Effect of Transgene Location, Transcriptional Control Elements and Transgene Features in Armed Oncolytic Adenoviruses
Abstract
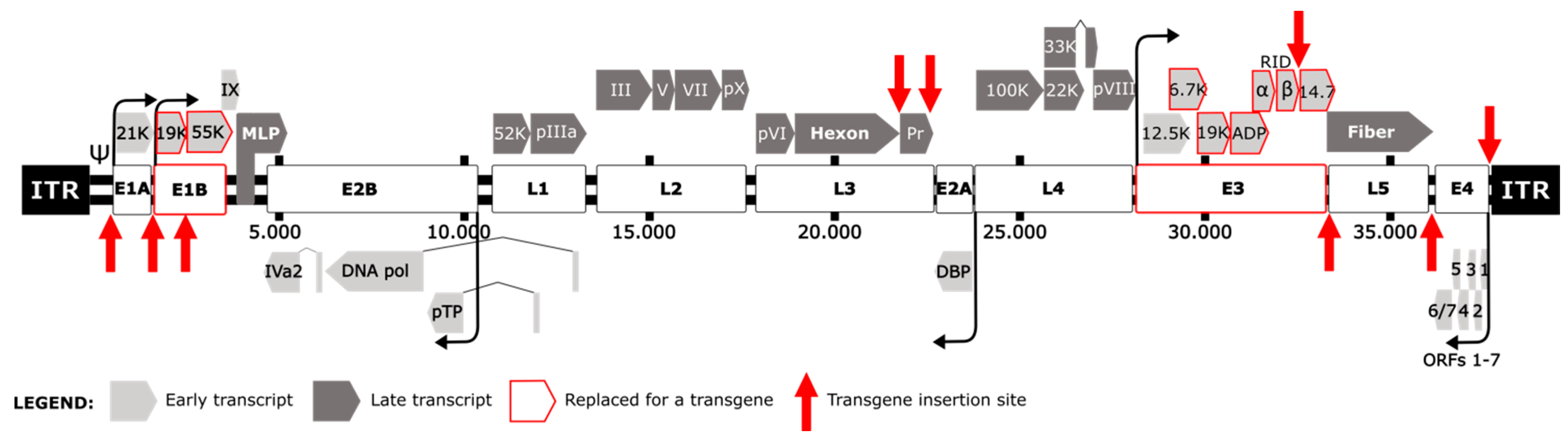
1. Introduction
2. Transgene Location Affects Expression Levels and Fitness of Armed OAds
3. Genetic Elements that Control Transgene Expression in the Context of Armed OAds
3.1. Exogenous Promoters
3.2. Internal Ribosome Entry Sites (IRES) and 2A Sequences
3.3. Splice Acceptors
4. Transgene-Related Parameters that Influence the Outcome of Armed OAds
4.1. Virocentric Transgenes
4.2. Immunocentric Transgenes
5. Conclusions
Funding
Conflicts of Interest
References
- Huebner, R.J.; Bell, J.A.; Rowe, W.P.; Ward, T.G.; Suskind, R.G.; Hartley, J.W.; Paffenbarger, R.S. Studies of adenoidal-pharyngeal-conjunctival vaccines in volunteers. J. Am. Med. Assoc. 1955, 159, 986–989. [Google Scholar] [CrossRef] [PubMed]
- Bischoff, J.R.; Kirn, D.H.; Williams, A.; Heise, C.; Horn, S.; Muna, M.; Ng, L.; Nye, J.A.; Sampson-Johannes, A.; Fattaey, A.; et al. An adenovirus mutant that replicates selectively in p53-deficient human tumor cells. Science 1996, 274, 373–376. [Google Scholar] [CrossRef]
- Liang, M. Oncorine, the World First Oncolytic Virus Medicine and its Update in China. Curr. Cancer Drug Targets 2018, 18, 171–176. [Google Scholar] [CrossRef]
- Larson, C.; Oronsky, B.; Scicinski, J.; Fanger, G.R.; Stirn, M.; Oronsky, A.; Reid, T.R. Going viral: A review of replication-selective oncolytic adenoviruses. Oncotarget 2015, 6, 19976–19989. [Google Scholar] [CrossRef] [PubMed]
- Niemann, J.; Kühnel, F. Oncolytic viruses: Adenoviruses. Virus Genes 2017, 53, 700–706. [Google Scholar] [CrossRef] [PubMed]
- Stepanenko, A.A.; Chekhonin, V.P. A compendium of adenovirus genetic modifications for enhanced replication, oncolysis, and tumor immunosurveillance in cancer therapy. Gene 2018, 679, 11–18. [Google Scholar] [CrossRef] [PubMed]
- García, M.; Moreno, R.; Gil-Martin, M.; Cascallò, M.; de Olza, M.O.; Cuadra, C.; Piulats, J.M.; Navarro, V.; Domenech, M.; Alemany, R.; et al. A Phase 1 Trial of Oncolytic Adenovirus ICOVIR-5 Administered Intravenously to Cutaneous and Uveal Melanoma Patients. Human Gene Ther. 2018, 30, 352–364. [Google Scholar] [CrossRef]
- DeWeese, T.L.; van der Poel, H.; Li, S.; Mikhak, B.; Drew, R.; Goemann, M.; Hamper, U.; DeJong, R.; Detorie, N.; Rodriguez, R.; et al. A phase I trial of CV706, a replication-competent, PSA selective oncolytic adenovirus, for the treatment of locally recurrent prostate cancer following radiation therapy. Cancer Res. 2001, 61, 7464–7472. [Google Scholar]
- Chen, Y.; DeWeese, T.; Dilley, J.; Zhang, Y.; Li, Y.; Ramesh, N.; Lee, J.; Pennathur-Das, R.; Radzyminski, J.; Wypych, J.; et al. CV706, a prostate cancer-specific adenovirus variant, in combination with radiotherapy produces synergistic antitumor efficacy without increasing toxicity. Cancer Res. 2001, 61, 5453–5460. [Google Scholar]
- Lang, F.F.; Conrad, C.; Gomez-Manzano, C.; Yung, W.K.A.; Sawaya, R.; Weinberg, J.S.; Prabhu, S.S.; Rao, G.; Fuller, G.N.; Aldape, K.D.; et al. Phase I Study of DNX-2401 (Delta-24-RGD) Oncolytic Adenovirus: Replication and Immunotherapeutic Effects in Recurrent Malignant Glioma. J. Clin. Oncol. 2018, 36, 1419–1427. [Google Scholar] [CrossRef]
- Small, E.J.; Carducci, M.A.; Burke, J.M.; Rodriguez, R.; Fong, L.; van Ummersen, L.; Yu, D.C.; Aimi, J.; Ando, D.; Working, P.; et al. A phase I trial of intravenous CG7870, a replication-selective, prostate-specific antigen-targeted oncolytic adenovirus, for the treatment of hormone-refractory, metastatic prostate cancer. Mol. Ther. 2006, 14, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Hermiston, T.W.; Kuhn, I. Armed therapeutic viruses: Strategies and challenges to arming oncolytic viruses with therapeutic genes. Cancer Gene Ther. 2002, 9, 1022–1035. [Google Scholar] [CrossRef]
- Chroboczek, J.; Bieber, F.; Jacrot, B. The sequence of the genome of adenovirus type 5 and its comparison with the genome of adenovirus type 2. Virology 1992, 186, 280–285. [Google Scholar] [CrossRef]
- Larsson, S.; Svensson, C.; Akusjärvi, G. Control of adenovirus major late gene expression at multiple levels. J. Mol. Biol. 1992, 225, 287–298. [Google Scholar] [CrossRef]
- Akusjarvi, G. Temporal regulation of adenovirus major late alternative RNA splicing. Front. Biosci. 2008, 5006. [Google Scholar] [CrossRef] [PubMed]
- Berkner, K.L.; Sharp, P.A. Generation of adenovirus by transfection of plasmids. Nucl. Acids Res. 1983, 11, 6003–6020. [Google Scholar] [CrossRef]
- Chanda, P.K.; Natuk, R.J.; Mason, B.B.; Brat, B.M.; Greenberg, L.; Dheer, S.K.; Molnar-kimber, K.L.; Mizutani, S.; Lubeck, M.D.; Davis, A.R.; et al. High level expression of the envelope glycoproteins of the human immunodeficiency virus type I in presence of rev gene using helper-independent adenovirus type 7 recombinants. Virology 1990, 175, 535–547. [Google Scholar] [CrossRef]
- Haj-Ahmad, Y.; Graham, F.L. Development of a helper-independent human adenovirus vector and its use in the transfer of the herpes simplex virus thymidine kinase gene. J. Virol. 1986, 57, 267–274. [Google Scholar] [CrossRef]
