Maximizing the Clinical Benefit of Radiotherapy in Solitary Plasmacytoma: An International Multicenter Analysis
Abstract
:1. Objective
2. Materials and Methods
3. Statistical Analysis
Definition of Response
4. Results
4.1. Treatments Administered
4.2. Response Rates
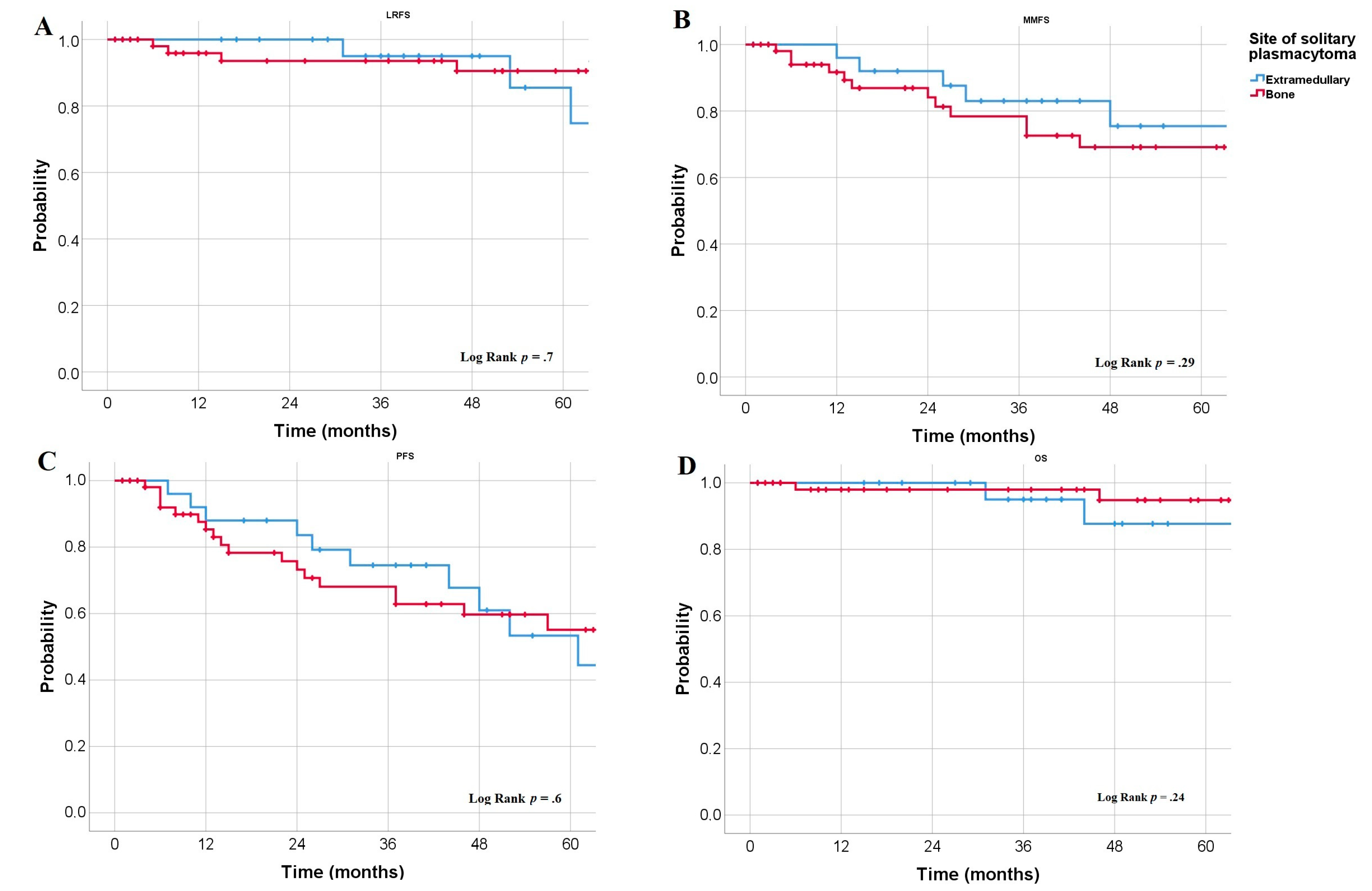
4.3. Survival Rates
4.4. Relapse Pattern
4.5. Toxicities
4.6. Cox proportional Hazard Model
5. Discussion
6. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Kumar, S.K.; Paiva, B.D.L.; Anderson, K.C.; Durie, B.; Landgren, O.; Moreau, P.; Munshi, N.V.; Lonial, S.; Bladé, J.; Mateos, M.-V.; et al. International Myeloma Working Group consensus criteria for response and minimal residual disease assessment in multiple myeloma. Lancet Oncol. 2016, 17, e328–e346. [Google Scholar] [CrossRef]
- Tsang, R.; Campbell, B.A.; Goda, J.S.; Kelsey, C.R.; Kirova, Y.M.; Parikh, R.R.; Ng, A.K.; Ricardi, U.; Suh, C.-O.; Mauch, P.M.; et al. Radiation Therapy for Solitary Plasmacytoma and Multiple Myeloma: Guidelines From the International Lymphoma Radiation Oncology Group. Int. J. Radiat. Oncol. Biol. Phys. 2018, 101, 794–808. [Google Scholar] [CrossRef] [Green Version]
- Goyal, G.; Bartley, A.C.; Funni, S.; Inselman, J.; Shah, N.D.; Marshall, A.L.; Ashrani, A.A.; Kapoor, P.; Durani, U.; Hashmi, S.K.; et al. Treatment approaches and outcomes in plasmacytomas: Analysis using a national dataset. Leukemia 2018, 32, 1414–1420. [Google Scholar] [CrossRef]
- Fotiou, D.; Dimopoulos, M.A.; Kastritis, E. How We Manage Patients with Plasmacytomas. Curr. Hematol. Malig. Rep. 2018, 13, 227–235. [Google Scholar] [CrossRef] [PubMed]
- Rajkumar, S.V.; Dimopoulos, M.A.; Palumbo, A.; Blade, J.; Merlini, G.; Mateos, M.V.; Kumar, S.; Hillengass, J.; Kastritis, E.; Richardson, P.; et al. International Myeloma Working Group updated criteria for the diagnosis of multiple myeloma. Lancet Oncol. 2014, 15, e538–e548. [Google Scholar] [CrossRef]
