaoui-Jamali, M.A. Cyclin D1 is essential for neoplastic transformation induced by both E6/E7 and E6/E7/ErbB-2 cooperation in normal cells. Oncogene 2004, 23, 5252–5256. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Holzinger, D.; Flechtenmacher, C.; Henfling, N.; Kaden, I.; Grabe, N.; Lahrmann, B.; Schmitt, M.; Hess, J.; Pawlita, M.; Bosch, F.X. Identification of oropharyngeal squamous cell carcinomas with active HPV16 involvement by immunohistochemical analysis of the retinoblastoma protein pathway. Int. J. Cancer 2013, 133, 1389–1399. [Google Scholar] [CrossRef] [PubMed]
- Plath, M.; Broglie, M.A.; Förbs, D.; Stoeckli, S.J.; Jochum, W. Prognostic significance of cell cycle-associated proteins p16, pRB, cyclin D1 and p53 in resected oropharyngeal carcinoma. J. Otolaryngol. Head Neck Surg. 2018, 47, 1–9. [Google Scholar] [CrossRef]
- McLaughlin-Drubin, M.E.; Park, D.; Munger, K. Tumor suppressor p16INK4A is necessary for survival of cervical carcinoma cell lines. Proc. Natl. Acad. Sci. USA 2013, 110, 16175–16180. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.J.; Anders, L. Signaling through cyclin D-dependent kinases. Oncogene 2014, 33, 1890–1903. [Google Scholar] [CrossRef]
- Michel, L.; Ley, J.; Wildes, T.M.; Schaffer, A.; Robinson, A.; Chun, S.-E.; Lee, W.; Lewis, J.; Trinkaus, K.; Adkins, D. Phase I trial of palbociclib, a selective cyclin dependent kinase 4/6 inhibitor, in combination with cetuximab in patients with recurrent/metastatic head and neck squamous cell carcinoma. Oral Oncol. 2016, 58, 41–48. [Google Scholar] [CrossRef]
- Adkins, D.; Ley, J.; Neupane, P.; Worden, F.; Sacco, A.G.; Palka, K.; Grilley-Olson, J.E.; Maggiore, R.; Salama, N.N.; Trinkaus, K.; et al. Palbociclib and cetuximab in platinum-resistant and in cetuximab-resistant human papillomavirus-unrelated head and neck cancer: A multicentre, multigroup, phase 2 trial. Lancet. Oncol. 2019, 20, 1295–1305. [Google Scholar] [CrossRef]
- Valach, J.; Fík, Z.; Strnad, H.; Chovanec, M.; Plzák, J.; Čada, Z.; Szabo, P.; Šáchová, J.; Hroudová, M.; Urbanová, M.; et al. Smooth muscle actin-expressing stromal fibroblasts in head and neck squamous cell carcinoma: Increased expression of galectin-1 and induction of poor prognosis factors. Int. J. Cancer 2012, 131, 2499–2508. [Google Scholar] [CrossRef]
- Szabó, P.; Kolář, M.; Dvořánková, B.; Lacina, L.; Štork, J.; Vlček, Č.; Strnad, H.; Tvrdek, M.; Smetana, K. Mouse 3T3 fibroblasts under the influence of fibroblasts isolated from stroma of human basal cell carcinoma acquire properties of multipotent stem cells. Biol. Cell 2011, 103, 233–248. [Google Scholar] [CrossRef]
- R Core Team R: A Language and Environment for Statistical Computing. Available online: http://www.r-project.org/ (accessed on 22 March 2020).
- Ritchie, M.E.; Phipson, B.; Wu, D.; Hu, Y.; Law, C.W.; Shi, W.; Smyth, G.K. Limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 2015, 43, e47. [Google Scholar] [CrossRef]
- Huber, W.; Carey, V.J.; Gentleman, R.; Anders, S.; Carlson, M.; Carvalho, B.S.; Bravo, H.C.; Davis, S.; Gatto, L. Orchestrating high-throughput genomic analysis with Bioconductor. Nat. Methods 2015, 12, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Leek, J.T.; Johnson, W.E.; Parker, H.S.; Fertig, E.J.; Jaffe, A.E.; Storey, J.D.; Zhang, Y.; Torres, L.C. sva: Surrogate Variable Analysis. R package version 3.34.0. Available online: http://bioconductor.org/packages/release/bioc/html/sva.html (accessed on 22 March 2020).
- Hahne, F.; Ivanek, R. Visualizing Genomic Data Using Gviz and Bioconductor. In Statistical Genomics; Mathé, E., Davis, S., Eds.; Humana Press: New York, NY, USA, 2016; Volume 1418, pp. 391–416. ISBN 9780849331664. [Google Scholar]
- Gu, Z.; Eils, R.; Schlesner, M. Complex heatmaps reveal patterns and correlations in multidimensional genomic data. Bioinformatics 2016, 32, 2847–2849. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Lichtenberg, T.; Hoadley, K.A.; Poisson, L.M.; Lazar, A.J.; Cherniack, A.D.; Kovatich, A.J.; Benz, C.C.; Levine, D.A.; Lee, A.V.; et al. An Integrated TCGA Pan-Cancer Clinical Data Resource to Drive High-Quality Survival Outcome Analytics. Cell 2018, 173, 400–416. [Google Scholar] [CrossRef]
- Cerami, E.; Gao, J.; Dogrusoz, U.; Gross, B.E.; Sumer, S.O.; Aksoy, B.A.; Jacobsen, A.; Byrne, C.J.; Heuer, M.L.; Larsson, E.; et al. The cBio Cancer Genomics Portal: An Open Platform for Exploring Multidimensional Cancer Genomics Data. Cancer Discov. 2012, 2, 401–404. [Google Scholar] [CrossRef]
- Gao, J.; Aksoy, B.A.; Dogrusoz, U.; Dresdner, G.; Gross, B.; Sumer, S.O.; Sun, Y.; Jacobsen, A.; Sinha, R.; Larsson, E.; et al. Integrative Analysis of Complex Cancer Genomics and Clinical Profiles Using the cBioPortal. Sci. Signal 2014, 6, 1–34. [Google Scholar] [CrossRef]
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 1–21. [Google Scholar] [CrossRef]
- Therneau, T. A Package for Survival Analysis in S. version 2.38. Available online: https://github.com/therneau/survival (accessed on 22 March 2020).
- Kassambara, A.; Kosinski, M.; Biecek, P.; Scheipl, F. Survminer: Drawing Survival Curves using “ggplot2.”. Available online: https://rpkgs.datanovia.com/survminer/index.html (accessed on 22 March 2020).
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).