- Saito, I.; Oya, Y.; Yamamoto, K.; Yuasa, T.; Shimojo, H. Construction of nondefective adenovirus type 5 bearing a 2.8-kilobase hepatitis B virus DNA near the right end of its genome. J. Virol. 1985, 54, 711–719. [Google Scholar] [CrossRef]
- Kelly, T.J.; Lewis, A.M. Use of Nondefective Adenovirus-Simian Virus 40 Hybrids for Mapping the Simian Virus 40 Genome. J. Virol. 1973, 12, 643–652. [Google Scholar] [CrossRef]
- Flint, S.J.; Wewerka-Lutz, Y.; Levine, A.S.; Sambrook, J.; Sharp, P.A., II. RNA Sequences Complementary to Simian Virus 40 and Adenovirus 2 DNA in Ad2+NDr- and Ad2+ND3-Infected Cells. J. Virol. 1975, 16, 12. [Google Scholar] [CrossRef]
- Bett, A.J.; Prevec, L.; Graham, F.L. Packaging capacity and stability of human adenovirus type 5 vectors. J. Virol. 1993, 67, 5911–5921. [Google Scholar] [CrossRef] [PubMed]
- Freytag, S.O.; Rogulski, K.R.; Paielli, D.L.; Gilbert, J.D.; Kim, J.H. A Novel Three-Pronged Approach to Kill Cancer Cells Selectively: Concomitant Viral, Double Suicide Gene, and Radiotherapy. Hum. Gene Ther. 1998, 9, 1323–1333. [Google Scholar] [CrossRef] [PubMed]
- Rogulski, K.R.; Wing, M.S.; Paielli, D.L.; Gilbert, J.D.; Kim, J.H.; Freytag, S.O. Double Suicide Gene Therapy Augments the Antitumor Activity of a Replication-Competent Lytic Adenovirus through Enhanced Cytotoxicity and Radiosensitization. Hum. Gene Ther. 2000, 11, 67–76. [Google Scholar] [CrossRef]
- Wildner, O.; Blaese, R.M.; Morris, J.C. Therapy of Colon Cancer with Oncolytic Adenovirus Is Enhanced by the Addition of Herpes Simplex Virus-thymidine kinase. Cancer Res. 1999, 59, 410–413. [Google Scholar]
- Hawkins, L.; Johnson, L.; Bauzon, M.; Nye, J.; Castro, D.; Kitzes, G.; Young, M.; Holt, J.; Trown, P.; Hermiston, T. Gene delivery from the E3 region of replicating human adenovirus: Evaluation of the 6.7 K/gp19 K region. Gene Ther. 2001, 8, 1123–1131. [Google Scholar] [CrossRef]
- Hawkins, L.; Hermiston, T. Gene delivery from the E3 region of replicating human adenovirus: Evaluation of the ADP region. Gene Ther. 2001, 8, 1132–1141. [Google Scholar] [CrossRef][Green Version]
- Hawkins, L.; Hermiston, T. Gene delivery from the E3 region of replicating human adenovirus: Evaluation of the E3B region. Gene Ther. 2001, 8, 1142–1148. [Google Scholar] [CrossRef]
- Sauthoff, H.; Heitner, S.; Rom, W.N.; Hay, J.G. Deletion of the Adenoviral E1b-19kD Gene Enhances Tumor Cell Killing of a Replicating Adenoviral Vector. Hum. Gene Ther. 2000, 11, 379–388. [Google Scholar] [CrossRef]
- Harrison, D.; Sauthoff, H.; Heitner, S.; Jagirdar, J.; Rom, W.N.; Hay, J.G. Wild-Type Adenovirus Decreases Tumor Xenograft Growth, but Despite Viral Persistence Complete Tumor Responses Are Rarely Achieved—Deletion of the Viral E1b-19-kD Gene Increases the Viral Oncolytic Effect. Hum. Gene Ther. 2001, 12, 1323–1332. [Google Scholar] [CrossRef]
- Rohmer, S.; Quirin, C.; Hesse, A.; Sandmann, S.; Bayer, W.; Herold-Mende, C.; Haviv, Y.S.; Wildner, O.; Enk, A.H.; Nettelbeck, D.M. Transgene expression by oncolytic adenoviruses is modulated by E1B19K deletion in a cell type-dependent manner. Virology 2009, 395, 243–254. [Google Scholar] [CrossRef] [PubMed]
- Yan, W.; Kitzes, G.; Dormishian, F.; Hawkins, L.; Sampson-Johannes, A.; Watanabe, J.; Holt, J.; Lee, V.; Dubensky, T.; Fattaey, A.; et al. Developing Novel Oncolytic Adenoviruses through Bioselection. J. Virol. 2003, 77, 2640–2650. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, T.; Vijayalingam, S.; Chinnadurai, G. Genetic Identification of Adenovirus Type 5 Genes That Influence Viral Spread. J. Virol. 2006, 80, 2000–2012. [Google Scholar] [CrossRef] [PubMed]
- Gros, A.; Martínez-Quintanilla, J.; Puig, C.; Guedan, S.; Molleví, D.G.; Alemany, R.; Cascallo, M. Bioselection of a Gain of Function Mutation that Enhances Adenovirus 5 Release and Improves Its Antitumoral Potency. Cancer Res. 2008, 68, 8928–8937. [Google Scholar] [CrossRef]
- Bauzon, M.; Castro, D.; Karr, M.; Hawkins, L.K.; Hermiston, T.W. Multigene expression from a replicating adenovirus using native viral promoters. Mol. Ther. 2003, 7, 526–534. [Google Scholar] [CrossRef]
- Kretschmer, P.J.; Jin, F.; Chartier, C.; Hermiston, T.W. Development of a transposon-based approach for identifying novel transgene insertion sites within the replicating adenovirus. Mol. Ther. 2005, 12, 118–127. [Google Scholar] [CrossRef]
- Jin, F.; Kretschmer, P.J.; Hermiston, T.W. Identification of novel insertion sites in the Ad5 genome that utilize the Ad splicing machinery for therapeutic gene expression. Mol. Ther. 2005, 12, 1052–1063. [Google Scholar] [CrossRef]
- Youil, R.; Toner, T.J.; Su, Q.; Casimiro, D.; Shiver, J.W.; Chen, L.; Bett, A.J.; Rogers, B.M.; Burden, E.C.; Tang, A.; et al. Comparative Analysis of the Effects of Packaging Signal, Transgene Orientation, Promoters, Polyadenylation Signals, and E3 Region on Growth Properties of First-Generation Adenoviruses. Hum. Gene Ther. 2003, 14, 1017–1034. [Google Scholar] [CrossRef]
- Bramson, J.; Hitt, M.; Gallichan, W.S.; Rosenthal, K.L.; Gauldie, J.; Graham, F.L. Construction of a Double Recombinant Adenovirus Vector Expressing a Heterodimeric Cytokine: In Vitro and In Vivo Production of Biologically Active Interleukin-12. Hum. Gene Ther. 1996, 7, 333–342. [Google Scholar] [CrossRef]