- Albano, D.; Bosio, G.; Treglia, G.; Giubbini, R.; Bertagna, F. 18F-FDG PET/CT in solitary plasmacytoma: Metabolic behavior and progression to multiple myeloma. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 77–84. [Google Scholar] [CrossRef] [PubMed]
- Caers, J.; Paiva, B.D.L.; Zamagni, E.; Leleu, X.; Bladé, J.; Kristinsson, S.Y.; Touzeau, C.; Abildgaard, N.; Terpos, E.; Heusschen, R.; et al. Diagnosis, treatment, and response assessment in solitary plasmacytoma: Updated recommendations from a European Expert Panel. J. Hematol. Oncol. 2018, 11, 10. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Li, H.; Liu, C.; Chen, C.; Yan, J. Solitary Plasmacytoma of Bone of the Spine: Results From Surveillance, Epidemiology, and End Results (SEER) Registry. Spine 2019, 44, E117–E125. [Google Scholar] [CrossRef]
- De Waal, E.G.; Leene, M.; Veeger, N.; Vos, H.J.; Ong, F.; Smit, W.G.; Hovenga, S.; Hoogendoorn, M.; Hogenes, M.; Beijert, M.; et al. Progression of a solitary plasmacytoma to multiple myeloma. A population-based registry of the northern Netherlands. Br. J. Haematol. 2016, 175, 661–667. [Google Scholar] [CrossRef]
- Ozsahin, M.; Tsang, R.; Poortmans, P.; Belkacemi, Y.; Bolla, M.; Dincbas, F.O.; Landmann, C.; Castelain, B.; Buijsen, J.; Curschmann, J.; et al. Outcomes and patterns of failure in solitary plasmacytoma: A multicenter Rare Cancer Network study of 258 patients. Int. J. Radiat. Oncol. Biol. Phys. 2006, 64, 210–217. [Google Scholar] [CrossRef]
- Mignot, F.; Schernberg, A.; Arsène-Henry, A.; Vignon, M.; Bouscary, D.; Kirova, Y. Solitary plasmacytoma: Lenalidomide-Dexamethasone combined with radiation therapy improves progression-free survival and multiple myeloma-free survival. Int. J. Radiat. Oncol. Biol. Phys. 2020, 106, 589–596. [Google Scholar] [CrossRef] [PubMed]
- Mheidly, K.; Lamy De La Chapelle, T.; Hunault, M.; Benboubker, L.; Benchalal, M.; Moreau, P.; Baugier de Materre, A.; Decaux, O.; Laribi, K. New insights in the treatment of patients with solitary bone plasmacytoma. Leuk. Lymphoma 2019, 60, 2810–2813. [Google Scholar] [CrossRef] [PubMed]