- Schneider, M.; Graham, F.L.; Prevec, L. Expression of the Glycoprotein of Vesicular Stomatitis Virus by Infectious Adenovirus Vectors. J. Gen. Virol. 1989, 70, 417–427. [Google Scholar] [CrossRef]
- Mittal, S.K.; McDermott, M.R.; Johnson, D.C.; Prevec, L.; Graham, F.L. Monitoring foreign gene expression by a human adenovirus-based vector using the firefly luciferase gene as a reporter. Virus Res. 1993, 28, 67–90. [Google Scholar] [CrossRef]
- Yarosh, O.K.; Wandeler, A.I.; Graham, F.L.; Campbell, J.B.; Prevec, L. Human adenovirus type 5 vectors expressing rabies glycoprotein. Vaccine 1996, 14, 1257–1264. [Google Scholar] [CrossRef]
- Wang, Y.; Hallden, G.; Hill, R.; Anand, A.; Liu, T.-C.; Francis, J.; Brooks, G.; Lemoine, N.; Kirn, D. E3 gene manipulations affect oncolytic adenovirus activity in immunocompetent tumor models. Nat. Biotechnol. 2003, 21, 1328–1335. [Google Scholar] [CrossRef] [PubMed]
- Bortolanza, S.; Bunuales, M.; Alzuguren, P.; Lamas, O.; Aldabe, R.; Prieto, J.; Hernandez-Alcoceba, R. Deletion of the E3-6.7K/gp19K region reduces the persistence of wild-type adenovirus in a permissive tumor model in Syrian hamsters. Cancer Gene Ther. 2009, 16, 703–712. [Google Scholar] [CrossRef]
- Suzuki, K.; Alemany, R.; Yamamoto, M.; Curiel, D.T. The Presence of the Adenovirus E3 Region Improves the Oncolytic Potency of Conditionally Replicative Adenoviruses. Clin. Cancer Res. 2002, 8, 3348–3359. [Google Scholar]
- García-Castro, J.; Martínez-Palacio, J.; Lillo, R.; García-Sánchez, F.; Alemany, R.; Madero, L.; Bueren, J.A.; Ramírez, M. Tumor cells as cellular vehicles to deliver gene therapies to metastatic tumors. Cancer Gene Ther. 2005, 12, 341–349. [Google Scholar] [CrossRef]
- Fuerer, C.; Iggo, R. 5-Fluorocytosine increases the toxicity of Wnt-targeting replicating adenoviruses that express cytosine deaminase as a late gene. Gene Ther. 2004, 11, 142–151. [Google Scholar] [CrossRef]
- Robinson, M.; Ge, Y.; Ko, D.; Yendluri, S.; Laflamme, G.; Hawkins, L.; Jooss, K. Comparison of the E3 and L3 regions for arming oncolytic adenoviruses to achieve a high level of tumor-specific transgene expression. Cancer Gene Ther. 2008, 15, 9–17. [Google Scholar] [CrossRef]
- Nettelbeck, D.M. Cellular genetic tools to control oncolytic adenoviruses for virotherapy of cancer. J. Mol. Med. 2008, 86, 363–377. [Google Scholar] [CrossRef]
- Brachtlova, T.; van Beusechem, V. Unleashing the Full Potential of Oncolytic Adenoviruses against Cancer by Applying RNA Interference: The Force Awakens. Cells 2018, 7, 228. [Google Scholar] [CrossRef]
- Suzuki, T.; Sakurai, F.; Nakamura, S.; Kouyama, E.; Kawabata, K.; Kondoh, M.; Yagi, K.; Mizuguchi, H. miR-122a-regulated expression of a suicide gene prevents hepatotoxicity without altering antitumor effects in suicide gene therapy. Mol. Ther. 2008, 16, 1719–1726. [Google Scholar] [CrossRef] [PubMed]
- Card, P.B.; Hogg, R.T.; Gil Del Alcazar, C.R.; Gerard, R.D. MicroRNA silencing improves the tumor specificity of adenoviral transgene expression. Cancer Gene Ther. 2012, 19, 451–459. [Google Scholar] [CrossRef] [PubMed]
- Geisler, A.; Fechner, H. MicroRNA-regulated viral vectors for gene therapy. World J. Exp. Med. 2016, 6, 37–54. [Google Scholar] [CrossRef] [PubMed]
- Davis, J.J.; Wang, L.; Dong, F.; Zhang, L.; Guo, W.; Teraishi, F.; Xu, K.; Ji, L.; Fang, B. Oncolysis and suppression of tumor growth by a GFP-expressing oncolytic adenovirus controlled by an hTERT and CMV hybrid promoter. Cancer Gene Ther. 2006, 13, 720–723. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.J.; Galoforo, S.S.; Battle, P.; Lee, H.; Corry, P.M.; Jessup, J.M. Replicating adenoviral vector–mediated transfer of a heat-inducible double suicide gene for gene therapy. Cancer Gene Ther. 2001, 8, 397–404. [Google Scholar] [CrossRef]
- Rohmer, S.; Mainka, A.; Knippertz, I.; Hesse, A.; Nettelbeck, D.M. Insulated hsp70B′ promoter: Stringent heat-inducible activity in replication-deficient, but not replication-competent adenoviruses. J. Gene Med. 2008, 10, 340–354. [Google Scholar] [CrossRef]
- Rivera, A.A.; Wang, M.; Suzuki, K.; Uil, T.G.; Krasnykh, V.; Curiel, D.T.; Nettelbeck, D.M. Mode of transgene expression after fusion to early or late viral genes of a conditionally replicating adenovirus via an optimized internal ribosome entry site in vitro and in vivo. Virology 2004, 320, 121–134. [Google Scholar] [CrossRef]
- Sauthoff, H.; Pipiya, T.; Heitner, S.; Chen, S.; Norman, R.G.; Rom, W.N.; Hay, J.G. Late Expression of p53 from a Replicating Adenovirus Improves Tumor Cell Killing and Is More Tumor Cell Specific than Expression of the Adenoviral Death Protein. Hum. Gene Ther. 2002, 13, 1859–1871. [Google Scholar] [CrossRef]
- Lukashev, A.N.; Fuerer, C.; Chen, M.J.; Searle, P.; Iggo, R. Late Expression of Nitroreductase in an Oncolytic Adenovirus Sensitizes Colon Cancer Cells to the Prodrug CB1954. Hum. Gene Ther. 2005, 16, 1473–1483. [Google Scholar] [CrossRef]
- Cascante, A.; Abate-Daga, D.; Garcia-Rodríguez, L.; González, J.R.; Alemany, R.; Fillat, C. GCV modulates the antitumoural efficacy of a replicative adenovirus expressing the TAT8-TK as a late gene in a pancreatic tumour model. Gene Ther. 2007, 14, 1471–1480. [Google Scholar] [CrossRef][Green Version]
- Donnelly, M.L.L.; Mehrotra, A.; Gani, D.; Hughes, L.E.; Luke, G.; Li, X.; Ryan, M.D. Analysis of the aphthovirus 2A/2B polyprotein ‘cleavage’ mechanism indicates not a proteolytic reaction, but a novel translational effect: A putative ribosomal ‘skip’. J. Gen. Virol. 2001, 82, 1013–1025. [Google Scholar] [CrossRef] [PubMed]
- Szymczak, A.L.; Workman, C.J.; Wang, Y.; Vignali, K.M.; Dilioglou, S.; Vanin, E.F.; Vignali, D.A.A. Correction of multi-gene deficiency in vivo using a single “self-cleaving” 2A peptide–based retroviral vector. Nat. Biotechnol. 2004, 22, 589–594. [Google Scholar] [CrossRef] [PubMed]
- Funston, G.M.; Kallioinen, S.E.; de Felipe, P.; Ryan, M.D.; Iggo, R.D. Expression of heterologous genes in oncolytic adenoviruses using picornaviral 2A sequences that trigger ribosome skipping. J. Gen. Virol. 2008, 89, 389–396. [Google Scholar] [CrossRef] [PubMed]
- Quirin, C.; Rohmer, S.; Fernández-Ulibarri, I.; Behr, M.; Hesse, A.; Engelhardt, S.; Erbs, P.; Enk, A.H.; Nettelbeck, D.M. Selectivity and Efficiency of Late Transgene Expression by Transcriptionally Targeted Oncolytic Adenoviruses Are Dependent on the Transgene Insertion Strategy. Hum. Gene Ther. 2011, 22, 389–404. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Tan, Y.; Lei, W.; Wang, Y.; Zhou, X.; Jia, X.; Zhang, K.; Chu, L.; Liu, X.; Qian, W. Complete Eradication of Xenograft Hepatoma by Oncolytic Adenovirus ZD55 Harboring TRAIL-IETD-Smac Gene with Broad Antitumor Effect. Hum. Gene Ther. 2012, 23, 992–1002. [Google Scholar] [CrossRef]
- Zhang, R.; Zhang, X.; Ma, B.; Xiao, B.; Huang, F.; Huang, P.; Ying, C.; Liu, T.; Wang, Y. Enhanced antitumor effect of combining TRAIL and MnSOD mediated by CEA-controlled oncolytic adenovirus in lung cancer. Cancer Gene Ther. 2016, 23, 168–177. [Google Scholar] [CrossRef]
- Berget, S.M.; Moore, C.; Sharp, P.A. Spliced segments at the 5′ terminus of adenovirus 2 late mRNA. Proc. Natl. Acad. Sci. USA 1977, 74, 3171–3175. [Google Scholar] [CrossRef]
- Muhlemann, O.; Yue, B.G.; Petersen-Mahrt, S.; Akusjarvi, G. A Novel Type of Splicing Enhancer Regulating Adenovirus Pre-mRNA Splicing. Mol. Cell. Biol. 2000, 20, 2317–2325. [Google Scholar] [CrossRef]
- Guedan, S.; Rojas, J.J.; Gros, A.; Mercade, E.; Cascallo, M.; Alemany, R. Hyaluronidase Expression by an Oncolytic Adenovirus Enhances Its Intratumoral Spread and Suppresses Tumor Growth. Mol. Ther. 2010, 18, 1275–1283. [Google Scholar] [CrossRef]
- Fajardo, C.A.; Guedan, S.; Rojas, L.A.; Moreno, R.; Arias-Badia, M.; de Sostoa, J.; June, C.H.; Alemany, R. Oncolytic Adenoviral Delivery of an EGFR-Targeting T-cell Engager Improves Antitumor Efficacy. Cancer Res. 2017, 77, 2052–2063. [Google Scholar] [CrossRef]
- de Sostoa, J.; Fajardo, C.A.; Moreno, R.; Ramos, M.D.; Farrera-Sal, M.; Alemany, R. Targeting the tumor stroma with an oncolytic adenovirus secreting a fibroblast activation protein-targeted bispecific T-cell engager. J. Immunother. Cancer 2019, 7, 19. [Google Scholar] [CrossRef]
- Carette, J.E.; Graat, H.C.A.; Schagen, F.H.E.; Abou El Hassan, M.A.I.; Gerritsen, W.R.; van Beusechem, V.W. Replication-dependent transgene expression from a conditionally replicating adenovirus via alternative splicing to a heterologous splice-acceptor site. J. Gene Med. 2005, 7, 1053–1062. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Ulibarri, I.; Hammer, K.; Arndt, M.A.E.; Kaufmann, J.K.; Dorer, D.; Engelhardt, S.; Kontermann, R.E.; Hess, J.; Allgayer, H.; Krauss, J.; et al. Genetic delivery of an immunoRNase by an oncolytic adenovirus enhances anticancer activity: ImmunoRNase-expressing oncolytic adenovirus. Int. J. Cancer 2015, 136, 2228–2240. [Google Scholar] [CrossRef] [PubMed]
- Hermening, S.; Kügler, S.; Bähr, M.; Isenmann, S. Increased protein expression from adenoviral shuttle plasmids and vectors by insertion of a small chimeric intron sequence. J. Virol. Methods 2004, 122, 73–77. [Google Scholar] [CrossRef]
- Villanueva, E.; Martí-Solano, M.; Fillat, C. Codon optimization of the adenoviral fiber negatively impacts structural protein expression and viral fitness. Sci. Rep. 2016, 6, 27546. [Google Scholar] [CrossRef]
- Akbulut, H.; Zhang, L.; Tang, Y.; Deisseroth, A. Cytotoxic effect of replication-competent adenoviral vectors carrying L-plastin promoter regulated E1A and cytosine deaminase genes in cancers of the breast, ovary and colon. Cancer Gene Ther. 2003, 10, 388–395. [Google Scholar] [CrossRef]
- Chen, M.J.; Green, N.K.; Reynolds, G.M.; Flavell, J.R.; Mautner, V.; Kerr, D.J.; Young, L.S.; Searle, P.F. Enhanced efficacy of Escherichia coli nitroreductase/CB1954 prodrug activation gene therapy using an E1B-55K-deleted oncolytic adenovirus vector. Gene Ther. 2004, 11, 1126–1136. [Google Scholar] [CrossRef] [PubMed]
- Singleton, D.C.; Li, D.; Bai, S.Y.; Syddall, S.P.; Smaill, J.B.; Shen, Y.; Denny, W.A.; Wilson, W.R.; Patterson, A.V. The nitroreductase prodrug SN 28343 enhances the potency of systemically administered armed oncolytic adenovirus ONYX-411 NTR. Cancer Gene Ther. 2007, 14, 953–967. [Google Scholar] [CrossRef]