- Aviles, A.; Huerta-Guzman, J.; Delgado, S.; Fernandez, A.; Diaz-Maqueo, J.C. Improved outcome in solitary bone plasmacytomata with combined therapy. Hematol. Oncol. 1996, 14, 111–117. [Google Scholar] [CrossRef]
- Mayr, N.A.; Wen, B.-C.; Hussey, D.H.; Burns, C.P.; Staples, J.J.; Doornbos, J.F.; Vigliotti, A.P. The role of radiation therapy in the treatment of solitary plasmacytomas. Radiother. Oncol. J. Eur. Soc. Ther. Radiol. Oncol. 1990, 17, 293–303. [Google Scholar] [CrossRef]
- Holland, J.; Trenkner, D.A.; Wasserman, T.H.; Fineberg, B. Plasmacytoma. Treatment results and conversion to myeloma. Cancer 1992, 69, 1513–1517. [Google Scholar] [CrossRef]
- Paiva, B.D.L.; Chandia, M.; Vidriales, M.-B.; Colado, E.; Caballero-Velázquez, T.; Escalante, F.; De Coca, A.G.; Montes, M.-C.; García-Sanz, R.; Ocio, E.M.; et al. Multiparameter flow cytometry for staging of solitary bone plasmacytoma: New criteria for risk of progression to myeloma. Blood 2014, 124, 1300–1303. [Google Scholar] [CrossRef] [PubMed]
- Hill, Q.A.; Rawstron, A.C.; Tute RM de Owen, R.G. Outcome prediction in plasmacytoma of bone: A risk model utilizing bone marrow flow cytometry and light-chain analysis. Blood 2014, 124, 1296–1299. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Elhammali, A.; Amini, B.; Ludmir, E.B.; Gunther, J.R.; Milgrom, S.A.; Pinnix, C.C.; Andraos, T.; Yoder, A.; Weber, D.; Orlowski, R.; et al. New Paradigm for Radiation in Multiple Myeloma: Lower yet effective dose to avoid radiation toxicity. Haematologica 2020. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oertel, M.; Elsayad, K.; Haverkamp, U.; Stelljes, M.; Eich, H.T. Radiotherapy for extramedullary leukaemic manifestation (Chloroma). Strahlenther. Onkol. 2018, 194, 164–173. [Google Scholar] [CrossRef] [PubMed]
- Elsayad, K.; Reinartz, G.; Oertel, M.; Rehn, S.; Eismann, J.; Scobioala, S.; Berssenbrügge, H.; Eter, N.; Weishaupt, C.; Schmidt, H.H.; et al. Radiotherapy of extranodal low-grade follicular and marginal zone lymphomas: Long-term follow-up of 159 patients. Strahlenther. Onkol. 2020, 196, 117–125. [Google Scholar] [CrossRef]