- Stubdal, H.; Perin, N.; Lemmon, M.; Holman, P.; Bauzon, M.; Potter, P.M.; Danks, M.K.; Fattaey, A.; Dubensky, T.; Johnson, L. A Prodrug Strategy Using ONYX-015-Based Replicating Adenoviruses to Deliver Rabbit Carboxylesterase to Tumor Cells for Conversion of CPT-11 to SN-38. Cancer Res. 2003, 63, 6900–6908. [Google Scholar] [PubMed]
- Schepelmann, S.; Hallenbeck, P.; Ogilvie, L.M.; Hedley, D.; Friedlos, F.; Martin, J.; Scanlon, I.; Hay, C.; Hawkins, L.K.; Marais, R.; et al. Systemic Gene-Directed Enzyme Prodrug Therapy of Hepatocellular Carcinoma Using a Targeted Adenovirus Armed with Carboxypeptidase G2. Cancer Res. 2005, 65, 5003–5008. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Erbs, P.; Regulier, E.; Kintz, J.; Leroy, P.; Poitevin, Y.; Exinger, F.; Jund, R.; Mehtali, M. In Vivo Cancer Gene Therapy by Adenovirus-mediated Transfer of a Bifunctional Yeast Cytosine Deaminase/Uracil Phosphoribosyltransferase Fusion Gene. Cancer Res. 2000, 60, 3813–3822. [Google Scholar]
- Qian, C.Y.; Wang, K.L.; Fang, F.F.; Gu, W.; Huang, F.; Wang, F.Z.; Li, B.; Wang, L.N. Triple-controlled oncolytic adenovirus expressing melittin to exert inhibitory efficacy on hepatocellular carcinoma. Int. J. Clin. Exp. Pathol. 2015, 8, 10403–10411. [Google Scholar]
- Li, B.; Ling, C.; Zhang, C.; Gu, W.; Li, S.; Huang, X.; Zhang, Y.; Yu, C. The induced apoptosis of recombinant adenovirus carrying melittin gene for hepatocellular carcinoma cell. Chin. J. Hepatol. 2004, 12, 453–455. [Google Scholar]
- Mi, J.; Li, Z.Y.; Ni, S.; Steinwaerder, D.; Lieber, A. Induced Apoptosis Supports Spread of Adenovirus Vectors in Tumors. Hum. Gene Ther. 2001, 12, 1343–1352. [Google Scholar] [CrossRef]
- van Beusechem, V.W.; van den Doel, P.B.; Grill, J.; Pinedo, H.M.; Gerritsen, W.R. Conditionally Replicative Adenovirus Expressing p53 Exhibits Enhanced Oncolytic Potency. Cancer Res. 2002, 62, 6165–6171. [Google Scholar] [PubMed]
- Kurihara, T.; Brough, D.E.; Kovesdi, I.; Kufe, D.W. Selectivity of a replication-competent adenovirus for human breast carcinoma cells expressing the MUC1 antigen. J. Clin. Investig. 2000, 106, 763–771. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sova, P.; Ren, X.W.; Ni, S.; Bernt, K.M.; Mi, J.; Kiviat, N.; Lieber, A. A Tumor-Targeted and Conditionally Replicating Oncolytic Adenovirus Vector Expressing TRAIL for Treatment of Liver Metastases. Mol. Ther. 2004, 9, 496–509. [Google Scholar] [CrossRef]
- Han, Z.; Lee, S.; Je, S.; Eom, C.Y.; Choi, H.J.; Song, J.J.; Kim, J.H. Survivin silencing and TRAIL expression using oncolytic adenovirus increase anti-tumorigenic activity in gemcitabine-resistant pancreatic cancer cells. Apoptosis 2016, 21, 351–364. [Google Scholar] [CrossRef]
- Oh, E.; Hong, J.; Kwon, O.-J.; Yun, C.O. A hypoxia- and telomerase-responsive oncolytic adenovirus expressing secretable trimeric TRAIL triggers tumour-specific apoptosis and promotes viral dispersion in TRAIL-resistant glioblastoma. Sci. Rep. 2018, 8. [Google Scholar] [CrossRef] [PubMed]
- Zou, H.; Zheng, Y.F.; Ge, W.; Wang, S.B.; Mou, X.Z. Synergistic Anti-tumour Effects of Quercetin and Oncolytic Adenovirus expressing TRAIL in Human Hepatocellular Carcinoma. Sci. Rep. 2018, 8, 2182. [Google Scholar] [CrossRef]
- Gao, H.; Zhang, X.; Ding, Y.; Qiu, R.; Hong, Y.; Chen, W. Synergistic Suppression Effect on Tumor Growth of Colorectal Cancer by Combining Radiotherapy with a TRAIL-Armed Oncolytic Adenovirus. Technol. Cancer Res. Treat. 2019, 18. [Google Scholar] [CrossRef]
- Fang, L.; Cheng, Q.; Liu, W.; Zhang, J.; Ge, Y.; Zhang, Q.; Li, L.; Liu, J.; Zheng, J. Selective effects of a fiber chimeric conditionally replicative adenovirus armed with hep27 gene on renal cancer cell. Cancer Biol. Ther. 2016, 17, 664–673. [Google Scholar] [CrossRef]
- Xie, W.; Hao, J.; Zhang, K.; Fang, X.; Liu, X. Adenovirus armed with VGLL4 selectively kills hepatocellular carcinoma with G2/M phase arrest and apoptosis promotion. Biochem. Biophys. Res. Commun. 2018, 503, 2758–2763. [Google Scholar] [CrossRef]
- Jiang, H.; Guo, S.; Xiao, D.; Bian, X.; Wang, J.; Wang, Y.; Zhou, H.; Cai, J.; Zheng, Z. Arginine deiminase expressed in vivo, driven by human telomerase reverse transcriptase promoter, displays high hepatoma targeting and oncolytic efficiency. Oncotarget 2017, 8, 37694–37704. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.B.; Shu, J.; Chen, L.; Chen, X.; Zhao, J.; Li, S.; Mou, X.; Tong, X. Synergistic suppression effect on tumor growth of ovarian cancer by combining cisplatin with a manganese superoxide dismutase-armed oncolytic adenovirus. OncoTargets Ther. 2016, 9, 6381–6388. [Google Scholar] [CrossRef] [PubMed]
- Wakabayashi, K.; Sakurai, F.; Ono, R.; Fujiwara, T.; Mizuguchi, H. Development of a Novel Oncolytic Adenovirus Expressing a Short-hairpin RNA against Cullin 4A. Anticancer Res. 2020, 40, 161–168. [Google Scholar] [CrossRef] [PubMed]
- Rovira-Rigau, M.; Raimondi, G.; Marín, M.Á.; Gironella, M.; Alemany, R.; Fillat, C. Bioselection Reveals miR-99b and miR-485 as Enhancers of Adenoviral Oncolysis in Pancreatic Cancer. Mol. Ther. 2019, 27, 230–243. [Google Scholar] [CrossRef]
- Luo, Q.; Basnet, S.; Dai, Z.; Li, S.; Zhang, Z.; Ge, H. A novel E1B55kDa-deleted oncolytic adenovirus carrying microRNA-143 exerts specific antitumor efficacy on colorectal cancer cells. Am. J. Transl. Res. 2016, 8, 3822–3830. [Google Scholar]
- Xu, Y.; Chu, L.; Yuan, S.; Yang, Y.; Yang, Y.; Xu, B.; Zhang, K.; Liu, X.Y.; Wang, R.; Fang, L. RGD-modified oncolytic adenovirus-harboring shPKM2 exhibits a potent cytotoxic effect in pancreatic cancer via autophagy inhibition and apoptosis promotion. Cell Death Dis. 2017, 8, e2835. [Google Scholar] [CrossRef]
- Li, Y.; Zhuo, B.; Yin, Y.; Han, T.; Li, S.; Li, Z.; Wang, J. Anti-cancer effect of oncolytic adenovirus-armed shRNA targeting MYCN gene on doxorubicin-resistant neuroblastoma cells. Biochem. Biophys. Res. Commun. 2017, 491, 134–139. [Google Scholar] [CrossRef]
- Li, X.; Su, Y.; Sun, B.; Ji, W.; Peng, Z.; Xu, Y.; Wu, M.; Su, C. An Artificially Designed Interfering lncRNA Expressed by Oncolytic Adenovirus Competitively Consumes OncomiRs to Exert Antitumor Efficacy in Hepatocellular Carcinoma. Mol. Cancer Ther. 2016, 15, 1436–1451. [Google Scholar] [CrossRef]
- Zhang, Y.A.; Nemunaitis, J.; Samuel, S.K.; Chen, P.; Shen, Y.; Tong, A.W. Antitumor Activity of an Oncolytic Adenovirus-Delivered Oncogene Small Interfering RNA. Cancer Res. 2006, 66, 9736–9743. [Google Scholar] [CrossRef] [PubMed]
- Chu, L.; Gu, J.; Sun, L.; Qian, Q.; Qian, C.; Liu, X. Oncolytic adenovirus-mediated shRNA against Apollon inhibits tumor cell growth and enhances antitumor effect of 5-fluorouracil. Gene Ther. 2008, 15, 484–494. [Google Scholar] [CrossRef] [PubMed]
- Yoo, J.Y.; Kim, J.H.; Kwon, Y.G.; Kim, E.C.; Kim, N.K.; Choi, H.J.; Yun, C.O. VEGF-specific Short Hairpin RNA–expressing Oncolytic Adenovirus Elicits Potent Inhibition of Angiogenesis and Tumor Growth. Mol. Ther. 2007, 15, 295–302. [Google Scholar] [CrossRef]
- Kuriyama, N.; Kuriyama, H.; Julin, C.M.; Lamborn, K.; Israel, M.A. Pretreatment with protease is a useful experimental strategy for enhancing adenovirus-mediated cancer gene therapy. Hum. Gene Ther. 2000, 11, 2219–2230. [Google Scholar] [CrossRef]
- Kim, J.H.; Lee, Y.S.; Kim, H.; Huang, J.H.; Yoon, A.R.; Yun, C.O. Relaxin Expression from Tumor-Targeting Adenoviruses and Its Intratumoral Spread, Apoptosis Induction, and Efficacy. J. Natl. Cancer Inst. 2006, 98, 1482–1493. [Google Scholar] [CrossRef]
- Ganesh, S.; Edick, M.G.; Idamakanti, N.; Abramova, M.; VanRoey, M.; Robinson, M.; Yun, C.O.; Jooss, K. Relaxin-Expressing, Fiber Chimeric Oncolytic Adenovirus Prolongs Survival of Tumor-Bearing Mice. Cancer Res. 2007, 67, 4399–4407. [Google Scholar] [CrossRef]
- Rodriguez-Garcia, A.; Gimenez-Alejandre, M.; Rojas, J.J.; Moreno, R.; Bazan-Peregrino, M.; Cascallo, M.; Alemany, R. Safety and Efficacy of VCN-01, an Oncolytic Adenovirus Combining Fiber HSG-Binding Domain Replacement with RGD and Hyaluronidase Expression. Clin. Cancer Res. 2015, 21, 1406–1418. [Google Scholar] [CrossRef]
- Garcia-Moure, M.; Martinez-Velez, N.; Gonzalez-Huarriz, M.; Marrodán, L.; Cascallo, M.; Alemany, R.; Patiño-García, A.; Alonso, M.M. The oncolytic adenovirus VCN-01 promotes anti-tumor effect in primitive neuroectodermal tumor models. Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef]
- Martínez-Vélez, N.; Xipell, E.; Vera, B.; de la Rocha, A.A.; Zalacain, M.; Marrodán, L.; Gonzalez-Huarriz, M.; Toledo, G.; Cascallo, M.; Alemany, R.; et al. The Oncolytic Adenovirus VCN-01 as Therapeutic Approach against Pediatric Osteosarcoma. Clin. Cancer Res. 2016, 22, 2217–2225. [Google Scholar] [CrossRef]
- Pascual-Pasto, G.; Bazan-Peregrino, M.; Olaciregui, N.G.; Restrepo-Perdomo, C.A.; Mato-Berciano, A.; Ottaviani, D.; Weber, K.; Correa, G.; Paco, S.; Vila-Ubach, M.; et al. Therapeutic targeting of the RB1 pathway in retinoblastoma with the oncolytic adenovirus VCN-01. Sci. Transl. Med. 2019, 11. [Google Scholar] [CrossRef] [PubMed]
- Tedcastle, A.; Illingworth, S.; Brown, A.; Seymour, L.W.; Fisher, K.D. Actin-resistant DNAse I Expression from Oncolytic Adenovirus Enadenotucirev Enhances Its Intratumoral Spread and Reduces Tumor Growth. Mol. Ther. 2016, 24, 796–804. [Google Scholar] [CrossRef]
- Hoffmann, D.; Bangen, J.-M.; Bayer, W.; Wildner, O. Synergy between expression of fusogenic membrane proteins, chemotherapy and facultative virotherapy in colorectal cancer. Gene Ther. 2006, 13, 1534–1544. [Google Scholar] [CrossRef]
- Guedan, S.; Grases, D.; Rojas, J.J.; Gros, A.; Vilardell, F.; Vile, R.; Mercade, E.; Cascallo, M.; Alemany, R. GALV expression enhances the therapeutic efficacy of an oncolytic adenovirus by inducing cell fusion and enhancing virus distribution. Gene Ther. 2012, 19, 1048–1057. [Google Scholar] [CrossRef] [PubMed]
- Bateman, A.R.; Harrington, K.J.; Kottke, T.; Ahmed, A.; Melcher, A.A.; Gough, M.J.; Linardakis, E.; Riddle, D.; Dietz, A.; Lohse, C.M.; et al. Viral fusogenic membrane glycoproteins kill solid tumor cells by nonapoptotic mechanisms that promote cross presentation of tumor antigens by dendritic cells. Cancer Res. 2002, 62, 6566–6578. [Google Scholar] [PubMed]