- Engert, A.; Plütschow, A.; Eich, H.T.; Lohri, A.; Dörken, B.; Borchmann, P.; Berger, B.; Greil, R.; Willborn, K.C.; Wilhelm, M.; et al. Reduced treatment intensity in patients with early-stage Hodgkin’s lymphoma. N. Engl. J. Med. 2010, 363, 640–652. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lowry, L.; Smith, P.; Qian, W.; Falk, S.; Benstead, K.; Illidge, T.; Linch, D.; Robinson, M.; Jack, A.; Hoskin, P. Reduced dose radiotherapy for local control in non-Hodgkin lymphoma: A randomised phase III trial. Radiother. Oncol. J. Eur. Soc. Ther. Radiol. Oncol. 2011, 100, 86–92. [Google Scholar] [CrossRef] [PubMed]
- Le Ray, E.; Belin, L.; Plancher, C.; Anract, P.; Babinet, A.; Dumaine, V.; Tamburini, J.; Fischer, B.D.; Willems, L.; Magro, L.; et al. Our experience of solitary plasmacytoma of the bone: Improved PFS with a short-course treatment by IMiDs or proteasome inhibitors combined with intensity-modulated radiotherapy. Leuk. Lymphoma 2018, 59, 1756–1758. [Google Scholar] [CrossRef] [PubMed]
- Mendenhall, C.M.; Thar, T.L.; Million, R.R. Solitary plasmacytoma of bone and soft tissue. Int. J. Radiat. Oncol. Biol. Phys. 1980, 6, 1497–1501. [Google Scholar] [CrossRef]
- Tsang, R.; Gospodarowicz, M.K.; Pintilie, M.; Bezjak, A.; Wells, W.; Hodgson, D.C.; Stewart, A. Solitary plasmacytoma treated with radiotherapy: Impact of tumor size on outcome. Int. J. Radiat. Oncol. Biol. Phys. 2001, 50, 113–120. [Google Scholar] [CrossRef]
- Knobel, D.; Zouhair, A.; Tsang, R.; Poortmans, P.; Belkacémi, Y.; Bolla, M.; Dinçbas, F.O.; Landmann, C.; Castelain, B.; Ozsahin, M. Prognostic factors in solitary plasmacytoma of the bone: A multicenter Rare Cancer Network study. BMC Cancer 2006, 6, 118. [Google Scholar] [CrossRef] [Green Version]
- Wen, G.; Wang, W.; Zhang, Y.; Niu, S.; Li, Q.; Li, Y. Management of extramedullary plasmacytoma: Role of radiotherapy and prognostic factor analysis in 55 patients. Chin. J. Cancer Res. 2017, 29, 438–446. [Google Scholar] [CrossRef] [Green Version]
- Suh, Y.-G.; Suh, C.-O.; Kim, J.S.; Kim, S.-J.; Pyun, H.O.; Cho, J. Radiotherapy for solitary plasmacytoma of bone and soft tissue: Outcomes and prognostic factors. Ann. Hematol. 2012, 91, 1785–1793. [Google Scholar] [CrossRef]
- Reed, V.; Medeiros, L.J.; Ha, C.S.; Arzu, I.; Shihadeh, F.; Dabaja, B.S.; Shah, J.; Mazloom, A.; Weber, D.; Orlowski, R.Z.; et al. Solitary plasmacytomas. Cancer 2011, 117, 4468–4474. [Google Scholar] [CrossRef]
- Sasaki, R.; Yasuda, K.; Abe, E.; Uchida, N.; Kawashima, M.; Uno, T.; Fujiwara, M.; Shioyama, Y.; Kagami, Y.; Shibamoto, Y.; et al. Multi-institutional analysis of solitary extramedullary plasmacytoma of the head and neck treated with curative radiotherapy. Int. J. Radiat. Oncol. Biol. Phys. 2012, 82, 626–634. [Google Scholar] [CrossRef] [PubMed]
- Moreno, A.C.; Frank, S.J.; Garden, A.S.; Rosenthal, D.I.; Fuller, C.D.; Gunn, G.B.; Reddy, J.P.; Morrison, W.H.; Williamson, T.D.; Holliday, E.B.; et al. Intensity modulated proton therapy (IMPT)—The future of IMRT for head and neck cancer. Oral Oncol. 2019, 88, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Elsayad, K.; Stockmann, D.; Channaoui, M.; Scobioala, S.; Grajda, A.; Berssenbrügge, H.; Huss, S.; Moustakis, C.; Haverkamp, U.; Kleinheinz, J.; et al. Using Image-guided Intensity-modulated Radiotherapy on Patients With Head and Neck Soft-tissue Sarcoma. In Vivo 2019, 33, 1293–1300. [Google Scholar] [CrossRef] [PubMed]
- Chesi, M.; Bergsagel, P.L. Molecular pathogenesis of multiple myeloma: Basic and clinical updates. Int. J. Hematol. 2013, 97, 313–323. [Google Scholar] [CrossRef] [PubMed]
- Mateos, M.-V.; Hernández, M.-T.; Giraldo, P.; De La Rubia, J.; De Arriba, F.; López-Corral, L.; Rosiñol, L.; Paiva, B.D.L.; Palomera, L.; Bargay, J.; et al. Lenalidomide plus dexamethasone for high-risk smoldering multiple myeloma. N. Engl. J. Med. 2013, 369, 438–447. [Google Scholar] [CrossRef] [PubMed] [Green Version]
| Characteristic | Nr. (% or Range) | Site of Solitary Plasmacytoma | ||