- Errington, F.; Jones, J.; Merrick, A.; Bateman, A.; Harrington, K.; Gough, M.; O’Donnell, D.; Selby, P.; Vile, R.; Melcher, A. Fusogenic membrane glycoprotein-mediated tumour cell fusion activates human dendritic cells for enhanced IL-12 production and T-cell priming. Gene Ther. 2006, 13, 138–149. [Google Scholar] [CrossRef] [PubMed]
- Ranki, T.; Joensuu, T.; Jäger, E.; Karbach, J.; Wahle, C.; Kairemo, K.; Alanko, T.; Partanen, K.; Turkki, R.; Linder, N.; et al. Local treatment of a pleural mesothelioma tumor with ONCOS-102 induces a systemic antitumor CD8 + T-cell response, prominent infiltration of CD8 + lymphocytes and Th1 type polarization. OncoImmunology 2014, 3, e958937. [Google Scholar] [CrossRef]
- Vassilev, L.; Ranki, T.; Joensuu, T.; Jäger, E.; Karbach, J.; Wahle, C.; Partanen, K.; Kairemo, K.; Alanko, T.; Turkki, R.; et al. Repeated intratumoral administration of ONCOS-102 leads to systemic antitumor CD8 + T-cell response and robust cellular and transcriptional immune activation at tumor site in a patient with ovarian cancer. OncoImmunology 2015, 4, e1017702. [Google Scholar] [CrossRef]
- Koski, A.; Kangasniemi, L.; Escutenaire, S.; Pesonen, S.; Cerullo, V.; Diaconu, I.; Nokisalmi, P.; Raki, M.; Rajecki, M.; Guse, K.; et al. Treatment of Cancer Patients with a Serotype 5/3 Chimeric Oncolytic Adenovirus Expressing GMCSF. Mol. Ther. 2010, 18, 1874–1884. [Google Scholar] [CrossRef]
- Ramesh, N.; Ge, Y.; Ennist, D.L.; Zhu, M.; Mina, M.; Ganesh, S.; Reddy, P.S.; Yu, D.C. CG0070, a conditionally replicating granulocyte macrophage colony-stimulating factor--armed oncolytic adenovirus for the treatment of bladder cancer. Clin. Cancer Res. 2006, 12, 305–313. [Google Scholar] [CrossRef]
- Burke, J.M.; Lamm, D.L.; Meng, M.V.; Nemunaitis, J.J.; Stephenson, J.J.; Arseneau, J.C.; Aimi, J.; Lerner, S.; Yeung, A.W.; Kazarian, T.; et al. A first in human phase 1 study of CG0070, a GM-CSF expressing oncolytic adenovirus, for the treatment of nonmuscle invasive bladder cancer. J. Urol. 2012, 188, 2391–2397. [Google Scholar] [CrossRef]
- Ahn, H.M.; Hong, J.; Yun, C.O. Oncolytic adenovirus coexpressing interleukin-12 and shVEGF restores antitumor immune function and enhances antitumor efficacy. Oncotarget 2016, 7, 84965–84980. [Google Scholar] [CrossRef] [PubMed]
- Oh, E.; Choi, I.K.; Hong, J.; Yun, C.O. Oncolytic adenovirus coexpressing interleukin-12 and decorin overcomes Treg-mediated immunosuppression inducing potent antitumor effects in a weakly immunogenic tumor model. Oncotarget 2017, 8, 4730–4746. [Google Scholar] [CrossRef] [PubMed]
- Huang, J.H.; Zhang, S.N.; Choi, K.J.; Choi, I.K.; Kim, J.H.; Lee, M.; Kim, H.; Yun, C.O. Therapeutic and tumor-specific immunity induced by combination of dendritic cells and oncolytic adenovirus expressing IL-12 and 4-1BBL. Mol. Ther. 2010, 18, 264–274. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Xu, H.; Wang, J.; Wu, X.; Wen, W.; Liang, Y.; Wang, L.; Liu, F.; Du, X. Inhibition of breast cancer cells by targeting E2F-1 gene and expressing IL15 oncolytic adenovirus. Biosci. Rep. 2019, 39. [Google Scholar] [CrossRef]
- Ye, J.; Qi, W.; Liu, M.; Li, Y. The combination of NK and CD8+ T cells with CCL20/IL15-armed oncolytic adenoviruses enhances the growth suppression of TERT-positive tumor cells. Cell. Immunol. 2017, 318, 35–41. [Google Scholar] [CrossRef]
- Li, Y.; Li, Y.; Si, C.; Zhu, Y.; Jin, Y.; Zhu, T.; Liu, M.; Liu, G. CCL21/IL21-armed oncolytic adenovirus enhances antitumor activity against TERT-positive tumor cells. Virus Res. 2016, 220, 172–178. [Google Scholar] [CrossRef]
- Sarkar, D.; Lebedeva, I.V.; Su, Z.; Park, E.S.; Chatman, L.; Vozhilla, N.; Dent, P.; Curiel, D.T.; Fisher, P.B. Eradication of Therapy-Resistant Human Prostate Tumors Using a Cancer Terminator Virus. Cancer Res. 2007, 67, 5434–5442. [Google Scholar] [CrossRef]
- Ashshi, A.M.; El-Shemi, A.G.; Dmitriev, I.P.; Kashentseva, E.A.; Curiel, D.T. Combinatorial strategies based on CRAd-IL24 and CRAd-ING4 virotherapy with anti-angiogenesis treatment for ovarian cancer. J. Ovarian Res. 2016, 9, 38. [Google Scholar] [CrossRef]
- Mao, L.; Ding, M.; Xu, K.; Pan, J.; Yu, H.; Yang, C. Oncolytic Adenovirus Harboring Interleukin-24 Improves Chemotherapy for Advanced Prostate Cancer. J. Cancer 2018, 9, 4391–4397. [Google Scholar] [CrossRef]
- Yuan, S.; Fang, X.; Xu, Y.; Ni, A.; Liu, X.Y.; Chu, L. An oncolytic adenovirus that expresses the HAb18 and interleukin 24 genes exhibits enhanced antitumor activity in hepatocellular carcinoma cells. Oncotarget 2016, 7, 60491–60502. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Gu, J.; Dong, A.; Zhang, Y.; Zhong, L.; He, L.; Wang, Y.; Zhang, J.; Zhang, Z.; Huiwang, J.; et al. Potent Antitumor Activity of Oncolytic Adenovirus Expressing mda-7/IL-24 for Colorectal Cancer. Hum. Gene Ther. 2005, 16, 845–858. [Google Scholar] [CrossRef] [PubMed]
- Wei, X.; Liu, L.; Wang, G.; Li, W.; Xu, K.; Hu, X.; Qian, C.; Shao, J. Targeting eradication of chronic myeloid leukemia using chimeric oncolytic adenovirus to drive IL-24 expression. Int. J. Clin. Exp. Pathol. 2015, 8, 3775–3784. [Google Scholar]
- Cervera-Carrascon, V.; Siurala, M.; Santos, J.M.; Havunen, R.; Tähtinen, S.; Karell, P.; Sorsa, S.; Kanerva, A.; Hemminki, A. TNFa and IL-2 armed adenoviruses enable complete responses by anti-PD-1 checkpoint blockade. OncoImmunology 2018, 7, e1412902. [Google Scholar] [CrossRef]