|---|---|---|---|---|
| Extramedullary | Bone | p-value | ||
| Patients | 84 | 30 (36%) | 54 (64%) | |
| Median Age | 57 y (22–81) | 53 (22–81) | 60 (28–77) | 0.2 |
| Gender | M: 55 (65 %) F: 29 (35 %) | 22 (73%) 8 (27%) | 33 (61%) 21 (39%) | 0.3 |
| Immunohistochemical analysis | 0.3 | |||
| Kappa light chain restriction | 25 (30%) | 12 (40%) | 13 (24%) | |
| Lambda light chain restriction | 25 (30%) | 10 (33%) | 15 (28%) | |
| Both | 2 (2%) | 0 (0%) | 2 (4%) | |
| Maximum diameter | 0.16 | |||
| In cm | 4 (0.7–9.6) | 3.6 (0.7–7.9) | 4.1 (1.3–9.6) | |
| Serum Beta-2-microglobulin | 0.1 | |||
| Elevated | 4 (5%) | 1 (3%) | 3 (6%) | |
| Serum protein | 0.17 | |||
| Elevated | 7 (8%) | 2 (7%) | 5 (9%) | |
| Low | 5 (6%) | 3 (10%) | 2 (4%) | |
| Serum albumin | 0.02 | |||
| g/dL | 4.5 (2.6–5.4) | 4.6 (3.6–5.4) | 2.8 (2.6–3) | |
| Hemoglobin | 0.06 | |||
| g/dL | 13.3 (9–16.6) | 14.1 (9.6–16.6) | 13.2 (9–14.8) | |
| Serum protein immunofixation | 0.3 | |||
| Positive | 36 (43%) | 11 (37%) | 25 (46%) | |
| Negative | 29 (34%) | 13 (43%) | 16 (30%) | |
| Unknown | 19 (23%) | 6 (20%) | 13 (24%) | |
| Serum LDH | 0.13 | |||
| Elevated | 5 (6%) | 5 (17%) | 0 (0%) | |
| Bence-Jones proteinuria | 0.12 | |||
| Yes | 12 (14%) | 6 (20%) | 6 (11%) | |
| Sites of RT | <0.001 | |||
| Head and neck | 29 (34%) | 24 (80%) | 5 (9%) | |
| Thorax | 28 (33%) | 2 (7%) | 26 (48%) | |
| Abdomen | 2 (2%) | 0 | 2 (4%) | |
| Pelvis | 21 (25%) | 4 (13%) | 17 (32%) | |
| Extremities | 4 (5%) | 0 | 4 (7%) | |
| Characteristic | Nr. (% or Range) | Site of Solitary Plasmacytoma | ||
|---|---|---|---|---|
| Extramedullary | Bone | p-value | ||
| Type of surgery | <0.001 | |||
| Resection | 20 (24%) | 15 (50%) | 5 (9%) | |
| Biopsy | 64 (76%) | 15 (50%) | 49 (91%) | |
| Systemic therapy | 0.004 | |||
| Yes | 28 (33%) | 4 (13%) | 24 (44%) | |
| No | 56 (67%) | 26 (87%) | 30 (56%) | |
| Timing of systemic therapy | ||||
| Prior to RT alone | 5/28 (18%) | 2/4 (50%) | 3/24 (12%) | <0.001 |
| Adjuvant alone | 21/28 (75%) | 0/4 (0%) | 21/24 (88%) | |
| Both | 2 (7%) | 2/4 (50%) | 0/24 (0%) | |
| Radiation parameters | ||||
| Med. radiation dose (range), Gy | 42 (30–59.4) | 50 (36–59.4) | 40 (30–59.4) | <0.001 |
| Radiation doses | <0.001 | |||
| ≤40 Gy | 44 (53%) | 3 (10%) | 41 (76%) | |
| >40–50 Gy | 22 (26%) | 16 (53%) | 6 (11%) | |
| >50 Gy | 18 (21%) | 11 (37%) | 7 (13%) | |
| Med. fraction dose (range), Gy | 2 (1.8–3) | 2 (1.8–3) | 2 (1.8–2) | 0.03 |
| Follow-up, m | 46 (2–154) | 40 (2–154) | 52 (2–149) | 0.4 |
| Follow-up, IQR | 53 | 48 | 55 | |
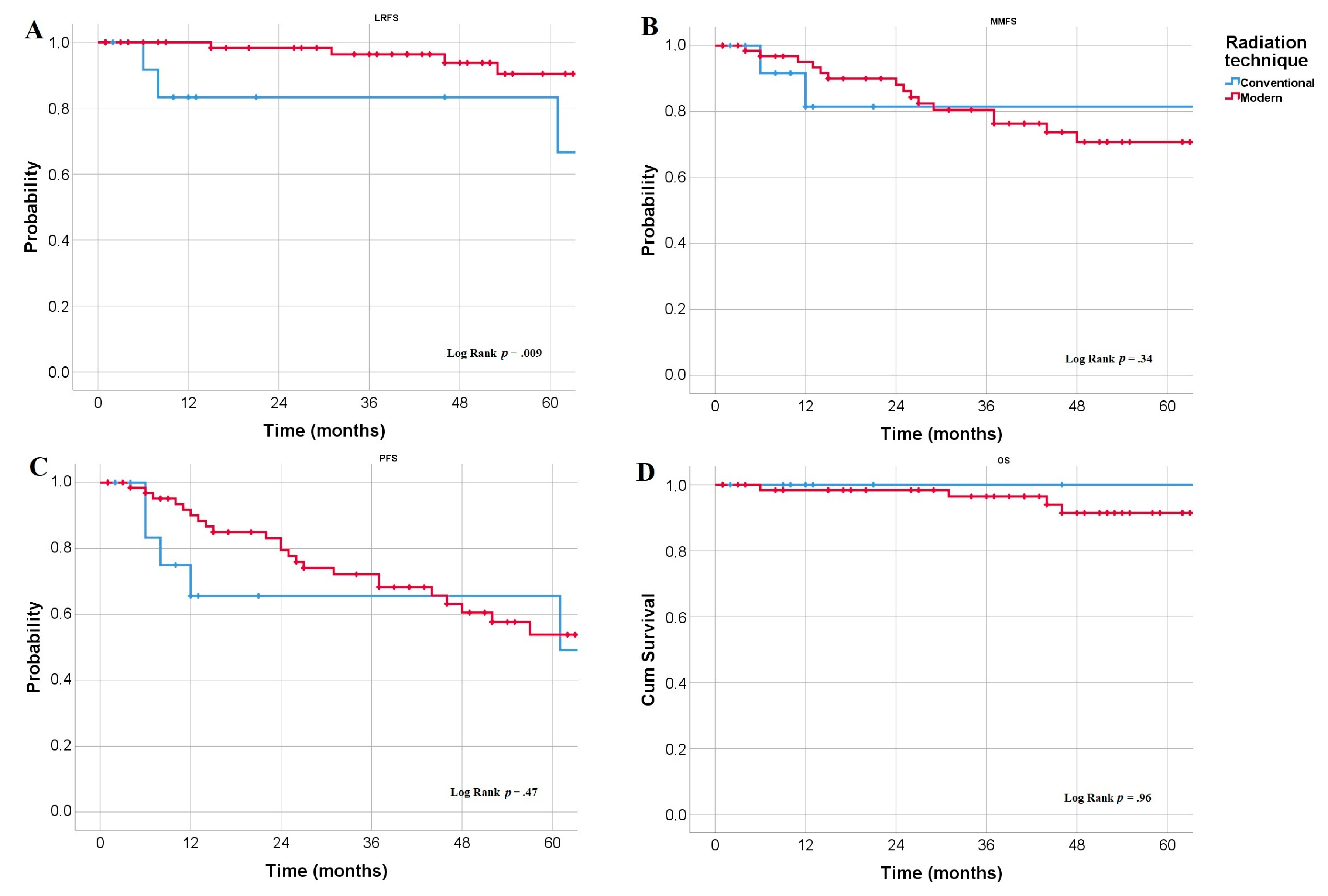
| RT technique | 0.02 | |||
| Proton therapy | 4 (5%) | 4 (13%) | 0 | |
| IMRT | 64 (76%) | 22 (74%) | 42 (78%) | |
| 3D-CRT | 16 (19%) | 4 (13%) | 12 (22%) | |
| Response to RT | 0.7 | |||
| Yes | 81 (96%) | 29 (97%) | 52 (96%) | |
| No | 3 (4%) | 1 (3%) | 2 (4%) | |
| Type of response | <0.001 | |||
| CR | 39/81 (48%) | 25/29 (86%) | 14/52 (27%) | |
| PR | 8/81 (10%) | 4/29 (14%) | 4/52 (8%) | |
| SD | 34/81 (42%) | 0 | 34/52 (65%) | |
| Relapse | 0.7 | |||
| Yes | 29 (35%) | 9 (30%) | 20 (37%) | |
| No | 55 (65%) | 21 (70%) | 34 (63%) | |
| Pattern of relapse | 0.6 | |||
| Local | 8 (10%) | 3/30 (10%) | 5/54 (9%) | |
| Multiple myeloma | 21 (25%) | 6/30 (20%) | 15/54 (28%) | |
| Risk Factor | LRFS | MMFS | PFS | OS | ||||
|---|---|---|---|---|---|---|---|---|
| HR | p | HR | p | HR | p | HR | p | |
| Univariate model | ||||||||
| Age (years) | 1.06 | 0.09 | 1.005 | 0.8 | 1.034 | 0.06 | 1.085 | 0.08 |
| EMP vs. SBP | 1.26 | 0.7 | 0.602 | 0.3 | 0.82 | 0.6 | 2.85 | 0.3 |
| Diameter (cm) | 0.89 | 0.7 | 1.17 | 0.3 | 1.06 | 0.6 | 1.74 | 0.07 |
| Bone erosion | 4.24 | 0.15 | 0.302 | 0.26 | 0.99 | 0.9 | 0.71 | 0.8 |
| Surgical resection | 2.29 | 0.26 | 0.67 | 0.5 | 0.91 | 0.8 | 0.49 | 0.4 |
| RT dose (>40 Gy vs. ≤40 Gy) | 0.38 | 0.2 | 1.39 | 0.5 | 1.12 | 0.7 | 1.92 | 0.5 |
| Modern RT technique | 0.83 | 0.02 | 2.08 | 0.4 | 0.72 | 0.5 | 1.06 | 0.9 |
| CR after RT | 1.39 | 0.6 | 0.42 | 0.09 | 0.67 | 0.3 | 1.01 | 0.9 |