- Eriksson, E.; Milenova, I.; Wenthe, J.; Ståhle, M.; Leja-Jarblad, J.; Ullenhag, G.; Dimberg, A.; Moreno, R.; Alemany, R.; Loskog, A. Shaping the Tumor Stroma and Sparking Immune Activation by CD40 and 4-1BB Signaling Induced by an Armed Oncolytic Virus. Clin. Cancer Res. 2017, 23, 5846–5857. [Google Scholar] [CrossRef]
- Dias, J.D.; Hemminki, O.; Diaconu, I.; Hirvinen, M.; Bonetti, A.; Guse, K.; Escutenaire, S.; Kanerva, A.; Pesonen, S.; Löskog, A.; et al. Targeted cancer immunotherapy with oncolytic adenovirus coding for a fully human monoclonal antibody specific for CTLA-4. Gene Ther. 2012, 19, 988–998. [Google Scholar] [CrossRef]
- Liikanen, I.; Ta htinen, S.; Guse, K.; Gutmann, T.; Savola, P.; Oksanen, M.; Kanerva, A.; Hemminki, A. Oncolytic Adenovirus Expressing Monoclonal Antibody Trastuzumab for Treatment of HER2-Positive Cancer. Mol. Cancer Ther. 2016, 15, 2259–2269. [Google Scholar] [CrossRef]
- Jiang, H.; Rivera-Molina, Y.; Gomez-Manzano, C.; Clise-Dwyer, K.; Bover, L.; Vence, L.M.; Yuan, Y.; Lang, F.F.; Toniatti, C.; Hossain, M.B.; et al. Oncolytic adenovirus and tumor-targeting immune modulatory therapy improve autologous cancer vaccination. Cancer Res. 2017, 77, 3894–3907. [Google Scholar] [CrossRef]
- Rivera-Molina, Y.; Jiang, H.; Fueyo, J.; Nguyen, T.; Shin, D.H.; Youssef, G.; Fan, X.; Gumin, J.; Alonso, M.M.; Phadnis, S.; et al. GITRL-armed Delta-24-RGD oncolytic adenovirus prolongs survival and induces anti-glioma immune memory. Neuro-Oncol. Adv. 2019, 1, vdz009. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhang, H.; Wei, M.; Mou, T.; Shi, T.; Ma, Y.; Cai, X.; Li, Y.; Dong, J.; Wei, J. Recombinant Adenovirus Expressing a Soluble Fusion Protein PD-1/CD137L Subverts the Suppression of CD8+ T Cells in HCC. Mol. Ther. 2019, 27, 1906–1918. [Google Scholar] [CrossRef]
- Yang, Y.; Xu, W.; Peng, D.; Wang, H.; Zhang, X.; Wang, H.; Xiao, F.; Zhu, Y.; Ji, Y.; Gulukota, K.; et al. An Oncolytic Adenovirus Targeting Transforming Growth Factor β Inhibits Protumorigenic Signals and Produces Immune Activation: A Novel Approach to Enhance Anti-PD-1 and Anti-CTLA-4 Therapy. Hum. Gene Ther. 2019, 30, 1117–1132. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Xiao, F.; Zhang, A.; Zhang, D.; Nie, W.; Xu, T.; Han, B.; Seth, P.; Wang, H.; Yang, Y.; et al. Oncolytic adenovirus targeting TGF-β enhances anti-tumor responses of mesothelin-targeted chimeric antigen receptor T cell therapy against breast cancer. Cell. Immunol. 2020, 348, 104041. [Google Scholar] [CrossRef] [PubMed]
- Wing, A.; Fajardo, C.A.; Posey, A.D.; Shaw, C.; Da, T.; Young, R.M.; Alemany, R.; June, C.H.; Guedan, S. Improving CART-Cell Therapy of Solid Tumors with Oncolytic Virus–Driven Production of a Bispecific T-cell Engager. Cancer Immunol. Res. 2018, 6, 605–616. [Google Scholar] [CrossRef] [PubMed]
- Freedman, J.D.; Hagel, J.; Scott, E.M.; Psallidas, I.; Gupta, A.; Spiers, L.; Miller, P.; Kanellakis, N.; Ashfield, R.; Fisher, K.D.; et al. Oncolytic adenovirus expressing bispecific antibody targets T-cell cytotoxicity in cancer biopsies. EMBO Mol. Med. 2017, 9, 1067–1087. [Google Scholar] [CrossRef] [PubMed]
| Virus | Backbone | Deletion | Ins. Site | Promoter | Transgene | NCT Identifier |
|---|---|---|---|---|---|---|
| Ad5yCD/mutKSR39rep-hIL12 | Ad5 | E1bΔ55K, ΔE3 | E1b55K, E3 | CMV | yCD/TK, hIL-12 | NCT02555397 NCT03281382 |
| VCN-01 | Ad5 | E1aΔ24 | After L5 | IIIa SA | PH20 | NCT02045602 NCT03284268 NCT03799744 |
| CG0070 | Ad5 | E1a Δ24 E3Δ19K | Between 6.7 and ADP | End. P | GM-CSF | NCT02143804 |
| ONCOS-102 | Ad5; Ad3 fiber knob | E1aΔ24 E3Δ6.7/19K | E3 6.7/19K | End. P. | GM-CSF | NCT03514836 NCT03003676 NCT02963831 NCT02879669 |
| TILT-123 | Ad5; Ad3 fiber knob | E1aΔ24 ΔE3 | E3 | End.P. | TNFα-IRES- -IL-2 | NCT04217473 |
| LoAd703 | Ad5 | E1aΔ24; E3Δ6.7/19K | After L5 | CMV | 4-1BB CD40L | NCT03225989 NCT02705196 NCT04123470 |
| NG-641 | Ad11/3 | NA | NA | NA | IFNα, CXCL9, CXCL10, FAP-BiTE | NCT04053283 |
| NG-350A | Ad11/3 | NA | NA | NA | CD40 agonist mAb | NCT03852511 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Farrera-Sal, M.; Fillat, C.; Alemany, R. Effect of Transgene Location, Transcriptional Control Elements and Transgene Features in Armed Oncolytic Adenoviruses. Cancers 2020, 12, 1034. https://doi.org/10.3390/cancers12041034
Farrera-Sal M, Fillat C, Alemany R. Effect of Transgene Location, Transcriptional Control Elements and Transgene Features in Armed Oncolytic Adenoviruses. Cancers. 2020; 12(4):1034. https://doi.org/10.3390/cancers12041034
Chicago/Turabian StyleFarrera-Sal, Martí, Cristina Fillat, and Ramon Alemany. 2020. "Effect of Transgene Location, Transcriptional Control Elements and Transgene Features in Armed Oncolytic Adenoviruses" Cancers 12, no. 4: 1034. https://doi.org/10.3390/cancers12041034
APA StyleFarrera-Sal, M., Fillat, C., & Alemany, R. (2020). Effect of Transgene Location, Transcriptional Control Elements and Transgene Features in Armed Oncolytic Adenoviruses. Cancers, 12(4), 1034. https://doi.org/10.3390/cancers12041034