| B2-microglobulin (normal) | 6.0 | 0.2 | 3.22 | 0.1 | 0.31 | 0.08 | 0.285 | 0.3 |
| Serum protein (normal) | 0.024 | 0.002 | 0.94 | 0.9 | 0.24 | 0.02 | 28.1 | 0.7 |
| Hemoglobin (g/dl) | 1.32 | 0.5 | 0.67 | 0.006 | 0.68 | 0.004 | 0.91 | 0.7 |
| Systemic therapy | 1.09 | 0.9 | 0.44 | 0.19 | 0.47 | 0.1 | 0.03 | 0.5 |
| Multivariate model | ||||||||
| Age (years) | - | - | - | - | - | - | 1.14 | 0.1 |
| Diameter | - | - | - | - | - | - | 2.29 | 0.03 |
| Modern RT technique | 0.036 | 0.012 | - | - | - | - | - | - |
| Serum protein (normal) | 0.019 | 0.008 | - | - | - | - | - | - |
| Hemoglobin (g/dl) | - | - | 0.676 | 0.006 | 0.655 | 0.004 | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Elsayad, K.; Oertel, M.; König, L.; Hüske, S.; Le Ray, E.; Meheissen, M.A.M.; Elsaid, A.A.; Elfaham, E.; Debus, J.; Kirova, Y.; et al. Maximizing the Clinical Benefit of Radiotherapy in Solitary Plasmacytoma: An International Multicenter Analysis. Cancers 2020, 12, 676. https://doi.org/10.3390/cancers12030676
Elsayad K, Oertel M, König L, Hüske S, Le Ray E, Meheissen MAM, Elsaid AA, Elfaham E, Debus J, Kirova Y, et al. Maximizing the Clinical Benefit of Radiotherapy in Solitary Plasmacytoma: An International Multicenter Analysis. Cancers. 2020; 12(3):676. https://doi.org/10.3390/cancers12030676
Chicago/Turabian StyleElsayad, Khaled, Michael Oertel, Laila König, Sebastian Hüske, Emmanuelle Le Ray, Mohamed A.M. Meheissen, Amr Abdelaziz Elsaid, Essam Elfaham, Jürgen Debus, Youlia Kirova, and et al. 2020. "Maximizing the Clinical Benefit of Radiotherapy in Solitary Plasmacytoma: An International Multicenter Analysis" Cancers 12, no. 3: 676. https://doi.org/10.3390/cancers12030676
APA StyleElsayad, K., Oertel, M., König, L., Hüske, S., Le Ray, E., Meheissen, M. A. M., Elsaid, A. A., Elfaham, E., Debus, J., Kirova, Y., Herfarth, K., & Eich, H. T. (2020). Maximizing the Clinical Benefit of Radiotherapy in Solitary Plasmacytoma: An International Multicenter Analysis. Cancers, 12(3), 676. https://doi.org/10.3390/cancers12030676







